Abstract

In the past few years since our viewpoint on carbon nanoparticles was first published in 2013 (Kumar, V.; Toffoli, G.; Rizzolio, F. ACS Med. Chem. Lett.2013, 4 (11), 1012–1013), a considerable progress has been made in the area of synthesis, functionalization, and applications of fluorescent carbon nanoparticles (CNPs). This update aims to highlight some key points achieved in the last 4 years in the development of CNPs with a particular emphasis on the approaches to ameliorate clinical applications of CNPs as therapeutics, diagnostics, and theranostics agents.
Keywords: Precision medicine, cancer, carbon nanoparticles, imaging, drug delivery, theranostics
Cancer theranostics, a combination of both bioimaging diagnostics and cancer therapy, is an emerging field toward personalized medicine aiming at increasing the efficacy of the drug and limiting the associated adverse effects. Several nanotheranostics agents have been developed in the past, although with certain limitations. Carbon nanoparticle (CNP)-based theranostics systems have the potential to fill the present gap.1
For biological applications, it is imperative that the imaging probe be nontoxic. Ideally, it should exhibit fluorescence in the red or near-infrared (NIR) region in order to minimize the background fluorescence from the endogenous fluorophores present in the biological specimen and be photostable.2 A plethora of carbonaceous materials as precursors and various synthetic conditions have been used to yield high-quality CNPs with tunable fluorescence and surface properties.3,4 It has been pointed out that the mechanisms responsible for photoluminescence could refer to surface properties, chemical groups, charge, size, graphitization, or a combination. Because multiple approaches and starting materials were utilized, it is difficult to combine the results and formulate a leading theory. In 2016, Ding et al. published a paper in which the fluorescence of carbon dots (CDs) was tuned under controlled and comparable conditions to determine the luminescence mechanisms.5 It was discovered that the increased oxidation of the surface of CDs allows a red shift in emission peaks from 440 to 625 nm that is independent from particle size and different from the traditional inorganic semiconductor quantum dots. The quantum yield was similar to molecular fluorophores and, consequently, suitable for in vivo imaging. Unlike semiconductor quantum dots, these CDs were not toxic in vitro and in vivo. In vivo imaging of these CNPs shows that a clear fluorescence was observed in nude mice when injected subcutaneously or through tail vein. Of particular note is that all mice remain healthy after the injections, demonstrating the excellent biocompatibility of the CNPs.
CNPs could be used both as imaging and therapeutic materials. Among promising applications, photodynamic (PDT) and photothermal (PTT) therapies are the best candidates for the treatment of local and small tumors accessible from the outside due to the low penetration of light through tissues. In PDT, the drug is a photosensitizer that when exposed to a specific wavelength of light produces reactive oxygen species (ROS) such as low singlet oxygen to kill cells locally. Ge et al. prepared C dots using polythiophene phenyl acid (PPA) as precursors exhibiting a broad absorption band from visible to NIR region with emission maxima at 640 nm, high photostability, and a quantum yield of 2.3%.6 Different spectroscopic analyses suggested that the surface of these nanoparticles was carboxylated and sulfur-doped. Interestingly, fluorescence imaging of CNPs in HeLa cells showed intense red fluorescence even after 120 min of laser irradiation, while fluorescein isothiocyanate (FITC) becomes undetectable after 10 min. These particles were used for PTT and fluorescence and photoacoustic imaging in vivo on nude mice. After irradiation with a 671 nm laser, a significant suppression of tumor growth was observed. The fluorescence analysis of organs 24 h postinjection showed that CNPs accumulated mostly in the liver and tumor followed by lung, kidney, spleen, and heart. No obvious inflammation, cell necrosis, or apoptosis were observed in the heart, liver, spleen, lung, and kidney. The same group prepared CNPs from another conjugated polymer (polythiophenes derivates, PT2) with excellent 1O2 generation capability for both imaging (deep red emission) and PDT.7 These brilliant examples demonstrate the potential use of CNPs as theranostic particles for innovative biomedical applications.
The third aspect that we are pointing out is the use of CNPs as vehicle for drugs. As suggested by Hanahan and Weinberg, the tumor is an organ made by cancer cells and the surrounding stroma that “intelligently” interact to grow and invade other tissues.8 To understand the biology of the tumor, it is necessary to disentangle the relationship among cancer cells and tumor microenvironment that could be targeted by drugs. In this scenario, drug delivery vehicles with high biocompatibility and sensitivity toward tumor microenvironment could greatly enhance their translational prospects.
In 2016, Feng et al. developed a tumor microenvironment-responsive drug nanocarrier based on a charge convertible anionic polymer with dimethylmaleic acid (PAH/DMMA) and cisplatin(IV) prodrug-loaded CDs (CDs-Pt(IV)@PEG-(PAH/DMMA)) for cancer theranostics.9 The mildly acidic condition of tumor microenvironment triggered the conversion of PEG–PAH/DMMA from negative to positive charge. Due to electrostatic repulsion, the positively charged CDs-Pt(IV) were released from the polymer and exhibited higher uptake through negatively charged cell membrane, which ultimately enhanced activation of PT(IV) drug in the cytosol. This work provides a potential strategy to optimize and increase clinical application of CNPs for cancer therapeutics.
