Abstract
An efficient screening assay was developed and validated for simultaneous assessment of compound-mediated inhibition of six major human cytochrome P450 (CYP) enzymes. This method employed a cocktail of six probe substrates (i.e., phenacetin, amodiaquine, diclofenac, S-mephenytoin, dextromethorphan and midazolam for CYP1A2, 2C8, 2C9, 2C19, 2D6 and 3A4, respectively) as well as individual prototypical inhibitors of the six CYP enzymes in human liver microsomes under optimized incubation conditions. The corresponding marker metabolites (i.e., acetaminophen, N-desethylamodiaquine, 4-OH-diclofenac, 4-OH-S-mephenytoin, dextrorphan and 1-OH-midazolam) in the incubates were quantified using LC–MS/MS methods either by an internal standard (IS) calibration curve or a simplified analyte-to-IS peak area ratio approach. The results showed that the IC50 values determined by the cocktail approach were in good agreement with those obtained by the individual substrate approach as well as those reported in the literature. Besides, no remarkable difference was observed between the two quantification approaches. In conclusion, this new cocktail assay can be used for reliable screening of compound-mediated CYP inhibition.
Keywords: LC–MS/MS, Cytochrome P450, Cocktail-probe, Inhibition assessment, Drug screenning
1. Introduction
The cytochrome P450 (CYP) enzyme system is a large and diverse super family of heme-containing enzymes that catalyze the oxidation of a wide variety of compounds. The primary function of CYP enzymes lies in the elimination of xenobiotics including therapeutic drugs and environmental toxins. These CYPs may play critical roles in the process of drug metabolism (e.g., drug clearance). Alterations in drug metabolism caused by co-administered drugs may lead to metabolic drug–drug interaction (DDI). The DDI effect may be due to either inhibition or induction of responsible CYP enzyme(s) [1], [2], [3]. In general, CYP inhibition is more common and more serious than CYP induction.
CYP enzyme activities can be assessed via two approaches, the individual probe substrate method and the cocktail probe substrate method (also known as cassette or N-in-1 method). With an increasing number of new chemical entities synthesized, quick methods for assessing CYP-mediated DDI potential are highly desirable. Besides, it is commonly recommended that the CYP-mediated DDI screening should be done as early as possible, in order to move forward projects more efficiently, where throughput matters more [4], [5]. For this reason, a probe-substrate cocktail approach was first developed for the assessment of in vivo DDI potential in the late of discovery stage [6], [7], and introduced to in vitro studies later on. Currently, the application of cocktail strategy has gained more and more popularity for both in vitro and in vivo DDI studies, mainly due to its high efficiency. It enables the simultaneous determination of activities of several CYP isozymes in one assay, thus saving significant amount of time and resources [8], [9], [10], [11], [12], [13]. However, prior to its use, a cocktail method should be well verified to clarify the concern in terms of interactions among the probe substrates.
To cope with the probe substrate cocktail strategy, rapid analytical methods with no or little compromise of specificity and sensitivity are required. Nowadays, liquid chromatography–tandem mass spectrometry (LC–MS/MS), which is highly sensitive and selective, has become an instrumental tool in CYP-mediated DDI assessment. Multi-reaction monitoring (MRM) function of the MS/MS system makes it possible to monitor multiple compounds simultaneously in a single run, enabling concurrent analysis of multiple probe substrates and/or their marker metabolites. Half maximal inhibitory concentration (IC50), representing the inhibitory potency of a compound, is generated based on fraction of activity inhibited or uninhibited, which is normally calculated by measuring concentrations of marker metabolite(s) using calibration curves. There is, however, a simpler way to calculate fraction of activity inhibited or uninhibited by substituting the concentration with analyte-to-IS peak area ratios. In this way, throughput can be further increased, but only after it is validated.
In the present study, a 6-probe cocktail incubation (i.e., phenacetin, amodiaquine, diclofenac, S-mephenytoin, dextromethorphan and midazolam for CYP1A2, 2C8, 2C9, 2C19, 2D6 and 3A4, respectively) was established for screening those compounds-mediated CYP inhibition in early stage of drug research. The inhibitory potency was presented with IC50 calculated by the amount of marker metabolites qualified by fully validated, rapid, and accurate LC–MS/MS method. The IC50 values determined by the cocktail approach would be compared with those obtained by the individual substrate approach.
2. Materials and methods
2.1. Chemicals and reagents
Phenacetin, dextromethorphan, midazolam and ketoconazole were purchased from the National Institutes for Food and Drug Control (Beijing, China). Amodiaquine, sulfaphenazole, nootakatone, quinidine, qucertin, α-naphthoflavone, diclofenac and β-NADPH were purchased from Sigma Chemical Company (Santa, USA). Dibasic potassium phosphate and magnesium chloride hexahydrate were purchased from SCRC (Beijing, China). Human liver microsomes were purchased from BD (Franklin Lakes, USA). Acetaminophen, N-desethylamodiaquine, 4-OH-diclofenac, 4-OH-S-mephenytoin, dextrorphan and 1-OH-midazolam were purchased from TRC (Toronto, Canada). Acetonitrile and formic acid were purchased from Merck (Darmstadt, Germany). Methanol was purchased from Tedia (Fairfield, USA). MgCl2, K2HPO4 and KH2PO4 were purchased from SCRC (Shanghai, China).
