ABSTRACT
Wnt signalling regulates cardiogenesis during specification of heart tissue and the morphogenetic movements necessary to form the linear heart. Wnt11-mediated non-canonical signalling promotes early cardiac development whilst Wnt11-R, which is expressed later, also signals through the non-canonical pathway to promote heart development. It is unclear which Frizzled proteins mediate these interactions. Frizzled-7 (fzd7) is expressed during gastrulation in the mesodermal cells fated to become heart, and then in the primary heart field. This expression is complementary to the expression of wnt11 and wnt11-R. We further show co-localisation of fzd7 with other early- and late-heart-specific markers using double in situ hybridisation. We have used loss of function analysis to determine the role of fzd7 during heart development. Morpholino antisense oligonucleotide-mediated knockdown of Fzd7 results in effects on heart development, similar to that caused by Wnt11 loss of function. Surprisingly, overexpression of dominant-negative Fzd7 cysteine rich domain (Fzd7 CRD) results in a cardia bifida phenotype, similar to the loss of wnt11-R phenotype. Overexpression of Fzd7 and activation of non-canonical wnt signalling can rescue the effect of Fzd7 CRD. We propose that Fzd7 has an important role during Xenopus heart development.
KEY WORDS: Xenopus laevis, Cardiogenesis, Wnt signalling, Fzd7
Summary: Wnt signalling has been shown to be important in heart development. Here, we demonstrate that the wnt receptor fzd7 is required in mediating these Wnt signals.
INTRODUCTION
During embryogenesis, the heart is one of the first organs to form. Development of the heart includes specification of cardiac progenitors and formation of the linear heart tube by cell migration and morphogenetic movements (Mohun et al., 2000). In Xenopus, the heart begins to form during early gastrula stages when the cardiac progenitors arise in the dorsolateral mesoderm. Cell movements during gastrulation result in the dorso-anterior translocation of these regions and subsequent ventral migration during neurulation. The heart progenitors, which comprise cells fated to become primary or secondary heart field, form a linear heart tube at the ventral midline before looping and remodelling to form the beating heart (Kriegmair et al., 2013). Understanding the processes underlying heart development and morphogenesis are important for understanding congenital heart disease.
Heart formation is controlled by many signalling pathways including wnt signalling. Wnt6, wnt11, and wnt11-R have all been implicated in Xenopus heart development (Garriock et al., 2005; Gessert et al., 2008; Lavery et al., 2008a; Pandur et al., 2002). Wnt antagonists such as Dickkopf-1, Crescent and Sfrp1 have also been reported to control early heart formation (David et al., 2008; Foley and Mercola, 2005; Gibb et al., 2013; Marvin et al., 2001; Schneider and Mercola, 2001). Little is known however about which Frizzled proteins mediate these signals. Frizzled-7 (fzd7) has been well characterised in Xenopus laevis and other species. It has been shown to be involved in numerous developmental processes as well as being shown to be active in several forms of cancer (Huang and Klein, 2004; Liu et al., 2016; Schiffgens et al., 2016; Xu et al., 2016). Fzd7 has been demonstrated to interact with several wnts including Wnt5a (animal cap elongation assays), Wnt6 (in somite development), Wnt8 (co-immunoprecipitation assays, Xenopus axis duplication) and Wnt11 (gastrulation movements, neural crest development) (Hsieh et al., 1999; Linker et al., 2005; Medina et al., 2000; Medina and Steinbeisser, 2000; Umbhauer et al., 2000; Witzel et al., 2006). It has also been shown to genetically interact with the co-receptors ror2 and ryk (Hikasa et al., 2002; Kim et al., 2008). Xenopus fzd7 has been implicated in gastrulation movements, tissue separation, and neural crest induction (Abu-Elmagd et al., 2006; Djiane et al., 2000; Wheeler et al., 2000; Winklbauer et al., 2001). We have previously shown fzd7 to be expressed in the cardiac region throughout development (Wheeler and Hoppler, 1999). It has also been shown that specific depletion of fzd7 function in Xenopus foregut leads to impaired cardiac morphogenesis, but has no effect on heart specification (Zhang et al., 2013). Here, we further characterise its expression relative to known heart markers, and then use whole-embryo experiments to show that fzd7 is required for heart formation during early embryonic development.
RESULTS
fzd7 expression overlaps with early heart markers
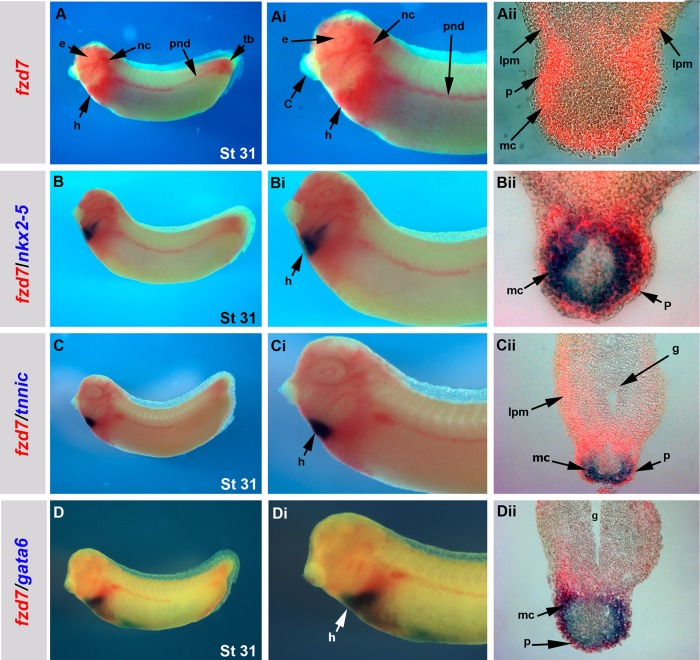
Expression pattern analysis shows Xenopus fzd7 is expressed in the heart-forming regions throughout development (Wheeler and Hoppler, 1999). At stage 10.5 fzd7 is expressed in the dorsal mesoderm from which cardiac tissue originates (Wheeler and Hoppler, 1999) (Fig. 1A). As development progresses, fzd7 expression at stage 25 is maintained in the presumptive cardiac mesoderm as it migrates dorso-laterally to the ventral midline (Fig. 1C-Cii). By stage 29, fzd7 is expressed throughout the cardiac crescent in the cardiac mesoderm (Fig. 1E,Ei). fzd7 expression correlates with that of wnt11 (Fig. 1B, stage 10.5) where expression of both genes seem to be complementary in the presumptive heart region in the dorsal side of the embryo. fzd7 expression also correlates to that of wnt11-R (Fig. 1D-Dii,F,Fi, stages 25 and 29, respectively) where it is expressed in the anterior endoderm at stage 25 when fzd7 is expressed in the heart field. By stage 29, the expression of fzd7 and wnt11-R overlaps (Fig. 1E-Fi). As the heart continues to form, fzd7 is strongly expressed in the lateral plates of mesoderm, cardiac mesoderm, myocardium, and over time, is restricted to the pericardium (Wheeler and Hoppler, 1999) (Fig. 2A-Aii, Bii, Cii, Dii). Using double in situ hybridisation, we analysed fzd7 expression in correlation to that of early heart markers including nkx2-5, troponin-ic (tnnic) and gata6, which are all known to be required for Xenopus cardiogenesis (Afouda and Hoppler, 2011; Afouda et al., 2008; Drysdale et al., 1994; Flaherty and Dawn, 2008; Fu et al., 1998; Garriock et al., 2005; Jiang and Evans, 1996; Martin et al., 2010). fzd7 expression overlaps with that of nkx2-5 (Fig. 2B-Bii), tnnic (Fig. 2C-Cii) and gata6 (Fig. 2D-Dii) in the forming heart. Interestingly, none of these markers are seen in the pericardium except for fzd7 (Fig. 2Aii,Bii,Cii,Dii).
