Abstract
Liquid chromatography tandem mass chromatography (LC–MS/MS) is an important hyphenated technique for quantitative analysis of drugs in biological fluids. Because of high sensitivity and selectivity, LC–MS/MS has been used for pharmacokinetic studies, metabolites identification in the plasma and urine. This manuscript gives comprehensive analytical review, focusing on chromatographic separation approaches (column packing materials, column length and mobile phase) as well as different acquisition modes (SIM, MRM) for quantitative analysis of glucocorticoids and stimulants. This review is not meant to be exhaustive but rather to provide a general overview for detection and confirmation of target drugs using LC–MS/MS and thus useful in the doping analysis, toxicological studies as well as in pharmaceutical analysis.
Keywords: LC–MS/MS, Ionization techniques, Glucocorticoids, Stimulants, Hyphenated techniques, Biological fluid
1. Introduction
Corticosteroids and stimulants are the class of compounds that are illicitly used by professionals owing to their anti-inflammatory and mood elevating as well as euphoric properties respectively. They are widespread abused among sports persons [1], stimulant addiction among teenagers and deliberate counterfeiting in herbal products to enhance their effects [2], [3]. Hence there is a need for more sensitive analytical tools to detect and confirm these classes of drugs in biological fluids [3], [4]. These analytical tools would serve to fulfill demands in forensic, toxicological and food safety departments. Glucocorticoid belongs to steroid family, particularly of pregnane class containing C-21 derivatives. Glucocorticoids have important functions upon carbohydrate, protein and calcium metabolism, potent anti-inflammatory and immunosuppressive activities [5], [6], [7], [8]. The activity of glucocorticoids largely depends upon the substituent attached to the nucleus. Substituent has been found to significantly increase both glucocorticoids and mineralocorticoids activities. It has been found that Δ1, 2 corticosteroids enhance anti-inflammatory activity and decrease salt retaining activity [9]. Some selected glucocorticoids and their structures are shown in Table 1. Stimulants are the class of drugs that have a marked effect on mental function and behavior, producing euphoria and reduced fatigue. They are diverse class of compounds that exhibit their action through different mechanism. This class of drugs is often abused by teenagers leading to addiction and risk of health hazards. Some of selected stimulants are modafinil, famprofazone, tuaminoheptane, amiphenazole, amphetamine, methamphetamine, dimethylamphetamine, methylphenidate, 3,4-methylene-dioxy-N-amphetamine, 3,4-methylene-dioxy-N-ethylamphetamine, strychnine and 3,4-methylene-dioxy-N-methamphetamine etc.
Table 1.
General nucleus of corticoids and different substitutions of different glucocorticoids drugs.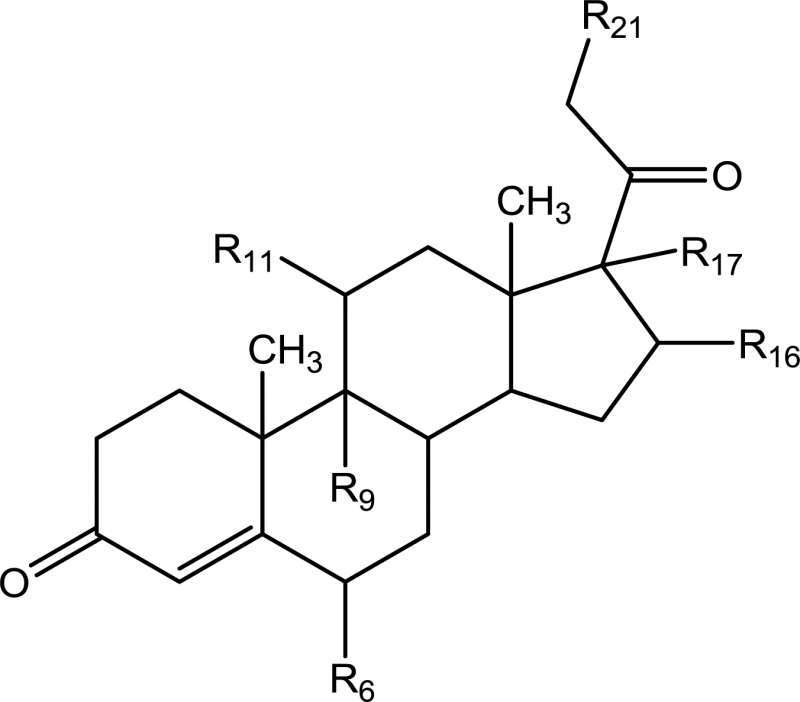 .
.