These studies show that CNPs are emerging as ideal candidates for theranostic applications (Figure 1). However, there are issues that need to be addressed before their clinical realization such as to better control surface properties, batch to batch reproducibility, purification, and characterization so that their interaction with biological systems and subsequent clinical outcome could be optimized. Though the quest is still ongoing, nevertheless, these studies harbor prospects for the development of new bioimaging probes and theranostics nanosystems based on carbon nanoparticles exhibiting high potential for their translation in biomedical laboratories and clinics in the very near future.
Figure 1.
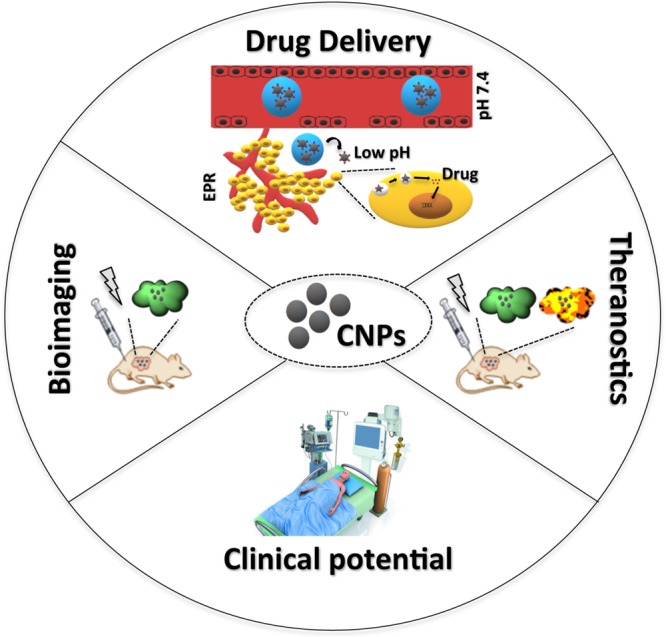
CNPs as ideal candidates for theranostic applications.
Acknowledgments
V.K. and F.R. are thankful to SERB (ECR/2016/001624) and AIRC (My First AIRC no. 1569) respectively, for funding.
Views expressed in this editorial are those of the authors and not necessarily the views of the ACS.
The authors declare no competing financial interest.
References
- Kumar V.; Toffoli G.; Rizzolio F. Fluorescent Carbon Nanoparticles in Medicine for Cancer Therapy. ACS Med. Chem. Lett. 2013, 4 (11), 1012–1013. 10.1021/ml400394a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayda S.; Hadla M.; Palazzolo S.; Kumar V.; Caligiuri I.; Ambrosi E.; Pontoglio E.; Agostini M.; Tuccinardi T.; Benedetti A.; Riello P.; Canzonieri V.; Corona G.; Toffoli G.; Rizzolio F. Bottom-up Synthesis of Carbon Nanoparticles with Higher Doxorubicin Efficacy. J. Controlled Release 2017, 248, 144–152. 10.1016/j.jconrel.2017.01.022. [DOI] [PubMed] [Google Scholar]
- Lim S. Y.; Shen W.; Gao Z. Carbon Quantum Dots and Their Applications. Chem. Soc. Rev. 2015, 44 (1), 362–381. 10.1039/C4CS00269E. [DOI] [PubMed] [Google Scholar]
- Dong Y.; Pang H.; Yang H. B.; Guo C.; Shao J.; Chi Y.; Li C. M.; Yu T. Carbon-Based Dots Co-Doped with Nitrogen and Sulfur for High Quantum Yield and Excitation-Independent Emission. Angew. Chem., Int. Ed. 2013, 52 (30), 7800–7804. 10.1002/anie.201301114. [DOI] [PubMed] [Google Scholar]
- Ding H.; Yu S.-B.; Wei J.-S.; Xiong H.-M. Full-Color Light-Emitting Carbon Dots with a Surface-State-Controlled Luminescence Mechanism. ACS Nano 2016, 10 (1), 484–491. 10.1021/acsnano.5b05406. [DOI] [PubMed] [Google Scholar]
- Ge J.; Jia Q.; Liu W.; Guo L.; Liu Q.; Lan M.; Zhang H.; Meng X.; Wang P. Red-Emissive Carbon Dots for Fluorescent, Photoacoustic, and Thermal Theranostics in Living Mice. Adv. Mater. 2015, 27 (28), 4169–4177. 10.1002/adma.201500323. [DOI] [PubMed] [Google Scholar]
- Ge J.; Lan M.; Zhou B.; Liu W.; Guo L.; Wang H.; Jia Q.; Niu G.; Huang X.; Zhou H.; Meng X.; Wang P.; Lee C.-S.; Zhang W.; Han X. A Graphene Quantum Dot Photodynamic Therapy Agent with High Singlet Oxygen Generation. Nat. Commun. 2014, 5, 4596. 10.1038/ncomms5596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanahan D.; Weinberg R. A. Hallmarks of Cancer: The next Generation. Cell 2011, 144 (5), 646–674. 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- Feng T.; Ai X.; An G.; Yang P.; Zhao Y. Charge-Convertible Carbon Dots for Imaging-Guided Drug Delivery with Enhanced in Vivo Cancer Therapeutic Efficiency. ACS Nano 2016, 10 (4), 4410–4420. 10.1021/acsnano.6b00043. [DOI] [PubMed] [Google Scholar]