Fig. 1.
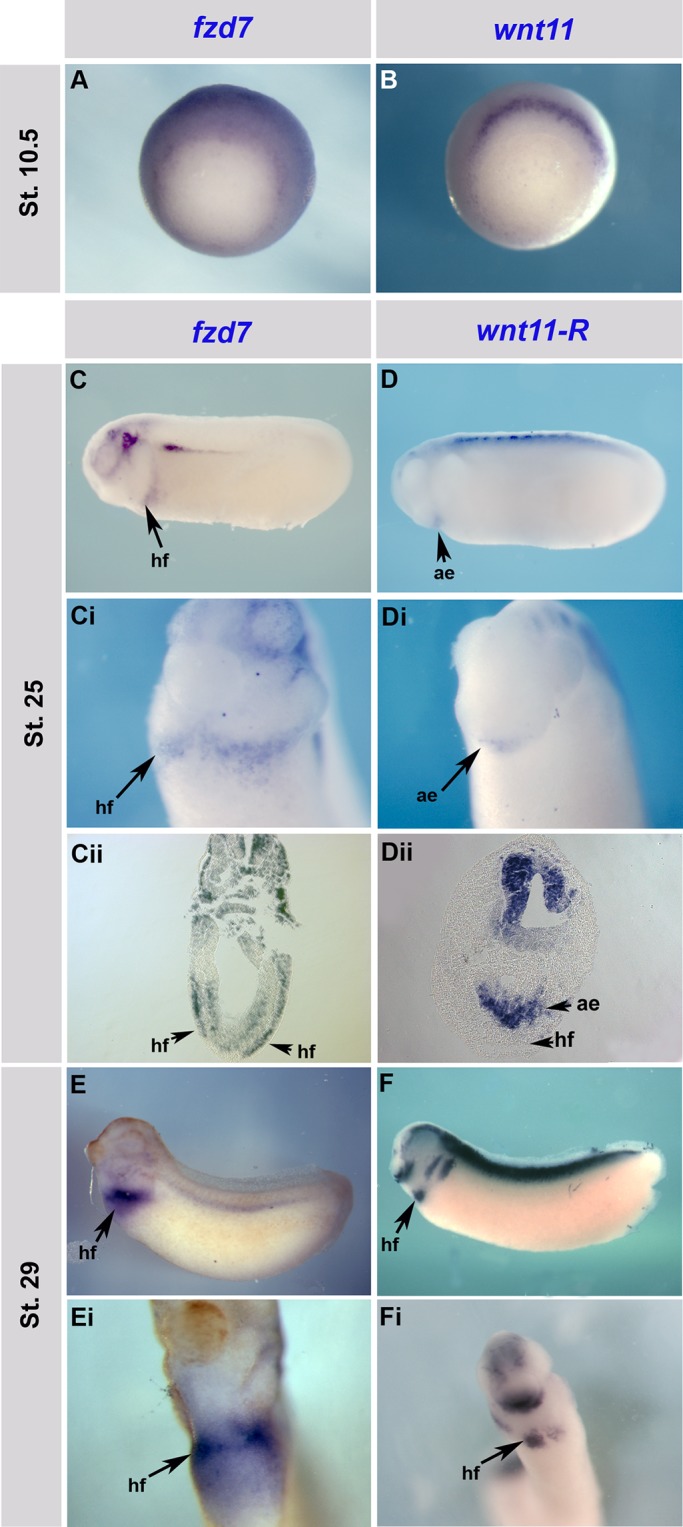
Endogenous expression of fzd7 in Xenopus heart and relative to wnt11 and wnt11-R expression. (A,B) Stage 10.5 (mid-gastrula) fzd7 and wnt11 expression detected at the dorsal side of the embryo and appear complementary in the presumptive heart region. (C-Cii,D-Dii) fzd7 and wnt11-R expression at stage 25. fzd7 is seen in the heart field and wnt11-R in the anterior endoderm. fzd7 and wnt11-R expression are complementary in the heart region (Cii,Dii). (E,Ei,F,Fi) Stage 29 embryos with fzd7 and wnt11-R expression in the heart field. hf, heart field; ae, anterior endoderm. Magnification 20×.
