Abstract
Subsets of human tumors are infiltrated with tumor antigen–specific CD8+ T cells (TILs) despite tumor progression. These TILs are thought to be inactivated by the immunosuppressive tumor microenvironment, through the engagement of inhibitory receptors such as CTLA-4 and PD-1. However, antigen-specific CD8+ TILs are not functionally inert, but are undergoing activation in situ. Here, we show that antigen-specific CD8+ TIL are actively proliferating, yet also undergo high rates of apoptosis, leading to a vicious cycle of activation and death that limits immune efficacy. Preventing CD8+ TIL apoptosis by Bcl-xL overexpression enabled accumulation and improved tumor control. Effective combination immunotherapy with an agonist 4-1BB monoclonal antibody plus either CTLA-4 or PD-L1 neutralization led to a marked accumulation of specific CD8+ TILs through decreased apoptosis rather than increased T-cell entry or proliferation. Our data suggest that antigen-driven apoptosis of CD8+ TILs is a barrier to effective spontaneous antitumor immunity and should be considered as a critical factor in the development of cancer immunotherapies.
Introduction
Tumors from many different cancer types can be infiltrated by CD8+ T cells (TILs (1), and their presence has positive prognostic value (2). Despite their prognostic importance, most T cell-infiltrated tumors grow progressively. Evidence has suggested that TILs become functionally impaired through a multitude of immune suppressive mechanisms within the tumor microenvironment, including engagement of the inhibitory receptor PD-1 by its major ligand PD-L1 (3), the activity of metabolic enzymes such as indoleamine-2,3-dioxygenase (3), and extrinsic suppression by FoxP3+ regulatory T cells (Tregs) (3). However, we have found that antigen-specific TILs appear activated, produce IFNγ in situ, and retain cytolytic potential despite expression of PD-1, LAG-3, and other inhibitory receptors (4). These results indicate that TILs within progressing tumors may not be as functionally inert as once thought, and raise the question of why these TILs are not able to control progressing tumors.
Immunotherapy that targets T-cell inhibitory receptors PD-1 or CTLA-4 can lead to impressive tumor control in patients (5, 6). Data from mouse models and human cancer patients suggest that blockade of inhibitory receptors predominantly acts on the pre-existing T-cell infiltrate residing in the tumor microenvironment at the time of treatment onset (7, 8). In mouse models, efficacy of anti-CTLA-4 and anti-PD-L1 combination therapies did not require new migration of T cells to the tumor from lymphoid tissues (7). Similarly, clinical studies demonstrated that patients with tumors infiltrated by CD8+ T cells are much more likely to benefit from checkpoint blockade immunotherapy (8). The efficacy of treatment has been assumed to correlate with a proliferative burst in TILs (8). However, our data indicated that TILs are already proliferating in situ (4). This led us to hypothesize that other mechanisms might account for failed T cell-mediated tumor elimination at baseline, as well as improved tumor control in the context of immunotherapy.
To test these hypotheses, we utilized the B16.SIY murine melanoma model. B16.SIY is a transplantable melanoma cell line that was originally isolated from a spontaneous murine melanoma, and then engineered to expresses the model antigen SIYRYYGL (SIY). B16.SIY will grow progressively when transplanted subcutaneously into wild type, syngeneic C57BL/6 mice (9). This system allows us to study the immune response against a progressing tumor. The SIY peptide is presented to CD8+ T cells in the context of H2-Kb (10), which enables monitoring of SIY-specific T cell responses in tumor-bearing hosts using IFN-γ ELISPOT, as well as SIY-pentamer staining and flow cytometry. CD8+ TILs in B16.SIY tumors express receptors that are targets for antibody-based immunotherapy, including PD-1, CTLA-4, and 4-1BB, allowing us to study how immunotherapy influences tumor antigen-specific T cell responses (4).
In the current study we found that antigen-specific TILs were not only undergoing continuous proliferation, but also apoptosis within the tumor microenvironment. This cycle of activation and death restrained T-cell numbers within the tumor and led to inadequate tumor control. In contrast to progressor tumors, spontaneously rejecting tumors showed five-fold higher numbers of SIY-specific TILs without evidence of apoptosis. Overexpressing the anti-apoptotic molecule Bcl-xL in T cells reduced TIL apoptosis and increased TIL accumulation in progressor tumors, and anti-4-1BB combination immunotherapies promoted tumor control by a mechanism associated with prevention of TIL apoptosis. Therefore, tumor antigen–specific TIL apoptosis appears to be a critical limiting factor of T-cell immunity against tumors.
Materials and Methods
Mice
C57BL/6 and Rag2−/− mice were from Taconic. Lckpr–Bcl-xL mice were a gift from Dr. M. Alegre (U. Chicago). Transgenic 2C TCR mice were bred in our facility (11). All mice were housed at University of Chicago in specific pathogen-free conditions in accordance with the National Institute of Health animal care guidelines. Autochthonous melanoma mice were described previously (11). All experiments were approved by the Institutional Animal Care and Use Committee at The University of Chicago and followed international guidelines.
Cell Culture and Inoculation
B16.F10, MC57, and 1969 cells were engineered to express SIYRYYGL, a peptide isolated from a random peptide library that binds H-2Kb. The resulting cell lines B16.SIY, MC57.SIY, and 1969.SIY, respectively, were cultured in DMEM with 10% FBS and penicillin and streptomycin, as described (4). Cells (2 × 106) were inoculated subcutaneously into the right flank of each animal.
Cells were cultured for one month after thawing. Cells were tested yearly for mycoplasma contamination using the HEK-Blue system (Invivogen). Our laboratory previously generated B16.SIY and 1969.SIY cell lines (4, 9). MC57.SIY cells were a gift from Dr. Hans Schreiber (U. Chicago) (12). Cell lines were not re-authenticated or tested for cell line cross-contamination in the past year.
Antibody Treatments
All therapeutic and depleting monoclonal antibodies (mAbs) were purchased from Bio X Cell (West Lebanon, NH). Antibodies (100 µg) to 4-1BB (LOB.12.3), CTLA-4 (UC10-4F10-11), and PD-L1 (10F.9G2) were injected intraperitoneally seven days after tumor inoculation. For tumor outgrowth, mAbs were given on days 7, 10, 13, and 16 after tumor inoculation. To deplete CD4−, CD8− or NK1.1-expressing cells, mAbs (250 µg) to CD4 (GK1.5), CD8 (2.43), NK1.1 (PK136) were given 24 hours before tumor inoculation, and then every seven days.
FTY720 Administration
FTY720 (5 µg per mouse, Enzo) was dissolved in DMSO and then diluted in PBS before administration daily by oral gavage.
Flow Cytometry
Cells were analyzed on either a BD Fortessa or LSR-II cytometer. SIY-loaded pentamers were from ProImmune. The following antibodies were used in analyses: BD Biosciences: CD45 (30-F11), CD3 (145-2C11), CD4 (RM4-5), CD8 (53-6.7), BrdU (Bu20a), Ki-67 (35/Ki-67), and active caspase-3 (C92-605); eBioscience: LAG-3 (C9B7W), 4-1BB (17B5); Biolegend: PD-1 (RMPI-30); Thermo-Fisher: γH2AX (CR55T33). Fixable viability dyes were used to gate out dead cells and were purchased from eBioscience. Tumors, lymph nodes and spleens were dissociated through a 70 µM cell strainer to generate cell suspensions. Tumor suspensions were centrifuged over a Ficoll-hypaque gradient to isolate live mononuclear cells. Cell suspensions were stained with antibodies in PBS containing 1% FBS for 20 min at room temperature. For intracellular antigens, cells were fixed and permeabilized in FoxP3 buffer (eBioscience) for 30 min at room temperature, washed, and stained with intracellular antibodies for 30 min at room temperature. For annexin V staining, cells were first stained with extracellular antibodies and fixable viability dyes. Immediately before analysis, cells were stained using the annexin V staining kit (BD Biosciences, catalog number 559763).
