Abstract
W-18 (4-chloro-N-[1-[2-(4-nitrophenyl)ethyl]-2-piperidinylidene]-benzenesulfonamide) and W-15 (4-chloro-N-[1-(2-phenylethyl)-2-piperidinylidene]-benzenesulfonamide) represent two emerging drugs of abuse chemically related to the potent opioid agonist fentanyl (N-(1-(2-phenylethyl)-4-piperidinyl)-N-phenylpropanamide). Here, we describe the comprehensive pharmacological profiles of W-18 and W-15, as examination of their structural features predicted that they might lack opioid activity. We found W-18 and W-15 to be without detectible activity at μ, δ, κ, and nociception opioid receptors in a variety of assays. We also tested W-18 and W-15 for activity as allosteric modulators at opioid receptors and found them devoid of significant positive or negative allosteric modulatory activity. Comprehensive profiling at essentially all the druggable GPCRs in the human genome using the PRESTO-Tango platform revealed no significant activity. Weak activity at the sigma receptors and the peripheral benzodiazepine receptor was found for W-18 (Ki = 271 nM). W-18 showed no activity in either the radiant heat tail-flick or the writhing assays and also did not induce classical opioid behaviors. W-18 is extensively metabolized, but its metabolites also lack opioid activity. Thus, although W-18 and W-15 have been suggested to be potent opioid agonists, our results reveal no significant activity at these or other known targets for psychoactive drugs.
Keywords: Neuroscience
Keywords: Drug screens, Drug therapy
The emerging drugs of abuse W-18 and W-15 were found to be pharmacologically inactive.
Introduction
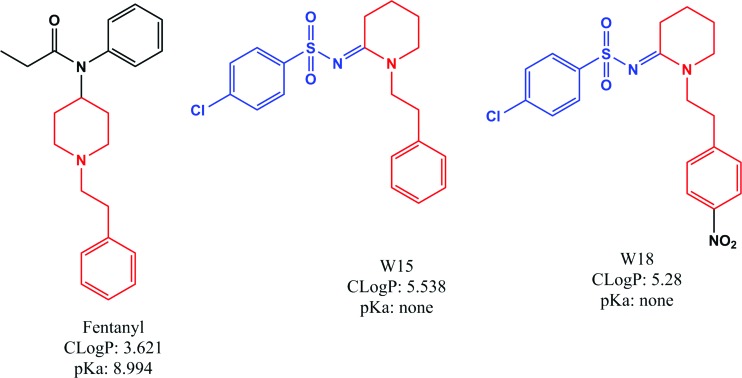
W-18 (4-chloro-N-[1-[2-(4-nitrophenyl)ethyl]-2-piperidinylidene]-benzenesulfonamide) and W-15 (4-chloro-N-[1-(2-phenylethyl)-2-piperidinylidene]-benzenesulfonamide) were originally identified in the patent literature as analogs of the potent opioid agonist fentanyl (N-(1-(2-phenylethyl)-4-piperidinyl)-N-phenylpropanamide) (Figure 1) (1). Given the fact that W-18 and W-15 differ in key respects chemically from fentanyl (Figure 1), it could be predicted that they might lack appreciable opioid receptor agonist actions. The presence of an aryl sulfonamide group renders the piperidine nitrogen atom nonbasic. A basic tertiary amine is commonly seen in most opioid templates. It could thereby be predicted that both W-15 and W-18 might lack appreciable opioid receptor agonist activity. In the original description, W-18 and W-15 (along with several other analogs) were described to be extraordinarily potent at inhibiting phenylquinone-induced writhing — a mouse model useful for assessing potential analgesic actions for a wide variety of drugs, such as aspirin, antihistamines, opioids, antidepressants, sympathomimetics, acetylcholinesterase inhibitors, and psychotomimetic opioids, such as cyclazocine (2). Thus, the inhibition of phenylquinone-induced writhing is a relatively nonspecific test for drugs with potential analgesic and other activities. In the original report, there was no evidence that the activity in this model by either W-18 or W-15 was antagonized by the prototypical opioid antagonist naloxone, although the related compound W-20 (1-cyclopropylmethylpiperidylidene-2-(4-chlorophenyl)sulfonamide) was reported to have modest naloxone-reversible activity.
Figure 1. Structure of fentanyl, W-15, and W-18.
Shown are the chemical structures of fentanyl (left), W-15 (center), and W-18 (right). The structural similarities between fentanyl and W-15/W-18 are shown in red. The presence of aryl sulfonamide group (in blue) in both W-15/W-18 makes the piperidine nitrogen not charged at physiological pH compared with the fentanyl piperidine nitrogen.
Recently, W-18 (3–7) and perhaps W-15 have emerged as potential drugs of abuse; W-18 is scheduled as a controlled substance in Canada (8) and is being considered for inclusion in Schedule 1 control in the US. To our knowledge, there are no other published reports on the pharmacology of W-18 or W-15. Accordingly, we here report a comprehensive evaluation of the in vitro molecular pharmacology of W-18 and W-15.
Results
Radioligand-binding assays reveal no activity of W-18 at human, cloned opioid receptors.
