Abstract.
Digital breast tomosynthesis (DBT) acquires a series of projection images from different angles as an x-ray source rotates around the breast. Such imaging geometry lends DBT naturally to stereoscopic viewing as two projection images with a reasonable separation angle can easily form a stereo pair. This simulation study assessed the efficacy of stereo viewing of DBT projection images. Three-dimensional computational breast phantoms with realistically shaped synthetic lesions were scanned by three simulated DBT systems. The projection images were combined into a sequence of stereo pairs and presented to a stereomatching-based model observer for deciding lesion presence. Signal-to-noise ratio was estimated, and the detection performance with stack viewing of reconstructed slices was the benchmark. We have shown that: (1) stereo viewing of projection images may underperform stack viewing of reconstructed slices for current DBT geometries; (2) DBT geometries may impact the efficacy of the two viewing modes differently: narrow-arc and wide-arc geometries may be better for stereo viewing and stack viewing, respectively; (3) the efficacy of stereo viewing may be more robust than stack viewing to reductions in dose. While in principle stereo viewing is potentially effective for visualizing DBT data, effective stereo viewing may require specifically optimized DBT image acquisition.
Keywords: digital breast tomosynthesis, model observer, stereoscopic viewing, stereo matching, disparity, reconstruction
1. Introduction
Digital breast tomosynthesis (DBT) is an emerging three-dimensional (3-D) breast imaging technique in which an x-ray source rotates over a limited angular span around the breast (usually 15 deg to 50 deg), acquiring a series of projection images (typically 15 to 30) from different angles.1 A stack of reconstructed slices from these DBT projection images offers a reduction of overlapping out-of-plane tissue structures.2 This provides DBT the potential to achieve high diagnostic accuracy.3,4 For example, clinical studies have shown DBT to be advantageous over two-dimensional (2-D) mammography for the detection of noncalcified lesions, particularly in dense breasts.5 There are multiple ways one could visualize 3-D DBT imaging data, and the visualization strategy could significantly impact reader performance. The most common viewing mode for DBT is stack mode, in which 3-D DBT imaging data are displayed as a stack of 2-D reconstructed slices for radiologist to scroll through. As a vast amount of imaging data is generated, one challenging point of interpreting DBT is that radiologists are faced with reviewing hundreds of slices for a four-view exam, which can be repetitive and fatiguing. For example, the average time required for interpreting a DBT exam is greater than that of interpreting a 2-D mammography exam.6,7 Thus, it is necessary to optimize DBT visualizing strategy so that 3-D information of the breast can be perceived accurately and quickly.
This study focused on evaluating an entirely different approach to visualize DBT imaging data: stereoscopic viewing of DBT projection images. The technical background and motivation of this visualization method derives from prior research with stereoscopic digital mammography that consists of two projection images of the breast acquired from two different points of view (6 deg to 10 deg apart).8,9 When the two images of a stereoscopic mammogram are viewed on a stereo display, radiologists are able to view the breast in true depth and see the internal structures in 3-D. Such stereo acquisition and viewing of mammograms has shown to improve the sensitivity of breast cancer detection and reduces unnecessary patient recalls when compared to conventional mammograms.8,9 These advantages of stereoscopic mammography were more evident for dense breasts.8 The imaging geometry of DBT naturally lends itself to stereo visualization, provided that the angular separation between the projection images is carefully selected.2 One of the potential advantages of stereo viewing of DBT projection images over stereoscopic mammograms is that multiple stereo pairs can be formed with DBT projection images, whereas a stereo mammogram only presents one pair. This enables alternative ways of presenting those DBT projection images (e.g., creating a rotating sequence of stereo pairs10).
Unlike the common stack viewing of reconstructed slices, stereo viewing of DBT projection images has the potential to reveal the true 3-D structure of the breast, thus providing additional 3-D visual information to radiologists. A previous study11 showed that radiologists performed significantly better at finding simulated breast masses on DBT projection images viewed on a stereo display than those viewed on 2-D mammograms. Our pilot human observer study12–14 that compared stereoscopic and monoscopic detection of masses also showed that stereoscopic viewing could yield better performance, particularly at sensitivities greater than 90%. Different DBT system designs can yield different image characteristics such as spatial resolution (e.g., Refs. 15 and 16), lesion detectability (e.g., Refs. 17 and 18), and contrast-to-noise ratios (e.g., Ref. 19). An optimal acquisition geometry balances tradeoffs among such image characteristics. Stereo viewing of DBT projection images raises new questions. For example, would presenting more stereo pairs lead to higher lesion detectability? Also, would stereo viewing likely achieve the nearly equivalent detection performance at a reduced total x-ray exposure? These questions remain to be answered.
As conducting psychophysical studies are resource-demanding, it would be valuable to use a reliable numerical model observer as a surrogate. An ideal model observer is desirable and especially valuable during early stages of technology development and optimization, because its performance offers an upper limit of imaging system performance for a given task of interest, and such feasibility measurement is not subject to the impact of human observer variability.20 Prior studies (e.g., Refs. 20–24) have shown that the performance trend of an ideal observer is highly correlated with that of human observers (e.g., a correlation of 0.92 in a tumor detection task with SPECT liver images21). In previous work,25 we developed a stereo matching-based model observer that incorporates the characteristics of stereopsis and approximates the ideal observer performance of stereo viewing in a location-know-exactly binary detection task. With respect to varying configurations of stereo viewing, the performance trends of the model observer were generally consistent with previously reported human observer performance trend of breast lesion detection using stereo viewing.8,9,11,13,14 For example, in the simulation study of detecting 3-D Gaussian objects from volumes of random power-law noise, the model observer achieved the highest accuracy when the two views of a stereo pair were separated by 8 deg. This agreed with the optimal separation angle of a stereo pair reported in a clinical trial of stereoscopic mammography for breast cancer screening.9 We are aware of a few of other model observer studies on task-based assessment of stereoscopic imaging data, particularly Zafar et al. for evaluating stereoscopic display devices.26 However, as these alternative model observers were also designed for trending ideal observer performance, using them rather than our stereo model observer would not alter any qualitative conclusion of our study.
In our prior work,10,27 we investigated the feasibility of stereo viewing of DBT projection images by employing the stereo model observer to detect synthetic breast lesions from simulated DBT images. The preliminary results suggest that breast lesion detectability may be inferior on stereo viewing of DBT projection images relative to stack viewing of reconstructed DBT slices. However, there were several pitfalls in these studies that may have degraded the detection performance of the stereo model observer and limited our ability to draw any definitive conclusion. For example, both studies used simple spheres as the synthetic breast lesions, which are relatively unrealistic and easy to detect. Also, the semiglobal stereo matching algorithm28 sometimes failed to generate an accurate disparity map for a stereo pair of two projection images. In this extended work, we further evaluated the efficacy of stereo viewing with the following three key modifications to the design of the visual assessment: (1) a new set of realistically shaped synthetic lesions;29 (2) a disparity estimation algorithm30 specifically designed for creating cyclopean views from DBT projection images; (3) a multislice channelized Hotelling observer (CHO) with 3-D partial least squares (PLS)31 channels for making detection decisions. We compared the performance of stereo viewing of DBT projection images to that of stack viewing of DBT reconstructed slices. We also compared the performance across three different DBT geometries to identify any performance trend when DBT system configurations change.