Adoptive Transfer
Splenocytes were isolated from 2C or 2C Bcl-xL mice and red blood cells were lysed with Gey’s solution. Cells were stimulated with plate-bound CD3 and CD28 antibodies for 4–6 days to generate activated effector cells. One million cells were then transferred intravenously to mice with 7-day established tumors.
BrdU Administration
BrdU (0.8 mg, BD) was given intraperitoneally 24 h before animal sacrifice. After staining extracellular antigens, cells were fixed and permeabilized with FoxP3 buffer (eBioscience) for 30 min at room temperature. Cells were resuspended in PBS with DNAse I (300 µg/mL, Roche) at 37 °C for 1 h. Cells were subsequently stained with anti-BrdU antibody at room temperature for 30 min.
Gene Expression Profiling
The microarray data are available in the Gene Expression Omnibus database under accession no. GSE79919.
Statistics
GraphPad Prism was used to compute all statistical tests. Data represent mean ± SEM. Mann-Whitney U, Kruskal-Wallis, and Two-way ANOVA statistical tests were calculated in GraphPad Prism: *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Results
Tumor antigen-specific CD8+ T cells proliferate in the tumor microenvironment
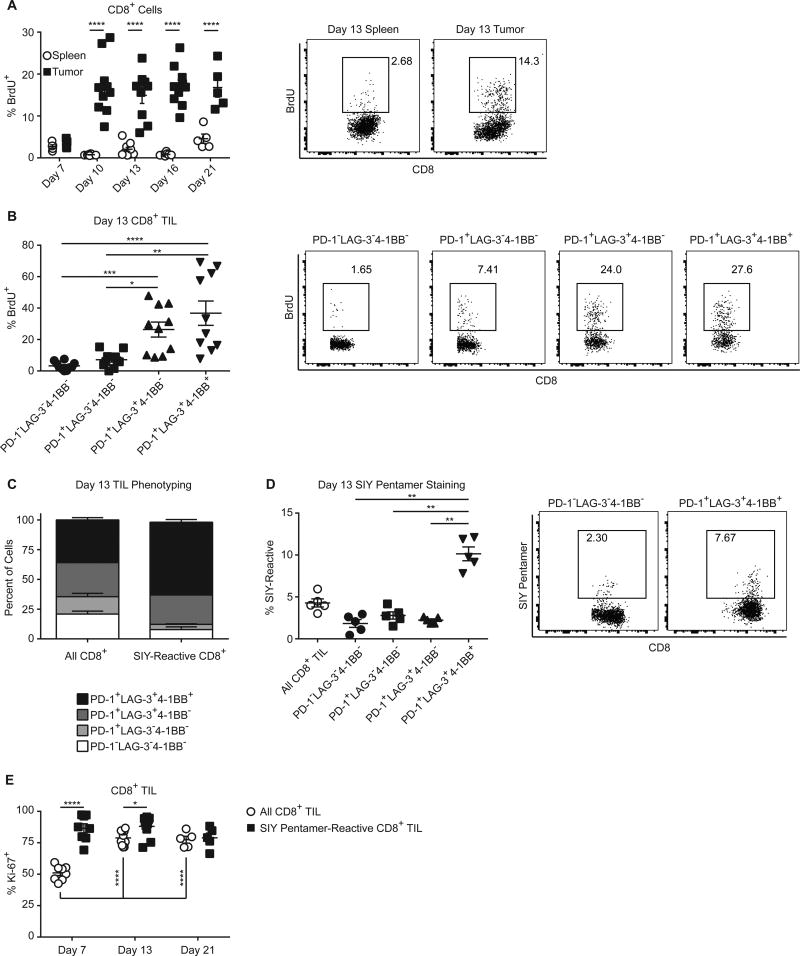
We previously found that antigen-specific TILs acquired a phenotype that included PD-1, LAG-3, and 4-1BB expression (4). Despite inhibitory receptor expression, however, these TILs proliferated within the tumor microenvironment (4). To rule out the possibility that TILs proliferated at early stages of tumor growth but lost proliferation upon persistent tumor antigen exposure, we examined TIL proliferation over time as tumor growth was progressing. To detect TIL proliferation, we pulsed mice bearing subcutaneous B16.SIY tumors with BrdU 24 hours before sacrifice at various time points (Fig. 1A). Although little BrdU was incorporated seven days post tumor injection, we observed consistent BrdU incorporation by TILs 10 – 21 days after tumor injection. Incorporation of BrdU was significantly greater in the TILs than in the spleen, indicating that TILs were constantly proliferating in the tumor microenvironment. To analyze antigen-specific TILs, we used Kb/SIY pentamers. Unfortunately, the BrdU staining interfered with the detection of pentamer-reactive cells. Expression of the surface receptors LAG-3 and 4-1BB identifies antigen-specific TILs (4), therefore we phenotyped TILs for LAG-3 and 4-1BB while measuring BrdU incorporation (example phenotyping in Supplementary Fig. S1A). The most actively proliferating subpopulations of TILs expressed LAG-3 (Fig. 1B), suggesting that antigen-specific TILs were proliferating within the tumor microenvironment during tumor progression. LAG-3+ populations remained the most proliferative subset of TILs over the course of tumor outgrowth (Supplementary Fig. S1B). Consistent with our previous finding that this surface phenotype enriches for antigen specificity (4), 80%–90%of SIY-reactive TIL expressed LAG-3, or LAG-3, and 4-1BB (Fig. 1C), and conversely the PD-1+LAG-3+4-1BB+ population was 3-fold more highly enriched for SIY-reactive TIL than the other phenotypic subsets (Fig. 1D). This phenotype was consistent over all time points analyzed (Supplementary Fig. S1C), indicating that antigen-specific TILs proliferate for the duration of tumor outgrowth. To confirm this directly, we analyzed Ki-67 expression as an alternative indicator of proliferation, as Ki-67 staining was compatible with SIY staining. Nearly all SIY-reactive TILs expressed Ki-67 at every time point analyzed (Fig. 1E). Despite this constant TIL proliferation, total TILs or SIY-reactive TILs did not significantly accumulate over the time points analyzed (Supplementary Fig. S1D and 1E). Therefore, some other process was restraining TIL accumulation despite constant proliferation.
Figure 1. CD8+ T cells proliferate in the tumor microenvironment.
(A) C57BL/6 mice were injected subcutaneously with 2×106 B16.SIY tumor cells on day 0 and sacrificed at the indicated time points. One day prior to sacrifice, mice were injected intraperitoneally with BrdU (0.8 mg). Spleens and tumors of mice were analyzed via flow cytometry for BrdU incorporation in CD8+ T cells. Day 7, n = 5; day 10, n = 10; day 13, n = 10; day 16, n = 10; day 21, n = 5. Data pooled from 3 independent experiments, two-way ANOVA. (B) Analysis of TILs for the incorporation of BrdU on day 13, combined with phenotyping for the expression of PD-1, LAG-3, and 4-1BB. Data pooled from 2 independent experiments. One-way ANOVA, n = 10. (C) and (D) TILs were stained with SIY pentamer and analyzed for the expression of PD-1, LAG-3, and 4-1BB on day 13 post tumor injection. Representative data from greater than 3 independent experiments, n = 5, one-way ANOVA. (E) Tumors of mice bearing B16.SIY tumors for 13 days were analyzed for SIY pentamer-reactive cells and Ki-67 expression via flow cytometry, day 7, n = 8; day 13, n = 10; day 21, n = 5. Data pooled from 3 independent experiments. Two-way ANOVA.
