Abstract
The association of parathyroid hormone (PTH) and vitamin D with Metabolic syndrome (MetS) was evaluated using representative data from the Korean population. Data from 7004 subjects aged 50 or older with preserved renal function (excluding chronic kidney disease stage 3b to 5) who were included in the Korean National Health and Nutrition Examination Survey between 2008 and 2010 were analysed. Higher PTH levels (pg/ml) were observed in subjects with MetS than in those without MetS among both genders (60.1 (58.6–61.6) vs. 62.4 (60.7–64.2) in males p = 0.018, 60.7 (59.4–62.1) vs. 63.9 (62.4–65.6) in females, p < 0.001). For females, PTH levels were significantly higher in subjects with MetS than in those without MetS after adjustment for possible covariates. Lower 25(OH)D levels were significantly associated with MetS only in male subjects (p = 0.004). As the number of MetS components increased, a significant rise in PTH levels (p for trend 0.005 in males and 0.024 in females) and a decrease in 25(OH)D levels (p for trend < 0.001 in males and 0.053 in females) were observed. In conclusion, among subjects with preserved renal function, PTH levels were possibly associated with MetS in females, whereas vitamin D levels exhibited a possible link to MetS in males.
Introduction
Parathyroid hormone (PTH) and vitamin D are the primary regulators of calcium homeostasis. In addition to their conventional role, both have been associated with insulin resistance in subjects with and without primary hyperparathyroidism (PHP)1, and an inverse relationship between PTH and insulin sensitivity was observed2. Vitamin D has also been suggested to be related to beta cell function3, and insulin resistance is related to vitamin D status4,5.
Metabolic syndrome (MetS), a cluster of symptoms that include hypertension, high blood glucose and triglyceride levels, low high-density lipoprotein (HDL) cholesterol, and increased waist circumference, is suspected to be related to PTH status or vitamin D levels, as insulin resistance is an underlying mechanism. Several studies have evaluated the relationship between PTH/vitamin D and MetS in large population-based datasets4,6–15, but the results were inconsistent. This inconsistency may be due to the heterogeneity of the included study populations and confounding factors in the analysis. Specifically, with the exception of two studies6,10, the aforementioned large-scale studies did not include exclusion criteria for subjects with reduced glomerular filtration rates (GFR). Recent results from the Korean National Health and Nutrition Examination Survey (KNHANES) had no exclusion criteria for subjects with renal impairment, and the status of renal function based on the GFR was not considered in the evaluation of the relationship between PTH/vitamin D and MetS15.
However, the status of both PTH and vitamin D is closely related to impaired renal function. For example, a steep increase in serum PTH levels during moderate chronic kidney disease (CKD) (GFR <60 ml/min/1.73 m2) is well known16. Vitamin D deficiency, which is a stimulator of PTH secretion, has been frequently observed in subjects with CKD7. As an insufficient vitamin D status in CKD subjects is associated with unfavourable outcomes, vitamin D supplementation along with calcium in cases of secondary hyperparathyroidism is recommended by clinical guidelines17. The incidence of MetS was also observed to increase with a decline in the GFR in a study examining a nationally representative population18. Besides CKD 4 and 5, CKD stage 3b (GFR <45 ml/min/1.73 m2) patients exhibited an increased risk of MetS compared to stage 3a (45 ≤ GFR <60 ml/min/1.73 m2) patients19. Therefore, exclusion of subjects with impaired renal function is necessary for the evaluation of the association of PTH/vitamin D with MetS. In addition, because gender could affect body composition changes during ageing20 and calcium homeostasis21, and both of these criteria can possibly influence PTH and vitamin D, the associations of PTH and vitamin D with MetS were evaluated separately for each gender.
The aim of this study is to elucidate the associations of PTH and vitamin D with MetS in subjects with normal kidney function using representative datasets of the Korean population.
Methods
Study population
The study examined data from subjects aged 50 years or older from the fourth and fifth KNHANES (n = 10,238) collected from 2008 to 2010. The survey was cross-sectional and representative of non-institutionalized Korean people. The KNHANES, which was designed based on stratified multistage probability sampling, is conducted by the Korean Centre for Disease Control and Prevention. The detailed design of sampling and variables are described elsewhere22. The collected data consisted of information about health and nutritional status as well as socioeconomic status. Subjects with fasting times less than 8 hours before laboratory tests, treatment for osteoporosis, or missing covariates were excluded. In addition, subjects with stage 3b to 5 CKD (GFR ≤45 ml/min/1.73 m2, moderate to severe CKD) were excluded from the study. Finally, data from 7004 subjects were analysed. All participants provided written informed consent for the survey, and the study protocol was approved by Institutional Review Board of The Catholic University of Korea (No. PC16EISI0037). The study was conducted based on the Declaration of Helsinki for biomedical research.
Demographic characteristics and Survey on status of health and nutrition
Demographic data and medical histories were obtained from self-reported questionnaires and personal interviews by trained personnel. A 24-hour recall method and Can-Pro 2.0 nutrient intake assessment software, developed by the Korean Nutrition Society, were used for assessment of dietary intake and daily energy intake, respectively. Regular exercise was defined as exercise for at least 20 minutes 3 times per week with moderate to severe intensity.
Drinking status was classified as non-to-moderate drinking and heavy drinking. Heavy drinking was defined as intake of more than 30 g/day of alcohol. Smoking status was divided into current smokers and non-smokers (including ex-smokers). Low educational status was defined as those with elementary to middle school as their highest academic achievement. The lowest quartile of income was defined as low income.
Anthropometric and laboratory measurements
Height, body weight, and waist circumference were measured by a standardized protocol23. Briefly, waist circumference was defined as the distance from the midline of the lower rib margin to the iliac crest. The weight and height of subjects were measured with light clothing after removing their shoes. The weight (kg) per square of height (m2) was calculated as the BMI. Blood pressure was measured three times using a mercury sphygmomanometer (Baumanometer; W.A. Baum Co. Inc., Copiague, NY, USA) with patients in a seated position after 5 minutes of rest.
