Saalwächter et al. commented on our publication regarding the effect of the disulfide/polysulfide ratio on the healing behavior of natural rubber.1 They argue that our interpretation of self-healing processes by means of Raman spectroscopy and electron-spin resonance (ESR) should be handled with care. We agree that chain interdiffusion plays an important role on the healing behavior of lowly cross-linked rubbers and that more detailed insight and systematic experiments are needed to confirm the role of sulfur-based radicals in self-healing processes. Here, we explain and discuss the correctness of our approach as reported in our original publication.
They also stated that a correlation of the healing efficiency with the cross-linking density would be rationally more convincing than a correlation to the disulfide/polysulfide ratio. They supported this with an illustrative plot. Their “claim” is that one would expect higher potential of chain interdiffusion across a damage zone in a lowly cross-linked sample, thereby displaying higher healing capability. We agree with Saalwächteret al. We had addressed this well-known fact in our manuscript and several of our previous publications.2−7 As we clearly stated in our original paper, one of our conclusions is that the disulfide/polysulfide ratio is not the main parameter in the healing of NR. Paraphrasing ourselves in the original paper: “the limiting factor seems to be the sulfur content and the degree of curing rather than the disulfide/polysulfide ratio”. Interestingly, we did not rule out the interdiffusion hypothesis, and we had stated in all our previous reports including the original paper that interdiffusion is necessary to explain the current results. However, for interdiffusion to occur, the mobility of the relevant species must be present via the opening of disulfide bridges. Therefore, we believe Saalwächter et al. could be correct, but they only highlighted what was described in our paper, which is part of our conclusions but plotted differently.
Saalwächter et al. argued that our Raman-based results need to be re-examined. We understand that the lack of similar studies in the literature makes it difficult to arrive at a definite conclusion (for or against); thus, we presented our data supported by previous research on proteins.8−10 We believe and agree with Saalwächter et al. that more dedicated studies and measurements are needed.
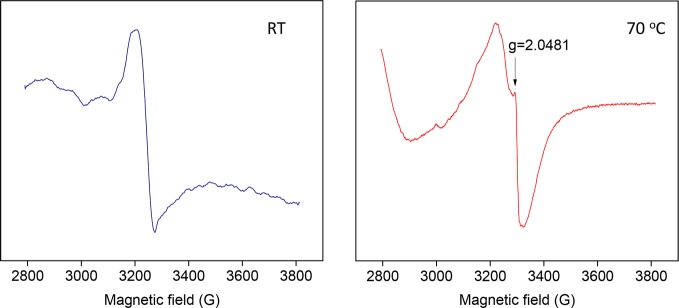
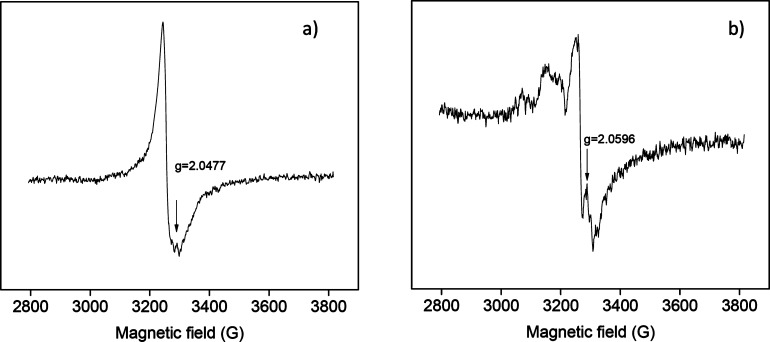
Saalwächter et al. doubted our ESR evidence presented in favor of a sulfur disulfide opening based healing mechanism. We ran ESR tests on our 90% cured NR samples at room temperature and at 70 °C, considering that the latter is the temperature where disulfide exchange reactions take place. In our paper, we discuss the presence of a small resonance at g = 2.0481 at 70 °C assigned to polysulfanyl radicals and the obvious absence of this peak at room temperature. In order to confirm our results, we repeated the tests with samples healed at 70 °C. These samples contain disulfide bonds (epoxy–silane and epoxy–thiol polymers), and they have different chemical structures. The same peak was detected under the same conditions, as evident in Figure S3 in the Supporting Information of the original paper. It is relevant to highlight here that the epoxy–silane sample is a highly cross-linked polymer with disulfides still showing a high degree of healing at 70 °C (healing promoted by the mobility gained due to S–S opening). However, Saalwächter et al. do not consider our multiple tests as sufficient evidence for the sulfur exchange reaction and consequently their influence on the healing mechanism. We understand their doubts, which may not be based on inaccuracy of the method or evaluation, but rather on the obvious process in a scientific study, where limited research data are available.11 We agree that further dedicated experimental study is needed.