| Steroid name | Δ1, 2 | R6 | R9 | R11 | R16 | R17 | R21 |
|---|---|---|---|---|---|---|---|
| Hydrocortisone | Saturated | 3H | 3H | 3–OH | 3H | 3–OH | 3–OH |
| Cortisone | Saturated | 3H | 3H | åO | 3H | 3–OH | 3–OH |
| Corticosterone | Saturated | 3H | 3H | 3–OH | 3H | 3H | 3–OH |
| Fludrocortisone | Saturated | 3H | 3F | 3–OH | 3H | 3–OH | 3–OH |
| Prednisolone | Double bond | 3H | 3H | 3–OH | 3H | 3–OH | 3–OH |
| Prednisone | Double bond | 3H | 3H | åO | 3H | 3–OH | 3–OH |
| Methylprednisolone | Double bond | 3–CH3 | 3H | 3–OH | 3H | 3–OH | 3–OH |
| Prednisolone acetate | Double bond | 3H | 3H | 3–OH | 3H | 3–OH | 3–OCOCH3 |
| Dexamethasone | Double bond | 3H | 3F | 3–OH |
 (á isomer) (á isomer) |
3–OH | 3–OH |
| Betamethasone | Double bond | 3H | 3F | 3–OH |
 (â isomer) (â isomer) |
3–OH | 3–OH |
| Beclomethasone | Double bond | 3H | 3Cl | 3–OH |
 (â isomer) (â isomer) |
3–OH | 3–OH |
| Triamcinolone | Double bond | 3H | 3F | 3–OH | 3–OH | 3–OH | 3–OH |
| Triamcinolone acetonide | Double bond | 3H | 3F | 3–OH | 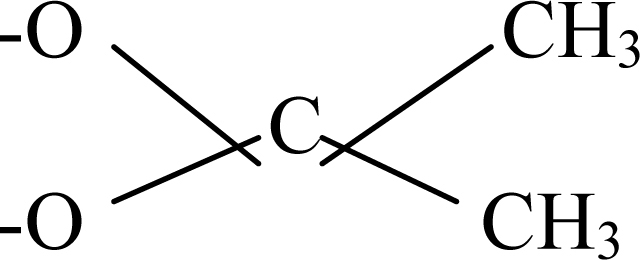 |
3–OH | |
Thus there is need for sophisticated and robust analytical strategy to confirm their presence in biological fluids. A number of different analytical approaches are available for this purpose. Analytical techniques such as high performance liquid chromatography (HPLC), ultra high performance liquid chromatography (UHPLC), mass spectrometry, gas chromatography, electrochemical detection and enhanced confirmatory procedures are used in detection of these classes of drugs. Recent development and advancement in analytical technologies has emerged with more sophisticated hyphenated techniques, to detect nanograms of drugs or their metabolites in biological fluids. Among hyphenated techniques, LC–MS/MS is the choice of interest because it is highly sophisticated and considerably powerful tool for detection of low and high molecular weight analytes. New methods have enabled the determination of drugs that were formerly difficult to detect by conventional methods of analysis as well as time consuming procedures have been replaced by faster, more comprehensive and robust assays. Good sensitivity and high throughput are key factors for the LC–MS/MS approaches used in drug analysis.
In this review, we present an overview of the methodologies that are reported in literature for detection, confirmation of corticosteroids and stimulants in biological fluids. Reported screening methods using LC–MS/MS approach highlight chromatographic separation and different modes of instrumental acquisition (selective ion monitoring (SIM), multiple reaction monitoring (MRM), precursor ion scan) for MS detection. Proposed mass fragmentation pathways of selected drugs are incorporated in this review.
2. Screening methods for detection of glucocorticoids and stimulants in biological fluids using liquid chromatography mass spectrometry
LC–MS/MS techniques provide specific, selective and sensitive quantitative results with reduced sample preparation. Other techniques such as electrochemical detection were also explored for the analysis of drugs. Goyal et al. [10], [11] investigated electrochemical behavior of dexamethasone and triamcinolone at the fullerene-C60-modified pyrolytic graphite electrode (PGE) using Osteryoung square wave voltammetry (SWV) and they illustrated quantitative determination of dexamethasone and triamcinolone in several commercially available pharmaceutical formulations and human blood plasma of patients being treated with dexamethasone. LC–MS/MS is currently most ideal tools for screening and quantifying corticosteroids in biological fluids as compared to other conventional techniques. This technique is widely used for pharmacokinetic (PK) studies, metabolites identification in plasma and urine, doping analysis and forensic studies. The introduction of commercial hyphenated instruments in which liquid chromatography is coupled with different mass analyzers such as time of flight mass spectrometry (LC–TOFMS), triple quadrupole mass spectrometer and soft ionization techniques [electrospray ionization (ESI), atmospheric pressure chemical ionization (APCI), atmospheric pressure photo ionization (APPI), and matrix assisted laser desorption ionization (MALDI) etc.] has simplified the laboratory analysis and also decreased the cost of analysis to some extent. The polarity and functionalities of corticosteroids and stimulants allow the use of ionization techniques in positive ion or negative ion modes and different modes of instrumental acquisition for mass detection. Table 2, Table 3 summarize the information on methods used for identification and confirmation of corticosteroids [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25] and selected stimulants [26], [27], [28], [29], [30].
Table 2.
Selected confirmatory methods for detecting glucocorticoids in biological fluids.
| Analyte | Matrix | Pretreatment | Column; mobile phase | Detection mode | LOD | Ref. |
|---|---|---|---|---|---|---|
| 9 Synthetic corticosteroids | Urine | LLE with Extrelut-NT3 columns | Inertsil ODS-3 C18 column (150 mm×3 mm, 3 μm); 1 mM ammonium acetate pH 6.8/ACN (60:40, v/v) at 0.4 mL/min | ESI negative SIM mode | 1 ng/mL | [12] |
| 20 Corticosteroids | Urine | Enzymatic hydrolysis C-8 SCX SPE Cartridge | Supelcosil LC-8-DBcolumn (2.1 mm×3.3 cm, 3 μm); 5 mM acetic acid/10 mM ammonium acetate (pH 3.8)/methanol, at 0.2 mL/min | ESI positive MRM | 0.1–10 ng/mL | [13] |