For laboratory measurements, all subjects fasted for at least 8 hours before sampling. The samples were processed immediately and then refrigerated for transportation to the central testing institute (NeoDin Medical Institute, Seoul, Korea). Detailed information about measurement methods for laboratory parameters including glucose, cholesterol, PTH and serum 25-hydroxyvitamin D (25(OH)D), which reflects the status of vitamin D, were described in previous studies5,15. The Friedewald formula was used for the calculation of low-density lipoprotein (LDL) cholesterol24. The modification of diet in renal disease (MDRD) equation was used to calculate the estimated GFR (eGFR)25.
Definition of metabolic syndrome and its components
The criteria for the presence of MetS were determined based on the criteria suggested by the American Heart Association (AHA)/National Heart, Lung, and Blood Institute (NHLBI) and the World Health Organization (WHO) Asia–Pacific region criteria for abdominal obesity. Specifically, MetS was defined when subjects had at least 3 of the 5 following components:
Waist circumference ≥90 cm for men and ≥80 cm for women
Triglycerides ≥150 mg/dl
HDL cholesterol <40 mg/dl for men and <50 mg/dl for women
Blood pressure ≥130/85 mmHg or current use of antihypertensive medication
Fasting glucose ≥100 mg/dl or current use of antidiabetic medication/insulin
When subjects were reported to take medication for dyslipidemia, it was regarded as the presence of both high triglyceride and low HDL cholesterol levels.
Statistical analysis
To provide nationally representative prevalence estimates, statistical procedures were performed to reflect the complex sampling design and sampling weights of the KNHANES. The SAS PROC SURVEY module was used to consider strata, clusters, and weights. Based on the characteristics of the data, the results were expressed as the means ± standard error (SE), geometric means (95% confidence interval [CI]) or percentages ( ± SE), as appropriate. Characteristics of each gender were compared with the chi-squared test for dichotomous variables and independent t-tests for continuous variables. Logarithmic transformations of variables with skewed distributions were performed before statistical analysis. To compare the mean of PTH and vitamin D levels between groups according to the absence or presence of MetS and each of its components, analysis of covariance (ANCOVA) was performed to adjust several covariates. Multivariate logistic regression analysis for the presence of metabolic syndrome was performed. To acquire clinical significance from the analysis, the population was categorized into two groups; population with the 1st to 3rd vs. 4th quartiles of PTH levels (cut-off values for men: 76.8 pg/ml, for female: 77.6 pg/ml) and those with less than 20 ng/ml vs. 20 ng/ml or above of vitamin D levels. All statistical analyses were performed using SAS version 9.3 software (SAS Institute Inc., Cary, NC, USA).
Results
Baseline characteristics of the population
The mean ages of male and female participants were 60.8 ± 0.2 and 61.7 ± 0.2 years, respectively (Table 1). In contrast to incidence of diabetes, MetS was more frequently observed in females (52.3%) than in males (43.1%). Although the intake of calcium was significantly higher in females than that in males, significantly increased levels of PTH and decreased levels of 25(OH)D were observed in female subjects compared to male subjects. Other parameters including MetS components and socioeconomic status are shown in Table 1.
Table 1.
Baseline characteristics total population
| Male (n = 3190) | Female (n = 3814) | P-value | |
|---|---|---|---|
| Age (years) | 60.8 ± 0.2 | 61.7 ± 0.2 | < 0.0001 |
| Smoking (%) | 35.5(1.1) | 4.8(0.5) | < 0.0001 |
| Drinking (%) | 17.8(0.9) | 0.8(0.2) | < 0.0001 |
| Regular exercise (%) | 28.3(1.1) | 23.8(1) | 0.0004 |
| Low Education status (%) | 46.9(1.3) | 23.3(1.1) | < 0.0001 |
| Low income (%) | 23.7(1) | 31.3(1.1) | < 0.0001 |
| Calcium intake (mg) | 431.5 + 8.4 | 559.0 + 9.2 | < 0.0001 |
| DM (%) | 18.1(0.8) | 14.4(0.7) | 0.0008 |
| HTN (%) | 51.6(1.2) | 49.1(1) | 0.103 |
| Metabolic syndrome (%) | 43.1(1.1) | 52.3(1.1) | < 0.0001 |
| BMI (kg m−2) | 23.9 ± 0.1 | 24.3 ± 0.1 | < 0.0001 |
| Waist circumference (cm) | 85.4 ± 0.2 | 82.3 ± 0.2 | < 0.0001 |
| Systolic blood pressure (mmHg) | 127.2 ± 0.4 | 126.8 ± 0.4 | 0.5328 |
| Diastolic blood pressure (mmHg) | 81 ± 0.3 | 78.2 ± 0.2 | < 0.0001 |
| Fasting glucose (mmol l−1) | 104.8 ± 0.5 | 101.1 ± 0.5 | < 0.0001 |
| Total cholesterol (mmol l−1) | 186.7 ± 0.8 | 201.4 ± 0.7 | < 0.0001 |
| Triglyceride (mmol l−1) | 133.9(130.3–137.6) | 118.5(115.9–121.2) | < 0.0001 |
| HDL cholesterol (mmol l−1) | 48.4 ± 0.3 | 52.5 ± 0.3 | < 0.0001 |
| LDL cholesterol(mmol l−1) | 112 ± 0.7 | 126.1 ± 0.6 | < 0.0001 |
| Glomerular filtration rate ( | 91.37 + 0.4 | 88.9 ± 0.4 | < 0.0001 |
| PTH (pg/mL) | 61.1(59.8–62.4) | 62.4(61.2–63.6) | 0.0457 |
| Serum 25(OH)D (ng/mL) | 21.6 + 0.3 | 18.5 + 0.2 | < 0.0001 |
BMI, body mass index; LDL, low-density lipoprotein; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment of insulin resistance; PTH, parathyroid hormone; Normal range of PTH is 8–76 pg/mL. Data are expressed as the means ± standard error (SE), percentage (SE) or geometric means (95% confidence interval).
Low educational status: subject with elementary to middle school.
Low income: the lowest quartile.
Comparison of PTH and vitamin D levels of subjects with and without MetS (and its components)
Higher PTH levels were observed in populations with MetS than in those without MetS among both genders (model 1 in Table 2). For females, PTH levels were significantly higher in subjects with MetS than in those without MetS after adjustment for possible covariates (model 2–4 in Table 2). In contrast to PTH levels, differences in 25(OH)D levels were observed only in male subject groups (model 1–3 in Table 3)
Table 2.