2. Materials and Methods
2.1. Image Dataset
2.1.1. Structured image background
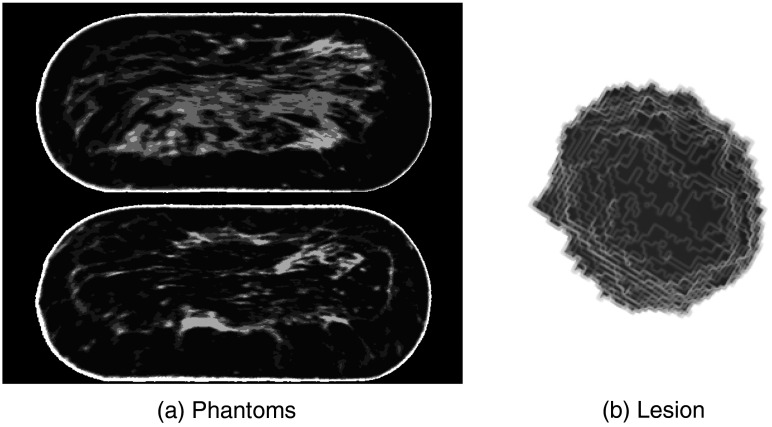
For simulating structured image background, we used a set of patient-based, 3-D anthropomorphic computational phantoms, based upon the dedicated breast-computed tomography imaging data of normal patients.32 The phantoms have been utilized in a number of previous studies of breast imaging system optimization and/or evaluation (e.g., Refs. 27, 33). The voxelized phantoms are generated after being realistically compressed using a finite element model.34 144 different breast phantoms were generated, and they were compressed into , 6, 7, or 8 cm, based on the original breast volume. The phantoms were processed to have an isotropic resolution of . Using the approach of Zeng et al.,23 trilinear interpolation was applied to upsample the phantoms by a factor of 2 to match the phantom voxel size with the detector element size of . Figure 1(a) shows the coronal views of two example breast phantoms with 7 cm compressed thickness.
Fig. 1.
(a) Coronal view of two example breast phantoms with 7 cm compressed thickness. The two phantoms had different spatial distributions of segmented breast tissue classes. (b) Example synthetic breast lesion to be detected. The central mass contained some surface irregularities.
2.1.2. Synthetic breast lesions
We simulated synthetic 3-D breast lesions following the framework in Ref. 29. That is, a modified stochastic Gaussian random sphere (GRS) model was used first to construct the shape of a central mass by defining a parametric surface using the radial distance of the surface from the origin. Varying the statistics of the GRS shapes, controlled by the maximum order of spherical harmonics and the variance of radial distance, led to different levels of surface irregularities. Then, set of low-frequency modifications, denoted as “bumps” and “spikes,” and high-frequency modifications denoted as a “fuzzy” surface texture, were added to modify lesion surface for a greater degree of surface variations (e.g., lobulated surface and pointy surface), and hence better realism. In this study, we used the example combinations of user-defined parameters as listed in Ref. 29 for simulating our lesions. The lesions had an approximate diameter of , and approximate volume of . The size of the lesions was chosen to mimic the typical size of small breast tumors detected in breast cancer patients.35,36 Figure 1(b) shows an example lesion used in the evaluation study.
For each of the 114 lesion-free phantoms, we also created the counterpart lesion-present phantoms by inserting three simulated lesions into three different regions of the breast (i.e., upper inner region, near the nipple, and lower outer region). Hence, for each of the three regions of interest (ROIs), the image ensemble contained a total of 228 cases (i.e., 114 lesion-absent and 114 lesion-present). The contrast between the lesions and the background was set to around to simulate the typical appearance of a lesion embedded in normal breast tissues.11 To quantify local anatomical variations across different breast regions, we estimated the power law noise coefficient at each of the three ROIs by fitting a power function to the anatomical noise power spectrum (NPS) of the ROI.37–39 Specifically, for the central projection image that was acquired vertical to the detector, and the central reconstructed slice that contained the center of the inserted lesion, we extracted the one-dimensional (1-D) NPS by averaging radially over the 2-D NPS, and then performed linear regression on the log-log data for estimating .37
2.1.3. Simulated DBT scanners
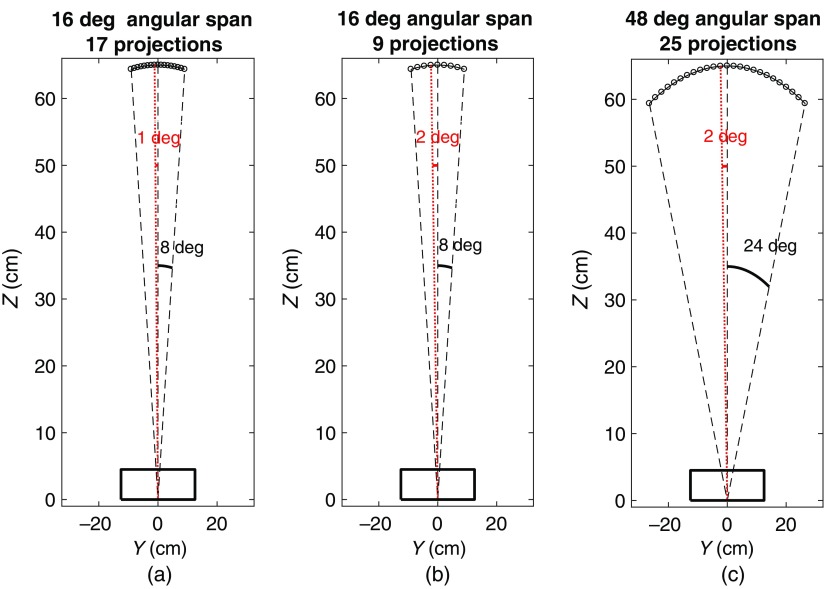
As a starting point to assess the efficacy of stereo viewing, we evaluated three simulated DBT system geometries (Fig. 2) that cover a representative range of geometries across current clinical, prototype, and research systems.1 Specifically, we simulated: (1) two narrow-arc geometries (16 deg of angular span, 17 projections per scan) [Fig. 2(a)] and (16 deg of angular span, nine projections per scan) [Fig. 2(b)] that are similar to that of Hologic Selenia Dimensions system (Hologic, Marlborough, Massachusetts), and GE SenoClair system (General Electric Healthcare, Milwaukee, Wisconsin), respectively, and (2) one wide-arc geometry (48 deg of angular span, 25 projections per scan) [Fig. 2(c)] that mimics the Siemens Inspiration system (Siemens Healthineers, Erlangen, Germany). Each of the three simulated DBT systems had an ideal photon-counting flat panel detector and a point x-ray source that moved along an arc trajectory in a step-and-shoot mode. The distance between the x-ray source and the detector was 65 cm, and the rotation center of the x-ray tube was 6 cm above the detector. When the x-ray source rotated, the detector moved along the plane. The x-ray scatter in photon transport was ignored, and only Poisson noise was considered. A 20-keV monoenergetic x-ray spectrum with a fixed total exposure in all scan settings was simulated. The total amount of exposure at the detector was , which matched with the exposure level measured in similar DBT scan settings (e.g., a mean glandular dose of 1.6 mGy to a 4-cm-thick breast as measured in Ref. 40). These acquisition configurations were similar to the simulated DBT scanners used in previous studies.23,27,41 Besides saving all the projection images for stereo viewing, we also generated stacks of DBT reconstructed slices with the thickness of 1 mm and the in-plane resolution of . To investigate potential impact of different reconstruction algorithms on lesion detection in stack viewing, three reconstruction algorithms were explored: (1) conventional filtered backprojection (FBP) with a Hann filter,42 (2) simultaneous algebraic reconstruction technique (SART),19 and (3) maximum likelihood expectation maximization (MLEM).17 The step size in SART and MLEM reconstruction was manually tuned to achieve a fast convergence of the cost function while still being able to reach a stable solution. The iteration started with an initial guess obtained with back projection, and the maximum number of iterations was set to 20. Figure 3(d) shows an example DBT reconstructed slice using the FBP method.