Proliferating CD8+ T cells undergo apoptosis in the tumor microenvironment
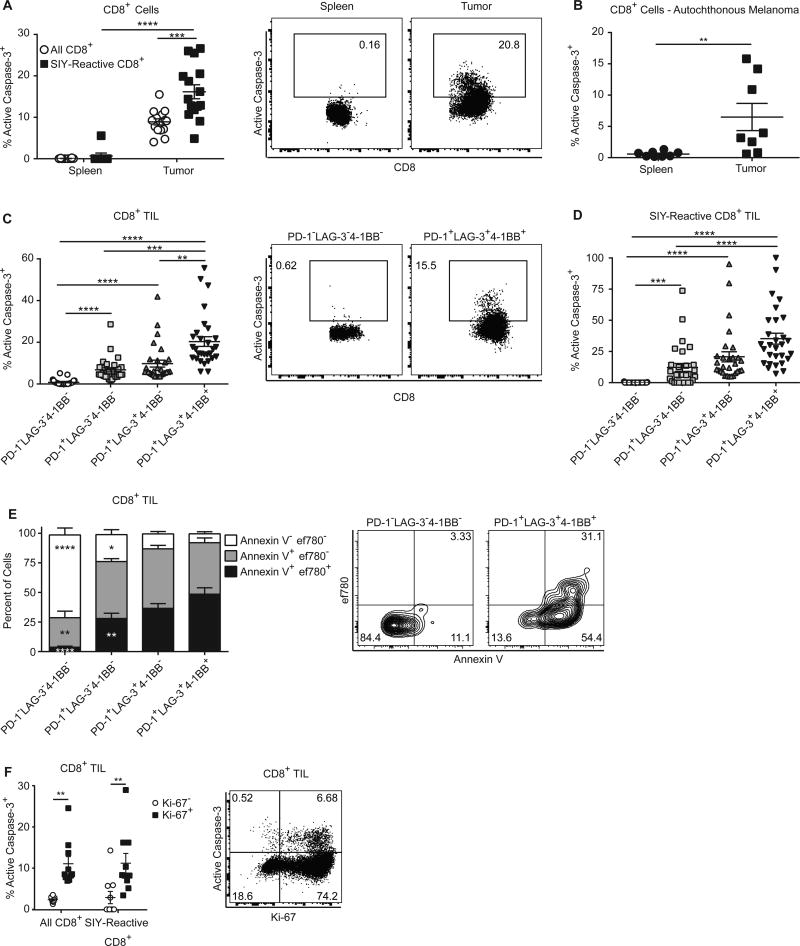
To investigate how TILs can constantly proliferate without accumulating, we asked whether these cells were simultaneously undergoing apoptosis within the tumor microenvironment. Caspase-3 is activated via cleavage during apoptosis, and effector T cells containing active caspase-3 are undergoing cell death (13, 14). Therefore, we utilized flow cytometry to detect intracellular active caspase-3. We found that at day 13 post tumor injection, a large fraction of TIL and SIY-reactive TIL contained active caspase-3 within the tumor but not in the spleen (Fig. 2A). Caspase-3 staining among TILs was consistent throughout tumor growth (Supplementary Fig. S2A). To test if TILs apoptosed in non-transplantable tumor settings, we used an autochthonous, inducible B-RafV600E/PTEN−/− melanoma model. CD8+ T cells within RafV600E/PTEN−/− melanomas also underwent apoptosis (Fig. 2B) (11). Thus, CD8+ T-cell apoptosis occurs specifically in the tumor microenvironment of tumors models with very different developmental origins and latency times.
Figure 2. CD8+ T cells undergo apoptosis in the tumor microenvironment.
(A) CD8+ cells from tumors and spleens of mice bearing B16.SIY tumors for 13 days were stained with SIY pentamer and analyzed for the presence of the active form of caspase-3 via flow cytometry. Data pooled from 3 independent experiments. Two-way ANOVA, n=15. (B) An autochthonous melanoma model driven by the melanocyte specific, inducible expression of activated BRAF and deletion of PTEN was analyzed for the presence of active caspase-3 in CD8+ T cells from spleens and melanomas via flow cytometry. Data pooled from 2 independent experiments. Mann-Whitney U test. (C) TIL from B16.SIY tumor-bearing mice were analyzed for active caspase-3 as in (A) and for expression of PD-1, LAG-3, and 4-1BB via flow cytometry. Data pooled from 6 independent experiments. One-way ANOVA, n = 29. (D) TIL were stained with SIY pentamer and analyzed as in (C), n = 29. Data pooled from 6 independent experiments. (E) TIL from B16.SIY were isolated and analyzed for the expression of PD-1, LAG-3, and 4-1BB, as well as binding to fluorescently labeled annexin V and Fixable Viability Dye eFluor 780, n = 10. Data pooled from 2 independent experiments. Two-way ANOVA. The indicated statistical differences are compared to the PD-1+LAG-3+4-1BB+ populations. (F) TILs from mice bearing 13 day-established B16.SIY tumors were stained with SIY pentamer and analyzed for Ki-67 and active caspase-3 expression via flow cytometry, n = 10. Data pooled from 2 independent experiments, two-way ANOVA.
We wanted to better understand the relationship between TIL surface phenotype, proliferation, and apoptosis. Phenotypic analysis revealed that TIL co-expressing LAG-3 and 4-1BB showed the highest rate of apoptosis (Fig. 2C), which suggested that antigen-specific TILs were simultaneously proliferating and dying. LAG-3+4-1BB+ TILs maintained the highest rates of apoptosis over time (Supplementary Fig. S2B). The rate of apoptosis of LAG-3+4-1BB+ cells was not significantly changed over the analyzed time points, indicating that even at fairly early times, LAG-3+4-1BB+ TIL underwent apoptosis. This suggested that the acquisition of this phenotype in the tumor environment was associated with high rates of proliferation and apoptosis. Direct measurement of active caspase-3 in SIY-reactive TIL revealed similar patterns of increased apoptosis in LAG-3+4-1BB+ cells (Fig. 2D and Supplementary Fig. S2B). Antigen-specific TILs were therefore undergoing both proliferation and apoptosis in the tumor microenvironment, and both proliferation and apoptosis correlated with LAG-3 and 4-1BB expression.
We also stained TILs with annexin V and a cell-impermeable viability dye to measure different stages of apoptosis. The LAG-3+4-1BB+ population had decreased non-apoptotic, annexin V− cells, and far more cells in late apoptosis (Fig. 2E), indicating that many of the LAG-3+4-1BB+ were in the final stages of cell death, thus confirming that the ultimate fate for these TILs was death.
Both proliferating and dying TILs were LAG-3+4-1BB+, suggesting that the same TILs were undergoing both proliferation and apoptosis. To directly confirm this, we measured active caspase-3 and Ki-67 expression in total TILs and SIY-reactive TILs. Active caspase-3 was highest among TILs that were Ki-67+, directly indicating that apoptosis of proliferating cells was occurring (Fig. 2F). Antigen-specific CD8+ TILs were therefore trapped in a cycle of proliferation and apoptosis that potentially limited TIL accumulation and antitumor immunity.
