Abstract
Previous studies have demonstrated that Licochalcone A possesses anti-inflammatory, anticancer, anti-bacterial, anti-malarial and anti-parasitic activities. In the present study the potential anticancer effects of Licochalcone A on MCF-7 cells were investigated. Licochalcone A significantly decreased cell viability and promoted autophagy and apoptosis, as demonstrated by an MTT assay, acridine orange staining and Annexin V-fluorescein isothiocyanate staining, respectively. Western blot analyses demonstrated that Licochalcone A treatment activated the LC3-II signaling pathway while suppressing the phosphoinositide 3-kinase (PI3K)/RAC-α serine-threonine-protein kinase (Akt)/mammalian target of rapamycin (mTOR) signaling pathway. In addition, Licochalcone A significantly increased caspase-3 activity and significantly decreased B-cell lymphoma-2 expression. The results from the present study indicate that Licochalcone A inhibits PI3K/Akt/mTOR activation, and promotes autophagy and apoptosis in MCF-7 cells.
Keywords: Licochalcone A, breast cancer, autophagy, phosphoinositide 3-kinase/RAC-α serine-threonine-protein kinase/mammalian target of rapamycin
Introduction
Breast cancer is a severe disease that presents a great threat to female health worldwide (1). Breast cancer-associated morbidity increases each year and ~1.2 million individuals are diagnosed with breast cancer annually (2). Although surgery, radiotherapy, chemotherapy and endocrine therapy are widely applied in clinics, long-term survival rates have not significantly improved (3). Each year ~500,000 individuals succumb to the disease; the leading cause of breast cancer-associated mortality is tumor metastasis, which remains a challenge for the prophylaxis and treatment of breast cancer (4).
Autophagy is an important process that aids with the turnover of intracellular proteins. During autophagy, proteins or organelles are encased by double-membrane structures, which eventually fuse with the lysosome in order for protein degradation to take place (2). Major functions of autophagy include cell clearance, and the degradation of impaired organelles and excessive biomacromolecules (5). The products of degradation can be used to provide energy and reconstruct cellular structures in order to maintain metabolic balance and homeostasis. A previous study has reported important functions for autophagy in the metastasis of breast cancer (6). Certain molecules and anti-cancer drugs have been reported to influence the proliferation and metastasis of breast cancer by regulating autophagy (6). For instance, B-cell lymphoma-2 (Bcl-2) can promote the proliferation of MCF-7 cells, an effect that is associated with the inhibition of autophagy (7). In addition, a previous study demonstrated that inhibition of the phosphoinositide 3-kinase (PI3K)/RAC-α serine-threonine-protein kinase (Akt)/mammalian target of rapamycin (mTOR) signaling pathway inhibits MDA-MB-231 cell proliferation, and that the PI3K/Akt/mTOR cascade is a key signaling pathway that regulates autophagy (8).
Licochalcone A is a chalconoid (a type of natural phenol) that can be isolated from Glycyrrhiza glabra (licorice) and Glycyrrhiza inflata (Chinese licorice) (9), although the quantity of Licochalcone A that can be extracted from licorice is the highest. Previous research has demonstrated that Licochalcone A possesses antimalarial and antitumor effects, in addition to antioxidant, anti-inflammatory, anti-bacterial, anti-leishmaniasis and estrogenic effects (10,11). Additionally, Licochalcone A has wide applications in the food and medical industry (10,11).
Materials and methods
Reagents and chemicals
Dulbecco's modified Eagle's medium (DMEM) and fetal bovine serum (FBS) were obtained from Gibco (Thermo Fisher Scientific, Inc., Waltham, MA, USA). MTT was obtained from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). The Annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI) kit was purchased from BD Biosciences (San Jose, CA, USA). The chemical structure of Licochalcone A (purchased from Sigma-Aldrich; Merck KGaA) is illustrated in Fig. 1.
Figure 1.
