Abstract
Introduction: Pulsed dye laser (PDL) for treatment of port wine stain (PWS) usually causes some acute adverse effects, including pain, erythema, scabbing and swelling. This study aimed to determine whether topical halometasone can be used to reduce these acute adverse effects for post-PDL care of patients.
Methods: A total of 40 PWS subjects were enrolled in this study and randomly assigned into two regimens: PDL alone and PDL + halometasone. All subjects were given a single treatment of PDL with wavelength of 595 nm, fluence of 8.0~13.5 J/cm2, pulse duration of 0.45~20 ms (We mainly used purpuric pulse duration for PWS) and spot size of 7 mm. Subjects in the PDL + halometasone group received topical application of halometasone daily for 3 days. Subjects were followed-up on days 3, 7 and one month post-PDL to evaluate the reduction of adverse effects.
Results: Subjects in the PDL + halometasone group had significantly improved erythema and scab scores and had less erythema duration days as compared to those in the PDL alone group. Topical halometasone also showed a decrease in pain index among subjects, but was statistically insignificant. Topical halometasone did not affect the ultimate efficacy of PDL treatment for PWS.
Conclusion: Topical halometasone can significantly reduce acute adverse effects induced by PDL for treatment of PWS birthmarks, thus providing a safe and efficacious solution for post-PDL care of PWS patients.
Keywords: Port wine stain, Pulsed dye laser, Halometasone, Adverse effects
Introduction
Port wine stain (PWS) is a congenital, progressive vascular malformation of human skin involving a superficial vascular plexus with an occurrence of 3~5 children per 1000 live births.1-3 Personality development is adversely affected in patients since most malformations occur on the face. Low self-esteem in such patients and problems with interpersonal relationships have been reported in many studies.4-6 The pathogenesis of PWS remains incompletely understood. A sporadic somatic guanine nucleotide-binding protein, G alpha subunit q (GNAQ) mutation (R183Q), has been identified in PWS lesions with an average mutation frequency lower than 5%.7,8 Tan et al have further found that the c-Jun N-terminal kinases and extracellular signal regulated kinases are consecutively activated in both infantile and adult PWS blood vessels,9 which may partially be a result from GNAQ mutation (R183Q) in a subset of PWS patients.10
The dilation of PWS blood vessels in patients is progressive with aging: PWS lesions are flat red macules in childhood, but become darken to purple, and often develop into vascular nodules by middle ages.11,12 Pulsed dye laser (PDL) is the current standard treatment for PWS, which converts incoming photo energy to heat, thus inducing blood vessel wall necrosis.13-19 However, PDL usually induces some acute adverse effects on patients after laser treatment, including pain, erythema, scabbing and swelling. These adverse effects cause discomfort to patients, especially infantile and pediatric subjects. Therefore, efficient reduction of these adverse effects is very important for post-PDL care of those young subjects. Numerous types of corticosteroids have been used with PDL to reduce the post-PDL inflammatory response or prevent scar formation.20-22 However, high incidence of adverse sequelae following application of intralesional corticosteroid (triamcinolone acetonide, 5-FU)20 and high cost of these agents to many impoverished patients visiting our clinic have driven us to look for a more affordable corticosteroid with fewer side effects. Halometasone is a low-cost synthetic tri-halogenated corticosteroid that shows anti-inflammatory properties in treating dermatitis and chronic psoriasis vulgaris.23,24 In this study, we hypothesized that topical halometasone could significantly reduce PDL-induced acute adverse effects for treatment of PWS birthmarks.