| Methylprednisolone methylprednisolone acetate | Plasma | Protein precipitation | Sinergy Max RP C-12 column (2.0 mm×150 mm, 4 μm); 0.01% formic acid in water/ACN (50:50, v/v) at 0.2 mL/min | ESI positive MRM | 6–300 ng/mL | [14] |
| 17 Synthetic glucocorticoids | Urine | Enzymatic hydrolysis followed by LLE | BEH C18 column(2.1 mm×50 mm, 1.7 μm); 0.1% formic acid in ACN/0.1% formic acid in water, at 0.35 mL/min | TOF-MS with ESI positive | 0.1–3.3 μg/L | [15] |
| Methylprednisolone | Methylprednisolone Plasma | LLE | Pursuit C-18 column (2.0 mm×150 mm, 5 μm); ACN/0.5% formic acid aq. solution (85:15, v/v) at 0.2 mL/min | ESI positive SRM | 20–2000 ng/mL | [16] |
| 5 Corticosteroids: dexamethasone, flumethasone, fluorometholone, beclomethasone, triamcinolone acetonide | Urine | Enzymatic hydrolysis Oasis-HLB SPE column | Alltima C-18 column (2.1 mm × 150 mm, 5 µm); ACN/water(40:60, v/v) at 0.3 mL/min | APCI positive SRM mode | 2–3 ng/mL | [17] |
| Cortisol, cortisone, prednisolone, prednisone | Plasma | Protein precipitation | Zorbax-SB phenyl, HT rapid resolution column (2.1 mm×100 mm, 1.8 μm); ACN/H2O/ formic acid(32:68:0.1, v/v) at 0.140 mL/min | APCI positive MRM mode | 0.5–2 ng/mL | [18] |
| Prednisone, prednisolone, dexamethasone, cortisol | Human serum | SPE | Symmetry C-18 column (2.1 mm×30 mm, 3.5 μm); methanol/5 mM acetate buffer pH 3.25 at 400 μL/min | ESI negative MRM mode | 0.2–0.5 ng/mL | [19] |
| Cortisol, dexamethasone, prednisone, prednisolone, methylprednisolone | Plasma | SPE Oasis HLB Cartridge | Symmetry C-18 column (2.1 mm×30 mm, 3.5 μm); 5 mM ammonium acetate buffer pH 3.5 and methanol (95:5, v/v)/methanol and 5 mM ammonium acetate buffer pH 3.5 (95:5, v/v), at 400 μL/min | ESI negative MRM mode | 3–7 ng/mL | [20] |
| 21 Synthetic corticosteroids | Urine | Enzymatic hydrolysis followed by LLE | RP DB-8 column (4.6 mm×75 mm, 3 μm); 1% acetic acid/ methanol, at 1 mL/min | APCI positive full scan mode | 5 ng/mL | [21] |
| Cortisol, cortisone, prednisolone, dexamethasone, 11-deoxycortisol | Plasma, urine saliva, plasma-ultra filtrate | SPE Oasis HLB 1 cm3 Cartridge | Acquity UPLC BEH C-18 column (2.1 mm×50 mm, 1.7 μm); A=2 mM ammonium acetate in water with 0.1% formic acid, B=2 mM ammonium acetate in methanol with 0.1% formic acid, at 0.4 mL/min, total run time 3 min. | ESI positive MRM | 0.6–5 nM | [22] |
| 16 Glucocorticoids | Urine | Enzymatic hydrolysis followed by LLE | Halo C-18 column(2.1 mm×150 mm, 2.7 μm and 2.1 mm×100 mm, 2.7 μm); water with 0.1% formic acid/ACN with 0.1% formic acid at 400 μL/min | ESI positive MRM | 5–15 ng/mL | [23] |
| 15 Synthetic glucocorticoids | Urine | Enzymatic hydrolysis followed by LLE | Zorbax C-18 column (2.1 mm×50 mm, 1.8 μm); 0.1% acetic acid/ACN with 0.1% acetic acid at 0.3 mL/min | ESI positive MRM | 1–30 ng/mL | [24] |
| 14 Glucocorticoids | Urine | Enzymatic hydrolysis followed by LLE | Inertsil ODS-3 C18 column(50 mm×4.6 mm, 3 μm); 1% formic acid/ACN, at 700 μL/min | ESI positive MRM | 1–5 ng/mL | [25] |
Ref.—References.
Table 3.
Selected confirmatory methods for detecting stimulants in biological fluids.
| Analyte | Matrix | Pretreatment | Column; Mobile phase | Detection mode | LOD | Ref. |
|---|---|---|---|---|---|---|
| 9 Stimulants including strychnine, methylphenidate, amiphenazole, famprofazone, tuaminoheptan | Urine | Enzymatic hydrolysis followed by LLE | Halo C-18 column (2.1 mm ×150 mm, 2.7 μm and 2.1 mm×100 mm, 2.7 μm); water with 0.1% formic acid/ACN with 0.1% formic acid at 400 μL/min | ESI positive MRM | 70–300 ng/mL | [23] |
| Mesocarb, modafinil, formoterol | Urine | Enzymatic hydrolysis followed by LLE | Zorbax C-18 column (2.1 mm×50 mm,1.8 μm); 0.1% acetic acid/ACN with 0.1% acetic acid at 0.3 mL/min | ESI positive MRM | 100–200 ng/mL | [24] |
| Methylphenidate, mesocarb | Urine | Enzymatic hydrolysis followed by LLE | Inertsil ODS-3 C18 column(50 mm×4.6 mm, 3 μm); 1% formic acid/ACN, at 700 μL/min | ESI positive MRM | 5 ng/mL | [25] |
| 49 Stimulants | Urine | Enzymatic hydrolysis SPE cartridge | Phenomex Luna C-18 column( 2 mm ×100 mm, 3 μm); 5 mM ammonium acetate with 0.1% formic acid/ ACN, at 0.3 mL/min | ESI positive | 0.025 μg/mL | [26] |
| 27 Amphetamine and amphetamine like drugs | Urine | LLE | Omnispher C-18 column (3.0 mm×50 mm, 3 μm); 0.1% formic acid/methanol, at 0.4 mL/min | APCI positive full scan MS | 1–25 ng/mL | [27] |
| 7 Amphetamine derivatives: AP, MA, MDA, MDMA,MDEA, DMA, DMANO | Urine | Oasis HLB SPE | Capcell C-18 MG-II column (2.0 mm×150 mm, 5 μm); 5 mM ammonium formate pH 4.0/ACN, at 230 μL/min | ESI positive MRM | 1.95 ng/mL | [28] |
| Mesocarb | Urine | Enzymatic hydrolysis followed by LLE | Thermo-Hyper Gold C-18 (2.1 mm×50 mm, 3 μm ); 15 mM ammonium formate containing 1% formic acid/ACN at 200 μL/min | SRM positive | 5 ng/mL | [29] |
| AP, MA, MDA, MDMA, MDEA, PMA, ephedrine | Urine | LLE | Acquity UPLC HSS C-18 column(2.1 mm×100 mm, 1.8 μm ); 5 mM ammonium formate buffer containing 0.05% formic acid/methanol, at 0.3 mL/min | ESI positive MRM | 0.5–2.5 ng/mL | [30] |
AP: amphetamine, MA: methamphetamine, MDA: 3,4-methylenedioxy-N-amphetamine, DMA: N, N-dimethylamphetamine, PMA: p-hydroxymethamphetamine, MDMA: 3,4-methylenedioxy-N-methamphetamine, MDEA: 3,4-methylenedioxy-N-ethylamphetamine, DMANO: N, N-dimethyl amphetamine-N-oxide, SPE: solid phase extraction, LLE: liquid–liquid extraction, ACN: acetonitrile, Ref: references.