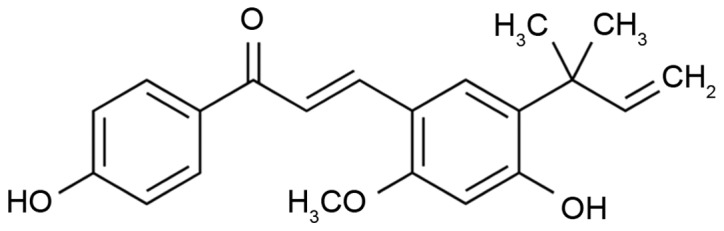
Chemical structure of Licochalcone A.
Cell culture
The human MCF-7 cell line was obtained from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China) and cultured in DMEM supplemented with 10% FBS at 37°C with 5% CO2.
Cell viability assay
The effect of Licochalcone A on cell viability was determined using an MTT assay and untreated cells were used as a comparison. A total of 8,000–10,000 MCF-7 cells/well were seeded into 96-well plates and cultured with 20 µl MTT for 4 h at 37°C. Following the removal of culture medium, 150 µl dimethyl sulfoxide was added to each well to dissolve the formazan crystals. The results were assessed by measuring the absorbance at 495 nm.
Annexin V-FITC/PI staining
The effect of Licochalcone A on the apoptosis rate of MCF-7 cells was determined using an Annexin V-FITC/PI kit (BD Biosciences). A total of 1–2×106 MCF-7 cells/well were seeded in 6-well plates and cultured with 100 µl Annexin V-FITC at 4°C in the dark for 30 min. Then, 10 µl PI was added to each well and incubated in the dark for 5 min at 37°C.
Acridine orange (AO) staining of autophagic cells
A total of 1–2×106 MCF-7 cells/well were seeded in 6-well plates and washed twice with ice-cold PBS. Then, MCF-7 cells were incubated with 1 µg/ml AO (Sigma-Aldrich; Merck KGaA) for 30 min at 37°C. MCF-7 cells were observed with fluorescence microscopy using 490-nm band-pass blue excitation filters and a 515-nm long-pass barrier filter.
Western blot analysis
A total of 1–2×106 MCF-7 cells/well were seeded in 6-well plates and washed twice with ice-cold PBS. Then, MCF-7 cells were harvested at 2,000 × g for 10 min at 4°C and gently lysed for 1 h in ice-cold cell lysis buffer (Beijing Dingguo Biotechnology, Co., Ltd., Beijing, China). Supernatants were collected following centrifugation at 12,000 × g for 10 min at 4°C. Protein concentrations were measured using a BCA assay. The samples (50 µg protein) were loaded onto a 10–12% SDS-PAGE gel and then transferred to a polyvinylidene difluoride (PVDF) membrane. The PVDF membrane was blocked with PBS containing 5% non-fat milk and 0.1% Tween-20 for 1 h at 37°C. Then, the PVDF membrane was incubated with anti-PI3K (cat. no. 4249; dilution, 1:2,000), anti-Akt (cat. no. 4691; dilution, 1:2,000), anti-phosphorylated (p)-Akt (cat. no. 4060; dilution, 1:2,000), anti-p-mTOR (cat. no. 5536; dilution, 1:2,000), anti-mTOR (cat. no. 2983; dilution, 1:2,000), anti-GFP-microtubule-associated proteins 1A/1B light chain 3 (LC3-II), anti-LC3-II, anti-Bcl-2 (cat. no. 3498; dilution, 1:2,000) and anti-β-actin (cat. no. 4970; dilution, 1:5,000) antibodies (all from Cell Signaling Technology, Inc., Danvers, MA, USA) at a dilution of 1:1,000 overnight at 4°C. Following washing with TBST for 20 min, the membrane was incubated with a horseradish peroxidase-conjugated anti-mouse antibody (cat. no. 14708; dilution, 1:10,000; Cell Signaling Technology, Inc.) at room temperature for 2 h. Protein blank was visualized using BeyoECL Plus (Beyotime Institute of Biotechnology, Haimen, China).
