Abstract
The current study aimed to summarize the clinical manifestations and identify the epigenetic mechanisms of gastric mucosa associated lymphoid tissue (MALT) lymphoma, as well as evaluate the long-term effects of Helicobacter pylori (H. pylori) eradication. A total of 122 patients with marginal zone B-cell lymphoma of primary gastric MALT lymphoma were enrolled in the present study. The clinical manifestations of gastric MALT lymphoma, including symptoms, H. pylori state and endoscopic type, were summarized. The response to therapy was evaluated in patients that underwent H. pylori eradication. Survival analysis was estimated using the Kaplan-Meier method. The expression of microRNA-383 (miR-383) in tumor tissues and cell lines was determined using reverse transcription quantitative polymerase chain reaction. Furthermore, bioinformatic analyses, luciferase reporter assays. and western blot analysis identified zinc finger E-box binding homeobox 2 (ZEB2) as a direct target gene of miR-383. An MTT assay was used to examine the function of miR-383 and ZEB2 in MALT lymphoma. The clinical symptoms of patients with gastric MALT lymphoma were non-specific and included epigastric pain, abdominal discomfort and bleeding. The majority of endoscopic types were classified as ulcer, erosion and mucosa edema. The H. pylori infection rate was 79.5% (97/122) and a total of 47 patients underwent eradication therapy. Lymphoma remission was achieved in 93.6% (44/47) of patients and complete remission (CR) was achieved in 74.4% (35/47). The median follow-up time was 38 months (range, 10–132 months) and the median time taken to achieve CR was 4 months (range, 3–7 months). The estimated 3-year survival rate was 90.3% and the 5-year survival rate was 76.2%. Therefore, it was determined that patients with stage I or II gastric MALT lymphoma are able to undergo H. pylori eradication as a first-line treatment and that the survival rate of patients undergoing this treatment is high. Furthermore, it was determined that the mechanism by which miR-383 and ZEB2 contribute to MALT lymphoma progression is by the targeting of ZEB2 by miR-383, which inhibits the proliferation of cancer cells.
Keywords: gastric mucosa associated lymphoid tissue lymphoma, Helicobacter pylori eradication, survival, microRNA-383, zinc-finger E-box binding homeobox 2, epigenetic mechanism
Introduction
Numerous studies have investigated gastric mucosa associated lymphoid tissue (MALT) lymphoma. It accounts for 20–40% of extranodal lymphomas and 1–6% of all gastric malignancies (1–3). Gastric MALT lymphoma is defined as a distinct clinicopathological entity in the World Health Organization classification of malignant lymphomas (2,4). It is widely accepted that Helicobacter pylori (H. pylori) infection is the primary pathogenic factor stimulating the development of low-grade, marginal zone B-cell lymphoma of the stomach. In China, the incidence of H. pylori infection is high, as is the morbidity rate of patients with gastric MALT lymphoma (5). There have been few reports investigating the long-term effect of H. pylori eradication therapy. Previous studies have demonstrated that H. pylori eradication leads to the regression of early stage lymphoma (6–8). However, the traditional strategies used to treat gastric MALT lymphoma are surgery and chemotherapy. Long-term follow-up studies investigating the characteristics of gastric MALT lymphoma and the therapeutic effect of different treatments remain rare, particularly in China. Therefore, the current study was conducted on a large number of patients with gastric MALT lymphoma with the aim of summarizing the manifestations of the disease and evaluating the effect of H. pylori eradication on gastric MALT lymphoma.
microRNAs (miRNA) are a group of small noncoding RNAs 19–24 nucleotides long that regulate gene expression at the post-transcriptional level (9,10). They regulate the expression of their target mRNAs by binding to them (11,12), Previous studies have demonstrated that miRNAs and their epigenetic mechanisms serve a role in gastric lymphoma (13,14), however to the best of our knowledge, the role served by miRNAs in the pathogenesis of gastric MALT lymphoma remains unknown.
Zinc-finger E-box-binding homeobox (ZEB) 1 and ZEB2 are transcription factors for marginal zone B cells, which induce the epithelial-mesenchymal transition and promote metastasis in different types of cancer (15,16). In H. pylori-positive gastric diffuse large B-cell lymphoma, the inhibition of ZEB1 by miR-200 leads to a less aggressive form of the disease (17). However, it remains unclear whether ZEB2 serves a role in gastric MALT lymphoma and whether the regulation of ZEB2 is mediated by miRNA. The current study demonstrates that H. pylori eradication therapy is effective in the treatment of early gastric MALT lymphoma.
Materials and methods
Patients
A total of 122 patients (56 females and 66 males; median age, 56.5 years) with marginal zone B-cell lymphoma of the primary gastric MALT lymphoma (i.e. disease confined to the stomach with or without involvement of the paragastric lymph nodes and with no distant lymph node involvement) who were admitted to Tianjin Medical University General Hospital (Tianjin, China) were enrolled in the present study between January 2000 and January 2013. All patients had stage I1E (confined to the stomach) or II1E (involvement of paragastric lymph nodes) MALT lymphoma according to the Ann Arbor classification system modified by Musshoff (6,18,19). H. pylori status was determined using the rapid urease test, Giemsa staining or the 13C urea breath test (7). H. pylori infection was classed as positive if the result of ≥1 test was positive. Gastroendoscopy was performed in all the 122 patients. The endoscopic type of gastric lymphoma was classified as superficial, ulcerative or polypoid based on the classification reported previously (20). Endoscopic ultrasonography (EUS) was performed to evaluate the depth of tumor invasion and the degree of perigastric lymphadenopathy in 47 patients.
Initially, 47 patients accepted H. pylori eradication therapy. First-line treatment consisted of omeprazole (20 mg; Changzhou Siyao Pharmaceuticals Co., Ltd., Changzhou, China) or rabeprazole (10 mg; Zhuhai Rundu Pharmacy Ltd., Co., Zhuhai, China), amoxicillin (1,000 mg; Beijing Qin Wutian Pharmaceutical Co., Ltd., Beijing, China) or metronidazole (500 mg; Qin Wutian Pharmaceutical Co., Ltd.) and clarithromycin (500 mg; Hainan Puli Pharmacy Co., Ltd., Hainan, China) twice a day for 14 days. Second-line treatment consisted of omeprazole (20 mg) or rabeprazole (10 mg), levofloxacin (400 mg; Daiichi Saikyo Pharmaceutical Co., Ltd., Beijing, China) or furazolidone (100 mg; Shuanghe Pharmaceutical Co., Ltd., Beijing, China) and clarithromycin (500 mg) or amoxicillin (1,000 mg) twice a day for 14 days. H. pylori status, endoscopic controls and biopsies were performed 1 month following H. pylori eradication and were continued every 3 months in the first year, 6 months in the second year and then once a year from the third year onwards.