Patients and Methods
Forty patients with diagnosis of PWS were recruited from the Outpatient Clinic of Dermatology at the author’s institution. The subjects were randomly assigned into 2 groups: PDL alone and PDL + halometasone. All subjects received a single dose of PDL treatment on their PWS lesions with a wavelength of 595 nm, fluence of 8.0~13.5 J/cm2, pulse duration of 0.45~20 ms (We mainly used purpuric pulse duration for PWS), DCD 30/20 and spot size of 7 mm (Vbeam-Perfecta, Candela, Wayland, MA). The subjects in the PDL + halometasone group were instructed to apply a 1 mm thin layer of topical halometasone cream (0.05% w/w [6α, 11β, 16α]-2-chloro-6, 9-difluoro-11, 17, 21-trihydroxy-16-methylpregna-1, 4-diene-3,20-dione; Bright Future Pharmaceutical Laboratories Ltd., Hong Kong, China) over PDL-treated areas daily for 3 days. The subjects in the PDL alone group received topical vaseline cream as the placebo control. All subjects were followed-up on days 3, 7 and one month post-PDL to evaluate reduction of adverse effects. Photos were taken during each follow-up visit. The duration days of existing pain, erythema, scabbing and swelling were recorded. The indexes of pain, swelling, erythema and scabbing were scored on the thirdday post-PDL based on severity on a 1 to 4 scale (1: none or 1~25% severity degree; 2: mild or 25~50%; 3: moderate or 50~75%; 4: severe or 75~100%). The evaluations were performed by two independent dermatologists. Statistical analysis was performed using a paired t test with SPSS software (version 15.0, Chicago, IL). P value < 0.05 was considered statistically significant.
Results and Discussion
All 40 subjects completed the 1-month protocol and follow-up evaluations. There was no statistical significance in subject age, lesion size and lesion color between the 2 groups (Table 1). All the subjects reported that the pain from the PWS lesions post-PDL treatment lasted for about one day. Topical halometasone showed a reduction in the pain index of the PDL + halometasone group as compared to the PDL alone group, but it was not statistically significant (1.2 ± 0.41 vs. 1.4 ± 0.50, P = 0.0881, Table 1). There was no difference in swelling days and scores between the 2 groups (Table 1). The subjects in the PDL + halometasone group showed a significantly lower erythema score and shorter erythema duration time than those in the PDL alone group (erythema scores: 0.4 ± 0.82 vs. 0.9 ± 0.91, P = 0.0381; erythema duration dates: 3.7 ± 8.11 vs. 7.3 ± 10.49, P = 0.0494; Table 1 & Figure 1). Topical halometasone also showed a significant reduction of scab scores in the PDL + halometasone group as compared to the PDL alone group (1.15 ± 0.77 vs. 1.65 ± 1.22, P = 0.0486, Table 1 & Figure 1). There was no significant change in the duration time of scabbing between the 2 groups (Table 1). No hyperpigmentation or hypopigmentation was observed in any of the subjects. Halometasone 0.05% showed good tolerability in the study; no obvious adverse event was reported from the subjects. There was no significant difference in the improvement of bleached PWS lesion one month post-PDL between the 2 groups, suggesting topical halometasone did not affect the ultimate efficacy of PDL treatment on PWS lesions among the subjects.
Table 1. Comparison of PDL-Induced Acute Adverse Effects Between the PDL Alone and PDL + Halometasone Groups .
| Treatment Group | PWS Alone | PWS + Halometasone | P Value |
| N | 20 | 20 | |
| Age | 16 ± 10.814 | 13 ± 8.146 | 0.2207 |
| Lesion size (cm2) | 29.9 ± 23.510 | 37.9 ± 19. 488 | 0.1243 |
| Lesion color score | 4.85 ± 1.136 | 5.6 ± 1.903 | 0.0692 |
| Days for pain | 1.0 ± 0.00 | 1.0 ± 0.00 | |
| Pain index | 1.4 ± 0.50 | 1.2 ± 0.41 | 0.0881 |
| Days for swelling | 3.9 ± 1.21 | 3.9 ± 2.29 | 0.5000 |
| Swelling score | 1.0 ± 0.00 | 1.0 ± 0.00 | |
| Erythema score | 0.9 ± 0.91 | 0.4 ± 0.82 | 0.0381 |
| Days for erythema | 7.3 ± 10.49 | 3.7 ± 8.11 | 0.0494 |
| Scab score | 1.65 ± 1.22 | 1.15 ± 0.77 | 0.0486 |
| Days for scabbing | 7.2 ± 5.82 | 8.7 ± 3.58 | 0.1744 |
Abbreviations: PDL, pulsed dye laser; PWS, port wine stain.