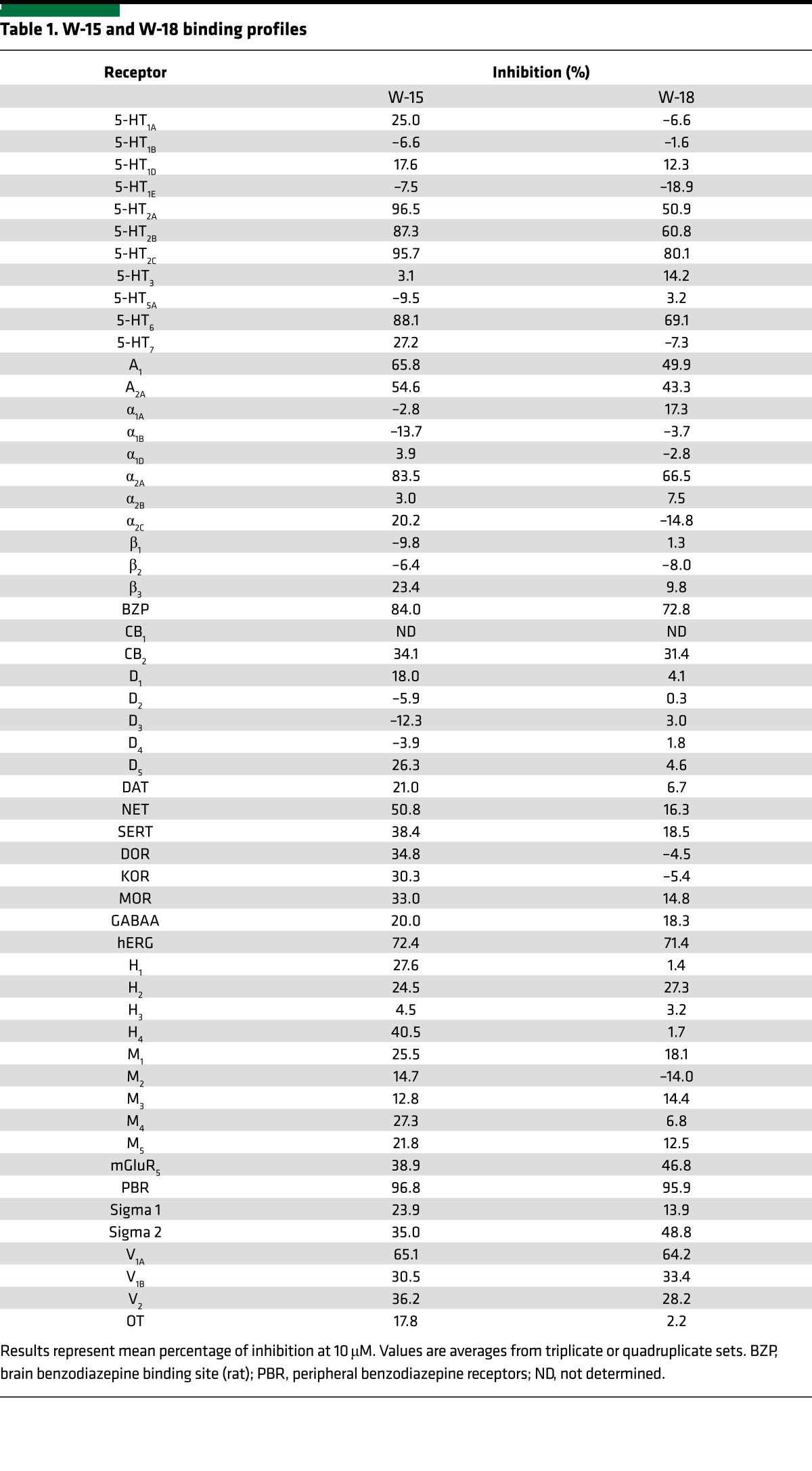
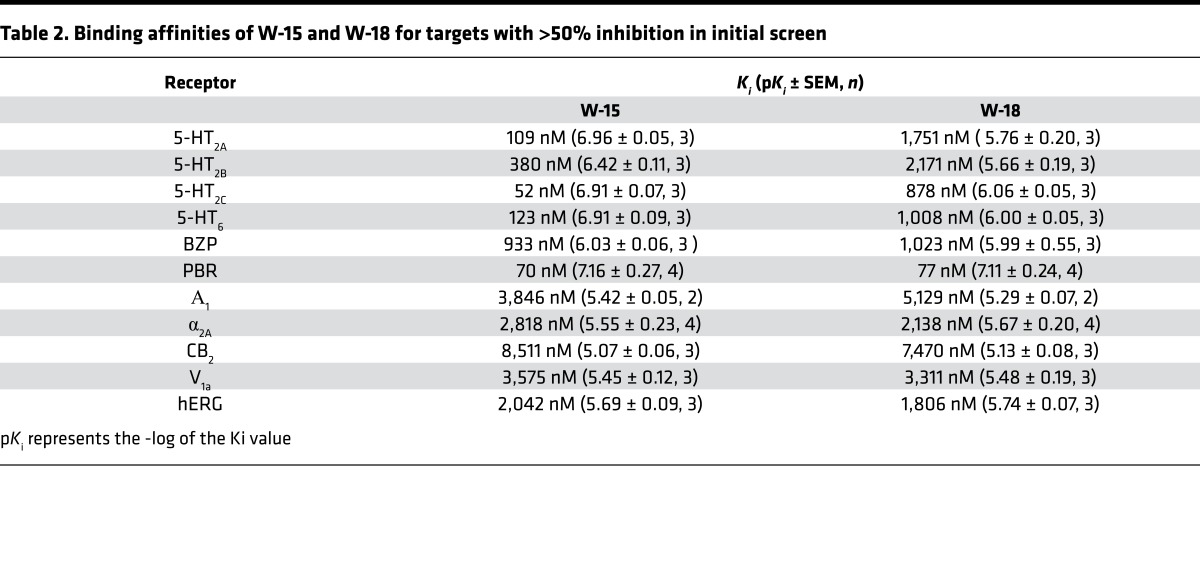
In our initial studies, we evaluated the ability of W-15 and W-18 to interact with 3 canonical opioid receptors via radioligand-binding studies performed as previously described using cloned, human κ (9, 10), μ (11), and δ opioid receptors (KOR, MOR, and DOR, respectively) (12, 13). No significant inhibition of radioligand binding was measured up to concentrations as high as 10,000 nM (Table 1). These studies were followed by investigation of effects against the murine MOR, KOR, and DOR, with similar results. No inhibition of binding with concentrations as high as 1 μM were observed. W-15 and/or W-18 inhibited radioligand binding at several nonopioid receptors with low affinities, including the 5-HT2A, 5-HT2B, 5-HT2C, and 5-HT6 serotonin receptors; benzodiazepine receptors (BZP and PBR) (Table 1); and other miscellaneous targets (Table 2).
Table 1. W-15 and W-18 binding profiles.
Table 2. Binding affinities of W-15 and W-18 for targets with >50% inhibition in initial screen.
Functional assays reveal no agonist, antagonist, or allosteric modulator activity of W-18 or W-15 at human cloned opioid receptors.
