Abstract
Certain microRNAs (miRs) regulate the progression and metastasis of various cancer types. In the present study, the role of miR-370 in the progression and proliferation of human astrocytoma and glioblastoma cells was assessed and the underlying molecular mechanism was investigated. miR-370 levels in clinical specimens of human glioma and peritumoral tissues were determined by reverse-transcription quantitative PCR. Oligonucleotide mimics and inhibitors were transfected into the U-251MG human astrocytoma cell line and the and U-87MG glioblastoma cell line and the cell viability of was determined by an MTT assay. The expression of β-catenin and forkhead box protein (FOX)O3a was determined by western blot analysis. The results revealed that the expression of miR-370 in human glioma tissues was significantly decreased compared with that in peritumoral tissues. The miR-370 levels in patients with grade III/IV gliomas were significantly decreased compared with those in grade I/II. Transfection with miR-370 mimics inhibited the proliferation of U-251MG and U-87MG cells. Furthermore, the miR-370 levels were negatively correlated with β-catenin and positively correlated with nuclear FOXO3a. In conclusion, miR-370 inhibited the proliferation of human glioma cells by regulating the levels of β-catenin and the activation of FOXO3a, suggesting that miR-370 was a tumor suppressor in the progression of human astrocytoma and glioblastoma cells.
Keywords: gliomas, microRNA-370, β-catenin, forkhead box protein O3a
Introduction
Human glioma is derived from the neural ectoderm and is the most common type of primary malignant tumor in human brains (1). Also in China, human gliomas are the most common type of intracranial tumor, accounting for 40–50% (2). In recent years, the risk for gliomas has been gradually increasing in young adults (3). On the World Health Organization tumor grading scale, primary gliomas are normally divided into low-grade glioma, such as fibrillary astrocytomas and pilocytic astrocytomas, and high-grade glioma, including glioblastoma and anaplastic astrocytoma (4,5).
Clinical therapy of glioma depends on the size, type, grade and location of the tumor, as well as the age and overall health of the patient, and mainly consists of surgical resection followed by radiotherapy, chemotherapy, Chinese medicine treatment, gene therapy, immunotherapy and psychotherapy (6,7). However, the overall mortality rate remains high in glioma patients (8,9). It is therefore important and urgent to clarify the mechanisms of the genesis of human gliomas, which may be helpful for providing approaches for the therapy of human gliomas.
MicroRNAs (miRNAs/miRs) are a class of small non-coding RNA molecules of approximately 21–25 nucleotides in length, which are conserved in plants, animals and certain types of virus (10). miRNAs are involved in the regulation of gene expression through RNA silencing and post-transcriptional gene silencing via binding to the 3′-untranslated region (3′-UTR) of specific mRNAs (11,12). miRNAs regulate diverse aspects of development and physiology, such as metabolic diseases (13), cardiovascular disease (14,15), immune dysfunction (16,17) and cancer (18,19). A large number of studies have reported that miRNAs are key factors in regulating cell differentiation and growth, migration, apoptosis and necrosis. miR-370 is one of the endogenous non-coding RNAs that has a critical role in carcinogenesis. However, contradictory effects of miR-370 on malignancies have been identified among various human cancer types. miR-370 was reported to function as a tumor suppressor by targeting forkhead box protein (FOX)M1 to inhibit the development and progression of acute myeloid leukemia (20). By contrast, Mollainezhad et al (21) identified miR-370 as an onco-miR and observed six-fold up-regulation of miR-370 in breast cancer tissue compared with that in normal adjacent tissue. Considering that β-catenin was previously reported to be a target of miR-370 (22), and miR-370a is a key oncogene in the progression of numerous human cancers, it was thus assessed in the present study whether there was a regulatory mechanism between miR-370 and FOXO3a in human gliomas. Thus, the present study explored the role of miR-370 in the progression and proliferation of human gliomas and investigated the underlying mechanisms.
Materials and methods
Patients
A total of 16 clinical specimens were obtained by surgical resection of the glioma tissues and collected from Shandong Cancer Hospital affiliated to Shandong University (Jinan, China) between February 2014 and October 2015. The patients were diagnosed by pathological identification. The glioma and peritumoral tissues 2 cm adjacent from the tumors were collected by surgical resection. The tissues were immediately frozen in liquid nitrogen. In compliance with the Helsinki Declaration, the subjects or their families were well informed of the details and signed relevant consent forms prior to the study. The experiment was approved by the Ethics Committee of Shandong Cancer Hospital affiliated to Shandong University (Jinan, China).