Fig. 2.
Schematic of the three DBT system geometries that differ in the angular span and the number of projections per scan. (a) and (b) used narrow-arc geometries with 16 deg of angular span, whereas (c) used a wide-arc geometry with 48 deg of angular span. The angular interval between two consecutive projections (e.g., red dotted line and black dash line) indicates the angular increment of the geometry. (a) used 1 deg of angular increment, whereas (b) and (c) used 2 deg of angular increment.
Fig. 3.
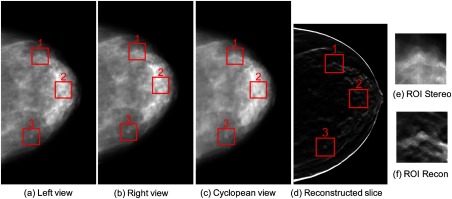
(a, b) An example of a stereo pair with a separation angle of 8 deg. (c) The corresponding cyclopean view constructed from the stereo pair in (a) and (b). (d) An example FBP reconstructed slice. The red boxes in (a)–(d) indicate the locations of the three ROIs on projections, cyclopean view, and reconstructed slices, respectively. (e)–(f) Example ROI 1 extracted from the cyclopean view in (c) and from the reconstructed slice in (d), respectively. The ROIs were of size around the center of the lesions.
2.2. Stereo Model Observer
2.2.1. Cyclopean views
Following the approach described in our previous study,10 we generated a sequence of stereo pairs using DBT projection images. The perceived effect of navigating through the sequence of stereo pairs was rotation of the breast in the stereo display. The two views of one stereo pair, denoted as the left view and the right view , were separated by an angle of 8 deg. We previously demonstrated this separation angle to be optimal for depth perception of the breast.14,25 Cyclopean views, defined as mental images created in the brain by fusing two images received from the two eyes,43 appeared as seen from a virtual eye placed midway between the two eyes. Stereo matching was used to incorporate this property into the model observer.44 First, for each stereo pair, a disparity map , defined as the difference in the location of an object in and was computed by a disparity estimation algorithm. Second, was shifted based on to match with the corresponding pixels in
where was the pixel location and and were the direction of shift in and coordinates. Finally, a 2-D cyclopean view was constructed as the average of the left view and the disparity-compensated right view
Specifically, we used the disparity estimation algorithm proposed by our lab in Muralidhar et al.30 for estimating . Unlike other canonical algorithms (e.g., the semiglobal stereo matching we used in the previous studies10,25,27) that are designed to exploit natural scene statistics, Muralidhar’s disparity estimation algorithm incorporates distinctive characteristics of stereo mammogram images. By adding a steerable multiscale singularity index45 as the constraint in global energy minimization process, it is able to obtain more accurate estimates of disparity along critical curvilinear structures (e.g., vasculature and spicules) that are salient in the breast. Hence, it would help overcome potential issues due to the limited accuracy in matching stereo pairs for cyclopean views. Figures 3(a) and 3(b) show an example stereo pair that was symmetric to the central projection view. Figure 3(c) shows the corresponding estimated cyclopean view for the stereo pair.
2.2.2. Multislice channelized Hotelling observer
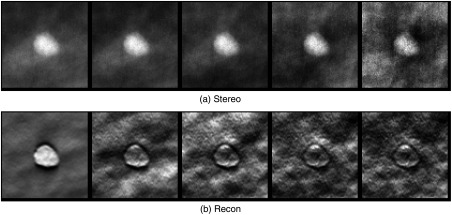
Given a sequence of stereo pairs, the detection task of the model observer is to make a binary decision regarding the presence or absence of the simulated lesion. CHO uses the mean and covariance of channelized image data to optimally differentiate among classes of images.20 It computes a decision statistic that determines whether lesion-presence or lesion-absence is more likely to be true, and the performance of a well-designed CHO (with carefully chosen efficient channels for the given task) approximates the performance of an ideal observer that uses all available statistical information in the images. In this study, through the process of stereo matching discussed in Sec. 2.2, most of the 3-D information conveyed in a stereo pair and is captured by the corresponding 2-D cyclopean view . Thus, the detection task with the sequence of stereo pairs can be simplified into finding the lesion (if any) in the sequence of cyclopean views; in other words, the decision statistic approximates the decision statistics . To efficiently integrate the information from multiple images, our stereo model observer adapted the design of multislice CHO (msCHO)46 that utilizes the channelized multislice data directly for computing a final decision statistics . This approach was chosen for this study because Platiša et al.46 demonstrated that this particular design performed the best among the three existing designs of msCHO. We extracted three ROIs of size around the centers of the lesions on the cyclopean views [Fig. 3(e)], and considered the sequence of the cyclopean view ROIs as the 3-D image data . We applied five 3-D PLS channels31 as the channels for the msCHO. 3-D PLS channels, estimated directly from the images, maximize the covariance between and the truth-label. They have shown to be efficient for approximating the ideal observer in task-based assessments of breast tomosynthesis (e.g., Refs. 31, 41, and 47), as the channels capture not only lesion characteristics and anatomical background statistics but also spatial correlations among multiple slices for making detection decisions. Figure 4(a) shows examples of the estimated PLS channels for stereo viewing. We explored the impact of the number of PLS channels on the performance trend of the msCHO, and the results suggest that increasing did not significantly improve the detection performance (i.e., five channels were sufficient for the detection task).
Fig. 4.
Example of five PLS channels [first (left) to fifth (right) channel] for: (a) the sequence of cyclopean ROIs; (b) the reconstructed ROIs. Results with ROI 3 from the geometry of 16 deg angular span and 17 projections per scan are shown here. PLS channels captured lesion characteristics and local anatomical background.
2.3. Evaluation Metrics
We computed the signal-to-noise ratio (SNR) to quantify the ability of msCHO in making binary classifications.20 SNR is defined as
where () is the mean of under lesion-present () or lesion-absent hypothesis (), and () is the variance of under each hypothesis. Bootstrapping was conducted to obtain a more statistically stable SNR.
For each evaluation trial, we randomly divided the 228 phantom cases into two nonoverlapping sets: of the cases (i.e., 180 cases) for training, and the remaining unseen 20% of the cases (i.e., 48 cases) for testing. In both the training and testing sets, there were equal numbers of lesion-present and lesion-absent cases. Stratified sampling was used for the grouping process, and hence the prevalence of the four breast thickness groups in each subset was approximately the same. For each ensemble of the three ROIs, we trained an msCHO by estimating the PLS channels and the decision templates based on the training data set, and then applied the msCHO to the testing data set for computing the decision statistics. These steps were repeated for 200 evaluation trials, and the average and standard deviation of bootstrap-adjusted SNRs were reported for each of the three ROIs.