Fig. 2.
fzd7 expression coincides with expression of the early heart markers nkx2-5, tnnic and gata6. (A-Aii) Lateral view of Xenopus laevis embryos at stage 31 showing fzd7 expression detected in red and co-localised by double in situ hybridisation with other heart markers in dark blue including nkx2-5 (B-Bii), tnnic (C-Cii) and gata6 (D-Dii). (Ai,Bi,Ci,Di) Magnified lateral view of the same embryos in A, B, C and D, respectively. (Aii,Bii,Cii,Dii) Cross sections through the heart region of the embryos in A, B, C and D, respectively. fzd7 is expressed in the myocardium and pericardium (Aii) and in other structures including neural crest, eye, pronephric duct and tail bud. fzd7 expression shows a high degree of overlapping with the heart markers in the myocardium but not in the pericardium (Bii,Cii,Dii). h, heart; c, cement gland; e, eye; nc, neural crest; pnd, pronephric duct; tb, tail bud; mc, myodcardium; lpm, lateral plate of mesoderm. Magnification: 20× in A, B, C and D; 30× in Ai, Bi, Ci and Di; 200× in Aii, Bii, Cii and Dii.
fzd7 is required for heart induction or specification
Microinjection into Xenopus embryo dorsal blastomeres at the 4- or 8-cell stage targets prospective mesoderm including cardiac tissue. In order to test the role of fzd7 in heart development, we inhibited its function by injecting either fzd7 morpholino (fzd7 MO) or its dominant-negative form expressing only the extracellular domain (cysteine rich domain, fzd7 CRD), which would disrupt fzd7 mediated signalling (Abu-Elmagd et al., 2006).
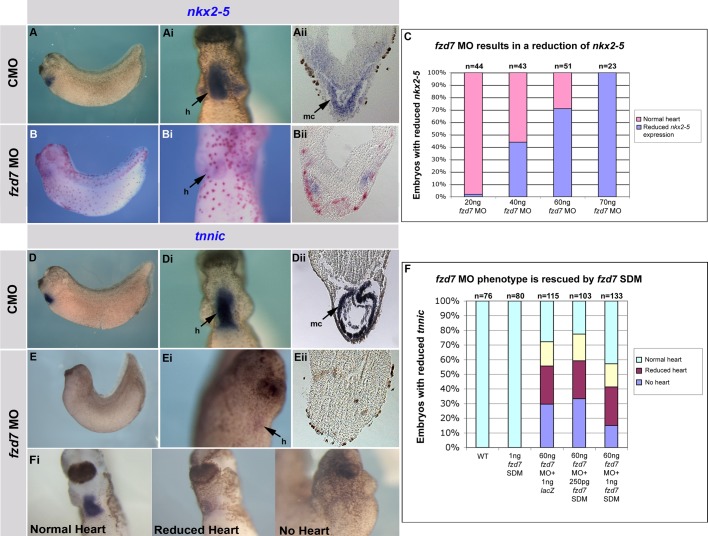
Microinjection of fzd7 MO into the dorsal blastomeres of 4- or 8-cell embryos leads to a reduction of both early cardiac marker nkx2-5 (Fig. 3B-Bii) and later cardiac marker tnnic expression (Fig. 3E-Eii). Adding increasing amounts of fzd7 MO leads to a progressively more severe phenotype with a greater number of embryos affected (Fig. 3C). In situ hybridisation for nkx2-5 and tnnic show embryos with mild convergent extension phenotypes (Fig. 3B,E), but a severe decrease in cardiac gene expression (Fig. 3Bi,Ei) while control morpholino (CMO) show normal heart (Fig. 3A,Ai,D,Di). Some embryos also showed anterior defects (data not shown). Sections through the cardiac region showed not only a decrease of nkx2-5 and tnnic expression, but an absence of recognisable heart structures (Fig. 3Bii,Eii) compared to CMO (Fig. 3Aii,Dii). The number of embryos injected with fzd7 MO which showed heart and/or convergent extension and anterior defects are shown in Table S1.
Fig. 3.
fzd7 is required for Xenopus heart development. (A,Ai,D,Di) Lateral and ventral views of embryos injected in the dorsal blastomeres (DB) at 4-cell stage with control morpholino (CMO) showing normal nkx2-5 (A,Ai) and tnnic (D,Di) expression. (Aii,Dii) Cross sections in the heart region of the embryos in A and D, respectively, showing normal nkx2-5 and tnnic expression in the myocardium. (B,Bi,E,Ei) Lateral and ventral views of embryos injected in the DB at 4-cell stage with fzd7 MO showing loss of nkx2-5 (B-Bi) and tnnic (E,Ei) expression. (Bii,Eii) Cross sections in the heart region of the embryos in B and E, respectively, showing loss of the heart. (C) Graph showing that fzd7 MO phenotype leads to reduction of nkx2-5 expression in a dose-dependent manner. (F,Fi) fzd7 MO phenotype can be rescued by fzd7 SDM full-length, panel Fi is the key for the phenotype scoring. Red staining in B-Bii is due to lac-Z lineage tracing using Red-Gal.
Overexpression of fzd7 full-length (fzd7 FL) results in severe convergent extension defects, but no cardiac phenotype (Fig. S1A,B). Knockdown with fzd7 MO can also cause a mild convergent extension phenotype and anterior defects (Abu-Elmagd et al., 2006). In order to test whether this cardiac effect is specific to fzd7, we rescued the fzd7 MO cardiac phenotype with fzd7 FL that has been mutated to not bind the fzd7 MO (fzd7 SDM, as described in Abu-Elmagd et al., 2006). Titrating increasing amounts of fzd7 SDM capped RNA from 250 pg to 1 ng results in a modest rescue of the cardiac phenotype (Fig. 3F,Fi; Table S2), thus showing that fzd7 is required for normal heart development.
Interestingly, injecting fzd7 FL at 8-cell stage embryos shows detectable expression of tnnic and nkx2-5, despite some of these embryos showing severe convergent extension movements phenotype (head arrows in Fig. S1A,B). This leads to the suggestion that heart phenotypes are not necessarily due to convergent extension secondary effects.