Cell lines and agents
Human astrocytes isolated from a human brain (cerebral cortex) were purchased from ScienCell Research Laboratories (cat. no. 1800; San Diego, CA, USA) and cultured in astrocyte medium (cat. no. 1801; ScienCell Research Laboratories). The U-251MG human astrocytoma (grade III glioma) cell line was purchased from Jining Shiye Corp. (cat. no. JN-B1757; Shanghai, China) and the U-87MG glioblastoma cell line was from the American Type Culture Collection (ATCC; Manassas, VA, USA; ATCC® HTB-14™). Recently, the U-87MG ATCC cell line was reported to be misidentified; it is not the original glioblastoma cell line established in 1968 at the University of Uppsala, but is most probably also a glioblastoma cell line whose origin is unknown. Although it is a contaminated cell line, the contamination is unlikely to affect the interpretation of the results or conclusions of the present study (23). The cells were cultured in Dulbecco's modified Eagle's medium (Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal calf serum (Thermo Fisher Scientific, Inc.) with penicillin and streptomycin in a humidified atmosphere containing 5% CO2 at 37°C. MTT agent was obtained from Sigma-Aldrich (Merck KGaA). Lipofectamine 2000 was from Invitrogen (Thermo Fisher Scientific, Inc.). micrON™ Homo sapiens (hsa)-miR-370 mimics (cat. no. miR10000722-1-5) and micrON™ agomir Negative Control #24 (cat. no. miR04201-1-2) were obtained from Ribobio Corp. (Guangzhou, China).
Reverse-transcription quantitative polymerase chain reaction (RT-qPCR) analysis
Total RNA was extracted with an RNApure kit (cat. no. RP1201; Bioteke, Beijing, China) according to the manufacturer's protocols for human glioma, peritumoral tissues, as well as the glioblastoma and astrocytoma cell lines and human normal astrocytes. RT was performed using the PrimeScript-RT reagent kit (cat. no. RR037A; Takara Co., Lt d., Tokyo, Japan) in a 20-ml final reaction volume according to the protocols provided by the manufacturer. The miR-370 levels were determined by a qPCR assay performed using SYBR Premix ExTaq™ II (Takara Co., Ltd.) on ABI 7500 system (Applied Biosystems; Thermo Fisher Scientific, Inc.). Thermal cycling conditions were 30 cycles of 10 sec at 94°C and 1 min at 56°C Each sample was tested in triplicate. Templates and RT were omitted for the negative controls. U6 small nuclear RNA was used for normalization. The sequences of the primers were as follows: hsa-mir-370 forward, 5′-GCCUGCUGGGGUGGAACCUGGU-3′ and reverse, 5′-CAGGUUCCACCCCAGCAGGCUU-3′; U6 forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse, 5′-AACGCTTCACGAATTTGCGT-3′. Expression levels were quantified using the 2−∆∆Cq method (24).
MTT assay
The cell viability was determined by an MTT assay. The U-251MG and U-87MG cell lines were seeded in 48-well plates and following 8 h of incubation, they were transfected with micrON™ hsa-miR-370 mimics and micrON™ agomir Negative Control #24 for 1–5 days. Each sample was set up as two replicates. At the end of the incubation, 5 mg/ml MTT agent was added to each well, followed by culture for 4 h. The purple crystals were dissolved in 100 µl dimethyl sulfoxide. The optical density was determined at a wavelength of 490 nm.
Western blot analysis
The levels of β-catenin and FOXO3a in clinical specimens or astrocytoma and glioblastoma cell lines were determined by western blot analysis as previously described (25,26). The antibodies used in the present study were as follows: mouse anti-β-catenin monoclonal immunoglobulin (Ig)G1 (1:1,000 dilution; cat. no. sc-133239; Santa Cruz Biotechnology, Inc., Dallas, TX, USA), rabbit anti-phosphorylated (p)-FOXO3a (S253) monoclonal IgG (1:1,000 dilution; cat. no. ab154786; Abcam, Cambridge, MA, USA), rabbit FOXO3a monoclonal antibody (1:1,000 dilution; cat. no. 2497; Cell Signaling Technology, Inc., Danvers, MA, USA), mouse lamin B1 monoclonal IgG1 (1:1,000 dilution; cat. no. sc-374015; Santa Cruz Biotechnology, Inc.) and mouse anti-β-actin monoclonal antibody (1:1,000 dilution; cat. no. sc-47778; Santa Cruz Biotechnology, Inc.). The secondary antibodies included horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (cat. no. sc-2004) and HRP-conjugated goat anti-mouse IgG (cat. no. sc-2005; both from Santa Cruz Biotechnology, Inc.).
