Abstract
Melatonin has been reported to alleviate chilling symptoms in postharvest peach fruit during cold storage, however, the mechanism involved is largely unknown. To better understand its role in chilling tolerance, here we investigated the effects of melatonin on oxidative damage in peach fruit subjected to chilling after harvest. Chilling injury of peaches was dramatically reduced by melatonin treatment. Melatonin induced hydrogen peroxide (H2O2) content at the early stage of storage but inhibited its accumulation thereafter. Meanwhile, melatonin also up-regulated the expression of genes involved in antioxidant responses in peaches. In addition, compared to the control fruit, peaches treated with melatonin displayed higher transcript abundance of ascorbic acid (AsA) biosynthetic genes and consequently increased the AsA content. Our results suggested that in response to melatonin during chilling, the high H2O2 level in the treated peaches at the initial time of storage, may work as a signaling molecule to induce protective mechanisms via up-regulating the expression of antioxidative genes and increasing AsA content. On the other hand, after the transient increase in the treated peaches, H2O2 was efficiently removed because of the activated antioxidant systems, which was associated with the higher chilling tolerance induced by melatonin.
Introduction
Temperature is one of the most predominant factors for maintaining quality of peach fruit after harvest. Some of metabolic activities such as ripening and decay are reduced by lowering temperatures1,2. However, storage at temperature range of 2.2–7.6 °C causes certain physiological disorders known as chilling injury. Classic symptoms of chilling injury in peach fruit include flesh browning, reddening/bleeding and woolliness/mealiness1,2. Due to this phenomenon, fruit quality is deteriorated significantly during postharvest storage and the storage life is limited.
The appearance of chilling damage is often associated with oxidative stress resulted from an elevated production of reactive oxygen species (ROS) such as superoxide anion (O2−), hydrogen peroxide (H2O2), hydroxyl radical, nitric oxide, and peroxynitrite3. Oxidative damage is considered to be an early response of sensitive tissues to chilling4. If the production of ROS increases dramatically, as occurs under environmental stress, hydroxyl radical reacts with membrane lipids, which leads to lipid peroxidation and membrane degradation5. Malondialdehyde (MDA) is a product of this lipid peroxidation, and is used as an indicator of stress in some tissues6. In order to cope with ROS, plants have evolved an efficient antioxidant defense system to respond to oxidative stress and prevent the accumulation of ROS and repair oxidative damage7. This system involves both lipid-soluble antioxidants (α-tocopherol and carotene), water-soluble reductants including ascorbate acid (AsA) and glutathione (GSH), and enzymes such as catalase (CAT), ascorbate peroxidase (APX), superoxide dismutase (SOD) and glutathione reductase (GR)8.
Melatonin, (N-acetyl-5-methoxytiyptamine), a neural hormone secreted by the pineal gland in mammals, has been found in several plant tissues9. It has been reported to be involved in growth, development and stress responses in plants10–22. Recently, melatonin has been shown promising in regulating fruit ripening and preventing postharvest disease and disorders in horticultural crops. For example, grape berries treated with melatonin at pre-veraison exhibited higher endogenous melatonin accumulation, which not only increased berry size and weight, but also increased synchronicity of berry ripening23. A postharvest application of melatonin effectively delayed senescence and maintained quality of peach fruit stored at ambient temperature24. Exogenous melatonin pre-treatment improved anthocyanin accumulation by regulating gene expressions and resulted in high reactive oxygen species scavenging capacity in cabbage sprouts25. Treatment with melatonin delayed cassava roots postharvest physiological deterioration, which was resulted from lowering H2O2 accumulation26. The beneficial impacts of melatonin treatment on attenuating postharvest decay has also been demonstrated in strawberry fruit27. Additional, a label-free differential proteomics analysis revealed the effect of melatonin on promoting fruit ripening and anthocyanin accumulation upon postharvest in tomatoes28.
Our previous work has shown that melatonin could inhibit chilling injury and enhance the defense response against chilling in cold stored peach fruit29. However, the underlying physiological and molecular mechanism of melatonin in the induction of tolerance to cold stresses remains unclear. As a positive regulator of anti-ROS process, solid evidence suggests that melatonin can not only directly scavenge some ROS, but can also modulate antioxidant enzymes and enhance cellular antioxidants30,31. This study was therefore conducted to investigate whether the mitigating effect of melatonin is associated with its effect on the antioxidant response of postharvest peaches exposed to cold stress.
Results
Melatonin increased chilling tolerance in postharvest peaches
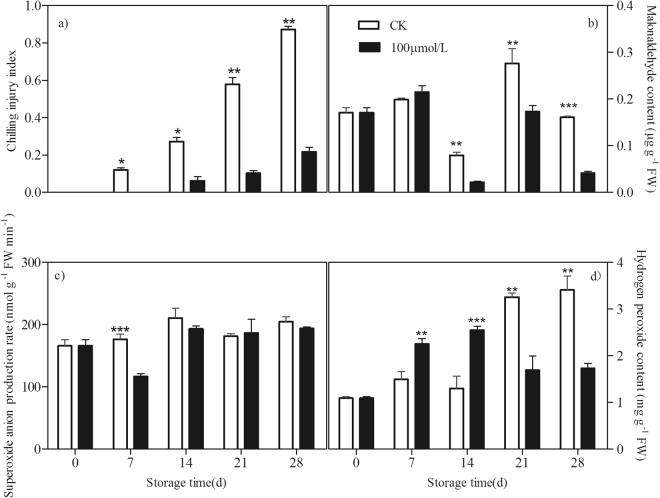
Compared with the control peaches, melatonin treatment delayed and reduced the chilling injury in peach fruit during the cold storage. Chilling was expressed as internal browning (Fig. 1a). The accumulation of MDA is the marker of lipid peroxidation and damage of cellular membranes. The MDA content of the control fruit decreased during the first 14 days and then sharply increased to a peak at the 21st day before declining. Melatonin treatment significantly inhibited MDA content after 14 days of storage (Fig. 1b).
Figure 1.