Capase-3 activity assay
A total of 1–2×106 MCF-7 cells/well were seeded in 6-well plates and washed twice with ice-cold PBS. Then, cells were incubated with caspase-3 activity kits (C1115; Beyotime Institute of Biotechnology) according to the manufacturer's protocol for 2 h at room temperature. The caspase-3 activity was detected at 405 nm using a Sunrise absorbance reader (Tecan Group, Ltd., Männedorf, Switzerland).
Statistical analysis
Data was calculated by mean ± standard deviation. All statistical analyses were performed with SPSS software (version 17.0; SPSS, Inc., Chicago, IL, USA) using an unpaired Student's t-test. Experiments were repeated three times. P<0.05 was considered to indicate a statistically significant difference.
Results
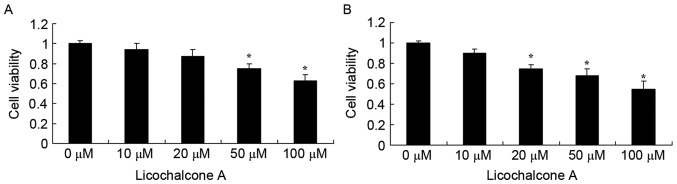
Licochalcone A decreases the viability of MCF-7 cells
Prior to investigating the anticancer effects of Licochalcone A on a culture of MCF-7 cells, the effect of Licochalcone A on cell viability was measured using an MTT assay. Licochalcone A treatment decreased MCF-7 cell viability in a dose- and time-dependent manner at 24 and 48 h (Fig. 2A and B, respectively). At 24 h, 50 or 100 µM Licochalcone A significantly decreased cell viability compared with the 0 µM control group (P<0.05; Fig. 2A). At 48 h, 20, 50 or 100 µM Licochalcone A significantly decreased the cell viability of MCF-7 cells compared with the 0 µM control group (P<0.01; Fig. 2B). Subsequently, 10, 20 and 50 µM Licochalcone A treatment for 48 h was selected to evaluate the underlying molecular mechanisms of Licochalcone A on breast cancer cells.
Figure 2.
Licochalcone A significantly inhibits MCF-7 cell viability. The viability of MCF-7 cells following treatment with the indicated concentrations of Licochalcone A for (A) 24 h and (B) 48 h. *P<0.01, compared with the 0 µM control group.
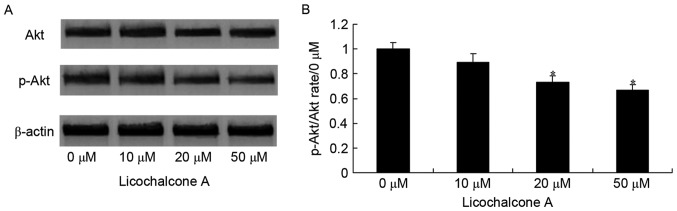
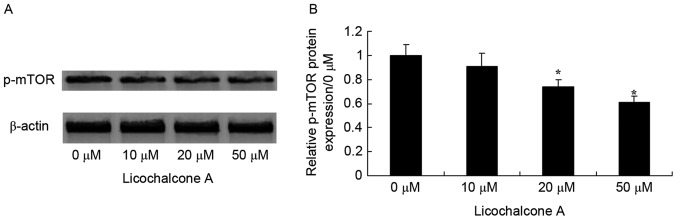
Licochalcone A inhibits Akt and mTOR signaling in MCF-7 cells
To determine whether the anticancer effect of Licochalcone A on MCF-7 cells was mediated by the Akt/mTOR signaling pathway, p-Akt and Akt expression was detected in MCF-7 cells incubated with various concentrations of Licochalcone A. The expression of p-Akt/Akt and p-mTOR/mTOR was significantly reduced in MCF-7 cells following treatment with 20 and 50 µM Licochalcone A, compared with the 0 µM control group (P<0.01; Figs. 3 and 4).
Figure 3.
Licochalcone A significantly inhibits Akt signaling in MCF-7 cells. (A) Representative western blot analysis and (B) statistical analysis of p-Akt and total Akt expression in MCF-7 cells following treatment with the indicated concentrations of Licochalcone A for 48 h. *P<0.01 compared with the 0 µM control group. Akt, RAC-α serine-threonine-protein kinase; p-, phosphorylated.