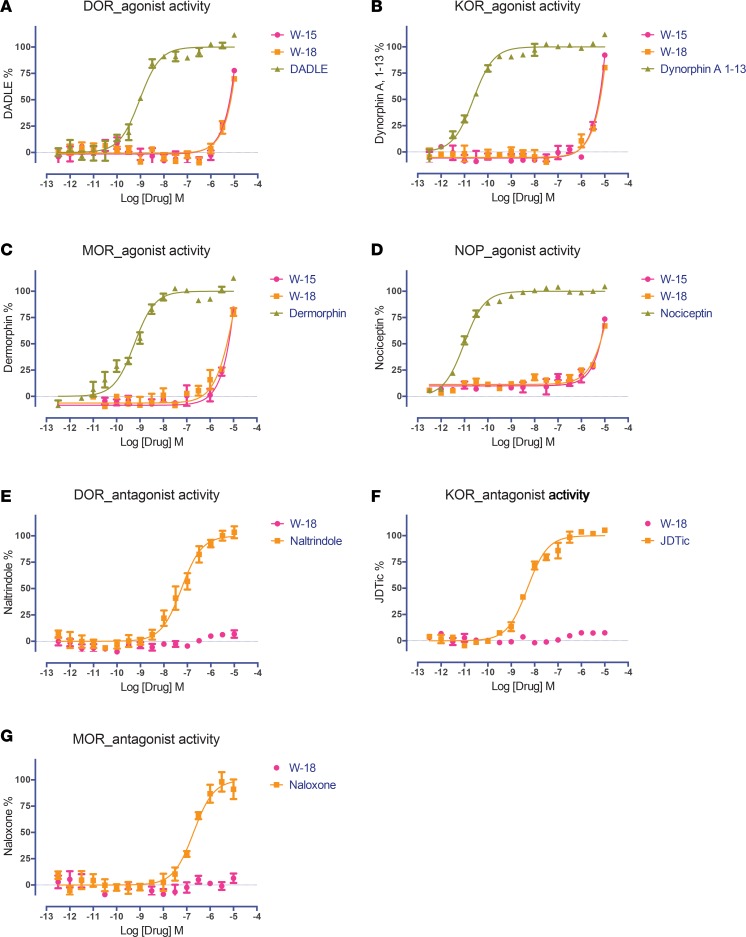
We next evaluated W-18 and W-15 for agonist and antagonist activity at cloned human opioid receptors in both Gi-dependent inhibition of cAMP production and G protein–independent arrestin translocation assays (Figures 2 and 3). No significant agonist or antagonist activity was measured up to doses as high as 10,000 nM, although nonspecific agonist effects were evident at high concentrations in these assays (Figure 2, A–D). We also evaluated the potential of W-18 to be an allosteric modulator of opioid receptors and found only weak and potentially nonspecific negative allosteric modulator actions in either Gi-cAMP or arrestin signaling (Figures 4 and 5).
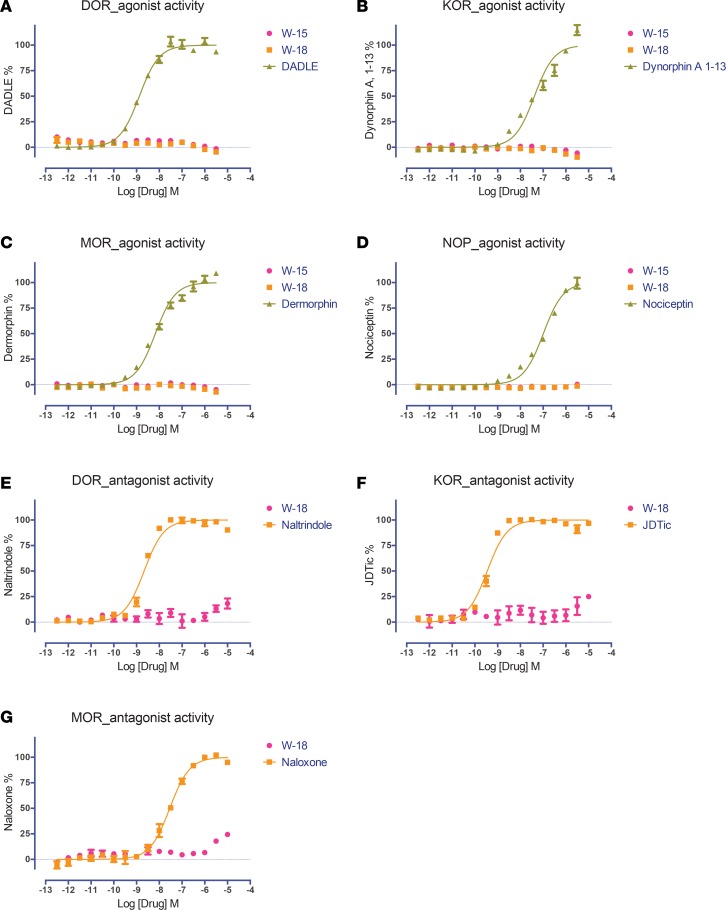
Figure 2. Effect of W-15 and W-18 on opioid receptor–mediated Gαi signaling.
The Gi-mediated inhibition of cAMP production was determined as previously described (9–11). Both W-15 and W-18 showed no agonist activity at DOR (A), KOR (B), MOR (C), and nociceptin (NOP) (D) receptors and no antagonist activity at DOR (E), KOR (F), or MOR (G) receptors. Normalized results represent mean ± SEM of 3 independent experiments, each performed in triplicate (0% activity for vehicle activity; 100% for reference activity), and were analyzed in Prism.
Figure 3. Effect of W-15 and W-18 on opioid receptor–mediated β-arrestin recruitment.
G protein–independent arrestin recruitment was determined in the GPCR Tango assay, as outlined in the Methods. Both W-15 and W-18 were tested and showed no agonist activity at DOR (A), KOR (B), MOR (C), and NOP (D) receptors and no antagonist effect at DOR (E), KOR (F), or MOR (G) receptors. Normalized results represent mean ± SEM of 3 independent experiments, each performed in triplicate (0% for vehicle activity, and 100% for reference activity), and were analyzed in Prism.
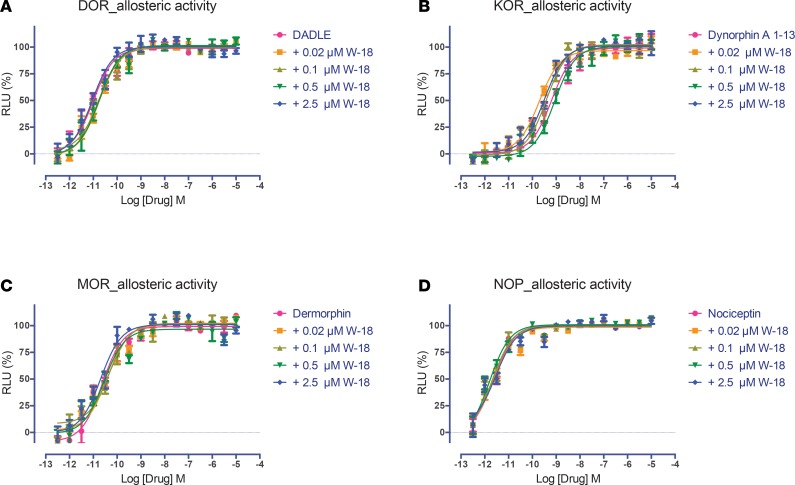
Figure 4. Allosteric activity of W-18 on agonist stimulated inhibition of cAMP accumulation at opioid receptors.
The Gi-mediated inhibition of cAMP production was determined as previously described (9–11) and in Methods. W-18 failed to modify the effect of DADLE at DOR (A), dynorphin A at KOR (B), dermorphin at MOR (C), or nociceptin at NOP (D) receptors. Normalized results represent mean ± SEM of 3 independent experiments, each performed in triplicate (0% for vehicle activity and 100% for reference activity), and were analyzed in Prism.
