Abstract
Glucocorticoids are adrenally produced hormones critically involved in development, general physiology, and control of inflammation. Since their discovery, glucocorticoids have been widely used to treat a variety of inflammatory conditions. However, high doses or prolonged use leads to a number of side effects throughout the body, which preclude their clinical utility. The primary actions of glucocorticoids are mediated by the glucocorticoid receptor (GR), a transcription factor that regulates many complex signaling pathways. Although GR is nearly ubiquitous throughout the body, glucocorticoids exhibit cell- and tissue-specific effects. For example, glucocorticoids stimulate glucose production in the liver, reduce glucose uptake in the skeletal muscle, and decrease insulin secretion from the pancreatic β-cells. Mouse models represent an important approach to understanding the dynamic functions of GR signaling in normal physiology, disease, and resistance. In the absence of a viable GR null model, gene-targeting techniques utilizing promoter-driven recombination have provided an opportunity to characterize the tissue-specific actions of GR. The aim of the present review is to describe the organ systems in which GR has been conditionally deleted and summarize the functions ascribed to glucocorticoid action in those tissues.
Tissue-specific knockout models have provided insight into the functions of the glucocorticoid receptor. This review examines the reported models, highlighting discoveries in each organ system.
Glucocorticoids are steroid hormones released from the adrenal glands in response to activation of the hypothalamic–pituitary–adrenal (HPA) axis. Glucocorticoid release occurs in a diurnal pattern but is also induced acutely in response to a variety of physiological or psychological stresses or inflammatory stimuli. These hormones function to critically regulate physiological homeostasis through actions in nearly every organ and tissue in the body. Glucocorticoids were originally named for their ability to promote gluconeogenesis in the liver, but they also function to coordinate the inflammatory and immune response, metabolic homeostasis, cognitive function, reproduction, and development. Their potent anti-inflammatory and immunosuppressive actions are the basis for their pervasive use clinically. However, systemic therapeutic uses of these hormones and synthetic derivatives escape negative feedback mechanisms that regulate the duration and extent of glucocorticoid release, triggering side effects in many organ systems. Therefore, an understanding of selective functions of glucocorticoids within distinct cell types in various organ systems could be of enormous clinical benefit. Such insight may enhance the safety and efficacy of treatment regimens for currently used therapeutic glucocorticoids or lead to the development of novel more selective glucocorticoid derivatives.
The actions of glucocorticoids are mediated by the glucocorticoid receptor (GR), the first identified member of the nuclear receptor superfamily of transcription factors (1). Members of this superfamily share common functional domains, including an N-terminal transactivation domain, a central DNA-binding domain, and a C-terminal ligand-binding domain (2). The N-terminal transactivation domain is the least conserved domain and contains regulatory regions that allow binding to diverse coregulators and components of the transcriptional machinery through activation function domain 1 (AF-1). The most conserved region within the nuclear receptor superfamily is the DNA-binding domain, containing two zinc finger motifs that mediate dimerization and dictate binding to specific genomic sequences. Transactivation by genomic GR binding has been well characterized at sequences termed the glucocorticoid response element (GRE), a palindrome composed of two hexameric repeats separated by a three-nucleotide spacer (3). Glucocorticoid-bound GR can also repress transcription through association with negative GREs, which facilitates recruitment of corepressors and histone deacetlyases (4). GR can also interact with inverted-repeat GREs and sequences that contain only a single half-site of a palindromic consensus GRE (5). Finally, the well-conserved C-terminal ligand-binding domain forms a high-affinity hormone-binding pocket that dictates ligand selectivity, a second dimerization domain, and a second activation function domain (AF-2) that supports the transcriptional regulatory activities of GR through associations with coactivators (6).
In the absence of ligand, GR is predominantly located in the cytoplasm in a large multiprotein complex that includes heat shock proteins (hsp90, hsp70, and hsp56), immunophilins, and p23 (7, 8). GR can shuttle between the cytoplasm and nucleus, but ligand binding shifts the balance toward nuclear accumulation. This occurs in part through ligand-induced conformational changes that expose nuclear localization sequences and alter the association of GR-interacting proteins that influence its subcellular trafficking (9, 10). Within the nucleus, GR–DNA binding depends on several factors, including the chromatin environment, cofactor availability, and genomic sequence. Therefore, several mechanisms for transcriptional regulation by GR have been described (5). The classical model of GR transactivation involves GR dimerization and binding at GREs leading to coactivator recruitment and activation of transcription from proximate promoters (11). However, alternative modes of GR transactivation and transrepression have been defined based on recent results from genome-wide analysis of GR gene occupancy, chromatin structure, superresolution microscopy, and computational analysis (12–14). For example, monomeric GR can bind DNA in association with other transcription factors that are located proximal to the direct GR binding site creating what is termed a “composite GRE.” GR can also localize to genomic sites through protein–protein interactions with other transcription factors without directly binding DNA in a tethering mechanism. Transrepression can result from GR binding to composite GREs or unique negative GREs, tethering to or sequestering other transcription factors, or utilizing the dominant-negative activity of the β isoform of GR (GRβ) (15). Finally, cell type–specific GR transactivation and transrepression can use sequences in close proximity to a gene promoter or use lineage-specific enhancer regulatory sequences, which may be located several kilobases to megabases away from their target promoters. The functional diversity of glucocorticoid signaling is further extended by posttranslational modifications of GR, which can regulate GR nuclear translocation, determine protein interaction surfaces, and dictate the transcriptional response to GR binding (16, 17).
The biology of glucocorticoid signaling is influenced by physiological and pathophysiological contexts, and thus allows GR the capacity to regulate various biological functions depending on physiological status of the organism. This phenomenon allows glucocorticoids to induce apoptosis in several cell types, including thymocytes, bone osteoblasts and osteocytes, and cells of the hematopoietic system, but promote the expression of prosurvival factors in cancer cells (18–20). Within the immune system, glucocorticoids can exert both anti-inflammatory and proinflammatory effects depending on the stimulus (21). The physiological response to glucocorticoids is also determined by independent endocrine signals. For example, in the reproductive system, glucocorticoids can modulate female fertility through altering hypothalamic–pituitary activity, as well as via direct actions in the uterus (22). Although the diversity signaling mechanisms allow glucocorticoids to regulate a myriad of physiological processes, the pleiotropic actions of glucocorticoids have made it difficult to define the signal transduction pathways responsible for these processes within individual cells or organ systems.
Transgenic Mice Identify Tissue-Specific Functions of GR
Gene targeting technologies have provided a powerful tool to study the physiological functions of GR; however, the utility of conventional GR knockout mice has been restricted due to neonatal mortality in null mice. Born at the expected Mendelian frequency, GR-null pups die within hours due to severe respiratory distress caused by impaired lung development (23). Whereas the global GR-null mice have been used to study the function of GR during embryonic development and for donor tissue in transplant experiments, their use for postnatal studies has been limited (24). Early studies to assess the tissue-specific function of GR in vivo relied on promoter-driven transgenes encoding an antisense GR sequence (25, 26). The first tissue-restricted transgenic mouse used the human neurofilament gene promoter and reported transgene expression in the cortex, hypothalamus, heart, kidney, and adrenal (25). Disruption of the HPA axis by this antisense GR transgene resulted in mice that were obese, with increased fat deposits and elevated corticosterone and adrenocorticotropic hormone levels. However, expression of the transgene was not restricted to the brain as expected, and in general, the nucleotide antisense sequence was subjected to nuclease degradation and potential activation of the immune system unrelated to its GR-specific activities (27). The arrival of Cre/lox technology allowed promoter-driven DNA excision with high fidelity across generations (Fig. 1). As a result, several mouse models have been developed to study the tissue- or cell-specific roles of GR (Fig. 2). These models have used several different floxed constructs, flanking different exons of the GR gene (Nr3c1) (Fig. 3). For example, lymphocyte protein tyrosine kinase (Lck) Cre has been used to remove GR from T cells with three different floxed models, two in which exon 3 was floxed and one where exon 2 was floxed (28, 29, 32). Some of these mouse models, which use the same Cre transgenic mouse, present with differing phenotypes depending on which exon was excised (33). Likewise, the same organ has been targeted for GR deletion through the use of alternative Cre lines, which results in various physiological outcomes depending on the timing of Cre excision during development or the cell type targeted within the tissue. Importantly, both the floxed and Cre model should be considered when interpreting the phenotype of the tissue-specific GR knockout mice.
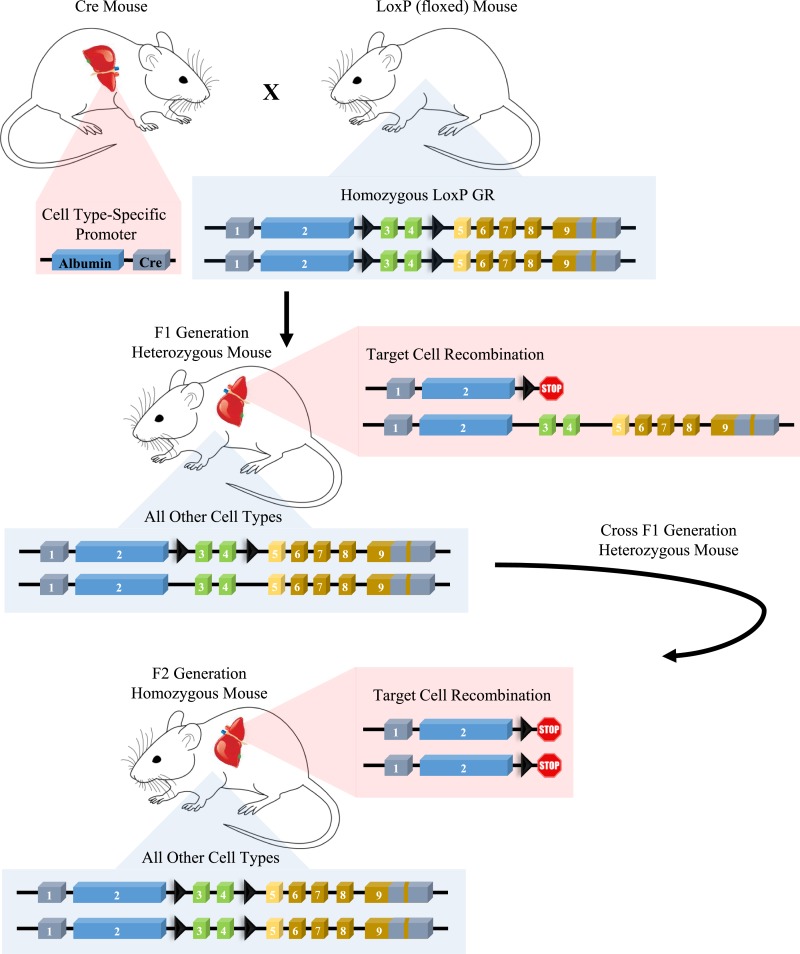
Figure 1.
The principles of Cre-LoxP technology. Creating animal models of cell- or tissue-specific gene deletion requires an animal expressing Cre recombinase under the control of a gene promoter (transgenic or endogenous). This animal line must be crossed to an animal model with an allele containing loxP sites (“floxed”). The offspring (F1 generation) are heterozygous for the deletion and must be crossed with other heterozygous animals to achieve full deletion of the target gene in the target cell or tissue.