Figure 1.
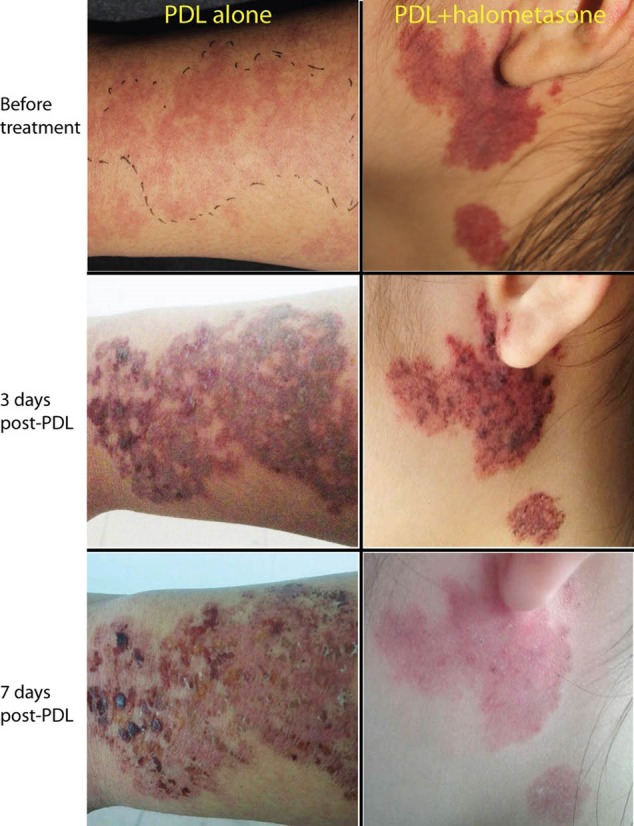
Topical halometasone significantly reduces PDL-induced erythema and scabbing. The individual subjects in the PDL alone group (16-year-old male with PWS lesions on the right arm, left panel) and PDL + halometasone group (20-year-old female with PWS lesions on the left lateral face, right panel) received one pass of PDL treatment, followed by topical application of Vaseline cream (placebo) or 0.05% halometasone daily, respectively, for three days. The images were taken before PDL treatment, 3 days and 7 days post-PDL treatment.
Many acute adverse effects can be produced on PWS lesions after PDL treatment, such as pain, swelling, erythema, scabbing, and changes in pigmentation. The majority of them will diminish within days to 2 weeks post-PDL therapy without further treatment. However, these adverse effects can cause temporary, sometimes severe, discomfort to patients in the first week, especially to infantile and pediatric subjects. Therefore, how to reduce PDL-induced acute adverse effects is a very important step for post-PDL care. Many types of corticosteroids have been used with PDL to reduce the post-PDL inflammatory response or prevent scar formations.20-22 However many adverse sequelae, such as hypopigmentation, telangiectasis, and skin atrophy, have been observed in a substantial portion (50%) of the segments treated by corticosteroid (triamcinolone acetonide, 5-FU) intralesionally alone.20 Furthermore, affordability of the prescribed corticosteroid is also a consideration for patients from rural China. Therefore, looking for a more affordable corticosteroid with fewer side effects is very important for many patients visiting our clinic. Halometasone is a potent synthetic corticosteroid that has been shown to be more effective than betamethasone in treating dermatitis.22 Due to its profound and strong anti-inflammatory functions, we hypothesized that topical Halometasone could effectively reduce PDL-induced acute adverse effects including pain, swelling, scabbing and erythema. Indeed, our results showed topical halometasone could significantly reduce the severity of scabbing and erythema induced by PDL for the treatment of PWS. Many clinical studies have shown halometasone is well tolerated.23-26 Some mild adverse events, such as a stinging and burning sensation, have been reported in some patients when halometasone is applied for more than 18 days.26 In this study, our purpose was to reduce PDL-induced adverse effects which were instant so that halometasone was applied only for 3 days. We did not observe any side effects in the participants during the short time of topical application. Therefore, topical halometasone has a good safety and tolerability profile for patients. Furthermore, the low cost of halometasone also offers an economical benefit for patients.