Statistical analysis
All of the results were analyzed by SPSS software version 20.0 (SPSS, Inc., IBM Corp., Armonk, NY, USA). Two independent groups of samples were performed by t-test. The multiple comparisons were analyzed by analysis of variance and Dunnett's post hoc test. Values are expressed as the mean ± standard deviation. P<0.05 was considered to indicate a statistically significant difference.
Results
miR-370 is downregulated in glioma tissues
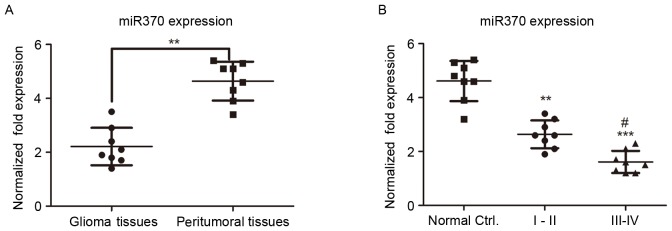
miRs are 21–25 nucleotides in length and are involved in post-transcriptional gene silencing. In order to clarify the role of miR-370 in human glioma, RT-qPCR was used to determine the levels of miR-370 in human glioblastoma and paired peritumoral tissue samples. U6 small nuclear RNA was used as an internal control in the experiment. As presented in Fig. 1A, miR-370 levels were significantly decreased in human glioma tissues compared with those in peritumoral control tissues (P<0.01), suggesting that miR-370 has a tumor suppressor function in gliomas.
Figure 1.
(A) miR-370 levels in 8 clinical specimens of glioma and paired peritumoral tissues as determined by reverse-transcription quantitative polymerase chain reaction. miR-370 levels in glioma tissues were downregulated compared with those in peritumoral tissues. **P<0.01. (B) miR-370 expression in grade I/II and grade III/IV gliomas as well as paired peritumoral tissue was negatively associated with the glioma grade. Values are expressed as the mean ± standard deviation (n=8 per group). **P<0.01 and ***P<0.001, compared with peritumoral tissues.#P<0.05, compared with grade I/II. miR, microRNA; Ctrl., control (peritumoral tissue).
miR-370 expression is negatively associated with the glioma grade
Gliomas are classified into four grades (I–IV); tumor growth is slow in low-grade gliomas (grades I and II) and progresses rapidly in high-grade gliomas (grades III and IV). In order to investigate whether the miR-370 levels were associated with the glioma grade, the miR-370 levels were detected by RT-qPCR in patients with low- and high-grade glioma. As presented in Fig. 1B, the expression of miR-370 in high-grade glioma was decreased compared with that in low-grade glioma tissues, suggesting that miR-370 expression was negatively associated with the glioma grade.
miR-370 expression is decreased in human astrocytoma and glioblastoma cell lines
Next, miR-370 expression in astrocytoma and glioblastoma cell lines was determined by RT-qPCR. Normal human astrocytes were used as normal controls. As presented in Fig. 2, miR-370 was significantly decreased in U-87MG cells and U-251MG cells, compared with that in normal human astrocytes (P<0.01). These results demonstrated that decreased levels of miR-370 were associated with the malignant transformation of astrocytes into glioblastoma and astrocytoma (grade III glioma).
Figure 2.
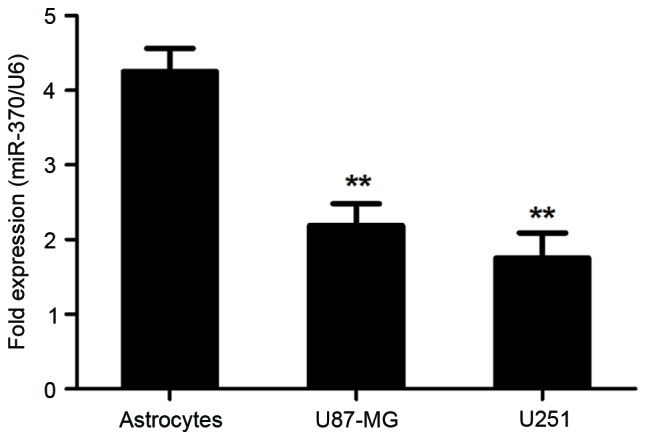
miR-370 expression is decreased in U-87MG human glioblastoma and U-251MG astrocytoma cell lines as well as in astrocytes (normal controls). The levels of miR-370 were detected by reverse-transcription quantitative polymerase chain reaction. **P<0.01, compared with astrocytes. miR, microRNA.