Effect of exogenous melatonin on chilling injury index (a) and contents of MDA (b), O2− (c) and H2O2 (d) of peach fruit during storage at 4 °C. All data about the chilling injury index is presented as a mean of fifteen biological replicates and error bars represent ± standard errors. Data about contents of MDA, O2− and H2O2 is presented as a mean of three biological replicates and error bars represent ± standard errors. Asterisks (*) indicate significant differences among melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
O2− and H2O2 are major ROS induced by chilling stress. In control fruit, no significant changes in O2− content was observed. However, under chilling stress, H2O2 level accumulated within storage time and a dramatically increase happened at 21st day. As compared to the control fruit, lower O2− was found at the 7th day after melatonin treatment (Fig. 1c). However, the treatment induced the increase in H2O2 content during the first 14 days of storage but inhibited its accumulation thereafter (Fig. 1d).
Melatonin increased expression of genes involved in antioxidative pathway
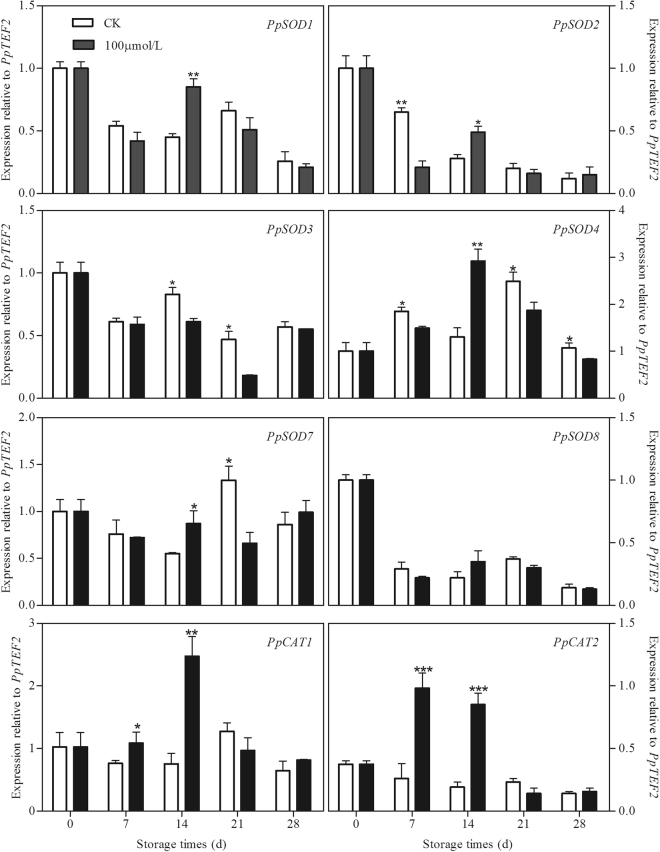
The effect of melatonin application on the transcripts of PpSODs in peach fruit is shown in Fig. 2. Among the 6 PpSODs, the expression of PpSOD1 and PpSOD7 in the control peaches decreased during the first 14 days of storage and then increased and reached a peak before a decline. The transcripts of PpSOD2, PpSOD3 and PpSOD8 decreased along with the storage time, while the expression of PpSOD4 increased during storage then declined at the end of storage. Melatonin up-regulated PpSOD1, PpSOD2, PpSOD4 and PpSOD7 at the 14th day but inhibited the expression of PpSOD2 at the 7th day and PpSOD4 during the whole storage except day 14. In addition, higher expression of PpSOD3 could also be observed in the control peaches at 14 and 21 days than in treated fruit. There was no significantly difference in PpSOD8 expression between the control and treated peaches. No significant changes in expression of PpCATs was observed in control peach subjected to chilling during storage. However, in melatonin treated fruit, the expression of PpCAT1 and PpCAT2 increased dramatically and reached their peaks at 14th and 7th day of storage, respectively. As compared to the control fruit, higher transcripts of these two PpCATs could be found at 7th and 14th days (Fig. 2).
Figure 2.
Effect of exogenous melatonin treatment on expression of PpSODs and PpCATs in peach fruit during storage at 4 °C. All data is presented as a mean of three biological replicates and error bars represent ± standard errors. Transcript abundance was determined using qPCR and was normalized using PpTEF2. SOD, superoxide dismutase. CAT, catalase. Asterisks (*) indicate significant differences between melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
Melatonin increased AsA content and upregulated genes involved in ASA-GSH cycle
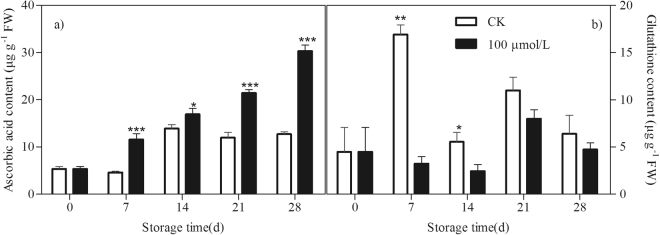
AsA and GSH can directly detoxify ROS and thus contribute to non-enzymatic ROS scavenging. During storage, in the control peaches, the AsA level increased slightly under chilling stress condition. GSH content increased sharply at the first 7 days of storage but declined thereafter. Melatonin treatment induced the AsA accumulation during the whole storage but maintained a lower level of GSH content at 7th and 14th days of storage as compared to the control fruit (Fig. 3).
Figure 3.