fzd7 CRD mimics wnt11-R morpholino cardia bifida phenotype and is required for non-canonical signalling
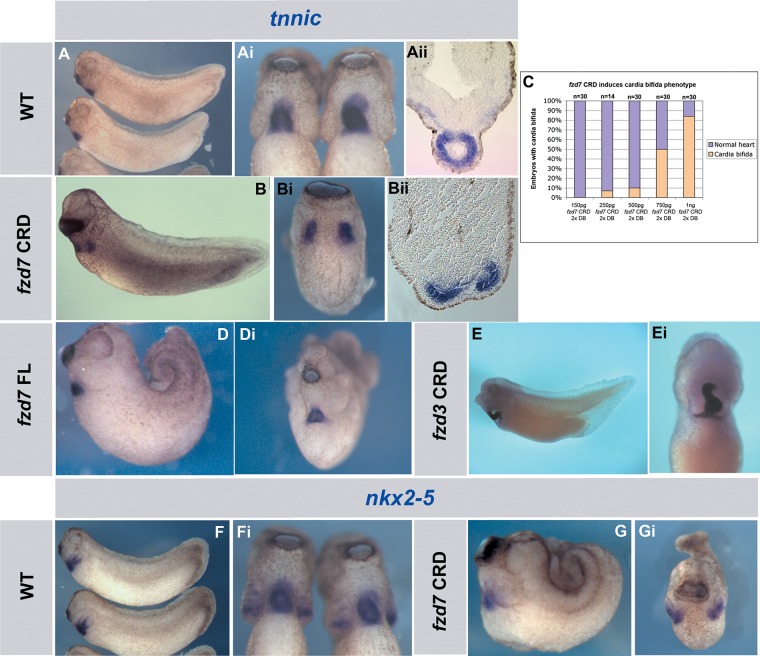
To further look at the effect of inhibiting fzd7 function, we took a dominant-negative approach using fzd7 CRD. Surprisingly, this did not give a similar result to the MO knockdown. Instead, increasing amounts of fzd7 CRD results in a dose-dependent increase in frequency and severity of cardia bifida. This was very similar to the phenotype seen for wnt-11R knockdown (Garriock et al., 2005). Embryos with very mild convergent extension movement defects displayed a severe cardia bifida phenotype as shown by tnnic (Fig. 4B-Bii,C) and nkx2-5 (Fig. 4G,Gi) expression. Control embryos showed normal expression of tnnic (Fig. 4A-Aii) and nkx2-5 (Fig. 4F,Fi). These results suggest that the cardia bifida phenotype is not a secondary effect of the convergent extension defect. Overexpression of fzd7 FL gives a severe convergent extension phenotype but no cardiac phenotype (Fig. S1A,B). Embryos with cardia bifida were unable to recover and form a normal heart when incubated up to stage 41 (n=23, data not shown). Embryos injected with a dominant-negative form of fzd3 (fzd3 CRD) into the dorsal blastomeres at 4-cell stage did not show cardia bifida (n= 27, Fig. 4E,Ei) indicating that the cardia bifida phenotype is specific to fzd7 CRD. Furthermore, this phenotypic specificity to fzd7 CRD was confirmed by rescuing the cardia bifida with fzd7 FL-capped RNA (Fig. 5A-D,F).
Fig. 4.
A dominant-negative fzd7 induces cardia bifida phenotype. (A,Ai,F,Fi) Lateral and ventral views of wild-type embryos at stage 29 showing normal tnnic (A, Ai) and nkx2-5 (F-Fi) expression in the heart. (B,Bi,G,Gi) Lateral and ventral views of embryos injected in the DB at 4-cell stage with dominant-negative fzd7 (fzd7 CRD). The cardia bifida phenotype is shown by tnnic (B,Bi) and nkx2-5 (G,Gi) expression. These embryos were fixed at the same stage as the control embryos in A and F. (C) Graph showing fzd7 CRD cardia bifida phenotype percentages indicated by tnnic expression. (D,Di) Lateral and ventral views of embryos injected in the DB at 4-cell stage with full-length of fzd7 (fzd7 FL) showing normal heart tube. Note that embryos in D and G are showing severe convergent extension defects but cardia bifida phenotype is only induced by fzd7 CRD. (E,Ei) Lateral and ventral views of injected embryo in the DB at 4-cell stage with fzd3 dominant-negative form (fzd3 CRD) showing normal heart looping at stage 38 indicating that fzd7 CRD cardia bifida phenotype is specific to fzd7. Magnification 20×.
Fig. 5.
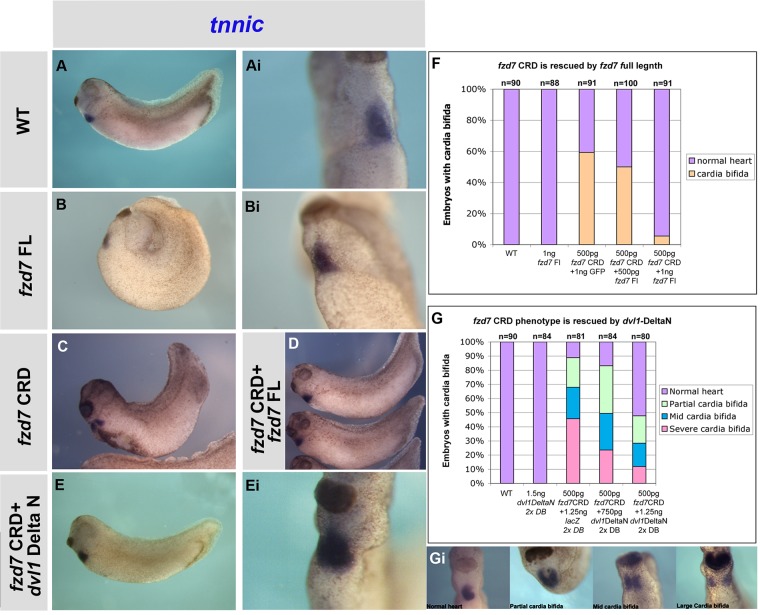
Activation of non-canonical wnt signalling rescues fzd7 CRD-induced cardia bifida. (A,Ai) Wild-type control embryos showing normal tnnic expression in the heart. (B,Bi) fzd7 full-length (fzd7 FL) overexpression (500 pg) injected into the dorsal blastomeres (DB) at the 4-cell stage show normal heart expression of tnnic despite suffering a severe extension movement defect. (C) Embryos injected with 500 pg fzd7 CRD show cardia bifida phenotype, note that embryos have normal to mild convergent extension defects. (D) Rescue of the fzd7 CRD (250 pg) cardia bifida phenotype with 250 pg fzd7 FL, embryos show normal morphology as well as normal tnnic expression. (F) Graph of fzd7 CRD cardia bifida phenotype rescue with fzd7 FL. (E,Ei) Rescue of fzd7 CRD (500 pg) cardia bifida phenotype with dishevelled1-Delta-N (Dvl1ΔN, 1.25 ng) indicating that fzd7 is required for the non-canonical signalling in the heart. (G) Graph of fzd7 CRD cardia bifida phenotype rescue with dvl1ΔN, panel Gi is the key for the cardia bifida phenotype scoring in G. Magnification 20×.
It has been previously reported that a Jun N-terminal kinases (Jun) inhibitor phenocopies the wnt11-R cardiac phenotype of effects on cardiac morphogenesis and heart tube fusion, suggesting signalling through the non-canonical pathway (Garriock et al., 2005; Gessert et al., 2008). We therefore determined to rescue the fzd7 CRD phenotype with dishevelled1-Delta-N (dvl1ΔN)-capped RNA. Dvl1ΔN-capped RNA can rescue fzd7 CRD (Fig. 5E,Ei,G,Gi; Table S3), suggesting that fzd7 is required for non-canonical wnt signalling during heart development.