Prevention of TIL apoptosis leads to improved tumor control
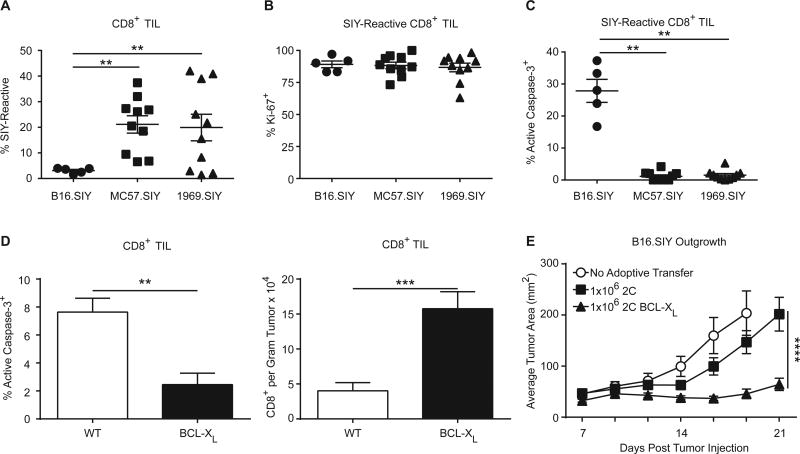
To determine the relationship between apoptosis, accumulation of TILs, and immune-mediated tumor control, we studied TIL apoptosis in syngeneic tumor cell lines (MC57.SIY and 1969.SIY) which are spontaneously rejected by the adaptive immune system (15, 16). This allowed comparison of TIL apoptosis in progressing versus regressing tumors, to determine if TIL apoptosis was specific to tumor progression. When compared to progressing B16.SIY tumors, a 4–5 fold expansion of SIY-reactive TIL was observed in MC57.SIY and 1969.SIY tumors (Fig. 3A). This expansion did not appear to be the result of changes in proliferation, since the Ki-67+ fraction of T cells was similar in progressing vs regressing tumors (Fig. 3B). However, apoptosis was only observed among TILs from B16.SIY progressing tumors (Fig. 3C). We previously found that CD8+ TILs in MC57.SIY and 1969.SIY tumors did not significantly express LAG-3 or 4-1BB (4). Less CD8+ TIL apoptosis in regressing tumors was consistent with decreased LAG-3+4-1BB+ CD8+ TIL in regressing tumors, as LAG-3+4-1BB+ CD8+ TIL are the most apoptotic (Fig. 1C). These results indicate that spontaneous TIL apoptosis is a feature of progressing and not regressing tumors.
Figure 3. Decreased TIL apoptosis leads to increased tumor control.
(A) C57BL/6 mice were injected with 2 × 106 B16.SIY, 2 × 106 MC57.SIY, or 2 × 106 1969.SIY cells on day 0. On day 7, mice were sacrificed and analyzed for SIY-reactive TIL via flow cytometry. One-way ANOVA, B16.SIY, n = 5; MC57.SIY and 1969.SIY, n = 10. (B) Similar conditions to (A) Ki-67 expression of SIY-reactive TIL is shown. One-way ANOVA; B16.SIY, n = 5; MC57.SIY and 1969.SIY, n = 10. (C) As in (A), the fraction of SIY-reactive cells with active caspase-3 is shown. One-way ANOVA; B16.SIY, n = 5; MC57.SIY and 1969.SIY, n = 10. All data in (A) – (C) is pooled from 2 independent experiments. (D) Bcl-xL or WT mice were injected with 2 × 106 B16.SIY cells on day 0. On day 13, mice were sacrificed and analyzed for active caspase-3 expression and to enumerate the number of TIL. Data pooled from 2 independent experiments. Mann-Whitney U test, n = 9. (E) 2C or 2C Bcl-xL splenocytes were activated in vitro with plate-bound antibodies to CD3 and CD28 to generate activated effector CD8+ T cells. T cells (1 × 106) were transferred to mice bearing B16.SIY tumors on day 7 post-tumor injection. No Adoptive Transfer, n = 10; 2C, n = 10; 2C Bcl-xL, n = 20. Data pooled from 2 independent experiments, two-way ANOVA.
To modulate apoptosis in T cells, we used the Lckpr-Bcl-xL mouse that over-expresses the anti-apoptotic molecule Bcl-xL in T cells (Bcl-xL mice). Bcl-xL mice challenged with B16.SIY tumors had significantly reduced apoptosis of TIL, and an increase in the number of infiltrating TIL (Fig. 3D). Proliferation between TILs from WT and Bcl-xL mice was not different, indicating the increase of TILs was, indeed, correlated with a reduction in apoptosis (Supplementary Fig. S3A). However, these mice paradoxically had a reduced percentage of SIY-reactive TILs (Supplementary Fig. S2A), likely because T cells of multiple specificities were allowed to accumulate. In order to restrict Bcl-xL expression to tumor antigen–specific CD8+ T cells, we crossed Bcl-xL mice to 2C TCR transgenic mice, the T cells from which recognize the SIY peptide in the context of Kb (2C Bcl-xL mice) (17). Splenocytes from either 2C or 2C Bcl-xL mice were activated in vitro with CD3/CD28 antibodies to generate primed effector T cells, which were then transferred intravenously to mice bearing seven-day established B16.SIY tumors. Bcl-xL expression in 2C T cells resulted in significantly improved tumor control than WT 2C cells, indicating that inhibiting effector T-cell apoptosis is sufficient for more effective immune-mediated tumor destruction in vivo (Fig. 3E).