Effect of exogenous melatonin treatment on contents of AsA (a) and GSH (b) in peach fruit during storage at 4 °C. All data is presented as a mean of three biological replicates and error bars represent ± standard errors. Asterisks (*) indicate significant differences between melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
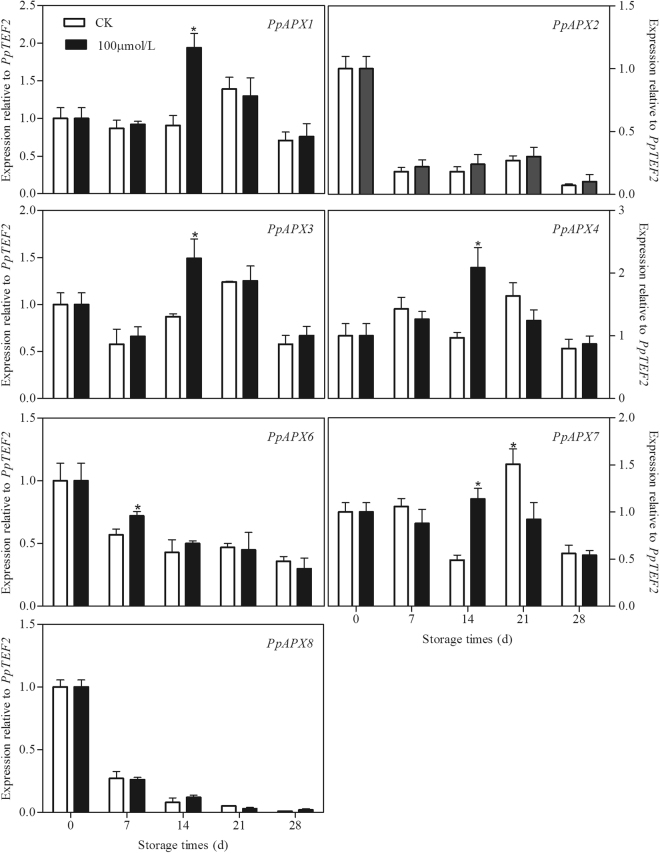
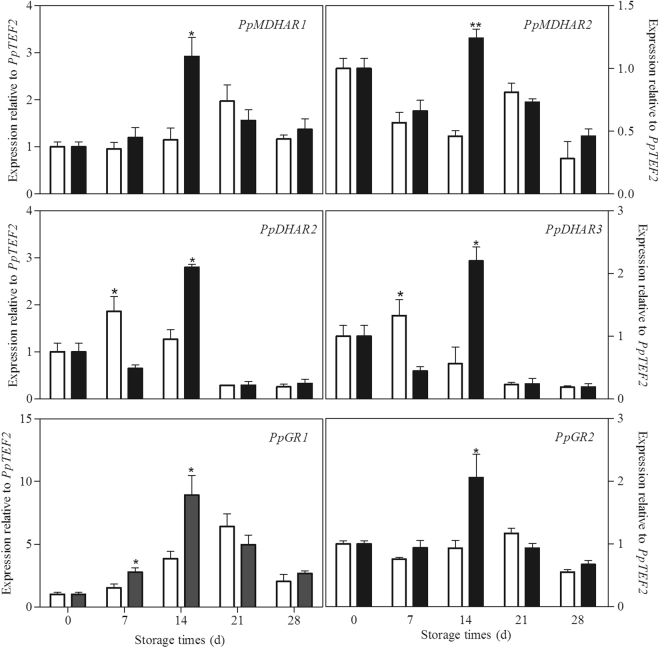
Seven PpAPXs in peach fruit were analysed. As shown in Fig. 4, in the control peaches under chilling stress, PpAPX2, PpAPX6 and PpAPX8 decreased gradually with the storage, but the other four APXs increased firstly and declined at the end of storage. Melatonin treatment induced the expression of PpAPX1, PpAPX3, PpAPX4 and PpAPX7 at 14th days of storage. A higher transcript of PpAPX6 was found at 7th day in treated peaches as compared to control fruit. There was no significant difference on the PpAPX2 and PpAPX8 between the control and treated fruit. The expression of two PpMDHARs in control peaches increased gradually during the first 21 days of storage and then declined. The two PpDHARs also experienced a similar change pattern during storage. The transcripts of the two genes increased at 7th day of storage and then decreased during the remaining storage time. The expression of PpGR1 increased firstly and then declined at the end of storage. No significant change was observed in the PpGR2 expression in the control fruit during storage. Interestingly, melatonin treatment up-regulated the transcripts of all the six genes at 14th day, but down-regulated the expression of two PpDHARs at 7th day of storage (Fig. 5).
Figure 4.
Effect of exogenous melatonin treatment on expression of PpAPX genes in peach fruit during storage at 4 °C. All data is presented as a mean of three biological replicates and error bars represent ± standard errors. Transcript abundance was determined using qPCR and was normalized using PpTEF2. APX, ascorbate peroxidase. Asterisks (*) indicate significant differences between melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
Figure 5.
Effect of exogenous melatonin treatment on expression of PpMDHAR, PpDHAR and PpGR genes in peach fruit during storage at 4 °C. All data is presented as a mean of three biological replicates and error bars represent ± standard errors. Transcript abundance was determined using qPCR and was normalized using PpTEF2. MDHAR, monodehydroascorbate reductase. DHAR, dehydroascorbate reductase. GR, glutathione reductase. Asterisks (*) indicate significant differences between melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
Melatonin increased transcript abundance of AsA biosynthetic genes
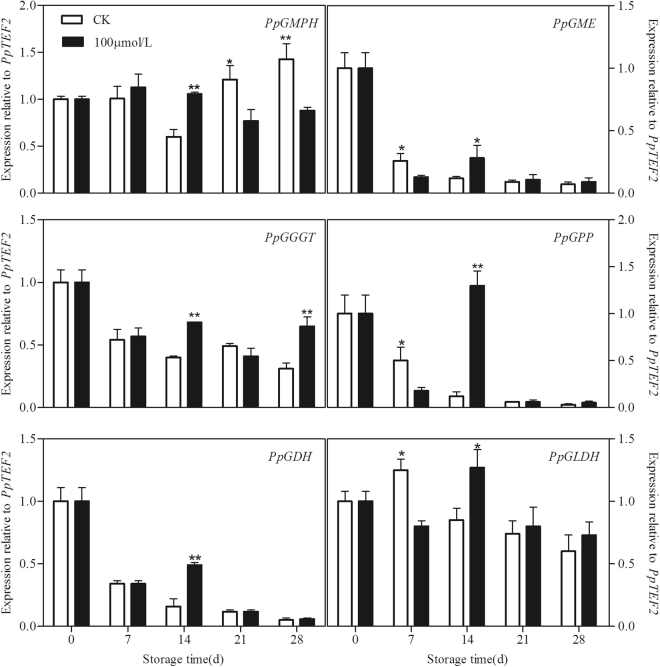
Degree of gene expression by GMPH, GME, GGGT, GPP, GDH and GLDH involved in AsA biosynthesis was analysed in peach fruit during cold storage. Expression of most of the AsA biosynthetic genes such as PpGGGT, PpGME, PpGPP and PpGDH decreased during the while storage in control fruit subjected to chilling stress. Transcripts of PpGMPH in control peaches declined firstly but increased after 21 days of storage but an increase in PpGLDH expression was observed at 7th of storage followed by a decline during the remaining time. Melatonin treatment upregulated the expression of all the biosynthetic genes at 14th day of storage. However, as compared to treated fruit, the control peaches showed a higher transcript of PpGME, PpGPP and PpGLDH at 7th and PpGMPH at 21st and 28th days of storage (Fig. 6).