2.2. Analytical method
The MS/MS system was an Applied Biosystems API-3000 mass spectrometer with an electrospray ionization (ESI) probe (Toronto, Canada). MS/MS signals of analytes were first automatically optimized for generating compound-dependent parameters such as declustering potential, focusing potential, collision energy and collision cell exit potential, and then manually adjusted for obtaining other parameters such as curtain gas, nebulizer gas, heater gas, collision gas, source voltage and temperature. For construction of an MS/MS method simultaneously detecting six metabolites and IS, the mutual parameters (source-dependent parameters) were set on behalf of 4-OH-S-mephenytoin which represented the most challenging compound for MS/MS analysis. The optimized ion spray voltage and temperature were set at 3000 V and 400 °C. Nitrogen gas was used for the curtain gas, nebulizer gas (gas 1), heater gas (gas 2), collision gas, which were set at 14 psi, 12 psi, 700 mL/min, and 8 psi, respectively. Besides these mutual parameters, the ion transitions and other detection parameters for the analytes and IS are summarized in Table 1.
Table 1.
Summary of MS/MS parameters of metabolites and IS.
| Isoform | Probe | Metabolite | Precursor | Product | CE | CXP | DP | FP |
|---|---|---|---|---|---|---|---|---|
| CYP1A2 | Phenacetin | Acetaminophen | 152.0 | 110.0 | 23 | 8 | 26 | 110 |
| CYP2C8 | Amodiaquine | Desethyl-amodiaquine | 328.2 | 219.4 | 61 | 22 | 46 | 160 |
| CYP2C9 | Diclofenac | 4-OH-diclofenac | 312.0 | 231.0 | 29 | 14 | 31 | 200 |
| CYP2C19 | S-mephenytoin | 4-OH-S-mephenytoin | 235.2 | 150.1 | 25 | 12 | 21 | 110 |
| CYP2D6 | Dextromethorphan | Dextrophan | 258.1 | 199.1 | 53 | 14 | 31 | 200 |
| CYP3A4 | Midazolam | 1-OH-midazolam | 342.0 | 203.0 | 42 | 14 | 31 | 200 |
| IS | Verapamil | NA | 455.3 | 165.1 | 30 | 15 | 30 | 200 |
CE: collision energy (V); CXP: collision cell exit potential (V); DP: declustering potential (V); and FP: focus potential (V).
Separation was performed on a Shimadzu LC-20AD system equipped with two binary pumps (Kyoto, Japan), and a SIL-HTA (Shimadzu, Japan). Commonly used organic mobile phases such as methanol/acetonitrile and modifiers such as formic acid/ammonium acetic were tested on three columns for the optimal chromatographic separation and sensitivity, as well as the peak shape. A synergy Hydro-RP (2.0 mm×50 mm, 3 μm) column (Phenomenex, Torrance, USA) was finally chosen. The mobile phases chosen for this study were 0.1% (v/v) formic acid in water designated as A, and 0.1% (v/v) formic acid in methanol designated as B. The flow rate was 0.5 mL/min. Gradient program was as follows: mobile phase B was held at 5% for 0.80 min; a linear gradient was run to 60% B in 1.30 min; solvent composition was held for 0.5 min; a second linear gradient was run to 80% B in 0.7 min; solvent composition was held for 1.4 min and solvent composition was returned to 5% B in 0.1 min for re-equilibration. The total run time was 5.0 min. Data were collected and processed using Analyst 1.1 data collection and integration software.
The validation of the LC–MS/MS method was based on the FDA guidance for bioanalytical method validation [14], [15]. A blank matrix was prepared from a blank incubation system following the same processing steps as for other incubation systems, and assessed for any significant interference in the chromatographic regions of the six metabolites and the IS. The six marker metabolites were pooled to prepare the calibrators (0.5–100 ng/mL for all six metabolites). Area ratio of metabolite to IS was plotted against metabolite concentration, then weighted (1/x2) linear least-square regression was used to generate calibration curves. The fitting equation was y=ax+b, where y represents analyte-to-IS peak ratio and x donates nominal concentration of analytes in the pooled calibrators. The goodness of the calibration curve fitting was checked by the back-calculated concentration of the standards and the determination coefficient (r2).
2.3. Microsomal incubation
The incubations were conducted in three experimental sessions: (1) incubation conditions in terms of incubation time and microsomal protein concentration were tested and optimized in the first session, (2) DMSO effect on CYP activity was then investigated, and (3) the CYP inhibition experiment was performed finally. Each incubation system (200 μL) contained phosphate buffer (100 mM, pH 7.4), MgCl2 (3 mM), NADPH (1 mM), human liver microsomes, probe substrate(s), and a (or no) selective inhibitor. The incubation concentrations of the probe substrates (in individual or cocktail manner) were set around their reported Km values, i.e., 40, 2.0, 5.0, 40, 5.0 and 2.0 μM for phenacetin, amodiaquine, diclofenac, S-mephenytoin, dextromethorphan and midazolam, respectively. The reactions were initiated by the final addition of NADPH to the incubation systems, and then conducted at 37 °C. Following the incubations, the reactions were terminated by adding 400 μL ice-cold acetonitrile spiked with verapamil as the IS. The samples were allowed for protein precipitation on ice for 15 min, then vortexed shortly, and centrifuged at 3000g for 10 min. Supernatants were transferred into 96-well plates for LC–MS/MS analysis.
2.3.1. Incubation condition optimization
Incubation conditions were optimized by incubations of the six probe substrates in cocktail manner in human liver microsomes at 0.05, 0.1 and 0.2 mg microsomal proteins/mL for different times (5, 10 and 20 min). The remaining substrates and formed metabolites in the processed samples were analyzed and conditions were chosen according to the following criteria: (1) metabolite formation was linear with time and enzyme concentration, (2) substrate consumption was no more than 20% of the initial amount, and (3) all the metabolites formed could be easily detected by LC–MS/MS methods. The microsomal concentration and the incubation time were optimized as 0.1 mg microsomal proteins/mL and 10 min, respectively.