The present study was approved by the Ethics Committee of Tianjin Medical University General Hospital (Tianjin, China) and written informed consent was obtained from all participants.
Remission evaluation following H. pylori eradication
Complete remission (CR) was defined as no macroscopic findings of lymphoma and negative histology in two subsequent follow-up investigations. Partial remission (PR) was defined as normalization or reduction of macroscopic findings, histological signs of lymphoma regression and no signs of disease progression. Stable disease (SD) was characterized by unmodified macroscopy and/or unmodified histology. Progressive disease (PD) was defined by worsening of macroscopic findings, dissemination of gastric MALT lymphoma or transformation into DLBCL. Relapse was defined as persistent histological confirmation of lymphoma following documentation of CR (4).
Cell lines and reagents
HEK-293T cells, Daudi and U2932 cells (American Type Culture Collection, Manassas, VA, USA) were cultured in Dulbecco's modified Eagles medium and NCU-L-4 and Raji cells (American Type Culture Collection, ATCC; Manassas, VA, USA) were cultured in RPMI-1640 medium (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) with 10% fetal bovine serum (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C with 5% CO2.
Transfection of cells with miRs
Synthetic hsa-miR-383, scrambled control RNA (miR-control), hsa-miR-383 inhibitor or negative control RNA (miR-NC) were purchased from Shanghai GeneChem Co., Ltd. (Shanghai, China). A total of 3×105 cells were seeded in each well of 6-well plates and transfected with 2 µg ZEB2 overexpression construct (Shanghai GenePharma Co., Ltd., Shanghai, China), using Lipofectamine™ 2000 (Invitrogen; Thermo Fisher Scientific, Inc.). Transfected cells were harvested for RNA or protein extraction 1–2 days following treatment.
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
Total RNA containing miRNA was extracted from tissues using a miRNeasy Mini kit (Qiagen China Co., Ltd., Shanghai, China). cDNA was synthesized using a miScript Reverse Transcription kit (Qiagen China Co., Ltd.), following the manufacturer's instructions. Levels of miRNA expression were analyzed by quantification using SYBR Green miRNA RT-qPCR on a 7500 Fast Real-Time system (both from Applied Biosystems; Thermo Fisher Scientific, Inc.). U6 was used for normalization in miRNA studies. Experiments were performed in triplicate and the results were expressed as the mean ± standard deviation. The genes expression was normalized against that of GAPDH and relative fold changes were calculated using the 2−ΔΔCq (21). The amplification was performed as follows: Denaturation at 94°C for 20 sec, annealing at 60°C for 45 sec followed by an extension at 72°C for 30 sec for 40 cycles. The following primers were used: GAPDH forward, 5′-GAGAAGTATGACAACAGCCTC-3′ and reverse, 5′-ATGGACTGTGGTCATGAGTC-3′; and ZEB2 forward, 5′-GTGAGGTCTCCCCGAGGTGT-3′ and reverse, 5′-ATTAGGGTGTGTGCATGATGAAT-3′.
Luciferase reporter assay
Luciferase assays in 293T cells and U2932 cells were performed with 0.02 mg firefly luciferase reporter construct with the wild-type ZEB2 pCI-neo-RL-ZEB2 (plasmid #35536) or mutant ZEB2 pCI-neo-RL-ZEB2 200b mut (plasmid #35538) (both from Addgene, Inc., Cambridge, MA, USA) and 0.02 mg control vector containing Renilla luciferase pRL-CMV (Promega Corporation, Madison, WI, USA) in a 24-well plate. Transfection was conducted using Lipofectamine 2000. HEK-293T cells and NCU-L-4 cells were co-transfected with miR-control, miR-383, miR-NC and miR-383 inhibitor at a final concentration of 100 nM. Luciferase activity was measured using a Luciferase Assay system (Promega Corporation). A total of 48 h post transfection, the activity of firefly luciferase was normalized to that of Renilla luciferase. Each experiment was performed in triplicate.
Western blot analysis
Following transfection, cells were cultured for 2 days and protein was extracted using radioimmunoprecipitation assay lysis buffer (Promega Corporation). Protein concentration was determined using the BCA protein assay kit (Thermo Fisher Scientific, Inc.). A total of 40 µg protein from each group was loaded per lane and underwent 10% SDS-polyacrylamide gel electrophoresis. Subsequently, proteins were transferred onto a PVDF membrane (Merck KGaA). Following blocking with 5% non-fat dried milk at room temperature for 30 min, membranes were incubated with primary antibodies against ZEB2 (cat no. ab13222; 1:1,000; Abcam, Cambridge, UK) and GAPDH (cat no. SC81545; 1:1,000; Santa Cruz Biotechnology, Inc., Dallas, TX, USA) overnight at 4°C. Following washing with TBST, membranes were incubated with peroxidase-conjugated goat anti-rabbit secondary antibodies (cat no. sc-2004, 1:5,000; Santa Cruz Biotechnology, Inc.) at room temperature for 1 h. Immunoreactive bands were analyzed using the ECL chemiluminescence system (Thermo Fisher Scientific, Inc.) and quantified using ImageI software (version 2x; Rawak Software, Inc., Stuttgart, Germany).
MTT assay
An MTT assay was performed to determine the viability of cancer cells. The transfected cells were seeded in triplicate in a 96-well plate, at a concentration of 500 cells/well. At 24, 48, 72 and 96 h following transfection, 20 µl MTT (5 mg/ml; Sigma-Aldrich; Merck KGaA) was added to each well and the cells were further incubated for 4 h at 37°C prior to harvest. A total of 150 µl dimethyl sulfoxide was used to terminate the reaction and absorbance was measured at a wavelength of 570 nm using a microplate reader.
Statistical analysis
Statistical analysis was conducted using the SPSS 17.0 software (SPSS Inc., Chicago, IL, USA) and differences between groups were assessed using Student's t-test. Survival time was calculated according to the revised criteria for malignant lymphoma from the time of diagnosis to that of mortality from any cause or lost follow-up (22). Follow-up was calculated as the median time to censoring using reverse Kaplan-Meier analysis and survival curves were estimated using the Kaplan-Meier method. RT-qPCR and the luciferase reporter assay were performed in triplicate. Data are presented as the mean ± standard deviation and P<0.05 was considered to indicate a statistically significant difference.