Figure 4.
Licochalcone A significantly inhibits mTOR signaling in MCF-7 cells. (A) Representative western blot analysis and (B) statistical analysis of p-mTOR expression in MCF-7 cells following treatment with the indicated concentrations of Licochalcone A for 48 h. *P<0.01 compared with the 0 µM control group. mTOR, mammalian target of rapamycin; p, phosphorylated.
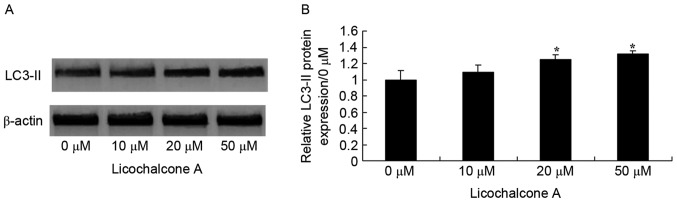
Licochalcone A promotes autophagy of MCF-7 cells
To determine the association between the anticancer effects of Licochalcone A and autophagy following treatment of MCF-7 cells with Licochalcone A, AO staining was performed. Fluorescence microscopy indicated that 20 or 50 µM Licochalcone A induces autophagy of MCF-7 cells (Fig. 5). In addition, western blot analysis demonstrated that the expression of LC3-II, a protein recruited to autophagosomal membranes, was significantly increased in MCF-7 cells treated with 20 or 50 µM Licochalcone A, as compared with in the 0 µM control group (P<0.01; Fig. 6).
Figure 5.
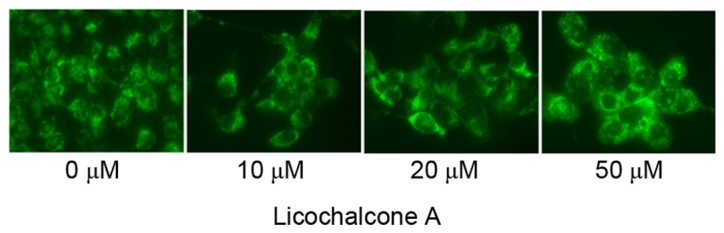
Licochalcone A induces autophagy of MCF-7 cells. Representative images of acridine orange staining of MCF-7 cells treated with the indicated concentrations of Licochalcone A (magnification, ×40).
Figure 6.
Licochalcone A significantly increases LC3-II expression in MCF-7 cells. (A) Representative western blot analysis and (B) statistical analysis of LC3-II expression in MCF-7 cells following treatment with the indicated concentrations of Licochalcone A for 48 h. *P<0.01, compared with the 0 µM control group. LC3-II, conjugated form of microtubule-associated proteins 1A/1B light chain 3.
Licochalcone A promotes apoptosis of MCF-7 cells
To elucidate the effects of Licochalcone A on the apoptosis rate of MCF-7 cells, an Annexin V-FITC/PI staining assay was performed on MCF-7 cells treated with various concentrations of Licochalcone A. Concentrations of 20 or 50 µM Licochalcone A significantly increased the apoptosis rate of MCF-7 cells compared with the 0 µM control group (P<0.01; Fig. 7).
Figure 7.
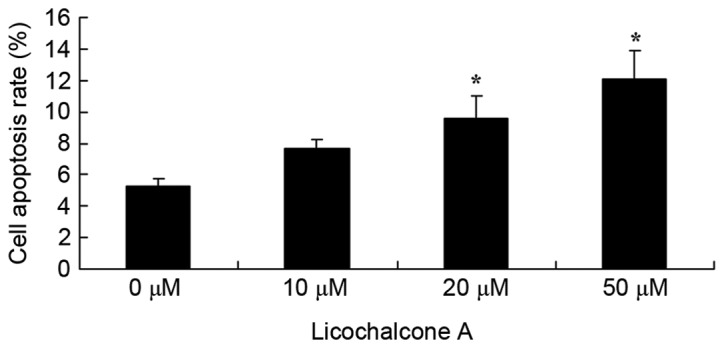
Licochalcone A induces apoptosis of MCF-7 cells. Apoptosis rate of MCF-7 cells treated with the indicated concentrations of Licochalcone A for 48 h. *P<0.01, compared with the 0 µM control group.