Comparison of parathyroid hormone levels between groups according to presence or absence of metabolic syndrome or its components.
| MODEL 1 | MODEL 2 | MODEL 3 | MODEL 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Male | Female | Male | Female | |
| Metabolic syndrome | ||||||||
| Absent | 60.1(58.6–61.6) | 60.7(59.4–62.1) | 60.3(58.8–61.9) | 61.8(60.4–63.2) | 60.3(58.7–61.9) | 61.5(60.1–62.9) | 60.4(58.8–62) | 61.3(59.9–62.7) |
| Present | 62.4(60.7–64.2) | 63.9(62.4–65.6) | 62.7(60.9–64.5) | 63.6(62–65.2) | 62(60.2–63.8) | 63.5(61.8–65.2) | 61.6(59.9––63.4) | 63.2(61.6–64.7) |
| P value | 0.0188 | 0.0003 | 0.0188 | 0.05 | 0.1066 | 0.0394 | 0.2289 | 0.0493 |
| High waist circumference | ||||||||
| Absent | 60.9(59.6–62.2) | 59.9(58.4–61.4) | 61.1(59.8–62.4) | 60.7(59.1–62.2) | 61(59.6–62.4) | 60.4(58.8–62) | 60.8(59.5–62.2) | 60.2(58.7–61.8) |
| Present | 61.6(59.4–63.9) | 64.1(62.6–65.6) | 61.8(59.6–64.1) | 64.1(62.7–65.7) | 61.1(59–63.3) | 64(62.4–65.7) | 61.1(59–63.3) | 63.7(62.2–65.2) |
| P value | 0.5032 | < 0.0001 | 0.5042 | 0.0005 | 0.9189 | 0.0006 | 0.7687 | 0.0008 |
| High blood pressure | ||||||||
| Absent | 58.7(57–60.5) | 58.4(56.9–59.8) | 59(57.3–60.8) | 59.5(58.1–61.1) | 58.7(56.8–60.6) | 59(57.5–60.6) | 58.9(57–60.8) | 59(57.5–60.5) |
| Present | 62.5(61–64) | 65.3(63.8–66.8) | 62.6(61.2–64.1) | 64.9(63.4–66.4) | 62.4(60.9–63.9) | 64.9(63.3–66.5) | 62.1(60.6–63.5) | 64.4(63–65.9) |
| P value | 0.0001 | <0.0001 | 0.0002 | < 0.0001 | 0.0005 | < 0.0001 | 0.0026 | < 0.0001 |
| High blood gluose | ||||||||
| Absent | 60.9(59.2–62.7) | 61.9(60.6–63.3) | 61.2(59.5–62.9) | 62.6(61.3–64) | 60.9(59.3–62.7) | 62.6(61.2–64.1) | 61(59.4–62.7) | 62.4(61–63.8) |
| Present | 61.2(59.7–62.8) | 63.1(61.4–64.9) | 61.4(59.9–63.1) | 62.9(61.2–64.7) | 61.1(59.5–62.8) | 62.5(60.7–64.3) | 60.8(59.2–62.4) | 62.2(60.5–63.9) |
| P value | 0.7611 | 0.2066 | 0.8005 | 0.7557 | 0.8553 | 0.8693 | 0.8275 | 0.8544 |
| Hypertriglyceridemia | ||||||||
| Absent | 60.6(59.2–62.1) | 62.8(61.5–64.2) | 60.8(59.4–62.2) | 63.5(62.2–64.9) | 60.9(59.5–62.5) | 63.3(61.9–64.7) | 61(59.6–62.5) | 63.1(61.8–64.5) |
| Present | 61.7(59.8–63.6) | 61.7(59.9–63.5) | 62(60.1–64) | 61.5(59.8–63.4) | 61.1(59.3–63.1) | 61.5(59.6–63.4) | 60.8(59–62.6) | 61(59.2–62.8) |
| P value | 0.3086 | 0.238 | 0.2334 | 0.0532 | 0.8485 | 0.0951 | 0.8349 | 0.0345 |
| Low HDL cholesterol | ||||||||
| Absent | 60.2(58.7–61.8) | 61.7(60.1–63.4) | 60.5(58.9–62.1) | 62.8(61.2–64.5) | 60(58.4–61.7) | 62.8(61.1–64.6) | 60.2(58.6–61.8) | 62.8(61.1–64.5) |
| Present | 62.2(60.5–63.9) | 62.7(61.4–64.1) | 62.5(60.8–64.2) | 62.7(61.4–64.1) | 62.3(60.6–64.1) | 62.4(61–63.9) | 61.9(60.2–63.6) | 62(60.7–63.4) |
| P value | 0.0406 | 0.2837 | 0.042 | 0.9316 | 0.0281 | 0.6669 | 0.092 | 0.448 |
MODEL 1 non–adjusted.
MODEL 2 age, sex.
MODEL 3 age, sex, smoke, exercise, education levels, income status, calcium intake.
MODEL 4 age, sex, smoke, exercise, education levels, income status, calcium intake, vitamin D.
Table 3.
Comparison of vitamin D levels between groups according to presence or absence of metabolic syndrome or its components.