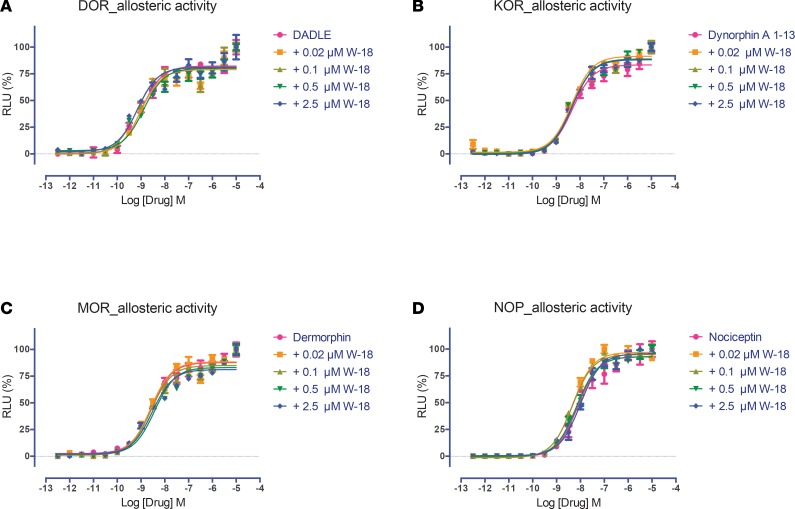
Figure 5. Allosteric activity of W-18 on agonist stimulated β-arrestin recruitment at opioid receptors.
The arrestin recruitment activity was determined in the GPCR Tango assays, as outlined in the Methods. W-18 failed to modify the effect of DADLE at DOR (A), dynorphin A at KOR (B), dermorphin at MOR (C), or nociceptin at NOP (D) receptors. Normalized results represent mean ± SEM of 3 independent experiments, each performed in triplicate (0% activity for vehicle and 100% for reference activity), and were analyzed in Prism.
Functional assays reveal antagonist activity at human 5-HT receptors.
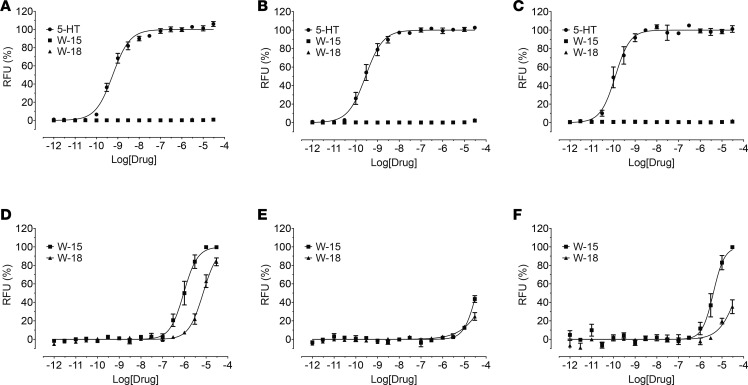
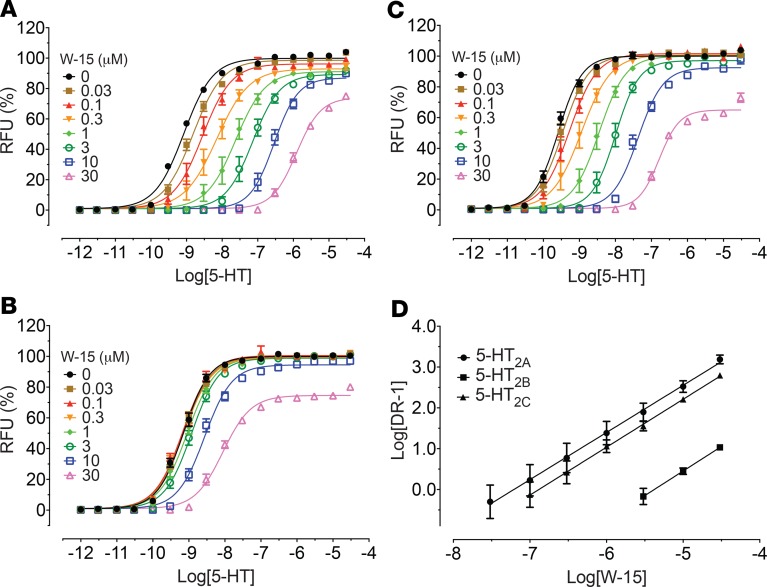
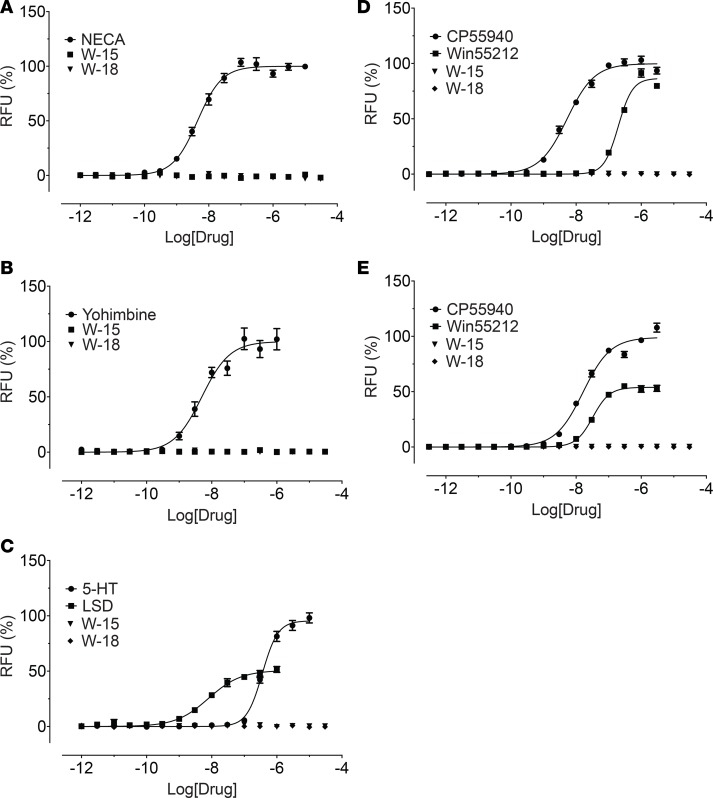
Since W-15 and W-18 displayed binding activities at 5-HT2 and 5-HT6 receptors, we examined their functional activity at 5-HT2A, 5-HT2B, and 5-HT2C receptors for calcium mobilization and at 5-HT6 receptors for arrestin signaling. W-15 and W-18 showed no agonist activity and modest antagonist activity at 5-HT2 receptors (Figure 6). Schild analysis with W-15 indicated competitive antagonism against 5-HT at 5-HT2 receptors, as shown by the parallel shifts of the agonist dose-response curves (Figure 7). At 5-HT6 receptors, W-15 and W-18 displayed no agonist activity (Figure 8). W-15 and W-18 also displayed weak binding at A1-adenosine, α2A-adrenergic, and CB2 cannabinoid receptors; we therefore examined the nature of their activity at these receptors. Our results (Figure 8) indicated that the drugs had no agonist activity at any of these examined receptors.