2.3.2. Effect of DMSO on CYP activity
To test the effect of DMSO on CYP activity, the individual-probe incubations with different DMSO concentration levels of 0%, 0.1%, 0.5% and 1% (v/v) were conducted under the optimized conditions. After the incubations, the formed metabolites were analyzed and the loss of activity was calculated as the percent decrease of metabolite formation.
2.3.3. CYP inhibition assay
The inhibitory effects of selective inhibitors on CYP activities were investigated in both individual-probe and cocktail-probe incubation systems. The probe substrates were co-incubated with each of the selective inhibitors with a series of concentration levels under the optimized conditions. The inhibitor concentrations were set sufficiently covering the reported IC50 values, i.e., 0.01, 0.03, 0.10, 0.30, 1.0, and 3.0 µM for α-naphthoflavone, quinidine and ketoconazole; 1.0, 3.0, 10, 30, 100, and 300 µM for quercetin; and 0.10, 0.30, 1.0, 3.0, 10, and 30 µM for sulfaphenazole and nootkatone. For the control incubation, simply no inhibitor was co-incubated with the probe substrates. The final DMSO concentration in the reaction systems was kept constant and not more than 0.5%. After the incubation, the formed metabolites in the incubates were analyzed.
2.4. Data analysis
The uninhibited fraction of the CYP activity (remaining activity fraction) was calculated as CI/C0, where CI and C0 donate the concentrations of generated metabolites after incubation in the presence and absence of the inhibitors, respectively. With the area ratios substituting the concentrations, the uninhibited fraction can be transformed as AI/A0, where AI and A0 represent the analyte-to-IS peak area ratios after incubation in the presence and absence of the inhibitors, respectively.
The IC50 values of the inhibitors were determined as appropriate by fitting a non-linear curve of uninhibited fraction versus incubation concentration of the inhibitors, using the following four-parameter model (Hill equation):
where Top, Bottom, S, x and y donate the maximum inhibited fraction, the minimum inhibited fraction, the slope factor, the inhibitor concentration, and the uninhibited fraction, respectively. The parameters Top and Bottom were limited between 0 and 1 by the software.
3. Results and discussion
3.1. Probe substrate combination
The primary purpose of this study was to develop and validate a cocktail method for efficient assessment of CYP-mediated inhibition potential. The probe substrates used for the cocktail assay included phenacetin (CYP1A2), amodiaquine (CYP2C8), diclofenac (CYP2C9), S-mephenytoin (CYP2C19), dextromethorphan (CYP2D6), and midazolam (CYP3A4). According to an investigation on the elimination pathways of top 200 most prescribed drugs in 2002, CYP1A2, CYP2C9, CYP2C19, CYP2D6 and CYP3A4 contributed over 95% of the overall metabolism [16]. FDA has recommended in vitro DDI studies targeting 6 major human CYP enzymes, i.e., CYP1A2, 2C8, 2C9, 2C19, 2D6 and 3A4, while PhRMA suggested 5 CYP enzymes, i.e., CYP1A2, 2C9, 2C19, 2D6 and 3A4, to be considered for DDI evaluation in its white paper. In addition, CYP2C8 is referred as a P450 enzyme of emerging importance [17]. In this regard, it would be a good idea to develop an in vitro cocktail method covering the 6 major human CYP enzymes for DDI screening. All the six probe substrates used in our cocktail assay are FDA-preferred for corresponding CYPs. To the best of our knowledge, the six-probe substrate cocktail was reported for the first time in this paper.