| MODEL 1 | MODEL 2 | MODEL 3 | MODEL 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | Male | Female | Male | Female | |
| Metabolic syndrome | ||||||||
| Absent | 22 ± 0.3 | 18.7 ± 0.2 | 22.1 ± 0.3 | 18.6 ± 0.2 | 22.1 ± 0.3 | 18.7 ± 0.3 | 22 ± 0.3 | 18.6 ± 0.3 |
| Present | 21.1 ± 0.3 | 18.4 ± 0.3 | 21.2 ± 0.3 | 18.4 ± 0.3 | 21.4 ± 0.3 | 18.4 ± 0.3 | 21.4 ± 0.3 | 18.5 ± 0.3 |
| P value | 0.0044 | 0.3512 | 0.0045 | 0.4098 | 0.0312 | 0.4767 | 0.0613 | 0.7903 |
| High waist circumference | ||||||||
| Absent | 21.6 ± 0.3 | 18.5 ± 0.3 | 21.6 ± 0.3 | 18.5 ± 0.3 | 21.7 ± 0.3 | 18.7 ± 0.3 | 21.7 ± 0.3 | 18.5 ± 0.3 |
| Present | 21.7 ± 0.4 | 18.5 ± 0.3 | 21.8 ± 0.4 | 18.5 ± 0.3 | 22 ± 0.4 | 18.5 ± 0.3 | 22 ± 0.4 | 18.6 ± 0.3 |
| P value | 0.5612 | 0.9223 | 0.5602 | 0.97 | 0.2814 | 0.5441 | 0.271 | 0.8591 |
| High blood pressure | ||||||||
| Absent | 22.2 ± 0.3 | 18.9 ± 0.3 | 22.3 ± 0.4 | 19 ± 0.3 | 22.4 ± 0.3 | 19 ± 0.3 | 22.2 ± 0.3 | 18.8 ± 0.3 |
| Present | 21.3 ± 0.3 | 18.3 ± 0.2 | 21.3 ± 0.3 | 18.2 ± 0.3 | 21.4 ± 0.3 | 18.3 ± 0.3 | 21.5 ± 0.3 | 18.4 ± 0.2 |
| P value | 0.0024 | 0.009 | 0.0012 | 0.0153 | 0.0039 | 0.0148 | 0.0291 | 0.267 |
| High blood gluose | ||||||||
| Absent | 21.8 ± 0.3 | 18.6 ± 0.2 | 21.9 ± 0.3 | 18.6 ± 0.2 | 22.1 ± 0.3 | 18.6 ± 0.2 | 22.1 ± 0.3 | 18.6 ± 0.2 |
| Present | 21.4 ± 0.3 | 18.4 ± 0.3 | 21.4 ± 0.3 | 18.4 ± 0.3 | 21.4 ± 0.3 | 18.5 ± 0.3 | 21.4 ± 0.3 | 18.5 ± 0.3 |
| P value | 0.1961 | 0.6449 | 0.1784 | 0.6955 | 0.0608 | 0.8803 | 0.06 | 0.8524 |
| Hypertriglyceridemia | ||||||||
| Absent | 22 ± 0.3 | 18.7 ± 0.2 | 22 ± 0.3 | 18.7 ± 0.2 | 22.1 ± 0.3 | 18.7 ± 0.2 | 22.1 ± 0.3 | 18.8 ± 0.2 |
| Present | 21.1 ± 0.3 | 18.2 ± 0.3 | 21.2 ± 0.3 | 18.2 ± 0.3 | 21.3 ± 0.3 | 18.2 ± 0.3 | 21.3 ± 0.3 | 18.2 ± 0.3 |
| P value | 0.005 | 0.0879 | 0.0077 | 0.0999 | 0.0164 | 0.0572 | 0.0174 | 0.014 |
| Low HDL cholesterol | ||||||||
| Absent | 22.1 ± 0.3 | 18.8 ± 0.2 | 22.1 ± 0.3 | 18.8 ± 0.2 | 22.2 ± 0.3 | 18.7 ± 0.3 | 22.1 ± 0.3 | 18.8 ± 0.3 |
| Present | 21 ± 0.3 | 18.4 ± 0.3 | 21.1 ± 0.3 | 18.4 ± 0.3 | 21.2 ± 0.3 | 18.4 ± 0.3 | 21.3 ± 0.3 | 18.5 ± 0.3 |
| P value | 0.0016 | 0.1444 | 0.0015 | 0.1603 | 0.0062 | 0.339 | 0.0132 | 0.2629 |
MODEL 1 non–adjusted.
MODEL 2 age, sex.
MODEL 3 age, sex, smoke, exercise, education levels, income status, calcium intake.
MODEL 4 age, sex, smoke, exercise, education levels, income status, calcium intake, PTH.
For both genders, subjects with hypertension showed higher PTH levels (model 1–4 in Table 2) and lower 25(OH)D levels (model 1–3 for females, model 1–4 for males in Table 3) than those without hypertension. Higher PTH levels were found in female subjects with the waist circumference component of MetS (model 1–4 in Table 2). For male subjects, those with the HDL cholesterol component (low HDL cholesterol levels) of MetS showed higher PTH and lower 25(OH)D levels than those with normal HDL levels. Male subjects with the triglyceridxe component of MetS presented lower 25(OH)D levels than those without this component.
In multivariate logistic regression analysis (Table 4), gender specific association patterns of PTH and vitamin D with metabolic syndrome were observed. After adjustment of possible confounding factors, the highest quartiles of PTH was significantly associated with increased risk of MetS in females, while vitamin D deficiency (less than 20 ng/ml) was related to increased risk in males.
Table 4.
Multiple logistic regression analysis (Presence of Metabolic syndrome – PTH or Vitamin D).
| Unadjusted | Adjusted | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| MODEL 1 | MODEL 2 | MODEL 3 | MODEL 4 | ||||||
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | ||||||
| Male | Female | Male | Female | Male | Female | Male | Female | ||
| PTH* | |||||||||
| 1–3rd quartiles | reference | reference | reference | reference | reference | reference | reference | reference | |
| 4th quartile | 1.24 (1.022–1.505) | 1.382 (1.15–1.661) | 1.241 (1.022–1.507) | 1.213(1.005,1.463) | 1.148(0.929,1.417) | 1.238(1.012,1.514) | 1.105(0.894,1.365) | 1.227(1.002,1.504) | |
| P value | 0.0295 | 0.0006 | 0.0295 | 0.044 | 0.201 | 0.0382 | 0.3549 | 0.0482 | |
| Vitamin D | |||||||||
| Less than 20 ng/ml | reference | reference | reference | reference | reference | reference | reference | reference | |
| 20 ng/ml or more | 0.795(0.675,0.937) | 0.97(0.832,1.132) | 0.795(0.675,0.937) | 0.963(0.825,1.124) | 0.812(0.679,0.97) | 0.947(0.798,1.125) | 0.824(0.688,0.988) | 0.978(0.823,1.163) | |
| P value | 0.0062 | 0.6993 | 0.0063 | 0.632 | 0.0219 | 0.5378 | 0.0362 | 0.8038 | |
Statistics were carried out using Logistic regression. adjusted for age, sex, self-pay of cancer screening, weight variation, waist-height ratio, physical activity, drink, smoking.
MODEL 1 non-adjusted.
MODEL 2 age, sex.
MODEL 3 age, sex, smoke, exercise, education levels, income status, calcium intake.
MODEL 4 age, sex, smoke, exercise, education levels, income status, calcium intake, PTH (or vitamin D).