Figure 6. Agonist activities and antagonist effects of W-15 and W-18 at 5-HT2A, 5-HT2B, and 5-HT2C receptors.
Gq-mediated calcium mobilization activity was determined using HEK293 cells stably expressing corresponding 5-HT2 subtypes on FLIPRTETRA. Normalized results (relative fluorescence unit in fold of basal, basal was set as 0% and 5-HT Emax was set as 100%) represented mean values ± SEM from 2 independent assays, each performed in quadruplicate, and were analyzed in Prism. In antagonist activity assays, the reference agonist (5-HT) was used at a final concentration of 10 nM and was set as 0% inhibition and basal was set as 100% inhibition. Panels A–C represent dose-response curves to 5-HT, W-15 and W-18 at 5-HT2A (A), 5-HT2B (B), or 5-HT2C (C) receptors. Panels D–F shows the antagonist activity of W-15 and W-18 at 5-HT2A (D), 5-HT2B (E), or 5-HT2C (F) receptors.
Figure 7. Concentration-dependent effect of W-15 on 5-HT activity at 5-HT2A, 5-HT2B, and 5-HT2C receptors.
5-HT–mediated calcium mobilization was determined in the absence and presence of increasing concentrations of W-15 using HEK293 cells stably expressing corresponding 5-HT2 subtypes on FLIPRTETRA. Normalized results (relative fluorescence unit in fold of basal, basal was set as 0%, and 5-HT Emax in the absence of W-15 was set as 100%) represented mean values ± SEM from 3 independent assays, each performed in triplicate, and were analyzed in Prism. Schild plot analysis (D) indicated W-15 is a competitive antagonist against 5-HT. The average pA2 values are 7.20 (5-HT2A), 5.38 (5-HT2B), and 6.88 (5-HT2C). Panels A–C represent oncentration response curves for W-15 with increasing concentrations of 5-HT at 5-HT2A (A), 5-HT2B (B), or 5-HT2C (C) receptors.
Figure 8. W-15 and W-18 have no agonist activity at A1, α2A, 5-HT6, CB1, or CB2 receptors.
Agonist activity (arrestin recruitment) was determined in GPCR Tango assays, as outlined in the Methods. Normalized results (basal was set as 0% and reference Emax was set as 100%) represented mean ± SEM from minimum of n assays (n = 2 for A and B, n = 3 for C–E), each performed in triplicate, and were analyzed in Prism. Panels A–E represent a dose response curve for A1-adenosine (A), a2-adrenergic (B), 5-HT6 receptors (C), CB1 receptors (D), or CB2 receptors (E).
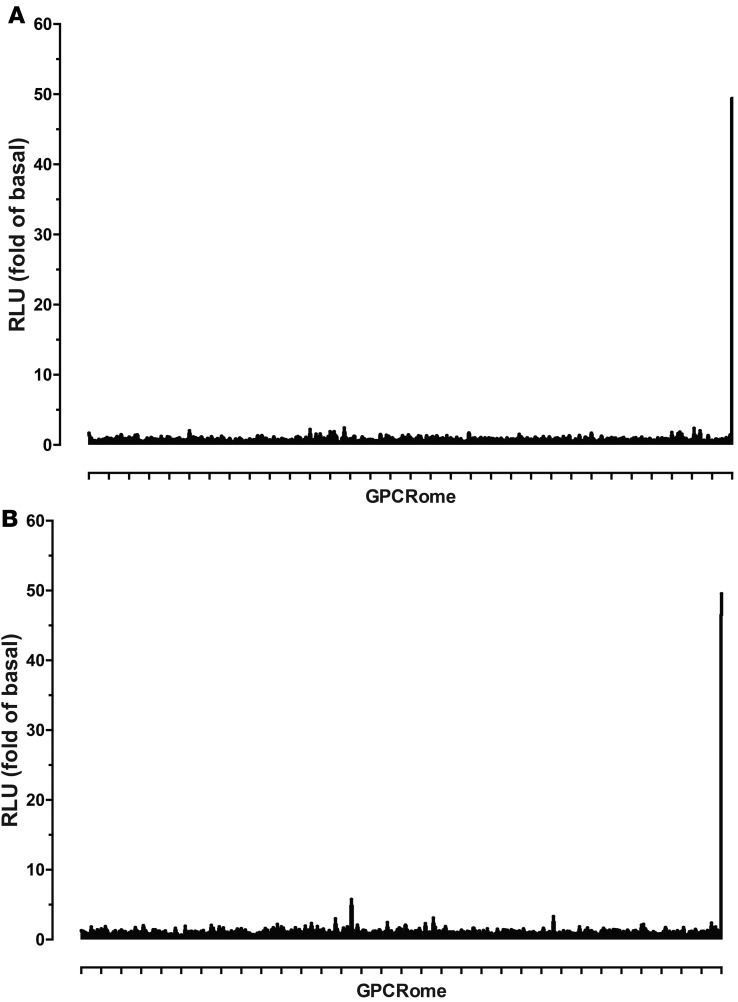
Screening of W-18 and W-15 against the druggable GPCR-ome revealed no significant agonist activity at any human druggable GPCR.
We next screened W-18 and W-15 against the druggable human GPCR-ome using our recently developed PRESTO-Tango resource (14). Although no significant agonist activity was reproducibly found, we did note that concentrations of W-18 higher than 1 μM tended to cause a decrease in the overall baseline activity for nearly every target, consistent with a toxic activity at HEK cells with prolonged incubation (Figure 9).
Figure 9. GPCR-ome screening with W-18 and W-15.
GPCR-ome screening was conducted according to the PRESTO-Tango assays, as outlined in the Methods. W-15 and W-18 were screened at a final concentration of 1 μM, in quadruplicate for both sample and basal, and results are presented in fold of basal. Dopamine (D2) receptor with 100 nM quinpirole served as an assay control. A represents GPCRome screen of W-18. B represents GPCRome screen of W-15.
We examined the metabolism of W-18. W-18 was extensively metabolized by both human and mouse liver microsomes (Supplemental Figures 1–3), resulting in multiple monohydroxylated and dihydroxylated metabolites as well as a dealkylated and an amino metabolite from reduction of the nitro group. To determine if W-18 was a prodrug, releasing an active compound through metabolism, we incubated W-18 with liver microsomes, extracted the remaining W-18 and its metabolites, and tested the mixture in opioid receptor–binding assays at a concentration corresponding to 1 μM W-18 prior to the microsomal incubation. The mixture of W-18 and metabolites failed to significantly inhibit binding to murine MOR, DOR, or KOR expressed in Chinese hamster ovary (CHO) cells.