Saalwächter et al. have further doubts on the comparison of our results with previous literature data. First, they considered that our similarity with the paper by Dondi et al.12 is misleading. We believe they have misread the literature. Dondi et al. reported that the spectra of sulfur vulcanized polybutadiene in the presence of a sulfenamide type accelerant (just like the one used in our study) possessed three observable components, one of which is centered at g = 2.037 assigned to polysulfanyl radicals. Second, the materials used by Dondi et al.12 and by Posadas et al.13 were not vulcanized. In their case, the vulcanization process was carried out simultaneously with the ESR measurements, and the subsequent detection of polysulfanyl species associated with the curing process occurred at temperatures near the vulcanization temperature (T= 150 °C). In our study, NR samples were 90% vulcanized prior to the ESR tests. Therefore, the detection of the polysulfanyl species (g = 2.0481) at 70 °C (well below the vulcanization temperature) is strong and clear evidence to support that these radicals come from the disulfide exchange reactions and not from the curing process. Third, Posadas et al. clearly stated that “the g-values around 2.004 correspond to carbon or sulfur radicals generated in the course of the cross-linking reaction or during the post vulcanization stage”. None of these situations are applicable to our samples; thus, there is no reason to detect this resonance. We agree that it is too early to make a conclusive statement on the use of ESR to confirm the role of sulfur-based radicals in self-healing systems. The suggestions of Saalwächter et al. for using high-field/high-frequency ESR spectroscopy will be considered in future studies.
Finally, Saalwächter et al. pointed out an error in the x-axis of ESR spectra (Figure 8 of the original paper and Figure S3 of Supporting Information). We checked the calculations and corrected the graphs as shown below.
Figure 8.
ESR spectra of 90%-cured NR compound at room temperature and at 70 °C.
Figure S3.
ESR spectra of disulfide based healing systems: (a) dual organic–inorganic cross-linked sol–gel-based polymer and (b) epoxy-based thermoset.
Acknowledgments
M. Hernández Santana acknowledges the European Commission for a Marie Curie fellowship (PIEF-GA-2013-623379).
The authors declare no competing financial interest.