*The cut-off values for the 4th quartiles of PTH was 76.8 pg/ml in male and 77.6 pg/ml in female.
Number of MetS components and PTH levels
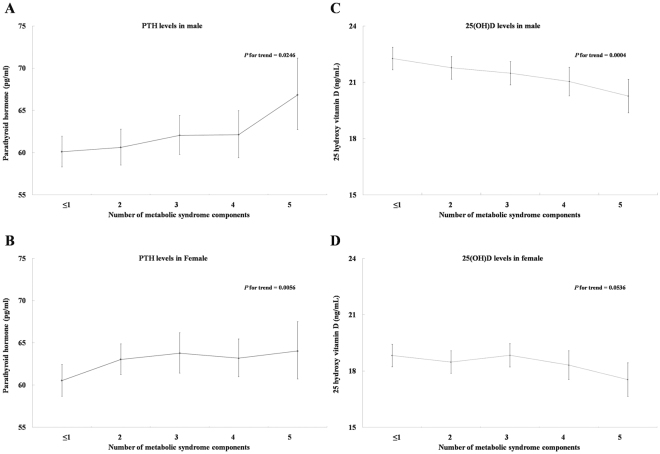
While significant decreases in 25(OH)D levels were observed only in male subjects, an increase in PTH levels was observed for both genders with an increase in the number of MetS components (Fig. 1).
Figure 1.
PTH (A and B) and 25(OH)D levels (C and D) according to the number of metabolic syndrome components.
Discussion
A significant association of PTH and vitamin D with MetS was found in an analysis of representative nationwide data of the middle-aged Korean population with preserved renal function. Specifically, PTH levels in females and vitamin D levels in males with MetS were significantly different from those of subjects without MetS after adjustment for possible confounding factors. The association patterns of PTH and vitamin D with each component of MetS were different between males and females. In addition, significant increases in PTH levels in both genders and a decrease in vitamin D levels in males were observed based on the number of MetS components present.
In terms of the association of PTH levels with MetS, our study showed similar results to previous studies6,8, which excluded subjects with impaired renal function. Even in a study of obese populations, which did not exclude CKD subjects10, an increase in PTH was associated with the presence of MetS after adjustment for confounding factors including the GFR. However, although an association of PTH levels with MetS was found in our study, statistically significant differences after adjusting several confounding factors remained only for females, unlike prior studies that showed a significant relevance in men6,8 or in both genders10. Some studies failed to show a relationship between PTH and MetS in large population datasets9,14,15. Among them, a study by Ford et al.9 included relatively young subjects with a mean age of under 50 years and no adjustment of GFR or exclusion criteria for CKD subjects. More than 50% of the subjects were under 50 years old, and the incidence of MetS was less than 30% in a study by Li et al.14. As mentioned previously, a recent study using data from the KNHANES15 that found higher levels of PTH than our study did not consider the effects of the GFR on PTH and vitamin D in the analysis. An association of vitamin D with MetS was observed for males in our study. Among previous large-scale population studies, some have shown an inverse relationship between vitamin D status and the presence of MetS7,8,11,13,15,26,27, but others did not show this association6,28,29. Four studies7,11,13,26 showed significant associations of vitamin D with MetS but did not measure the levels of PTH. Inconsistent results regarding the association of PTH and vitamin D levels with MetS may be caused by several factors, including whether the GFR was considered in the analysis. As differences in vitamin D metabolism have been observed among various ethnicities30, and a prior study examined the effect of ethnicity on the risk of MetS29, the effect of the ethnicity of the studied population should be considered.
The mechanism by which PTH and vitamin D affect the development MetS has been explained in several ways. As summarized in a review article31, PTH could increase intracellular calcium in skeletal muscle and adipocytes, which would consequently suppress glucose uptake by those tissues and induce insulin resistance. In a recent study, increased lipolysis induced by PTH could cause insulin reisstance32. Additionally, a prosclerotic effect on vascular smooth muscle cells33 has been suggested. Likewise, vitamin D deficiency also harbours a possible mechanistic link to the development of MetS because it influences insulin secretion34,35, adipogenesis36 and renin-angiotensin system37.
The gender difference in the association of PTH and vitamin D with MetS is not fully understood. Differences in the prevalence of vitamin D deficiency in males and females38 may contribute to the different degrees of association of PTH and vitamin D with MetS. Importantly, vitamin D deficiency is prevalent in the Korean female population39. As oestrogen could act as a stimulator for calcium absorption in the intestinal tract40,41, oestrogen deficiency in females may contribute to the difference in association patterns that we observed. Although no detailed data for hormone replacement in postmenopausal women was obtained, most of the female subjects would likely have oestrogen deficiency considering the mean age of the studied female population (61.7 ± 0.2 years).
Based on the results, gender specific application of both hormones might be considered as a biomarker or therapeutic target of MetS. PTH in females could be suggested as an additional surrogate marker for development of MetS. Though it would not be possible to provide a certain cut-off values of PTH in terms of increasing MetS, at least, increasing PTH with normal calcium and vitamin D homeostasis might be warning sign for MetS in females. In addition, it would be very interesting to elucidate the role of intervention, that is supplementation of calcium and vitamin D for female with higher PTH and for male with lower vitamin D, to prevent MetS. There is no interventional study to identify the role of calcium supplement in prevention of MetS yet. Though possible benefit of vitamin D supplement on glycemic control or insulin resistance was suggested42, it is not conclusive43. Therefore, further well-organized studies are required to determine the MetS preventive effect of calcium and vitamin D.
Interestingly, according to our results, as the number of MetS components increased, the levels of PTH increased in both females and males (p for trend 0.0056 in females, 0.0246 in males). A statistically significant decrease in vitamin D levels was observed only in males (p for trend 0.0004). From a clinical perspective, elevated PTH levels may be a useful surrogate marker for the risk of MetS in subjects with preserved renal function.
Several limitations could be noted in our study. First, no causal relationship could be deduced due to the cross-sectional design of the study. Second, no detailed data were provided on the supplementation of calcium and vitamin D (i.e., dose) in the studied subjects, and the effect of supplementation could not be estimated. Third, subjects with primary aldosteronism or renovacaulr hypertension, which would lead to secondary hyperparathyroidism and increase the risk of Mets44, could not be identified and excluded. Lastly, information about sun exposure and seasonal variation, which are related to the status of vitamin D, was unavailable for the analysis.