Results
Patient characteristics
The characteristics of the patients participating in the current study are presented in Table I. The median age of initial diagnosis was 56.5 years (range, 19–84 years) and 54.1% of the patients were male. All patients exhibited gastrointestinal symptoms. The most common symptom experienced was epigastric pain (39.4%), however abdominal discomfort (23.8%) and bleeding (15.6%) were also common. MALT lymphoma was most often localized in the antrum of patients (40.2%) and was determined as multifocal (situated at two different sites out of the antrum, body and fundus) in 26.2% of patients. Results indicated that from all the 122 patients who underwent gastroendoscopy, 46.7% of tumors were classified as ulcerative type, 20.5% as superficial and 32.8% as polypoid. Among the 122 patients, 97 of them tested positive for H. pylori. With the exception of those 75 patients who decided to undergo the surgery or the chemotherapy, 47 received the H. pylori eradication treatment following diagnosis (Table I).
Table I.
Clinical manifestations of gastric mucosa associated lymphoid tissue lymphoma at diagnosis.
| Features | n (%) |
|---|---|
| Age (years) | |
| ≥60 | 52 (42.6) |
| <60 | 70 (57.4) |
| Sex | |
| Female | 56 (45.9) |
| Male | 66 (54.1) |
| Symptoms | |
| Epigastric pain | 48 (39.4) |
| Abdominal discomfort | 29 (23.8) |
| Bleeding | 19 (15.6) |
| Abdominal distention | 11 (9.0) |
| Poor appetite | 8 (6.5) |
| Nausea and vomiting | 7 (5.7) |
| Helicobacter pylori status | |
| Positive | 97 (79.5) |
| Negative | 25 (20.5) |
| Gastric location | |
| Antrum | 49 (40.2) |
| Body | 27 (22.1) |
| Fudus | 14 (11.5) |
| Multifocal | 32 (26.2) |
| Endoscopic type | |
| Ulcerative | 57 (46.7) |
| Superficial | 25 (20.5) |
| Polypoid | 40 (32.8) |
Treatment outcomes
The clinical course following eradication of H. pylori is summarized in Fig. 1. A total of 47 patients received H. pylori eradication treatment. All these patients were infected with H. pylori, their median age was 52 years old (range, 24–69 years old) and they all underwent EUS. Among the 47 patients, the depth of lymphoma infiltration in the gastric wall was: The mucosa in 13, subcutaneous in 14, muscularis propria in 13 and the entire gastric wall in 7 patients. The paragastric lymph nodes were involved in the case of 1 patient with lymphoma that had infiltrated the entire gastric wall. Overall, lymphoma remission was achieved in 44/47 (93.6%) patients and 35/47 patients achieved CR (74.4%). The median time taken to achieve CR was 4 months (range, 3–7 months). Among the 9 patients that achieved PR, 8 chose to undergo chemotherapy and 1 patient experienced relapse. Following relapse, this patient underwent further H. pylori eradication and achieved CR in 4 months. A total of 2 out of 47 patients continued to exhibit SD following H. pylori eradication and subsequently underwent chemotherapy 6 months later. One patient presented with DLBCL following H. pylori eradication treatment and subsequently underwent a gastrectomy (Fig. 1).
Figure 1.
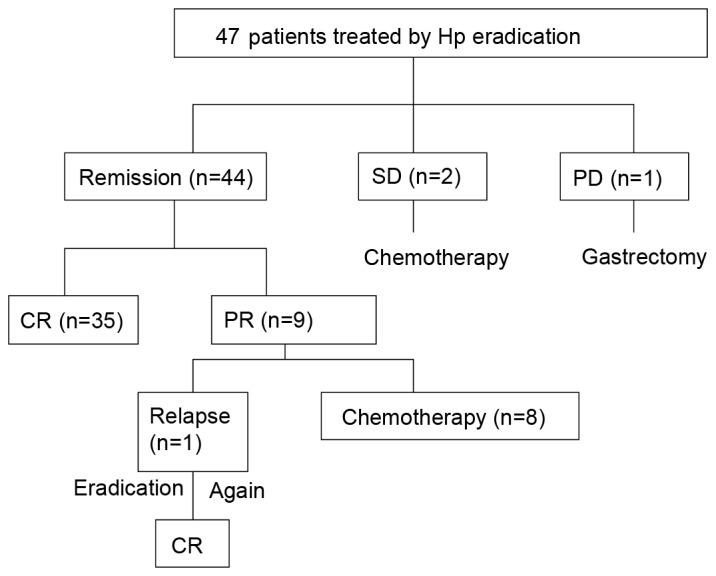
Response and follow-up of the 47 patients that accepted eradication. The clinical course following eradication of H. pylori is presented. Hp, Helicobacter pylori; SD, stable disease; PD, progressive disease; CR, complete remission; PR, partial remission.
Overall survival
All mortalities, regardless of cause, were recorded. The results presented are a conservative estimate of survival in patients with gastric MALT lymphoma. The median follow-up time was 38 months (range, 10–132 months; Kaplan-Meier analysis; Fig. 2). Among the 47 patients, 7 succumbed. One patient with PD (DLBC lymphoma) succumbed 20 months following surgery. Another patient with SD also succumbed 36 months following surgery due to a progression of lymphoma. Other causes of mortality included coronary syndrome (n=2), stroke (n=2) and chest infection (n=1). The estimated 3-year survival rate was 90.3% and the 5-year survival rate was 76.2%.
Figure 2.
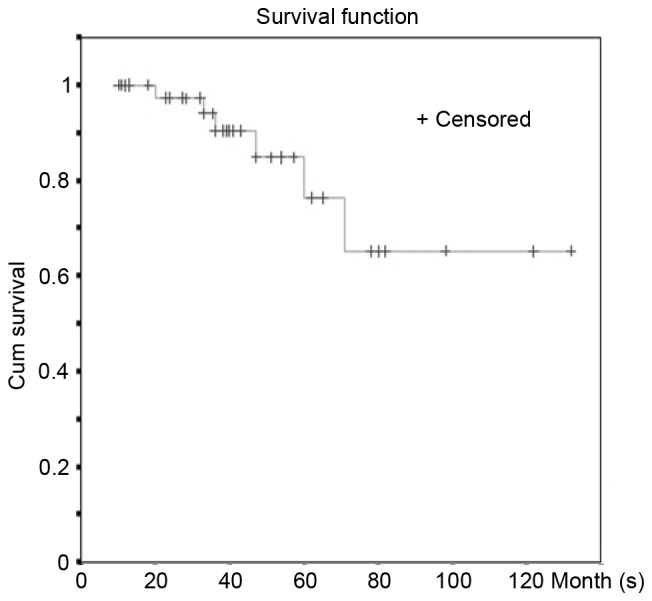
Kaplan-Meier analysis of 47 patients with gastric MALT lymphoma following Helicobacter pylori eradication. Survival time was calculated using the Kaplan-Meier analysis. MALT, mucosa associated lymphoid tissue; Cum survival, cumulative survival.