Figure 6.
Effect of exogenous melatonin treatment on expression of AsA biosynthetic genes in peach fruit during storage at 4 °C. All data is presented as a mean of three biological replicates and error bars represent ± standard errors. Transcript abundance was determined using qPCR and was normalized using PpTEF2. GMPH, Mannose-1-phosphate guanylyltransferase. GME, GDP-D-mannose-3′,5′-epimerase. GGGT, GDP-L-galactose guanylyltransferase. GPP L-galactose-1-phosphate phosphatase. GDH, L-galactose-1-dehydrogenase. GLDH, L-galactono-1,4-lactone dehydrogenase. Asterisks (*) indicate significant differences between melatonin-treated and control samples (Student’s unpaired T test; *P < 0.05, **P < 0.01, and ***P < 0.001).
Discussion
It is well known that the major negative effect of chilling is that it causes oxidative stress in plants. At least some of the effects of chilling stress and tolerance mechanisms are believed to be mediated by chilling-induced H2O23,4. H2O2 has been reported to operate as a signal, secondary messenger, and regulator of some gene expression for the activation of stress responses and defense pathways to regulate the cellular redox status32,33. Similarly to our previous study29, melatonin treatment at 100 µM alleviated chilling injury and induced tolerance against chilling in postharvest peach fruit during cold storage (Fig. 1a). A transient increase in H2O2 was suggested to signal activation of the protective mechanism for acclimation to chilling34,35. In our present study, melatonin-treated peach fruit also maintained a higher level of H2O2 than control ones at the initial time of cold stress (Fig. 1d), suggesting the transiently-induced H2O2 by melatonin may act as a diffusible signal molecule to up-regulate downstream defense genes encoding many antioxidants. Thus, H2O2 implicated an important contribution to the redox state of peaches, which ultimately played a crucial role in chilling tolerance36.
However, H2O2 plays a dual role in response to chilling and this response is dependent on the severity of the stress. When the generation of H2O2 exceeds the capacity of the plant to maintain cellular redox homeostasis, or when the production of H2O2 exceeds the capacity of the plant to scavenge them, the oxidative stress from lipid peroxidation, and oxidation of protein and DNA occurs37–40. Lipid peroxidation from excessive H2O2 leads to structural abnormalities and cell dysfunction5. Melatonin is a well-documented antioxidant and plays important roles in alleviating environmental stress-induced oxidation20,22. Exogenous application of melatonin has been found to improve plant chilling tolerance. Melatonin played a positive role in Bermudagrass to protect against cold stress through modulating photosynthesis and metabolism related pathways41. Melatonin could protect cucumber seeds and young seedlings against oxidative stress directly and indirectly detoxifying ROS, thereby plants grew better even in harmful environmental conditions42,43. Melatonin-treated rice seedlings inhibited the accumulation of ROS and alleviated the cold stress-induced inhibition to plant growth44. In present study, followed a transient induction of H2O2 content by melatonin, contents of H2O2 and MDA in peach fruit were significantly reduced by the melatonin treatment (Fig. 1b and c), indicating lower lipid peroxide and oxidative damage in the treated peaches. Our results suggested that melatonin treatment could reduce the chilling-induced oxidative stress and thus enhance tolerance against chilling in peach fruit. Similarly, application of melatonin improved plant resistance to cold stress in wheat seedlings in which H2O2 level was much lower than in non- treated ones45.
Plants have evolved enzymatic antioxidant system or the non-enzymatic antioxidant system to take control over the physiological ROS production. Among enzymatic antioxidant defense systems, SOD, the first line of defense against the oxidative stresses, dismutates O2− to O2 and H2O2, while CAT can consequently convert H2O2 to H2O in plant cells7. Previous studies have established that melatonin promotes the activities of antioxidant enzymes to counteract the harmful effects caused by various environmental stresses. For instance, exogenous melatonin alleviated ROS accumulation and cold-induced oxidative damages by directly scavenging ROS and enhancing antioxidative enzymes in bermudagrass18. The applications of exogenous melatonin induced cold tolerance in rice seedlings by enhancing the activities of antioxidative enzymes44. Consistently, in our study, the high transcripts of PpSODs and PpCATs in melatonin-treated peach fruit further highlighted the role of melatonin in improving antioxidant capacity of postharvest peaches under chilling stress (Fig. 2).
Another important and efficient ROS-scavenging pathway is the AsA-GSH cycle, composed of antioxidant enzymes, including APX, monodehydroascorbate reductase (MDHAR), dehydroascorbate reductase (DHAR), and GR, and reducing substances, including AsA and reduced GSH46. After O2− has been reduced by SOD to H2O2, APX catalyzes the conversion of H2O2 to monodihydroascorbate (MDHA) using AsA as substrate and thus removes toxic H2O247. AsA and GSH can be recovered by a few relevant metabolic reactions catalyzed by MDHAR, DHAR and GR. Maintaining the efficient recycling of AsA and GSH via APX, MDHAR, DHAR and GR is crucial to maintaining the redox state at a high level so that they scavenge H2O2 efficiently48,49. There are several lines of evidence suggesting that promoting the AsA-GSH cycle contributes to the increase in cold tolerance of postharvest peaches50,51. Alleviation of chilling by hypobaric treatment was related to its effect on triggering the AsA-GSH cycle by up-regulating transcriptional expression of antioxidant related enzymes52. Melatonin has been also reported to increase the chilling tolerance in cucumber seedlings by accelerating the AsA-GSH cycle to enhance ROS scavenging ability43. In present study, accompanying with more AsA accumulation in melatonin-treated peaches (Fig. 3a), the transcript levels of genes in ASA-GSH cycle including PpAPXs, PpMDHARs, PpDHARs and PpGRs were also higher than in control fruit (Figs 4 and 5). Our results demonstrated that melatonin activated the expression of genes involved in ASA-GSH cycle in postharvest peaches under chilling stress, leading to the higher levels of AsA and subsequently lower H2O2 contents after the transient increase of H2O2 in the treated fruit (Figs 1a and 3a). This study confirmed that the AsA-GSH cycle plays an important role in implementing the function of exogenous melatonin in harvested peach fruit under chilling conditions.