Fluri et al. [12] reported confirmatory method for 11 corticosteroids in urine samples based on LC–ESI–MS. The selective and sensitive method for confirmation and identification of nine synthetic corticosteroids assured the exclusion of false positive results obtained by corticosteroid group ELISA screening tests. Emmie et al. [13] developed two high throughput LC–MS methods for the screening of anabolic steroids, corticosteroids, and acidic drugs using a high efficiency LC column coupled with a fast scanning triple quadrupole mass spectrometer. The detection of 40 anabolic steroids, corticosteroids, and 52 acidic drugs were achieved within a 6.5 min and a 4.5 min LC–MRM run, respectively and all the targets were detected at low amount. Validation data showed that both methods have acceptable precision to be used on a routine basis and no interference from sample matrix was observed.
A rapid, sensitive and specific method for the simultaneous detection and quantization of methylprednisolone acetate (MPA) and methylprednisolone (MP) in rat plasma, using a triple stage quadrupole was developed and validated by Panusa et al. [14] using LC–ESI–MS/MS. Its excellent applicability in PK studies was demonstrated. It was proved to be highly sensitive, allowing detection and quantization of the analytes at lower concentrations.
Method was developed by Touber et al. [15] using the latest high-resolution LC column technology, UPLC™, and ESI interface in the positive ion mode. Gradient UPLC separation conditions were optimized for a group of 22 analytes including 17 glucocorticosteroids. The UPLC–TOF–MS separation obtained required 5.5 min only for all the substances tested. Even the critical pair of dexamethasone and betamethasone isomers was almost completely resolved. The authors recommended that dedicated UPLC–TOF–MS criteria regarding the number of identification points (IPs), mass accuracy of parent, fragment ions, ion ratio, and relative retention time have not been assessed, in order to allow application of this new technology for confirmation of identity as well, that should be considered and thus needs improvement.
Mazzarino et al. [23] performed screening of several classes of substances in a single chromatographic method with a run-time of 11 min, inclusive of post-run and reconditioning times. The effectiveness of this approach was evaluated by LC–ESI–MS/MS in the positive mode, using 20 blank urine samples spiked with 45 compounds prohibited in sport including 16 glucocorticoids and 9 stimulants. All of the analytes were clearly distinguishable in urine, with limits of detection ranging from 5 ng/mL to 350 ng/mL. All the compounds of interest were separated, including synthetic and endogenous glucocorticoids with similar retention times and fragmentation patterns.
Kolmonen et al. [26] developed a general screening method based on solid phase extraction (SPE), LC–TOF/MS and validated 124 different doping agents including stimulants in urine. The result indicated that compared with conventional doping control methods, this method was more flexibility in identification, database management and reduced the time required for analysis.
A selective and sensitive method for the qualitative screening of urine samples for 27 amphetamine and amphetamine-type drugs was described by Deventer et al. [27] using mass spectrometer equipped with APCI interface, operated in positive ionization mode. They reported that the amount of urine routinely used in their laboratory for the extraction of these stimulants (5 mL) was reduced to 2 mL and thus reduced sample volume. The detection limits for all the compounds were lower than 25 ng/mL except for chlorphentermine, thus it was good alternative to gas chromatography with nitrogen phosphorus detector (GC-NPD).
Kim et al. [28] developed and validated LC–ESI–MS/MS method for the simultaneous detection and quantification of seven amphetamine derivatives amphetamine (AP), methamphetamine (MA), 3,4-methylenedioxy-N-amphetamine (MDA), 3,4-methylenedioxy-N-methamphetamine (MDMA), 3,4-methylenedioxy-N-ethylamphetamine (MDEA), N,N-dimethylamphetamine (DMA) and N,N-dimethylamphetamine-N-oxide (DMANO) in human urine. The paper reported that the SPE step was assayed to detect and quantify seven target analytes in urine samples without any significant interference from the matrix components.
Counterfeiting of herbal drugs with synthetic agents can also be detected by LC–MS/MS [31], [32]. Different mass analyzers (TOF, IT (ion trap)) have been coupled with LC and extensively used for screening and characterization of different analytes in plasma and herbal extracts [33], [34].
3. Focusing on different strategies for detection of glucocorticoids and stimulants in biological fluids
Based on the reported MS-fragmentation data, a common fragmentation could be developed in screening of corticosteroids and stimulants. The basic information regarding empirical formula, exact masses, Log P and absorption maxima (λmax) is also summarized in Table 4. P and Log P are partition coefficient or logarithm of the partition coefficient of a drug. These parameters express the relative distribution of drug between oil and water under specified conditions for example, octanol/water at 37 °C and pH 7.4. Drugs with higher P or Log P are more lipophilic, generally distribute more rapidly and to a greater degree into bodily tissues and fluids. These physicochemical properties help in prediction of drug transport. These data render to presume interaction of analyte with stationary phase, thereby allowing a good evaluation of its chromatographic performance. UV spectra can help as a preliminary screening to distinguish different analytes on the basis of their absorption maxima (λmax). The super-imposable nature of UV spectra of an analyte with a certified reference material (CRM) can give fair idea about their structural similarity to much extent. This approach has been employed for detection of adulteration using HPLC with UV detector.