Anti-4-1BB plus checkpoint blockade therapy leads to tumor regression and decreased TIL apoptosis
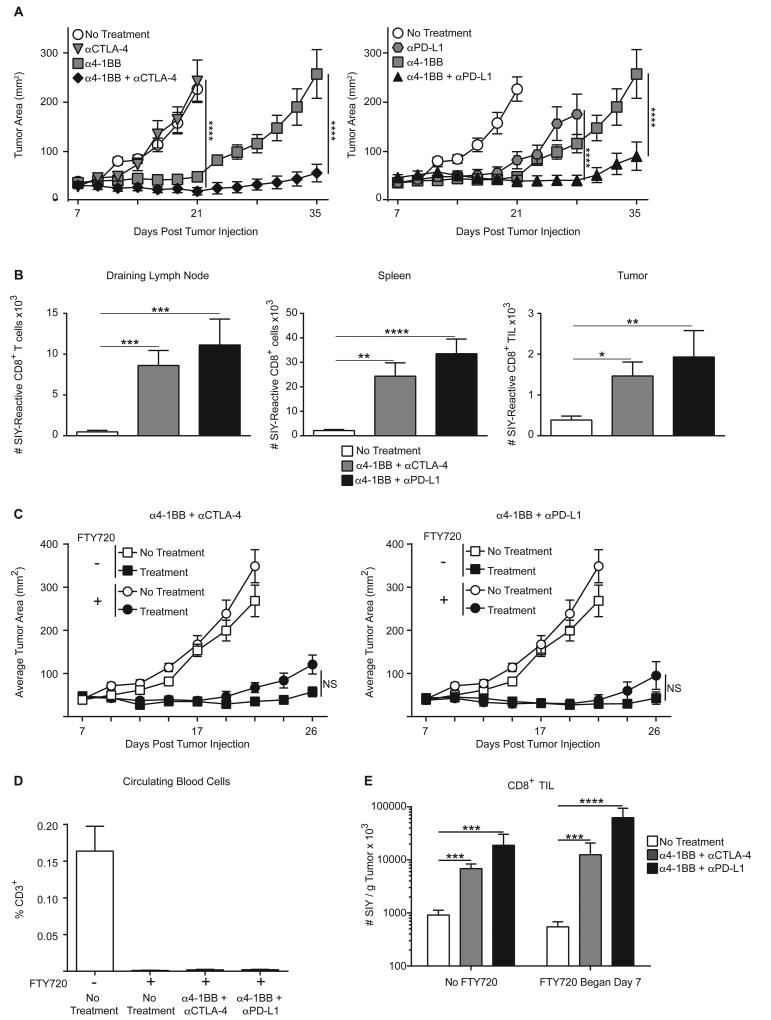
Because many TILs undergoing apoptosis express 4-1BB (Fig. 2C), and agonist antibodies that induce 4-1BB signaling drive activation of NF-κB, a pro-survival transcription factor (18), we investigated whether engagement of 4-1BB could reduce TIL apoptosis and improve antitumor immunotherapy. Clinical trials are testing anti–4-1BB in combination with blockade of immune inhibitory receptors, so we focused on an agonist 4-1BB antibody with either CTLA-4 or PD-L1 blockade. These combinations led to enhanced tumor control of established B16.SIY tumors (Fig. 4A). Immunotherapy led to increased SIY-reactive CD8+ T cells in tumor-draining lymph node, spleen, and within the tumor of treated mice (Fig. 4B). CD8+ cells were required for rejection, as RAG2−/− animals had no response to immunotherapy, and depletion of CD8+ cells, but not of CD4+ or NK1.1+ cells, abrogated the therapeutic effects (Supplementary Fig. S3A–D). Because we found SIY-reactive CD8+ cell accumulation both in secondary lymphoid tissues and in the tumor, it was important to determine if the trafficking of newly primed cells to the tumor was necessary for TIL accumulation and tumor regression. The trafficking of lymphocytes from lymph nodes is dependent on the receptor S1P1, whose action can be inhibited by the molecule FTY720. FTY720 treatment has been shown to effectively deplete circulating lymphocytes by preventing their ability to exit lymphoid tissues and enter back into circulation. Therefore, we treated B16.SIY tumor-bearing mice with FTY720 along with administration of anti-4-1BB combination immunotherapy. Daily FTY720 beginning on day 7 post-tumor injection did not significantly impact tumor control (Fig. 4C), indicating that therapeutic efficacy was largely mediated by TILs already present within the tumor when immunotherapy began. FTY720 was confirmed to efficiently block trafficking of T cells into the blood (Fig. 4D). However, tumor-bearing mice treated with FTY720 and immunotherapy had no deficit in the numbers of SIY-reactive CD8+ T cells within the tumor microenvironment compared to vehicle-treated mice receiving immunotherapy (Fig. 4E), indicating that TILs were expanding within the tumor site independently of cells expanding in the lymph nodes. Therefore, TIL accumulation was being driven by an intratumoral process.
Figure 4. 4-1BB combination immunotherapy leads to tumor regression and TIL accumulation through an intratumoral process.
(A) C57BL/6 mice were subcutaneously inoculated with B16.SIY cells (2 × 106). Tumors were established for seven days, then cohorts were treated with either single antibodies against 4-1BB, CTLA-4, or PD-L1, or combinations of anti–4-1BB + anti–CTLA-4 or anti–4-1BB + anti–PD-L1. Antibodies were given intraperitoneally on days 7, 10, 13 and 16 post-tumor injection. Each mouse received 100 µg of each indicated antibody at each time point, n = 10 mice per cohort. Data pooled from 2 independent experiments, two-way ANOVA. (B) Combination immunotherapy was given as in (A), but mice were sacrificed on day 13 and tumor draining lymph nodes, spleens and tumors were analyzed for SIY-reactive CD8+ T cells via flow cytometry. Data pooled from 2 independent experiments. One-way ANOVA, n = 10. (C) C57BL/6 mice were injected subcutaneously with B16.SIY cells (2 × 106). Tumors were established for 7 days, at which point daily oral administration of FTY720 was begun. Mice received anti–4-1BB (100 µg) + anti–CTLA-4 (100 µg) or anti–4-1BB (100 µg) + anti–PD-L1 (100 µg) intraperitoneally on days 7, 10, 13, and 16 post tumor injection, n = 10. Data pooled from 2 independent experiments, two-way ANOVA. (D) Blood from mice from (C) was analyzed at the endpoint of the experiment for circulating CD3+ cells using flow cytometry. (E) C57BL/6 mice were injected subcutaneously with B16.SIY cells (2 × 106). Tumors were established for 7 days, at which point daily oral administration of FTY720 was begun. Mice received anti–4-1BB (100 µg) + anti–CTLA-4 (100 µg) or anti–4-1BB (100 µg) + anti–PD-L1 (100 µg) intraperitoneally on days 7, and 10 post tumor injection, and were analyzed on day 13. The accumulation of SIY-reactive TIL was analyzed with flow cytometry. Data pooled from 3 independent experiments, n = 15, one-way ANOVA.
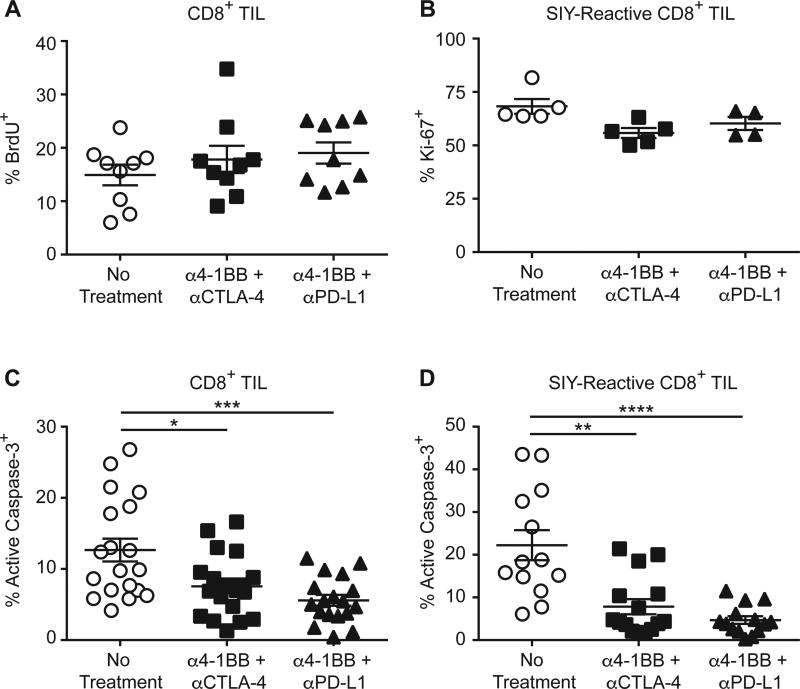
Because trafficking from lymphoid tissues was not required for SIY-reactive TIL accumulation after immunotherapy, we reasoned that either increased proliferation or decreased apoptosis of antigen-specific TILs was occurring. After immunotherapy, we found no difference in the proliferation of TILs (Fig. 5A). SIY-reactive TILs also showed no increase in Ki-67 expression (Fig. 5B). However, both the overall population of TILs and the SIY-reactive TIL subpopulation showed a significant reduction in active caspase-3 (Fig. 5C and 5D). Agonistic anti-4-1BB combination immunotherapy therefore appears to mediate tumor regression by boosting antigen-specific TIL accumulation via reduction of TIL apoptosis.
