Abstract
Objective
HIV self-testing (HIVST) is now officially recommended by the WHO, yet much of HIVST evidence to date has focused on quantitative data and hypothetical concerns. Effective scale up of HIVST in diverse local contexts requires qualitative data from experiences using and organizing HIVST. This qualitative systematic review aims to appraise and synthesize research evidence on experiences using and organizing HIVST.
Methods
We conducted a systematic search of seven primary literature databases, four grey literature sources, and reference lists reporting qualitative evidence on HIVST. Data extraction and thematic analysis were used to synthesize findings. Quality of studies was assessed using the CASP tool. Confidence in review findings was evaluated using the CERQual approach. The review protocol was registered (CRD42015027607).
Results
From 1,266 potential articles, we included eighteen. Four studies were conducted in low-income countries, three in middle-income countries, ten in high-income countries, and one in multiple countries. Generally, HIVST increased capacity to reach priority populations and expanded opportunities for service delivery. Self-testing was preferred to facility-based testing due to increased convenience and confidentiality, especially among stigmatized populations. HIVST decreased test-associated stigma compared to facility-based testing. HIVST generally empowered people because it provided greater control over individual testing needs. At the same time, HIVST rarely allowed husbands to coerce their wives to test.
Conclusions
This review suggests that HIVST should be offered as an additional HIV testing option in order to expand testing and empower testers. Adapting national policies to incorporate HIVST will be necessary to guide scale-up.
Keywords: CASP, CERQual, HIV self-testing, Qualitative research, Systematic review
INTRODUCTION
Despite UNAIDS 90-90-90 targets[1], only an estimated 60% of people living with HIV globally knew their status in 2016[2]. This disappointment has spurred interest in expanding decentralized testing strategies[3, 4] such as HIV self-testing (HIVST)[5]. The WHO defines HIVST as an individual collecting their own test specimen, performing an HIV test, and interpreting the result [5]. HIVST offers confidentiality and convenience, which may expand access among those who would not otherwise test[6]. Mathematical models[7] and several implementation pilots suggest that HIVST could expand HIV testing and save money. The WHO now recommends that HIVST be offered as an additional HIV testing approach[5].
However, there are unresolved questions about how self-testing can be scaled up in countries with diverse environments[8]. Adverse outcomes related to the social context of HIVST may influence the overall effectiveness of the approach[9]. These lingering doubts about HIVST have contributed to policies in Germany[10] and elsewhere[11] making HIVST explicitly illegal. Although many countries are re-examining policies now, only 40 countries currently have national policies that support HIVST[12].
Much of the existing HIVST social science research has focused on perceptions of self-testing[13–19] that may not reflect experiences. Examining HIVST experiences is important for three reasons. First, HIVST is more decentralized compared to facility-based testing, increasing the number of settings and potential risks of testing. Second, the unsupervised nature of HIVST introduces the possibility for coercion[20, 21]. Third, HIVST social science research can capture experiences from marginalized individuals who may not be reached by conventional surveys[22].
A qualitative systematic review brings together data collected from individual qualitative research studies[23]. This method has been used to synthesize qualitative data and inform WHO guidelines[24–28]. This systematic review appraises and synthesizes qualitative research evidence on experiences using or organizing HIVST.
METHODS
Our methods were informed by methodology described in the Cochrane Handbook[29]. Our reporting of items is based on PRISMA guidelines[30].
Search Strategy
A comprehensive literature search was performed to identify qualitative studies of HIVST. The search was initially conducted in seven primary databases from each database’s inception through November 30, 2015: MEDLINE/PubMed, EMBASE, CINAHL, PsycINFO, the Cochrane Library, Global Health, and Scopus. Additional searches were conducted on February 25, 2016 for grey literature and unpublished reports in: Conference on Retroviruses and Opportunistic Infections (CROI), International AIDS Society (IAS), OpenGrey, and EThOS (British Library). An updated search was conducted in all resources on November 17, 2016. The search strategy was developed with input from members of the research team, and an experienced librarian conducted the searches. A more detailed search algorithm with subject headings and keywords can be found attached in supplementary material (S1).
Screening and Selection Process
All citations were screened by two independent reviewers. Full texts were assessed for eligibility based on standardized inclusion criteria: (a) presented primary research data; (b) used qualitative methodology; (c) evaluated direct experiences with HIVST; (e) published in English; and (f) not a thesis or dissertation. Discrepancies during each stage of screening were resolved after discussion and final input by a third reviewer. For the purposes of this review, direct experience with HIVST meant that study participants themselves received an HIVST or organized a program that provided HIVST services to other people.
Data Extraction
Basic study descriptors were extracted: study setting, year of study, population researched, research aims, study design, type of data collection, and study context (whether HIV self-test was performed outside of or within research context). Conclusions, themes, and primary data were extracted from manuscripts. Data on hypothetical concerns, preferences, and perspectives were excluded.
Synthesis
A framework thematic synthesis approach was used to analyze and synthesize the data[31]. All extracted data were collected in a spreadsheet and subjected to open coding using line-by-line analysis to develop preliminary descriptive themes. Inductive analysis of all themes developed into an axial coding framework with first, second, and third order themes. Each review finding was assessed using the Confidence in the Evidence from Reviews of Qualitative Research (CERQual) approach[24, 32]. The CERQual approach provides a transparent method for evaluating qualitative evidence syntheses, similar to the GRADE approach for quantitative systematic reviews[33].
Reporting
The protocol for this study was registered on PROSPERO, the International Prospective Register of Systematic Reviews (CRD42015027607).
