Rotavirus is the leading cause of severe childhood gastroenteritis. Each year, rotavirus is responsible for about 25 million clinic visits, two million hospital admissions, and 180 000–450 000 deaths in children under 5 years of age globally.1–3 Although rotavirus infection is prevalent worldwide, most deaths from this infection occur in developing countries (fig 1). Gastroenteritis caused by rotavirus cannot be clinically distinguished from that caused by other enteric pathogens; diagnosis requires testing of fecal specimens with commercially available assays. However, rotavirus is not routinely tested for in patients with gastroenteritis because the results do not alter clinical management, which relies mainly on appropriate rehydration therapy. Orally administered live attenuated vaccines that mimic natural infection offer the best protection against rotavirus. Two licensed rotavirus vaccines have been available since 2006 and have been implemented in many countries. We review approaches to diagnosis, management, and prevention of rotavirus gastroenteritis.
Fig 1.
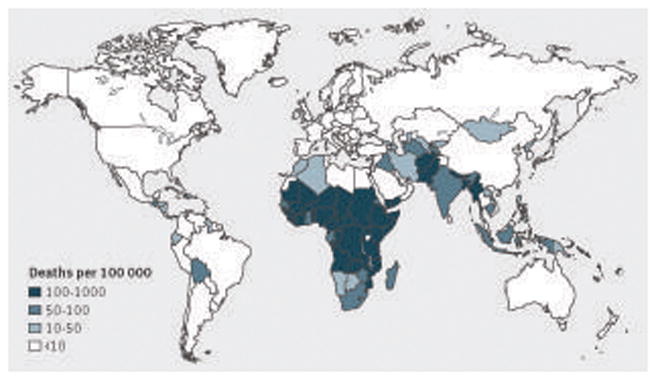
World Health Organization estimates of rotavirus mortality (deaths/100 000 children under 5 years of age) by country in 2008. Reproduced, with permission, from Lancet Infectious Diseases2
How does rotavirus gastroenteritis present clinically?
The clinical spectrum of rotavirus infection ranges from subclinical illness or mild watery diarrhea of limited duration to frequent profuse diarrhea with vomiting and fever that can result in dehydration with shock, electrolyte imbalance, and death. Rotavirus illness usually begins with acute onset of fever and vomiting, followed one or two days later by frequent watery stools. About 30–40% of children may have a moderate fever (temperature >39°C). Vomiting usually lasts for only one or two days and other gastrointestinal symptoms generally resolve in three to seven days.
Although gastroenteritis is the chief manifestation of rotavirus infection, neurologic features—including benign convulsions (usually afebrile but febrile in some cases), encephalitis or encephalopathy, and cerebellitis—have also been described.4 For example, a multicentre study from Canada reported that 7% of 1359 children admitted to hospital with laboratory confirmed rotavirus had seizures at presentation.5 A variety of other clinical conditions (such as sudden infant death syndrome, necrotizing enterocolitis, intussusception, Kawasaki’s disease, and type 1 diabetes) have been associated with rotavirus gastroenteritis, but a causal association has not been confirmed. A transient rise in serum transaminase concentrations is also often seen in patients with rotavirus gastroenteritis.
Who gets rotavirus disease?
Rotavirus infects nearly all children in developed and developing countries by 3–5 years of age. Neonatal infections occur but are often asymptomatic or mild, possibly because of protection from maternal antibody. The incidence of clinical illness peaks in children aged 4–23 months, who are also at greatest risk of severe disease that requires hospital admission. Although repeat infections are common (three or more rotavirus infections occurred in about 42% of children by 2 years of age in one follow-up study in a cohort of Mexican children), symptoms are milder with each subsequent infection.6,7 Therefore, rotavirus infections are usually subclinical or mild in adults, but they can be severe, particularly in immunocompromised and older people.8 Rotavirus is also an important cause of nosocomial diarrhea.
Case-control studies in industrialized countries have shown that lack of breast feeding, prematurity, and low birth weight are associated with increased risk of hospital admission for rotavirus gastroenteritis.9,10 Protracted rotavirus diarrhea with prolonged viral excretion and, in rare instances, systemic dissemination has been described in severely immunodeficient children, particularly those with severe T cell and combined T and B cell deficiencies. People who are immunosuppressed in preparation for bone marrow transplantation are also at risk for severe or even fatal rotavirus disease.