β-catenin is upregulated in human glioma tissues
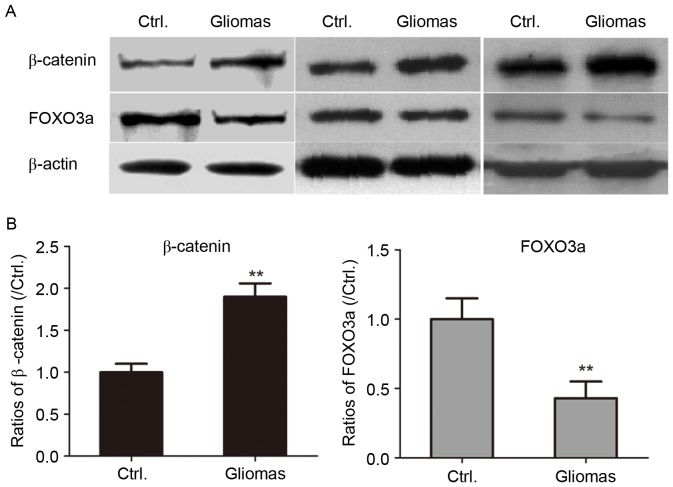
It has been reported that the canonical Wingless-type MMTV integration site family (Wnt)/beta-catenin signaling pathway is aberrantly activated in human gliomas. β-catenin was reported to be significantly associated with the histological malignancy grade and with an unfavorable prognosis for patients with glioma (27). The levels of β-catenin in the glioblastoma and peritumoral control tissue specimens were determined by western blot analysis. As presented in Fig. 3, the expression of β-catenin was significantly upregulated in glioma tissues compared with that in peritumoral tissues (P<0.01).
Figure 3.
Aberrant expression of β-catenin and FOXO3a in human glioma and peritumoral tissues as determined by western blot analysis. (A) Western blot images of three pairs of glioma (grade I/II) and peritumoral tissues are presented. (B) Quantified expression levels of β-catenin and FOXO3a. **P<0.01 compared with paired peritumoral tissues. Ctrl., control; FOX, forkhead box protein.
FOXO3a expression is significantly decreased in human glioma specimens
The Akt/FOXO3a/Bim pathway was reported to be critical in cancer progression in terms of elevation of Akt activity and inactivation of FOXO3a through phosphorylation, which led to downregulation of Bim (28). Therefore, the present study assessed the levels of FOXO3a in glioma and paired peritumoral tissues. As presented in Fig. 3, the levels of FOXO3a in human glioma tissues were significantly decreased compared with those in control specimens (P<0.01).
Transfection with miR-370 mimics suppresses the proliferation of human astrocytoma and glioblastoma cells in vitro
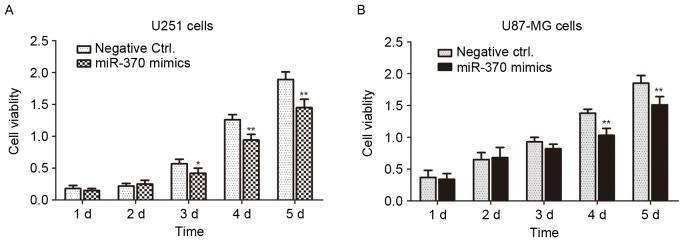
U-251MG and U-87MG cells were used as cell models of astrocytoma and glioblastoma, respectively. The miR-370 mimics and negative controls were transfected into the cells for 1–5 days and the cell viability was determined by an MTT assay. As presented in Fig. 4, the cell viability of miR-370-transfected cells was significantly decreased compared with that of negative control-transfected cells (P<0.01 or P<0.05). Overexpression of miR-370 led to an obvious inhibition of the proliferation of human astrocytoma and glioblastoma cells.
Figure 4.
Transfection with miR-370 mimics suppresses the proliferation of (A) the U-251MG astrocytoma cell line and (B) the U-87MG human glioblastoma cell line. The viability of negative control or miR-370 mimics-transfected cells was determined by an MTT assay at 1, 2, 3, 4 and 5 days. *P<0.05, **P<0.01, compared with negative control group. miR, microRNA; Ctrl., control; d, days.