LC–MS/MS, with its high sensitivity and specificity, has become the routine analytical method for P450-involved DDI studies. A major purpose of this study was to develop an analytical method coping with the cocktail incubation protocol, aiming to keep the whole assay system in an highly efficient manner.
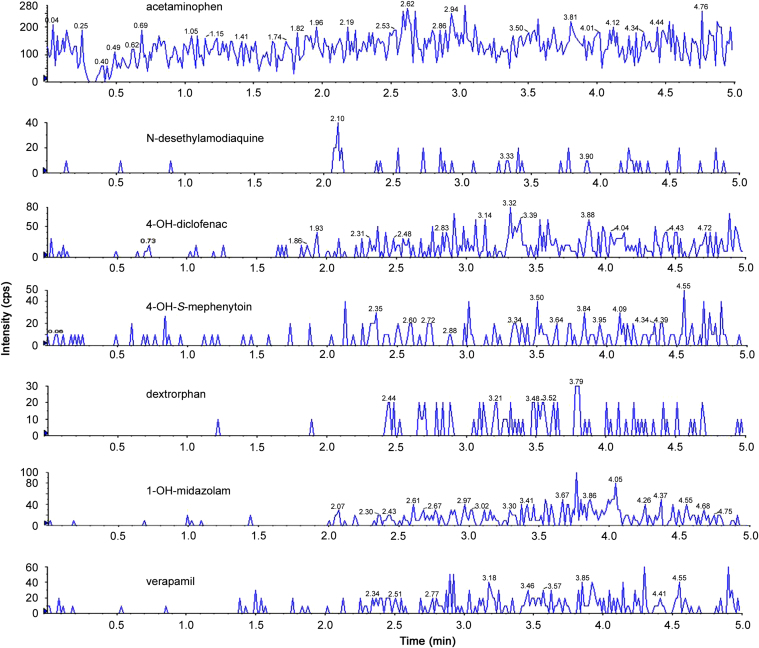
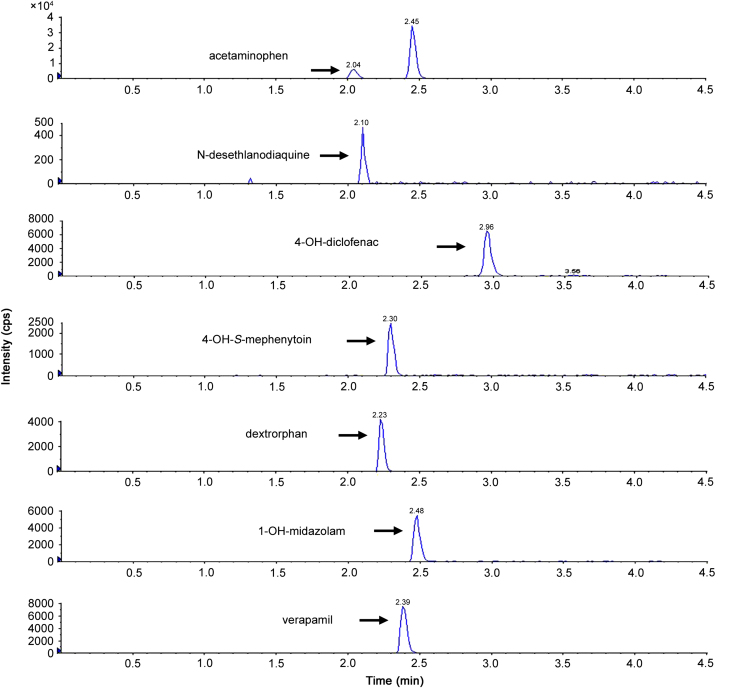
The single LC–MS/MS method for the simultaneous determination of multiple analytes was validated in terms of specificity, linearity and accuracy. Specificity was investigated with a blank matrix prepared from the substrate/inhibitor-free incubation system. As shown in Fig. 1, no significant interfering peaks were observed over the chromatographic regions of analytes. As shown in a representative chromatograph (Fig. 2), the retention times of acetaminophen, N-desethylamodiquine, 4-OH-diclofenac, 4-OH-S-mephenytoin, dextrorphan, 1-OH-midazolam and verapamil (IS) were 2.04, 2.11, 2.96, 2.30, 2.23, 2.48 and 2.39 min, respectively.
Fig. 1.
Chromatograms of metabolites and IS in blank matrix observed in detection channels of acetaminophen, N-desethylamodiquine, 4-hydroxydiclofenac, 4-hydroxymephenytoin, dextrophan, 1-hydroxymidazolam, and verapamil. No significant interfere peaks were observed at the same retention time in the chromatograms of marker metabolites.
Fig. 2.
Typical chromatographic profiling of acetaminophen, N-desethylamodiquine, 4-hydroxydiclofenac, 4-hydroxymephenytoin, dextrophan, 1-hydroxymidazolam, and verapamil; two significant peaks were observed in the channel of acetaminophen and the peak with earlier retention time was identified as acetaminophen (see Fig. 3).
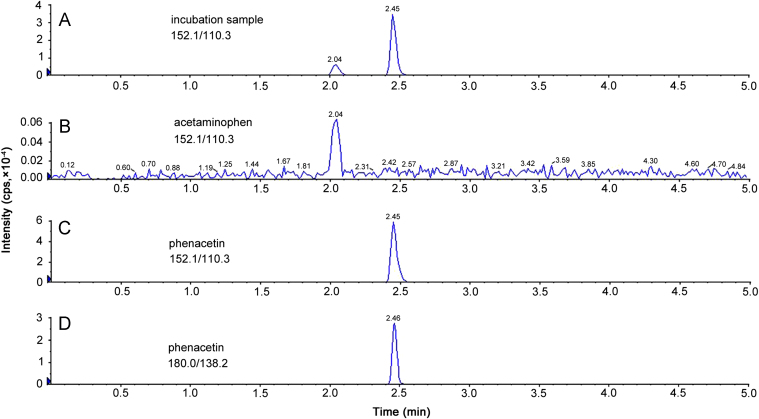
Two peaks with the retention times of 2.04 and 2.45 min were observed in the MRM channel of acetaminophen (152.1/110.3) using the incubation sample shown in Fig. 2. The earlier one was recognized as acetaminophen according to the chromatograph of standard (Fig. 3B). The retention time of the second one was identical with the phenacetin standard (Fig. 3C), indicating that the second peak might be generated from phenacetin. This hypothesis was verified by checking the chromatograph of phenacetin standard using the acetaminophen MRM channel (Fig. 3D). Actually, such interference had been reported previously and was presumed to be caused by substantial in-source fragmentation of phenacetin in the ESI source, yielding an ion structurally identical to acetaminophen [18], [19]. Since acetaminophen and phenacetin were well separated, the in-source fragmentation of phenacetin would not affect the detection of acetaminophen.
Fig. 3.
Chromatograms of acetaminophen and phenacetin detected in two detection channels. (A) Chromatogram of incubation sample observed in acetaminophen channel (152.1/110.3). Two peaks with the retention time of 2.04 min and 2.45 min were observed. (B) Chromatogram of acetaminophen standard observed in acetaminophen detection channel with the retention time 2.04 min, indicating that the peak with earlier retention time in A was the target. (C) Chromatogram of phenacetin standard observed in phenacetin channel. A peak was observed at the retention time of 2.45 min. (D) Chromatogram of phenacetin standard observed in acetaminophen transmit channel (180.0/138.2). A peak was observed at the retention time of 2.45 min, demonstrating the cross channel interference between phenacetin and acetaminophen.