Expression of miR-383 is downregulated in gastric MALT lymphoma and cell lines
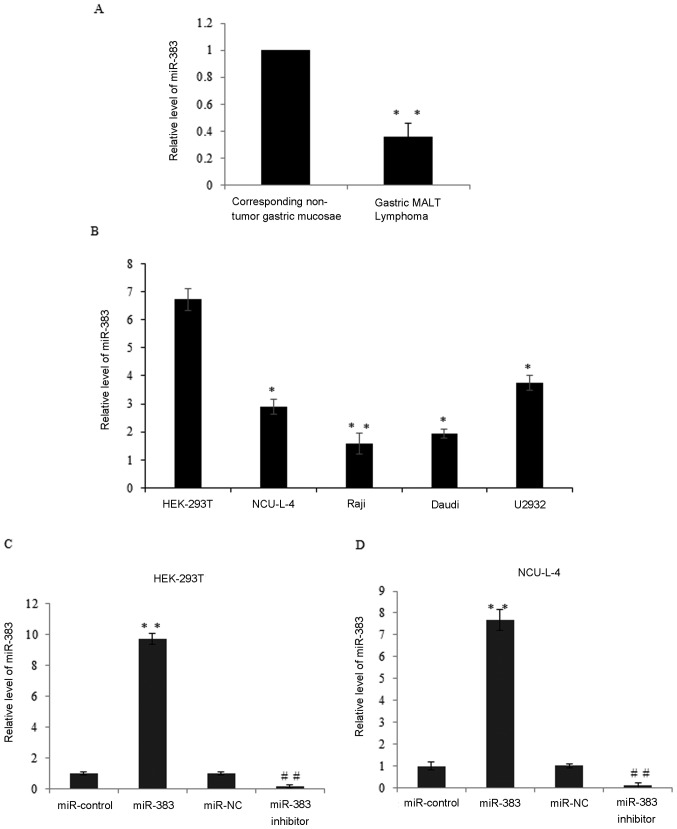
miR-383 is a tumor suppressor in various types of cancer, therefore the present study was conducted to identify whether miR-383 contributes to the progression of gastric MALT lymphoma. RT-qPCR was conducted to measure miR-383 expression in 47 cases of gastric MALT lymphoma. miR-383 expression in gastric MALT lymphoma lesions was significantly lower than in the corresponding non-tumor gastric mucosae (P<0.01; Fig. 3A). miR-383 expression in malignant lymphoma cell lines was also examined using RT-qPCR. The results demonstrated that miR-383 was significantly downregulated in the NCU-L-4, Raji, Daudi lymphoma and U2932 cells compared with HEK-293T cells (P<0.05; Fig. 3B). To identify the role of miR-383 in gastric MALT lymphoma, the expression of miR-383 was measured in HEK-293T and NCU-L-4 cells transfected with miR-control, miR-383, miR-NC and miR-383 inhibitor (Fig. 3C and D). The overexpression or knockdown efficiency was demonstrated. As compared with the miR-control, miR-183 significantly increased the expression levels of miR-183 (P<0.05). Furthermore, compared with the miR-NC, miR-383 inhibitor significantly reduced the miR-183 expression levels (P<0.05) in HEK-293T cells and NCU-L-4 cells.
Figure 3.
Expression of miR-383 is downregulated in gastric MALT lymphoma and cell lines. (A) Expression of miR-383 in 47 cases of gastric MALT lymphoma and corresponding non-tumor gastric mucosae by RT-qPCR. **P<0.01 vs. corresponding non-tumor gastric mucosae. (B) Relative miR-383 expression was detected in the NCU-L-4, Raji, Daudi lymphoma and U2932 cells compared with HEK-293T cells by RT-qPCR; U6 was used as an internal control. *P<0.05, **P<0.01 vs. HEK-293T. (C) Relative miR-383 expression was detected in miR-control, miR-383, miR-NC and miR-383 inhibitor transfected HEK-293T cells by RT-qPCR. **P<0.01 vs. miR-control and ##P<0.01 vs. miR-NC. (D) Relative miR-383 expression was detected in miR-control, miR-383, miR-NC and miR-383 inhibitor transfected NCU-L-4 cells by RT-qPCR. **P<0.01 vs. miR-control and ##P<0.01 vs. miR-NC. Results are presented as the mean ± standard deviation. miR-383, microRNA-383; MALT, mucosa associated lymphoid tissue; RT-qPCR, reverse transcription quantitative polymerase chain reaction.
Validation of ZEB2 as a direct target of miR-383
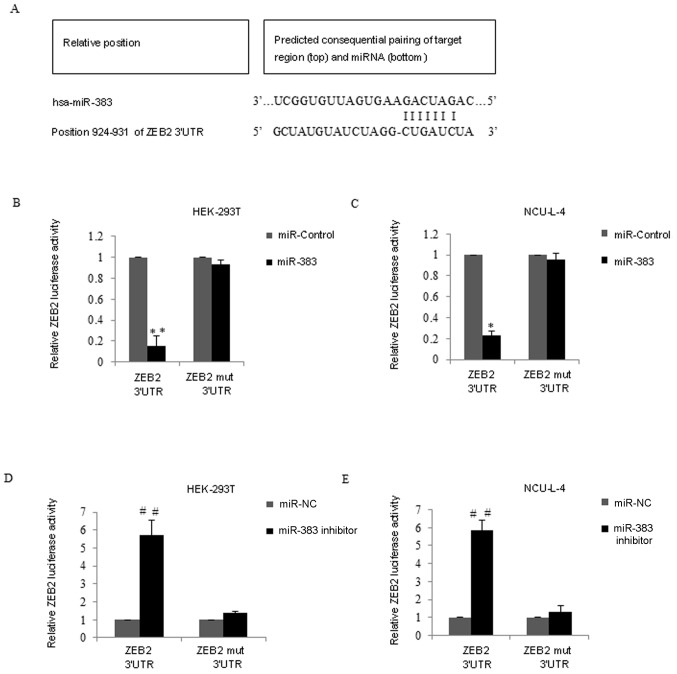
It was hypothesized that miR-383 may be a tumor suppressor in gastric MALT lymphoma therefore, the target gene of miR-383 was screened using TargetScan 6.2 (http://www.targetscan.org). ZEB2 was considered to be a potential target with miR-383-binding sites (Fig. 4A). To confirm this possibility, the ZEB2 3′UTR wild-type (wt) or ZEB2 mutant site (ZEB2 3′ UTR-mut) was cotransfected with miR-383 mimics into HEK-293T cells (Fig. 4B) and NCU-L-4 cells (Fig. 4C). Compared with the miR-control group, the luciferase reporter activity of the cells containing the ZEB2-UTR-wt was significantly decreased in HEK-293T and NCU-L-4 miR-383 transfected cells (P<0.01; Fig. 4B; P<0.05; Fig. 4C). However, the luciferase activity of the ZEB2 3′-UTR-mut reporter was unaffected by the simultaneous transfection of miR-383 in the two cell lines. By contrast, co-transfection of ZEB2 3′UTR wt and miR-383 inhibitor into HEK-293T cells and NCU-L-4 cells resulted in significantly higher luciferase activity compared with the miR-NC groups (P<0.01) however, ZEB2 3′-UTR-mut reporter activity was unaffected following transfection with miR-383 inhibitor (Fig. 4D and E).