Figure 5. Anti-4-1BB combination immunotherapy decreases TIL apoptosis.
(A) C57BL/6 mice were inoculated subcutaneously with 2 × 106 B16.SIY cells and treated with immunotherapy on day 7 and day 10 post tumor cell injection. BrdU (0.8 mg per mouse) was administered intraperitoneally 24 h before sacrifice of each cohort. On day 13 after tumor cell injection the tumors were analyzed using flow cytometry to detect CD8+ T cells with incorporated BrdU; n = 9. Data pooled from 2 independent experiments. (B) As in (A), but Ki-67 expression was analyzed via flow cytometry. No Treatment, n = 5; ant–4-1BB + anti-CTLA-4, n = 5; anti-4-1BB + anti-PD-L1, n = 4. (C) As in (A), but TILs were analyzed for the presence of active capase-3 via flow cytometry, Data pooled from 4 independent experiments. No Treatment, n = 19; anti-4-1BB + anti-CTLA-4, n = 20; anti-4-1BB + anti-PD-L1, n = 18; one-way ANOVA. (D) As in (A), but TIL were analyzed for the presence of active capase-3 and SIY-reactivity via flow cytometry. Data pooled from 3 independent experiments. No Treatment, n = 13; anti-4-1BB + anti-CTLA-4, n = 15; anti-4-1BB + anti-PD-L1, n = 14; one-way ANOVA.
TILs Have Increased DNA Damage
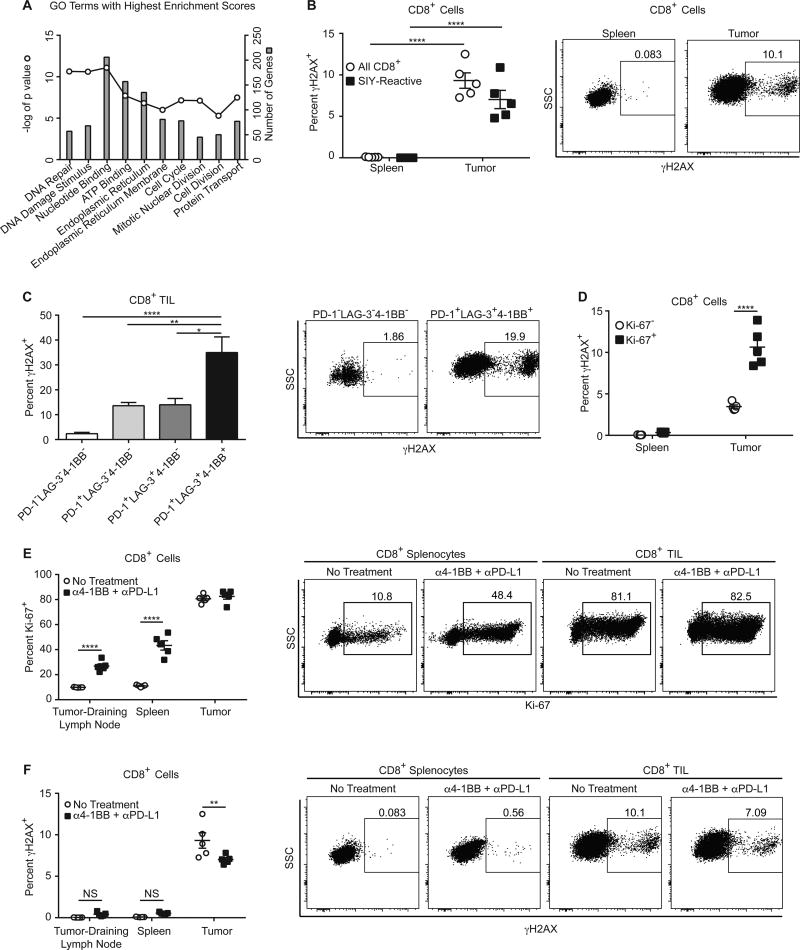
To better understand the mechanisms behind TIL apoptosis, we compared gene expression profiles from the PD-1+LAG-3−4-1BB− cell population to the PD-1+LAG-3+4-1BB+ cell population (4). We chose these populations as their shared expression of PD-1 indicates they have recently seen their cognate antigen, but the PD-1+LAG-3+4-1BB+ population is both much more proliferative and apoptotic. Using The Database for Annotation, Visualization and Integrated Discovery (DAVID) v6.8, we determined enriched gene ontology (GO) terms from 1616 genes upregulated 2-fold or more in the PD-1+LAG-3+4-1BB+ TIL. The two GO terms with the highest enrichment scores were associated with DNA damage (Fig. 6A). As DNA damage can lead to apoptosis, we measured DNA damage in TILs. Phosphorylated histone 2A, also known as γH2AX, is a marker of double-stranded DNA breaks readily detectable by robust flow cytometry antibodies. Using intracellular staining for γH2AX, we found significantly greater γH2AX staining in the total CD8+ and SIY-reactive TIL, as compared to the spleen (Fig. 6B). γH2AX detection was the highest among the PD-1+LAG-3+4-1BB+ TIL (Fig. 6C). Ki-67+ TILs contained significantly more γH2AX than did Ki-67− TIL (Fig. 6D). However, γH2AX levels between CD8+ splenocytes that were Ki-67+ and Ki-67− were the same, indicating that proliferation itself was not sufficient to explain positive staining, but rather the accumulation of DNA damage was only found among proliferating T cells within the tumor microenvironment. We wanted to test more directly whether increased proliferation alone was responsible for increased detectable DNA damage, so we used 4-1BB combination immunotherapy to induce high proliferation in the CD8+ T cells in peripheral lymphoid organs. Immunotherapy greatly increased the proliferation of these T cells, as measured by increased Ki-67 expression (Fig. 6E). However, γH2AX levels in CD8+ T cells in the spleen and lymph nodes were not significantly increased after immunotherapy (Fig. 6F). Therefore, DNA damage in proliferating CD8+ T cells occurs at high levels specifically within the tumor microenvironment, providing a potential cause of TIL apoptosis.
Figure 6. TIL Have Increased DNA Damage.
(A) Depicted are the top 10 enriched GO terms identified by DAVID from genes upregulated in PD-1+LAG-3+4-1BB+ TIL compared to PD-1+LAG-3−4-1BB− TIL, the −log(P value) of each, and the number of genes from the upregulated genes that were included the each GO term. (B) Day 13 CD8+ cells from the spleens and tumors of mice were analyzed for γH2AX; n = 5, two-way ANOVA. (C) TIL from day 13 tumors were analyzed for expression of PD-1, LAG-3, 4-1BB, and γH2AX; n = 10, one-way ANOVA. Data pooled from 2 independent experiments. (D) Day 13 CD8+ cells were analyzed for expression of Ki-67 and γH2AX; n = 5, two-way ANOVA. (E) C57BL/6 mice were injected subcutaneously with B16.SIY cells (2 × 106) on day 0. On days 7 and 10, mice were treated with 100 µg anti-4-1BB plus 100 µg anti-PD-L1. On day 13, CD8+ cells were analyzed for expression of Ki-67; n = 5, two-way ANOVA. (F) The same CD8+ cells from (E) were also analyzed for γH2AX; n = 5, two-way ANOVA.