Table 4.
Empirical formula, exact masses, MS fragmentation data, Log P and λmax of selected glucocorticoids and stimulants.
| Drug name | Empirical formula | Exact mass | [M+H]+ | m/z of major fragments reported | Log Pa | λmax (nm) | Ref. |
|---|---|---|---|---|---|---|---|
| Amphetamine | C9H13N | 135.10480 | 136.2 | 119.2, 91.2 | 1.8 | 257, 263 | [28] |
| MDA | C10H13NO2 | 179.09463 | 180.2 | 163, 134.9, 105 | 1.64 | 233, 285 | [28], [30] |
| MDMA | C11H15 NO2 | 193.11028 | 194.1 | 163.2, 105.1 | 2.15 | 234, 285 | [28], [30] |
| Beclomethasone | C22H29ClO5 | 408.17035 | 409.18 | 391, 373, 337 | 2.03 | 239 | [17], [24] |
| Betamethasone | C22H29FO5 | 392.19990 | 393 | 337, 355, 241, 147 | 1.94 | 240 | [24], [25], [35], [37] |
| Budesonide | C25H34O6 | 430.23554 | 431 | 413, 341, 323, 173 | 2.18 | 248 | [24], [32], [36], [37] |
| Dexamethasone | C22H29FO5 | 392.19990 | 393 | 373, 355, 337, 237, 147 | 1.83 | 240 | [17], [24], [35], [37] |
| Fludrocortisone | C21H29FO5 | 380.19990 | 381 | 343, 239 | 1.67 | 240 | [24], [36] |
| Flumethasone | C22H28F2O5 | 410.19047 | 411 | 371, 335, 253, 121 | 1.9 | 238 | [17], [24], [32], [35] |
| Fluocortolone | C22H29FO4 | 376.20499 | 377 | 321, 303 | 2.1 | 242 | [24] |
| Methylphenidate | C14H19NO2 | 233.14158 | 234 | 174, 129, 84, 56 | 0.20 | 264 | [23], [25], [35] |
| Methylprednisolone | C22H30O5 | 374.20932 | 375 | 357, 339, 161 | 1.82 | 240 | [16], [25], [36], [37] |
| Prednisolone | C21H28O5 | 360.19367 | 361 | 343, 325, 147, 307 | 1.62 | 240 | [24], [25], [35], [36], [37] |
| Prednisone | C21H26O5 | 358.17802 | 359 | 341, 323, 267, 147, 34 | 1.46 | 240 | [24], [25], [35], [36], [37] |
| Strychnine | C21H22N2O2 | 334.16813 | 335 | 264, 184, 156 | 1.9 | 255 | [21], [23] |
| Triamcinolone | C21H27FO6 | 394.17917 | 395 | 357, 321, 225 | 1.16 | 238 | [24], [25], [36], [37] |
| Triamcinolone | C24H31FO6 | 434.21047 | 435 | 415, 397, 213 | 1.2 | 238 | [24], [25], [36], [37] |
| Acetonide |
Values obtained from online SRC Phys Prop Database, Ref.—References.
The precursor and products ions summarized in Table 4, are based on positive ionization. Depending upon the nature of target analytes, ionization mode is optimized. Fluri et al. [12] studied both negative and positive ionization modes for examined glucocorticoids. Due to less fragmentation and a better signal-to-noise ratio (4:1), sensitivity increased for measurements in the negative ionization mode. They assessed that fragmentation of corticosteroids in the ESI negative mode is simple as few ions were produced. The products ions of ten selected corticosteroids by their study are presented in Table 5.
Table 5.
Characteristics fragment ions of glucocorticoids obtained in ESI negative mode LC–MS/MS.
| Analyte | m/z fragments |
|---|---|
| Cortisone | 329.2a, 301.2, 311.2 |
| Dexamethasone | 361.2a, 307.2, 325.2 |
| Hydrocortisone | 331.2a, 297.2, 282.2 |
| Flumethasone | 379.2a, 325.2, 305.2 |
| Flunisolide | 375.2a, 357.2, 433.2 |
| Methylpednisolone | 343.2a, 309.2, 294.2 |
| Prednisolone | 329.2a, 295.2, 280.2 |
| Prednisone | 327.2a, 299.2, 285.2 |
| Triamcinolone | 345.2a, 325.2, 393.2 |
| Triamcinolone acetonide | 413.2a, 337.2, 375.2, 469.2 |
Indicate base peak.