RESULTS
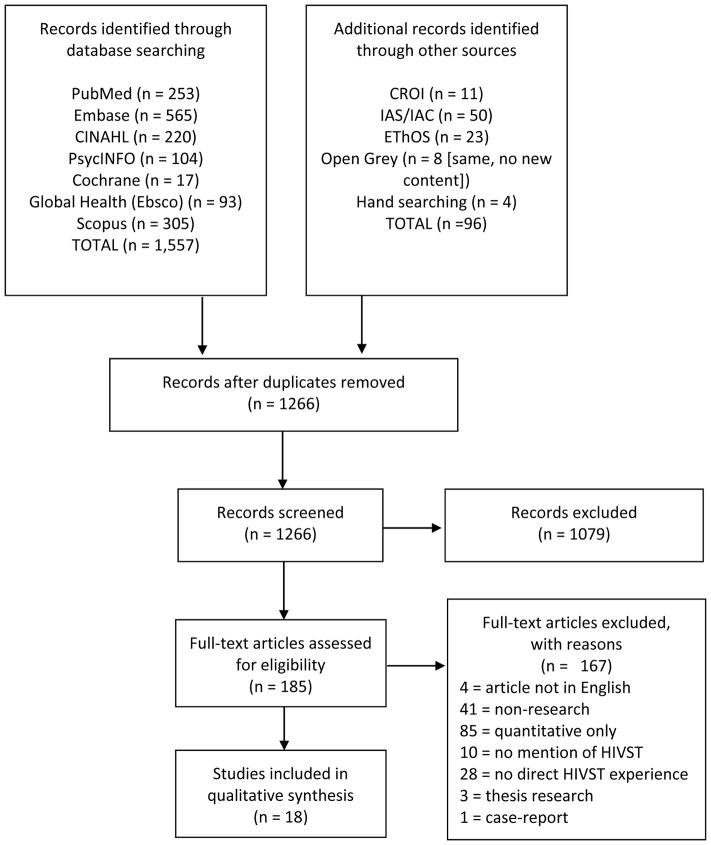
Initial screening yielded 1,266 potential articles, from which 185 were selected for full text review (Figure 1). The eighteen included studies examined research conducted in eight countries (USA, UK, Ethiopia, Malawi, South Africa, Canada, Kenya, India) with 15 studies focused on adults[34–48] and three on youths[49–51]. Seven studies were focused on key populations: six on men who have sex with men (MSM)[34–37, 41, 46] and one on transgender (TG) individuals[40]. Four studies looked at the special population of health care workers (HCWs)[38, 42, 43, 47], one study evaluated pregnant women[45], and five studies investigated partner-testing among couples[34, 35, 37, 39, 41]. Seventeen studies reported experiences using HIVST and one study reported experiences organizing HIVST[36]. Five studies were conducted in low-income countries (LIC)[38, 39, 42, 47, 48], two in a middle-income country (MIC)[43, 45], ten in high-income countries (HIC)[34–37, 40, 41, 46, 49–51], and one study spanned three countries (2 LIC, 1 MIC)[44].
Figure 1.
Study flow diagram
Table 1 presents a classification of HIVST themes based on synthesis of qualitative evidence. First-order themes were primarily descriptive and coded directly from the text line-by-line. Second- and third-order themes provided higher level analysis that grouped findings into logical classifications.
Table 1.
Emerging themes from direct experiences with HIVST
| Third Order Themes | Second Order Themes | First Order Themes |
|---|---|---|
| Increased capacity to reach priority populations | HIVST was more convenient than facility-based testing | Autonomy over test conditions (timing, location, setting) Avoided hassle of traveling to clinics, making appointments, and waiting in line |
| HIVST was more confidential than facility-based testing. | Avoided stigmatizing experiences at testing facilities Privacy was better protected Reached high-risk and untested populations |
|
| HIVST reduced test-associated stigma | Normalized testing Best promoted through social networks |
|
|
| ||
| Expanded opportunities for service delivery | HIVST brought HIV testing into new settings and contexts | Obtained from vending machines, community centers, bars/clubs, pharmacies, online, research programs Used at home, bars/clubs, supermarket, cars, on-the-go Used during casual hookups Used for antenatal care |
| HIVST facilitated partner-testing | Logistically easier than facility-based partner testing Allowed for point-of sex testing with casual partners Allowed for monitoring in serodiscordant couples Allowed partners to provide support during & after testing |
|
| HIVST supplemented existing resources in the community and clinic to improve HIV care | Mental health services Community-based organizations and LGBT centers Healthcare workers and counselors Confirmatory testing and re-testing Start or re-enter HIV care/treatment Peer networks and online communities |
|
| Lay persons were trained to correctly operate and teach HIVST | HIVST kits were easy to operate, particularly oral kits Oral kits were preferred to blood-based kits Clear instructions facilitated correct operation of kits |
|
|
| ||
| Social and ethical considerations for implementation and scale-up | HIVST generally promoted agency, but sometimes generated coercion. | Increased HIV testing (both self and partners) Increased HIV status disclosure Reduced risk for coercion when freedom to refuse was explicit or implicit Increased risk for coercion within context of gender and power imbalances |
| HIVST informed sexual decision-making | Condom-use Partner screening |
|
| Positive HIVST results did not lead to adverse outcomes (suicide, violence), but in certain contexts had strong emotional impact | Anticipatory anxiety Psychological distress Verbal aggression |
|
The first-order themes are derived from specific instances in the text of included articles. The second-order themes represent higher-level syntheses that meaningfully groups first-order themes into key concepts. The third-order themes highlight overarching core messages for future policy and research.
Results are grouped under three main findings: (1) HIVST increased capacity to reach priority populations; (2) HIVST provides expanded opportunities for service delivery; and (3) social and ethical considerations for implementation and scale-up of HIVST. Table 2 summarizes the review findings and the confidence rating of each finding. Study characteristics are presented in Table 3.
Table 2.