Figure 4.
Validation of ZEB2 as a direct target of miR-383. (A) A putative miR-383-binding site in ZEB2 3′ UTR was determined using bioinformatic analyses. (B) Luciferase report assays of HEK-293T cells. Relative luciferase activity was analyzed following the co-transfection of ZEB2 3′UTR-wt or ZEB2 3′ UTR-mut vector (mock reporter plasmid) with miR-control or miR-383 mimics into HEK-293T cells. The luciferase activity was normalized to Renilla luciferase activity. (C) The relative luciferase reporter assays were performed in NCU-L-4 cells. (D) Luciferase report assays of HEK-293T cells. Relative luciferase activity was analyzed following the co-transfection of ZEB2 3′UTR wild-type (wt) or ZEB2 mutant site (ZEB2 3′ UTR-mut) vector (mock reporter plasmid) with miR-NC or miR-383 inhibitor into HEK-293T cells. Luciferase activity was normalized to Renilla luciferase activity. (E) Luciferase reporter assays were performed in NCU-L-4 cells. Results are presented as the mean ± standard deviation. miR-383, microRNA-383; ZEB2, zinc-finger E-box-binding homeobox 2; UTR, untranslated region; wt, wild type; mut, mutant; MALT, mucosa associated lymphoid tissue; RT-qPCR, reverse transcription quantitative polymerase chain reaction; NC, negative control. *P<0.05 and **P<0.01, vs. miR-control and ##P<0.01 vs. miR-NC.
miR-383 inhibits proliferation through the repression of ZEB2 expression
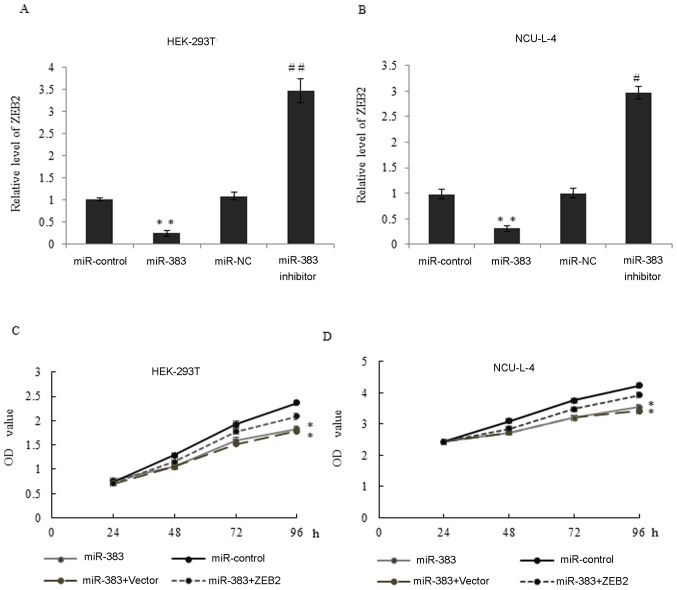
Consistent with the results of the luciferase report assay, RT-qPCR was used to measure expression of ZEB2 mRNA in HEK-293T and NCU-L-4 cells transfected with miR-control, miR-383, miR-NC and miR-383 inhibitor (Fig. 5A and B). Compared with the control and NC groups, overexpression of miR-383 significantly inhibited ZEB2 expression (both P<0.01; Fig. 5A and B). By contrast, inhibition of miR-383 significantly increased ZEB2 expression in HEK-293T and NCU-L-4 cells (P<0.01; Fig. 5A; P<0.05; Fig. 5B). Western blot analysis was also used to measure levels of ZEB2 mRNA in the two cell lines and a similar trend was observed (data not shown).
Figure 5.
miR-383 inhibits proliferation through the repression of ZEB2 expression. (A) The expression of ZEB2 mRNA in HEK-293T cells transfected with miR-control, miR-383, miR-NC or miR-383 inhibitor was measured. *P<0.05, **P<0.01, vs. miR-control. #P<0.05, ##P<0.01 vs. miR-NC. (B) The level of ZEB2 mRNA expression in NCU-L-4 cells transfected with miR-control, miR-383, miR-NC or miR-383 inhibitor was measured. *P<0.05, **P<0.01, vs. miR-control. #P<0.05, ##P<0.01 vs. miR-NC. (C) Cell viability was assessed in miR-control, miR-383, miR-383 plus vector and miR-383 + ZEB2 overexpression construct transfected HEK-293T cells by MTT assay at 0, 24, 48, 72 and 96 h time points. *P<0.05, **P<0.01 vs. miR-control. (D) The relative MTT assay was performed in NCU-L-4 cells. Results are presented at the mean ± standard deviation. miR-383, microRNA-383; ZEB2, zinc-finger E-box-binding homeobox 2; NC, negative control.
To examine whether miR-383 regulates cancer cell proliferation by suppressing ZEB2 in vitro, an MTT assay was performed in HEK-293T and NCU-L-4 cells transfected with miR-control, miR-383, miR-383+vector and miR-383+ZEB2 overexpression vector. The overexpression of miR-383 significantly inhibited the viability of cells compared with the miR-control group and this may also be blocked by the additional ZEB2 overexpression (Fig. 5C and D). Therefore, miR-383 may inhibit the proliferation of cancer cell lines by suppressing ZEB2 expression in vitro.
The expression of ZEB2 is upregulated and is negatively associated with miR-383 expression
The expression of ZEB2 mRNA in patients with gastric MALT lymphoma was measured (Fig. 6A). The expression of ZEB2 mRNA was significantly increased in gastric MALT lymphoma tumor tissues compared with corresponding non-tumor tissues (P<0.01; Fig. 6A). In addition, the expression of ZEB2 mRNA in NCU-L-4, Raji, Daudi lymphoma and U2932 cells was measured and compared with expression in HEK-293T cells. The expression of ZEB2 mRNA was also increased in those cancer cell lines compared with the HEK-293T cells (all P<0.05; Fig. 6B). Furthermore, there was an inverse association between the expression of ZEB2 mRNA and miR-383 in clinical gastric MALT lymphoma tissues (Fig. 6C). These results indicate that miR-383 may directly target ZEB2 by interacting with its 3′-UTR binding site, thus regulating the endogenous expression of ZEB2 at the transcriptional and translational levels.