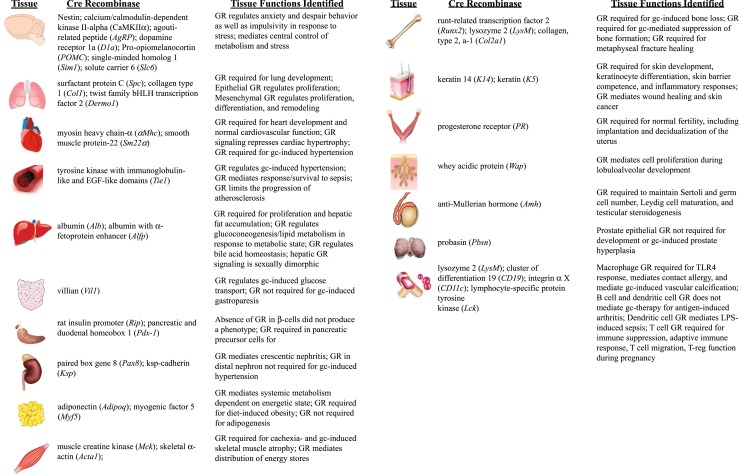
Figure 2.
Tissues targeted for GR deletion. Tissue-specific GR knockouts have been developed in several tissues throughout the body. Tissues or organs in which GR expression has been targeted are listed, along with the Cre recombinase used to achieve the deletion and a brief summary of the reported findings. EGF, epidermal growth factor; gc, glucocorticoid; T-reg, regulatory T-cell.
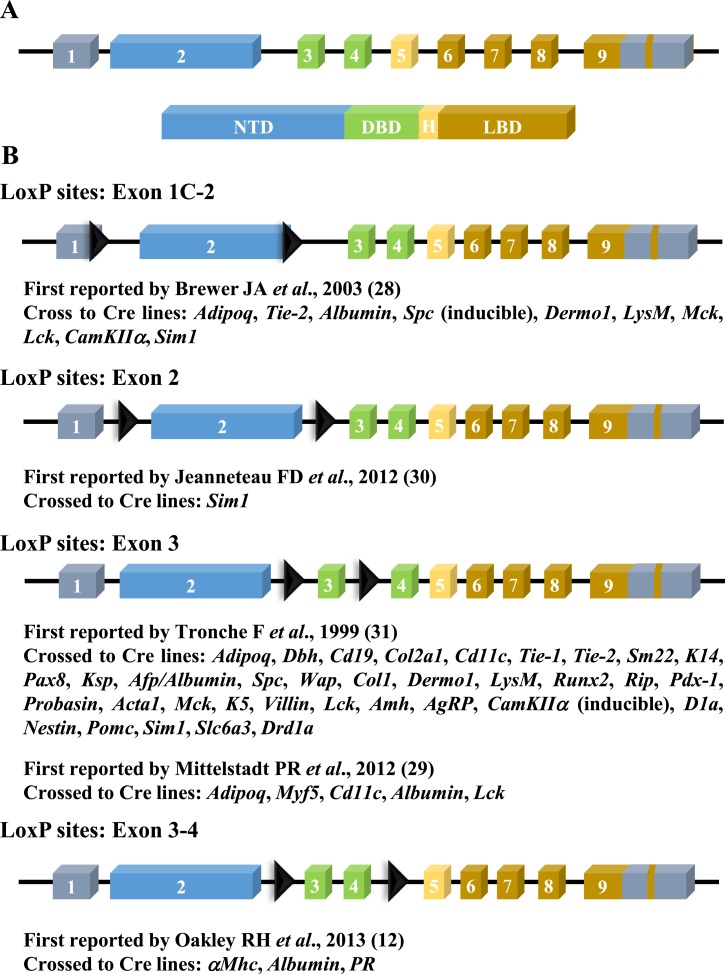
Figure 3.
Organization of the human GR gene and reported floxed contructs. (A) The human GR genes (Nr3c1) are composed of nine exons on chromosome 5 (5q31) that encode functionally unique domains. These domains can be divided into the DNA-binding domain (DBD), hinge region (H), ligand-binding domain (LBD), and N-terminal domain (NTD). Located within these domains are regions that regulate transcription, nuclear trafficking, and protein–protein interactions. (B) Multiple floxed mice have been developed with loxP sites flanking different exons of Nr3c1. The location of the loxP sites in the different models has been depicted, and the reference to the first reported use of each floxed mouse model is listed.
Central Nervous System
Many studies have exploited Cre/lox technology to investigate the role of GR within specific neuronal populations in regulating stress responses. The first “brain-specific” knockout of GR was performed using floxed exon 3 of GR deleted essentially in all cells of the CNS via a Cre recombinase driven from the rat Nestin promoter and enhancer (31). These mice exhibited increased basal corticosterone levels as expected from loss of central feedback inhibition and showed reduced anxiety as assessed by a forced swim test, dark–light crossing task, and elevated zero maze (31). A follow-up analysis of the brain GR knockout revealed complex effects on energy metabolism that were characterized by reduced lean body mass following suckling and an initial increase in body fat content until weaning, after which this trend was reversed (34). The systemic increase in corticosterone and elevations of corticotropin-releasing hormone (CRH) within parvocellular neurons of the paraventricular nucleus (PVN) of the hypothalamus likely contributed to the metabolic alterations in the brain-specific knockout of GR. Other metabolic hormones (e.g., insulin, leptin) were also dysregulated and likely to directly or indirectly influence the alterations in energy metabolism in these mice.
The next phase of brain-specific GR knockouts used region-specific Cre drivers allowing the assessment of more selective actions of glucocorticoids in the brain. For example, a number of studies used a calcium/calmodulin-dependent kinase II-α (CaMKIIα) promoter driving Cre expression to selectively knockout GR in the forebrain (35–37). This promoter is activated at approximately 3 weeks of age in mouse neonates and led to selective deletion of floxed GR in the hippocampus and cortex with intact GR in the central nucleus of the amygdala, pituitary, and the PVN of the hypothalamus (35). Male mice deleted of GR within the forebrain demonstrated altered behavioral and hormonal responses to stress with an increase in despair activity and increased impulsivity in response to stressful stimuli (36). Even though hypothalamic GR, particularly in the PVN, was intact in forebrain-specific GR knockouts, compensatory changes in the hypothalamic expression of selected neuropeptides (e.g., arginine/vasopressin) or neuropeptide receptors (i.e., CRH receptor-1) are likely to contribute to the differences in behavioral responses to stress in mice devoid of forebrain GR (36). Interestingly, the depressive-like phenotype observed in forebrain-specific GR knockouts in male mice was not observed in females (38).
When a much larger segment of the CaMKIIα promoter, which included 43-kb upstream and 100-kb downstream sequences, was used to drive Cre expression, exon 3 of GR was knocked out in the embryonic pituitary, resulting in early postnatal lethality and severe disruption of the HPA axis that generated system-wide histopathologies in many organs (38). A more selective pituitary-specific knockout of GR was attained using a pro-opiomelanocortin (Pomc) promoter driving Cre expression, which did not result in premature postnatal lethality (40). Mice lacking GR expression in the pituitary develop hypercorticolism owing to impaired negative feedback and have enhanced stress coping behaviors with no detectable cognitive deficits (40).
Because the initial forebrain-specific GR knockouts were on mixed genetic backgrounds (i.e., C57BL/6 × 129 × CBA), more recent studies have examined various depressive-like behaviors in a CaMKIIα Cre–driven knockout in a pure C57BL/6 background (41). Although baseline depressivelike behavior was not observed in the forebrain-specific GR knockout in a C57BL/6 background, antidepressants maintained their effect in forebrain-specific GR knockouts in mixed and pure backgrounds (40). Forebrain-specific GR knockout mice in a pure background also uniquely exhibited increased mineralocorticoid receptor expression in the hippocampus, which could also contribute to differences in depressive-like phenotypes observed in different strain backgrounds (41).
The single-minded homolog 1 (Sim1) gene promoter driving Cre expression was used by different groups to knockout GR in selected nuclei of the hypothalamus, particularly the PVN (33, 42). The Sim1 promoter activity is active during the initiation of hypothalamic neurogenesis in mice (approximately embryonic day 10.5), contributing to the nearly 90% reduction in GR expression in the PVN of GR mice with exon 3 floxed GR, including Sim1-driven Cre expression (33). Deletion of floxed exon 1C and exon 2 in these mice does not generate a robust reduction in GR expression in the PVN and leads to only modest metabolic phenotypes (31). Moreover, in a separate GR exon 1C and exon 2 deletion in the PVN generated by a Sim1 promoter driving Cre, GR expressed was reduced by only 60% and led to sex-specific differences in HPA axis function with females but not males exhibiting an increase in corticosterone levels at the nadir of the circadian cycle and males but not females exhibiting an elevated corticosterone response to acute stress (42). However, exon 3–deleted GR in the hypothalamus of male and female mice has an altered HPA axis characterized by increased corticosterone levels at the nadir and peak of the circadian cycle and elevations in response to acute stress (33). The hypercorticosterolemia in these mice could derive from alterations in negative feedback, as CRH expression in the PVN and plasma adrenocorticotropic hormone levels are elevated in the exon 3–deleted GR within the PVN (30, 33).
Although PVN-specific deletion of GR does not lead to alterations in baseline despair or anxiety behaviors, it is associated with stunted growth at birth and development of obesity during adulthood (33). A PVN and supraoptic nucleus–specific knockout of exon 2 of GR generated by the Sim1 Cre driver revealed the role of nuclear GR in rapid nongenomic effect of glucocorticoids on excitatory and inhibitory synapses in hypothalamic neuroendocrine cells (43). Finally, a selective GR exon 3 knockout in agouti-related protein (AgRP)–expressing neurons in the arcuate nucleus of the hypothalamus reduced body weight gain in mice fed a high-fat diet and increased energy expenditure, highlighting the role of GR in this selected population of neuroendocrine cells in regulating system-wide energy expenditure (44).
To assess the role of GR in addictive behaviors, exon 3 of GR was deleted specifically in dopamine-producing neurons using the Slc6a3 dopamine transporter gene promoter driving Cre expression or in postsynaptic dopaminoceptive neurons using the dopamine receptor 1a (Drd1a) promoter (45). Loss of GR expression in dopaminoreceptive neurons in the nucleus accumbens, dorsal striatum, and prefrontal cortex, but not from dopamine-producing cells, resulted in a reduced efficiency of cocaine self-administration (45). Electrophysiological studies suggest that GR in dopaminoreceptive neurons regulates cocaine self-administration via feedback control on the firing rate of dopaminergic neurons in ventral tegmental area (45). These mice were also used to reveal a role of GR in dopaminoreceptive, but not dopaminergic, neurons in stimulating social aversion that results from chronic stress (46) and neuronal and behavioral responses to amphetamine exposure (47).
Deletion on GR from noradrenergic neurons in the periphery and central nervous system was accomplished using the dopamine β-hydroxylase (Dbh) gene driving Cre expression. These studies established a role for GR in survival of adrenergic chromaffin cells (48) as well as for centrally driven, sex-specific regulation of anxiety and depressive behaviors (49).