One of our main objectives was to compare the efficacy of stereo viewing of projection images to that of stack viewing of reconstructed slices. Accordingly, for each evaluation trial, in addition to measuring the performance of the stereo model observer, we also measured the performance of a benchmark msCHO for stack viewing. This was done as follows: (1) create the counterpart ensemble of a stack of reconstructed ROIs [Fig. 3(f)] based on the same training/testing grouping as for stereo viewing; (2) train the msCHO by estimating five PLS channels [Fig. 4(b)] and the decision template based on the training cases; (3) compute the SNR after applying the msCHO to the testing cases. In this way, for each of the three ROIs, we had paired SNRs for stereo viewing and stack viewing, respectively. Furthermore, to investigate the potential impact of DBT system geometries on the efficacy of stereo viewing, we compared the SNRs across the three geometries (Sec. 2.1.3) to identify performance trends.
3. Results
3.1. Stereo Viewing Versus Stack Viewing
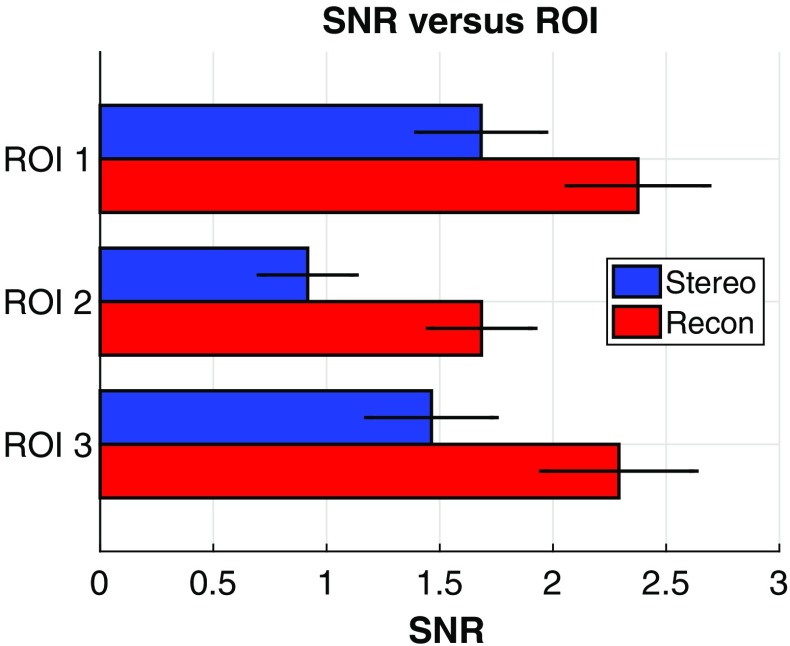
Figure 5 shows the SNRs achieved at the three ROIs using stereo viewing of projection images (blue), and that using stack of FBP reconstructed slices (red). Example results with the DBT geometry of 16 deg angular span and 17 projections per scan demonstrate that the average SNRs for stereo viewing of projection images (blue) were statistically lower than the average SNRs for using stack of reconstructed slices (red). For example, for ROI 3, the SNR for stereo viewing was , which was significantly lower than the SNR of for stack viewing [, ]. These results suggest that stereo viewing of projection images may be less efficient than stack viewing of reconstructed slices.
Fig. 5.
The SNRs achieved at the three ROIs using stereo viewing of projection images (blue), and that using stack viewing of FBP reconstructed slices (red). Results with the DBT geometry of 16 deg angular span and 17 projections per scan are shown here. The SNRs for stereo viewing (blue) were statistically lower than those for stack viewing (red). The SNRs for both stereo viewing and stack viewing were different across the three ROIs.
Table 1 shows the computed power law noise coefficient of the three ROIs. For both projection images and FBP reconstructed slices, ROI 2 near the nipple had the highest , while ROI 1 in the upper outer region had the lowest . As previous research has shown that a higher indicates a higher local breast density,48 ROI 2 was subject to the highest level of local anatomical noise. Hence, the mean SNRs achieved by the model observers at ROI 2 and ROI 1 were the lowest and the highest, respectively (Fig. 5).
Table 1.
of the three ROIs for DBT projection images, and FBP reconstructed slices. ROI 2 near the nipple had the highest and consequently the lowest SNRs achieved by the model observer, while ROI 1 in the upper outer region has the lowest , and the highest SNRs.
| ROI 1 | ROI 2 | ROI 3 | |
|---|---|---|---|
| (projection image) | 2.81 | 3.20 | 3.09 |
| (reconstructed slice) | 2.47 | 2.78 | 2.69 |
3.2. Comparisons across DBT Geometries
3.2.1. Number of projections per scan
We compared the two narrow-arc geometries (16 deg of angular span, 17 projections per scan) and (16 deg of angular span, nine projections per scan) to investigate the impact of number of projections on the efficacy of the two viewing modes. For illustration, we present here the results with ROI 3 and FBP reconstruction, but similar trends are observed with the other two ROIs and reconstruction algorithms. For stereo viewing of projection images, the SNR of for 17 projections per scan was not statistically different from the SNR of for nine projections per scan [, ]. Similarly, for stack viewing of reconstructed slices, the SNR of for 17 projections per scan was not statically different from the SNR of for nine projections per scan [, ].
3.2.2. Angular span
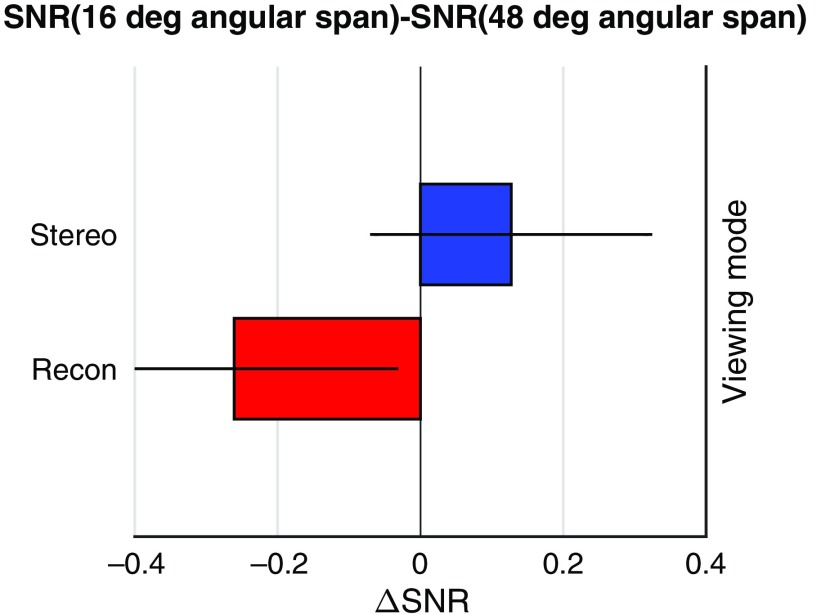
We compared the narrow-arc geometry (16 deg of angular span, 17 projections per scan) and the wide-arc geometry (18 deg of angular span, 25 projections per scan) to investigate the impact of angular span on the efficacy of the two viewing modes. Again, for illustration, we present here the results with ROI 3 and FBP reconstruction (Fig. 6), but similar trends are observed with the other two ROIs and reconstruction algorithms. For stereo viewing of projection images, the SNR of for 16 deg of angular span was significantly higher than the SNR of for 48 deg of angular span [, ]. However, for stack viewing of reconstructed slices, the SNR of for 16 deg of angular span was significantly lower than the SNR of for 48 deg of angular span [, ].
Fig. 6.