In fact, it has been firmly established that melatonin is in favor of AsA production in plants under various stress conditions18,41,53. In agreement with these findings, despite the upregulation of all the genes involved in AsA-GSH cycle in peach fruit treated with melatonin, only higher AsA content was observed (Fig. 3a), which suggested that AsA may play a more dominant role in ROS scavenging than GSH in melatonin treated fruit. Therefore, the expression profiles of six L-galactose (Gal) pathway-related genes involved in AsA biosynthesis were also analysed in order to explain the higher AsA content in the treated peaches54. In our present study, expression of most of genes in control peach declined with storage time, indicating the loss of capacity to AsA accumulation under chilling stress. However, the transcripts of all these genes were up-regulated in peach fruit treated with melatonin (Fig. 6). It could thus be seen that melatonin might affect the AsA biosynthetic pathway in peaches subjected to chilling and consequently its final content during storage. To the best of our knowledge, this is the first report on the roles of the last six genes in the Gal pathway in AsA biosynthesis for chilling tolerance regulation in melatonin-treated peaches at the molecular level.
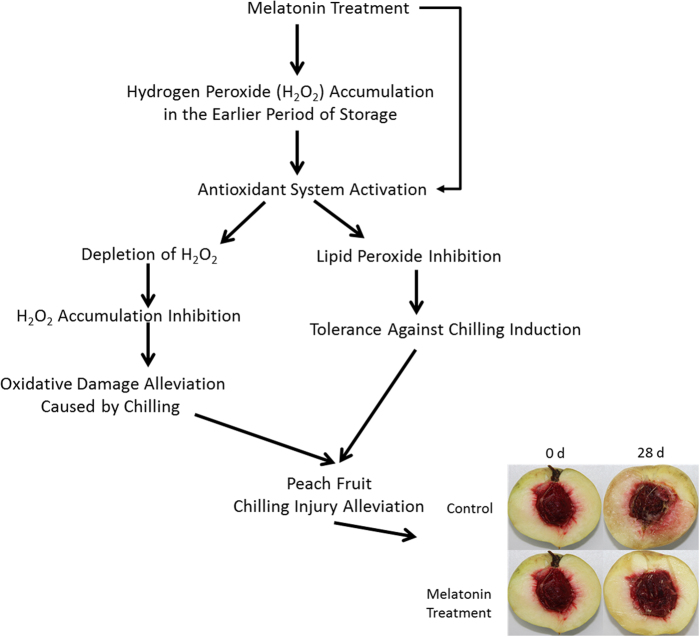
Based on evidence provided herein, a novel model for melatonin and chilling tolerance in postharvest peaches subjected to chilling is proposed (Fig. 7). As mentioned above, treatment with melatonin induced chilling tolerance in peach after harvest. The higher level of H2O2 in melatonin treated peaches in the earlier period of storage might serve as a signal to activate antioxidant system and subsequently induce tolerance against chilling. When the chilling progressed, the activated antioxidant system in turn scavenged excess H2O2, thereby inhibited H2O2 accumulation and alleviated the oxidative damage caused by chilling in the treated peaches, which might be partially accountable for the induced chilling tolerance. However, it is well documented that melatonin can not only directly scavenge some ROS, but can also modulate antioxidant enzymes and enhance cellular antioxidants. Therefore, the induced transcripts of genes involved antioxidant systems and AsA biosynthesis was likely to the results of transiently increased-H2O2 induced by melatonin or the direct effect of melatonin or both. However, as a direct scavenger of ROS, why melatonin enhanced H2O2 accumulation at the initial storage time and which signal pathway was involved still remain unknown, which is deserved to further investigation.
Figure 7.
Proposed model for melatonin-mediated chilling tolerance in postharvest peach fruit.
Methods
Melatonin treatment
Peach fruit (Prunus persica Batsch cv. Hujing) were harvested at commercial maturity from a local orchard in Fenghua, China. Trees were subjected to standard horticultural practices and fruit ripening stage was defined according to the previous study55. Twenty fruit per each of the ten plants at 135 DAFB (days after full bloom) were picked and quickly transferred to the laboratory. The peaches were selected for uniformity without any damage and randomly divided into two groups. The first group was immersed into solutions of 100 µM melatonin for 120 min, whereas the second group of fruit was soaked in sterile deionized water for 120 min and considered as the control. Thereafter, all fruit were air-dried at room temperature for approximately 30 min, then transferred to 4 °C and 80% relative humidity for 28 d. Fruit samples were taken before melatonin treatment (time 0) and at 7 d intervals during storage for measurements of chilling injury index, levels of O2−, H2O2, AsA and GSH and molecular analysis. Each treatment was repeated three times, and the experiment was conducted twice.
Chilling injury index
The degree of chilling was visually assessed on the mesocarp surface, following a double cut parallel to the axial diameter. The extent of flesh browning was divided into four classes: 0, no browning; 1, browning covering <25% of the cut surface; 2, browning covering ≥25% but <50% of cut surface; 3, browning covering ≥50%. Chilling injury index was calculated using the following formula:
Chilling injury index = ∑ [(browning level) × (number of fruit at the browning level)]/(total number of fruit in the treatment).
MDA, O2− and H2O2 analysis
To analysis MDA content, 2 g of flesh tissue was homogenized with 5 mL of 0.5% (w/v) trichloroacetic acid (TCA) and then centrifuged at 10,000 × g for 10 min at 4 °C. MDA content was determined and expressed as μg g−1 FW fresh weight (FW)56. O2− production was measured following previously described procedures57. Briefly, 5 g of flesh tissue was ground in 5 ml of 50 mM phosphate buffer (pH 7.8). The homogenate was centrifuged at 10 000 × g for 20 min at 4 °C. The supernatant was used for the determination of O2− production. O2− production was calculated against the standard curve using sodium nitrite as a standard and expressed as nmol g−1 FW min−1. For H2O2 determination, 2 g of fresh tissue was homogenized with 5 mL of chilled 100% acetone and then centrifuged at 10 000 × g for 20 min at 4 °C. The supernatant was collected immediately for H2O2 analysis by a method based on titanium oxidation58. H2O2 concentration was determined by a standard graph constructed by direct addition of H2O2 to the titanium solution and expressed as mg g−1 FW.