In silico studies.
We also used the similarity ensemble approach to determine if any molecular targets might be predicted with high expectancies to have activity for W-18. No target(s) emerged with high confidence, although weak predictions (E = 4.67 e–1) suggested activity were for H3-histamine for W-18, which was not confirmed experimentally (Table 1).
In vivo evaluation.
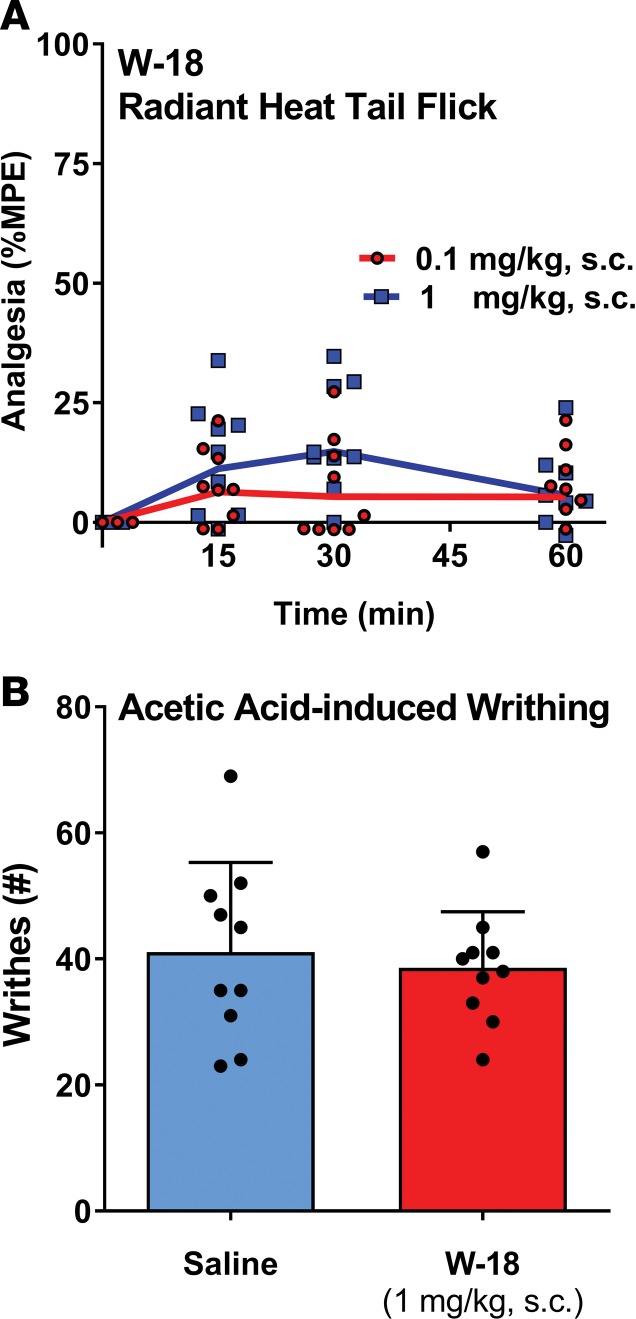
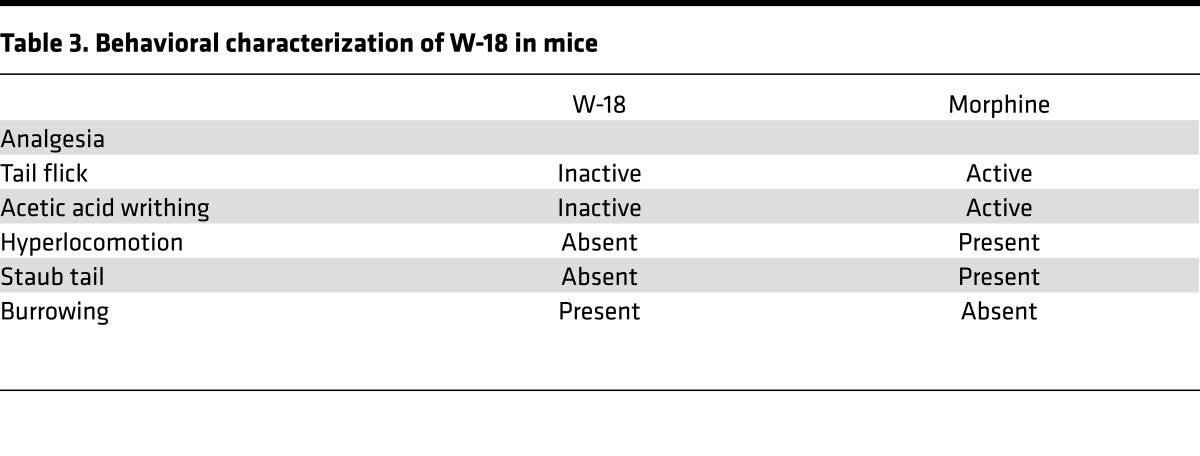
In the initial patent report, W-18 was active in the writhing assay, with a potency many times that of morphine. When administered in mice at doses as high as 1 mg/kg, s.c., W-18 failed to demonstrate any activity in either the radiant heat tail-flick assay or the writhing assay (Figure 10 and Table 3). Treated mice failed to display classical opioid behaviors, such as hyperlocomotion or Straub tail (Table 3). A general behavioral action of burrowing into the bedding was observed that is not associated with classical opioids, and the animals were clearly atypical in their overall behavior, showing a general tunneling behavior. Administration of naloxone failed to counteract these behaviors. Similar burrowing behaviors were seen in rats after s.c. administration of 0.3 and 1.0 mg/kg.
Figure 10. Analgesic activity of W-18 in tail-flick and writhing assays.
(A) Radiant heat tail-flick: W-18 was tested in the radiant heat tail-flick assay at 0.1 and 1 mg/kg, s.c., as previously described (25). Groups of mice (n = 10) received the indicated dose of W-18, after determining baseline latencies, and were tested at the indicated times. ANOVA revealed no significant differences among groups or times. W-18 (0.1 mg/ml) was dissolved in DMSO (10%), Tween 80 (10%)/water for use at the 1 mg/kg dose. The solution was diluted 10-fold to 0.01 mg/ml for use in the 0.1 mg/kg dose. (B) Acetic acid–induced writhing: groups of mice (n = 10) received either saline or W-18 (1 mg/kg, s.c.). Fifteen minutes later, all mice received 1% acetic acid (10 μl/g), and writhes were counted from 5–15 minutes after acetic acid injection. There were no significant differences between the groups by Student’s t test.
Table 3. Behavioral characterization of W-18 in mice.