Figure 6.
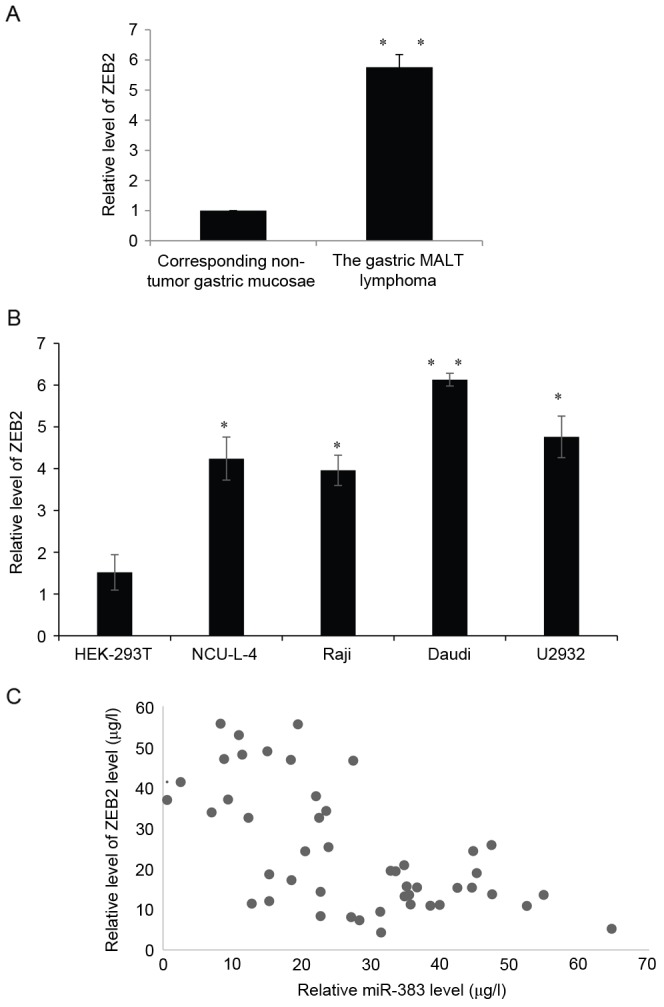
The expression of ZEB2 is upregulated and negatively correlated with miR-383 expression. (A) Expression of ZEB2 in 47 cases of gastric MALT lymphoma and corresponding non-tumor gastric mucosae by RT-qPCR analysis. **P<0.01 vs. corresponding non-tumor gastric mucosae. (B) Relative ZEB2 expression was detected in NCU-L-4, Raji, Daudi lymphoma and U2932 cell lines compared with HEK-293T cells by RT-qPCR analysis; U6 was used as an internal control. *P<0.05, **P<0.01 vs. HEK-293T. (C) The association between miR-383 and ZEB2 expression in patients with gastric MALT lymphoma was assessed. All results are presented as the mean ± standard deviation. miR-383, microRNA-383; ZEB2, zinc-finger E-box-binding homeobox 2; MALT, mucosa associated lymphoid tissue; RT-qPCR, reverse transcription quantitative polymerase chain reaction.
Discussion
Previous studies have demonstrated that H. pylori infection serves a role in the pathogenesis of MALT lymphomas of the gastrointestinal tract. Asenjo and Gisbert (23) indicated that the infection rate of H. pylori in patients with gastric MALT lymphoma patients was 90% compared with the 79.5% infection rate observed in the present study. This difference between rates may be due to the different dietary habits of patients in the respective countries in which the studies were conducted.
H. pylori infection stimulates immune lymphocytes in the gastric mucosa and induces the formation of MALTs, from which MALT lymphomas of B-cell origin develop. Cytotoxin-associated gene A may be translocated into human B lymphocytes through the bacterial type-IV secretion system. H. pylori colonization induces systemic and mucosal immune responses (24). Bacterial colonization of the gastric mucosa triggers lymphoid infiltration (25) and the formation of acquired MALTs. H. pylori infection induces and sustains an actively proliferating B-cell population via direct and indirect immunologic stimulation and infiltrating T cells serve an important role in the development of MALT lymphomas (24,26). Thus, gastric MALT lymphoma may be the only malignant tumor induced by bacteria infection.
The diagnosis of gastric MALT lymphoma depends primarily on endoscopic and pathological examination. However, it is difficult to distinguish between gastric MALT lymphoma, gastric ulcer, gastritis and gastric cancer as they exhibit similar endoscopic characteristics and sometimes the pathologic results reveal inflammation, which may be a result of insufficient or inadequate biopsy materials. The clinical symptoms of gastric MALT lymphoma are non-specific and include epigastric pain, abdominal discomfort and bleeding. Lymphoma is often localized in the antrum, body and multifocal sites of the stomach and the common endoscopic types are ulcer, erosion and mucosa edema (27). A large number of biopsies should be taken from visible lesions and normal mucosa. In cases where gastric MALT lymphoma is suspected but insufficient or inadequate initial biopsy materials have been obtained, an EUS and endoscopic mucosa resection may be required (28). Previous studies have recommended examining patients for signs of translocation t (11;18) (4,27), however, this testing is not common in the institution in which the current study was performed. The fact that none of the patients participating in the current study were tested for translocation t (11;18) was a limitation.
Insufficient knowledge of the association between H. pylori and gastric MALT lymphoma and patient concerns leads to a large proportion of patients initially choosing surgery or chemotherapy to treat gastric MALT lymphoma, which was the case in the present study. Extranodal marginal zone lymphoma of gastric MALT lymphoma has been defined as a distinct clinicopathological entity in the World Health Organization's classification of malignant lymphomas (2). Early low-grade gastric MALT lymphoma rarely transforms into secondary DLBCL or progresses beyond the stomach (3,2). Without transformation or progression, patients with early low-grade gastric MALT lymphoma rarely succumb and should be treated conservatively (29). Furthermore, it has been demonstrated that H. pylori eradication may induce the regression of low-grade gastric MALT lymphoma in the majority of patients (27). In the present study, CR was achieved in 74.4% of the patients who underwent H. pylori eradication. It is essential to evaluate the lymphoma response following effective H. pylori eradication. CR is usually achieved within 6–12 months following eradication, however in some cases, this may be delayed up to 24–72 months (7). In the present study, the median time taken to achieve CR was 4 months (range, 3–7 months), which is shorter than the length of time cited in previous studies (3,8). This may have been due to patients opting to undergo chemotherapy or surgery when CR was not achieved 6 months following eradication, rather than maintaining a ‘watch and wait’ strategy. The 3-year survival rate was 90.3% and the 5-year survival rate was 76.2%. This is consistent with previous studies conducted in different countries (3,5,6). Recently, it has been demonstrated that patients with H. pylori-negative gastric MALT lymphomas achieve CR following exclusive anti-H. pylori treatment (30).