In all regions of the world, rotavirus is the leading cause of hospital admission for gastroenteritis. A systematic review of 131 surveillance studies published from 2001 to 2011 found that rotavirus accounted for 33–49% of hospital admissions for gastroenteritis in countries in different geographic regions and with varying levels of child mortality.2 However, more than 90% of global deaths from rotavirus occur in low income countries in sub-Saharan Africa and South Asia, mainly because of suboptimal access to healthcare, including basic hydration therapy. In addition, compared with industrialized countries, severe rotavirus gastroenteritis occurs at a younger age in developing countries (up to two thirds of all paediatric disease occurs in the first year of life) and coinfections with other enteric pathogens are more common.
Summary points.
Rotavirus is the leading cause of severe gastroenteritis in children worldwide, accounting for 35–40% of hospital admissions for gastroenteritis
Each year, 180 000–450 000 children under 5 years die from rotavirus gastroenteritis, with more than 90% of deaths occurring in developing countries
Because nearly all children are affected by rotavirus by age 5 years, good sanitation and hygiene alone are inadequate for prevention
Orally administered live attenuated vaccines offer the best protection against rotavirus; as of December 2013, national immunization programs of 51 countries include rotavirus vaccine
Such programs have greatly reduced morbidity and mortality from gastroenteritis
A low risk of intussusception has also been documented post-licensure in some countries, but this risk is greatly exceeded by the health benefits of vaccination
Sources and selection criteria.
We looked at recent conference proceedings and searched PubMed, the Cochrane Database of Systematic Reviews, and Clinical Evidence online using the terms “rotavirus”, “rotavirus gastroenteritis”, and “rotavirus vaccines”. We focused on systematic reviews, meta-analyses, and high quality randomized controlled trials published in English within the past 10 years (2004–13).
In temperate climates, rotavirus gastroenteritis shows prominent seasonality, occurring mainly during the fall and winter, with little disease activity during summer months.11 In tropical countries, rotavirus occurs all year round, although incidence often increases during the cool dry months.
How do rotaviruses cause diarrhea?
Mechanisms of rotavirus pathogenesis have been studied in animal models and humans. The infectious dose of rotavirus is estimated to be 100–1000 viral particles.12 Transmission of rotavirus occurs mainly through the fecal-oral route, and viral spread can occur through contaminated hands, environmental surfaces and objects, and occasionally food and water. Rotavirus exclusively infects the mature differentiated enterocytes found at the tips of the villi in the small intestine (fig 2). Attachment of rotavirus to its sialoglycoprotein and integrin receptors is mediated mainly by viral protein 4 (VP4), but neutralizing antibodies directed against VP4 or VP7 (or both) can prevent viral binding and penetration. Infection of the enterocyte leads to virus entry, uncoating of the virus, transcription of nucleic acid, translation of viral proteins, formation of viroplasms, and apical release of the virus and viral protein by a non-classic secretory pathway.13 The progeny virus are produced after 10–12 hours and released in large numbers into the intestinal lumen. Rotavirus can infect neighboring cells, leading to continued replication and shedding, which consists initially of a high viral load. In children, the viral load decreases rapidly as diarrhea resolves, but viral nucleic acid can be detected at low levels for several weeks.14
Fig 2.