Discussion
The major findings of these studies are that both W-18 and W-15 are devoid of significant opioid receptor activity. Although W-18 and W-15 were described in the initial patent report as having potent analgesic activity in the phenylquinone-induced writhing assay, it should be noted that the test used cannot definitively identify the molecular target for the presumed “analgesic activity.” The nociceptive stimulus is relatively weak, with opioids active at doses far below those needed in thermal assays. Thus, as originally reported (2), a large assortment of pharmacological agents show activity in this assay, including cholinesterase inhibitors, antihistamines, and antidepressants (15). In our hands, W-18 revealed no antinociceptive activity in either the thermal radiant heat tail-flick assay or the writhing assay. Furthermore, the atypical behaviors observed with the drugs (i.e., tunneling) failed to be reversed by naloxone, indicating that opioid receptors are not involved.
It is conceivable that W-18 and W-15 are prodrugs that require metabolic transformation for their effects in humans, as has been seen for toxic pharmaceuticals, including fenfluramine (16) and methysergide (17). Even the prescribed analgesic codeine requires demethylation to generate morphine for activity in both receptor-binding assays and in vivo. W-18 undergoes extensive metabolism in both human and mouse liver microsomes; however, these metabolites do not reveal any affinity for opioid receptors. Behaviorally, W-18 failed to elicit any naloxone-sensitive actions. In addition, we saw no inhibition of murine opioid receptor–binding from the W-18 metabolite mixture. The hepatic microsomal incubations provide an assessment of in vivo relevant metabolites but may not be comprehensive with respect to conjugation reactions, such as glucuronidation of primary W-18 metabolites. Although further studies aimed at identifying the individual metabolite(s) and determining their pharmacological activities are needed, and may uncover mechanisms contributing to the abuse potential of W-18, our studies strongly indicate that opioid systems are not involved.
In conclusion, comprehensive evaluation of the potential in vitro molecular pharmacology of W-18 and W-15 revealed no consensus targets for interaction. Although apparently devoid of opioid receptor activity, weak activity of W-18 at the peripheral benzodiazepine receptor was found, and the drugs appear to exhibit toxicity in HEK cells with overnight incubation at concentrations higher than 1 μM. The literature has suggested that W-18 falls into the opioid class of drugs, possibly providing a false sense of security for those abusing the compound who may rely upon its reversibility by naloxone. Given the lack of apparent activity at human or mouse cloned opioid receptors for the parent compounds or the metabolites, the utility of naloxone and other opioid antagonists in treating overdose in humans is unlikely to be successful. This needs to be understood by both first responders and those abusing the compound.
Methods
Drugs.
W-18 and W-15 were purchased from Cayman Chemicals and supplied as >98% pure. Independent LC-MS quality control revealed the compounds were both pure and authentic.
Mice.
CD-1 mice (20–30 g) were obtained from Charles River Laboratories and maintained on normal chow.
Cell lines.
HEK93T cells were obtained from ATCC (catalog CRL-3216), and HTLA cells were obtained from Richard Axel (Columbia University).
Radioligand-binding assays.
Radioligand-binding studies were performed as previously described using cloned, human receptors (18). Comprehensive profiling was performed using the resources of the National Institute of Mental Health Psychoactive Drug Screening Program as described previously (18). Initial screens were performed at 10,000 nM in quadruplicate. For targets with inhibition >50%, we also carried out concentration-response binding assays to determine affinities (Ki). Follow-up assays on murine opioid receptors expressed in CHO cells were carried out as previously described (19).
Metabolism studies.
Metabolism was assessed using both mouse liver microsomes and pooled human liver microsomes incubated with NADPH (1 mM). Heat-inactivated microsomes and incubations without NADPH were used as controls. At various time points, 2 times the volume of acetonitrile was added to stop the incubation and precipitate the protein so the sample could be analyzed by mass spectrometry. Samples were analyzed using multiple methods on a Sciex 6500, operating in multiple reaction monitoring mode using predicted mass transitions, using precursor ion and product ion scans. Samples were further analyzed on a Thermo Scientific Q Exactive high-resolution mass spectrometer using full-scan mode operating at 70,000 resolution. Samples were also evaluated by HPLC using UV detection. HPLC-UV samples were processed by solid-phase extraction, dried using a vacuum centrifuge and reconstituted in a minimal volume of mobile phase.
In addition, a W-18 mixed-metabolite sample was prepared by incubating 40 μl of a 20 mM W-18 DMSO stock with 5 ml of 1 mg/ml human liver microsomes or mouse liver microsomes and 1 mM NADPH for 90 minutes. Metabolites were isolated using solid-phase extraction and dried using a speed vacuum. The mixed-metabolite sample was resolubilized, and the pooled metabolites were examined in opioid receptor–binding assays using the murine receptors expressed in CHO cells at concentrations corresponding to an initial concentration of W-18 an initial concentration of W-18 of 1 μM.
Opioid receptor functional assays.
Assays for agonist and antagonist activity at cloned opioid receptors were performed as previously described using cloned, human κ (9, 10), μ (11), δ, and nociceptin opioid receptors (12, 13). Assays for evaluating the allosteric modulation of opioid receptors were performed using graded concentrations of potential allosteric modulators in the absence and presence of increasing concentrations of the orthosteric ligand in a manner similar to a manner we recently described (20).
Profiling of W-18 and W-15 against the druggable GPCR-ome.
These studies were performed using our recently described PRESTO-Tango resource (14), which allows for the unbiased interrogation of drugs against the entire, druggable GPCR-ome.
Analgesic studies.
Antinociception was assessed using the writhing assay (15) and the radiant heat tail-flick assay (21). The writhing assay assesses the response to an i.p. irritant, either acetic acid or phenylquinone, which yield similar responses. It is a weak stimulus, with activity shown by a wide range of drugs as inactive in thermal assays, such as tail-flick and hot plate. In brief, groups of mice received 1% acetic acid (10 μl/g, i.p.) 15 minutes after receiving the test drug, and writhes were counted from 5–15 minutes after acetic acid injection The radiant heat tail-flick assay involves placing the tail under a radiant heat source and determining the latency, defined as the time the mouse permits its tail to remain within the light beam. After establishing baseline latencies for each mouse, they receive the test drug and were tested again. A maximal exposure of 10 seconds was used to minimize any tissue damage. Results are provided as percentage maximal effects (%MPE), defined as (test latency – baseline latency)/(10 – baseline latency) × 100.
Predictive pharmacology.
We used the similarity ensemble approach (22–24), essentially as previously described (22, 23, 25), to predict potential molecular targets for W-18 and W-15.
Data integrity and reproducibility.
All dose-response assays were performed several times and replicated independently, while the GPCR-ome analysis was performed twice in quadruplicate at different concentrations.
Statistics.
For the behavioral assays, ANOVA and the 2-tailed Student’s t test were used with significance predetermined to be at the 0.05 level, thus P < 0.05 was considered to be significant.
Study approval.