AsA and GSH analysis
To determine the AsA content, 2 g of flesh tissue was homogenized in ice-cold 5% (w/v) trichloroacetic acid (TCA) and then centrifuged at 10,000 × g for 20 min at 4°C. AsA content was measured following previously described procedures59. The results were expressed in μg g−1 FW. For GHS determination, each flesh tissue (2 g) was homogenized in 50 mM sodium phosphate buffer (pH 7.0) and then centrifuged at 10,000 × g for 20 min at 4 °C. The supernatant was used. GSH was assayed following previously described procedures60. The results were expressed in μg−1g FW.
Total RNA extraction and cDNA synthesis
Frozen tissues were carefully grounded in liquid nitrogen. Total RNA was extracted using Plant RNA Kit (Omega Bio-Tek Inc., Norcross, GA, USA) according to the manufacturer’s instructions. The RNA was treated with amplification grade RNase-free DNase1 (Omega Bio-Tek Inc., Norcross, GA, USA) to remove any DNA contamination prior to cDNA synthesis. Reverse transcription (RT) was carried out using 2 µg of total RNA and the SuperRT First Strand cDNA Synthesis Kit (CWBIO, Beijing, China) as recommended by the manufacturer.
Quantitative Real-Time PCR (qPCR)
For each primer pair, a fragment was amplified from cDNA and purified. A 10-fold dilution series of the fragment wa. s prepared over 7 orders of magnitude and used as a template for qRT-PCR in duplicate and generate standard curves. The reaction efficiency (E) of each primer pair was determined using the formula E = 10−1/slopeThe primers used for qRT-PCR are detailed in Table S1. QPCR reactions were performed with an Mx3000P qPCR System (Agilent Stratagene, Santa Clara, CA, USA) and the reactions consisted of 250 nM forward and reverse primer, SYBR Green PCR master mix (Thermo Fisher Scientific Inc., Pittsburgh, PA, USA), and 2 µl of cDNA template (diluted 1 in 5). Two-step qPCR analysis was performed, and the thermal cycling conditions consisted of an initial denaturation at 95 °C for 7 min, and then for 40 cycles as follows: denaturation at 95 °C for 15 s combined with each primer specific annealing temperature ranged from 50 °C to 60 °C for 30 s, then completed with a melting curve analysis program. QPCR data was calibrated relative to PpTEF2 (JQ732180) expression level at zero time for each treatment, following the 2−ΔΔCt method for relative quantification. Three independent biological replicates were analysed for each sample.
Statistical analysis
Experiments were performed using a completely randomized design. All statistical analyses were performed with SPSS package program version 16.0 (SPSS Inc., Chicago, IL, USA). Data were analysed by one-way analysis of variance (ANOVA). Student’s unpaired T test was used to compare the means at P < 0.05.
Electronic supplementary material
Primers Sequences Used for q-PCR and Amplicon Characteristics
Acknowledgements
This study was supported by the National Natural Science Foundation of China (31371866) to ZFY and National Natural Science Foundation of China (31571905) to SFC, the Natural Science Foundation of Zhejiang Province (Q15C200013) to WC and the Natural Science Foundation of Jiangsu Province (BK20171127) to SFC.
Author Contributions
S.C. and Z.Y. designed and performed this experiment, with assistance from J.S., L.S., L.X. and Z.S., S.C., J.S. and W.C. analysed the data and wrote the manuscript. Z.Y. and S.C. provided financial support, the research guidance, critical revision of the manuscript, and approval of the article.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-19363-5.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Crisosto C, Mitchell F, Ju Z. Susceptibility to chilling injury of peach, nectarine, and plum cultivars grown in California. Hort Sci. 1999;34:1116–1168. [Google Scholar]
- 2.Lurie S, Crisosto C. Chilling injury in peach and nectarine. Postharvest Biol. Technol. 2005;37:195–208. doi: 10.1016/j.postharvbio.2005.04.012. [DOI] [Google Scholar]
- 3.Wise RR. Chilling-enhanced photooxidation: the production, action and study of reactive oxygen species produced during chilling in the light. Photosynth. Res. 1995;45:79–97. doi: 10.1007/BF00032579. [DOI] [PubMed] [Google Scholar]
- 4.Hariyadi P, Parkin KL. Chilling-induced oxidative stress in cucumber fruits. Postharvest Biol. Technol. 1991;1:33–45. doi: 10.1016/0925-5214(91)90017-6. [DOI] [Google Scholar]
- 5.Gill SS, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010;48:909–930. doi: 10.1016/j.plaphy.2010.08.016. [DOI] [PubMed] [Google Scholar]
- 6.Xi Z, et al. Effects of 24- epibrassinolide on antioxidation defense and osmoregulation systems of young grapevines (V. Vinifera L.) under chilling stress. Plant Growth Regul. 2013;71:57–65. doi: 10.1007/s10725-013-9809-4. [DOI] [Google Scholar]
- 7.Mittler R. Reactive oxygen gene network of plants. Trends Plant Sci. 2004;9:490–498. doi: 10.1016/j.tplants.2004.08.009. [DOI] [PubMed] [Google Scholar]
- 8.Schieber M, Chandel NS. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014;24:453–462. doi: 10.1016/j.cub.2014.03.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dubbels R, et al. Melatonin in edible plants identified by radioimmunoassay and by high performance performance liquid chromatography-mass spectrometry. J. Pineal Res. 1995;18:28–31. doi: 10.1111/j.1600-079X.1995.tb00136.x. [DOI] [PubMed] [Google Scholar]
- 10.Wang P, et al. Delayed senescence of apple leaves by exogenous melatonin treatment: toward regulating the ascorbate-glutathione cycle. J. Pineal Res. 2012;53:11–20. doi: 10.1111/j.1600-079X.2011.00966.x. [DOI] [PubMed] [Google Scholar]
- 11.Wang P, et al. Delay in leaf senescence of Malus hupehensis by long-term melatonin application is associated with its regulation of metabolic status and protein degradation. J. Pineal Res. 2013;55:424–434. doi: 10.1111/jpi.12069. [DOI] [PubMed] [Google Scholar]