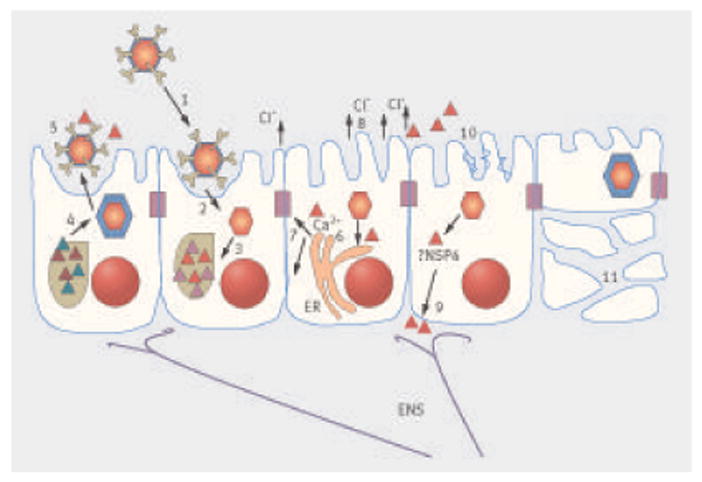
Mechanisms of rotavirus pathogenesis. Most information on rotavirus pathogenesis is derived from small animal models. Infection leads to attachment of the rotavirus to enterocytes (1), uncoating of the virus (2), followed by transcription and translation of viral proteins (3). This leads to the formation of a viroplasm, from which double layered particles assemble (4) and acquire outer coat proteins to form triple layered mature particles, which are shed by a non-classical secretory pathway (5). The viral enterotoxin non-structural protein 4 (NSP4; red triangles) is also released with the mature virions. NSP4 induces the release of intracellular calcium (Ca2+) (6) from the endoplasmic reticulum (ER) and disrupts tight junctions (7), resulting in paracellular leakage of fluid and electrolytes. NSP4 can act on crypt cells to induce chloride (Cl−) secretion directly (8) or through the enteric nervous system (ENS) (9), both of which draw water into the lumen. A decrease in brush border enzymes induced by infection (10) results in accumulation of sugars in the lumen and osmotic fluid loss. Destruction of intestinal cells later in infection results in loss of surface area and a malabsorptive diarrhea (11)
The pathogenesis of rotavirus diarrhea includes effects on absorption and on secretion. Morphologically, rotavirus infection causes enterocyte death and desquamation, leading to the loss of absorptive villous cells and the proliferation of secretory crypt cells.15 Malabsorption may also result from a virus induced reduction in the expression of absorptive enzymes, as well as paracellular leakage as a consequence of functional changes in tight junctions between enterocytes, which may be mediated by the viral non-structural protein 4 (NSP4). Biopsies show atrophy of the villi and mononuclear cell infiltrates in the lamina propria. Increased chloride secretion and loss of consequent water and electrolyte are mediated by NSP4, which acts as a viral enterotoxin, activating cellular calcium channels and inducing secretory diarrhea. Activation of the enteric nervous system (also thought to be dependent on NSP4) induces secretory diarrhea and increases intestinal motility.13 Villous brush border enzymes such as sucrase and isomaltase also decrease, resulting in accumulation of undigested sugars in the intestinal lumen. This increases the osmotic gradient, which in turn favours further fluid secretion.
Rotavirus infection was previously thought to be limited to the intestine, but several studies over the past decade have shown that rotavirus causes short term viremia in immunocompetent infants as well as in experimentally infected animals.16 The clinical relevance of this systemic spread of rotavirus remains unclear, but it may be linked to the extraintestinal clinical manifestations associated with rotavirus infection.
How is rotavirus diagnosed?
Rotavirus can be detected in stool specimens from children with gastroenteritis by several techniques, including electron microscopy, polyacrylamide gel electrophoresis, antigen detection assays, reverse transcription polymerase chain reaction (RT-PCR), and virus isolation. Diagnosis of rotavirus was initially by electron microscopy, with and without agglutination by immune sera. Large numbers of rotavirus particles (up to 1011/g feces) are excreted during the acute phase of infection, and children with severe diarrhea seem to excrete a greater number of viruses.17 Polyacrylamide gel electrophoresis detects rotavirus RNA extracted directly from stool specimens; the electrophoretic migration pattern of the 11 segments of the double stranded RNA genome permits analysis of the relatedness of circulating strains.18
Children with gastroenteritis are not routinely tested for rotavirus because the results do not alter treatment. When testing is performed, antigen detection tests—including commercially available enzyme linked immunosorbent assays (ELISAs) and immunochromatographic assays—are widely used. Most of these tests have high sensitivity and specificity (90–95%).19 RT-PCR is widely used in research laboratories to detect the viral genome.20 It provides data on the VP7 and VP4 genotypes that form the basis of binary classification (G and P type, respectively) of rotavirus strains.