Cardiovascular and Pulmonary System
Stress is a significant risk factor for cardiovascular disease (50). In fact, exposure to elevated endogenous or exogenous glucocorticoids has been associated with an increased risk of heart failure, ischemic heart disease, and hypertension (51, 52). However, both high and insufficient levels of glucocorticoids have been reported to adversely affect cardiovascular function, indicating that an appropriate balance of glucocorticoid levels is essential for this organ system (51, 53). In 1855, Dr. Thomas Addison reported “remarkable feebleness of the heart’s actions” in patients with adrenal insufficiency (54). Therefore, it is not surprising that mouse models harboring a deletion of GR in cells of the heart reported significant cardiovascular pathologies (12, 55, 56). Similar to GR-null mice, embryonic development of the heart in mice deficient in GR specifically in cardiomyocytes and vascular smooth muscle was impaired (55). The hearts of both GR-null and smooth muscle–specific knockout mice exhibited irregularly shaped and disorganized myofibrils at embryonic day 17.5. The expression of genes critical to cardiac development and function was diminished in the hearts of GR-null and smooth muscle–specific knockout mice compared with controls, likely contributing to the cardiac phenotype. Postnatally, deletion of GR in cardiomyocytes and smooth muscle cells resulted in larger hearts, cardiac fibrosis, and decreased early-life survival (56). In this model, cardiomyocyte hypertrophy was present in males but not females, suggesting that the sexually dimorphic actions of glucocorticoids, which have been demonstrated in other tissues, also occur in the heart (57). Another group used the same mating strategy to evaluate GR functions in rapid glucocorticoid-induced hypertension (58). Overall dexamethasone-induced hypertension was diminished in the mice with a targeted deletion of GR in the vascular smooth muscle, although a rise in blood pressure was evident following chronic dexamethasone treatment. The mechanism by which glucocorticoids mediate blood pressure through vascular GR was further investigated using Tie-2 Cre, which targets the vascular endothelium (59). Attenuation of glucocorticoid-induced hypertension was more pronounced in the endothelial-specific GR knockouts compared with the mice that targeted GR in vascular smooth muscle (58, 59). Endothelial GR was also found to be protective in response to lipopolysaccharide (LPS)-induced sepsis, by repressing the release of the inflammatory cytokines nuclear factor κB and interleukin-6 and acting as a negative regulator of nitric oxide release (60).
Cardiomyocyte-specific deletion of GR was accomplished utilizing the α-myosin heavy chain promoter. Maximal excision in this model occurs postnatally, and, accordingly, severe cardiac pathology is manifested in adult mice (12, 61). This conditional knockout of GR in cardiomyocytes resulted in cardiac hypertrophy, left ventricular systolic dysfunction, and premature death due to cardiovascular complications. Interestingly, the survival phenotype related to inactivation of cardiomyocyte GR also exhibited sexual dimorphism. The α-myosin heavy chain promoter was also used to drive overexpression of GR in cardiomyocytes (62). Overexpression of GR was associated with bradycardia and atrioventricular block, which was reversible when physiological levels of GR were restored. Cardiovascular dysfunctions in the mouse models of GR deletion and overexpression are analogous to clinical findings from patients with aberrant levels of glucocorticoids, and they provide important models to further examine the pathology of heart disease.
The primary mechanism of postnatal death in GR-null neonates is immature lung development contributing to respiratory failure. Therefore, it was of primary interest to identify the lung cell type responsible for this lethal phenotype. Early rescue studies with GR hypomorphic mice, where GR expression was restored to the lung epithelium under the regulation of the rat surfactant protein C (Spc) promoter, indicated that epithelial GR alone was not sufficient to prevent neonatal mortality (63). However, this rescue experiment was unable to restore GR to endogenous levels. The requirement of epithelial GR for lung development was also evaluated in mice harboring a lung epithelial cell–specific deletion of GR under the regulation of a doxycycline-inducible Spc promoter and utilizing Spc Cre mice (64–66). These models were produced independently and although utilizing a comparable strategy, they resulted in conflicting results. In one report, doxycycline was fed to dams starting at embryonic day 6.5 through day 18.5, which induced the deletion of lung epithelial GR and resulted in ∼50% mortality at birth, with a lung phenotype similar to GR-null neonates (64). Others utilizing the doxycycline-inducible system with doxycycline administered from embryonic day 6.5 to day 14.5 or mice with Cre-driven recombination under the control of the human Spc promoter reported no mortality phenotype in the absence of epithelial GR, but instead suggested that GR expression in mesenchymal cells was required for lung maturation (65–67). Deletion of GR in mesenchyme-derived cells phenocopied the GR-null mice. Mesenchymal GR was found to regulate the morphological differentiation, development, and remodeling of the neonatal lung. Severe lung cell hyperplasia was a hallmark of both the mesenchymal and GR-null mice, suggesting that one of the primary functions of glucocorticoid signaling during lung development is to halt proliferation and allow thinning of the interstitial cells for functional gas exchange.
Immune System
The immunosuppressive effects of glucocorticoids have driven their widespread use clinically and led to the production of mouse models to determine the cell types contributing to their therapeutic benefits. The first immune cell targeted was the T cell and used an Lck Cre driver (28). This fundamental study revealed that glucocorticoid signaling in T cells was responsible for governing potentially lethal cytokine production in response to immune activation. Subsequently, glucocorticoid-driven T cell functions have been studied in a variety of altered immunological states, including antigen-induced arthritis, autoimmune encephalomyelitis, LPS-induced sepsis, Toxoplasma gondii infection, graft-versus-host disease, and pregnancy (68–75). The first clinical use of glucocorticoids was for the treatment of rheumatoid arthritis; however, it was not until the development of Cre/lox technology that the primary target of this intervention could be identified (71). Antigen-induced arthritis was used to model rheumatoid arthritis in mice harboring a deletion of GR in myeloid cells (neutrophils and macrophages), dendritic cells, B cells, and T cells. The authors found that only the T cell–specific deletion of GR attenuated the therapeutic benefit of glucocorticoids. T cell–specific GR knockouts have also been used in an experimental model of autoimmune encephalomyelitis, a rodent model of multiple sclerosis, and indicated that the therapeutic effects of glucocorticoids in this disease are mediated by diverting the migration of peripheral T cells away from the central nervous system (68, 70). In response to sepsis, GR signaling in thymocytes/T cells was reported to be necessary for protection from septic death (72). Another group found that glucocorticoid signaling in dendritic cells provided protection from LPS-induced septic lethality, although the Cd11c Cre used for these experiments was also active in T cells (76). Graft-versus-host disease requires an immunological response by the recipient to donor T cells. Transplantation of GR-deficient donor T cells led to rapid and fatal graft-versus-host disease in mice, indicating that glucocorticoid signaling in T cells mediates the severity of this pathology (73). During pregnancy, T cell–specific glucocorticoid signaling was reported to mediate the expansion of regulatory T cells and contribute to pregnancy-related protection from autoimmunity (74). T cell–specific GR knockout mice were also used to study the physiological role of endogenous glucocorticoids, where it was found that glucocorticoid signaling in T cells regulates the adaptive immune response through mediating T cell selection (29).
GR regulation of the innate immune response was evaluated through cell-specific deletion in myeloid cells utilizing a Cre recombinase inserted into the endogenous M lysozyme locus (LysM Cre) (30, 77, 78). Macrophage GR knockout mice were more susceptible to LPS-induced mortality, indicating that glucocorticoid signaling in macrophages restricts their proinflammatory activities (77). It was also shown that GR expression in macrophages is required for the anti-inflammatory effects of glucocorticoids in response to contact dermatitis, by mediating the release of proinflammatory cytokines and chemokines (30). Results from this model also demonstrated that glucocorticoid signaling in macrophages may favor vascular calcification in atherosclerosis.
Musculoskeletal System
Skeletal muscle atrophy results from prolonged exposure to high levels of exogenous glucocorticoids. Skeletal muscle-specific GR knockout mice, generated with a muscle creatine kinase (Mck) promoter-driven Cre, established that GR directly regulates muscle atrophy in response to high-dose glucocorticoid treatment (79). Notably, the induction of atrophy-associated genes in the skeletal muscle of control mice was absent in the muscle-specific GR knockout mice. Disease-associated atrophy, or cachexia, is driven by systemic inflammation and the release of endogenous glucocorticoids, although the relative contribution of systemic vs local glucocorticoid action in the pathophysiology has been contested. Skeletal muscle–specific GR knockout mice were protected from muscle wasting in response to elevated endogenous glucocorticoids related to nutritional deprivation, endotoxin exposure, and cytotoxic chemotherapy, indicating that the cachexia results from direct actions of GR in the muscle (79–81). The physiological significance of skeletal muscle GR in energy homeostasis and metabolism was evaluated using skeletal muscle-specific GR knockout mice where recombination was driven by the α-actin 1 (Acta1) promoter, which targets striated muscle (82). Interestingly, these mice have increased muscle mass but decreased fat depots and altered metabolic profiles, suggesting that skeletal muscle GR plays a physiological role in regulating the systemic energy supply.
Certain disorders of the musculoskeletal system reflect an imbalance between the bone-resorptive activities of osteoclasts and the bone-forming activities of osteoblasts. In the early 1900s, Cushing described the effects of high levels of glucocorticoids on bone formation (83). Glucocorticoids have been shown to stimulate osteoclast-mediated bone resorption and reduce osteoblast-mediated bone formation, leading to an overall decrease in bone mass (84). However, paradoxical observations in vitro and in vivo indicate that both cell types are sensitive to glucocorticoid-mediated apoptosis (85). Attempting to dissect the relative contribution of osteoblasts and osteoclasts to glucocorticoid-induced bone loss, investigators have developed lineage-specific mouse models with a targeted deletion of GR (86, 87). Osteoclasts derive from the monocyte lineage, and therefore LysM Cre, which is active in myeloid cells, was used to conditionally delete GR in osteoclasts (88). In these mice, dexamethasone did not suppress bone formation (87). Deletion of GR in osteoblasts utilizing the runt-related transcription factor 2 (Runx2) gene promoter-driven Cre mice also spared these mice from prednisone-induced bone loss, although this group found no such protection when GR was deleted in osteoclasts (86). These effects appear independent of development, as GR-null embryos do not display gross differences in growth plate dimensions or amount of calcified bone (86). The discrepant data with respect to glucocorticoid-induced bone loss may be a result of different background strains (mixed 129 × C57BL/6 background vs backcrossed on the Balb/c background), experimental glucocorticoid (dexamethasone vs prednisolone), or GR floxed model (floxed exon 1C-2 vs floxed exon 3). Interestingly, disruption of glucocorticoid signaling in osteoblasts in a mouse model overexpressing the glucocorticoid-inactivating enzyme 11β-hydroxysteroid dehydrogenase type II under the rat collagen type I promoter prevented glucocorticoid-induced weight gain and the development of altered glucose and insulin sensitivity (89). From these studies, it is evident that GR in the skeletal system directly regulates glucocorticoid-induced osteoporosis and contributes to metabolic disorders that are related to glucocorticoids.