Summary of qualitative findings and CERQual assessments
| Review finding | Relevant papers | Confidence in the evidence | Explanation of confidence in the evidence assessment |
|---|---|---|---|
| Increased ability to reach priority populations | |||
| HIVST was more convenient than facility-based testing: Across different populations, countries, and income-levels, HIVST offered users freedom of choice over timing, location, and test setting. | [36–40, 42–47, 49–51] | High | 14 studies with minor to serious methodological limitations. Thick data from 8 countries across income levels. High coherence. |
| HIVST was more confidential than facility-based testing: The privacy of HIVST allowed users to avoid stigmatizing experiences at facility-based testing sites. This encouraged populations that don’t usually engage with sexual health services to test. | [36, 38–40, 44, 46, 47, 49–51] | High | 13 studies with minor to serious methodological limitations. Thick data from 6 countries across all income levels. High coherence. |
| HIVST reduced test-associated stigma: Dissemination of HIVST to the public normalized testing in multiple settings, encouraging uptake of HIV testing. Effective service delivery approaches relied on tapping into social networks and/or normalizing point of sale process (with other products in vending machines, pharmacies, community centers). | [36, 37, 39, 40, 43, 46, 47, 49, 51] | Moderate | 9 studies with minor to moderate methodological limitations. Thick data from 5 countries, but particularly Kenya, Malawi, and USA. Reasonable level of coherence. |
| Expanded opportunities for service delivery | |||
| HIVST brought HIV testing into new settings and contexts: HIVST moved testing out of the medical environment and into people’s homes and lives. HIVST occurred at a growing number of different places, including homes, bars/clubs, supermarkets, cars, on-the-go, casual sex venues, and antenatal clinics. This indicates that HIVST can build on the current testing norms. | [34, 35, 37, 39, 43, 45–47, 49, 51] | High | 10 studies with minor to moderate methodological limitations. Fairly thick data from 5 countries. High coherence. |
| HIVST facilitated partner-testing: This form of promoting HIV testing was effective and generally well-received by partners, often resulting in mutual support. HIVST may increase HIV testing among casual partners (point-of-sex testing) and non-casual partners (monitoring among serodiscordant couples). | [34, 35, 38, 39, 41, 47, 51] | High | 7 studies with minor to serious methodological limitations. Thick data from 4 countries across all income levels. High coherence. |
| HIVST supplemented existing resources in the community and clinic to improve HIV care: HIVST programs successfully provided linkage to care when implemented properly. They did so by utilizing resources such as community-based organizations/LGBT centers, healthcare workers, counselors, peer networks, and web-based platforms to connect people with mental health services, confirmatory testing and re-testing, and starting or re-entering HIV care/treatment. | [34, 35, 39–41, 45–47, 49, 51] | Moderate | 9 studies with minor to moderate methodological limitations. Adequate data from 4 countries. Reasonable level of coherence. |
| Lay persons were trained to correctly operate and teach HIVST: HIVST kits were easy to operate, particularly oral kits, which were preferred to blood-based kits. Clear instructions, either written or verbal, facilitated correct operation of test kits. | [35, 39, 40, 43–47, 49–51] | High | 11 studies with minor to moderate methodological limitations. Adequate data from 6 countries, predominantly middle- and high-income countries. High coherence. |
| Social and ethical considerations for implementation and scale-up | |||
| HIVST generally promoted agency, but sometimes generated coercion: In most cases, users felt greater control over their HIV testing needs, making the experience more self-directed. This led to increased testing of oneself and others (with HIVST kits, at facilities, or both). Many individuals reported increased ownership of serostatus, as demonstrated through increased disclosure of serostatus (to partners, friends, family, etc). | [34, 35, 37–39, 41–47, 49–51] | High | 15 studies with minor to serious methodological limitations. Fairly thick data from 8 countries. High coherence. |
| Positive HIVST results did not lead to adverse outcomes (suicide, violence), but in certain contexts had strong emotional impact: Preliminary positive results with HIVST can trigger strong feelings of emotional distress, but in no case was there mention of suicide as coping mechanism. No cases of physical violence were reported. | [34, 35, 39–43, 46–48] | High | 10 studies with minor to moderate methodological limitations. Thick data from 4 countries. High coherence. |
| HIVST informed sexual decision-making: HIVST impacted individual attitudes, decision-making, and actions by increasing awareness of sexual risk behaviors and encouraging safe practices. High-risk MSM and marginalized youth populations reported increased condom use and employing HIVST as a tool to screen sexual partners. | [34, 35, 37, 40, 41, 43, 51] | Moderate | 7 studies with minor to moderate methodological limitations. Thick data from 2 countries. High coherence. |
A summary of the review findings from the qualitative synthesis is presented here, with contributing studies and the overall confidence in the evidence using the CERQual assessment. A full evidence profile is available in supplemental materials.
HCW = health care workers, MSM = men who have sex with men, TG = transgender
Table 3.
Characteristics of included studies
| #* | Author (Year) | Setting | Population | Research aims | Data collection & analysis | Findings | Quality |
|---|---|---|---|---|---|---|---|
| 1 | Balán (2014) | USA | 27 HIV negative MSM | Explore how HIVST use can affect sexual decision making. | IDI Content analysis, thematic analysis |
HIVST was used by participants to screen sexual partners in cases where partners refused HIVST or received positive HIVST results. Participants generally did not decide to forgo condom use following negative HIVST results, and three main themes were identified: (1) heightened awareness of HIV risk; (2) concerns about the window period; and (3) distrust of the sexual partner. HIVST provided a more concrete tool than direct inquiry for HIV risk reduction. | High |
| 2 | Brown (2016) | USA | 21 high-risk youths aged 18–24 | Understand how young adults perceive and use HIVST. | IDI Content analysis |
Participants generally preferred HIVST to facility testing and identified important motivating factors for HIVST use: (1) peer influence and (2) perceived social norms. | Medium |
| 3 | Carballo-Diéguez (2012) | USA | 27 HIV negative MSM | Test whether high-risk MSM would use HIVST to screen sexual partners. | IDI, telephone audio recordings Content analysis |
Over the course of 3 months, participants proposed HIVST to approximately 124 sexual partners of whom 101 accepted and 23 refused. Most partners were receptive to using HIVST prior to sex but two participants reported experiencing verbal aggression. Participants reported a range of settings where HIVST use occurred (home, partner’s home, public). | High |
| 4 | Flowers (2017) | UK | 12 FGs with 55 stakeholders (all involved in using, offering, or implementing self-testing) | Explore key barriers and facilitators to the effective use of HIVST among MSM | FG Thematic analysis |
Facilitators to effective use of HIVST among MSM: (1) convenience/speed; (2) discretion and privacy; (3) provider perceptions of rationing clinical time and resources; (4) test’s ability to reach new and potentially vulnerable populations; (5) avoiding stigma of using facility services. Barriers: (1) lost opportunities for engagement with range of services and staff; (2) deracinating HIV from wider and holistic health | High |
| 5 | Frasca (2014) | USA | 27 HIV negative MSM | Explore attitude and behavior changes among MSM after use of HIVST to screen sexual partners | IDI Content analysis |
Among MSM who described changes in their thinking or feelings about sexual risk and/or a reduction in risk practices after HIVST use, the following patterns were observed: (1) greater awareness of HIV and sexual risk; (2) reexamination of past behavior; (3) increased awareness of partner subjectivity; (4) fewer partners. | High |
| 6 | Kebede (2013) | Ethiopia | 307 HCWs | Gather evidence on HIVST practices among HCWs | IDI Thematic analysis |
HCWs often engaged in informal HIVST in order to avoid stigma, | Low |
| 7 | Kumwenda (2014) | Malawi | 17 heterosexual couples (34 men and women from the intervention arm of an HIV/TB prevention trial) | Explore concerns relating to coercive testing and how gender roles and relations impact experiences of HIVST for both women and men within couples. | IDI Thematic analysis |