Each calibration curve was constructed by linear fitting with 6 non-zero concentration points with quantification range of 2–100, 2–100, 1–100, 2–100, 1–100 and 1–100 ng/mL for acetaminophen, N-desethylamodiaquine, 4-OH-diclofenac, 4-OH-S-mephenytoin, dextrorphan, and 1-OH-midazolam, respectively. Good linearity was observed. The fitted equations were y=0.0389x−0.0106 (r=0.9943), y=0.0079x+0.00418 (r=0.9943), y=0.0517x+0.13 (r=0.9805), y=0.0197x−0.00411 (r=0.9855), y=0.0293x+0.00273 (r=0.9980) and y=0.0781x+0.069 (r=0.9977), for acetaminophen, N-desethylamodiaquine, 4-OH-diclofenac, 4-OH-S-mephenytoin, dextrorphan and 1-OH-midazolam, respectively. All the calibrators had their measured values within ±20%.
3.2. Optimization of incubation condition
Technically, a reliable in vitro CYP inhibition study should be conducted under a steady-state, linear condition, which means the metabolite formation should be linearly correlated with enzyme concentration and incubation time under the chosen incubation conditions. Meeting this requirement allows straightforward data interpretation. High microsomal protein concentration level may complicate the data interpretation because of the non-specific binding of inhibitors or substrates to the microsomal proteins. Microsomal protein concentration has been recommended to be no more than 1 mg/mL by FDA or no more than 0.5 mg/mL by PhRMA. Industrial scientists [20], [21], [22], [23] have suggested using a microsomal protein concentration as low as feasible for in vitro inhibition studies. Using lower microsomal protein concentration, however, longer incubation time might be needed to generate sufficient quantities of metabolites for quantification, thus compromising the assay robustness. In addition to the analytical requirement, an appropriate condition setting should also ensure that no significant depletion of substrates occurs during the incubation. PhRMA recommends the consumption of probe substrates no more than 20%, while FDA suggests that the probe substrate depletion is no more than 10–30%. For a probe substrate cocktail, incubation conditions (i.e., enzyme concentration and incubation time) may have to be chosen with some compromise regarding probe substrate M depletion, initial-rate reaction, and analytical requirements for all the probe substrates.
In our experiment, different microsomal protein concentration levels (0.05, 0.1, and 0.2 mg/mL) and incubation times (5, 10 and 20 min) were evaluated. Midazolam for CYP3A4 underwent rapid metabolism, while S-mephenytoin for CYP2C19 and phenecetin for CYP1A2 showed substantially low turnover rates. Formation of all the marker metabolites was linear with time (up to 20 min) at the protein concentration of 0.1 mg/mL, and linear with protein concentration (up to 0.2 mg/mL) with incubation time of 10 min for midazolam and 20 min for all other probe substrates. Based on these results, we decided to use 0.1 mg/mL of microsomal protein concentration and incubation time of 10 min for our cocktail assay. All other incubation conditions are described in Section 2.3.3.
3.3. Effect of DMSO on CYP activity
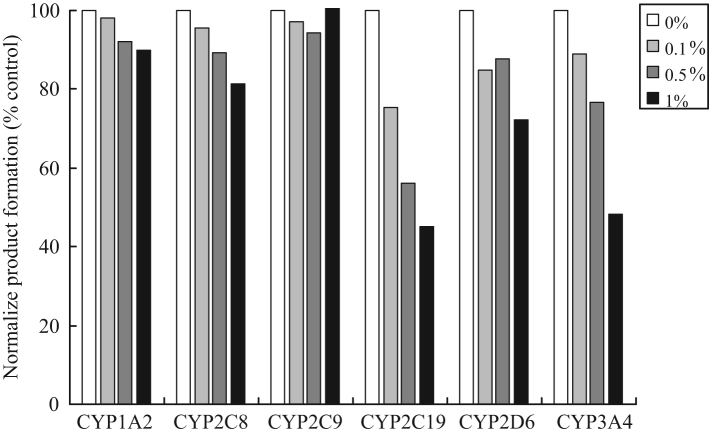
DMSO is routinely used to solubilize drug compounds due to its ability to solve chemicals in a broad range of physical properties. As shown in Fig. 4, CYP2C19 and 3A4 represent the most sensitive isoforms to DMSO with activity loss of 55.0% and 52.7% at 1% DMSO, and 44.8% and 24.4% at 0.5%. CYP1A2, 2C8 and 2C9 were not significantly affected by DMSO up to 1%. CYP2D6 was a mildly affected isoform compared to others, with activity loss of 27.8% at 1% DMSO. Since 0.1% DMSO caused no significant reduction of all the metabolite formation, DMSO concentration of no more than 0.1% in incubates would be preferred for our cocktail assay. The effect of DMSO on CYP activity has also been previously reported in several papers. For example, CYP1A2 and 2C9 were the two CYPs least affected by DMSO [24], while CYP2C19 and CYP3A4 were the two CYPs most sensitive to DMSO [25], [26]. Our results were consistent with the previous reports.
Fig. 4.
DMSO effect with different concentrations on the activities of CYPs in 6-substrate assay.
In a cocktail DDI assay, sometimes the use of DMSO may cause a problem because the pooling of several probe substrates may have to introduce more DMSO into the incubation system. This concern may represent a limitation of the cocktail approach, and sometimes it may be very important to make the effort to lower DMSO concentration. The DMSO concentration in our cocktail assay was set to be 0.5% at which the CYP activity was reduced by 8%, 11%, 6%, 44%, 12% and 23% for CYP1A2, 2C8, 2C9, 2C19, 2D6 and 3A4, respectively.