Difference in the SNRs achieved with the geometries (16 deg of angular span, 17 projections per scan) and (48 deg of angular span, 25 projections per scan). Results with ROI 3 and FBP reconstruction are shown here. with stereo viewing (blue) was significantly higher than zero, but with reconstructed slices (red) was significantly lower than zero. Narrow-arc geometries may be better for stereo viewing, while wide-arc geometries may be better for stack viewing.
3.3. Impact of Dose Reduction
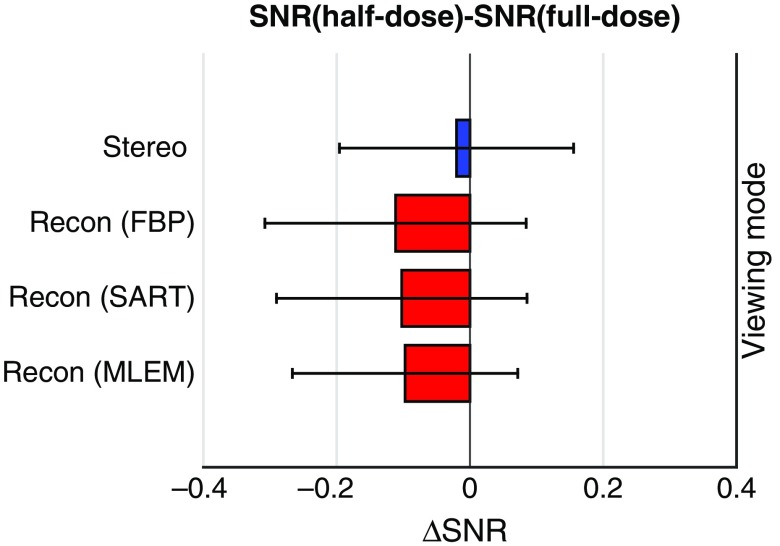
We investigated the impact of reducing the dose on stereo viewing and stack viewing of DBT images. From the image dataset with the DBT geometry (16 deg of angular span, 17 projections per scan), we extracted nine projections per scan (i.e., every other projection that led to an equivalent angular increment of 2 deg), and then used them for reconstruction. Consequently, we generated a new image dataset with an equivalent geometry (16 deg of angular span, nine projections per scan), but the total radiation dose was only of the original dose. We denoted this geometry as “half-dose” (16 deg of angular span, nine projections per scan) to distinguish from the geometry described in Sec. 2.1.3 with “full-dose.” We compared the msCHO performance with the two geometries. Again, for illustration, we present here the results with ROI 3 (Fig. 7), but similar trends are observed with the other two ROIs. For stereo viewing of projection images, the SNR of for “half-dose” nine projections per scan was not statistically different from the SNR of for “full-dose” nine projections per scan [, ]. For stack viewing of FBP and SART reconstructed slices, the SNRs of and for “half-dose” nine projections per scan were significantly lower than the SNRs of and for “full-dose” nine projections per scan, respectively [, for FBP and , for SART]. However, the difference in SNR for MLEM reconstructed images was not statistically different [, ].
Fig. 7.
Difference in the SNRs achieved with the geometries “half-dose” and “full-dose” (16 deg of angular span, nine projections per scan). Results with ROI 3 are shown here. with stereo viewing of projection images (blue), and with stack viewing of MLEM reconstructed slices (red) was statistically equivalent to zero, while with stack viewing of FBP and SART reconstructed slices (red) was significantly lower than zero. If a DBT geometry could be specifically optimized for stereo viewing, it may be possible to reduce the dose required for DBT image acquisition.
4. Discussion
For both stereo viewing of projection images and stack viewing of reconstructed slices, the SNRs across the three ROIs were significantly different (Fig. 5). This agrees with our expectation that the three ROIs with different local anatomical backgrounds would have different levels of lesion detectability. For example, ROI 2 was located closer to the nipple than ROIs 1 and 3 (Fig. 3), which usually contain more dense breast tissues and consequently has a higher local breast density.49 As previous studies (e.g., Ref. 48) have shown that breast density is negatively correlated with the lesion detectability, the SNRs for ROI 2 were the lowest. However, the general performance trends for comparing the two viewing modes were consistent across the three ROIs, and similar results are observed with the other two geometries. Moreover, the differences in the absolute values of SNR between the two viewing modes were also different across the three ROIs (Fig. 5). of the dense breast region ROI 2 was smaller than of the less dense breast regions ROIs 1 and 3. This suggests that stereo viewing might be more valuable for dense breasts than nondense breast. This aligns with the observations in the clinical trial8 that the detection accuracy of stereoscopic mammography was significantly higher than conventional mammography for dense breast subsets, but not for nondense breast subsets. More investigations are needed to investigate the potential of stereo viewing for dense breasts. For example, alternative image presentation methods (e.g., cine loop of stereo pairs50) could be studied.
In Sec. 3.2.1, we observed that for neither viewing mode did the detection performance change when 9 rather than 17 projections per scan were used. With the same total radiation exposure and angular span, increasing the number of projections per scan reduces the radiation exposure of each projection, which leads to a higher quantum noise in the projection images. Although quantum noise affects lesion detectability, especially for small-scale objects such as microcalcifications (e.g., Ref. 17), the variability of anatomical background has been shown as the dominant distracting factor for many detection tasks in breast imaging (e.g., Refs. 51 and 52). Given the total radiation exposure used by the two geometries being compared, the presence of anatomic noise may have overwhelmed the impact of quantum noise.
However, for the simulated “half-dose” geometry in Sec. 3.3 (Fig. 7), the total radiation exposure may be excessively low, and increased quantum noise may be much more noticeable in terms of degrading lesion detectability. Our results suggest that stereo viewing of projection images may be more robust to dose reduction than is stack viewing of reconstructed slices, especially when simple reconstruction algorithm such as FBP is used in stack viewing. These findings imply that stereo viewing has the potential to help reduce the dose required for DBT scans, if DBT image acquisition could be specifically designed to optimize the efficacy of stereo viewing. Many challenging questions require further investigation to be answered. For example, could DBT optimization potentially overcome the observed performance inferiority of stereo viewing? As shown in Sec. 3.2.1, the lesion detectability may not increase monotonically with decreasing number of projections per scan (i.e., increasing dose per projection).
In Sec. 3.2.2 (Fig. 6), we observed conflicting trends of the impact of angular span on the efficacy of the two viewing modes. This may be explained by the principle of parallax.53 With the same separation angle between the two views of a stereo pair, the displacement between the two projection images increases as the projection angle deviates further away from the central projection (i.e., more parallel to the detector). Accordingly, the two projection images would be much less similar from each other, and estimating disparity for fusing the two would be more error-prone and less physically meaningful. Hence, stereo viewing with wide-arc geometry (e.g., 48 deg of angular span) may be less efficient than narrow-arc geometry (e.g., 16 deg of angular span). However, this challenge is overcome in stack viewing of reconstructed slices because reconstruction algorithms typically account for such differences when combining multiple projection images; thus, for stack viewing, a larger angular span usually improves the depth perception of the breast.54 This may explain why the wide-arc geometry significantly outperformed the narrow-arc geometry in stack viewing.