Factors shaping initial decision to self-test among cohabitating couples presented against three key themes: (1) individual-level motivations; (2) couple power dynamics; and (3) serostatus disclosure. Participants felt HIVST offered advantages over facility-based testing. There was evidence of pressure from a partner to test in two of the 17 couples. In both cases it was a husband pressuring his wife to test with the justification that it was the husband’s right to know this information about his wife. | High |
| 8 | Lippman (2015) | USA | 50 HIV negative TG women (11 IDIs) | Pilot study to understand potential of HIVST among TG women and strategies for successful use. | IDI Content analysis |
Some participants liked self-testing because they found it empowering in addition to convenient and privacy. Participants found that partner-testing could offer more support, but also feared undesired disclosure and stigma. | High |
| 9 | Martinez (2014) | USA | 84 HIV negative MSM | Examine and compare anticipated and actual reactions to positive results through HIVST. | IDI Grounded theory, thematic analysis |
None of the participants tested positive but five individuals had partners who tested HIV-positive on seven occasions. Two major themes emerged from these experiences: (1) provision of emotional support and (2) connection to health services. | High |
| 10 | Namakhoma (2010) | Malawi | 906 HCWs (47 IDIs) | Describe experience of testing among HCWs and highlight ways in which social context of HIV influences access. | IDI Framework analysis |
Barriers to accessing HIV testing among HCWs were lack of confidentiality at facilities and fears about HIV testing: (1) fear of a positive HIV test; (2) the stigma attached to being positive; and (3) possible loss of status that might follow a positive result. | High |
| 11 | Pant Pai (2013) | South Africa | 251 HCWs | Pilot study to evaluate HIVST strategy among HCWs. | Open-ended qualitative questionnaire Thematic analysis |
Participants provided responses reflections on their self-testing experience that fell into three themes: (1) test administration; (2) privacy; and (3) factors influencing stress. | Medium |
| 12 | Pant Pai (2014) | Canada | 145 HIV negative university students | Pilot study to evaluate HIVST strategy among university students in Canada. | Open-ended qualitative questionnaire Thematic analysis |
The main themes that were retained reflected students’ concerns regarding the following issues: (1) self-test accuracy; (2) acceptability; (3) linking seropositive subjects with counselling and medical care; (4) increase in HIV testing with the self-testing option; (5) test administration; (6) time-saving process; (7) accessibility; and (8) privacy. | Medium |
| 13 | Peck (2014) | Kenya, Malawi, and South Africa | 150 lay users who never previously self-tested (Kenya = 49, Malawi = 47, South Africa = 54) | Usability study to identify use-related problems and hazards of test features in 5 different HIVST prototypes. | IDI, video observation Framework analysis |
Themes were centered around test attributes: (1) instructions; (2) oral swabbing or fingerstick sampling; (3) test liquid; (4) timing; (5) results interpretation; and (6) perception of tests [easy to use, enthusiasm due to time savings, reduced fear of HIV status disclosure, and convenience]. | High |
| 14 | Sarkar (2016) | India | 35 pregnant women | Explore acceptability, concordance, and feasibility of HIVST among pregnant women | IDI Thematic analysis |
Factors that influenced acceptability of oral fluid-based HIV testing fell into 6 major themes: (1) clarity of test instructions; (2) time-efficiency; (3) non-invasiveness; (4) convenience; (5) clarity of results interpretation; and (6) fear of incorrect results. | High |
| 15 | Schnall (2015) | USA | 21 high-risk youths aged 18–24 | Understand young adults’ ability use HIVST | IDI, video observation, think-aloud protocol observation Thematic analysis |
The following themes emerged regarding experience using an HIV self-test: (1) did I use it correctly? (2) can I trust the results? (3) how will my partner react? (4) what will people think? (5) quick, easy and blood free; and (6) Avoids the hassle of dealing with the healthcare system. | Medium |
| 16 | Young (2014) | USA | 8 MSM participants from an HIV prevention trial | Pilot study to determine acceptability of using vending machines to dispense HIVST kit among MSM | IDI Content analysis |
In general, participants expressed that the vending machine was an acceptable HIV test delivery method due to three main factors: (1) credibility; (2) confidentiality; and (3) convenience. All participants were interested in recommending this delivery model to others. | High |
| 17 | Kalibala (2011) | Kenya | 14 HIVST intervention coordinators 15 FGs with HCWs (participated in HIVST intervention) |
Explore feasibility and acceptability of an HIVST intervention among HCWs. | IDI, FG Thematic analysis |
While many of the health workers and their partners tested together, those couples who did not test together still revealed their test results to their partners. | High |
| 18 | Kumwenda (2016) | Malawi | 67 men who self-tested | Examine the role of masculinity on HIVST decisions and subsequent actions amongst couples | IDI Content analysis |
Conception of masculinity dictated men’s actions and prescribed specific behavioral attributes, namely having control, knowledge, strength and toughness, and both sexual and economic productivity. | Medium |
Increased capacity to reach priority populations
HIVST demonstrated increased capacity to reach priority populations in a wide variety of settings. Most individuals noted that HIVST was more convenient and more confidential than facility-based testing.
More convenient than facility-based testing
Individuals were able to employ HIVST in a way that catered to their own schedules and testing needs, offering a customized HIV testing experience that allowed for greater control over location and time[46]. MSM[46], TG women[40], youths[50], and pregnant women[45] commonly described HIVST as convenient. To some, convenience meant no longer having to wait in lines, make appointments, or travel long distances to clinics[38, 39, 44, 47, 49–51]. To others, convenience came in the form of greater control over the speed, efficiency, and location of testing[37, 39, 43, 45, 46, 49–51]. Individuals who organized HIVST for MSM similarly observed that these attributes reduced barriers to testing and allowed people from regions where getting HIV testing is difficult (e.g. rural areas) to access it[36].
More confidential than facility-based testing
MSM[41, 46], TG women[40], youths[49–51], HCWs[38, 42, 43, 47], and lay users[44] commented on better protection of privacy with HIVST compared to facility-based testing. In particular, HCWs[38, 42, 47] in Africa and TG women[40] in the USA wanted to avoid the stigma of testing in a public context, with the former being concerned about workplace discrimination and the latter about gossip within the relatively small TG community. Individuals from LICs[39, 44], MICs[44, 47], and HICs[40, 51] described being judged for seeking out HIV testing at a facility. Facility-based testing in Scotland was sometimes associated with connotations of immorality, promiscuity, and living with HIV. Individuals organizing HIVST confirmed that sexual health service facilities continue to be stigmatized, and thus HIVST reached new populations that don not traditionally test because of fear[36].
Reduced test-associated stigma
In several studies where HIVST kits were provided for participants to bring home, dissemination in public places normalized HIV testing across multiple settings. One pilot program in the USA used vending machines to dispense kits, delivering them along with other non-stigmatized products and through a local vending machine[46]. Among heterosexual couples in Malawi that were given free access to HIVST kits, participants often described self-test kits as toothbrushes, normalizing the testing kits within their daily routines[39]. Peer and social networks played an important role in influencing youths to self-test[46]. Positive experiences with HIVST led HCWs in South Africa[43] and MSM in the USA[46] to say they would recommend HIVST to friends, colleagues, family, and partners. Individuals that recommended HIVST to partners commented that in some instances, the physical presence of self-test kits served as an “ice-breaker” that facilitated discussions about health, fidelity and HIV-related concerns that were otherwise difficult to raise[35, 39]. Some MSM felt that they were more likely to stick together with partners that had agreed to self-test[37].