4. Proposed mass fragmentation pattern
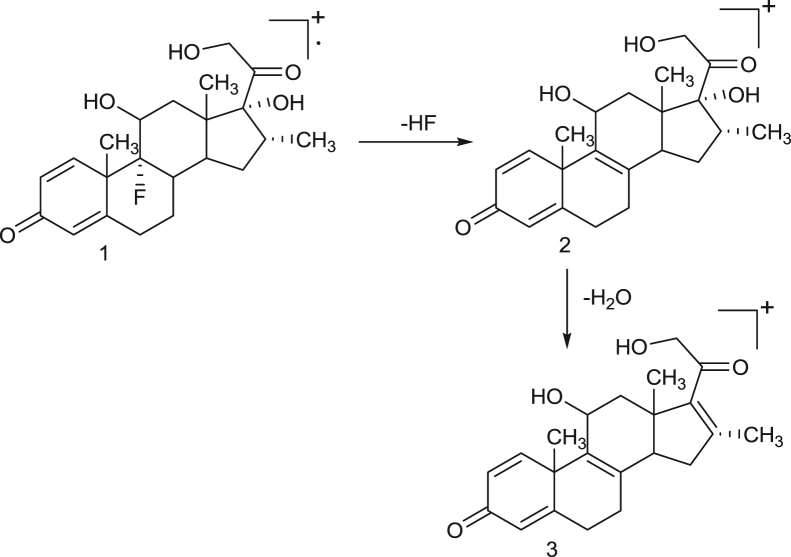
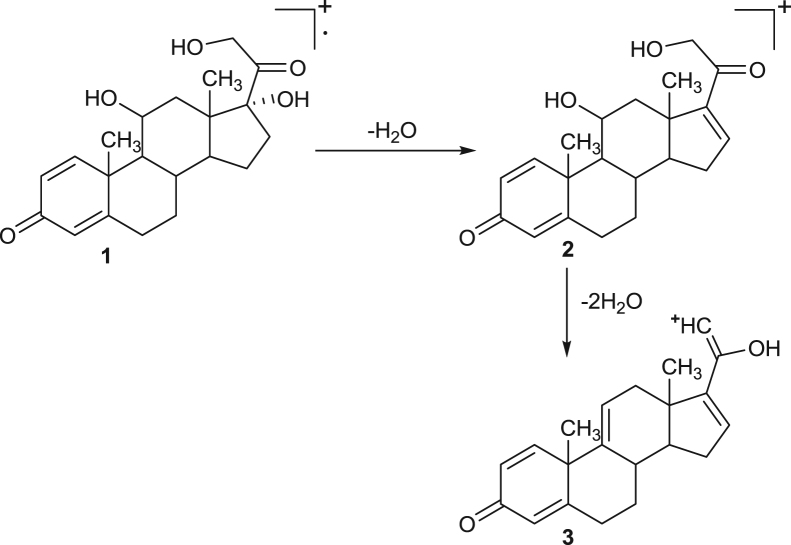
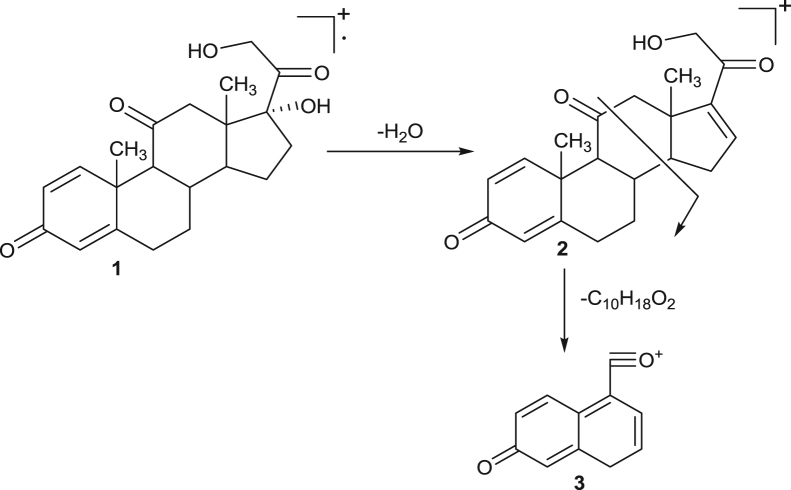
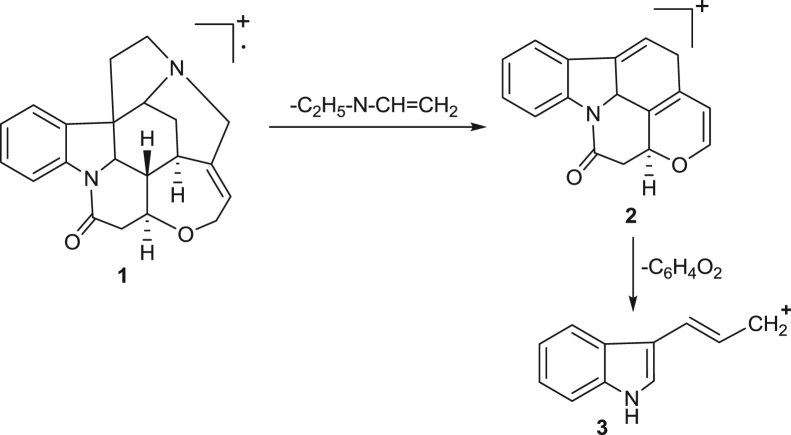
The hypothetical mass fragmentation pattern of some selected drugs like dexamethasone, prednisolone, prednisone and strychnine on the basis of MRM transitions has been incorporated and that helps in confirmation of analytes on the basis of diagnostic ions (Fig. 1, Fig. 2, Fig. 3, Fig. 4). The daughter ions generated from the parent ions help to predict the fragmentation pattern of the molecule and are useful in confirmation of the target analytes.
Fig. 1.
Proposed mass fragmentation pathway of dexamethasone drug. 1—Molecular ion peak at M+=393. 2—Base ion peak at m/z=373. 3—Daughter ion peak at m/z=355.
Fig. 2.
Proposed mass fragmentation pathway of prednisolone drug. 1—Molecular ion peak at M+=361. 2—Base ion peak at m/z=343. 3—Daughter ion peak at m/z=307.
Fig. 3.
Proposed mass fragmentation pathway of prednisone drug. 1—Molecular ion peak at M+=359. 2—Base ion peak at m/z=341. 3—Daughter ion peak at m/z=171.
Fig. 4.
Proposed mass fragmentation pathway of strychnine drug. 1—Molecular ion peak at M+=335. 2—Base ion peak at m/z=264. 3—Daughter ion peak at m/z=156.
5. Conclusion
With the advancement of hyphenated techniques, high resolution mass analyzers as well as high throughput separation approaches, quantitative analysis of glucocorticoids and stimulants can be achieved with good sensitivity. Newer methods can be developed for routine analysis of target analytes in biological fluids with shorter run time and good detectability. Application of new fused core columns for their effectiveness and use of both positive and negative polarities in a single run need to explore.
Footnotes
Peer review under responsibility of Xi'an Jiaotong University.