- 12.Wang P, et al. Long-term exogenous application of melatonin delays drought-induced leaf senescence in apple. J. Pineal Res. 2013;54:292–302. doi: 10.1111/jpi.12017. [DOI] [PubMed] [Google Scholar]
- 13.Wang P, et al. Melatonin regulates proteomic changes during leaf senescence in Malus hupehensis. J. Pineal Res. 2014;57:291–307. doi: 10.1111/jpi.12169. [DOI] [PubMed] [Google Scholar]
- 14.Zhang N, et al. The RNA-seq approach to discriminate gene expression profiles in response to melatonin on cucumber lateral root formation. J. Pineal Res. 2014;56:39–50. doi: 10.1111/jpi.12095. [DOI] [PubMed] [Google Scholar]
- 15.Weeda S, et al. Arabidopsis transcriptome analysis reveals key roles of melatonin in plant defense systems. PLoS ONE. 2014;9:e93462. doi: 10.1371/journal.pone.0093462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shi H, Chan Z. The cysteine2/histidine2-type transcription factor ZINC FINGER OF ARABIDOPSIS THALIANA 6-activated C-REPEAT-BINDING FACTOR pathway is essential for melatonin-mediated freezing stress resistance in Arabidopsis. J. Pineal Res. 2014;57:185–191. doi: 10.1111/jpi.12155. [DOI] [PubMed] [Google Scholar]
- 17.Shi H, et al. INDOLE-3-ACETIC ACID INDUCIBLE 17 positively modulates natural leaf senescence through melatonin-mediated pathway in Arabidopsis. J. Pineal Res. 2015;58:26–33. doi: 10.1111/jpi.12188. [DOI] [PubMed] [Google Scholar]
- 18.Shi H, et al. Comparative physiological, metabolomic, and transcriptomic analyses reveal mechanisms of improved abiotic stress resistance in bermudagrass [Cynodon dactylon (L). Pers.] by exogenous melatonin. J. Exp. Bot. 2015;66:681–694. doi: 10.1093/jxb/eru373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee HY, Byeon Y, Back K. Melatonin as a signal molecule triggering defense responses against pathogen attack in Arabidopsis and tobacco. J. Pineal Res. 2014;57:262–268. doi: 10.1111/jpi.12165. [DOI] [PubMed] [Google Scholar]
- 20.Bajwa VS, et al. Role of melatonin in alleviating cold stress in Arabidopsis thaliana. J. Pineal Res. 2014;56:238–245. doi: 10.1111/jpi.12115. [DOI] [PubMed] [Google Scholar]
- 21.Zhang N, et al. Melatonin promotes water-stress tolerance, lateral root formation, and seed germination in cucumber (Cucumis sativus L.) J. Pineal Res. 2013;54:15–23. doi: 10.1111/j.1600-079X.2012.01015.x. [DOI] [PubMed] [Google Scholar]
- 22.Wei W, et al. Melatonin enhances plant growth and abiotic stress tolerance in soybean plants. J. Exp. Bot. 2015;66:695–707. doi: 10.1093/jxb/eru392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meng JF, et al. Melatonin treatment of pre-veraison grape berries to increase size and synchronicity of berries and modify wine aroma components. Food Chem. 2015;185:127–134. doi: 10.1016/j.foodchem.2015.03.140. [DOI] [PubMed] [Google Scholar]
- 24.Gao H, et al. Melatonin treatment delays postharvest senescence and regulates reactive oxygen species metabolism in peach fruit. Postharvest Biol. Tech. 2016;118:103–110. doi: 10.1016/j.postharvbio.2016.03.006. [DOI] [Google Scholar]
- 25.Zhang N, et al. Melatonin improved anthocyanin accumulation by regulating gene expressions and resulted in high reactive oxygen species scavenging capacity in cabbage. Funct. Plant Sci. 2014;41:690–703. doi: 10.1071/FP13306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ma Q, et al. Melatonin attenuates postharvest physiological deterioration of cassava storage roots. J. Pineal Res. 2016;60:424–434. doi: 10.1111/jpi.12325. [DOI] [PubMed] [Google Scholar]
- 27.Aghdam MS, Fard JR. Melatonin treatment attenuates postharvest decay and maintains nutritional quality of strawberry fruits (Fragaria x anannasa cv. Selva) by enhancing GABA shunt activity. Food Chem. 2017;221:1650–1657. doi: 10.1016/j.foodchem.2016.10.123. [DOI] [PubMed] [Google Scholar]
- 28.Sun QQ, et al. A label-free differential proteomics analysis reveals the effect of melatonin on promoting fruit ripening and anthocyanin accumulation upon postharvest in tomato. J. Pineal Res. 2016;61:138–153. doi: 10.1111/jpi.12315. [DOI] [PubMed] [Google Scholar]
- 29.Cao S, et al. Exogenous melatonin treatment increases chilling tolerance and induces defence response in harvested peach fruit during cold storage. J. Agric. Food Chem. 2016;64:5215–5222. doi: 10.1021/acs.jafc.6b01118. [DOI] [PubMed] [Google Scholar]
- 30.Tan DX, et al. Novel rhythms of N1-acetyl-N2-formyl-5-methoxykynuramine and its precursor melatonin in water hyacinth: importance for phytoremediation. FASEB J. 2007;21:1724–1729. doi: 10.1096/fj.06-7745com. [DOI] [PubMed] [Google Scholar]
- 31.Manchester LC, et al. Melatonin: an ancient molecule that makes oxygen metabolically tolerable. J. Pineal Res. 2015;59:403–419. doi: 10.1111/jpi.12267. [DOI] [PubMed] [Google Scholar]
- 32.Neill SJ, et al. Hydrogen peroxide and nitric oxide as signalling molecules in plants. J. Exp. Bot. 2002;53:1237–1247. doi: 10.1093/jexbot/53.372.1237. [DOI] [PubMed] [Google Scholar]
- 33.Suzuki N, et al. ROS and redox signalling in the response of plants to abiotic stress. Plant Cell Environ. 2012;35:259–270. doi: 10.1111/j.1365-3040.2011.02336.x. [DOI] [PubMed] [Google Scholar]
- 34.Knight H, Trewavan AJ, Knight MR. Cold calcium signaling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell. 1996;8:489–503. doi: 10.1105/tpc.8.3.489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Prasad TK, et al. Evidence for chilling induced oxidative stress in maize seedlings and a regulatory role of H2O2. Plant Cell. 1994;6:65–74. doi: 10.1105/tpc.6.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bhattacharjee S. Reactive oxygen species and oxidative burst: roles in stress, senescence and signal transduction in plants. Current Sci. 2005;89:1113–1121. [Google Scholar]