In this study, we excluded subjects with impaired renal function for several reasons. First, PTH and vitamin D levels are closely related to the GFR45. Specifically, different patterns (steep increases) of PTH changes according to the GFR were noted in subjects with stage 3 CKD, and subjects with stage 3b CKD demonstrated an approximate 5-fold increased risk of high PTH (70 pg/ml) compared to individuals with a GFR ≥ 6016. In contrast to stage 3a CKD, stage 3b patients exhibit a higher rate of progression to end-stage renal disease46 and a higher mortality rate47. Moreover, stage 3b CKD patients have been shown to exhibit more metabolic complications than stage 3a patients48. In addition to the relationship between PTH and GFR, vitamin D deficiency, which is also related to PTH secretion, is prevalent in subjects with CKD49,50. Second, considering the current management guidelines of CKD patients17, the likelihood of patients undergoing vitamin D and calcium supplementation, which would interfere with the levels of PTH and vitamin D, would be high. Lastly, CKD itself is also a well-known risk factor for MetS18,51–57. Therefore, it would be reasonable to evaluate the association of PTH and vitamin D with MetS in subjects with preserved renal function separately from those with moderate to severe CKD. In a recent KNHANES study15 that evaluated the association of PTH and vitamin D with MetS, different results from ours were obtained. This discrepancy may have occurred because the GFR was not considered in the analysis of the former study. Because no exclusion criteria for CKD were applied, the prior study15 presented higher mean levels of PTH (approximately 70 pg/mL) than our study.
In summary, in a study of nationwide representative data of a middle-aged population with preserved renal function, gender-specific associations of PTH and vitamin D with MetS were found. PTH exhibited a possible link to MetS in females, whereas vitamin D showed a possible link to MetS in males. The number of MetS components was related to an increase in PTH and a decrease in vitamin D levels. Based on the results of this study, further investigations that are especially focused on gender specificity should be conducted to identify the mechanism that links these hormones to MetS.
Author Contributions
M.H.K. and J.M.L. were responsible for the conception and design of the study. M.H.K., J.L. and J.H. were responsible for acquisition of data. M.H.K., J.L., and J.H. performed the data analysis. M.H.K. drafted the manuscript. M.H.K., J.L., J.H., K.J., D.J.L., J.M.L., S.A.C., M.I.K., and B.Y.C. participated in interpretation of the findings. All authors read and approved the final version of the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kumar S, et al. Impaired glucose tolerance and insulin insensitivity in primary hyperparathyroidism. Clin. Endocrinol. (Oxf.) 1994;40:47–53. doi: 10.1111/j.1365-2265.1994.tb02442.x. [DOI] [PubMed] [Google Scholar]
- 2.Chiu KC, et al. Insulin sensitivity is inversely correlated with plasma intact parathyroid hormone level. Metabolism. 2000;49:1501–1505. doi: 10.1053/meta.2000.17708. [DOI] [PubMed] [Google Scholar]
- 3.Kayaniyil S, et al. Prospective associations of vitamin D with beta-cell function and glycemia: the PROspective Metabolism and ISlet cell Evaluation (PROMISE) cohort study. Diabetes. 2011;60:2947–2953. doi: 10.2337/db11-0465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhao G, Ford ES, Li C. Associations of serum concentrations of 25-hydroxyvitamin D and parathyroid hormone with surrogate markers of insulin resistance among U.S. adults without physician-diagnosed diabetes: NHANES, 2003–2006. Diabetes Care. 2010;33:344–347. doi: 10.2337/dc09-0924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kang JH, et al. Adiposity in the Relationship between Serum Vitamin D Level and Insulin Resistance in Middle-Aged and Elderly Korean Adults: The Korea National Health and Nutrition Examination Survey 2008. Endocrinol Metab (Seoul) 2013;28:96–102. doi: 10.3803/EnM.2013.28.2.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reis JP, von Mühlen D, Kritz-Silverstein D, Wingard DL, Barrett-Connor E. Vitamin D, parathyroid hormone levels, and the prevalence of metabolic syndrome in community-dwelling older adults. Diabetes Care. 2007;30:1549–1555. doi: 10.2337/dc06-2438. [DOI] [PubMed] [Google Scholar]
- 7.Hyppönen E, Boucher BJ, Berry DJ, Power C. 25-hydroxyvitamin D, IGF-1, and metabolic syndrome at 45 years of age: a cross-sectional study in the 1958 British Birth Cohort. Diabetes. 2008;57:298–305. doi: 10.2337/db07-1122. [DOI] [PubMed] [Google Scholar]
- 8.Reis JP, von Mühlen D, Miller ER. 3rd. Relation of 25-hydroxyvitamin D and parathyroid hormone levels with metabolic syndrome among US adults. Eur. J. Endocrinol. 2008;159:41–48. doi: 10.1530/EJE-08-0072. [DOI] [PubMed] [Google Scholar]
- 9.Ford ES, Zhao G, Li C, Pearson WS. Serum concentrations of vitamin D and parathyroid hormone and prevalent metabolic syndrome among adults in the United States. J. Diabetes. 2009;1:296–303. doi: 10.1111/j.1753-0407.2009.00046.x. [DOI] [PubMed] [Google Scholar]
- 10.Hjelmesaeth J, et al. Parathyroid hormone, but not vitamin D, is associated with the metabolic syndrome in morbidly obese women and men: a cross-sectional study. Cardiovasc. Diabetol. 2009;8:7. doi: 10.1186/1475-2840-8-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu L, et al. Plasma 25-hydroxyvitamin D concentration and metabolic syndrome among middle-aged and elderly Chinese individuals. Diabetes Care. 2009;32:1278–1283. doi: 10.2337/dc09-0209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.He JL, Scragg RK. Vitamin D, parathyroid hormone, and blood pressure in the National Health and Nutrition Examination Surveys. Am. J. Hypertens. 2011;24:911–917. doi: 10.1038/ajh.2011.73. [DOI] [PubMed] [Google Scholar]