References
- 1.Perrenoud L., Saugy M., Saudan C. Detection in urine of 4-methyl-2-hexaneamine a doping agent. J. Chromatogr. B. 2009;877(29):3767–3770. doi: 10.1016/j.jchromb.2009.09.013. [DOI] [PubMed] [Google Scholar]
- 2.C. Judkins, D. Hall, K. Hoffman, Investigation into Supplement Contamination Levels in the US Market, HFL. 2007. Available from: 〈http://www.supplementsafetynow.com/HFL-supplement-research-report.pdf〉.
- 3.Savaliya A.A., Prasad B., Raijada D.K. Detection and characterization of synthetic steroidal and non-steroidal anti-inflammatory drugs in Indian ayurvedic/herbal products using LC–MS/TOF. Drug Test Anal. 2009;1(8):372–381. doi: 10.1002/dta.75. [DOI] [PubMed] [Google Scholar]
- 4.Ahmed S., Riaz M. Quantitation of corticosteroids as common adulterants in local drugs by HPLC. Chromatographia. 1991;31(1–2):67–70. [Google Scholar]
- 5.Ramsay H.M., Goddard W., Gill S. Herbal creams used for atopic eczema in Birminghan, U.K. illegally contains potent corticosteroids. Arch. Dis. Child. 2003;88(12):1056–1057. doi: 10.1136/adc.88.12.1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beotra A., Gupta Y.K., Ahi S. Preliminary studies on detection of corticosteroids in adulterated herbal drugs. Recent Adv. Doping Anal. 2009;18:208–211. [Google Scholar]
- 7.Uchiyama N., Hanajiri R.K., Kawahara N. Identification of a cannabimimetic indole as a designer drug in a herbal product. Forensic Toxicol. 2009;27:61–66. [Google Scholar]
- 8.Deventer K., Thuyne W.V., Mikulcikova P. Detection of selected stimulants as contaminants in solid nutritional supplements by liquid chromatography–mass spectrometry. Food Chem. 2007;103:1508–1513. [Google Scholar]
- 9.Delgado J.N., Remers W.A. 10th ed. Lippincott Raven; Philadelphia, New York: 1998. Wilson and Gisvold's Textbook of Organic Medicinal and Pharmaceutical Chemistry. pp. 772–774. [Google Scholar]
- 10.Goyal R.N., Gupta V.K., Chatterjee S. Fullerene-C60-modified edge plane pyrolytic graphite electrode for the determination of dexamethasone in pharmaceutical formulations and human biological fluids. Biosens. Bioelectron. 2009;24:1649–1654. doi: 10.1016/j.bios.2008.08.024. [DOI] [PubMed] [Google Scholar]
- 11.Goyal R.N., Gupta V.K., Chatterjee S. A sensitive voltammetric sensor for determination of synthetic corticosteroid triamcinolone, abused for doping. Biosens. Bioelectron. 2009;24:356–3562. doi: 10.1016/j.bios.2009.05.016. [DOI] [PubMed] [Google Scholar]
- 12.Fluri K., Rivier L., Nagy A.D. Method for confirmation of synthetic corticosteroids in doping urine samples by liquid chromatography-electrospray ionisation mass spectrometry. J. Chromatogr. A. 2001;926:87–95. doi: 10.1016/s0021-9673(01)01065-2. [DOI] [PubMed] [Google Scholar]
- 13.Emmie N.M.H., Leung D.K.K., Terence S.M.W. Comprehensive screening of anabolic steroids, corticosteroids, and acidic drugs in horse urine by solid phase extraction and liquid chromatography–mass spectrometry. J. Chromatogr. A. 2006;1120:38–53. doi: 10.1016/j.chroma.2006.03.089. [DOI] [PubMed] [Google Scholar]
- 14.Panusa A., Orioli M., Aldinia G. A rapid and sensitive LC–ESI–MS/MS method for detection and quantitation of methylprednisolone and methylprednisolone acetate in rat plasma after intra-articular administration, J. Pharm. Biomed. Anal. 2010;51:691–697. doi: 10.1016/j.jpba.2009.09.041. [DOI] [PubMed] [Google Scholar]
- 15.Touber M.E., Engelen M.C.V., Georgakopoulus C. Multi-detection of corticosteroids in sports doping and veterinary control using high-resolution liquid chromatography/time-of-flight mass spectrometry. Anal. Chim. Acta. 2007;586:137–146. doi: 10.1016/j.aca.2006.09.058. [DOI] [PubMed] [Google Scholar]
- 16.Zhang S.Q., Thorsheim H.R., Penugonda S. Liquid chromatography–tandem mass spectrometry for the determination of methylprednisolone in rat plasma and liver after intravenous administration of its liver-targeted dextran prodrug. J. Chromatogr. B. 2009;877:927–932. doi: 10.1016/j.jchromb.2009.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Keeffe M.J.O., Martin S., Regan L. Validation of a multiresidue liquid chromatography–tandem mass spectrometric method for the quantitation and confirmation of corticosteroid residues in urine, according to the proposed SANCO 1085 criteria for banned substances. Anal. Chim. Acta. 2003;483:341–350. [Google Scholar]
- 18.Ionita I.A., Fast D.M., Akhlaghi F. Development of a sensitive and selective method for the quantitative analysis of cortisol, cortisone, prednisolone and prednisone in human plasma. J. Chromatogr. B. 2009;877:765–772. doi: 10.1016/j.jchromb.2009.02.019. [DOI] [PubMed] [Google Scholar]
- 19.Frerichs V.A., Tornatore K.M. Determination of the glucocorticoids prednisone, prednisolone, dexamethasone, and cortisol in human serum using liquid chromatography coupled to tandem mass spectrometry. J. Chromatogr. B. 2004;802:329–338. doi: 10.1016/j.jchromb.2003.12.015. [DOI] [PubMed] [Google Scholar]