How is rotavirus gastroenteritis treated?
The management of acute rotavirus gastroenteritis focuses on the treatment and prevention of dehydration. In most situations the clinician will not be aware at the start of treatment whether the gastroenteritis is caused by rotavirus or another pathogen. Initial assessment therefore focuses on determining the degree of dehydration because this will be used to guide and monitor treatment.
Many evidence based reviews and clinical guidelines are available on various aspects of the assessment and treatment of acute gastroenteritis in young children.21–36 However, a recent systematic review of eight guidelines noted considerable variation in their quality, inconsistencies between the recommendations, lack of evidence for many recommendations, and lack of generalisability to general practice.37 These were suggested as possible reasons why adherence to such guidelines is poor in high income countries. For example, the authors noted that in general practice it was unclear “how” or to “whom” oral rehydration therapy should be given. They concluded that future studies, particularly in general practice, need to determine the value of clinical signs and symptoms in assessing dehydration, the optimal dose of oral rehydration solution for each grade of dehydration, and the validity of reasons why clinicians prescribe other drugs. In contrast, another recent assessment of the quality of clinical practice guidelines for acute gastroenteritis, using the appraisal of guidelines for research and evaluation instrument, concluded that the overall quality of these guidelines was fair.38
How should dehydration be assessed?
Several scoring systems have been devised to assess dehydration.39 The World Health Organization scale is probably most widely used in the developing world (table 1),40 whereas the modified four point Gorelick score (box) and clinical dehydration scale (table 2) have been used in developed world settings.32–34,39 The clinical dehydration scale has been reported to help predict a longer length of stay and the need for intravenous fluid rehydration.32 It is generally accepted that conventional clinical signs of dehydration are valid and reliable when used collectively but individually they lack sensitivity and specificity.41 The Vesikari and Clark scores are used in rotavirus vaccine efficacy trials for assessing the severity of gastroenteritis but are not designed to guide clinical management according to the degree of dehydration.35
Table 1.
World Health Organization guidelines for assessing dehydration in children with acute gastroenteritis40
| A | B | C |
|---|---|---|
| Condition* | ||
| Well, alert | Restless, irritable | Lethargic or unconscious |
| Eyes† | ||
| Normal | Sunken | Sunken |
| Thirst | ||
| Drinks normally, not thirsty | Thirsty, drinks eagerly | Drinks poorly, or not able to drink |
| Skin pinch‡ | ||
| Skin goes back quickly | Skin goes back slowly | Skin goes back very slowly |
| Decision | ||
| No signs of dehydration | Some dehydration if there are ≥2 signs in B | Severe dehydration if there are ≥2 signs in C |
Being lethargic and sleepy are not the same. A lethargic child is not simply asleep: the child’s mental state is dull and the child cannot be fully awakened; the child may appear to be drifting into unconsciousness.
In some infants and children the eyes normally appear somewhat sunken. It is helpful to ask the mother if the child’s eyes are normal or more sunken than usual.
The skin pinch is less useful in infants or children with marasmus or kwashiorkor, and in obese children.
Table 2.
Clinical dehydration scale32
| Characteristic | Score* | ||
|---|---|---|---|
| 0 | 1 | 2 | |
| General appearance | Normal | Thirsty, restless, or sleepy; irritable when touched | Drowsy, limp, and cold; may be comatose |
| Eyes | Normal | Slightly sunken | Deeply sunken |
| Tongue | Moist | Sticky | Dry |
| Tears | Present | Decreased | Absent |
Total score: 0=no dehydration; 1–4=some dehydration; 5–8=moderate/severe dehydration.