- 13.Gagnon C, et al. Low serum 25-hydroxyvitamin D is associated with increased risk of the development of the metabolic syndrome at five years: results from a national, population-based prospective study (The Australian Diabetes, Obesity and Lifestyle Study: AusDiab) J. Clin. Endocrinol. Metab. 2012;97:1953–1961. doi: 10.1210/jc.2011-3187. [DOI] [PubMed] [Google Scholar]
- 14.Li LH, Yin XY, Yao CY, Zhu XC, Wu XH. Serum 25-hydroxyvitamin D, parathyroid hormone, and their association with metabolic syndrome in Chinese. Endocrine. 2013;44:465–472. doi: 10.1007/s12020-013-9885-2. [DOI] [PubMed] [Google Scholar]
- 15.Kim J. Association between serum vitamin D, parathyroid hormone and metabolic syndrome in middle-aged and older Korean adults. Eur. J. Clin. Nutr. 2015;69:425–430. doi: 10.1038/ejcn.2014.192. [DOI] [PubMed] [Google Scholar]
- 16.Muntner P, et al. Association of serum intact parathyroid hormone with lower estimated glomerular filtration rate. Clin. J. Am. Soc. Nephrol. 2009;4:186–194. doi: 10.2215/CJN.03050608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.National Kidney Foundation K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am. J. Kidney Dis. 2003;42:S1–201. [PubMed] [Google Scholar]
- 18.Beddhu S, Kimmel PL, Ramkumar N, Cheung AK. Associations of metabolic syndrome with inflammation in CKD: results From the Third National Health and Nutrition Examination Survey (NHANES III) Am. J. Kidney Dis. 2005;46:577–586. doi: 10.1053/j.ajkd.2005.06.014. [DOI] [PubMed] [Google Scholar]
- 19.Zammit AR, Katz MJ, Derby C, Bitzer M, Lipton RB. Metabolic Syndrome and Smoking Are Associated with Future Development of Advanced Chronic Kidney Disease in Older Adults. Cardiorenal Med. 2016;6:108–115. doi: 10.1159/000441624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sipilä S, et al. Sex hormones and skeletal muscle weakness. Biogerontology. 2013;14:231–245. doi: 10.1007/s10522-013-9425-8. [DOI] [PubMed] [Google Scholar]
- 21.Carnevale V, et al. Longitudinal evaluation of vitamin D status in healthy subjects from southern Italy: seasonal and gender differences. Osteoporos. Int. 2001;12:1026–1030. doi: 10.1007/s001980170012. [DOI] [PubMed] [Google Scholar]
- 22.Korean Ministry of Health and Welfare, Korea Centers for Disease Control and Prevention. The Korea National Health and Nutrition Examination Survey. https://knhanes.cdc.go.kr (2017).
- 23.Son JW, et al. Changing Clinical Characteristics according to Insulin Resistance and Insulin Secretion in Newly Diagnosed Type 2 Diabetic Patients in Korea. Diabetes Metab. J. 2015;39:387–394. doi: 10.4093/dmj.2015.39.5.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roberts WC. The Friedewald-Levy-Fredrickson formula for calculating low-density lipoprotein cholesterol, the basis for lipid-lowering therapy. Am. J. Cardiol. 1988;62:345–346. doi: 10.1016/0002-9149(88)90248-2. [DOI] [PubMed] [Google Scholar]
- 25.Matsushita K, Selvin E, Bash LD, Astor BC, Coresh J. Risk implications of the new CKD Epidemiology Collaboration (CKD-EPI) equation compared with the MDRD Study equation for estimated GFR: the Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Kidney Dis. 2010;55:648–659. doi: 10.1053/j.ajkd.2009.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ford ES, Ajani UA, McGuire LC, Liu S. Concentrations of serum vitamin D and the metabolic syndrome among U.S. adults. Diabetes Care. 2005;28:1228–1230. doi: 10.2337/diacare.28.5.1228. [DOI] [PubMed] [Google Scholar]
- 27.Kim MK, et al. The association of serum vitamin D level with presence of metabolic syndrome and hypertension in middle-aged Korean subjects. Clin. Endocrinol. (Oxf.) 2010;73:330–338. doi: 10.1111/j.1365-2265.2010.03798.x. [DOI] [PubMed] [Google Scholar]
- 28.Kim S, Lim J, Kye S, Joung H. Association between vitamin D status and metabolic syndrome risk among Korean population: based on the Korean National Health and Nutrition Examination Survey IV-2, 2008. Diabetes Res. Clin. Pract. 2012;96:230–236. doi: 10.1016/j.diabres.2012.01.001. [DOI] [PubMed] [Google Scholar]
- 29.George JA, Norris SA, van Deventer HE, Crowther NJ. The association of 25 hydroxyvitamin D and parathyroid hormone with metabolic syndrome in two ethnic groups in South Africa. PLoS One. 2013;8:e61282. doi: 10.1371/journal.pone.0061282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Awumey EM, Mitra DA, Hollis BW, Kumar R, Bell NH. Vitamin D metabolism is altered in Asian Indians in the southern United States: a clinical research center study. J. Clin. Endocrinol. Metab. 1998;83:169–173. doi: 10.1210/jcem.83.1.4514. [DOI] [PubMed] [Google Scholar]
- 31.McCarty MF, Thomas CA. PTH excess may promote weight gain by impeding catecholamine-induced lipolysis-implications for the impact of calcium, vitamin D, and alcohol on body weight. Med. Hypotheses. 2003;61:535–542. doi: 10.1016/S0306-9877(03)00227-5. [DOI] [PubMed] [Google Scholar]
- 32.Larsson S, Jones HA, Göransson O, Degerman E, Holm C. Parathyroid hormone induces adipocyte lipolysis via PKA-mediated phosphorylation of hormone-sensitive lipase. Cell. Signal. 2016;28:204–213. doi: 10.1016/j.cellsig.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 33.Perkovic V, et al. Parathyroid hormone has a prosclerotic effect on vascular smooth muscle cells. Kidney Blood Press. Res. 2003;26:27–33. doi: 10.1159/000069761. [DOI] [PubMed] [Google Scholar]
- 34.Bourlon PM, Billaudel B, Faure-Dussert A. Influence of vitamin D3 deficiency and 1,25 dihydroxyvitamin D3 on de novo insulin biosynthesis in the islets of the rat endocrine pancreas. J. Endocrinol. 1999;160:87–95. doi: 10.1677/joe.0.1600087. [DOI] [PubMed] [Google Scholar]