3.4. IC50 determination and comparison
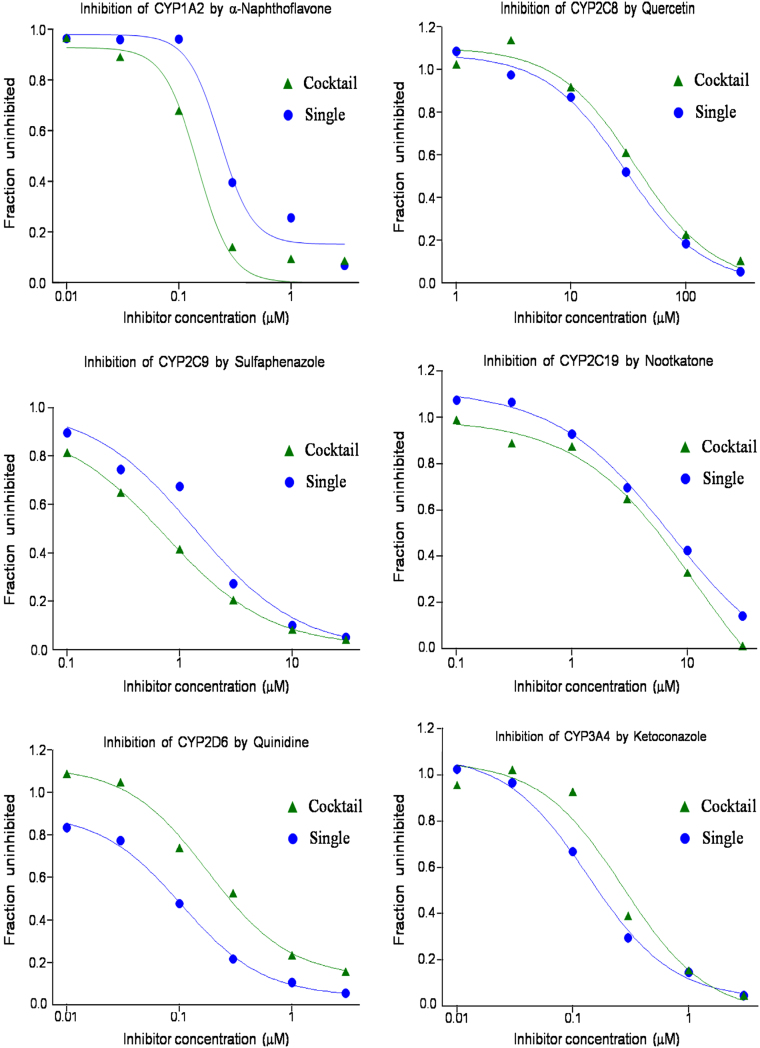
IC50 values of α-naphthoflavone, quercetin, sulfaphenazole, nootkatone, quinidine and ketoconazole were measured to be 0.149 vs. 0.244 µM, 33.5 vs. 27.6 µM, 0.719 vs. 1.16 µM, 6.48 vs. 6.18 µM, 0.182 vs. 0.129 µM and 0.236 vs. 0.138 µM, respectively, using the cocktail-probe vs. single-probe approach. The IC50 values determined by the cocktail assay were reasonably consistent with those reported in the literature which could be found with well-established ranges and used as solid references. α-Naphthoflavone (reported IC50: 0.04–0.2 µM [27]), quinidine (reported IC50: 0.02–0.22 µM [28]) and ketoconazole (reported IC50: 0.09–0.24 [27]) are potent inhibitors to CYP1A2, 2D6 and 3A4, respectively, and quercetin (reported IC50: 3.1–8.47 [27]), sulfaphenazole (reported IC50: 0.5–1.5 [28]) and nootkatone (reported Ki of 0.5 and IC50 of 9.56 [29], [30]) are potent-to-mild inhibitors to CYP2C8, CYP2C9 and CYP2C19, respectively. More importantly, the IC50 values obtained by cocktail-probe and single-probe approaches were in good agreement. As shown in Table 2, the difference in the IC50 values was within 2 folds. These results strongly suggest that the cocktail assay can be reasonably used as a highly efficient screening protocol for the assessment of compound-mediated CYP inhibition potential (Fig. 5).
Table 2.
Summary of IC50 values of inhibitors for six human cytochrome P450 enzymes in pooled human liver microsomes.
| Isoform | Inhibitor | IC50 calculated with SC (μM) |
IC50 calculated without SC (μM) |
||
|---|---|---|---|---|---|
| Single | Cocktail | Single | Cocktail | ||
| CYP1A2 | α-Naphthoflavone | 0.244 | 0.149 | 0.232 | 0.147 |
| CYP2C8 | Quercetin | 27.6 | 33.5 | 29.1 | 36.2 |
| CYP2C9 | Sulfaphenazole | 1.16 | 0.719 | 1.35 | 0.742 |
| CYP2C19 | Nootkatone | 6.18 | 6.48 | 7.19 | 6.85 |
| CYP2D6 | Qunidine | 0.129 | 0.182 | 0.104 | 0.180 |
| CYP3A4 | Ketoconazole | 0.138 | 0.236 | 0.138 | 0.273 |
Fig. 5.
Semi-log graphs obtained using individual or cocktail CYP probe substrates.
From an analytical perspective, good linearity and accuracy observed from the standards represent the essential part of the goodness of an analytical method. Therefore, utilization of the calibration curve constructed by the calibrators at different concentration levels is a routine way for quantification. Normally, IC50 is calculated by measuring concentrations of marker metabolite(s) using calibration curves. However, as an effort to further improve the analytical throughput with little influence on the final results, we also investigated the shifts of IC50 values after taking off the calibration curves from the analytical regimen. As described in Section 2.4, IC50 was calculated by substituting the concentration with analyte-to-IS peak area ratios. As shown in Table 2, no remarkable difference (within 1.2-fold) was made by doing so, which meant we could further cut the workload in the bioanalytical process with little compromise in data quality.