Oral and intravenous rehydration therapy
Oral and intravenous rehydration is the mainstay of treatment for rotavirus and other causes of acute gastroenteritis. Oral rehydration therapy is the preferred treatment of mild to moderate dehydration in children with acute diarrhea.27,29,41 For every 25 children (95% confidence interval 14 to 100) treated one will not respond and will require intravenous rehydration.27 Expert consensus recommends that children who are very ill, lethargic, drinking poorly, or have shock or near shock are initially treated intravenously. Oral rehydration therapy should not be given to children with intestinal ileus until bowel sounds are audible, or in the presence of glucose malabsorption.29
For more than two decades, WHO recommended the standard glucose based oral rehydration solution (90 mmol/L sodium, 111 mmol/L glucose, and a total osmolarity 311 mmol/L).28 However, subsequent studies have shown that when compared with the standard WHO solution, reduced osmolarity oral rehydration solution is associated with fewer unscheduled intravenous fluid infusions (odds ratio 0.59, 0.45 to 0.79), lower stool volume after randomization, less vomiting, and no increased risk of hyponatremia.28 In 2002, WHO recommended the routine use of a solution with reduced osmolarity (sodium 75 mmol/L, glucose 75 mmol/L, and total osmolarity 224 mmol/L) for non-cholera diarrhea.
To maintain hydration of children with “no signs of dehydration,” WHO recommends giving extra fluid or oral rehydration solution after each loose stool. The suggested volume is 50–100 mL (quarter to half a large cup) for children under 2 years of age and a 100–200 mL (half to one large cup) for those above 2.40 For children with “some signs of dehydration,” WHO recommends correcting hydration over four hours with 75 mL/kg of solution.40 However, a review of eight international guidelines showed that recommendations on the dose of oral rehydration solution were not evidence based and were inconsistent, reflecting the use of expert opinion and differences in how guidelines categorized dehydration.37 The authors concluded that the optimal regimen was unclear.
It is important that breast feeding should continue throughout the rehydration and maintenance phases of treatment.40 Feeding should be restarted as soon as possible because this will reduce the duration and severity of diarrhea, and there is no evidence that early refeeding increases the risk of unscheduled intravenous fluid use, episodes of vomiting, or development of persistent diarrhea.22 Although lactose-free feeds may reduce the duration of diarrhea in children with mild to severe dehydration compared with feeds containing lactose, there is no evidence that lactose containing feeds are harmful for most children with acute gastroenteritis.42
Randomized controlled trials—including some that specifically assessed efficacy against rotavirus gastroenteritis—have shown that other treatments such as probiotics, zinc, ondansetron, nitazoxanide, and some biological compounds are effective in the management of acute gastroenteritis.21,23,24,40–45 However, recommendations for the use of these treatments vary considerably between different developmental regions and different countries, and further discussion of their use is beyond the scope of this article.
How can rotavirus gastroenteritis be prevented?
Because rotavirus infects nearly all children in both industrialised and developing countries early in life, good hygiene and sanitation alone are considered inadequate for prevention. Observational studies have shown that breast feeding confers protection from rotavirus gastroenteritis, although one case-control study indicated that it may only postpone the occurrence of rotavirus gastroenteritis to the post-weaning period.46
Follow-up of birth cohorts indicates that, although children can be infected with rotavirus four to five times in the first two years of life, the incidence of severe rotavirus gastroenteritis is reduced with each repeat infection.6,7 Therefore, orally administered, live, attenuated rotavirus vaccines have been developed to mimic the effect of natural infection and prevent severe rotavirus disease.
A rhesus-human reassortant rotavirus vaccine (Rotashield, Wyeth) was licensed in the US in 1998 after showing high efficacy against severe rotavirus gastroenteritis in randomized clinical trials.47 Rotashield was recommended for routine immunization of US infants the same year but was abruptly withdrawn a year later because post-licensure observational studies found that it was associated with a severe adverse event, intussusception.48,49 Intussusception is a form of bowel obstruction that often requires surgery and is associated with high fatality if not treated. It was estimated that vaccination of 10 000 infants with Rotashield resulted in one excess case of intussusception.
Four point modified Gorelick score33,34.
1 point for each of the signs listed below:
Ill general appearance
Absent tears
Dry mucous membranes
Capillary refill >2 s
≤1 points: maintain hydration (<5% dehydration).
2 points: needs oral rehydration (5–10% dehydration).
3–4 points with normal vital signs: needs intravenous rehydration (>10% dehydration).
Abnormal vital signs (increased heart rate, decreased blood pressure, decreased level of consciousness, increased capillary refill time): needs resuscitation.