Metabolism and Digestive System
Under physiological conditions and in response to acute injury, glucocorticoids may serve as gastroprotective agents (90). In fact, Dr. Addison’s early characterization of adrenal insufficiency included a report of “irritability of the stomach” (54). However, chronic treatment with glucocorticoids is associated with an increased risk of stomach ulcers. To date, no model containing a conditional deletion of GR in the cells of the stomach has been reported. Intestine-specific GR deletion with villin 1 (Vil1) Cre did not protect mice from glucocorticoid-induced gastroparesis, suggesting that this pathology could be mediated by GR in the stomach (91). The role of glucocorticoid signaling in the stomach remains an important direction for future research.
Glucocorticoid actions in the gastrointestinal system and metabolic organs have been evaluated through tissue-specific deletion of GR in the intestine, adipocytes, pancreas, and liver (92–95). In the small intestine, specifically enterocytes, GR is necessary for glucocorticoid-induced glucose transport and hyperglycemia, potentially through regulating the expression of transport proteins (92). GR functions in adipocytes also contribute to metabolic homeostasis, specifically during periods of an altered metabolic state. Under standard feeding conditions, targeted deletion of GR in adipocytes, driven by Cre recombinase under the control of the adiponectin (Adipoq) promoter/enhancer, did not affect body weight or adipogenesis. Importantly, note that Adipoq Cre expression occurs postnatally and is restricted to mature adipocytes. A separate group, in which Cre is expressed from the myogenic factor 5 (Myf5) endogenous locus during the embryonic period, also found that GR was not required for adipogenesis (96). Instead, this group found that adipocyte GR regulates the timing of adipogenesis by promoting the expression of adipogenic transcription factors. In response to a high-fat diet, adipocyte GR mediates inflammation and diet-induced obesity (93, 97, 98). Mice with an adipocyte-specific deletion of GR were resistant to diet-induced obesity, with reduced expression of genes related to fatty acid storage, diminished hepatic lipid accumulation, and improved glucose tolerance (97). These mice also demonstrate a delay in posthypoglycemic recovery following fasting, providing further evidence that the lipolytic capacity of the liver is reduced in the absence of adipocyte GR. Attenuation of diet-induced obesity was independently confirmed in an adipocyte-specific GR knockout utilizing a floxed GR strategy targeting a different exon (1C-2) of Nr3c1 (98). These results suggest a gene-by-environment effect where the function of glucocorticoids in adipocytes is enhanced when mice are subjected to an altered metabolic state. An earlier study had reported that the adipocyte-specific GR knockout mice were not protected from diet-induced obesity, although the endpoint for that study was at an earlier age (93). The actions of GR in mediating obesity in response to metabolic excess were also linked to impaired HPA regulation (98). Specific deletion of GR in adipocytes resulted in altered hypothalamic expression of Crh, Avp, and Pomc messenger RNA, suggesting that adipose tissue contributes to central feedback responses.
The liver is a major target of glucocorticoid action, regulating glycogen metabolism and gluconeogenesis. Metabolic regulation by hepatocyte GR has been evaluated through the use of albumin (Alb) Cre in the presence and absence of a α-fetoprotein (AFP) enhancer (95, 99–107). AFP is an early marker of the hepatocyte lineage, and expression of AFP occurs at embryonic day 8.5 (108). Alb was initially detected at embryonic day 9.5 in hepatic precursor cells, and expression increased following the formation of the liver (109). When the AFP enhancer was included in the Alb Cre construct, hepatic GR was found to be critical for postnatal viability and growth (95). Approximately 50% of these mice die within 48 hours of birth, and growth deficits were evident in surviving mice shortly after weaning. The severity of the phenotype occurred in a sexually dimorphic manner, where males were more severely affected than females. The growth-promoting effects of hepatic glucocorticoids were shown to be mediated through interactions with signal transducer and activator of transcription (Stat)5, a member of the STAT family of transcription factors (95, 99). The surviving adult hepatic GR knockout mice driven by Alb Cre with the AFP enhancer have normal serum glucose levels but develop hypoglycemia following prolonged fasting (100). When crossed to liver-specific Stat5-deficient mice, the absence of GR and Stat5 leads to hypercortisolism, severe metabolic dysfunctions, and the spontaneous development of hepatocellular carcinoma (104). Viability defects were not reported in mice with a hepatocyte-specific deletion of GR driven by Alb Cre that lacked the AFP enhancer (102, 105). In this model of hepatic GR deficiency, mice demonstrated increased insulin sensitivity and reduced glucocorticoid-mediated glycogen storage compared with wild-types (106). Hepatocyte expression of GR was also found to be necessary for proliferation and adipogenesis during hepatic regeneration (105). The sexually dimorphic actions of glucocorticoids on hepatic gene expression were evaluated in mice with a hepatocyte-specific deletion of GR lacking the AFP enhancer (107). In response to immune challenge, hepatic GR drives liver gene expression in a sex-specific manner. In addition to the metabolic functions of GR in the liver, glucocorticoid signaling may regulate metabolism and the digestive system through direct actions in pancreas. Deletion of GR in pancreatic precursor cells [under the regulation of pancreas and duodenum homeobox 1 (Pdx-1) Cre] but not insulin-producing β-cells (driven by the rat insulin promoter Cre) increased β-cell numbers, indicating the sensitive nature of the developing pancreas and a potential link between early exposure to elevated glucocorticoids and adult metabolic disorders (94).
Renal System
Glucocorticoids are primary therapy for many pathological manifestations of the kidney, including glomerulonephritides and idiopathic nephrotic syndrome. However, the mechanisms by which glucocorticoids act as therapeutic agents in the kidney are still unclear. To understand the functions of glucocorticoid signaling in the kidney, GR was deleted from the distal nephron and kidney epithelial cells (110, 111). Conditional deletion of GR in the distal nephron was accomplished by Ksp-cadherin (Ksp) promoter-driven Cre expression (110). Mean baseline blood pressure was mildly elevated in these mice, but glucocorticoid-induced excretion of urinary electrolytes and hypertension was indistinguishable to that of controls, suggesting that glucocorticoid signaling in the distal nephron does not mediate this response. On the contrary, deletion of GR from kidney epithelial cells utilizing the paired box 8 (Pax8) Cre driver did not result in a phenotype under physiological conditions but was protective in an induced model of progressive glomerulonephritides (111). Interestingly, the deletion of GR prevented albuminuria and cellular crescent formation, hallmarks of progressive glomerulonephritides, as effectively as high-dose prednisone treatment in controls. However, the immunosuppressive effects of prednisone treatment were evident independent of genotype. The differential effects of GR deletion on the pathology of glomerulonephritides suggest a complex signaling pathway where the presence of GR in the kidney epithelium may be partially deleterious under certain conditions.
Reproductive System
The direct actions of GR in the reproductive tract and tissues associated with the reproductive tract have been largely overlooked, despite evidence indicating the profound effect of elevated glucocorticoids on reproductive function. In the male reproductive tract, GR has been specifically deleted in the testicular Sertoli cell and prostate epithelium (112, 113). Mice with a Sertoli cell–specific deletion of GR driven by promoter elements of the anti-Müllerian hormone (Amh) gene were fertile, although seminiferous tubules had fewer Sertoli cells, pachytene spermatocytes, and round spermatids (112). Circulating gonadotropin levels and expression of the gonadotropin receptors in the testis were also decreased in these mice, indicating that glucocorticoid signaling in the Sertoli cell is an important mediator of testicular steroidogenesis. Glucocorticoids as part of a combined therapy and selective GR modulators have been used clinically for the treatment of castration-resistant prostate cancer, supported by in vitro modeling that has indicated that glucocorticoid treatment inhibits proliferation of prostate cancer cells (114, 115). Probasin (Pbsn) Cre was used to delete GR from prostate epithelial cells to evaluate the in vivo effects of glucocorticoid signaling in the prostate (113). Under physiological conditions and in response to hormone treatment, the loss of GR did not alter prostate growth and proliferation. The absence of a phenotype may reflect the unique properties of glucocorticoids in the cellular context of castration-resistant prostate cancer, where glucocorticoids exert an antiproliferative effect.
In the female reproductive tract, GR deletion from mammary epithelial cells resulted in delayed pregnancy-associated lobuloalveolar development due to retarded epithelial cell proliferation (116). Milk composition was apparently normal in these mice, but pups weighed less during the early postnatal period, suggesting that mammary epithelial cell GR was not essential but contributes to the functions of the mammary glands during lactation.
The impact of chronic stress and elevated glucocorticoids on female fertility is mediated in part by the suppressive effects on the hypothalamus and pituitary, but the extent of local actions in the uterus were not clear (117). To evaluate the direct contribution of glucocorticoid signaling to female fertility, GR was conditionally deleted from the uterus using progesterone receptor (PR) Cre (118). In the absence of uterine GR, females were subfertile with fewer implantation sites and deficient decidualization of endometrial stromal cells, highlighting the importance of glucocorticoid signaling in the uterus to early stages of pregnancy. Absence of glucocorticoid signaling in the uterus led to an impaired molecular response to induced decidualization, which resulted in diminished stromal cell proliferation. These findings suggest a crucial role for GR in orchestrating the morphological changes required to support pregnancy.
Integumentary System
Glucocorticoids are one of most widely used therapies in dermatology, as they are highly effective for the topical treatment of inflammatory skin diseases. However, their long-term use topically and systemically is associated with common side-effects that include skin atrophy, disruption to cutaneous barrier function, and dermatitis (119). As with the other side-effects arising from prolonged exposure to elevated glucocorticoids, defects to skin function may result from local disruption to glucocorticoid signaling or systemic changes to HPA axis activity. Therefore, the contribution of GR signaling in skin development and function was evaluated in embryos of GR-null and GRdim mice, which harbor a point mutation in the DNA-binding domain of GR reported to be important for receptor dimerization, and compared with a keratinocyte-specific GR deficient mouse model and a mouse model constitutively overexpressing GR in keratinocytes (120–123).
Global GR-null mice at embryonic day 18.5 demonstrate significant defects in skin development, with incomplete epidermal stratification, impaired keratinocyte differentiation, and compromised skin barrier function, although the epidermis of GRdim mice appeared normal (120). Microarray analysis of the skin from GR-null embryos showed appreciable changes in the expression of genes related to keratinocyte terminal differentiation and ectoderm/epidermis development (124). Interestingly, gene expression analysis of the skin from an adult inducible knockout of GR in keratinocytes driven by the keratin 14 (K14) promoter revealed only partial overlap with the genes dysregulated in the embryonic skin of global GR-null mice, suggesting that certain functions of GR in the skin are specific to the developmental period (124). The phenotype of the keratinocyte-specific GR-deficient mice, where GR is excised at embryonic day 15.5 with keratin 5 (K5) Cre, closely resembled that of the GR-null mice (121). In addition to impaired skin barrier function and keratinocyte differentiation, the neonatal and postnatal keratinocyte-specific GR-deficient mice demonstrated epidermal hyperplasia. The phenotype was less pronounced in adult mice lacking GR in keratinocytes, but these mice were more sensitive to epidermal challenge, including increased susceptibility to skin tumorigenesis and tumor malignancy (125). However, removing GR from adult keratinocytes by the inducible keratin 14 promoter did not sensitize these mice to contact hypersensitivity, and dexamethasone was effective in reducing inflammation, indicating that the mechanism of GR-mediated inflammation in response to a dermal irritant is specific to the stimulus (30). Overexpression of GR in keratinocytes with keratin 5 Cre impaired proliferation, leading to a complete absence of skin in focal regions of postnatal mice (122). In adult mice, GR overexpression delayed wound healing similar to that of glucocorticoid-treated wild-type mice, which was related to deficient proliferation and immune activation. These mice were also unresponsive in tumor-promoting assays, suggesting that disruptions to glucocorticoid signaling in the skin may contribute to carcinogenesis.