Transfection of miR-370 mimics inhibits β-catenin expression in U251 cells
In order to clarify whether miR-370 regulates the Wnt/β-catenin signaling pathway in human astrocytoma cells, the U-251MG cell line was transfected with miR-370 mimics or negative controls for 48 h. As presented in Fig. 5, overexpression of miR-370 mimics led to a downregulation of the levels of β-catenin in human astrocytoma cells, as well as c-myc, the downstream target gene of β-catenin. The results revealed that overexpression of miR-370 obviously inhibited the activity of the Wnt/β-catenin signaling pathway in human astrocytoma cells.
Figure 5.
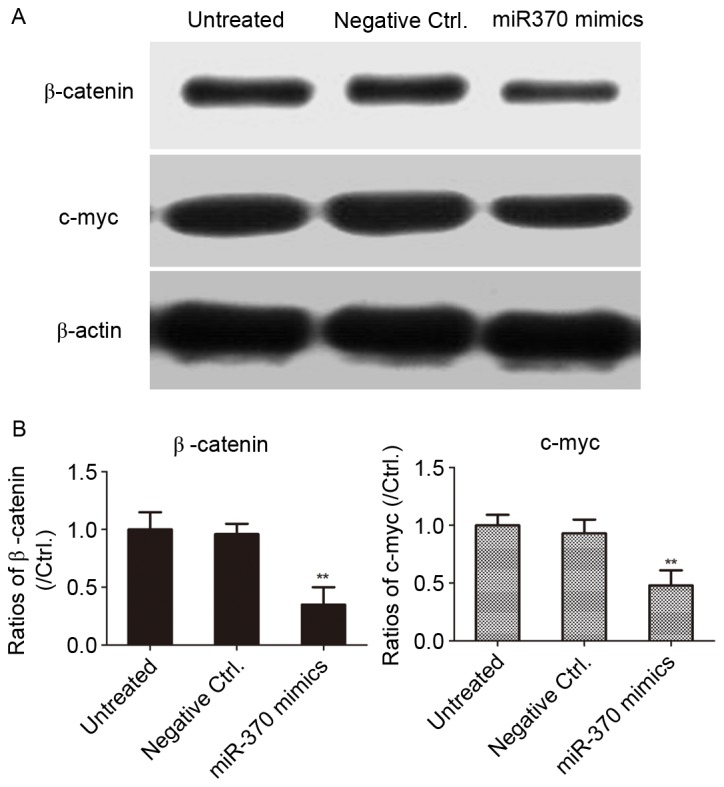
Transfection of miR-370 mimics inhibits β-catenin and c-myc expression in U-251MG cells. (A) Cells were transfected with miR-370 mimics or negative controls for 48 h. The levels of β-catenin and c-myc were determined by western blot analysis. (B) The ratios of β-catenin and c-myc were presented in a bar graph. **P<0.01, compared with the negative control group. miR, microRNA; Ctrl., control.
Transfection of miR-370 mimics leads to a decrease of p-FOXO3a and accumulation of FOXO3a in the nuclei of U251 cells
In order to investigate whether miR-370 was involved in the Akt/FOXO3a signaling pathway, western blot analysis was performed to detect the levels of p-FOXO3a, total FOXO3a and nuclear FOXO3a in human astrocytoma cells. As presented in Fig. 6, the p-FOXO3a levels were obviously decreased in miR-370 mimics-transfected cells; however, overexpression of miR-370 significantly promoted the accumulation of nuclear FOXO3a in U251 cells. These results may suggest that miR-370 inhibited the proliferation of human astrocytoma cells partly by regulating the phosphorylation and cellular localization of FOXO3a.
Figure 6.
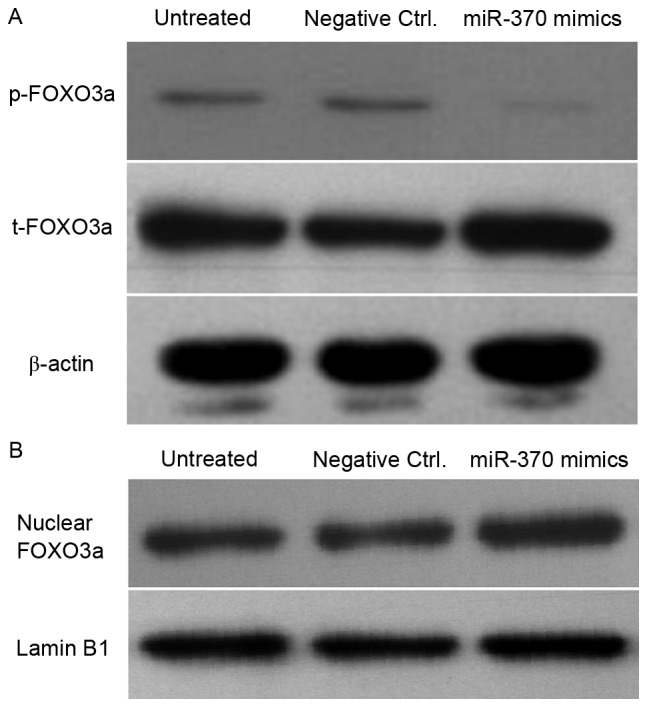
Transfection of miR-370 mimics leads to a decrease of phosphorylated FOXO3a and accumulation of FOXO3a in the nuclei of U-251MG cells. Cells were transfected with miR-370 mimics or negative controls for 48 h. (A) The levels of phosphorylated FOXO3a and total FOXO3a were detected by western blot analysis. β-actin was used as internal reference. (B) Nuclear FOXO3a was determined by western blot analysis in miR-370 mimics- and negative control-transfected cells. Lamin B1 was used as internal reference gene in the experiment. Ctrl., control; FOX, forkhead box protein; miR, microRNA.