Discussion
We have previously found that TILs can retain IFNγ production and cytolytic potential, yet tumors progress nonetheless (4), leaving as an open question why TILs fail to eradicate the tumors in which they reside. CD8+ TIL function and gene expression partially resemble exhausted CD8+ T cells from chronic LCMV infections, raising the question of whether a unique type of dysfunction occurs in TILs that enables tumor progression (4). Our current data suggest that TILs undergo a cycle of activation and proliferation followed by death within the tumor microenvironment that leads to their failed expansion, allowing tumor outgrowth. When this cycle is interrupted, either through the enforced expression of the anti-apoptotic factor Bcl-xL or through anti-4-1BB combination immunotherapy, antigen-specific TIL accumulation is rescued and immune-mediated tumor control ensues. The fact that TILs are not as functionally inert as once thought is supported by other findings both in mouse models and with human tumors (19, 20). Our data suggest that rescuing TIL from apoptosis may be a vital strategy to increase immune-mediated tumor control, either to increase TIL number or to allow TILs to carry out their functions for a longer period of time. Whether additional functional improvements also contribute to better TIL efficacy after immunotherapy is not formally known, and it is possible additional functional alterations could also contribute to tumor regression. Although our results clearly demonstrate that TIL apoptosis represents one mechanism by which proliferating T cells fail to accumulate in the tumor microenvironment, it is not excluded that T-cell exit from the tumor may contribute. However, the LAG-3+4-1BB+ phenotype characteristic of proliferating antigen-specific CD8+ T cells within the tumor microenvironment is not found in spleens or lymph nodes of tumor-bearing mice at any time point (4). Thus, if significant numbers of TILs do recirculate to other sites, they would need to change their surface phenotype rapidly, which seems unlikely.
We observed that simultaneous TIL proliferation and apoptosis occurred in progressively growing tumors, but not in tumors that underwent spontaneous rejection. In addition, TIL apoptosis was only seen in activated TILs expressing PD-1, with a higher percentage among those cells additionally expressing LAG-3 and 4-1BB. Apoptosis was not observed in the PD-1− TIL population, suggesting that the mere presence of T cells within the tumor microenvironment is not sufficient to result in T-cell death. Rather, those T cells undergoing antigen-driven activation in situ are those that show proliferation and apoptosis, and only under conditions when the tumor is not eliminated. These data suggest apoptosis results from an active process involving TCR engagement and is a critical component of T-cell dysfunction resulting from chronic antigen stimulation. The detailed biochemical mechanisms driving TIL apoptosis remain to be pursued in future studies, but our current data suggests that accumulated DNA damage in TILs may play a role. Such DNA damage could result from the reactive oxygen species (ROS) known to be produced in the context of a growing cancer, or the hypoxia and tissue stress that characterize solid tumors (21, 22). This result is consistent with published data indicating that p53−/− T cells are better able to control tumors upon adoptive transfer in vivo (23).
Our experiments with FTY720 indicate that major antitumor effects of anti-4-1BB combination immunotherapy can be exerted on the TILs present at baseline. This was the case despite the marked increase in tumor antigen-specific T cells in the tumor-draining lymph nodes in treated mice. These results suggest that 4-1BB expressed on TILs may be targeted directly by agonist 4-1BB immunotherapy. These data are also consistent with previous observations from both mouse and human studies that a preexisting T-cell infiltrate is associated with an increased likelihood of response to immunotherapy (7, 8). Thus, therapeutic blockade of inhibitory signals or enhancement of costimulatory signals may both act predominantly on tumor-infiltrating CD8+ cells for the immediate therapeutic effect observed.
The effects of costimulation in the tumor microenvironment are relatively under-studied, but are quickly becoming of interest. Costimulation through CD28 signaling is required for anti–PD-1 efficacy (24, 25). Blockade of CD28 inhibited the T-cell proliferation that normally occurred after PD-1 blockade (25). T cell–expressed ICOS is also necessary for the optimal effects of anti–CTLA-4–based immunotherapy (26). In our current study, we show that the delivery of additional costimulation signals through an agonist 4-1BB antibody increased the efficacy of PD-L1 or CTLA-4 blockade. However, 4-1BB combination immunotherapy did not increase the proliferation of TILs, but rather reduced TIL apoptosis. This is in line with previous findings that show that 4-1BB improves the survival of CD8+ T cells in various other model systems (27–29). A pro-survival role of 4-1BB is also supported by studies demonstrating increased persistence of CAR T cells that include a 4-1BB signaling domain (30). As a member of the TNFR superfamily, it is conceivable that 4-1BB ligation mediates enhanced TIL survival through the NF-κB pathway, which is critical for T cell–mediated tumor control in vivo (15, 31).
Supplementary Material
Acknowledgments
We would like to acknowledge Dr. Anita Chong, Dr. Maria-Luisa Alegre, and Dr. Justin Kline for insightful comments about this study.
Financial Support: This work was supported by R35CA210098 from the NCI. BH was funded by Cancer Biology Training Grant T32-CA09594. The flow cytometry core facility at the University of Chicago was funded by Cancer Center Support Grant (P30CA014599).
Footnotes
The authors declare no potential conflicts of interest.