Summary and Perspective
Glucocorticoids regulate numerous physiological processes with astonishing specificity by tissue and cell type, which has been exemplified in the tissue-specific GR knockout mice. These models have demonstrated that glucocorticoid signaling directly mediates physiological and pathophysiological functions within target tissues in addition to the indirect actions on the HPA/gonadal axis. For example, elevated glucocorticoid levels suppress reproductive function, which has been largely attributed to suppression of hypothalamic and pituitary function (126, 127). However, the phenotypes of the uterine- and Sertoli cell–specific GR knockout mice indicate that glucocorticoid signaling also directly regulates uterine and testicular function, respectively (112, 118). The direct actions of GR have been investigated in almost 40 unique Cre-driven knockout mice. However, several organs have yet to be targeted, and the relative contribution of GR in cell types of heterogeneous tissues is not well understood. Moreover, discrepant data have been reported for some tissues that have been studied by individual investigators using different experimental strategies, including various background strains, Cre drivers, and floxed GR mice. In tissues where the utilization of different promoter-driven Cre lines leads to discrepant data, insight is provided on the importance of GR expression during specific periods of embryonic development. Other differences may stem from the background strain of the mouse model, as recent studies in GR heterozygous-null mice have provided further evidence that the behavioral phenotype mediated by glucocorticoid signaling is sensitive to genetic background (128). Therefore, independent validation of an observed phenotype is of immense value when evaluating tissue-specific knockout mice.
Identifying the tissues and cells of direct GR action is a significant accomplishment of the last 20 years, but also represents the initial step in our understanding of how GR regulates context-specific gene expression. To interpret the phenotype of the tissue-specific GR knockout mice, the genes directly regulated by GR that contribute to the phenotype and the functional response elements responsible for their regulation must be identified. It is also important to determine the mechanism by which allosteric GR–DNA interactions lead to specific transcriptional profiles. The GRdim mice were generated to dissect the DNA binding–dependent and –independent activities of GR. However, recent evidence demonstrating GR dimerization and transactivation in a promoter-specific manner with the GRdim mutation suggests that the regulation of gene expression by GR is more complex and would benefit from additions mutational studies (129, 130). A gain-of-function point mutation in the hormone-binding domain of GR was previously described and may provide insight into the mechanism of glucocorticoid hypersensitivity (131). Reported polymorphisms and in vitro studies have evaluated other GR residues important for dimerization, phosphorylation, and transactivation/transrepression, although their functions in a physiological model have not been tested (129, 132–134). Targeted genome editing with clustered regularly interspersed short palindromic repeats (CRISPR)/Cas-9 technology will provide a mechanism for rapid, high-throughput manipulation of GR-binding sites in transcriptional regulatory regions or protein interaction domains required for transcriptional regulatory complexes. This technology has been successfully used in several species (zebrafish, monkeys, mice, rats, pigs, and human embryos), and could expand our understanding of glucocorticoid signaling in other relevant research models with applications to human health (135, 136). By identifying and targeting the primary transcripts of GR, we can begin to build the regulatory network responsible for GR actions in a given tissue. Importantly, understanding the signaling pathways responsible for health and disease may lead to new possibilities for treatment.
Acknowledgments
Financial Support: This work was supported in part by National Institutes of Health Grant 4R00 ES022983 (to S.W.), an Albert McKern Scholar Award (to S.W.), and National Institutes of Health Grant R01 HD087288 (to D.B.D.).
Acknowledgments
Disclosure Summary: The authors have nothing to disclose.
Footnotes
- AFP
- α-fetoprotein
- Alb
- albumin
- CaMKIIα
- calcium/calmodulin-dependent kinase II-α
- CRH
- corticotropin-releasing hormone
- GR
- glucocorticoid receptor
- GRE
- glucocorticoid response element
- HPA
- hypothalamic–pituitary–adrenal
- LPS
- lipopolysaccharide
- PVN
- paraventricular nucleus
- Sim1
- single-minded homolog 1
- Spc
- surfactant protein C
- Stat
- signal transducer and activator of transcription.
References
- 1.Hollenberg SM, Weinberger C, Ong ES, Cerelli G, Oro A, Lebo R, Thompson EB, Rosenfeld MG, Evans RM. Primary structure and expression of a functional human glucocorticoid receptor cDNA. Nature. 1985;318(6047):635–641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kumar R, Thompson EB. Gene regulation by the glucocorticoid receptor: structure:function relationship. J Steroid Biochem Mol Biol. 2005;94(5):383–394. [DOI] [PubMed] [Google Scholar]
- 3.Luisi BF, Xu WX, Otwinowski Z, Freedman LP, Yamamoto KR, Sigler PB. Crystallographic analysis of the interaction of the glucocorticoid receptor with DNA. Nature. 1991;352(6335):497–505. [DOI] [PubMed] [Google Scholar]
- 4.Surjit M, Ganti KP, Mukherji A, Ye T, Hua G, Metzger D, Li M, Chambon P. Widespread negative response elements mediate direct repression by agonist-liganded glucocorticoid receptor. Cell. 2011;145(2):224–241. [DOI] [PubMed] [Google Scholar]
- 5.Weikum ER, Knuesel MT, Ortlund EA, Yamamoto KR. Glucocorticoid receptor control of transcription: precision and plasticity via allostery. Nat Rev Mol Cell Biol. 2017;18(3):159–174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bledsoe RK, Montana VG, Stanley TB, Delves CJ, Apolito CJ, McKee DD, Consler TG, Parks DJ, Stewart EL, Willson TM, Lambert MH, Moore JT, Pearce KH, Xu HE. Crystal structure of the glucocorticoid receptor ligand binding domain reveals a novel mode of receptor dimerization and coactivator recognition. Cell. 2002;110(1):93–105. [DOI] [PubMed] [Google Scholar]
- 7.Kirschke E, Goswami D, Southworth D, Griffin PR, Agard DA. Glucocorticoid receptor function regulated by coordinated action of the Hsp90 and Hsp70 chaperone cycles. Cell. 2014;157(7):1685–1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yang J, DeFranco DB. Assessment of glucocorticoid receptor-heat shock protein 90 interactions in vivo during nucleocytoplasmic trafficking. Mol Endocrinol. 1996;10(1):3–13. [DOI] [PubMed] [Google Scholar]
- 9.Davies TH, Ning YM, Sánchez ER. A new first step in activation of steroid receptors: hormone-induced switching of FKBP51 and FKBP52 immunophilins. J Biol Chem. 2002;277(7):4597–4600. [DOI] [PubMed] [Google Scholar]
- 10.Echeverría PC, Mazaira G, Erlejman A, Gomez-Sanchez C, Piwien Pilipuk G, Galigniana MD. Nuclear import of the glucocorticoid receptor-hsp90 complex through the nuclear pore complex is mediated by its interaction with Nup62 and importin β. Mol Cell Biol. 2009;29(17):4788–4797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamamoto KR, Darimont BD, Wagner RL, Iñiguez-Lluhí JA. Building transcriptional regulatory complexes: signals and surfaces. Cold Spring Harb Symp Quant Biol. 1998;63:587–598. [DOI] [PubMed] [Google Scholar]
- 12.Oakley RH, Ren R, Cruz-Topete D, Bird GS, Myers PH, Boyle MC, Schneider MD, Willis MS, Cidlowski JA. Essential role of stress hormone signaling in cardiomyocytes for the prevention of heart disease. Proc Natl Acad Sci USA. 2013;110(42):17035–17040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Presman DM, Ball DA, Paakinaho V, Grimm JB, Lavis LD, Karpova TS, Hager GL. Quantifying transcription factor binding dynamics at the single-molecule level in live cells. Methods. 2017;123:76–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goldstein I, Baek S, Presman DM, Paakinaho V, Swinstead EE, Hager GL. Transcription factor assisted loading and enhancer dynamics dictate the hepatic fasting response. Genome Res. 2017;27(3):427–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Oakley RH, Sar M, Cidlowski JA. The human glucocorticoid receptor β isoform. Expression, biochemical properties, and putative function. J Biol Chem. 1996;271(16):9550–9559. [DOI] [PubMed] [Google Scholar]
- 16.Anbalagan M, Huderson B, Murphy L, Rowan BG. Post-translational modifications of nuclear receptors and human disease. Nucl Recept Signal. 2012;10:e001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wallace AD, Cidlowski JA. Proteasome-mediated glucocorticoid receptor degradation restricts transcriptional signaling by glucocorticoids. J Biol Chem. 2001;276(46):42714–42721. [DOI] [PubMed] [Google Scholar]
- 18.Wyllie AH. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature. 1980;284(5756):555–556. [DOI] [PubMed] [Google Scholar]
- 19.Viegas LR, Hoijman E, Beato M, Pecci A. Mechanisms involved in tissue-specific apopotosis regulated by glucocorticoids. J Steroid Biochem Mol Biol. 2008;109(3-5):273–278. [DOI] [PubMed] [Google Scholar]
- 20.Li Z, Dong J, Zou T, Du C, Li S, Chen C, Liu R, Wang K. Dexamethasone induces docetaxel and cisplatin resistance partially through up-regulating Krüppel-like factor 5 in triple-negative breast cancer. Oncotarget. 2017;8(7):11555–11565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cruz-Topete D, Cidlowski JA. One hormone, two actions: anti- and pro-inflammatory effects of glucocorticoids. Neuroimmunomodulation. 2015;22(1-2):20–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Whirledge S, Cidlowski JA. Glucocorticoids and reproduction: traffic control on the road to reproduction. Trends Endocrinol Metab. 2017;28(6):399–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cole TJ, Blendy JA, Monaghan AP, Krieglstein K, Schmid W, Aguzzi A, Fantuzzi G, Hummler E, Unsicker K, Schütz G. Targeted disruption of the glucocorticoid receptor gene blocks adrenergic chromaffin cell development and severely retards lung maturation. Genes Dev. 1995;9(13):1608–1621. [DOI] [PubMed] [Google Scholar]