Another limitation of our simulation study was that we extracted only three ROIs, rather than a whole phantom to represent the DBT imaging data. Also, we always made each of the breast lesions located in the center of each ROIs (i.e., the locations of the lesions were exactly known). These simplifications may have made the lesion detection task easier than it actually is, especially when the breast lesions were located in local anatomical backgrounds that help them stand out (e.g., less dense regions, more uniform distributed breast tissues). The model observers were also possibly biased because of the higher lesion prevalence than in reality. For example, in breast cancer screening, most of the cases are normal, and the total analysis time of these cases should be emphasized as an additional factor to be considered. On the other hand, in clinical practice breast lesion locations vary across patients, and lesions can have significantly different characteristics (e.g., shape, size, and symmetry). We set the size of all the synthetic lesions to be relatively small. This was to make the detection task harder for the model observer, mimicking the bigger challenge in detecting small tumors than large tumors.35,55,56 However, the simulated synthetic lesions did not represent the wide range of different types of tumor growth patterns.29 Hence, inclusion of additional mechanisms for handling such characteristics, such as lesion size, shape, and location uncertainty57,58 in the model observer would be helpful and clinically relevant.
5. Conclusions and Future Work
We present herein a model observer study in which we investigated the efficacy of stereoscopic viewing of DBT projection images. We employed the stereo matching msCHOs to detect synthetic lesions in the simulated DBT scans of digital breast phantoms. We showed that stereo viewing of DBT projection images significantly underperformed the benchmark stack viewing of DBT reconstructed slices using contemporary DBT designs. This trend was consistent across the three ROIs that the lesions may appear in the breast, and the trend was also consistent across the three simulated DBT system geometries. We also showed that stereo viewing of projection images would present distinctive challenges to different DBT systems. For the same angular span, changing the number of projections per scan may not change the detection performance with stereo viewing or with of stack viewing, but a narrow-arc geometry may be generally more effective than wide-arc geometry for stereo viewing, whereas a wide-arc geometry may be generally more effective for stack viewing of reconstructed slices. We observed that stereo viewing of projection images may be more robust to dose reduction than is stack viewing of reconstructed slices.
In future work, follow-up studies exploring the efficacy of stereo viewing of DBT projection images are necessary. First, one valuable step would be to verify the model performance with human-observer performance in similar tasks for DBT assessment. For example, experienced radiologists with good stereo perception (e.g., passing the Randot stereoacuity test59,60) could be recruited to interpret simulated DBT images that are displayed as a rotating sequence of stereo pairs. Also, an anthropomorphic module could be added to the stereo model observer to reflect the same internal noise in humans.61,62 This would enable the model observer to consistently predict human observer performance if given the same task of interest. Such psychophysical study would validate the clinical significance of the model observer studies, and may help achieve more definitive conclusions. Second, we only evaluated the viewing modes on cases with constant lesion locations, types, and shapes. However, in clinical practice, lesion locations vary across patients, and lesions have significantly different characteristics. Hence, inclusion of an additional mechanism for handling such lesion and location uncertainty in the model observer would be helpful (e.g., Refs. 57, 58, and 63). Third, the model observers only worked with the individual local ROIs, rather than the whole breast. This shortcoming may be critical for patients with multifocal multicentric breast cancer (i.e., presence of more than two tumors in the same breast).64–66 As locally varying anatomical backgrounds may notably impact lesion detectability,41,67–69 it would be interesting to consider, the impact of multiple lesions, and additional factors such as the distances between lesions. Moreover, similar model observer studies could be conducted to study other questions such as: (1) the efficacy of stereo viewing for detecting other types of breast abnormalities such as clusters of microcalcifications and architectural distortion, (2) the effective use of radiation dose in the context of stereo viewing, and (3) other feasible DBT configurations (not limited to current geometry designs in commercial DBT scanners) and visualization methods (e.g., cine loop of stereo pairs). Whereas this study suggests that it is better to view images acquired using contemporary DBT designs in stack mode rather than in stereo, there may exist DBT designs for which stereo viewing is more efficacious than stack mode.
Acknowledgments
This study was partially supported by NIH grant R01–CA134658 to Dr. Joseph Y. Lo. We thank Dr. Rongping Zeng for providing us the software for the simulated DBT scanner, and Dr. Christian Graff for sharing with us the lesion simulation software. We also thank Mr. Timothy Ho for his contributions to a prior implementation of the disparity estimation algorithm.
Biographies
Gezheng Wen received his BS degree in electronic and communications engineering from the University of Hong Kong in 2011 and his MS and PhD degrees in electrical and computer engineering from the University of Texas at Austin in 2014 and 2017, respectively. His research interests include medical image quality assessment, model observer, x-ray imaging, and visual search of medical images.
Ho-chang Chang is an undergraduate student currently studying electrical and computer engineering at the University of Texas at Austin. He is expected to obtain his BS degree in May 2018.
Jacob Reinhold is a PhD student in electrical engineering at Johns Hopkins University. He graduated from the University of Texas at Austin in 2016 with his BS degree in electrical engineering and worked at Applied Research Laboratories, the University of Texas at Austin, prior to starting graduate studies. His research interests are in biomedical signal and image processing.
Joseph Y. Lo is a professor and associate vice chair for Research of Radiology, Duke University School of Medicine. He is a also professor of biomedical engineering and electrical and computer engineering, Duke University Pratt School of Engineering, and faculty of the Graduate Program in Medical Physics.
Mia K. Markey is a professor of biomedical engineering and engineering foundation endowed faculty fellow in engineering at the University of Texas at Austin as well as adjunct professor of imaging physics at the University of Texas MD Anderson Cancer Center. She is a fellow of both the American Association for the Advancement of Science and the American Institute for Medical and Biological Engineering, a senior member of both the IEEE and SPIE.
Disclosures
No conflicts of interest, financial or otherwise, are declared by the authors.