Expanded opportunities for service delivery
HIVST facilitates a new testing paradigm in which the testing experience is uncoupled from a strictly medical environment, which provides opportunities for innovative and targeted service delivery.
HIVST brought HIV testing into new settings and contexts
Self-test kits could be obtained from a number of diverse venues, including vending machines[46], community centers[47], bars/clubs[35, 49], pharmacies[43], and online[46]. HIVST was also used at a number of diverse locations, including at home[39, 43, 46, 47, 49], the supermarket[35], bars/clubs[35], in cars[37, 46], at partners’ homes[35, 37, 51], and at work[35, 37, 42, 51]. Some individuals took to carrying test kits with them regularly so they could be used at any time, or “on the go”[35, 37, 51]. HIVST was used amidst new contexts, such as prior to casual sexual encounters, or “hookups,” among MSM in the USA[34] and antenatal screening for HIV among first trimester pregnant women supervised by auxiliary midwives in rural India[45].
HIVST facilitated partner-testing
Secondary distribution of HIVST kits to casual or long-term partners was explored among several populations: MSM[34, 35, 41], transgender individuals[40], heterosexual couples[39], youths[51], and HCWs[38, 47]. This method was generally well-received and allowed partners to provide support during HIVST in the USA, sometimes by providing a comforting presence before or while waiting for results[40] and other times after receiving a positive result[41]. The social support offered by testing with a partner was identified as a motivating factor to get tested for HIV[39, 51], seek confirmatory testing[35], and adhere to ART[39]. Women in Malawi contrasted the ease with which they could encourage their husbands to self-test with unsuccessful attempts to get their husbands to attend facility-based HIV testing[39]. Serodiscordant couples used HIVST kits as monitoring devices[39]. Distribution of HIVST kits to partners was noted even in studies where participants were not explicitly asked by researchers to distribute test kits to partners[40, 46, 47].
HIVST supplemented existing resources in the community and clinic to improve HIV care
HIVST supplemented existing resources in the community to provide a range of HIV services, including mental health counseling[35, 41], confirmatory testing or re-testing[34, 35], and starting or re-entering ART[39]. Peer networks, community organizations, and LGBT groups were important for both promoting HIVST and encouraging follow-up care after HIVST among MSM[34, 35, 41], youth,[51], and heterosexual couples in Malawi[39]. Youths, in particular, indicated that there was a strong influence of people in their lives (peer, intimate partner, and family referents) on their decision to self-test[49, 51]. HIVST programs in Malawi[39], Kenya[47], and India[45] trained community health workers to promote and support HIVST. The program in India collaborated with an antenatal clinic to provide HIV screening for pregnant women in their first trimester[45].
Lay persons were trained to correctly operate and teach HIVST
HIVST programs in HICs[35, 40, 46, 49–51], MICs[43–45], and LICs[39, 44, 47] successfully trained lay people to either operate or teach HIVST. Operating HIVST refers to the process of unpackaging, appropriately collecting the specimen, and correctly interpreting the test result. This was particularly welcomed in countries where a lack of adequate trained human resources impedes access to HIV testing[45, 47]. Members of the local communities in Kenya, Malawi, and South Africa were videotaped using oral and blood-based specimen HIVST kits and each type was described as easy to use[44]. Most participants correctly interpreted negative and positive HIVST results. HIVST kits were also described as easy and simple to use by youths[49, 51], HCWs, pregnant women[45], MSM[35, 46], TG women[40], and heterosexual couples in Malawi[39]. High-quality instructions, such as clear wording, local translations (where necessary), pictorial aids, or live demonstration, were valued and facilitated correct operation of self-test kits[44, 45, 47, 51].
Social and ethical considerations for implementation and scale-up
HIVST led to feelings of empowerment through control of one’s own testing experience and diagnosis. However, in the context of partner testing, this increased agency rarely was associated with coercion to test.
HIVST generally promoted agency, but sometimes generated coercion
HIVST was described as empowering for MSM[41, 46], TG individuals[40], HCWs[43, 47], pregnant women[45], youths[51] and the general population in Kenya, Malawi, and South Africa[44]. Among youth populations, many had never previously used HIVST, stating that they would have gotten tested sooner if they knew it were an option[50] and that completing the process provided positive reinforcement[49, 51]. TG women and MSM in the USA felt that HIVST allowed them to take ownership of their own serostatus through self-diagnosis and increased agency by providing a tool that allowed for continued self-monitoring[40, 46]. Lay users in Kenya, Malawi, and South Africa similarly indicated that they planned to use HIVST for routine monitoring in the future after having a good first experience[44]. Increased agency also manifested as increased testing of partners, notably among MSM[35, 37]. For women in relationships with underlying gender and power imbalances, the ability to bring HIVST into the household facilitated partner testing[39]. The introduction of HIVST allowed some couples to discuss topics that were previously difficult to bring up, such as fidelity, health, sexually transmitted infections and HIV serostatus[35, 37, 39]. In several instances, HIVST led to disclosure of HIV status between participants and their partners[35, 38, 47]. At the same time, this increased agency was rarely associated with coercive testing. One study in Malawi noted that women were pressured by their husbands to receive HIVST[39]. These instances of coercive HIVST were situated in underlying gender power imbalances.
HIVST informed sexual decision-making
Many individuals liked that HIVST provided a mechanism to detect the HIV status of their partners[34, 35, 37, 39–41]. MSM[34, 35, 37, 41] and TG women[40] used HIVST to screen partners and inform decisions about condom use. They also used HIVST to assess fidelity in their partners, a function also used by heterosexual couples in Malawi. Many individuals reported that their HIVST experience raised awareness about various aspects of sexual behavior, HIV knowledge, and general health and wellness[34]. HIVST created self-awareness about one’s membership in a group at increased risk for HIV[43].
Positive HIVST results did not lead to adverse outcomes (suicide, violence), but in certain contexts had strong emotional impact
Studies that explored adverse outcomes resulting from HIVST examined violence during partner testing[35]and psychological distress associated with self-testing[35, 41–43, 46, 50, 51]. For instance, in one study in Kenya[47], “while health workers expressed concern about potential abuse of the self-test kit, by for example testing minors or housekeepers without their consent, no such incident was reported.” Anxiety associated with HIV testing itself in Malawi[42] and suicidality associated with an HIV positive diagnosis in the USA and South Africa[41, 43] often blended with and carried over to discussions of HIVST, but participants did not specifically distinguish between self-testing and facility-based testing or attribute these emotions specifically to HIVST. Generally, participants described HIVST as a low stress experience compared to facility-based testing[46, 50].