- 24.Gesina E, Blondeau B, Milet A, Le Nin I, Duchene B, Czernichow P, Scharfmann R, Tronche F, Breant B. Glucocorticoid signalling affects pancreatic development through both direct and indirect effects. Diabetologia. 2006;49(12):2939–2947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pepin MC, Pothier F, Barden N. Impaired type II glucocorticoid-receptor function in mice bearing antisense RNA transgene. Nature. 1992;355(6362):725–728. [DOI] [PubMed] [Google Scholar]
- 26.Korte SM, De Kloet ER, Buwalda B, Bouman SD, Bohus B. Antisense to the glucocorticoid receptor in hippocampal dentate gyrus reduces immobility in forced swim test. Eur J Pharmacol. 1996;301(1-3):19–25. [DOI] [PubMed] [Google Scholar]
- 27.Verma IM, Somia N. Gene therapy—promises, problems and prospects. Nature. 1997;389(6648):239–242. [DOI] [PubMed] [Google Scholar]
- 28.Brewer JA, Khor B, Vogt SK, Muglia LM, Fujiwara H, Haegele KE, Sleckman BP, Muglia LJ. T-cell glucocorticoid receptor is required to suppress COX-2-mediated lethal immune activation. Nat Med. 2003;9(10):1318–1322. [DOI] [PubMed] [Google Scholar]
- 29.Mittelstadt PR, Monteiro JP, Ashwell JD. Thymocyte responsiveness to endogenous glucocorticoids is required for immunological fitness. J Clin Invest. 2012;122(7):2384–2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jeanneteau FD, Lambert WM, Ismaili N, Bath KG, Lee FS, Garabedian MJ, Chao MV. BDNF and glucocorticoids regulate corticotrophin-releasing hormone (CRH) homeostasis in the hypothalamus. Proc Natl Acad Sci USA. 2012;109(4):1305–1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tronche F, Kellendonk C, Kretz O, Gass P, Anlag K, Orban PC, Bock R, Klein R, Schütz G. Disruption of the glucocorticoid receptor gene in the nervous system results in reduced anxiety. Nat Genet. 1999;23(1):99–103. [DOI] [PubMed] [Google Scholar]
- 32.Tuckermann JP, Kleiman A, Moriggl R, Spanbroek R, Neumann A, Illing A, Clausen BE, Stride B, Förster I, Habenicht AJ, Reichardt HM, Tronche F, Schmid W, Schütz G. Macrophages and neutrophils are the targets for immune suppression by glucocorticoids in contact allergy. J Clin Invest. 2007;117(5):1381–1390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Laryea G, Schütz G, Muglia LJ. Disrupting hypothalamic glucocorticoid receptors causes HPA axis hyperactivity and excess adiposity. Mol Endocrinol. 2013;27(10):1655–1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kellendonk C, Eiden S, Kretz O, Schütz G, Schmidt I, Tronche F, Simon E. Inactivation of the GR in the nervous system affects energy accumulation. Endocrinology. 2002;143(6):2333–2340. [DOI] [PubMed] [Google Scholar]
- 35.Boyle MP, Brewer JA, Funatsu M, Wozniak DF, Tsien JZ, Izumi Y, Muglia LJ. Acquired deficit of forebrain glucocorticoid receptor produces depression-like changes in adrenal axis regulation and behavior. Proc Natl Acad Sci USA. 2005;102(2):473–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Boyle MP, Kolber BJ, Vogt SK, Wozniak DF, Muglia LJ. Forebrain glucocorticoid receptors modulate anxiety-associated locomotor activation and adrenal responsiveness. J Neurosci. 2006;26(7):1971–1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Furay AR, Bruestle AE, Herman JP. The role of the forebrain glucocorticoid receptor in acute and chronic stress. Endocrinology. 2008;149(11):5482–5490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Solomon MB, Furay AR, Jones K, Packard AE, Packard BA, Wulsin AC, Herman JP. Deletion of forebrain glucocorticoid receptors impairs neuroendocrine stress responses and induces depression-like behavior in males but not females. Neuroscience. 2012;203:135–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Erdmann G, Schütz G, Berger S. Loss of glucocorticoid receptor function in the pituitary results in early postnatal lethality. Endocrinology. 2008;149(7):3446–3451. [DOI] [PubMed] [Google Scholar]
- 40.Schmidt MV, Sterlemann V, Wagner K, Niederleitner B, Ganea K, Liebl C, Deussing JM, Berger S, Schütz G, Holsboer F, Müller MB. Postnatal glucocorticoid excess due to pituitary glucocorticoid receptor deficiency: differential short- and long-term consequences. Endocrinology. 2009;150(6):2709–2716. [DOI] [PubMed] [Google Scholar]
- 41.Vincent MY, Hussain RJ, Zampi ME, Sheeran K, Solomon MB, Herman JP, Khan A, Jacobson L. Sensitivity of depression-like behavior to glucocorticoids and antidepressants is independent of forebrain glucocorticoid receptors. Brain Res. 2013;1525:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Solomon MB, Loftspring M, de Kloet AD, Ghosal S, Jankord R, Flak JN, Wulsin AC, Krause EG, Zhang R, Rice T, McKlveen J, Myers B, Tasker JG, Herman JP. Neuroendocrine function after hypothalamic depletion of glucocorticoid receptors in male and female mice. Endocrinology. 2015;156(8):2843–2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nahar J, Haam J, Chen C, Jiang Z, Glatzer NR, Muglia LJ, Dohanich GP, Herman JP, Tasker JG. Rapid nongenomic glucocorticoid actions in male mouse hypothalamic neuroendocrine cells are dependent on the nuclear glucocorticoid receptor. Endocrinology. 2015;156(8):2831–2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shibata M, Banno R, Sugiyama M, Tominaga T, Onoue T, Tsunekawa T, Azuma Y, Hagiwara D, Lu W, Ito Y, Goto M, Suga H, Sugimura Y, Oiso Y, Arima H. AgRP neuron-specific deletion of glucocorticoid receptor leads to increased energy expenditure and decreased body weight in female mice on a high-fat diet. Endocrinology. 2016;157(4):1457–1466. [DOI] [PubMed] [Google Scholar]
- 45.Ambroggi F, Turiault M, Milet A, Deroche-Gamonet V, Parnaudeau S, Balado E, Barik J, van der Veen R, Maroteaux G, Lemberger T, Schütz G, Lazar M, Marinelli M, Piazza PV, Tronche F. Stress and addiction: glucocorticoid receptor in dopaminoceptive neurons facilitates cocaine seeking. Nat Neurosci. 2009;12(3):247–249. [DOI] [PubMed] [Google Scholar]
- 46.Barik J, Marti F, Morel C, Fernandez SP, Lanteri C, Godeheu G, Tassin JP, Mombereau C, Faure P, Tronche F. Chronic stress triggers social aversion via glucocorticoid receptor in dopaminoceptive neurons. Science. 2013;339(6117):332–335. [DOI] [PubMed] [Google Scholar]
- 47.Parnaudeau S, Dongelmans ML, Turiault M, Ambroggi F, Delbes AS, Cansell C, Luquet S, Piazza PV, Tronche F, Barik J. Glucocorticoid receptor gene inactivation in dopamine-innervated areas selectively decreases behavioral responses to amphetamine. Front Behav Neurosci. 2014;8:35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Parlato R, Otto C, Tuckermann J, Stotz S, Kaden S, Gröne HJ, Unsicker K, Schütz G. Conditional inactivation of glucocorticoid receptor gene in dopamine-β-hydroxylase cells impairs chromaffin cell survival. Endocrinology. 2009;150(4):1775–1781. [DOI] [PubMed] [Google Scholar]
- 49.Chmielarz P, Kuśmierczyk J, Parlato R, Schütz G, Nalepa I, Kreiner G. Inactivation of glucocorticoid receptor in noradrenergic system influences anxiety- and depressive-like behavior in mice. PLoS One. 2013;8(8):e72632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Oakley RH, Cidlowski JA. Glucocorticoid signaling in the heart: a cardiomyocyte perspective. J Steroid Biochem Mol Biol. 2015;153:27–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Souverein PC, Berard A, Van Staa TP, Cooper C, Egberts AC, Leufkens HG, Walker BR. Use of oral glucocorticoids and risk of cardiovascular and cerebrovascular disease in a population based case-control study. Heart. 2004;90(8):859–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pivonello R, De Leo M, Cozzolino A, Colao A. The treatment of Cushing’s disease. Endocr Rev. 2015;36(4):385–486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cruz-Topete D, Myers PH, Foley JF, Willis MS, Cidlowski JA. Corticosteroids are essential for maintaining cardiovascular function in male mice. Endocrinology. 2016;157(7):2759–2771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Addison T. On the Constitution and Local Effects of Disease of the Suprarenal Capsules. London, England: Samuel Highley; 1855. [Google Scholar]
- 55.Rog-Zielinska EA, Thomson A, Kenyon CJ, Brownstein DG, Moran CM, Szumska D, Michailidou Z, Richardson J, Owen E, Watt A, Morrison H, Forrester LM, Bhattacharya S, Holmes MC, Chapman KE. Glucocorticoid receptor is required for foetal heart maturation. Hum Mol Genet. 2013;22(16):3269–3282. [DOI] [PubMed] [Google Scholar]
- 56.Richardson RV, Batchen EJ, Thomson AJ, Darroch R, Pan X, Rog-Zielinska EA, Wyrzykowska W, Scullion K, Al-Dujaili EA, Diaz ME, Moran CM, Kenyon CJ, Gray GA, Chapman KE. Glucocorticoid receptor alters isovolumetric contraction and restrains cardiac fibrosis. J Endocrinol. 2017;232(3):437–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Duma D, Collins JB, Chou JW, Cidlowski JA. Sexually dimorphic actions of glucocorticoids provide a link to inflammatory diseases with gender differences in prevalence. Sci Signal. 2010;3(143):ra74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Goodwin JE, Zhang J, Geller DS. A critical role for vascular smooth muscle in acute glucocorticoid-induced hypertension. J Am Soc Nephrol. 2008;19(7):1291–1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Goodwin JE, Zhang J, Gonzalez D, Albinsson S, Geller DS. Knockout of the vascular endothelial glucocorticoid receptor abrogates dexamethasone-induced hypertension. J Hypertens. 2011;29(7):1347–1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Goodwin JE, Feng Y, Velazquez H, Sessa WC. Endothelial glucocorticoid receptor is required for protection against sepsis. Proc Natl Acad Sci USA. 2013;110(1):306–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Agah R, Frenkel PA, French BA, Michael LH, Overbeek PA, Schneider MD. Gene recombination in postmitotic cells. Targeted expression of Cre recombinase provokes cardiac-restricted, site-specific rearrangement in adult ventricular muscle in vivo. J Clin Invest. 1997;100(1):169–179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sainte-Marie Y, Nguyen Dinh Cat A, Perrier R, Mangin L, Soukaseum C, Peuchmaur M, Tronche F, Farman N, Escoubet B, Benitah JP, Jaisser F. Conditional glucocorticoid receptor expression in the heart induces atrio-ventricular block. FASEB J. 2007;21(12):3133–3141. [DOI] [PubMed] [Google Scholar]