In conclusion, topical halometasone is efficacious and safe to reduce PDL-induced acute adverse effects, such as pain, erythema and scabbing. The authors recommend its use for post-PDL care of PWS patients.
Conflict of Interests
None.
Ethical Considerations
The study protocol was approved by the Institutional Review Board (IRB) at the authors’ hospital (Registration number KY20150727-1), and therefore been performed in accordance with Declaration of Helsinki and its amendments.
Acknowledgements
This study was supported by National Natural Scientific Foundation of China (grant number 81301355 to LG and grants 81430073 and 81220108016 to GW).
Please cite this article as follows: Gao L, Qian L, Wang L, et al. Topical halometasone reduces acute adverse effects induced by pulsed dye laser for treatment of port wine stain birthmarks. J Lasers Med Sci. 2018;9(1):19-22. doi:10.15171/jlms.2018.05.
References
- 1. Mulliken JB, Young AR, Eds. Vascular Birthmarks--Hemangiomas and Malformations. Philadelphia: W. B. Saunders Co; 1988.
- 2.Jacobs AH, Walton RG. The incidence of birthmarks in the neonate. Pediatrics. 1976;58(2):218–222. [PubMed] [Google Scholar]
- 3.Pratt AG. Birthmarks in infants. AMA Arch Derm Syphilol. 1953;67(3):302–305. doi: 10.1001/archderm.1953.01540030065006. [DOI] [PubMed] [Google Scholar]
- 4.Heller A, Rafman S, Zvagulis I, Pless I. Birth defects and psychosocial adjustment. Am J Dis Child. 1985;139(3):257–263. doi: 10.1001/archpedi.1985.02140050051021. [DOI] [PubMed] [Google Scholar]
- 5.Malm M, Carlberg M. Port-wine stain--a surgical and psychological problem. Ann Plast Surg. 1988;20(6):512–516. doi: 10.1097/00000637-198806000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Kalick SM. Toward an interdisciplinary psychology of appearances. Psychiatry. 1978;41(3):243–253. [PubMed] [Google Scholar]
- 7.Shirley MD, Tang H, Gallione CJ. et al. Sturge-Weber syndrome and port-wine stains caused by somatic mutation in GNAQ. N Engl J Med. 2013;368(21):1971–1979. doi: 10.1056/NEJMoa1213507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lian CG, Sholl LM, Zakka LR. et al. Novel genetic mutations in a sporadic port-wine stain. JAMA Dermatol. 2014;150(12):1336–1340. doi: 10.1001/jamadermatol.2014.1244. [DOI] [PubMed] [Google Scholar]
- 9.Tan W, Chernova M, Gao L. et al. Sustained activation of c-Jun N-terminal and extracellular signal-regulated kinases in port-wine stain blood vessels. J Am Acad Dermatol. 2014;71(5):964–968. doi: 10.1016/j.jaad.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tan W, Nadora DM, Gao L, Wang G, Mihm MC Jr, Nelson JS. The somatic GNAQ mutation (R183Q) is primarily located within the blood vessels of port wine stains. J Am Acad Dermatol. 2016;74(2):380–383. doi: 10.1016/j.jaad.2015.09.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lever WF, Schaumburg-Lever G, eds. Histopathology of the Skin. 7th ed. Philadelphia: J B Lippincott Co; 1990.