References
- Hernández M.; Grande A. M.; Dierkes W.; Bijleveld J.; van der Zwaag S.; García S. J. Turning Vulcanized Natural Rubber into a Self-Healing Polymer: Effect of the Disulfide/Polysulfide Ratio. ACS Sustainable Chem. Eng. 2016, 4 (10), 5776–5784. 10.1021/acssuschemeng.6b01760. [DOI] [Google Scholar]
- Lafont U.; van Zeijl H.; van der Zwaag S. Influence of Cross-Linkers on the Cohesive and Adhesive Self-Healing Ability of Polysulfide-Based Thermosets. ACS Appl. Mater. Interfaces 2012, 4 (11), 6280–6288. 10.1021/am301879z. [DOI] [PubMed] [Google Scholar]
- Garcia S. J. Effect of Polymer Architecture on the Intrinsic Self-Healing Character of Polymers. Eur. Polym. J. 2014, 53, 118–125. 10.1016/j.eurpolymj.2014.01.026. [DOI] [Google Scholar]
- Bose R. K.; Hohlbein N.; Garcia S. J.; Schmidt A. M.; van der Zwaag S. Connecting Supramolecular Bond Lifetime and Network Mobility for Scratch Healing in Poly(Butyl Acrylate) Ionomers Containing Sodium, Zinc and Cobalt. Phys. Chem. Chem. Phys. 2015, 17 (3), 1697–1704. 10.1039/C4CP04015E. [DOI] [PubMed] [Google Scholar]
- Susa A.; Bose R. K.; Grande A. M.; van der Zwaag S.; Garcia S. J. Effect of the Dianhydride/Branched Diamine Ratio on the Architecture and Room Temperature Healing Behavior of Polyetherimides. ACS Appl. Mater. Interfaces 2016, 8 (49), 34068–34079. 10.1021/acsami.6b10433. [DOI] [PubMed] [Google Scholar]
- Grande A. M.; Bijleveld J. C.; Garcia S. J.; van der Zwaag S. A Combined Fracture Mechanical - Rheological Study to Separate the Contributions of Hydrogen Bonds and Disulphide Linkages to the Healing of Poly(Urea-Urethane) Networks. Polymer 2016, 96, 26–34. 10.1016/j.polymer.2016.05.004. [DOI] [Google Scholar]
- Abdolah Zadeh M.; Grande A. M.; van der Zwaag S.; Garcia S. J. Effect of Curing on the Mechanical and Healing Behaviour of a Hybrid Dual Network: A Time Resolved Evaluation. RSC Adv. 2016, 6 (94), 91806–91814. 10.1039/C6RA17799A. [DOI] [Google Scholar]
- David C.; Foley S.; Enescu M. Protein S-S Bridge Reduction: A Raman and Computational Study of Lysozyme Interaction with Tcep. Phys. Chem. Chem. Phys. 2009, 11 (14), 2532–2542. 10.1039/b815492a. [DOI] [PubMed] [Google Scholar]
- Vanwart H. E.; Scheraga H. A. Raman-Spectra of Strained Disulfides - Effect of Rotation About Sulfur-Sulfur Bonds on Sulfur-Sulfur Stretching Frequencies. J. Phys. Chem. 1976, 80 (16), 1823–1832. 10.1021/j100557a016. [DOI] [Google Scholar]
- Daly F. P.; Brown C. W. Raman-Spectra of Sodium Tetrasulfide in Primary Amines - Evidence for S42- and S8n- in Rhombic Sulfur-Amine Solutions. J. Phys. Chem. 1975, 79 (4), 350–354. 10.1021/j100571a013. [DOI] [Google Scholar]
- Xiang H. P.; Qian H. J.; Lu Z. Y.; Rong M. Z.; Zhang M. Q. Crack Healing and Reclaiming of Vulcanized Rubber by Triggering the Rearrangement of Inherent Sulfur Crosslinked Networks. Green Chem. 2015, 17 (8), 4315–4325. 10.1039/C5GC00754B. [DOI] [Google Scholar]
- Dondi D.; Buttafava A.; Zeffiro A.; Palamini C.; Lostritto A.; Giannini L.; Faucitano A. The Mechanisms of the Sulphur-Only and Catalytic Vulcanization of Polybutadiene: An Epr and Dft Study. Eur. Polym. J. 2015, 62, 222–235. 10.1016/j.eurpolymj.2014.11.012. [DOI] [Google Scholar]
- Posadas P.; Malmierca M. A.; Gonzalez-Jimenez A.; Ibarra L.; Rodriguez A.; Valentin J. L.; Nagaoka T.; Yajima H.; Toki S.; Che J.; Rong L.; Hsiao B. S. Esr Investigation of Nr and Ir Rubber Vulcanized with Different Cross-Linking Agents. eXPRESS Polym. Lett. 2016, 10 (1), 2–14. 10.3144/expresspolymlett.2016.2. [DOI] [Google Scholar]