- 63.Gagnon S, Atmodjo W, Humes D, McKerlie C, Kaplan F, Sweezey NB. Transgenic glucocorticoid receptor expression driven by the SP-C promoter reduces neonatal lung cellularity and midkine expression in GRhypo mice. Biol Neonate. 2006;90(1):46–57. [DOI] [PubMed] [Google Scholar]
- 64.Manwani N, Gagnon S, Post M, Joza S, Muglia L, Cornejo S, Kaplan F, Sweezey NB. Reduced viability of mice with lung epithelial-specific knockout of glucocorticoid receptor. Am J Respir Cell Mol Biol. 2010;43(5):599–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Habermehl D, Parkitna JR, Kaden S, Brügger B, Wieland F, Gröne HJ, Schütz G. Glucocorticoid activity during lung maturation is essential in mesenchymal and less in alveolar epithelial cells. Mol Endocrinol. 2011;25(8):1280–1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bird AD, Choo YL, Hooper SB, McDougall AR, Cole TJ. Mesenchymal glucocorticoid receptor regulates the development of multiple cell layers of the mouse lung. Am J Respir Cell Mol Biol. 2014;50(2):419–428. [DOI] [PubMed] [Google Scholar]
- 67.Li A, Hardy R, Stoner S, Tuckermann J, Seibel M, Zhou H. Deletion of mesenchymal glucocorticoid receptor attenuates embryonic lung development and abdominal wall closure. PLoS One. 2013;8(5):e63578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wüst S, van den Brandt J, Tischner D, Kleiman A, Tuckermann JP, Gold R, Lühder F, Reichardt HM. Peripheral T cells are the therapeutic targets of glucocorticoids in experimental autoimmune encephalomyelitis. J Immunol. 2008;180(12):8434–8443. [DOI] [PubMed] [Google Scholar]
- 69.Schweingruber N, Haine A, Tiede K, Karabinskaya A, van den Brandt J, Wüst S, Metselaar JM, Gold R, Tuckermann JP, Reichardt HM, Lühder F. Liposomal encapsulation of glucocorticoids alters their mode of action in the treatment of experimental autoimmune encephalomyelitis. J Immunol. 2011;187(8):4310–4318. [DOI] [PubMed] [Google Scholar]
- 70.Schweingruber N, Fischer HJ, Fischer L, van den Brandt J, Karabinskaya A, Labi V, Villunger A, Kretzschmar B, Huppke P, Simons M, Tuckermann JP, Flügel A, Lühder F, Reichardt HM. Chemokine-mediated redirection of T cells constitutes a critical mechanism of glucocorticoid therapy in autoimmune CNS responses. Acta Neuropathol. 2014;127(5):713–729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Baschant U, Frappart L, Rauchhaus U, Bruns L, Reichardt HM, Kamradt T, Bräuer R, Tuckermann JP. Glucocorticoid therapy of antigen-induced arthritis depends on the dimerized glucocorticoid receptor in T cells. Proc Natl Acad Sci USA. 2011;108(48):19317–19322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Guo L, Zheng Z, Ai J, Howatt DA, Mittelstadt PR, Thacker S, Daugherty A, Ashwell JD, Remaley AT, Li XA. Scavenger receptor BI and high-density lipoprotein regulate thymocyte apoptosis in sepsis. Arterioscler Thromb Vasc Biol. 2014;34(5):966–975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Theiss-Suennemann J, Jörß K, Messmann JJ, Reichardt SD, Montes-Cobos E, Lühder F, Tuckermann JP, AWolff H, Dressel R, Gröne HJ, Strauß G, Reichardt HM. Glucocorticoids attenuate acute graft-versus-host disease by suppressing the cytotoxic capacity of CD8+ T cells. J Pathol. 2015;235(4):646–655. [DOI] [PubMed] [Google Scholar]
- 74.Engler JB, Kursawe N, Solano ME, Patas K, Wehrmann S, Heckmann N, Lühder F, Reichardt HM, Arck PC, Gold SM, Friese MA. Glucocorticoid receptor in T cells mediates protection from autoimmunity in pregnancy. Proc Natl Acad Sci USA. 2017;114(2):E181–E190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kugler DG, Mittelstadt PR, Ashwell JD, Sher A, Jankovic D. CD4+ T cells are trigger and target of the glucocorticoid response that prevents lethal immunopathology in toxoplasma infection. J Exp Med. 2013;210(10):1919–1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li CC, Munitic I, Mittelstadt PR, Castro E, Ashwell JD. Suppression of dendritic cell-derived IL-12 by endogenous glucocorticoids is protective in LPS-induced sepsis. PLoS Biol. 2015;13(10):e1002269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bhattacharyya S, Brown DE, Brewer JA, Vogt SK, Muglia LJ. Macrophage glucocorticoid receptors regulate Toll-like receptor 4-mediated inflammatory responses by selective inhibition of p38 MAP kinase. Blood. 2007;109(10):4313–4319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Preusch MR, Rattazzi M, Albrecht C, Merle U, Tuckermann J, Schütz G, Blessing E, Zoppellaro G, Pauletto P, Krempien R, Rosenfeld ME, Katus HA, Bea F. Critical role of macrophages in glucocorticoid driven vascular calcification in a mouse-model of atherosclerosis. Arterioscler Thromb Vasc Biol. 2008;28(12):2158–2164. [DOI] [PubMed] [Google Scholar]
- 79.Watson ML, Baehr LM, Reichardt HM, Tuckermann JP, Bodine SC, Furlow JD. A cell-autonomous role for the glucocorticoid receptor in skeletal muscle atrophy induced by systemic glucocorticoid exposure. Am J Physiol Endocrinol Metab. 2012;302(10):E1210–E1220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Braun TP, Szumowski M, Levasseur PR, Grossberg AJ, Zhu X, Agarwal A, Marks DL. Muscle atrophy in response to cytotoxic chemotherapy is dependent on intact glucocorticoid signaling in skeletal muscle. PLoS One. 2014;9(9):e106489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Braun TP, Grossberg AJ, Krasnow SM, Levasseur PR, Szumowski M, Zhu XX, Maxson JE, Knoll JG, Barnes AP, Marks DL. Cancer- and endotoxin-induced cachexia require intact glucocorticoid signaling in skeletal muscle. FASEB J. 2013;27(9):3572–3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Shimizu N, Maruyama T, Yoshikawa N, Matsumiya R, Ma Y, Ito N, Tasaka Y, Kuribara-Souta A, Miyata K, Oike Y, Berger S, Schütz G, Takeda S, Tanaka H. A muscle-liver-fat signalling axis is essential for central control of adaptive adipose remodelling. Nat Commun. 2015;6:6693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cushing H. The basophil adenomas of the pituitary body and their clinical manifestations (pituitary basophilism). Bull Johns Hopkins Hosp. 1932;50:137–195. [Google Scholar]
- 84.Mitra R. Adverse effects of corticosteroids on bone metabolism: a review. PM R. 2011;3(5):466–471, quiz 471. [DOI] [PubMed] [Google Scholar]
- 85.Weinstein RS, Jilka RL, Parfitt AM, Manolagas SC. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Potential mechanisms of their deleterious effects on bone. J Clin Invest. 1998;102(2):274–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rauch A, Seitz S, Baschant U, Schilling AF, Illing A, Stride B, Kirilov M, Mandic V, Takacz A, Schmidt-Ullrich R, Ostermay S, Schinke T, Spanbroek R, Zaiss MM, Angel PE, Lerner UH, David JP, Reichardt HM, Amling M, Schütz G, Tuckermann JP. Glucocorticoids suppress bone formation by attenuating osteoblast differentiation via the monomeric glucocorticoid receptor. Cell Metab. 2010;11(6):517–531. [DOI] [PubMed] [Google Scholar]
- 87.Kim HJ, Zhao H, Kitaura H, Bhattacharyya S, Brewer JA, Muglia LJ, Ross FP, Teitelbaum SL. Glucocorticoids suppress bone formation via the osteoclast. J Clin Invest. 2006;116(8):2152–2160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Clausen BE, Burkhardt C, Reith W, Renkawitz R, Förster I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 1999;8(4):265–277. [DOI] [PubMed] [Google Scholar]
- 89.Brennan-Speranza TC, Henneicke H, Gasparini SJ, Blankenstein KI, Heinevetter U, Cogger VC, Svistounov D, Zhang Y, Cooney GJ, Buttgereit F, Dunstan CR, Gundberg C, Zhou H, Seibel MJ. Osteoblasts mediate the adverse effects of glucocorticoids on fuel metabolism. J Clin Invest. 2012;122(11):4172–4189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Filaretova L. Glucocorticoids are gastroprotective under physiologic conditions. Ther Adv Chronic Dis. 2011;2(5):333–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Reichardt SD, Weinhage T, Rotte A, Föller M, Oppermann M, Lühder F, Tuckermann JP, Lang F, van den Brandt J, Reichardt HM. Glucocorticoids induce gastroparesis in mice through depletion of l-arginine. Endocrinology. 2014;155(10):3899–3908. [DOI] [PubMed] [Google Scholar]
- 92.Reichardt SD, Föller M, Rexhepaj R, Pathare G, Minnich K, Tuckermann JP, Lang F, Reichardt HM. Glucocorticoids enhance intestinal glucose uptake via the dimerized glucocorticoid receptor in enterocytes. Endocrinology. 2012;153(4):1783–1794. [DOI] [PubMed] [Google Scholar]
- 93.Desarzens S, Faresse N. Adipocyte glucocorticoid receptor has a minor contribution in adipose tissue growth. J Endocrinol. 2016;230(1):1–11. [DOI] [PubMed] [Google Scholar]
- 94.Gesina E, Tronche F, Herrera P, Duchene B, Tales W, Czernichow P, Breant B. Dissecting the role of glucocorticoids on pancreas development. Diabetes. 2004;53(9):2322–2329. [DOI] [PubMed] [Google Scholar]
- 95.Tronche F, Opherk C, Moriggl R, Kellendonk C, Reimann A, Schwake L, Reichardt HM, Stangl K, Gau D, Hoeflich A, Beug H, Schmid W, Schütz G. Glucocorticoid receptor function in hepatocytes is essential to promote postnatal body growth. Genes Dev. 2004;18(5):492–497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Park YK, Ge K. Glucocorticoid receptor accelerates, but is dispensable for, adipogenesis. Mol Cell Biol. 2017;37(2):e00260-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mueller KM, Hartmann K, Kaltenecker D, Vettorazzi S, Bauer M, Mauser L, Amann S, Jall S, Fischer K, Esterbauer H, Müller TD, Tschöp MH, Magnes C, Haybaeck J, Scherer T, Bordag N, Tuckermann JP, Moriggl R. Adipocyte glucocorticoid receptor deficiency attenuates aging- and HFD-induced obesity and impairs the feeding-fasting transition. Diabetes. 2017;66(2):272–286. [DOI] [PubMed] [Google Scholar]