- 35.Kajikawa M, et al. An insulinotropic effect of vitamin D analog with increasing intracellular Ca2 + concentration in pancreatic beta-cells through nongenomic signal transduction. Endocrinology. 1999;140:4706–4712. doi: 10.1210/endo.140.10.7025. [DOI] [PubMed] [Google Scholar]
- 36.Zhuang H, Lin Y, Yang G. Effects of 1,25-dihydroxyvitamin D3 on proliferation and differentiation of porcine preadipocyte in vitro. Chem. Biol. Interact. 2007;170:114–123. doi: 10.1016/j.cbi.2007.07.012. [DOI] [PubMed] [Google Scholar]
- 37.Li YC, et al. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Invest. 2002;110:229–238. doi: 10.1172/JCI0215219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hyppönen E, Power C. Hypovitaminosis D in British adults at age 45 y: nationwide cohort study of dietary and lifestyle predictors. Am. J. Clin. Nutr. 2007;85:860–868. doi: 10.1093/ajcn/85.3.860. [DOI] [PubMed] [Google Scholar]
- 39.Ko MJ, Yun S, Oh K, Kim K. Relation of serum 25-hydroxyvitamin D status with skeletal muscle mass by sex and age group among Korean adults. Br. J. Nutr. 2015;114:1838–1844. doi: 10.1017/S0007114515003633. [DOI] [PubMed] [Google Scholar]
- 40.Heaney RP, Recker RR, Stegman MR, Moy AJ. Calcium absorption in women: relationships to calcium intake, estrogen status, and age. J. Bone Miner. Res. 1989;4:469–475. doi: 10.1002/jbmr.5650040404. [DOI] [PubMed] [Google Scholar]
- 41.Diaz de Barboza G, Guizzardi S, Tolosa de Talamoni N. Molecular aspects of intestinal calcium absorption. World J. Gastroenterol. 2015;21:7142–7154. doi: 10.3748/wjg.v21.i23.7142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.George PS, Pearson ER, Witham MD. Effect of vitamin D supplementation on glycaemic control and insulin resistance: a systematic review and meta-analysis. Diabet. Med. 2012;29:e142–150. doi: 10.1111/j.1464-5491.2012.03672.x. [DOI] [PubMed] [Google Scholar]
- 43.Jorde R, et al. Vitamin D 20,000 IU per Week for Five Years Does Not Prevent Progression From Prediabetes to Diabetes. J. Clin. Endocrinol. Metab. 2016;101:1647–1655. doi: 10.1210/jc.2015-4013. [DOI] [PubMed] [Google Scholar]
- 44.Hanslik G, et al. Increased prevalence of diabetes mellitus and the metabolic syndrome in patients with primary aldosteronism of the German Conn’s Registry. Eur J Endocrinol. 2015;173:665–675. doi: 10.1530/EJE-15-0450. [DOI] [PubMed] [Google Scholar]
- 45.Han SW, Kim SJ, Lee DJ, Kim KM, Joo NS. The Relationship between Serum 25-Hydroxyvitamin D, Parathyroid Hormone and the Glomerular Filtration Rate in Korean Adults: The Korea National Health and Nutrition Examination Survey between 2009 and 2011. Korean J. Fam. Med. 2014;35:98–106. doi: 10.4082/kjfm.2014.35.2.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Baek SD, et al. Does stage III chronic kidney disease always progress to end-stage renal disease? A ten-year follow-up study. Scand. J. Urol. Nephrol. 2012;46:232–238. doi: 10.3109/00365599.2011.649045. [DOI] [PubMed] [Google Scholar]
- 47.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 2004;351:1296–1305. doi: 10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- 48.Ito H, et al. The usefulness of the revised classification for chronic kidney disease by the KDIGO for determining the frequency of diabetic micro- and macroangiopathies in Japanese patients with type 2 diabetes mellitus. J. Diabetes Complications. 2012;26:286–290. doi: 10.1016/j.jdiacomp.2012.04.011. [DOI] [PubMed] [Google Scholar]
- 49.Levin A, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007;71:31–38. doi: 10.1038/sj.ki.5002009. [DOI] [PubMed] [Google Scholar]
- 50.Reichel H, Deibert B, Schmidt-Gayk H, Ritz E. Calcium metabolism in early chronic renal failure: implications for the pathogenesis of hyperparathyroidism. Nephrol. Dial. Transplant. 1991;6:162–169. doi: 10.1093/ndt/6.3.162. [DOI] [PubMed] [Google Scholar]
- 51.Kang YH, Jeong DW, Son SM. Prevalence of Reduced Kidney Function by Estimated Glomerular Filtration Rate Using an Equation Based on Creatinine and Cystatin C in Metabolic Syndrome and Its Components in Korean Adults. Endocrinol Metab (Seoul) 2016;31:446–453. doi: 10.3803/EnM.2016.31.3.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chen J, et al. The metabolic syndrome and chronic kidney disease in U.S. adults. Ann. Intern. Med. 2004;140:167–174. doi: 10.7326/0003-4819-140-3-200402030-00007. [DOI] [PubMed] [Google Scholar]
- 53.Kurella M, Lo JC, Chertow GM. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J. Am. Soc. Nephrol. 2005;16:2134–2140. doi: 10.1681/ASN.2005010106. [DOI] [PubMed] [Google Scholar]
- 54.Kanauchi M, Kanauchi K, Kimura K, Inoue T, Saito Y. Associations of chronic kidney disease with the metabolic syndrome in non-diabetic elderly. Nephrol. Dial. Transplant. 2006;21:3608–3609. doi: 10.1093/ndt/gfl435. [DOI] [PubMed] [Google Scholar]
- 55.Kang YU, et al. Metabolic syndrome and chronic kidney disease in an adult Korean population: results from the Korean National Health Screening. PLoS One. 2014;9:e93795. doi: 10.1371/journal.pone.0093795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Banerjee D, et al. Metabolic syndrome in chronic kidney disease and renal transplant patients in North India. Int. Urol. Nephrol. 2012;44:937–943. doi: 10.1007/s11255-011-9998-6. [DOI] [PubMed] [Google Scholar]
- 57.Johnson DW, et al. Metabolic syndrome in severe chronic kidney disease: Prevalence, predictors, prognostic significance and effects of risk factor modification. Nephrology (Carlton) 2007;12:391–398. doi: 10.1111/j.1440-1797.2007.00804.x. [DOI] [PubMed] [Google Scholar]