Discussion
Human glioma is one of the most malignant primary tumor type in the central nervous system (29). The present study assessed the levels of miR-370 in human glioma and peritumoral tissue. First, clinical specimens from the brains of 16 patients were obtained to detect the levels of miR-370 by RT-qPCR. The results obviously demonstrated that the miR-370 levels in human glioma tissues were significantly decreased compared with those in paired peritumoral tissues. In addition, the levels of miR-370 in clinical specimens of low-grade and high-grade gliomas were detected. Of note, miR-370 levels in grade III/IV glioma tissues were lower than those in in grade I/II specimens. All of these results obviously demonstrated that miR-370 has an important role as a tumor suppressor gene that is associated with the genesis and progression of human gliomas.
Next, the biological role of miR-370 in human astrocytoma and glioblastoma cell line was explored and the molecular mechanism was investigated. RT-qPCR analysis revealed that the levels of miR-370 were obviously decreased in the U-251MG human astrocytoma and the U-87MG glioblastoma cell line cell line compared with that in normal control astrocytes. This was consistent with the results obtained for the clinical specimens, suggesting that miR-370 was decreased in human astrocytoma and glioblastoma cells. Furthermore, oligonucleotide mimics of miR-370 were transfected into U-251MG and U-87MG cells, which inhibited the cell proliferation as demonstrated by an MTT assay. This was consistent with the results of a study by Peng et al (22), which reported that miR-370-3p inhibited human astrocytoma and glioblastoma cell proliferation and induced cell cycle arrest by directly targeting β-catenin. While this abovementioned and the present study were on miR-370 in human astrocytoma and glioblastoma cell proliferation, the present study also detected the expression of β-catenin and FOXO3a, which were two key factors in the progression of gliomas, apart from detecting miR-370 levels in clinical specimens. However, Peng et al (22) used a luciferase reporter assay to test the direct binding of miR-370-3p to a sequence from the 3′-UTR of β-catenin mRNA and also focused on cell cycle analysis, revealing that miR-370-3p induced cell cycle arrest at G0/G1 phase. The present study revealed that miR-370 mimics inhibited β-catenin/c-myc signaling. Furthermore, the phosphorylated and total FOXO3a levels as well as the nuclear localization of FOXO3a were assessed in miR-370 mimics-transfected cells, revealing that miR-370 mimics decreased the phosphorylation of FOXO3a promoted the accumulation of nuclear FOXO3a in U-251MG cells.
The Wnt/β-catenin and the Akt/FOXO3a signaling pathway are two important regulating pathways in human astrocytoma and glioblastoma cells. Aberrant expression of β-catenin and FOXO3a was found in astrocytoma and glioblastoma cells by western blot analysis. A reciprocal association between miR-370 and β-catenin and a positive association between miR-370 and FOXO3a were detected by western blot analysis. Of note, transfection of miR-370 mimics inhibited β-catenin expression in U-251MG cells. However, transfection of miR-370 mimics contributed to a decrease of p-FOXO3a and nuclear accumulation of FOXO3a in U251 cells. Thus, it was speculated that miR-370 inhibited the proliferation of human astrocytoma and glioblastoma cells by regulating β-catenin as well as FOXO3a-associated signaling pathways. All of the results of the present study demonstrated that miR-370 may be a tumor suppressor whose downregulation has a role in the genesis and progression of human astrocytoma and glioblastoma. miR-370 may represent a novel target for the molecular therapy of human astrocytoma and glioblastoma.