References
- 1.Sechopoulos I., “A review of breast tomosynthesis. Part I. The image acquisition process,” Med. Phys. 40(1), 014301 (2013). 10.1118/1.4770279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Smith A., “Fundamentals of breast tomosynthesis,” in Hologic Breast Imaging White Papers, Hologic Inc., Bedford, Mass: (2008). [Google Scholar]
- 3.Peppard H. R., et al. , “Digital breast tomosynthesis in the diagnostic setting: indications and clinical applications,” Radiographics 35(4), 975–990 (2015). 10.1148/rg.2015140204 [DOI] [PubMed] [Google Scholar]
- 4.Friedewald S. M., et al. , “Breast cancer screening using tomosynthesis in combination with digital mammography,” J. Am. Med. Assoc. 311(24), 2499–2507 (2014). 10.1001/jama.2014.6095 [DOI] [PubMed] [Google Scholar]
- 5.Alakhras M., et al. , “Digital tomosynthesis: a new future for breast imaging?” Clin. Radiol. 68(5), e225–e236 (2013). 10.1016/j.crad.2013.01.007 [DOI] [PubMed] [Google Scholar]
- 6.Wallis M. G., et al. , “Two-view and single-view tomosynthesis versus full-field digital mammography: high-resolution x-ray imaging observer study,” Radiology 262(3), 788–796 (2012). 10.1148/radiol.11103514 [DOI] [PubMed] [Google Scholar]
- 7.Good W. F., et al. , “Digital breast tomosynthesis: a pilot observer study,” Am. J. Roentgenol. 190(4), 865–869 (2008). 10.2214/AJR.07.2841 [DOI] [PubMed] [Google Scholar]
- 8.D’Orsi C. J., et al. , “Stereoscopic digital mammography: improved specificity and reduced rate of recall in a prospective clinical trial,” Radiology 266(1), 81–88 (2013). 10.1148/radiol.12120382 [DOI] [PubMed] [Google Scholar]
- 9.Getty D. J., Pickett R. M., D’Orsi C. J., “Stereoscopic digital mammography: improving detection and diagnosis of breast cancer,” in Int. Congress Series, Vol. 1230, pp. 538–544, Elsevier; (2001). [Google Scholar]
- 10.Wen G., Markey M. K., “Computational assessment of stereoscopic viewing a sequence of stereo pairs of breast tomosynthesis projection images,” in 36th Annual Int. Conf. of the IEEE Engineering in Medicine and Biology Society (EMBC ’14), pp. 6048–6051, IEEE; (2014). 10.1109/EMBC.2014.6945008 [DOI] [PubMed] [Google Scholar]
- 11.Webb L. J., et al. , “Comparative performance of multiview stereoscopic and mammographic display modalities for breast lesion detection,” Med. Phys. 38(4), 1972–1980 (2011). 10.1118/1.3562901 [DOI] [PubMed] [Google Scholar]
- 12.Muralidhar G. S., et al. , “Stereoscopic viewing of tomosynthesis projection images,” in XIV Conf. on Medical Image Perception Society (MIPS) (2011). [Google Scholar]
- 13.Muralidhar G. S., et al. , “Stereoscopic vs. monoscopic detection of masses on breast tomosynthesis projection images,” Proc. SPIE 8318, 831807 (2012). 10.1117/12.910927 [DOI] [Google Scholar]
- 14.Muralidhar G. S., et al. , “Stereoscopic interpretation of low-dose breast tomosynthesis projection images,” J. Digital Imaging 27(2), 248–254 (2014). 10.1007/s10278-013-9648-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhao B., Zhao W., “Three-dimensional linear system analysis for breast tomosynthesis,” Med. Phys. 35(12), 5219–5232 (2008). 10.1118/1.2996014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hu Y.-H., Zhao B., Zhao W., “Image artifacts in digital breast tomosynthesis: investigation of the effects of system geometry and reconstruction parameters using a linear system approach,” Med. Phys. 35(12), 5242–5252 (2008). 10.1118/1.2996110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Reiser I., Nishikawa R., “Task-based assessment of breast tomosynthesis: effect of acquisition parameters and quantum noise,” Med. Phys. 37(4), 1591–1600 (2010). 10.1118/1.3357288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Das M., et al. , “Evaluation of a variable dose acquisition technique for microcalcification and mass detection in digital breast tomosynthesis,” Med. Phys. 36(6), 1976–1984 (2009). 10.1118/1.3116902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lu Y., et al. , “Image quality of microcalcifications in digital breast tomosynthesis: effects of projection-view distributions,” Med. Phys. 38(10), 5703–5712 (2011). 10.1118/1.3637492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.He X., Park S., “Model observers in medical imaging research,” Theranostics 3(10), 774–786 (2013). 10.7150/thno.5138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gifford H. C., et al. , “Channelized Hotelling and human observer correlation for lesion detection in hepatic spect imaging,” J. Nucl. Med. 41(3), 514 (2000). [PubMed] [Google Scholar]
- 22.Das M., et al. , “Effect of postreconstruction filter strength on microcalcification detection at different imaging doses in digital breast tomosynthesis: human and model observer studies,” Proc. SPIE 8313, 831321 (2012). 10.1117/12.912807 [DOI] [Google Scholar]
- 23.Zeng R., et al. , “Evaluating the sensitivity of the optimization of acquisition geometry to the choice of reconstruction algorithm in digital breast tomosynthesis through a simulation study,” Phys. Med. Biol. 60(3), 1259–1288 (2015). 10.1088/0031-9155/60/3/1259 [DOI] [PubMed] [Google Scholar]
- 24.Zeng R., Badano A., Myers K. J., “Optimization of digital breast tomosynthesis (DBT) acquisition parameters for human observers: effect of reconstruction algorithms,” Phys. Med. Biol. 62(7), 2598–2611 (2017). 10.1088/1361-6560/aa5ddc [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wen G., Markey M. K., Muralidlhar G. S., “A stereo matching model observer for stereoscopic viewing of 3D medical images,” Proc. SPIE 9037, 90370Z (2014). 10.1117/12.2043175 [DOI] [Google Scholar]
- 26.Zafar F., Yesha Y., Badano A., “Computational observers and visualization methods for stereoscopic medical imaging,” Opt. Express 22(19), 22246–22267 (2014). 10.1364/OE.22.022246 [DOI] [PubMed] [Google Scholar]
- 27.Reinhold J., et al. , “Lesion detectability in stereoscopically viewed digital breast tomosynthesis projection images: a model observer study with anthropomorphic computational breast phantoms,” Proc. SPIE 10136, 101360W (2017). 10.1117/12.2255909 [DOI] [Google Scholar]
- 28.Hirschmuller H., “Accurate and efficient stereo processing by semi-global matching and mutual information,” in IEEE Computer Society Conf. on Computer Vision and Pattern Recognition (CVPR ’05), Vol. 2, pp. 807–814, IEEE; (2005). 10.1109/CVPR.2005.56 [DOI] [Google Scholar]
- 29.Sisternes L., et al. , “A computational model to generate simulated three-dimensional breast masses,” Med. Phys. 42(2), 1098–1118 (2015). 10.1118/1.4905232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Muralidhar G. S., Bovik A. C., Markey M. K., “Disparity estimation on stereo mammograms,” IEEE Trans. Image Process. 24(9), 2851–2863 (2015). 10.1109/TIP.2015.2432714 [DOI] [PubMed] [Google Scholar]
- 31.Park S., Zhang G., Myers K. J., “Comparison of channel methods and observer models for the task-based assessment of multi-projection imaging in the presence of structured anatomical noise,” IEEE Trans. Med. Imaging 35(6), 1431–1442 (2016). 10.1109/TMI.2016.2515027 [DOI] [PubMed] [Google Scholar]
- 32.Erickson D. W., et al. , “Population of 224 realistic human subject-based computational breast phantoms,” Med. Phys. 43(1), 23–32 (2016). 10.1118/1.4937597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kiarashi N., et al. , “Impact of breast structure on lesion detection in breast tomosynthesis, a simulation study,” J. Med. Imaging 3(3), 035504 (2016). 10.1117/1.JMI.3.3.035504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sturgeon G. M., et al. , “Finite-element modeling of compression and gravity on a population of breast phantoms for multimodality imaging simulation,” Med. Phys. 43(5), 2207–2217 (2016). 10.1118/1.4945275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Welch H. G., et al. , “Breast-cancer tumor size, overdiagnosis, and mammography screening effectiveness,” N. Engl. J. Med. 375(15), 1438–1447 (2016). 10.1056/NEJMoa1600249 [DOI] [PubMed] [Google Scholar]