The higher-throughput assay system for simultaneous evaluation of six major CYP enzymes validated in this study can be used for screening purpose based on IC50 measurement, or at a rougher level (for example, putative inhibitor is set at only two concentration levels and IC50 is not the end-point). However, caution should always be taken when results obtained from the cocktail method are used to make a decisive conclusion. The value of the cocktail methods lies in their cost/time-effectiveness with somewhat loss of reality. A sound strategy might be a combination of a screening step using cocktail incubation method and a sequential step using individual probe substrates where possible inhibitory DDI would be evaluated more accurately. This strategy has been utilized in our laboratory as a standard procedure for assessing compound-mediated inhibition potential and/or potency.
4. Conclusion
In this work, we developed a method for higher-throughput screening of compound-mediated inhibition of six major human CYP enzymes (CYP1A2, 2C8, 2C9, 2C19, 2D6 and 3A4) by employing six specific substrates and LC–MS/MS. The in vitro enzymatic reaction system was well optimized, including enzyme concentration, reaction time as well as the effect of DMSO. The amount of marker metabolites was qualified by a precise, sensitive and accurate LC–MS/MS method. The IC50 values of selective CYP inhibitors determined using the substrate cocktail were in good agreement with those obtained with individual substrates and with previously reported values in the literature, strongly suggesting that the cocktail approach can be reasonably used as a higher-throughput screening tool for the assessment of compound-mediated CYP inhibition potential. Besides, no remarkable difference was observed between the IC50 obtained by standard quantification with calibration curves and a simplified analytical approach with only analyte-to-IS peak area ratios, indicating that we may use the simplified analytical approach in our cocktail assay to further improve the throughput.
Footnotes
Peer review under responsibility of Xi׳an Jiaotong University.
References
- 1.Clarke S.E., Jones B.C. Human cytochrome P450 and their role in metabolism-based drug–drug interactions. In: Rodrigues A.D., editor. Drug and Drug Interactions. second ed. Informa healthcare Inc.; New York: 2008. pp. 53–86. [Google Scholar]
- 2.Lin J.H., Lu A.Y. Inhibition and induction of cytochrome P450 and the clinical implications. Clin. Pharmacokinet. 1998;35:361–390. doi: 10.2165/00003088-199835050-00003. [DOI] [PubMed] [Google Scholar]
- 3.Asha S., Vidyavathi M. Role of human liver microsomes in in vitro metabolism of drugs. Appl. Biochem. Biotechnol. 2010;160:1699–1722. doi: 10.1007/s12010-009-8689-6. [DOI] [PubMed] [Google Scholar]
- 4.Rutherford M.L., Stinger T. Recent trends in laboratory automation in the pharmaceutical industry. Curr. Opin. Drug Discov. Dev. 2001;4:343–346. [PubMed] [Google Scholar]
- 5.Smith D., Sadagopan N., Zientek M. Analytical approaches to determine cytochrome P450 inhibitory potential of new chemical entities in drug discovery. J. Chromatogr. B. 2007;850:455–463. doi: 10.1016/j.jchromb.2006.12.024. [DOI] [PubMed] [Google Scholar]
- 6.Schellens J.H., Van der Wart J.H., Brugman M. Influence of enzyme induction and inhibition on the oxidation of nifedipine, sparteine, mephenytoin and antipyrine in humans as assessed by a cocktail study design. J. Pharmacol. Exp. Ther. 1989;249:638–645. [PubMed] [Google Scholar]
- 7.Breimer D.D., Schellens J.H. A cocktail strategy to assess in vivo oxidative drug metaoblism in humans. Trends Pharmacol. Sci. 1990;11:223–225. doi: 10.1016/0165-6147(90)90245-4. [DOI] [PubMed] [Google Scholar]
- 8.O’Donnell C.J., Grime K., Courtney P. The development of a cocktail CYP2B6, CYP2C8, and CYP3A5 inhibition assay and a preliminary assessment of utility in a drug discovery setting. Drug Metab. Dispos. 2007;35:381–385. doi: 10.1124/dmd.106.012344. [DOI] [PubMed] [Google Scholar]
- 9.Alden P.G., Plumb R.S., Jones M.D. A rapid ultra-performance liquid chromatography/tandem mass spectrometric methodology for the in vitro analysis of pooled and cocktail cytochrome P450 assays. Rapid Commun. Mass Spectrom. 2010;24:147–154. doi: 10.1002/rcm.4364. [DOI] [PubMed] [Google Scholar]
- 10.Dixit V., Hariparsad N., Desai P. In vitro LC–MS cocktail assays to simultaneously determine human cytochrome P450 activities. Biopharm. Drug Dispos. 2007;28:257–262. doi: 10.1002/bdd.552. [DOI] [PubMed] [Google Scholar]
- 11.Yao M., Zhu M., Sinz M.W. Development and full validation of six inhibition assays for five major cytochrome P450 enzymes in human liver microsomes using an automated 96-well microplate incubation format and LC–MS/MS analysis. J. Pharm. Biomed. Anal. 2007;44:211–223. doi: 10.1016/j.jpba.2007.02.034. [DOI] [PubMed] [Google Scholar]
- 12.Zhang S., Song N., Li Q. Liquid chromatography/tandem mass spectrometry method for simultaneous evaluation of activities of five cytochrome P450s using a five-drug cocktail and application to cytochrome P450 phenotyping studies in rats. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2008;871:78–89. doi: 10.1016/j.jchromb.2008.06.034. [DOI] [PubMed] [Google Scholar]
- 13.Dierks E.A., Stams K.R., Lim H.K. A method for the simultaneous evaluation of the activities of seven major human drug-metabolizing cytochrome P450s using an in vitro cocktail of probe substrates and fast gradient liquid chromatography tandem mass spectrometry. Drug Metab. Dispos. 2001;29:23–29. [PubMed] [Google Scholar]
- 14.Yuan R., Madani S., Wei X.X. Evaluation of cytochrome P450 probe substrates commonly used by the pharmaceutical industry to study in vitro drug interactions. Drug Metab. Dispos. 2002;30:1311–1319. doi: 10.1124/dmd.30.12.1311. [DOI] [PubMed] [Google Scholar]
- 15.U.S. Food and Drug Administration, Guidance for Industry Bioanalytical Method Validation. Available at: 〈http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm368107.pdf〉.