DISCUSSION
Wnt signalling through the canonical and non-canonical pathways has been implicated in many aspects of heart development (Gessert and Kuhl, 2010; Ruiz-Villalba et al., 2016). How the wnt signals that arise from both non-cardiogenic and cardiogenic tissue are integrated into heart development is less well understood. Frizzled proteins are only a part of the increasingly complicated wnt-receptor complex found at the cell membrane, which can also include Lrp5/6, Ror2, Ryk and Kremen (Bryja et al., 2009; Korol et al., 2008; Mazzotta et al., 2016; van Wijk et al., 2009); however, Frizzled proteins are critical components of the Wnt receptor complex and so understanding their role in heart development is necessary to fully understand the signalling involved. We have previously shown that fzd7 is expressed throughout heart development, and in this study, we show that it is functionally required in both early and late heart development.
Morpholino knockdown of fzd7 leads to effects on heart development, including in some cases a complete loss of heart (Fig. 3). Overexpression of fzd7 gives rise to convergent extension defects as previously reported (Abu-Elmagd et al., 2006; Sumanas and Ekker, 2001; Winklbauer et al., 2001), but does not affect heart development. We can rescue the fzd7 MO phenotype by co-injecting site-directed mutagenized full-length fzd7 (Fig. 3). These results suggest that fzd7 is required for initial heart development, though we cannot exclude the possibility that it may also be playing a more general role in dorsoventral mesoderm patterning. Fzd7 could be interacting with Wnt11 (Kim et al., 2008; Tao et al., 2005; Witzel et al., 2006) , or another wnt ligand such as Wnt3a (Mazzotta et al., 2016), Wnt6 (Gibb et al., 2013; Lavery et al., 2008a, b) or Wnt8c (Ruiz-Villalba et al., 2016; Schneider and Mercola, 2001) during these stages of development.
As suggested, it is possible that the fzd7 morphant cardiac phenotype is a secondary effect of failures in mesoderm specification, patterning, gastrulation, axis formation and tissue separation. We have made efforts to inject embryos at the 4- and 8-cell stages to give as small a convergent extension phenotype as possible to generate normal-looking embryos but with clear heart phenotypes. The results suggest that the effect of fzd7 during early heart development is not secondary to convergent extension defects or mesoderm development, however, this cannot be ruled out completely (Fig. 3).
An interesting feature of the loss-of-function analysis using fzd7 Morpholino and a dominant-negative fzd7 (fzd7 CRD), is that they give different cardiac phenotypes. fzd7 morphants have anterior defects, convergent extension defects and reduction in nkx2-5 expression; whereas fzd7 CRD-capped RNA injections result in embryos with convergent extension defects and cardia bifida, but no head defects or loss of cardiac markers. Interestingly, it has been shown that the only way to replicate the anterior defect phenotype with a fzd7 CRD construct is to inject the capped RNA into oocytes (Medina et al., 2000). This could be because the relevant signalling event has been completed by time the product of mRNA injected at the 4- or 8-cell stage has been generated. It is possible that if we injected oocytes with fzd7 CRD then we might find embryos showing loss of the heart. Another possibility is that the Morpholino is able to disrupt all Wnt signalling through fzd7 by preventing translation of Fzd7 protein, but fzd7 CRD only disrupts non-canonical signalling in this context. The requirement for co-receptors in canonical signalling may allow the CRD to interact with endogenous fzd7 and any Lrps present allowing the receptor complex aggregates to form. In addition to this, it has been shown to be possible to activate canonical Wnt signalling using CRD constructs (Carron et al., 2003). Perhaps canonical Wnt signalling mediated by fzd7 early on during development is allowed to proceed by the Fzd7 CRD, but then when fzd7 switches to mediate non-canonical signalling, the CRD starts to behave as a dominant-negative. Other possibilities are that the Morpholino may have a broader specificity than thought or that the injected RNA of the fzd7 CRD construct may not be very stable, and thus only provide a short term effect compared to the Morpholino. These possibilities remain to be tested further.
The fzd7 CRD phenotype is very similar to the wnt11-R Morpholino phenotype (Garriock et al., 2005). It has previously been shown that DM-GRASP/alcam expression lies downstream of wnt11-R signalling and that DM-GRASP/alcam can mediate non-canonical wnt signalling effects on morphogenetic movements involved in the developing heart. The DM-GRASP/alcam Morpholino phenotype is also similar to the fzd7 CRD phenotype in that they both lead to a cardia bifida-like phenotype and a thickening of the myocardium. This suggests fzd7 could be mediating the wnt11-R control of DM-GRASP/alcam expression. This needs to be investigated further.
Ruiz-Villalba et al. (2016) suggest a model where periodic switching between proliferation and differentiation within the developing heart is mediated by the periodic and reciprocal activity of the canonical and non-canonical wnt pathways. fzd7 could be playing a crucial role in this process depending upon the Wnts and other receptors expressed at specific times.
In conclusion, we have shown fzd7 to be involved in heart development. Further investigation is required to determine the specific wnt(s) it is interacting with at different stages of heart development.
MATERIALS AND METHODS
Embryo manipulation
All experiments were performed in compliance with the relevant laws and institutional guidelines at the University of East Anglia. The research was approved by the local ethical review committee according to UK Home Office regulations. Xenopus laevis embryos were obtained as previously described (Harrison et al., 2004). Staging of the embryos was carried out according to the normal timetable of Nieuwkoop and Faber (Nieuwkoop and Faber, 1994). Embryos at the required stages were fixed in MEMFA, washed in PBS, dehydrated in ascending grades of Methanol/PBS, then stored in 100% MeOH at –20°C until processing for single or double in situ hybridisation.
Constructs
fzd7 full-length (fzd7 FL) and dominant-negative form fzd7-cysteine rich domain (fzd7 CRD) were sub-cloned into pCS2+ at Cla1–Xho1 restriction sites as described in Wheeler et al. (2000). fzd7 MO titration by RNA in the rescue experiments was avoided by creating a site-directed mutagenesis construct of the full-coding sequence of fzd7 (fzd7 SDM) as described in Abu-Elmagd et al. (2006). fzd3 full-length (fzd3 FL) and fzd3 CRD were kind gifts from Peter Klein (University of Pennsylvania). Dishevelled construct (Dvl1-Delta-N) was a gift from Roberto Mayor (University College, London) (De Calisto et al., 2005).