- 36.Narod S., et al. , “Are two-centimeter breast cancers large or small?” Curr. Oncol. 20(4), 205 (2013). 10.3747/co.20.1364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen L., Abbey C. K., Boone J. M., “Association between power law coefficients of the anatomical noise power spectrum and lesion detectability in breast imaging modalities,” Phys. Med. Biol. 58(6), 1663–1681 (2013). 10.1088/0031-9155/58/6/1663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Burgess A. E., Judy P. F., “Signal detection in power-law noise: effect of spectrum exponents,” J. Opt. Soc. Am. A 24(12), B52–B60 (2007). 10.1364/JOSAA.24.000B52 [DOI] [PubMed] [Google Scholar]
- 39.Burgess A. E., Jacobson F. L., Judy P. F., “Human observer detection experiments with mammograms and power-law noise,” Med. Phys. 28(4), 419–437 (2001). 10.1118/1.1355308 [DOI] [PubMed] [Google Scholar]
- 40.Zhao B., Zhao W., “Imaging performance of an amorphous selenium digital mammography detector in a breast tomosynthesis system,” Med. Phys. 35(5), 1978–1987 (2008). 10.1118/1.2903425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wen G., Park S., Markey M. K., “Digital breast tomosynthesis for detecting multifocal and multicentric breast cancer: influence of acquisition geometry on model observer performance in breast phantom images,” Proc. SPIE 10136, 101360V (2017). 10.1117/12.2254193 [DOI] [Google Scholar]
- 42.Chawla A. S., et al. , “Optimized image acquisition for breast tomosynthesis in projection and reconstruction space,” Med. Phys. 36(11), 4859–4869 (2009). 10.1118/1.3231814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Julesz B., Foundations of Cyclopean Perception, University of Chicago Press, Oxford, England: (1971). [Google Scholar]
- 44.Scharstein D., Szeliski R., “A taxonomy and evaluation of dense two-frame stereo correspondence algorithms,” Int. J. Comput. Vision 47(1–3), 7–42 (2002). 10.1023/A:1014573219977 [DOI] [Google Scholar]
- 45.Muralidhar G. S., Bovik A. C., Markey M. K., “A steerable, multiscale singularity index,” IEEE Signal Process. Lett. 20(1), 7–10 (2013). 10.1109/LSP.2012.2226027 [DOI] [Google Scholar]
- 46.Platiša L., et al. , “Channelized Hotelling observers for the assessment of volumetric imaging data sets,” J. Opt. Soc. Am. A 28(6), 1145–1163 (2011). 10.1364/JOSAA.28.001145 [DOI] [PubMed] [Google Scholar]
- 47.Zhang G., Myers K. J., Park S., “Investigating the feasibility of using partial least squares as a method of extracting salient information for the evaluation of digital breast tomosynthesis systems,” Proc. SPIE 8673, 867311 (2013). 10.1117/12.2008018 [DOI] [Google Scholar]
- 48.Mainprize J. G., Tyson A. H., Yaffe M. J., “The relationship between anatomic noise and volumetric breast density for digital mammography,” Med. Phys. 39(8), 4660–4668 (2012). 10.1118/1.4736422 [DOI] [PubMed] [Google Scholar]
- 49.Pereira S. M. P., et al. , “The spatial distribution of radiodense breast tissue: a longitudinal study,” Breast Cancer Res. 11(3), R33 (2009). 10.1186/bcr2318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Timberg P., et al. , “Investigation of viewing procedures for interpretation of breast tomosynthesis image volumes: a detection-task study with eye tracking,” Eur. Radiol. 23(4), 997–1005 (2013). 10.1007/s00330-012-2675-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Burgess A. E., “Visual signal detection with two-component noise: low-pass spectrum effects,” J. Opt. Soc. Am. A 16(3), 694–704 (1999). 10.1364/JOSAA.16.000694 [DOI] [PubMed] [Google Scholar]
- 52.Cederström B., Fredenberg E., “The influence of anatomical noise on optimal beam quality in mammography,” Med. Phys. 41(12), 121903 (2014). 10.1118/1.4900611 [DOI] [PubMed] [Google Scholar]
- 53.Rogers B., Graham M., “Similarities between motion parallax and stereopsis in human depth perception,” Vision Res. 22(2), 261–270 (1982). 10.1016/0042-6989(82)90126-2 [DOI] [PubMed] [Google Scholar]
- 54.Sechopoulos I., “A review of breast tomosynthesis. Part II. Image reconstruction, processing and analysis, and advanced applications,” Med. Phys. 40(1), 014302 (2013). 10.1118/1.4770281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Malich A., et al. , “Influence of breast lesion size and histologic findings on tumor detection rate of a computer-aided detection system,” Radiology 228(3), 851–856 (2003). 10.1148/radiol.2283011906 [DOI] [PubMed] [Google Scholar]
- 56.Rauscher G. H., et al. , “Beyond the mammography quality standards act: measuring the quality of breast cancer screening programs,” Am. J. Roentgenol. 202(1), 145–151 (2014). 10.2214/AJR.13.10806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Park S., et al. , “Ideal-observer performance under signal and background uncertainty,” in Biennial Int. Conf. on Information Processing in Medical Imaging, pp. 342–353, Springer; (2003). [DOI] [PubMed] [Google Scholar]
- 58.Popescu L. M., Lewitt R. M., “Small nodule detectability evaluation using a generalized scan-statistic model,” Phys. Med. Biol. 51(23), 6225–6244 (2006). 10.1088/0031-9155/51/23/020 [DOI] [PubMed] [Google Scholar]
- 59.Birch E., et al. , “Randot® preschool stereoacuity test: normative data and validity,” J. Am. Assoc. Pediatr. Ophthalmol. Strabismus 12(1), 23–26 (2008). 10.1016/j.jaapos.2007.06.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fu V. L., Birch E. E., Holmes J. M., “Assessment of a new distance randot stereoacuity test,” J. Am. Assoc. Pediatr. Ophthalmol. Strabismus 10(5), 419–423 (2006). 10.1016/j.jaapos.2006.06.013 [DOI] [PubMed] [Google Scholar]
- 61.Brankov J. G., “Optimization of the internal noise models for channelized Hotelling observer,” in IEEE Int. Symp. on Biomedical Imaging: From Nano to Macro, pp. 1788–1791, IEEE; (2011). [Google Scholar]
- 62.Zhang Y., Pham B. T., Eckstein M. P., “Evaluation of internal noise methods for Hotelling observer models,” Med. Phys. 34(8), 3312–3322 (2007). 10.1118/1.2756603 [DOI] [PubMed] [Google Scholar]
- 63.Banerjee K., et al. , “A visual-search model observer for multitarget volumetric spect images,” in 13th Int. Meeting on Fully Three-Dimensional Image Reconstruction in Radiology and Nuclear Medicine, pp. 114–121 (2015). [Google Scholar]
- 64.Buggi F., et al. , “Multicentric/multifocal breast cancer: overview, biology, and therapy,” in Cell and Molecular Biology of Breast Cancer, pp. 29–42, Springer; (2013). [Google Scholar]
- 65.Neri A., et al. , “Clinical significance of multifocal and multicentric breast cancers and choice of surgical treatment: a retrospective study on a series of 1158 cases,” BMC Surg. 15(1), 1 (2015). 10.1186/1471-2482-15-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kanumuri P., et al. , “Characteristics of multifocal and multicentric breast cancers,” Ann. Surg. Oncol. 22(8), 2475–2482 (2015). 10.1245/s10434-015-4430-6 [DOI] [PubMed] [Google Scholar]
- 67.Wen G., Markey M. K., Park S., “Model observer design for detecting multiple abnormalities in anatomical background images,” Proc. SPIE 9787, 97870S (2016). 10.1117/12.2217665 [DOI] [Google Scholar]
- 68.Wen G., Markey M. K., Park S., “Influence of local anatomical variations on detection of multifocal and multicentric breast cancer,” Med. Phys. 43(6), 3817–3817 (2016). 10.1118/1.4957863 [DOI] [Google Scholar]
- 69.Wen G., Markey M. K., Park S., “Model observer design for multi-signal detection in the presence of anatomical noise,” Phys. Med. Biol. 62(4), 1396 (2017). 10.1088/1361-6560/aa51e9 [DOI] [PubMed] [Google Scholar]