All animal studies were approved by the Institutional Animal Care and Use Committee of Memorial Sloan-Kettering Cancer Center and were conducted in strict accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Academies Press, 2011) in facilities accredited by the American Association for the Accreditation of Laboratory Animal Care.
Author contributions
GWP was responsible for study design, analysis of behavioral data, and murine receptor characterization and contributed to writing the paper. MHB contributed compounds and assisted with behavioral studies and writing the paper. SM supervised characterization of W-18 in radioligand-binding assays, and MDC supervised the generation of W-18 metabolites and performed metabolite profiling. XPH and TC performed functional assays and analyzed results. TJM performed radioligand-binding assays and analyzed results. BLR was responsible for overall project coordination and writing the paper. VLR and YXP provided chemical synthesis.
Supplementary Material
Acknowledgments
This work was supported in part by the National Institute of Mental Health Psychoactive Drug Screening Program Contract (HHSN-271-2013-00017-C) and PO1DA035764 from the National Institute of Drug Abuse to BLR, grants from the National Institute on Drug Abuse (DA06241), The MAYDAY Fund, and the Peter F. McManus Charitable Trust to GWP; a core grant from the National Cancer Institute (CA08748) to Memorial Sloan Kettering Cancer Center; and an equipment grant from the NIH (1S10OD010603) to MDC.
Version 1. 11/16/2017
Electronic publication
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information: JCI Insight. 2017;2(22):e97222. https://doi.org/10.1172/jci.insight.97222.
Contributor Information
Xi-Ping Huang, Email: ahuangxp@gmail.com.
Tao Che, Email: taoche@email.unc.edu.
Valerie Le Rouzic, Email: lerouziv@mskcc.org.
Ying-Xian Pan, Email: pany@mskcc.org.
Bryan L. Roth, Email: bryan_roth@med.unc.edu.
References
- 1. Knaus EE, Warren BK, Ondrus TA, inventors; Canadian Patents & Development Ltd., Assignee. Analgesic substituted piperidylidene-2-sulfon(cyan)amide derivatives. US patent 4,468,403. August 28, 1984.
- 2.Collier HO, Dinneen LC, Johnson CA, Schneider C. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br J Pharmacol Chemother. 1968;32(2):295–310. doi: 10.1111/j.1476-5381.1968.tb00973.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. EMCDDA. European Drug Report 2013: Trends and developments. http://www.emcdda.europa.eu/publications/edr/trends-developments/2013 Accessed November 6, 2017.
- 4. EMCDDA. European Drug Report 2014: Trends and developments. http://www.emcdda.europa.eu/publications/edr/trends-developments/2014 Accessed November 6, 2017.
- 5. EMCDDA. European Drug Report 2015: Trends and developments. http://www.emcdda.europa.eu/publications/edr/trends-developments/2015 Accessed November 6, 2017.
- 6.Gussow L. Toxicology Rounds: Designer Opiates 10,000x Stronger than Morphine. Emerg Med J. 2016;38(5):6 [Google Scholar]
- 7.Sustkova M. Synthetic Opioids, (re)Emerging Problem in Europe and North America. Int J Emerg Ment Health. 2015;17(4):694–695. [Google Scholar]
- 8. GONÇALVES J. Order Amending Schedules I and III to the Controlled Drugs and Substances Act (AH-7921, MT-45, W-18 and Lefetamine). Canada Gazette. http://gazette.gc.ca/rp-pr/p1/2016/2016-02-13/html/notice-avis-eng.php-nl3.2016.
- 9.White KL, et al. Identification of novel functionally selective κ-opioid receptor scaffolds. Mol Pharmacol. 2014;85(1):83–90. doi: 10.1124/mol.113.089649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu H, et al. Structure of the human κ-opioid receptor in complex with JDTic. Nature. 2012;485(7398):327–332. doi: 10.1038/nature10939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vardy E, et al. A New DREADD Facilitates the Multiplexed Chemogenetic Interrogation of Behavior. Neuron. 2015;86(4):936–946. doi: 10.1016/j.neuron.2015.03.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fenalti G, et al. Structural basis for bifunctional peptide recognition at human δ-opioid receptor. Nat Struct Mol Biol. 2015;22(3):265–268. doi: 10.1038/nsmb.2965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fenalti G, et al. Molecular control of δ-opioid receptor signalling. Nature. 2014;506(7487):191–196. doi: 10.1038/nature12944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kroeze WK, et al. PRESTO-Tango as an open-source resource for interrogation of the druggable human GPCRome. Nat Struct Mol Biol. 2015;22(5):362–369. doi: 10.1038/nsmb.3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Spiegel K, Kalb R, Pasternak GW. Analgesic activity of tricyclic antidepressants. Ann Neurol. 1983;13(4):462–465. doi: 10.1002/ana.410130418. [DOI] [PubMed] [Google Scholar]
- 16.Rothman RB, et al. Evidence for possible involvement of 5-HT(2B) receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation. 2000;102(23):2836–2841. doi: 10.1161/01.CIR.102.23.2836. [DOI] [PubMed] [Google Scholar]
- 17.Roth BL. Drugs and valvular heart disease. N Engl J Med. 2007;356(1):6–9. doi: 10.1056/NEJMp068265. [DOI] [PubMed] [Google Scholar]
- 18.Besnard J, et al. Automated design of ligands to polypharmacological profiles. Nature. 2012;492(7428):215–220. doi: 10.1038/nature11691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Majumdar S, et al. Generation of novel radiolabeled opiates through site-selective iodination. Bioorg Med Chem Lett. 2011;21(13):4001–4004. doi: 10.1016/j.bmcl.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang XP, et al. Allosteric ligands for the pharmacologically dark receptors GPR68 and GPR65. Nature. 2015;527(7579):477–483. doi: 10.1038/nature15699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lu Z, Xu J, Rossi GC, Majumdar S, Pasternak GW, Pan YX. Mediation of opioid analgesia by a truncated 6-transmembrane GPCR. J Clin Invest. 2015;125(7):2626–2630. doi: 10.1172/JCI81070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Laggner C, et al. Chemical informatics and target identification in a zebrafish phenotypic screen. Nat Chem Biol. 2011;8(2):144–146. doi: 10.1038/nchembio.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Keiser MJ, et al. Predicting new molecular targets for known drugs. Nature. 2009;462(7270):175–181. doi: 10.1038/nature08506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Keiser MJ, Roth BL, Armbruster BN, Ernsberger P, Irwin JJ, Shoichet BK. Relating protein pharmacology by ligand chemistry. Nat Biotechnol. 2007;25(2):197–206. doi: 10.1038/nbt1284. [DOI] [PubMed] [Google Scholar]
- 25.Yadav PN, et al. The presynaptic component of the serotonergic system is required for clozapine’s efficacy. Neuropsychopharmacology. 2011;36(3):638–651. doi: 10.1038/npp.2010.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.