Licochalcone A increases caspase-3 activity in MCF-7 cells
To determine the effects of Licochalcone A on the caspase-3 activity of MCF-7 cells, caspase-3 activity was detected using a caspase-3 activity kit. Following the administration of Licochalcone A at 20 or 50 µM in MCF-7 cells, caspase-3 activity was significantly enhanced compared with the 0 µM control group (P<0.01; Fig. 8).
Figure 8.
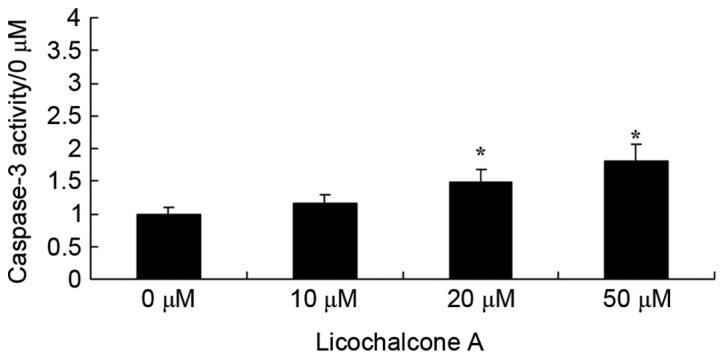
Licochalcone A significantly increases caspase-3 activity in MCF-7 cells. Caspase-3 activity in MCF-7 cells treated with the indicated concentrations of Licochalcone A for 48 h. *P<0.01, compared with the 0 µM control group.
Licochalcone A increases apoptosis regulator Bcl-2 signaling pathway activity in MCF-7 cells
To confirm the anticancer effects of Licochalcone A on the Bcl-2 signaling pathway activity of MCF-7 cells, Bcl-2 expression was examined in MCF-7 cells treated with various concentrations of Licochalcone A. Bcl-2 expression decreased in a dose-dependent manner and was significantly decreased following treatment with 20 and 50 µM Licochalcone A, as compared with the 0 µM control group (P<0.01; Fig. 9) These results suggest that Licochalcone A induces the apoptosis of human breast cancer cells.
Figure 9.
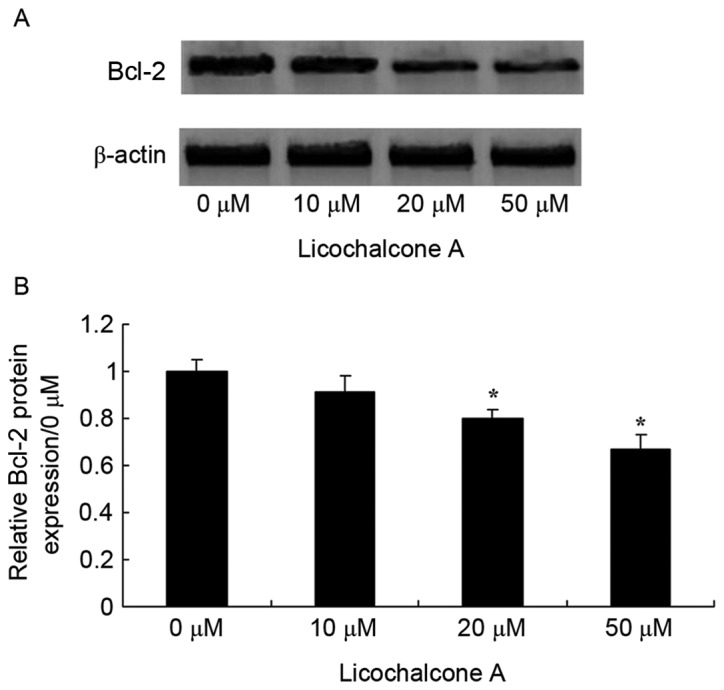
Licochalcone A significantly decreases Bcl-2 expression in MCF-7 cells. (A) Representative western blot analysis and (B) statistical analysis of Bcl-2 expression in MCF-7 cells following treatment with the indicated concentrations of Licochalcone A for 48 h. *P<0.01, compared with the 0 µM control group. Bcl-2, B-cell lymphoma-2.