In vitro capped mRNA synthesis and embryo microinjections
All capped mRNAs of all genes used for RNA injections were prepared according to the manufacturer's instructions using the SP6 mMessage mMachine Ambion kit (Invitrogen™ AM1340). Anti-sense oligonucleotides, morpholinos (MOs), were obtained and designed by Gene Tools (www.gene-tools.com, Oregon, USA) using the reported sequence for the control morpholino (CMO) (5′-CCTCTTACCTCAgTTACAATTTATA-3′) and fzd7MO (5′-GCGGAGTGAGCAGAAATCGGCTGA-3′) (Sumanas and Ekker, 2001). MOs were diluted, prepared before use according to the manufacturer's instructions and tested using the in vitro translation assay (TNT coupled reticulocyte lysate system, Promega-L4600). For targeting the heart, the DB of the 4- and 8-cell stage embryos were injected as previously described (Lavery et al., 2008a). Capped mRNA and MOs were co-injected with lac-Z for lineage tracing. Each experiment was carried out as an internally controlled group. Each experiment was carried out three times and the number of embryos in each class were pooled.
RNA probe synthesis and in situ hybridisation
fzd7 in pBluescript was linearised with XbaI and transcribed by T7; nkx2-5 was linearised with BamH1 and transcribed with T7; troponin-lC (tnnic) was linearised with XhoI and transcribed with T3; gata6 was linearised with XbaI and transcribed with T7. Promega probe synthesis manufacturing instructions were followed with fzd7 probe labelled with Fluorescene-substituted nucleotide (Fl-UTP) and for other heart makers labelled with DIG-substituted nucleotide. Each RNA probe was added to 10 ml hybridisation buffer and stored at –20˚C for in situ hybridisation. Single (Harland, 1991) or double (Knecht et al., 1995) in situ hybridisation was carried out as previously described (Abu-Elmagd et al., 2006). Anti-Fluorescein was detected using Fast Red tablets (Kelloff et al., 2006) while anti-Digoxigenin was detected with NBT/BCIP. Frozen and wax sectioning were carried out as described (Harrison et al., 2004; Hatch et al., 2016). Images were taken using Leica microscope and Axiovision software.
Supplementary Material
Acknowledgements
The authors would like to thank Stefan Hoppler, Boni Afouda and Andrea Münsterberg as well as members of the Wheeler and Münsterberg labs for many helpful discussions.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
Conceptualization: J.M., G.N.W.; Methodology: M.A.-E., J.M., G.N.W.; Validation: M.A.-E., J.M., G.N.W.; Formal analysis: M.A.-E., J.M., G.N.W.; Investigation: M.A.-E., J.M., G.N.W.; Resources: M.A.-E., J.M., G.N.W.; Data curation: M.A.-E., J.M., G.N.W.; Writing - original draft: M.A.-E., J.M., G.N.W.; Writing - review & editing: M.A.-E., J.M., G.N.W.; Visualization: J.M., G.N.W.; Supervision: G.N.W.; Project administration: G.N.W.; Funding acquisition: G.N.W.
Funding
J.M. was funded by a Biotechnology and Biological Sciences Research Council (BBSRC) Studentship. This work was funded by a Biotechnology and Biological Sciences Research Council (BBSRC) new investigator award to G.W. (grant no. G15793).
Supplementary information
Supplementary information available online at http://bio.biologists.org/lookup/doi/10.1242/bio.026963.supplemental
References
- Abu-Elmagd M., Garcia-Morales C. and Wheeler G. N. (2006). Frizzled7 mediates canonical Wnt signaling in neural crest induction. Dev. Biol. 298, 285-298. 10.1016/j.ydbio.2006.06.037 [DOI] [PubMed] [Google Scholar]
- Afouda B. A. and Hoppler S. (2011). Different requirements for GATA factors in cardiogenesis are mediated by non-canonical Wnt signaling. Dev. Dyn. 240, 649-662. 10.1002/dvdy.22570 [DOI] [PubMed] [Google Scholar]
- Afouda B. A., Martin J., Liu F., Ciau-Uitz A., Patient R. and Hoppler S. (2008). GATA transcription factors integrate Wnt signalling during heart development. Development 135, 3185-3190. 10.1242/dev.026443 [DOI] [PubMed] [Google Scholar]
- Bryja V., Andersson E. R., Schambony A., Esner M., Bryjova L., Biris K. K., Hall A. C., Kraft B., Cajanek L., Yamaguchi T. P. et al. (2009). The extracellular domain of Lrp5/6 inhibits noncanonical Wnt signaling in vivo. Mol. Biol. Cell 20, 924-936. 10.1091/mbc.E08-07-0711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carron C., Pascal A., Djiane A., Boucaut J. C., Shi D. L. and Umbhauer M. (2003). Frizzled receptor dimerization is sufficient to activate the Wnt/beta-catenin pathway. J. Cell Sci. 116, 2541-2550. 10.1242/jcs.00451 [DOI] [PubMed] [Google Scholar]
- David R., Brenner C., Stieber J., Schwarz F., Brunner S., Vollmer M., Mentele E., Müller-Höcker J., Kitajima S., Lickert H. et al. (2008). MesP1 drives vertebrate cardiovascular differentiation through Dkk-1-mediated blockade of Wnt-signalling. Nat. Cell Biol. 10, 338-345. 10.1038/ncb1696 [DOI] [PubMed] [Google Scholar]
- De Calisto J., Araya C., Marchant L., Riaz C. F. and Mayor R., (2005). Essential role of non-canonical Wnt signalling in neural crest migration. Development 132, 2587-2597. 10.1242/dev.01857 [DOI] [PubMed] [Google Scholar]
- Djiane A., Riou J., Umbhauer M., Boucaut J. and Shi D. (2000). Role of frizzled 7 in the regulation of convergent extension movements during gastrulation in Xenopus laevis. Development 127, 3091-3100. [DOI] [PubMed] [Google Scholar]
- Drysdale T. A., Tonissen K. F., Patterson K. D., Crawford M. J. and Krieg P. A. (1994). Cardiac troponin I is a heart-specific marker in the Xenopus embryo: expression during abnormal heart morphogenesis. Dev. Biol. 165, 432-441. 10.1006/dbio.1994.1265 [DOI] [PubMed] [Google Scholar]