- 12.Geronemus RG, Ashinoff R. The medical necessity of evaluation and treatment of port-wine stains. J Dermatol Surg Oncol. 1991;17(1):76–79. doi: 10.1111/j.1524-4725.1991.tb01597.x. [DOI] [PubMed] [Google Scholar]
- 13.Nelson JS, Milner TE, Anvari B. et al. Dynamic epidermal cooling during pulsed laser treatment of port-wine stain A new methodology with preliminary clinical evaluation. Arch Dermatol. 1995;131(6):695–700. [PubMed] [Google Scholar]
- 14.Nelson JS, Milner TE, Anvari B, Tanenbaum BS, Svaasand LO, Kimel S. Dynamic epidermal cooling in conjunction with laser-induced photothermolysis of port wine stain blood vessels. Lasers Surg Med. 1996;19(2):224– 229 . doi: 10.1002/(sici)1096-9101(1996)19:2<224::aidlsm15>3.0.co;2-6.. [DOI] [PubMed] [Google Scholar]
- 15.Chang CJ, Nelson JS. Cryogen spray cooling and higher fluence pulsed dye laser treatment improve port-wine stain clearance while minimizing epidermal damage. Dermatol Surg. 1999;25(10):767–772. doi: 10.1046/j.1524-4725.1999.99100.x. [DOI] [PubMed] [Google Scholar]
- 16.Chang CJ, Kelly KM, Van Gemert MJ, Nelson JS. Comparing the effectiveness of 585-nm vs 595-nm wavelength pulsed dye laser treatment of port wine stains in conjunction with cryogen spray cooling. Lasers Surg Med. 2002;31(5):352–358. doi: 10.1002/lsm.10102. [DOI] [PubMed] [Google Scholar]
- 17.Chiu CH, Chan HH, Ho WS, Yeung CK, Nelson JS. Prospective study of pulsed dye laser in conjunction with cryogen spray cooling for treatment of port wine stains in Chinese patients. Dermatol Surg. 2003;29(9):909–915. doi: 10.1046/j.1524-4725.2003.29255.x. [DOI] [PubMed] [Google Scholar]
- 18.Waldorf HA, Alster TS, McMillan K, Kauvar AN, Geronemus RG, Nelson JS. Effect of dynamic cooling on 585-nm pulsed dye laser treatment of port-wine stain birthmarks. Dermatol Surg. 1997;23(8):657–662. doi: 10.1111/j.1524-4725.1997.tb00385.x. [DOI] [PubMed] [Google Scholar]
- 19.Fiskerstran EJ, Ryggen K, Norvang LT, Svaasand LO. Clinical effects of dynamic cooling during pulsed laser treatment of port-wine stains. Lasers Med Sci. 1997;12(4):320–327. doi: 10.1007/bf02767154. [DOI] [PubMed] [Google Scholar]
- 20.Manuskiatti W, Fitzpatrick RE. Treatment response of keloidal and hypertrophic sternotomy scars: comparison among intralesional corticosteroid, 5-fluorouracil, and 585-nm flashlamp-pumped pulsed-dye laser treatments. Arch Dermatol. 2002;138(9):1149–1155. doi: 10.1001/archderm.138.9.1149. [DOI] [PubMed] [Google Scholar]
- 21.Alster T. Laser scar revision: comparison study of 585-nm pulsed dye laser with and without intralesional corticosteroids. Dermatol Surg. 2003;29(1):25–29. doi: 10.1046/j.1524-4725.2003.29024.x. [DOI] [PubMed] [Google Scholar]
- 22.Asilian A, Darougheh A, Shariati F. New combination of triamcinolone, 5-Fluorouracil, and pulsed-dye laser for treatment of keloid and hypertrophic scars. Dermatol Surg. 2006;32(7):907–15. doi: 10.1111/j.1524-4725.2006.32195.x. [DOI] [PubMed] [Google Scholar]
- 23.Galbiati G, Bonfacini V, Candiani F. Halometasone cream by day and halometasone ointment at night for the treatment of patients with chronic psoriasis vulgaris. J Int Med Res. 1983;11 Suppl 1:31–33. [PubMed] [Google Scholar]
- 24.Yawalkar SJ, Macarol V, Montanari C. An overview of international clinical trials with halometasone cream. J Int Med Res. 1983;11 Suppl 1:1–7. [PubMed] [Google Scholar]
- 25.Schuppli R, Dressler H, Yawalkar SJ, Weirich EG. [Comparative clinical trial of a new trihalogenated dermatocorticoid (halometasone) versus betamethasone dipropionate] Z Hautkr. 1983;58(4):230–237. [PubMed] [Google Scholar]
- 26.The Execare Working Group. Halometasone 005% cream in eczematous dermatoses. J Clin Aesthet Dermatol. 2013;6(11):39–44. [PMC free article] [PubMed] [Google Scholar]