Discussion
Breast cancer is a common type of malignant cancer with an incidence that increases each year (12). Although the incidence of breast cancer is low in China, compared with in western countries, the incidence in China is rising annually (13). Autophagy is a process that leads to the degradation of proteins and organelles in eukaryotic cells. A previous study suggested that autophagy is associated with a number of diseases, including cancer, neurodegeneration and cardiac hypertrophy (6). The data from the present study demonstrated that Licochalcone A decreases cell viability, induces apoptosis and increases the caspase-3 activity in MCF-7 cells. Previous studies have demonstrated that Licochalcone A suppresses growth of human esophageal carcinoma (14), human lung cancer (15) and human oral cancer cells (16).
Autophagy promotes the survival of cells and maintains homeostasis by degrading damaged organelles and proteins (17). The PI3K/Akt/mTOR signaling pathway promotes cellular growth, migration, protein synthesis, survival and metabolism in response to growth factors and nutrient availability (8). PI3K activates Akt, which leads to the phosphorylation of mTOR via a number of regulators (17). In the present study, Licochalcone A suppressed the PI3K/Akt/mTOR signaling pathway in MCF-7 cells. Tsai et al (18) demonstrated that Licochalcone A induces autophagy through the inhibition of the PI3K/Akt/mTOR signaling pathway in human cervical cancer cells (18). In addition, Hao et al (19) suggested that Licochalcone A induces the apoptosis of BGC-823 human gastric cancer cells via the PI3K/AKT signaling pathway.
Apoptosis and autophagy are forms of programmed cell death. The morphological manifestations of apoptosis include cell contraction, nuclear fragmentation, chromatin condensation and DNA fragmentation (20). mTOR complex 1 is a negative regulator of autophagy (21). LC3 and autophagy-related protein (Atg)8 serve essential functions in metastasis and the maturation of autophagosomes (21). Prior to induction of autophagy, LC3-I and phosphatidylethanolamine are combined under the actions of Atg3 and Atg7 (22), forming LC3-II. LC3-II is absorbed by the autophagosomes, which then degrade. At present, to the best of our knowledge, no cancer therapies currently exist that act on apoptosis and autophagy (23). Data from the present study demonstrated that Licochalcone A increases the expression of LC3-II, inhibits Bcl-2 expression and increases caspase-3 activity in MCF-7 cells. Tsai et al (18) demonstrated that Licochalcone A induces autophagy through LC3-II and the inactivation of the PI3K/Akt/mTOR signaling pathway in human cervical cancer cells. The results from the present study revealed that Licochalcone A could induce apoptosis in MCF-7 cells via the caspase-dependent Bcl-2 apoptosis signaling pathway.
In conclusion, in the present study, Licochalcone A was observed to suppress the viability of MCF-7 cells, induce autophagy and apoptosis, and significantly increase the levels of LC3-II protein expression in human breast cancer cells through the suppression of PI3K/Akt/mTOR signaling pathway activation.