- Flaherty M. P. and Dawn B. (2008). Noncanonical Wnt11 signaling and cardiomyogenic differentiation. Trends Cardiovasc. Med. 18, 260-268. 10.1016/j.tcm.2008.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foley A. C. and Mercola M. (2005). Heart induction by Wnt antagonists depends on the homeodomain transcription factor Hex. Genes Dev. 19, 387-396. 10.1101/gad.1279405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu Y., Yan W., Mohun T. J. and Evans S. M. (1998). Vertebrate tinman homologues XNkx2-3 and XNkx2-5 are required for heart formation in a functionally redundant manner. Development 125, 4439-4449. [DOI] [PubMed] [Google Scholar]
- Garriock R. J., D'Agostino S. L., Pilcher K. C. and Krieg P. A. (2005). Wnt11-R, a protein closely related to mammalian Wnt11, is required for heart morphogenesis in Xenopus. Dev. Biol. 279, 179-192. 10.1016/j.ydbio.2004.12.013 [DOI] [PubMed] [Google Scholar]
- Gessert S. and Kuhl M. (2010). The multiple phases and faces of wnt signaling during cardiac differentiation and development. Circ. Res. 107, 186-199. 10.1161/CIRCRESAHA.110.221531 [DOI] [PubMed] [Google Scholar]
- Gessert S., Maurus D., Brade T., Walther P., Pandur P. and Kühl M. (2008). DM-GRASP/ALCAM/CD166 is required for cardiac morphogenesis and maintenance of cardiac identity in first heart field derived cells. Dev. Biol. 321, 150-161. 10.1016/j.ydbio.2008.06.013 [DOI] [PubMed] [Google Scholar]
- Gibb N., Lavery D. L. and Hoppler S. (2013). sfrp1 promotes cardiomyocyte differentiation in Xenopus via negative-feedback regulation of Wnt signalling. Development 140, 1537-1549. 10.1242/dev.088047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harland R. M. (1991). In situ hybridization: an improved whole-mount method for Xenopus embryos. Methods Cell Biol. 36, 685-695. 10.1016/S0091-679X(08)60307-6 [DOI] [PubMed] [Google Scholar]
- Harrison M., Abu-Elmagd M., Grocott T., Yates C., Gavrilovic J. and Wheeler G. N. (2004). Matrix metalloproteinase genes in Xenopus development. Dev. Dyn. 231, 214-220. 10.1002/dvdy.20113 [DOI] [PubMed] [Google Scholar]
- Hatch V. L., Marin-Barba M., Moxon S., Ford C. T., Ward N. J., Tomlinson M. L., Desanlis I., Hendry A. E., Hontelez S., van Kruijsbergen I. et al. (2016). The positive transcriptional elongation factor (P-TEFb) is required for neural crest specification. Dev. Biol. 416, 361-372. 10.1016/j.ydbio.2016.06.012 [DOI] [PubMed] [Google Scholar]
- Hikasa H., Shibata M., Hiratani I. and Taira M. (2002). The Xenopus receptor tyrosine kinase Xror2 modulates morphogenetic movements of the axial mesoderm and neuroectoderm via Wnt signaling. Development 129, 5227-5239. [DOI] [PubMed] [Google Scholar]
- Hsieh J.-C., Rattner A., Smallwood P. M. and Nathans J. (1999). Biochemical characterization of Wnt-frizzled interactions using a soluble, biologically active vertebrate Wnt protein. Proc. Natl. Acad. Sci. USA 96, 3546-3551. 10.1073/pnas.96.7.3546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang H.-C. and Klein P. S. (2004). The Frizzled family: receptors for multiple signal transduction pathways. Genome Biol. 5, 234 10.1186/gb-2004-5-7-234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Y. and Evans T. (1996). The Xenopus GATA-4/5/6 genes are associated with cardiac specification and can regulate cardiac-specific transcription during embryogenesis. Dev. Biol. 174, 258-270. 10.1006/dbio.1996.0071 [DOI] [PubMed] [Google Scholar]
- Kelloff G. J., Lippman S. M., Dannenberg A. J., Sigman C. C., Pearce H. L., Reid B. J., Szabo E., Jordan V. C., Spitz M. R., Mills G. B. et al. (2006). Progress in chemoprevention drug development: the promise of molecular biomarkers for prevention of intraepithelial neoplasia and cancer--a plan to move forward. Clin. Cancer Res. 12, 3661-3697. 10.1158/1078-0432.CCR-06-1104 [DOI] [PubMed] [Google Scholar]
- Kim G.-H., Her J.-H. and Han J.-K. (2008). Ryk cooperates with Frizzled 7 to promote Wnt11-mediated endocytosis and is essential for Xenopus laevis convergent extension movements. J. Cell Biol. 182, 1073-1082. 10.1083/jcb.200710188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knecht A. K., Good P. J., Dawid I. B. and Harland R. M. (1995). Dorsal-ventral patterning and differentiation of noggin-induced neural tissue in the absence of mesoderm. Development 121, 1927-1935. [DOI] [PubMed] [Google Scholar]
- Korol O., Gupta R. W. and Mercola M. (2008). A novel activity of the Dickkopf-1 amino terminal domain promotes axial and heart development independently of canonical Wnt inhibition. Dev. Biol. 324, 131-138. 10.1016/j.ydbio.2008.09.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriegmair M. C. M., Frenz S., Dusl M., Franz W.-M., David R. and Rupp R. A. W. (2013). Cardiac differentiation in Xenopus is initiated by mespa. Cardiovasc. Res. 97, 454-463. 10.1093/cvr/cvs354 [DOI] [PubMed] [Google Scholar]
- Lavery D. L., Davenport I. R., Turnbull Y. D., Wheeler G. N. and Hoppler S. (2008a). Wnt6 expression in epidermis and epithelial tissues during Xenopus organogenesis. Dev. Dyn. 237, 768-779. 10.1002/dvdy.21440 [DOI] [PubMed] [Google Scholar]
- Lavery D. L., Martin J., Turnbull Y. D. and Hoppler S. (2008b). Wnt6 signaling regulates heart muscle development during organogenesis. Dev. Biol. 323, 177-188. 10.1016/j.ydbio.2008.08.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linker C., Lesbros C., Gros J., Burrus L. W., Rawls A. and Marcelle C. (2005). beta-Catenin-dependent Wnt signalling controls the epithelial organisation of somites through the activation of paraxis. Development 132, 3895-3905. 10.1242/dev.01961 [DOI] [PubMed] [Google Scholar]