References
- 1.Hirst TC, Vesterinen HM, Conlin S, Egan KJ, Antonic A, Lawson McLean A, Macleod MR, Grant R, Brennan PM, Sena ES, Whittle IR. A systematic review and meta-analysis of gene therapy in animal models of cerebral glioma: Why did promise not translate to human therapy? Evid Based Preclin Med. 2014;1:e00006. doi: 10.1002/ebm2.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wang X, Chen JX, Zhou Q, Liu YH, Mao Q, You C, Chen N, Xiong L, Duan J, Liu L. Statistical report of central nervous system tumors histologically diagnosed in the sichuan province of China from 2008 to 2013: A west China glioma center report. Ann Surg Oncol. 2016;23(Suppl 5):S946–S953. doi: 10.1245/s10434-016-5410-1. [DOI] [PubMed] [Google Scholar]
- 3.Bauer R, Kaiser M, Stoll E. A computational model incorporating neural stem cell dynamics reproduces glioma incidence across the lifespan in the human population. PLoS One. 2014;9:e111219. doi: 10.1371/journal.pone.0111219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Matyja E, Grajkowska W, Stepien K, Naganska E. Heterogeneity of histopathological presentation of pilocytic astrocytoma-diagnostic pitfalls. A review. Folia Neuropathol. 2016;54:197–211. doi: 10.5114/fn.2016.62530. [DOI] [PubMed] [Google Scholar]
- 5.Borghei-Razavi H, Shibao S, Schick U. Prechiasmatic transection of the optic nerve in optic nerve glioma: Technical description and surgical outcome. Neurosurg Rev. 2017;40:135–141. doi: 10.1007/s10143-016-0747-2. [DOI] [PubMed] [Google Scholar]
- 6.Luderer MJ, Muz B, de la Puente P, Chavalmane S, Kapoor V, Marcelo R, Biswas P, Thotala D, Rogers B, Azab AK. A hypoxia-targeted boron neutron capture therapy agent for the treatment of glioma. Pharm Res. 2016;33:2530–2539. doi: 10.1007/s11095-016-1977-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bouffet E, Ramaswamy V. Old chemotherapy makes a comeback: Dual alkylator therapy for pediatric high-grade glioma. Neuro Oncol. 2016;18:1333–1334. doi: 10.1093/neuonc/now200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cordier D, Krolicki L, Morgenstern A, Merlo A. Targeted radiolabeled compounds in glioma therapy. Semin Nucl Med. 2016;46:243–249. doi: 10.1053/j.semnuclmed.2016.01.009. [DOI] [PubMed] [Google Scholar]
- 9.Barajas RF, Jr, Butowski NA, Phillips JJ, Aghi MK, Berger MS, Chang SM, Cha S. The development of reduced diffusion following bevacizumab therapy identifies regions of recurrent disease in patients with high-grade glioma. Acad Radiol. 2016;23:1073–1082. doi: 10.1016/j.acra.2016.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cai Y, Yu X, Hu S, Yu J. A brief review on the mechanisms of miRNA regulation. Genomics Proteomics Bioinformatics. 2009;7:147–154. doi: 10.1016/S1672-0229(08)60044-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu X, Yang X, Xing C, Zhang S, Cao J. miRNA: The nemesis of gastric cancer (Review) Oncol Lett. 2013;6:631–641. doi: 10.3892/ol.2013.1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ma H, Wu Y, Yang H, Liu J, Dan H, Zeng X, Zhou Y, Jiang L, Chen Q. MicroRNAs in oral lichen planus and potential miRNA-mRNA pathogenesis with essential cytokines: A review. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122:164–173. doi: 10.1016/j.oooo.2016.03.018. [DOI] [PubMed] [Google Scholar]
- 13.Mehta R, Otgonsuren M, Younoszai Z, Allawi H, Raybuck B, Younossi Z. Circulating miRNA in patients with non-alcoholic fatty liver disease and coronary artery disease. BMJ Open Gastroenterol. 2016;3:e000096. doi: 10.1136/bmjgast-2016-000096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Srinivasan H, Das S. Mitochondrial miRNA (MitomiR): A new player in cardiovascular health. Can J Physiol Pharmacol. 2015;93:855–861. doi: 10.1139/cjpp-2014-0500. [DOI] [PubMed] [Google Scholar]