References
- 1.Bouvet V, Jans HS, Wuest M, Soueidan OM, Grant T, Mercer J, McEwan AJ, West FG, Cheeseman CI, Wuest F. Erratum: Automated synthesis and dosimetry of 6-deoxy-6-[F]fluoro-D-fructose (6-[F]FDF): A radiotracer for imaging of GLUT5 in breast cancer. Am J Nucl Med Mol Imaging. 2014;5:95. [PMC free article] [PubMed] [Google Scholar]
- 2.Zarzynska JM. The importance of autophagy regulation in breast cancer development and treatment. Biomed Res Int. 2014;2014:710345. doi: 10.1155/2014/710345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pu Z, Zhang X, Chen Q, Yuan X, Xie H. Establishment of an expression platform of OATP1B1 388GG and 521CC genetic polymorphism and the therapeutic effect of tamoxifen in MCF-7 cells. Oncol Rep. 2015;33:2420–2428. doi: 10.3892/or.2015.3864. [DOI] [PubMed] [Google Scholar]
- 4.Wang LW, Qu AP, Yuan JP, Chen C, Sun SR, Hu MB, Liu J, Li Y. Computer-based image studies on tumor nests mathematical features of breast cancer and their clinical prognostic value. PLoS One. 2013;8:e82314. doi: 10.1371/journal.pone.0082314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chukkapalli S, Amessou M, Dilly AK, Dekhil H, Zhao J, Liu Q, Bejna A, Thomas RD, Bandyopadhyay S, Bismar TA, et al. Role of the EphB2 receptor in autophagy, apoptosis and invasion in human breast cancer cells. Exp Cell Res. 2014;320:233–246. doi: 10.1016/j.yexcr.2013.10.022. [DOI] [PubMed] [Google Scholar]
- 6.Fang WB, Yao M, Jokar I, Alhakamy N, Berkland C, Chen J, Brantley-Sieders D, Cheng N. The CCL2 chemokine is a negative regulator of autophagy and necrosis in luminal B breast cancer cells. Breast Cancer Res Treat. 2015;150:309–320. doi: 10.1007/s10549-015-3324-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lamparska-Przybysz M, Gajkowska B, Motyl T. BID-deficient breast cancer MCF-7 cells as a model for the study of autophagy in cancer therapy. Autophagy. 2006;2:47–48. doi: 10.4161/auto.2149. [DOI] [PubMed] [Google Scholar]
- 8.Zhang H, Guo M, Chen JH, Wang Z, Du XF, Liu PX, Li WH. Osteopontin knockdown inhibits αv,β3 integrin-induced cell migration and invasion and promotes apoptosis of breast cancer cells by inducing autophagy and inactivating the PI3K/Akt/mTOR pathway. Cell Physiol Biochem. 2014;33:991–1002. doi: 10.1159/000358670. [DOI] [PubMed] [Google Scholar]
- 9.Kim KH, Yoon G, Cho JJ, Cho JH, Cho YS, Chae JI, Shim JH. Licochalcone A induces apoptosis in malignant pleural mesothelioma through downregulation of Sp1 and subsequent activation of mitochondria-related apoptotic pathway. Int J Oncol. 2015;46:1385–1392. doi: 10.3892/ijo.2015.2839. [DOI] [PubMed] [Google Scholar]
- 10.Chen M, Theander TG, Christensen SB, Hviid L, Zhai L, Kharazmi A. Licochalcone A, a new antimalarial agent, inhibits in vitro growth of the human malaria parasite Plasmodium falciparum and protects mice from P. yoelii infection. Antimicrob Agents Chemother. 1994;38:1470–1475. doi: 10.1128/AAC.38.7.1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shibata S, Inoue H, Iwata S, Ma RD, Yu LJ, Ueyama H, Takayasu J, Hasegawa T, Tokuda H, Nishino A, et al. Inhibitory effects of licochalcone A isolated from Glycyrrhiza inflata root on inflammatory ear edema and tumour promotion in mice. Planta Med. 1991;57:221–224. doi: 10.1055/s-2006-960078. [DOI] [PubMed] [Google Scholar]
- 12.Rovito D, Giordano C, Plastina P, Barone I, De Amicis F, Mauro L, Rizza P, Lanzino M, Catalano S, Bonofiglio D, Andò S. Omega-3 DHA- and EPA-dopamine conjugates induce PPARgamma-dependent breast cancer cell death through autophagy and apoptosis. Biochim Biophys Acta. 2015;1850:2185–2195. doi: 10.1016/j.bbagen.2015.08.004. [DOI] [PubMed] [Google Scholar]