- 37.Mishra S, Jha AB, Dubey RS. Arsenite treatment induces oxidative stress, upregulates antioxidant system, and causes phytochelatin synthesis in rice seedlings. Protoplasma. 2011;248:565–577. doi: 10.1007/s00709-010-0210-0. [DOI] [PubMed] [Google Scholar]
- 38.Srivastava S, Dubey RS. Manganese-excess induces oxidative stress, lowers the pool of antioxidants and elevates activities of key antioxidative enzymes in rice seedlings. Plant Growth Regul. 2011;64:1–16. doi: 10.1007/s10725-010-9526-1. [DOI] [Google Scholar]
- 39.Verma S, Dubey RS. Lead toxicity induces lipid peroxidation and alters the activities of antioxidant enzymes in growing rice plants. Plant Sci. 2003;164:645–655. doi: 10.1016/S0168-9452(03)00022-0. [DOI] [Google Scholar]
- 40.Meriga B, et al. Aluminium-induced production of oxygen radicals, lipid peroxidation and DNA damage in seedlings of rice (Oryza sativa) J. Plant Physiol. 2004;161:63–68. doi: 10.1078/0176-1617-01156. [DOI] [PubMed] [Google Scholar]
- 41.Fan J, et al. Alleviation of cold damage to photosystem II and metabolisms by melatonin in Bermudagrass. Front. Plant Sci. 2015;6:925. doi: 10.3389/fpls.2015.00925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bałabusta M, Szafranska K, Posmyk MM. Exogenous melatonin improves antioxidant defense in cucumber seeds (Cucumis sativus L.) germinated under chilling stress. Front. Plant Sci. 2016;7:575. doi: 10.3389/fpls.2016.00575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhao H, et al. Melatonin increases the chilling tolerance of chloroplast in cucumber seedlings by regulating photosynthetic electron flux and the ascorbate-glutathione cycle. Front. Plant Sci. 2016;7:1814. doi: 10.3389/fpls.2016.01814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Han QH, et al. Effects of melatonin on anti-oxidative systems and photosystem II in cold-stressed rice seedlings. Front. Plant Sci. 2017;8:785. doi: 10.3389/fpls.2017.00785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Turk H, Erdal S. Melatonin alleviates cold-induced oxidative damage in maize seedlings by up-regulating mineral elements and enhancing antioxidant activity. J. Plant Nutr. Soil Sci. 2015;178:433–439. doi: 10.1002/jpln.201400476. [DOI] [Google Scholar]
- 46.Palma JM, et al. Antioxidative enzymes from chloroplasts, mitochondria, and peroxisomes during leaf senescence of nodulated pea plants. J. Exp. Bot. 2006;57:1747–1758. doi: 10.1093/jxb/erj191. [DOI] [PubMed] [Google Scholar]
- 47.May MJ, et al. Glutathione homeostasis in plants: implications for environmental sensing and plant development. J. Exp. Bot. 1998;49:649–667. [Google Scholar]
- 48.Li YL, Liu YF, Zhang JG. Advance in researches on AsA-GSH cycle in horticultural crops. Front. Agric. Chin. 2010;4:84–90. doi: 10.1007/s11703-009-0089-8. [DOI] [Google Scholar]
- 49.Secenji M, et al. Transcriptional differences in gene families of the ascorbate-glutathione cycle in wheat during mild water deficit. Plant Cell Rep. 2010;29:37–50. doi: 10.1007/s00299-009-0796-x. [DOI] [PubMed] [Google Scholar]
- 50.Yang ZF, et al. Combined salicyclic acid and ultrasound treatments for reducing the chilling injury on peach fruit. J. Agric. Food Chem. 2012;60:1209–1212. doi: 10.1021/jf2041164. [DOI] [PubMed] [Google Scholar]
- 51.Yang AP, et al. γ-Aminobutyric acid treatment reduces chilling injury and activates the defence response of peach fruit. Food Chem. 2011;129:1619–1622. doi: 10.1016/j.foodchem.2011.06.018. [DOI] [Google Scholar]
- 52.Song LL, et al. Hypobaric treatment effects on chilling injury, mitochondrial dysfunction, and the ascorbate-glutathione (AsA-GSH) cycle in postharvest peach fruit. J. Agric. Food Chem. 2016;64:4665–4674. doi: 10.1021/acs.jafc.6b00623. [DOI] [PubMed] [Google Scholar]
- 53.Li C, et al. The mitigation effects of exogenous melatonin on salinity induced stress in Malus hupehensis. J. Pineal Res. 2012;53:298–306. doi: 10.1111/j.1600-079X.2012.00999.x. [DOI] [PubMed] [Google Scholar]
- 54.Imai T, et al. L-Ascorbate biosynthesis in peach: cloning of six L-galactose pathway-related genes and their expression during peach fruit development. Physiol. Plant. 2009;136:139–149. doi: 10.1111/j.1399-3054.2009.01213.x. [DOI] [PubMed] [Google Scholar]
- 55.Tonutti P, et al. Ethylene evolution and 1-aminocyclopropane-1-carboxylate oxidase gene expression during early development and ripening of peach fruit. J. Am. Soc. Hortic. Sci. 1997;122:642–647. [Google Scholar]
- 56.Hodges DM, et al. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissue containing anthocyanin and other interfering compounds. Planta. 1999;207:604–611. doi: 10.1007/s004250050524. [DOI] [PubMed] [Google Scholar]
- 57.Cao SF, et al. Effects of 1-methylcyclopropene on oxidative damage, phospholipases and chilling injury of loquat fruit. J. Sci. Food Agric. 2009;89:2214–2220. doi: 10.1002/jsfa.3710. [DOI] [Google Scholar]
- 58.Patterson BD, Mackae EA, Ferguson IB. Estimation of hydrogen peroxide in plant extracts using titanium. Anal. Biochem. 1984;139:487–492. doi: 10.1016/0003-2697(84)90039-3. [DOI] [PubMed] [Google Scholar]
- 59.Kampfenkel K, Van Montague M, Inze D. Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal. Biochem. 1995;225:165–167. doi: 10.1006/abio.1995.1127. [DOI] [PubMed] [Google Scholar]
- 60.Castillo FJ, Greppin H. Extracellular ascorbic acid and enzyme activities related to ascorbic acid metabolism in Sedum album L. leaves after ozone exposure. Environ. Exp. Bot. 1988;28:232–238. doi: 10.1016/0098-8472(88)90033-0. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Primers Sequences Used for q-PCR and Amplicon Characteristics