Authors' Contributions:
Conception and design: T.F. Gajewski, B.L. Horton
Development of methodology: B. L. Horton, J. B. Williams, S. Spranger, T. F. Gajewski
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): B. L. Horton, J. B. Williams, A. Cabanov, S. Spranger, T. F. Gajewski
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): B.L. Horton, T.F. Gajewski
Writing, review, and/or revision of the manuscript: B. L. Horton, S. Spranger, T. F. Gajewski
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): B.L. Horton, T.F. Gajewski
Study supervision: T.F. Gajewski
References
- 1.Spranger S, Luke JJ, Bao R, Zha Y, Hernandez KM, Li Y, et al. Density of immunogenic antigens does not explain the presence or absence of the T-cell-inflamed tumor microenvironment in melanoma. Proceedings of the National Academy of Sciences of the United States of America. 2016;113:E7759–E7768. doi: 10.1073/pnas.1609376113. published online EpubNov 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fridman WH, Pages F, Sautes-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nature reviews. Cancer. 2012;12:298–306. doi: 10.1038/nrc3245. published online EpubMar 15. [DOI] [PubMed] [Google Scholar]
- 3.Gajewski TF, Schreiber H, Fu YX. Innate and adaptive immune cells in the tumor microenvironment. Nature immunology. 2013;14:1014–1022. doi: 10.1038/ni.2703. published online EpubOct. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Williams JB, Horton BL, Zheng Y, Duan Y, Powell JD, Gajewski TF. The EGR2 targets LAG-3 and 4-1BB describe and regulate dysfunctional antigen-specific CD8+ T cells in the tumor microenvironment. The Journal of experimental medicine. 2017;214:381–400. doi: 10.1084/jem.20160485. published online EpubFeb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. The New England journal of medicine. 2012;366:2443–2454. doi: 10.1056/NEJMoa1200690. published online EpubJun 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. The New England journal of medicine. 2015;373:23–34. doi: 10.1056/NEJMoa1504030. published online EpubJul 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Spranger S, Koblish HK, Horton B, Scherle PA, Newton R, Gajewski TF. Mechanism of tumor rejection with doublets of CTLA-4, PD-1/PD-L1, or IDO blockade involves restored IL-2 production and proliferation of CD8(+) T cells directly within the tumor microenvironment. Journal for immunotherapy of cancer. 2014;2:3. doi: 10.1186/2051-1426-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJ, Robert L, et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature. 2014;515:568–571. doi: 10.1038/nature13954. published online EpubNov 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kline J, Brown IE, Zha YY, Blank C, Strickler J, Wouters H, et al. Homeostatic proliferation plus regulatory T-cell depletion promotes potent rejection of B16 melanoma. Clinical cancer research : an official journal of the American Association for Cancer Research. 2008;14:3156–3167. doi: 10.1158/1078-0432.CCR-07-4696. published online EpubMay 15. [DOI] [PubMed] [Google Scholar]
- 10.Udaka K, Wiesmuller KH, Kienle S, Jung G, Walden P. Self-MHC-restricted peptides recognized by an alloreactive T lymphocyte clone. Journal of immunology. 1996;157:670–678. published online EpubJul 15 ( [PubMed] [Google Scholar]
- 11.Spranger S, Bao R, Gajewski TF. Melanoma-intrinsic beta-catenin signalling prevents anti-tumour immunity. Nature. 2015;523:231–235. doi: 10.1038/nature14404. published online EpubJul 9. [DOI] [PubMed] [Google Scholar]
- 12.Spiotto MT, Yu P, Rowley DA, Nishimura MI, Meredith SC, Gajewski TF, et al. Increasing tumor antigen expression overcomes "ignorance" to solid tumors via crosspresentation by bone marrow-derived stromal cells. Immunity. 2002;17:737–747. doi: 10.1016/s1074-7613(02)00480-6. published online EpubDec ( [DOI] [PubMed] [Google Scholar]
- 13.Martin SJ, Amarante-Mendes GP, Shi L, Chuang TH, Casiano CA, O'Brien GA, et al. The cytotoxic cell protease granzyme B initiates apoptosis in a cell-free system by proteolytic processing and activation of the ICE/CED-3 family protease, CPP32, via a novel two-step mechanism. The EMBO journal. 1996;15:2407–2416. published online EpubMay 15 ( [PMC free article] [PubMed] [Google Scholar]
- 14.Garrod KR, Moreau HD, Garcia Z, Lemaitre F, Bouvier I, Albert ML, et al. Dissecting T cell contraction in vivo using a genetically encoded reporter of apoptosis. Cell reports. 2012;2:1438–1447. doi: 10.1016/j.celrep.2012.10.015. published online EpubNov 29. [DOI] [PubMed] [Google Scholar]
- 15.Barnes SE, Wang Y, Chen L, Molinero LL, Gajewski TF, Evaristo C, et al. T cell-NF-kappaB activation is required for tumor control in vivo. Journal for immunotherapy of cancer. 2015;3:1. doi: 10.1186/s40425-014-0045-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Woo SR, Fuertes MB, Corrales L, Spranger S, Furdyna MJ, Leung MY, et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity. 2014;41:830–842. doi: 10.1016/j.immuni.2014.10.017. published online EpubNov 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang L, Chen X, Liu X, Kline DE, Teague RM, Gajewski TF, et al. CD40 ligation reverses T cell tolerance in acute myeloid leukemia. The Journal of clinical investigation. 2013;123:1999–2010. doi: 10.1172/JCI63980. published online EpubMay. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Martinez-Forero I, Azpilikueta A, Bolanos-Mateo E, Nistal-Villan E, Palazon A, Teijeira A, et al. T cell costimulation with anti-CD137 monoclonal antibodies is mediated by K63-polyubiquitin-dependent signals from endosomes. Journal of immunology. 2013;190:6694–6706. doi: 10.4049/jimmunol.1203010. published online EpubJun 15. [DOI] [PubMed] [Google Scholar]
- 19.Boldajipour B, Nelson A, Krummel MF. Tumor-infiltrating lymphocytes are dynamically desensitized to antigen but are maintained by homeostatic cytokine. JCI insight. 2016;1:e89289. doi: 10.1172/jci.insight.89289. published online EpubDec 08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Daud AI, Loo K, Pauli ML, Sanchez-Rodriguez R, Sandoval PM, Taravati K, et al. Tumor immune profiling predicts response to anti-PD-1 therapy in human melanoma. The Journal of clinical investigation. 2016;126:3447–3452. doi: 10.1172/JCI87324. published online EpubSep 01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kondoh M, Ohga N, Akiyama K, Hida Y, Maishi N, Towfik AM, et al. Hypoxia-induced reactive oxygen species cause chromosomal abnormalities in endothelial cells in the tumor microenvironment. PloS one. 2013;8:e80349. doi: 10.1371/journal.pone.0080349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Klein TJ, Glazer PM. The tumor microenvironment and DNA repair. Seminars in radiation oncology. 2010;20:282–287. doi: 10.1016/j.semradonc.2010.05.006. published online EpubOct. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Banerjee A, Thyagarajan K, Chatterjee S, Chakraborty P, Kesarwani P, Soloshchenko M, et al. Lack of p53 Augments Antitumor Functions in Cytolytic T Cells. Cancer research. 2016;76:5229–5240. doi: 10.1158/0008-5472.CAN-15-1798. published online EpubSep 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hui E, Cheung J, Zhu J, Su X, Taylor MJ, Wallweber HA, et al. T cell costimulatory receptor CD28 is a primary target for PD-1-mediated inhibition. Science. 2017 doi: 10.1126/science.aaf1292. published online EpubMar 09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kamphorst AO, Wieland A, Nasti T, Yang S, Zhang R, Barber DL, et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science. 2017 doi: 10.1126/science.aaf0683. published online EpubMar 09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fan X, Quezada SA, Sepulveda MA, Sharma P, Allison JP. Engagement of the ICOS pathway markedly enhances efficacy of CTLA-4 blockade in cancer immunotherapy. The Journal of experimental medicine. 2014;211:715–725. doi: 10.1084/jem.20130590. published online EpubApr 07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hernandez-Chacon JA, Li Y, Wu RC, Bernatchez C, Wang Y, Weber JS, et al. Costimulation through the CD137/4-1BB pathway protects human melanoma tumor-infiltrating lymphocytes from activation-induced cell death and enhances antitumor effector function. Journal of immunotherapy. 2011;34:236–250. doi: 10.1097/CJI.0b013e318209e7ec. published online EpubApr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takahashi C, Mittler RS, Vella AT. Cutting edge: 4-1BB is a bona fide CD8 T cell survival signal. Journal of immunology. 1999;162:5037–5040. published online EpubMay 01 ( [PubMed] [Google Scholar]
- 29.Weigelin B, Bolanos E, Teijeira A, Martinez-Forero I, Labiano S, Azpilikueta A, et al. Focusing and sustaining the antitumor CTL effector killer response by agonist anti-CD137 mAb. Proceedings of the National Academy of Sciences of the United States of America. 2015;112:7551–7556. doi: 10.1073/pnas.1506357112. published online EpubJun 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Long AH, Haso WM, Shern JF, Wanhainen KM, Murgai M, Ingaramo M, et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nature medicine. 2015;21:581–590. doi: 10.1038/nm.3838. published online EpubJun. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Evaristo C, Spranger S, Barnes SE, Miller ML, Molinero LL, Locke FL, et al. Cutting Edge: Engineering Active IKKbeta in T Cells Drives Tumor Rejection. Journal of immunology. 2016;196:2933–2938. doi: 10.4049/jimmunol.1501144. published online EpubApr 01. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.