DISCUSSION
Forward progress to achieve 2020 UNAIDs targets will require development of HIV testing services that are adaptable to a wide range of priority populations. HIVST priorities are likely to differ based on HIV prevalence, with higher prevalence settings requiring more investment in services farther along the HIV care continuum. HIVST has been shown to reach populations that had not previously tested, with one study in Africa showing that between 20 to 30 percent of self-testers were first-time HIV testers[52]. Although individual user experiences varied, unifying themes applicable to many HIVST programs were identified.
Despite concerns about the availability of counselling and mixed views on the ability of HIVST to secure confirmatory testing and follow-up care, the preference for HIVST over facility-based testing was clear. Respondents consistently showed dissatisfaction with testing at clinics due to concerns about access and confidentiality. HIVST expanded access by providing greater autonomy over test conditions, including the timing, location, and setting. While increased confidentiality brought in more individuals who might not otherwise test, it also presented challenges in educating patients who test negative about avoiding risky behaviors and informing patients who test positive about the importance of care and treatment.
We found that HIVST was associated with less test-associated stigma compared to facility-based testing. HIVST kits brought the experience of HIV testing into people’s lives and homes, normalizing it and decreasing test-associated stigma. Given the importance of stigma as a barrier to expanding HIV testing around the world[53, 54], this could facilitate scaling up HIV testing in many locations, especially LMIC countries that have substantial HIV test-associated stigma. Persistent stigma associated with the physical presence of HIVST kits may be further minimized by anonymizing kit packaging and incorporating user preferences.
Although HIVST generally increased agency, it was not commonly associated with coercive testing. This risk increased in the context of gender power imbalances in heterosexual couples[39]. This finding is consistent with quantitative studies from Malawi[22] and China[20, 21] showing rare instances of HIVST-associated coercion. Our data suggests the potential for coercion directly related to HIVST, contrasting the interpretation of evidence from the WHO HIV Self-Testing Guidelines[5]. Further research on coercive testing is needed to better understand this potential harmful outcome. This emphasizes the need for accurate adverse event reporting among testers and their partners.
Our qualitative evidence synthesis has several limitations. First, many studies in our review were from heterosexual couples, MSM, and transgender individuals in high-income countries. At the same time, CERQual transparently reports data adequacy for each review finding (Table 2), allowing the reader to draw their own inferences about transferability. Further HIVST research among other vulnerable populations such as sex workers will be important to understand coercive testing. Second, only one of the identified studies included data from individuals organizing HIVST programs. Third, none of the identified qualitative studies assessed linkage to care. A previous quantitative systematic review of HIVST[55] also found no existing literature on issues related to seeking linkage to care. Fourth, none of the identified studies reported the price of HIVST kits. Finally, none of the identified qualitative studies assessed participant knowledge of the limitations of HIVST in the window period between HIV infection and the detection of antibodies. Future qualitative studies should consider follow-up of participants post-HIVST to examine outcomes along the entire cascade of HIV care.
Our systematic review has implications for research, policy, and programs. From a research perspective, our findings suggest the need for more extensive research on coercive HIV self-testing and linkage to care. From a policy perspective, this systematic review can be used to inform the development of local and national HIVST guidelines. Given the WHO HIV Self-Testing Guidelines[5], many policymakers are now re-evaluating HIVST policies. In July 2017 the WHO announced that 40 countries have now incorporated HIVST in their national policies, with 48 other countries currently developing new HIVST policies. This represents significant progress from only 16 countries supporting HIVST two years ago[12]. A system to collate test results into a national repository will help governments better assess the role of HIVST in national HIV prevention and treatment programs. From the perspective of HIVST programs, our data may be useful for optimizing pilot HIVST sites, service delivery models, and strategies. At the same time, implementers should be certain that HIV rapid diagnostic test kits for self-testing are approved by regulatory authorities and quality-assured. There will unlikely be a single “one-size-fits-all” HIV testing approach and the natural advantage of HIVST is that it could be adapted to a wide range of different settings. Our review findings provide practical details to enhance HIVST in diverse settings.
This systematic review provides synthesized data on experiences of using and organizing HIV self-testing. The advent of the self-test will usher in new opportunities and challenges for HIV testing programs. Our data confirm the importance of self-testing as a new tool that normalizes testing, decreases test-associated stigma, and expands HIV testing.
Supplementary Material
Acknowledgments
Funding: This work was supported by the National Institutes of Health [National Institute of Allergy and Infectious Diseases 1R01AI114310 to JT]; UNC-South China STD Research Training Centre [Fogarty International Centre 1D43TW009532 to JT]; UNC Center for AIDS Research [National Institute of Allergy and Infectious Diseases 5P30AI050410 to JT]; the UNC Chapel Hill, Johns Hopkins University, Morehead School of Medicine and Tulane University (UJMT) Fogarty Fellowship [FIC R25TW0093 to WT, FL]. The listed grant funders played no role in any step of this study.
We thank all staff members at SESH Global and the Guangdong Provincial Center for Skin Diseases and STI Control for their contributions. We thank Chongyi Wei for helpful edits on the manuscript.
Footnotes
Author Contributions
JT, WT, YL, AB, LH and FL conceived the study. JT and JW designed the search strategy. YL, AB, and JT screened and selected articles. YL, LH, and AB extracted and synthesized data. LH drafted the manuscript with input from JT, YL, HT, WT, AB, FL, and JW. All authors critically reviewed and revised the manuscript.
Declaration of interests
We declare no competing interests.
References
- 1.UNAIDS. 90-90-90-An ambitious treatment target to help end the AIDS epidemic. Geneva: 2014. [Google Scholar]
- 2.UNAIDS 2016 estimates
- 3.Suthar AB, Ford N, Bachanas PJ, Wong VJ, Rajan JS, Saltzman AK, et al. Towards universal voluntary HIV testing and counselling: a systematic review and meta-analysis of community-based approaches. PLoS Med. 2013;10:e1001496. doi: 10.1371/journal.pmed.1001496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sharma M, Ying R, Tarr G, Barnabas R. Systematic review and meta-analysis of community and facility-based HIV testing to address linkage to care gaps in sub-Saharan Africa. Nature. 2015;528:S77–S85. doi: 10.1038/nature16044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.WHO. Guidelines on HIV self-testing and partner notification: supplement to consolidated guidelines on HIV testing services. Geneva: World Health Organization; 2016. [PubMed] [Google Scholar]
- 6.Figueroa C, Johnson C, Verster A, Baggaley R. Attitudes and Acceptability on HIV Self-testing Among Key Populations: A Literature Review. AIDS Behav. 2015;19:1949–1965. doi: 10.1007/s10461-015-1097-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cambiano V, Ford D, Mabugu T, Napierala Mavedzenge S, Miners A, Mugurungi O, et al. Assessment of the Potential Impact and Cost-effectiveness of Self-Testing for HIV in Low-Income Countries. J Infect Dis. 2015;212:570–577. doi: 10.1093/infdis/jiv040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tucker JD, Wei C, Pendse R, Lo YR. HIV self-testing among key populations: an implementation science approach to evaluating self-testing. J Virus Erad. 2015;1:38–42. doi: 10.1016/S2055-6640(20)31145-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brown AN, Djimeu EW, Cameron DB. A review of the evidence of harm from self-tests. AIDS Behav. 2014;18(Suppl 4):S445–449. doi: 10.1007/s10461-014-0831-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Karcher HL. HIV home test kit banned in Germany. BMJ: British Medical Journal. 1997;315:627–627. doi: 10.1136/bmj.315.7109.623i. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.HIV self-testing research and policy hub: Policy map. In; 2017.