- Liu N., Zang S., Liu Y., Wang Y., Li W., Liu Q., Ji M., Ma D. and Ji C. (2016). FZD7 regulates BMSCs-mediated protection of CML cells. Oncotarget 7, 6175-6187. 10.18632/oncotarget.6742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin J., Afouda B. A. and Hoppler S. (2010). Wnt/beta-catenin signalling regulates cardiomyogenesis via GATA transcription factors. J. Anat. 216, 92-107. 10.1111/j.1469-7580.2009.01171.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marvin M. J., Di Rocco G., Gardiner A., Bush S. M. and Lassar A. B., (2001). Inhibition of Wnt activity induces heart formation from posterior mesoderm. Genes Dev. 15, 316-327. 10.1101/gad.855501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzotta S., Neves C., Bonner R. J., Bernardo A. S., Docherty K. and Hoppler S. (2016). Distinctive roles of canonical and noncanonical Wnt signaling in human embryonic cardiomyocyte development. Stem Cell Rep. 7, 764-776. 10.1016/j.stemcr.2016.08.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medina A. and Steinbeisser H. (2000). Interaction of Frizzled 7 and dishevelled in xenopus. Dev. Dyn. 218, 671-680. [DOI] [PubMed] [Google Scholar]
- Medina A., Reintsch W. and Steinbeisser H. (2000). Xenopus frizzled 7 can act in canonical and non-canonical Wnt signaling pathways: implications on early patterning and morphogenesis. Mech. Dev. 92, 227-237. 10.1016/S0925-4773(00)00240-9 [DOI] [PubMed] [Google Scholar]
- Mohun T. J., Leong L. M., Weninger W. J. and Sparrow D. B. (2000). The morphology of heart development in Xenopus laevis. Dev. Biol. 218, 74-88. 10.1006/dbio.1999.9559 [DOI] [PubMed] [Google Scholar]
- Nieuwkoop P. and Faber J. (1994). Normal Table of Xenopus laevis (Daudin)(Garland, New York). ISBN 0-8153-1896-0.
- Pandur P., Läsche M., Eisenberg L. M. and Kühl M. (2002). Wnt-11 activation of a non-canonical Wnt signalling pathway is required for cardiogenesis. Nature 418, 636-641. 10.1038/nature00921 [DOI] [PubMed] [Google Scholar]
- Ruiz-Villalba A., Hoppler S. and van den Hoff M. J. B. (2016). Wnt signaling in the heart fields: Variations on a common theme. Dev. Dyn. 245, 294-306. 10.1002/dvdy.24372 [DOI] [PubMed] [Google Scholar]
- Schiffgens S., Wilkens L., Brandes A. A., Meier T., Franceschi E., Ermani M., Hartmann C., Sandalcioglu I. E. and Dumitru C. A. (2016). Sex-specific clinicopathological significance of novel (Frizzled-7) and established (MGMT, IDH1) biomarkers in glioblastoma. Oncotarget 7, 55169-55180. 10.18632/oncotarget.10465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider V. A. and Mercola M. (2001). Wnt antagonism initiates cardiogenesis in Xenopus laevis. Genes Dev. 15, 304-315. 10.1101/gad.855601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumanas S. and Ekker S. C. (2001). Xenopus frizzled-7 morphant displays defects in dorsoventral patterning and convergent extension movements during gastrulation. Genesis 30, 119-122. 10.1002/gene.1044 [DOI] [PubMed] [Google Scholar]
- Tao Q., Yokota C., Puck H., Kofron M., Birsoy B., Yan D., Asashima M., Wylie C. C., Lin X. and Heasman J. (2005). Maternal wnt11 activates the canonical wnt signaling pathway required for axis formation in Xenopus embryos. Cell 120, 857-871. 10.1016/j.cell.2005.01.013 [DOI] [PubMed] [Google Scholar]
- Umbhauer M., Djiane A., Goisset C., Penzo-Mendez A., Riou J. F., Boucaut J. C. and Shi D. L. (2000). The C-terminal cytoplasmic Lys-thr-X-X-X-Trp motif in frizzled receptors mediates Wnt/beta-catenin signalling. EMBO J. 19, 4944-4954. 10.1093/emboj/19.18.4944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Wijk N. V., Witte F., Feike A. C., Schambony A., Birchmeier W., Mundlos S. and Stricker S. (2009). The LIM domain protein Wtip interacts with the receptor tyrosine kinase Ror2 and inhibits canonical Wnt signalling. Biochem. Biophys. Res. Commun. 390, 211-216. 10.1016/j.bbrc.2009.09.086 [DOI] [PubMed] [Google Scholar]
- Wheeler G. N. and Hoppler S. (1999). Two novel Xenopus frizzled genes expressed in developing heart and brain. Mech. Dev. 86, 203-207. 10.1016/S0925-4773(99)00117-3 [DOI] [PubMed] [Google Scholar]
- Wheeler G. N., Hamilton F. S. and Hoppler S. (2000). Inducible gene expression in transgenic Xenopus embryos. Curr. Biol. 10, 849-852. 10.1016/S0960-9822(00)00596-0 [DOI] [PubMed] [Google Scholar]
- Winklbauer R., Medina A., Swain R. K. and Steinbeisser H. (2001). Frizzled-7 signalling controls tissue separation during Xenopus gastrulation. Nature 413, 856-860. 10.1038/35101621 [DOI] [PubMed] [Google Scholar]
- Witzel S., Zimyanin V., Carreira-Barbosa F., Tada M. and Heisenberg C.-P. (2006). Wnt11 controls cell contact persistence by local accumulation of Frizzled 7 at the plasma membrane. J. Cell Biol. 175, 791-802. 10.1083/jcb.200606017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu R., Zeng S., Xie W., Sun C., Chen Y. L., Chen M. J. and Zhang L. (2016). The expression and function of Frizzled-7 in human renal cell carcinoma. Clin, Transl. Oncol. 18, 269-276. 10.1007/s12094-015-1362-3 [DOI] [PubMed] [Google Scholar]
- Zhang Z., Rankin S. A. and Zorn A. M. (2013). Different thresholds of Wnt-Frizzled 7 signaling coordinate proliferation, morphogenesis and fate of endoderm progenitor cells. Dev. Biol. 378, 1-12. 10.1016/j.ydbio.2013.02.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.