- 15.Daimiel-Ruiz L, Klett-Mingo M, Konstantinidou V, Micó V, Aranda JF, García B, Martínez-Botas J, Dávalos A, Fernández-Hernando C, Ordovás JM. Dietary lipids modulate the expression of miR-107, a miRNA that regulates the circadian system. Mol Nutr Food Res. 2015;59:1865–1878. doi: 10.1002/mnfr.201400616. [DOI] [PubMed] [Google Scholar]
- 16.Zhou X, Jeker LT, Fife BT, Zhu S, Anderson MS, McManus MT, Bluestone JA. Selective miRNA disruption in T reg cells leads to uncontrolled autoimmunity. J Exp Med. 2008;205:1983–1991. doi: 10.1084/jem.20080707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shaked A, Chang BL, Barnes MR, Sayre P, Li YR, Asare S, DesMarais M, Holmes MV, Guettouche T, Keating BJ. An ectopically expressed serum miRNA signature is prognostic, diagnostic, and biologically related to liver allograft rejection. Hepatology. 2017;65:269–280. doi: 10.1002/hep.28786. [DOI] [PubMed] [Google Scholar]
- 18.Cramer DW, Elias KM. A prognostically relevant miRNA signature for epithelial ovarian cancer. Lancet Oncol. 2016;17:1032–1033. doi: 10.1016/S1470-2045(16)30149-8. [DOI] [PubMed] [Google Scholar]
- 19.Lin YC, Lin JF, Tsai TF, Chou KY, Chen HE, Hwang TI. Tumor suppressor miRNA-204-5p promotes apoptosis by targeting BCL2 in prostate cancer cells. Asian J Surg. 2017;40:396–406. doi: 10.1016/j.asjsur.2016.07.001. [DOI] [PubMed] [Google Scholar]
- 20.Zhang X, Zeng J, Zhou M, Li B, Zhang Y, Huang T, Wang L, Jia J, Chen C. The tumor suppressive role of miRNA-370 by targeting FoxM1 in acute myeloid leukemia. Mol Cancer. 2012;11:56. doi: 10.1186/1476-4598-11-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mollainezhad H, Eskandari N, Pourazar A, Salehi M, Andalib A. Expression of microRNA-370 in human breast cancer compare with normal samples. Adv Biomed Res. 2016;5:129. doi: 10.4103/2277-9175.186987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peng Z, Wu T, Li Y, Xu Z, Zhang S, Liu B, Chen Q, Tian D. MicroRNA-370-3p inhibits human glioma cell proliferation and induces cell cycle arrest by directly targeting β-catenin. Brain Res. 2016;1644:53–61. doi: 10.1016/j.brainres.2016.04.066. [DOI] [PubMed] [Google Scholar]
- 23.Allen M, Bjerke M, Edlund H, Nelander S, Westermark B. Origin of the U87MG glioma cell line: Good news and bad news. Sci Transl Med. 2016;8:354re3. doi: 10.1126/scitranslmed.aaf6853. [DOI] [PubMed] [Google Scholar]
- 24.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 25.Yang M, Pi H, Li M, Xu S, Zhang L, Xie J, Tian L, Tu M, He M, Lu Y, et al. From the Cover: Autophagy induction contributes to cadmium toxicity in Mesenchymal stem cells via AMPK/FOXO3a/BECN1 signaling. Toxicol Sci. 2016;154:101–114. doi: 10.1093/toxsci/kfw144. [DOI] [PubMed] [Google Scholar]
- 26.Sun G, Hou YB, Jia HY, Bi XH, Yu L, Chen DJ. MiR-370 promotes cell death of liver cancer cells by Akt/FoxO3a signalling pathway. Eur Rev Med Pharmacol Sci. 2016;20:2011–2019. [PubMed] [Google Scholar]
- 27.Denysenko T, Annovazzi L, Cassoni P, Melcarne A, Mellai M, Schiffer D. WNT/β-catenin signaling pathway and downstream modulators in low- and high-grade glioma. Cancer Genomics Proteomics. 2016;13:31–45. [PubMed] [Google Scholar]
- 28.Guan H, Song L, Cai J, Huang Y, Wu J, Yuan J, Li J, Li M. Sphingosine kinase 1 regulates the Akt/FOXO3a/Bim pathway and contributes to apoptosis resistance in glioma cells. PLoS One. 2011;6:e19946. doi: 10.1371/journal.pone.0019946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Amberger VR, Hensel T, Ogata N, Schwab ME. Spreading and migration of human glioma and rat C6 cells on central nervous system myelin in vitro is correlated with tumor malignancy and involves a metalloproteolytic activity. Cancer Res. 1998;58:149–158. [PubMed] [Google Scholar]