- 12.UNITAID. Market and Technology Landscape: HIV Rapid Diagnostic Tests for Self-Testing. 3. WHO; 2017. [Google Scholar]
- 13.Bilardi JE, Walker S, Read T, Prestage G, Chen MY, Guy R, et al. Gay and bisexual men’s views on rapid self-testing for HIV. AIDS Behav. 2013;17:2093–2099. doi: 10.1007/s10461-012-0395-7. [DOI] [PubMed] [Google Scholar]
- 14.Ochako R, Vu L, Peterson K. Insights Into Potential Users and Messaging for HIV Oral Self-Test Kits in Kenya, 3ie Grantee Final Report. Washington, DC: International Initiative for Impact Evaluation (3ie); 2014. [Google Scholar]
- 15.Greacen T, Friboulet D, Blachier A, Fugon L, Hefez S, Lorente N, et al. Internet-using men who have sex with men would be interested in accessing authorised HIV self-tests available for purchase online. AIDS Care. 2013;25:49–54. doi: 10.1080/09540121.2012.687823. [DOI] [PubMed] [Google Scholar]
- 16.Rosales-Statkus M, Fuente L, Fernández-Balbuena S, Figueroa C, Fernàndez-López L, Hoyos J, et al. Approval and Potential Use of Over-the-Counter HIV Self-Tests: The Opinion of Participants in a Street Based HIV Rapid Testing Program in Spain. AIDS & Behavior. 2015;19:472–484. 413. doi: 10.1007/s10461-014-0975-9. [DOI] [PubMed] [Google Scholar]
- 17.van Rooyen H, Tulloch O, Mukoma W, Makusha T, Chepuka L, Knight LC, et al. What are the constraints and opportunities for HIVST scale-up in Africa? Evidence from Kenya, Malawi and South Africa. J Int AIDS Soc. 2015;18:19445. doi: 10.7448/IAS.18.1.19445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van Dyk AC. Client-initiated, provider-initiated, or self-testing for HIV: what do South Africans prefer? J Assoc Nurses AIDS Care. 2013;24:e45–56. doi: 10.1016/j.jana.2012.12.005. [DOI] [PubMed] [Google Scholar]
- 19.Kabiru CW, Sidze EM, Egondi T, Osok D, Izugbara CO. 3ie Grantee Final Report. Washington, DC: International Initiative for Impact Evaluation (3ie); 2014. Understanding Perceived Social Harms and Abuses of Oral HIV-Self-Testing in Kenya: Key Findings of a Cross-sectional Study. [Google Scholar]
- 20.Qin Y, Tang W, Nowacki A, Mollan K, Reifeis SA, Hudgens MG, et al. Benefits and Potential Harms of Human Immunodeficiency Virus Self-Testing Among Men Who Have Sex With Men in China: An Implementation Perspective. Sex Transm Dis. 2017;44:233–238. doi: 10.1097/OLQ.0000000000000581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ong J, Li H, Wu D, Fu H, Liu E, Ma W, et al. Coercion and HIV Self-Testing in Men Who Have Sex with Men: Implementation Data from a Cross-Sectional Survey in China. JAIDS. doi: 10.1097/QAI.0000000000001583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Choko AT, MacPherson P, Webb EL, Willey BA, Feasy H, Sambakunsi R, et al. Uptake, Accuracy, Safety, and Linkage into Care over Two Years of Promoting Annual Self-Testing for HIV in Blantyre, Malawi: A Community-Based Prospective Study. PLoS Med. 2015;12:e1001873. doi: 10.1371/journal.pmed.1001873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Boeije HR, van Wesel F, Alisic E. Making a difference: towards a method for weighing the evidence in a qualitative synthesis. J Eval Clin Pract. 2011;17:657–663. doi: 10.1111/j.1365-2753.2011.01674.x. [DOI] [PubMed] [Google Scholar]
- 24.Bohren MA, Vogel JP, Hunter EC, Lutsiv O, Makh SK, Souza JP, et al. The Mistreatment of Women during Childbirth in Health Facilities Globally: A Mixed-Methods Systematic Review. PLoS Med. 2015;12:e1001847. doi: 10.1371/journal.pmed.1001847. discussion e1001847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hall BJ, Sou K, Beanland R, Lackey M, Tso LS, Ma Q, et al. Barriers and facilitators to interventions improving retention in HIV care: A qualitative evidence meta-synthesis. AIDS and Behavior. 2016 doi: 10.1007/s10461-016-1537-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ma Q, Tso LS, Rich ZC, Hall BJ, Beanland R, Li H, et al. Barriers and facilitators of interventions for improving antiretroviral therapy adherence: a systematic review of global qualitative evidence. J Int AIDS Soc. 2016;19:21166. doi: 10.7448/IAS.19.1.21166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tso LS, Best J, Beanland R, Doherty M, Lackey M, Ma Q, et al. Facilitators and barriers in HIV linkage to care interventions: a qualitative evidence review. AIDS. 2016 doi: 10.1097/QAD.0000000000001101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li H, Marley G, Ma W, Wei C, Lackey M, Ma Q, et al. The Role of ARV Associated Adverse Drug Reactions in Influencing Adherence Among HIV-Infected Individuals: A Systematic Review and Qualitative Meta-Synthesis. AIDS Behav. 2016 doi: 10.1007/s10461-016-1545-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Higgins J, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0. The Cochrane Collaboration; 2011. [updated March 2011] [Google Scholar]
- 30.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100. doi: 10.1371/journal.pmed.1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Thomas J, Harden A. Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Med Res Methodol. 2008;8:45. doi: 10.1186/1471-2288-8-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lewin S, Glenton C, Munthe-Kaas H, Carlsen B, Colvin CJ, Gulmezoglu M, et al. Using qualitative evidence in decision making for health and social interventions: an approach to assess confidence in findings from qualitative evidence syntheses (GRADE-CERQual) PLoS Med. 2015;12:e1001895. doi: 10.1371/journal.pmed.1001895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Atkins D, Best D, Briss PA, Eccles M, Falck-Ytter Y, Flottorp S, et al. Grading quality of evidence and strength of recommendations. BMJ. 2004;328:1490. doi: 10.1136/bmj.328.7454.1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Balán I, Carballo-Diéguez A, Frasca T, Dolezal C, Ibitoye M. The Impact of Rapid HIV Home Test Use with Sexual Partners on Subsequent Sexual Behavior Among Men Who have Sex with Men. AIDS & Behavior. 2014;18:254–262. 