- 20.Francesco R.D., Frerichs V., Donnelly J. Simultaneous determination of cortisol, dexamethasone, methylprednisolone, prednisone, prednisolone, mycophenolic acid and mycophenolic acid glucuronide in human plasma utilizing liquid chromatography–tandem mass spectrometry. J. Chromatogr. B. 2007;859:42–51. doi: 10.1016/j.jchromb.2007.09.003. [DOI] [PubMed] [Google Scholar]
- 21.Tang P.W., Law W.C., Wan T.S.M. Analysis of corticosteroids in equine urine by liquid chromatography–mass spectrometry. J. Chromatogr. B. 2001;754:229–244. doi: 10.1016/s0378-4347(00)00613-7. [DOI] [PubMed] [Google Scholar]
- 22.Mc-Whinney B.C., Briscoe S.E., Ungerer J.P.J. Measurement of cortisol, cortisone, prednisolone, dexamethasone and 11-deoxycortisol with ultra high performance liquid chromatography–tandem mass spectrometry: application for plasma, plasma ultrafiltrate, urine and saliva in a routine laboratory. J. Chromatogr. B. 2010;878:2863–2869. doi: 10.1016/j.jchromb.2010.08.044. [DOI] [PubMed] [Google Scholar]
- 23.Mazzarino M., Torre X., Botre F. A rapid screening LC–MS/MS method based on conventional HPLC pumps for the analysis of low molecular weight xenobiotics: application to doping control analysis. Drug Test Anal. 2010;2:311–322. doi: 10.1002/dta.148. [DOI] [PubMed] [Google Scholar]
- 24.Mazzarino M., Botre F. A fast liquid chromatographic/mass spectrometric screening method for the simultaneous detection of synthetic glucocorticoids, some stimulants, anti-oestrogen drugs and synthetic anabolic steroids. Rapid Commun. Mass Spectrom. 2006;20:3465–3476. doi: 10.1002/rcm.2729. [DOI] [PubMed] [Google Scholar]
- 25.Reddy I.M., Beotra A., Jain S. A simple and rapid ESI–LC–MS/MS method for simultaneous screening of doping agents in urine samples. Ind. J. Pharm. 2009;41(2):80–86. doi: 10.4103/0253-7613.51347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kolmonen M., Leinonen A., Pelander A. A general screening method for doping agents in human urine by solid phase extraction and liquid chromatography/time-of-flight mass spectrometry. Anal. Chim. Acta. 2007;585:94–102. doi: 10.1016/j.aca.2006.12.028. [DOI] [PubMed] [Google Scholar]
- 27.Deventer K., Eenoo P.V., Delbeke F.T. Screening for amphetamine and amphetamine-type drugs in doping analysis by liquid chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2006;20:877–882. doi: 10.1002/rcm.2372. [DOI] [PubMed] [Google Scholar]
- 28.Kim J.Y., Cheong J.C., Ko B.J. Simultaneous determination of methamphetamine, 3,4-methylenedioxy- N-methylamphetamine, 3,4-methylenedioxy-N-ethylamphetamine, N,N-dimethylamphetamine and their metabolites in urine by liquid chromatography-electrospray ionization-tandem mass spectrometry. Arch. Pharm. Res. 2008;31(12):1644–1651. doi: 10.1007/s12272-001-2163-5. [DOI] [PubMed] [Google Scholar]
- 29.Kang M.J., Hwang Y.H., Lee W. Validation and application of a screening method for b2-agonists, anti-estrogenic substances and mesocarb in human urine using liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2007;21:252–264. doi: 10.1002/rcm.2834. [DOI] [PubMed] [Google Scholar]
- 30.Fernandez M.M.R., Wille S.M.R., Fazio V.D. Analysis of amphetamines and metabolites in urine with ultra performance liquid chromatography tandem mass spectrometry. J. Chromatogr. B. 2010;878:1616–1622. doi: 10.1016/j.jchromb.2010.03.048. [DOI] [PubMed] [Google Scholar]
- 31.Gratz S.R., Flurer C.L., Wolnik K.A. Analysis of undeclared synthetic phosphodiesterase-5 inhibitors in dietary supplements and herbal matrices by LC–ESI–MS and LC-UV. J. Pharm. Biomed. Anal. 2004;36(3):525–533. doi: 10.1016/j.jpba.2004.07.004. [DOI] [PubMed] [Google Scholar]
- 32.Wiergowski M., Tatarowicz K.G., Banasik L.N. Hazard for human health and life by unintentional use of synthetic sibutramine, which was sold as Chinese herbal product meizitanc. Przegl. Lek. 2007;64(4–5):268–272. [PubMed] [Google Scholar]
- 33.Polagania S.R., Pillib N.R., Gandu V. High performance liquid chromatography mass spectrometric method for the simultaneous quantification of pravastatin and aspirin in human plasma: pharmacokinetic application. J. Pharm. Anal. 2012;2(3):206–213. doi: 10.1016/j.jpha.2012.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen X.F., Wu H.T., Tan G.G. Liquid chromatography coupled with time-of-flight and ion trap mass spectrometry for qualitative analysis of herbal medicines. J. Pharm. Anal. 2011;1(4):235–245. doi: 10.1016/j.jpha.2011.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bogusz M.J., Hassan H., Enazi E.A. Application of LC–ESI–MS–MS for detection of synthetic adulterants in herbal remedies. J. Pharm. Biomed. Anal. 2006;41:554–564. doi: 10.1016/j.jpba.2005.12.015. [DOI] [PubMed] [Google Scholar]
- 36.Tseng M.C., Tsai M.J., Lin J.H. GC/MS analysis on anorectics adulterated in traditional Chinese medicines. J. Food Drug Anal. 2000;8(4):315–330. [Google Scholar]
- 37.Taylor R.L., Grebe S.K., Singh R.J. Quantitative highly sensitive liquid chromatography-tandem mass spectrometry method for detection of synthetic corticosteroids. Clin. Chem. 2004;50(12):2345–2352. doi: 10.1373/clinchem.2004.033605. [DOI] [PubMed] [Google Scholar]