The role of miRNAs and epigenetic mechanisms in primary gastric mucosa-associated lymphoid tissue lymphoma are being increasingly studied to improve understanding of its pathogenesis, prognosis and treatment (31,32). miR-383 has been identified as a tumor suppressor and potential prognostic biomarker in human non-small cell lung cancer (33). miR-383 inhibits anchorage-independent growth and induces glioma cell cycle arrest by targeting cyclin D1 (34). It is also associated with male infertility and inhibits testicular embryonal carcinoma cell proliferation by targeting interferon regulatory factor 1 (35). However, it has remains unknown whether miR-138 serves a function in the progression of gastric MALT lymphomas and what its target gene is.
To the best of our knowledge, the current study is the first to investigate the function of miR-383 and demonstrate that miR-383 inhibits ZEB2 in H. pylori-positive gastric MALT lymphoma. In the present study, 1 patient relapsed 3 months following achievement of PR and eradication treatment. This patient received further H. pylori eradication treatment and achieved CR in 4 months. The mechanism of lymphoma relapse may be associated with the re-infection of H. pylori, however this remains to be determined.
In conclusion, the results of the current study demonstrate that patients with stage I or II gastric MALT lymphoma may receive H. pylori eradication as a first-line treatment. Patients achieving CR following H. pylori eradication may be managed by follow-up endoscopy with multiple biopsies every 1–2 years. Other treatments, including chemotherapy and surgery, may be adopted if there is no change following eradication. Furthermore, the inhibition of ZEB2 by miR-383 contributes to a less aggressive form of the disease.
References
- 1.Nakamura S, Matsumoto T, Iida M, Yao T, Tsuneyoshi M. Primary gastrointestinal lymphoma in Japan: A clinicopathologic analysis of 455 patients with special reference to its time trends. Cancer. 2003;97:2462–2473. doi: 10.1002/cncr.11415. [DOI] [PubMed] [Google Scholar]
- 2.Sabattini E, Bacci F, Sagramoso C, Pileri SA. WHO classification of tumours of haematopoietic and lymphoid tissues in 2008: An overview. Pathologica. 2010;102:83–87. [PubMed] [Google Scholar]
- 3.Nakamura S, Sugiyama T, Matsumoto T, Iijima K, Ono S, Tajika M, Tari A, Kitadai Y, Matsumoto H, Nagaya T, et al. Long-term clinical outcome of gastric MALT lymphoma after eradication of Helicobacter pylori: A multicentre cohort follow-up study of 420 patients in Japan. Gut. 2012;61:507–513. doi: 10.1136/gutjnl-2011-300495. [DOI] [PubMed] [Google Scholar]
- 4.Ruskoné-Fourmestraux A, Fischbach W, Aleman BM, Boot H, Du MQ, Megraud F, Montalban C, Raderer M, Savio A, Wotherspoon A, EGILS group EGILS consensus report. Gastric extranodal marginal zone B-cell lymphoma of MALT. Gut. 2011;60:747–758. doi: 10.1136/gut.2010.224949. [DOI] [PubMed] [Google Scholar]
- 5.Cheng KS, Chan YK, Yeung YW. Treatment outcome of gastric extra-nodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue type in the Hong Kong Chinese population: A single centre experience. J Gastrointest Oncol. 2013;4:198–202. doi: 10.3978/j.issn.2078-6891.2013.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Andriani A, Miedico A, Tedeschi L, Patti C, Di Raimondo F, Leone M, Schinocca L, Romanelli A, Bonanno G, Linea C, et al. Management and long-term follow-up of early stage H. pylori-associated gastric MALT-lymphoma in clinical practice: An Italian, multicentre study. Dig Liver Dis. 2009;41:467–473. doi: 10.1016/j.dld.2008.09.009. [DOI] [PubMed] [Google Scholar]
- 7.Fischbach W, Goebeler-Kolve ME, Dragosics B, Greiner A, Stolte M. Long term outcome of patients with gastric marginal zone B cell lymphoma of mucosa associated lymphoid tissue (MALT) following exclusive Helicobacter pylori eradication therapy: Experience from a large prospective series. Gut. 2004;53:34–37. doi: 10.1136/gut.53.1.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wündisch T, Thiede C, Morgner A, Dempfle A, Günther A, Liu H, Ye H, Du MQ, Kim TD, Bayerdörffer E, et al. Long-term follow-up of gastric MALT lymphoma after Helicobacter pylori eradication. J Clin Oncol. 2005;23:8018–8024. doi: 10.1200/JCO.2005.02.3903. [DOI] [PubMed] [Google Scholar]
- 9.Ke XS, Liu CM, Liu DP, Liang CC. MicroRNAs: Key participants in gene regulatory networks. Curr Opin Chem Biol. 2003;7:516–523. doi: 10.1016/S1367-5931(03)00075-9. [DOI] [PubMed] [Google Scholar]
- 10.Herranz H, Cohen SM. MicroRNAs and gene regulatory networks: Managing the impact of noise in biological systems. Genes Dev. 2010;24:1339–1344. doi: 10.1101/gad.1937010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lytle JR, Yario TA, Steitz JA. Target mRNAs are repressed as efficiently by microRNA-binding sites in the 5′ UTR as in the 3′ UTR; Proc Natl Acad Sci USA; 2007; pp. 9667–9672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Standart N, Jackson RJ. MicroRNAs repress translation of m7Gppp-capped target mRNAs in vitro by inhibiting initiation and promoting deadenylation. Genes Dev. 2007;21:1975–1982. doi: 10.1101/gad.1591507. [DOI] [PubMed] [Google Scholar]
- 13.Matsushima K, Isomoto H, Inoue N, Nakayama T, Hayashi T, Nakayama M, Nakao K, Hirayama T, Kohno S. MicroRNA signatures in Helicobacter pylori-infected gastric mucosa. Int J Cancer. 2011;128:361–370. doi: 10.1002/ijc.25348. [DOI] [PubMed] [Google Scholar]
- 14.Gebauer N, Kuba J, Senft A, Schillert A, Bernard V, Thorns C. MicroRNA-150 is up-regulated in extranodal marginal zone lymphoma of MALT type. Cancer Genomics Proteomics. 2014;11:51–56. [PubMed] [Google Scholar]