259. doi: 10.1007/s10461-013-0497-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Carballo-Dieguez A, Frasca T, Balan I, Ibitoye M, Dolezal C. Use of a rapid HIV home test prevents HIV exposure in a high risk sample of men who have sex with men. AIDS Behav. 2012;16:1753–1760. doi: 10.1007/s10461-012-0274-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Flowers P, Riddell J, Park C, Ahmed B, Young I, Frankis J, et al. Preparedness for use of the rapid result HIV self-test by gay men and other men who have sex with men (MSM): a mixed methods exploratory study among MSM and those involved in HIV prevention and care. HIV Medicine. 2017;18:245–255. doi: 10.1111/hiv.12420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Frasca T, Balan I, Ibitoye M, Valladares J, Dolezal C, Carballo-Dieguez A. Attitude and behavior changes among gay and bisexual men after use of rapid home HIV tests to screen sexual partners. AIDS Behav. 2014;18:950–957. doi: 10.1007/s10461-013-0630-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kebede B, Abate T, Mekonnen D. HIV self-testing practices among Health Care Workers: feasibility and options for accelerating HIV testing services in Ethiopia. Pan Afr Med J. 2013;15:50. doi: 10.11604/pamj.2013.15.50.2328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kumwenda M, Munthali A, Phiri M, Mwale D, Gutteberg T, MacPherson E, et al. Factors shaping initial decision-making to self-test amongst cohabiting couples in urban Blantyre, Malawi. AIDS Behav. 2014;18(Suppl 4):S396–404. doi: 10.1007/s10461-014-0817-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lippman SA, Moran L, Sevelius J, Castillo LS, Ventura A, Treves-Kagan S, et al. Acceptability and Feasibility of HIV Self-Testing Among Transgender Women in San Francisco: A Mixed Methods Pilot Study. AIDS Behav. 2015 doi: 10.1007/s10461-015-1236-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Martinez O, Carballo-Dieguez A, Ibitoye M, Frasca T, Brown W, Balan I. Anticipated and actual reactions to receiving HIV positive results through self-testing among gay and bisexual men. AIDS Behav. 2014;18:2485–2495. doi: 10.1007/s10461-014-0790-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Namakhoma I, Bongololo G, Bello G, Nyirenda L, Phoya A, Phiri S, et al. Negotiating multiple barriers: health workers’ access to counselling, testing and treatment in Malawi. AIDS Care. 2010;22(Suppl 1):68–76. doi: 10.1080/09540121003607241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pant Pai N, Behlim T, Abrahams L, Vadnais C, Shivkumar S, Pillay S, et al. Will an unsupervised self-testing strategy for HIV work in health care workers of South Africa? A cross sectional pilot feasibility study. PLoS One. 2013;8:e79772. doi: 10.1371/journal.pone.0079772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Peck RB, Lim JM, van Rooyen H, Mukoma W, Chepuka L, Bansil P, et al. What should the ideal HIV self-test look like? A usability study of test prototypes in unsupervised HIV self-testing in Kenya, Malawi, and South Africa. AIDS and Behavior. 2014;18:422–432. doi: 10.1007/s10461-014-0818-8. [DOI] [PubMed] [Google Scholar]
- 45.Sarkar A, Mburu G, Shivkumar PV, Sharma P, Campbell F, Behera J, et al. Feasibility of supervised self-testing using an oral fluid-based HIV rapid testing method: a cross-sectional, mixed method study among pregnant women in rural India. J Int AIDS Soc. 2016;19:20993. doi: 10.7448/IAS.19.1.20993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Young SD, Daniels J, Chiu CJ, Bolan RK, Flynn RP, Kwok J, et al. Acceptability of using electronic vending machines to deliver oral rapid HIV self-testing kits: a qualitative study. PLoS One. 2014;9:e103790. doi: 10.1371/journal.pone.0103790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kalibala S, Tun W, Muraah W, Cherutich P, Oweya E, Oluoch P. ‘Knowing myself first’: Feasibility of self-testing among health workers in Kenya. Nairobi: Population Council; 2011. [Google Scholar]
- 48.Kumwenda MK. AIDS 2016. Durban, South Africa: 2016. The influence of masculinity on HIVST community intervention: a qualitative evaluation of empirical evidence from Blantyre, Malawi. [Google Scholar]
- 49.Brown W, 3rd, Carballo-Dieguez A, John RM, Schnall R. Information, Motivation, and Behavioral Skills of High-Risk Young Adults to Use the HIV Self-Test. AIDS Behav. 2016;20:2000–2009. doi: 10.1007/s10461-016-1309-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pant Pai N, Bhargava M, Joseph L, Sharma J, Pillay S, Balram B, et al. Will an unsupervised self-testing strategy be feasible to operationalize in Canada? Results from a pilot study in students of a large canadian university. AIDS Res Treat. 2014;2014:747619. doi: 10.1155/2014/747619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schnall R, John RM, Carballo-Dieguez A. Do High-Risk Young Adults Use the HIV Self-Test Appropriately? Observations from a Think-Aloud Study. AIDS Behav. 2015 doi: 10.1007/s10461-015-1240-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.UNITAID. Stimulating and shaping the market for HIV self-testing in Africa (STAR) 2016. [Google Scholar]
- 53.Treves-Kagan S, El Ayadi AM, Pettifor A, MacPhail C, Twine R, Maman S, et al. Gender, HIV Testing and Stigma: The Association of HIV Testing Behaviors and Community-Level and Individual-Level Stigma in Rural South Africa Differ for Men and Women. AIDS Behav. 2017 doi: 10.1007/s10461-016-1671-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Evangeli M, Pady K, Wroe AL. Which Psychological Factors are Related to HIV Testing? A Quantitative Systematic Review of Global Studies. AIDS Behav. 2016;20:880–918. doi: 10.1007/s10461-015-1246-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pant Pai N, Sharma J, Shivkumar S, Pillay S, Vadnais C, Joseph L, et al. Supervised and unsupervised self-testing for HIV in high- and low-risk populations: a systematic review. PLoS Med. 2013;10:e1001414. doi: 10.1371/journal.pmed.1001414. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.