- 16.Williams J.A., Hyland R., Jones B.C. Drug–drug interactions for UDP-glucuronosyltransferase substrates: a pharmacokinetic explanation for typically observed low exposure (AUCi/AUC) ratios. Drug Metab. Dispos. 2004;32:1201–1208. doi: 10.1124/dmd.104.000794. [DOI] [PubMed] [Google Scholar]
- 17.Bjornsson T.D., Callaghan J.T., Einolf H.J. The conduct of in vitro and in vivo drug–drug interaction studies: a pharmaceutical research and manufacturers of America (PhRMA) perspective. Drug Metab. Dispos. 2003;31:815–832. doi: 10.1124/dmd.31.7.815. [DOI] [PubMed] [Google Scholar]
- 18.Lim K.B., Ozbal C.C., Kassel D.B. Development of a high-throughput online-phase extraction/tandem mass spectrometry method for cytochrome P450 inhibition screening. J. Biomol. Screen. 2010;15:447–452. doi: 10.1177/1087057110362581. [DOI] [PubMed] [Google Scholar]
- 19.Weaver R., Graham K.S., Beattle I.G. Cytochrome P450 inhibition using recombinant proteins and mass spectrometry/multiple reaction monitoring technology in a cassette incubation. Drug Metab. Dispos. 2003;31:955–966. doi: 10.1124/dmd.31.7.955. [DOI] [PubMed] [Google Scholar]
- 20.Obach R.S. Nonspecific binding to microsomes: impact on scale-up of in vitro intrinsic clearance to hepatic clearance as assessed through examination of warfarin, imipramine, and propranolol. Drug Metab. Dispos. 1997;25:1359–1369. [PubMed] [Google Scholar]
- 21.Obach R.S., Baxter J.G., Liston T.E. The prediction of human pharmacokinetic parameters from preclinical and in vitro metabolism data. J. Pharmacol. Exp. Ther. 1997;283:46–58. [PubMed] [Google Scholar]
- 22.Obach R.S. The importance of nonspecific binding in in vitro matrices, its impact on enzyme kinetic studies of drug metabolism reactions, and implications for in vitro-in vivo correlations. Drug Metab. Dispos. 1996;24:1047–1049. [PubMed] [Google Scholar]
- 23.Tran T.H., Von Moltke L.L., Venkatakrishnan K. Microsomal protein concentration modifies the apparent inhibitory potency of CYP3A4 inhibitors. Drug Metab. Dispos. 2002;30:1441–1445. doi: 10.1124/dmd.30.12.1441. [DOI] [PubMed] [Google Scholar]
- 24.Busby W.F., Jr., Ackermann J.M., Crespi C.L. Effect of methanol, ethanol, dimethyl sulfoxide, and acetonitrile on in vitro activities of cDNA-expressed human cytochromes P-450. Drug Metab. Dispos. 1999;27:246–249. [PubMed] [Google Scholar]
- 25.Chauret N., Gauthier A., Nicoll-Griffith D.A. Effect of common organic solvents on in vitro cytochrome P450-mediated metabolic activities in human liver microsomes. Drug Metab. Dispos. 1998;26:1–4. [PubMed] [Google Scholar]
- 26.Easterbrook J., Lu C., Sakai Y. Effects of organic solvents on the activities of cytochrome P450 isoforms, UDP-dependent glucuronyl transferase, and phenol sulfotransferase in human hepatocytes. Drug Metab. Dispos. 2001;29:141–144. [PubMed] [Google Scholar]
- 27.Kim M.J., Kim H., Cha I.J., Park J.S. High-throughput screening of inhibitory potential of nine cytochrome P450 enzymes in vitro using liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2005;19:2651–2658. doi: 10.1002/rcm.2110. [DOI] [PubMed] [Google Scholar]
- 28.Testino S.A., Jr, Patonay G. High-throughput inhibition screening of major human cytochrome P450 enzymes using an in vitro cocktail and liquid chromatography-tandem mass spectrometry. J. Pharm. Biomed. Anal. 2003;30:1459–1467. doi: 10.1016/s0731-7085(02)00480-6. [DOI] [PubMed] [Google Scholar]
- 29.U.S. Food and Drug Administration, Guidance for Industry Drug Interaction Studies Study Design Data Analysis and Implications for Dosing and Lableling. Available at: 〈http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm072101.pdf〉.
- 30.J.Y. Lee, T.A. Moeller, T. Emig, et al., Unique Classed of Celsis InVitroCYPTM M-class and H-class Pooled Human Microsomes Used for in vitro ADME Studies, Bioreclamation IVT. Available at: 〈http://www.bioreclamationivt.com/images/pdfs/ps_invitrocypmclass.pdf〉.