- 13.Jain K, Paranandi KS, Sridharan S, Basu A. Autophagy in breast cancer and its implications for therapy. Am J Cancer Res. 2013;3:251–265. [PMC free article] [PubMed] [Google Scholar]
- 14.Yang P, Tuo L, Wu Q, Cao X. Licochalcone-A sensitizes human esophageal carcinoma cells to TRAIL-mediated apoptosis by proteasomal degradation of XIAP. Hepatogastroenterology. 2014;61:1229–1234. [PubMed] [Google Scholar]
- 15.Huang HC, Tsai LL, Tsai JP, Hsieh SC, Yang SF, Hsueh JT, Hsieh YH. Licochalcone A inhibits the migration and invasion of human lung cancer cells via inactivation of the Akt signaling pathway with downregulation of MMP-1/−3 expression. Tumour Biol. 2014;35:12139–12149. doi: 10.1007/s13277-014-2519-3. [DOI] [PubMed] [Google Scholar]
- 16.Zeng G, Shen H, Yang Y, Cai X, Xun W. Licochalcone A as a potent antitumor agent suppresses growth of human oral cancer SCC-25 cells in vitro via caspase-3 dependent pathways. Tumour Biol. 2014;35:6549–6555. doi: 10.1007/s13277-014-1877-1. [DOI] [PubMed] [Google Scholar]
- 17.Chang L, Graham PH, Hao J, Ni J, Bucci J, Cozzi PJ, Kearsley JH, Li Y. PI3K/Akt/mTOR pathway inhibitors enhance radiosensitivity in radioresistant prostate cancer cells through inducing apoptosis, reducing autophagy, suppressing NHEJ and HR repair pathways. Cell Death Dis. 2014;5:e1437. doi: 10.1038/cddis.2014.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tsai JP, Lee CH, Ying TH, Lin CL, Lin CL, Hsueh JT, Hsieh YH. Licochalcone A induces autophagy through PI3K/Akt/mTOR inactivation and autophagy suppression enhances Licochalcone A-induced apoptosis of human cervical cancer cells. Oncotarget. 2015;6:28851–28866. doi: 10.18632/oncotarget.4767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hao W, Yuan X, Yu L, Gao C, Sun X, Wang D, Zheng Q. Licochalcone A-induced human gastric cancer BGC-823 cells apoptosis by regulating ROS-mediated MAPKs and PI3K/AKT signaling pathways. Sci Rep. 2015;5:10336. doi: 10.1038/srep10336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu C, Xu P, Chen D, Fan X, Xu Y, Li M, Yang X, Wang C. Roles of autophagy-related genes Beclin-1 and LC3 in the development and progression of prostate cancer and benign prostatic hyperplasia. Biomed Rep. 2013;1:855–860. doi: 10.3892/br.2013.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pan ST, Qin Y, Zhou ZW, He ZX, Zhang X, Yang T, Yang YX, Wang D, Qiu JX, Zhou SF. Plumbagin induces G2/M arrest, apoptosis, and autophagy via p38 MAPK- and PI3K/Akt/mTOR-mediated pathways in human tongue squamous cell carcinoma cells. Drug Des Devel Ther. 2015;9:1601–1626. doi: 10.2147/DDDT.S76057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zou M, Lu N, Hu C, Liu W, Sun Y, Wang X, You Q, Gu C, Xi T, Guo Q. Beclin 1-mediated autophagy in hepatocellular carcinoma cells: implication in anticancer efficiency of oroxylin A via inhibition of mTOR signaling. Cell Signal. 2012;24:1722–1732. doi: 10.1016/j.cellsig.2012.04.009. [DOI] [PubMed] [Google Scholar]
- 23.Li JP, Yang YX, Liu QL, Zhou ZW, Pan ST, He ZX, Zhang X, Yang T, Pan SY, Duan W, et al. The pan-inhibitor of Aurora kinases danusertib induces apoptosis and autophagy and suppresses epithelial-to-mesenchymal transition in human breast cancer cells. Drug Des Devel Ther. 2015;9:1027–1062. doi: 10.2147/DDDT.S74412. [DOI] [PMC free article] [PubMed] [Google Scholar]