- 15.Brabletz S, Brabletz T. The ZEB/miR-200 feedback loop - a motor of cellular plasticity in development and cancer? EMBO Rep. 2010;11:670–677. doi: 10.1038/embor.2010.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Si W, Huang W, Zheng Y, Yang Y, Liu X, Shan L, Zhou X, Wang Y, Su D, Gao J, et al. Dysfunction of the reciprocal feedback loop between GATA3- and ZEB2-nucleated repression programs contributes to breast cancer metastasis. Cancer Cell. 2015;27:822–836. doi: 10.1016/j.ccell.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 17.Huang WT, Kuo SH, Cheng AL, Lin CW. Inhibition of ZEB1 by miR-200 characterizes Helicobacter pylori-positive gastric diffuse large B-cell lymphoma with a less aggressive behavior. Mod Pathol. 2014;27:1116–1125. doi: 10.1038/modpathol.2013.229. [DOI] [PubMed] [Google Scholar]
- 18.Bartl R, Frisch B, Burkhardt R, Kettner G, Mahl G, Fateh-Moghadam A, Sund M. Assessment of bone marrow histology in the malignant lymphomas (non-Hodgkin's): Correlation with clinical factors for diagnosis, prognosis, classification and staging. Br J Haematol. 1982;51:511–530. doi: 10.1111/j.1365-2141.1982.tb02815.x. [DOI] [PubMed] [Google Scholar]
- 19.Musshoff K. Clinical staging classification of non-Hodgkin's lymphomas. Strahlentherapie. 1977;153:218–221. (In German) [PubMed] [Google Scholar]
- 20.Roseau G. Role of echoendoscopy in the study of gastric diseases with fold thickening and in gastric lymphomas. Gastroenterol Hepatol. 2002;25:19–25. doi: 10.1016/S0210-5705(02)70236-7. (In Spanish) [DOI] [PubMed] [Google Scholar]
- 21.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 22.Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, Coiffier B, Fisher RI, Hagenbeek A, Zucca E, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25:579–586. doi: 10.1200/JCO.2006.09.2403. [DOI] [PubMed] [Google Scholar]
- 23.Asenjo LM, Gisbert JP. Prevalence of Helicobacter pylori infection in gastric MALT lymphoma: A systematic review. Rev Esp Enferm Dig. 2007;99:398–404. doi: 10.4321/S1130-01082007000700006. (In Spanish) [DOI] [PubMed] [Google Scholar]
- 24.Lin WC, Tsai HF, Kuo SH, Wu MS, Lin CW, Hsu PI, Cheng AL, Hsu PN. Translocation of Helicobacter pylori CagA into Human B lymphocytes, the origin of mucosa-associated lymphoid tissue lymphoma. Cancer Res. 2010;70:5740–5748. doi: 10.1158/0008-5472.CAN-09-4690. [DOI] [PubMed] [Google Scholar]
- 25.Wu YY, Tsai HF, Lin WC, Hsu PI, Shun CT, Wu MS, Hsu PN. Upregulation of CCL20 and recruitment of CCR6+ gastric infiltrating lymphocytes in Helicobacter pylori gastritis. Infect Immun. 2007;75:4357–4363. doi: 10.1128/IAI.01660-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tasaki K, Shichishima A, Furuta M, Yoshida S, Nakamura N, Abe M. CD5-positive mucosa-associated lymphoid tissue (MALT) lymphoma of ocular adnexal origin: Usefulness of fluorescence in situ hybridization for distinction between mantle cell lymphoma and MALT lymphoma. Pathol Int. 2007;57:101–107. doi: 10.1111/j.1440-1827.2006.02063.x. [DOI] [PubMed] [Google Scholar]
- 27.Stathis A, Chini C, Bertoni F, Proserpio I, Capella C, Mazzucchelli L, Pedrinis E, Cavalli F, Pinotti G, Zucca E. Long-term outcome following Helicobacter pylori eradication in a retrospective study of 105 patients with localized gastric marginal zone B-cell lymphoma of MALT type. Ann Oncol. 2009;20:1086–1093. doi: 10.1093/annonc/mdn760. [DOI] [PubMed] [Google Scholar]
- 28.Zucca E, Dreyling M, ESMO Guidelines Working Group Gastric marginal zone lymphoma of MALT type: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;5(Suppl 21):v175–v176. doi: 10.1093/annonc/mdq182. [DOI] [PubMed] [Google Scholar]
- 29.Liu TY, Dei PH, Kuo SH, Lin CW. Early low-grade gastric MALToma rarely transforms into diffuse large cell lymphoma or progresses beyond the stomach and regional lymph nodes. J Formos Med Assoc. 2010;109:463–471. doi: 10.1016/S0929-6646(10)60078-2. [DOI] [PubMed] [Google Scholar]
- 30.Gong EJ, Ahn JY, Jung HY, Park H, Ko YB, Na HK, Jung KW, Kim do H, Lee JH, Choi KD, et al. Helicobacter pylori eradication therapy is effective as the initial treatment for patients with H. pylori-negative and disseminated gastric Mucosa-Ass. Gut Liver. 2016;10:706–713. doi: 10.5009/gnl15510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vasilatou D, Sioulas AD, Pappa V, Papanikolaou IS, Triantafyllou K, Dimitriadis GD, Papageorgiou SG. The role of miRNAs and epigenetic mechanisms in primary gastric mucosa-associated lymphoid tissue lymphoma. Future Oncol. 2016;12:1587–1593. doi: 10.2217/fon-2016-0038. [DOI] [PubMed] [Google Scholar]
- 32.Thorns C, Kuba J, Bernard V, Senft A, Szymczak S, Feller AC, Bernd HW. Deregulation of a distinct set of microRNAs is associated with transformation of gastritis into MALT lymphoma. Virchows Arch. 2012;460:371–377. doi: 10.1007/s00428-012-1215-1. [DOI] [PubMed] [Google Scholar]
- 33.Shang Y, Zang A, Li J, Jia Y, Li X, Zhang L, Huo R, Yang J, Feng J, Ge K, et al. MicroRNA-383 is a tumor suppressor and potential prognostic biomarker in human non-small cell lung caner. Biomed Pharmacother. 2016;83:1175–1181. doi: 10.1016/j.biopha.2016.08.006. [DOI] [PubMed] [Google Scholar]
- 34.Xu Z, Zeng X, Tian D, Xu H, Cai Q, Wang J, Chen Q. MicroRNA-383 inhibits anchorage-independent growth and induces cell cycle arrest of glioma cells by targeting CCND1. Biochem Biophys Res Commun. 2014;453:833–838. doi: 10.1016/j.bbrc.2014.10.047. [DOI] [PubMed] [Google Scholar]
- 35.Lian J, Tian H, Liu L, Zhang XS, Li WQ, Deng YM, Yao GD, Yin MM, Sun F. Downregulation of microRNA-383 is associated with male infertility and promotes testicular embryonal carcinoma cell proliferation by targeting IRF1. Cell Death Dis. 2010;1:e94. doi: 10.1038/cddis.2010.70. [DOI] [PMC free article] [PubMed] [Google Scholar]