- 98.de Kloet AD, Krause EG, Solomon MB, Flak JN, Scott KA, Kim DH, Myers B, Ulrich-Lai YM, Woods SC, Seeley RJ, Herman JP. Adipocyte glucocorticoid receptors mediate fat-to-brain signaling. Psychoneuroendocrinology. 2015;56:110–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Engblom D, Kornfeld JW, Schwake L, Tronche F, Reimann A, Beug H, Hennighausen L, Moriggl R, Schütz G. Direct glucocorticoid receptor–Stat5 interaction in hepatocytes controls body size and maturation-related gene expression. Genes Dev. 2007;21(10):1157–1162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Opherk C, Tronche F, Kellendonk C, Kohlmüller D, Schulze A, Schmid W, Schütz G. Inactivation of the glucocorticoid receptor in hepatocytes leads to fasting hypoglycemia and ameliorates hyperglycemia in streptozotocin-induced diabetes mellitus. Mol Endocrinol. 2004;18(6):1346–1353. [DOI] [PubMed] [Google Scholar]
- 101.Van Bogaert T, Vandevyver S, Dejager L, Van Hauwermeiren F, Pinheiro I, Petta I, Engblom D, Kleyman A, Schütz G, Tuckermann J, Libert C. Tumor necrosis factor inhibits glucocorticoid receptor function in mice: a strong signal toward lethal shock. J Biol Chem. 2011;286(30):26555–26567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.He B, Cruz-Topete D, Oakley RH, Xiao X, Cidlowski JA. Human glucocorticoid receptor β regulates gluconeogenesis and inflammation in mouse liver. Mol Cell Biol. 2015;36(5):714–730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Rose AJ, Berriel Díaz M, Reimann A, Klement J, Walcher T, Krones-Herzig A, Strobel O, Werner J, Peters A, Kleyman A, Tuckermann JP, Vegiopoulos A, Herzig S. Molecular control of systemic bile acid homeostasis by the liver glucocorticoid receptor. Cell Metab. 2011;14(1):123–130. [DOI] [PubMed] [Google Scholar]
- 104.Mueller KM, Kornfeld JW, Friedbichler K, Blaas L, Egger G, Esterbauer H, Hasselblatt P, Schlederer M, Haindl S, Wagner KU, Engblom D, Haemmerle G, Kratky D, Sexl V, Kenner L, Kozlov AV, Terracciano L, Zechner R, Schuetz G, Casanova E, Pospisilik JA, Heim MH, Moriggl R. Impairment of hepatic growth hormone and glucocorticoid receptor signaling causes steatosis and hepatocellular carcinoma in mice. Hepatology. 2011;54(4):1398–1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shteyer E, Liao Y, Muglia LJ, Hruz PW, Rudnick DA. Disruption of hepatic adipogenesis is associated with impaired liver regeneration in mice. Hepatology. 2004;40(6):1322–1332. [DOI] [PubMed] [Google Scholar]
- 106.Bose SK, Hutson I, Harris CA. Hepatic glucocorticoid receptor plays a greater role than adipose GR in metabolic syndrome despite renal compensation. Endocrinology. 2016;157(12):4943–4960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Quinn MA, Cidlowski JA. Endogenous hepatic glucocorticoid receptor signaling coordinates sex-biased inflammatory gene expression. FASEB J. 2016;30(2):971–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Shiojiri N. Enzymo- and immunocytochemical analyses of the differentiation of liver cells in the prenatal mouse. J Embryol Exp Morphol. 1981;62:139–152. [PubMed] [Google Scholar]
- 109.Cascio S, Zaret KS. Hepatocyte differentiation initiates during endodermal-mesenchymal interactions prior to liver formation. Development. 1991;113(1):217–225. [DOI] [PubMed] [Google Scholar]
- 110.Goodwin JE, Zhang J, Velazquez H, Geller DS. The glucocorticoid receptor in the distal nephron is not necessary for the development or maintenance of dexamethasone-induced hypertension. Biochem Biophys Res Commun. 2010;394(2):266–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kuppe C, van Roeyen C, Leuchtle K, Kabgani N, Vogt M, Van Zandvoort M, Smeets B, Floege J, Grone HJ, Moeller MJ. Investigations of glucocorticoid action in GN. J Am Soc Nephrol. 2017;28(5):1408–1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hazra R, Upton D, Jimenez M, Desai R, Handelsman DJ, Allan CM. In vivo actions of the Sertoli cell glucocorticoid receptor. Endocrinology. 2014;155(3):1120–1130. [DOI] [PubMed] [Google Scholar]
- 113.Zhao B, Choi JP, Jaehne M, Gao YR, Desai R, Tuckermann J, Zhou H, Handelsman DJ, Simanainen U. Glucocorticoid receptor in prostate epithelia is not required for corticosteroid-induced epithelial hyperproliferation in the mouse prostate. Prostate. 2014;74(10):1068–1078. [DOI] [PubMed] [Google Scholar]
- 114.Nishimura K, Nonomura N, Satoh E, Harada Y, Nakayama M, Tokizane T, Fukui T, Ono Y, Inoue H, Shin M, Tsujimoto Y, Takayama H, Aozasa K, Okuyama A. Potential mechanism for the effects of dexamethasone on growth of androgen-independent prostate cancer. J Natl Cancer Inst. 2001;93(22):1739–1746. [DOI] [PubMed] [Google Scholar]
- 115.Kach J, Long TM, Selman P, Tonsing-Carter EY, Bacalao MA, Lastra RR, de Wet L, Comiskey S, Gillard M, VanOpstall C, West DC, Chan WC, Griend DV, Conzen SD, Szmulewitz RZ. Selective glucocorticoid receptor modulators (SGRMs) delay castrate-resistant prostate cancer growth. Mol Cancer Ther. 2017;16(8):1680–1692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wintermantel TM, Bock D, Fleig V, Greiner EF, Schütz G. The epithelial glucocorticoid receptor is required for the normal timing of cell proliferation during mammary lobuloalveolar development but is dispensable for milk production. Mol Endocrinol. 2005;19(2):340–349. [DOI] [PubMed] [Google Scholar]
- 117.Rabin D, Gold PW, Margioris AN, Chrousos GP. Stress and reproduction: physiologic and pathophysiologic interactions between the stress and reproductive axes. Adv Exp Med Biol. 1988;245:377–387. [DOI] [PubMed] [Google Scholar]
- 118.Whirledge SD, Oakley RH, Myers PH, Lydon JP, DeMayo F, Cidlowski JA. Uterine glucocorticoid receptors are critical for fertility in mice through control of embryo implantation and decidualization. Proc Natl Acad Sci USA. 2015;112(49):15166–15171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Schäcke H, Döcke WD, Asadullah K. Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther. 2002;96(1):23–43. [DOI] [PubMed] [Google Scholar]
- 120.Bayo P, Sanchis A, Bravo A, Cascallana JL, Buder K, Tuckermann J, Schütz G, Pérez P. Glucocorticoid receptor is required for skin barrier competence. Endocrinology. 2008;149(3):1377–1388. [DOI] [PubMed] [Google Scholar]
- 121.Sevilla LM, Latorre V, Sanchis A, Pérez P. Epidermal inactivation of the glucocorticoid receptor triggers skin barrier defects and cutaneous inflammation. J Invest Dermatol. 2013;133(2):361–370. [DOI] [PubMed] [Google Scholar]
- 122.Pérez P, Page A, Bravo A, Del Río M, Giménez-Conti I, Budunova I, Slaga TJ, Jorcano JL. Altered skin development and impaired proliferative and inflammatory responses in transgenic mice overexpressing the glucocorticoid receptor. FASEB J. 2001;15(11):2030–2032. [DOI] [PubMed] [Google Scholar]
- 123.Reichardt HM, Kaestner KH, Tuckermann J, Kretz O, Wessely O, Bock R, Gass P, Schmid W, Herrlich P, Angel P, Schütz G. DNA binding of the glucocorticoid receptor is not essential for survival. Cell. 1998;93(4):531–541. [DOI] [PubMed] [Google Scholar]
- 124.Sevilla LM, Bayo P, Latorre V, Sanchis A, Pérez P. Glucocorticoid receptor regulates overlapping and differential gene subsets in developing and adult skin. Mol Endocrinol. 2010;24(11):2166–2178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Latorre V, Sevilla LM, Sanchis A, Pérez P. Selective ablation of glucocorticoid receptor in mouse keratinocytes increases susceptibility to skin tumorigenesis. J Invest Dermatol. 2013;133(12):2771–2779. [DOI] [PubMed] [Google Scholar]
- 126.Politch JA, Herrenkohl LR. Postnatal ACTH and corticosterone: effects on reproduction in mice. Physiol Behav. 1984;32(3):447–452. [DOI] [PubMed] [Google Scholar]
- 127.Dubey AK, Plant TM. A suppression of gonadotropin secretion by cortisol in castrated male rhesus monkeys (Macaca mulatta) mediated by the interruption of hypothalamic gonadotropin-releasing hormone release. Biol Reprod. 1985;33(2):423–431. [DOI] [PubMed] [Google Scholar]
- 128.Vogt MA, Pfeiffer N, Le Guisquet AM, Brandwein C, Brizard B, Gass P, Belzung C, Chourbaji S. May the use of different background strains “strain” the stress-related phenotype of GR+/− mice? Behav Brain Res. 2017;335:71–79. [DOI] [PubMed] [Google Scholar]
- 129.Presman DM, Ogara MF, Stortz M, Alvarez LD, Pooley JR, Schiltz RL, Grøntved L, Johnson TA, Mittelstadt PR, Ashwell JD, Ganesan S, Burton G, Levi V, Hager GL, Pecci A. Live cell imaging unveils multiple domain requirements for in vivo dimerization of the glucocorticoid receptor. PLoS Biol. 2014;12(3):e1001813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Jewell CM, Scoltock AB, Hamel BL, Yudt MR, Cidlowski JA. Complex human glucocorticoid receptor dim mutations define glucocorticoid induced apoptotic resistance in bone cells. Mol Endocrinol. 2012;26(2):244–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zhang J, Ge R, Matte-Martone C, Goodwin J, Shlomchik WD, Mamula MJ, Kooshkabadi A, Hardy MP, Geller D. Characterization of a novel gain of function glucocorticoid receptor knock-in mouse. J Biol Chem. 2009;284(10):6249–6259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Banuelos J, Shin SC, Lu NZ. A hotspot in the glucocorticoid receptor DNA-binding domain susceptible to loss of function mutation. Steroids. 2015;96:115–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Koper JW, van Rossum EF, van den Akker EL. Glucocorticoid receptor polymorphisms and haplotypes and their expression in health and disease. Steroids. 2014;92:62–73. [DOI] [PubMed] [Google Scholar]
- 134.Galliher-Beckley AJ, Williams JG, Cidlowski JA. Ligand-independent phosphorylation of the glucocorticoid receptor integrates cellular stress pathways with nuclear receptor signaling. Mol Cell Biol. 2011;31(23):4663–4675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.DeMayo FJ, Spencer TE. CRISPR bacon: a sizzling technique to generate genetically engineered pigs. Biol Reprod. 2014;91(3):79. [DOI] [PubMed] [Google Scholar]
- 136.Ma H, Marti-Gutierrez N, Park S-W, Wu J, Lee Y, Suzuki K, Koski A, Ji D, Hayama T, Ahmed R, Darby H, Van Dyken C, Li Y, Kang E, Park AR, Kim D, Kim S-T, Gong J, Gu Y, Xu X, Battaglia D, Krieg SA, Lee DM, Wu DH, Wolf DP, Heitner SB, Belmonte JCI, Amato P, Kim J-S, Kaul S, Mitalipov S. Correction of a pathogenic gene mutation in human embryos. Nature. 2017;548(7668):413–419. [DOI] [PubMed] [Google Scholar]