Two other live oral rotavirus vaccines—a pentavalent bovine-human reassortant vaccine (RotaTeq, Merck) and a monovalent human vaccine (Rotarix, GSK Biologicals)—were in advanced stages of clinical testing when Rotashield was withdrawn (table 3). RotaTeq and Rotarix were each tested in randomized clinical trials of 60 000–70 000 infants to assess the risk of intussusception before licensure.50,51 No increase in risk was found during the 42 and 30 days post-vaccination after the three doses of RotaTeq or two doses of Rotarix, respectively. The vaccines showed 85–98% efficacy against severe rotavirus gastroenteritis in these trials conducted in the Americas and Europe, with good protection against disease caused by rotavirus strains not included in the vaccines (heterotypic immunity). These findings supported vaccine licensure and recommendations for use by policy groups in the US and Europe and by WHO.52
Table 3.
Features of Rotarix and RotaTeq rotavirus vaccines
| Feature | Rotarix | RotaTeq |
|---|---|---|
| Composition | Single human rotavirus strain (P1A[8], G1) | Five human G/P reassortants with bovine rotavirus strain WC3 (P7[5], G6): G1 × WC3; G2 × WC3; G3 × WC3; G3 × WC3; P1A[8] × WC3 |
| Number of doses needed | 2 oral doses | 3 oral doses |
| Schedule* | Dose 1: minimum 6 weeks of age; dose 2: ≥4 weeks later; complete by 24 weeks of age | Dose 1: 6–12 weeks of age; doses 2 and 3: 4–10 week intervals; complete by 32 weeks of age |
| Dose | Each dose (1–1.5 mL) contains at least 106 median cell culture infectious doses | Each dose (2 mL) contains at least 2.0–2.8×106 infectious units per reassortant |
| Shelf life | 36 months | 24 months |
| Storage | 2–8°C, protected from light | 2–8°C, protected from light |
| Contraindications | History of hypersensitivity to the vaccine or any component of the vaccine; history of uncorrected congenital malformation of the gastrointestinal tract that would predispose to intussusception; history of severe combined immunodeficiency disease; history of intussusception | History of hypersensitivity to the vaccine or any component of the vaccine; history of severe combined immunodeficiency disease; history of intussusception |
Ages for vaccine doses vary according to the recommendations of individual countries and vaccination schedules.
As of December 2013, 51 countries include rotavirus vaccines in their national immunization programs. A systematic review of ecologic studies from eight countries reported a 49–89% decline in laboratory confirmed rotavirus hospital admissions and 17–55% reduction in all cause hospital admissions for gastroenteritis in children under 5 years within two years of vaccine introduction.53 Unexpectedly, rotavirus vaccination of young infants has also resulted in decreases in rotavirus disease in children who missed vaccination, older children, and even adults who were not eligible for vaccination.54 This phenomenon, known as herd protection, is probably related to reduced transmission of rotavirus in the community as a result of vaccination. Reductions in nosocomial rotavirus infections have also been seen since the introduction of the vaccine.55 Lastly, ecologic studies from Mexico and Brazil have shown a 35% and 22% decline in childhood deaths from diarrhea, respectively, since the introduction of the vaccine,56,57 and these reductions have been sustained for four years in Mexico. These findings are particularly noteworthy because vaccine efficacy against death from diarrhea was not evaluated in pre-licensure trials.
Post-licensure observational studies in several countries, including the US, Australia, Mexico, and Brazil, have also identified a low risk of intussusception with both rotavirus vaccines.58–65 The evidence of risk for the two vaccines is difficult to compare directly because the study populations and designs differed. In general, the overall risk is one to five excess cases of intussusception per 100 000 vaccinated infants. Considering the substantial and well documented health benefits of vaccination against this low intussusception risk, policy makers in countries with documented risk, as well as global health authorities such as WHO, strongly support rotavirus vaccination of infants.
Live oral vaccines against many diseases, such as polio, typhoid, and cholera, have performed less well in developing countries than in industrialised ones. The reasons for this variability are not completely understood. Potential explanations include interference in vaccine uptake by greater levels of maternal antibody or concurrent enteric infections in developing countries, as well as reduced immune response in infants because of comorbidities or malnutrition, including micronutrient deficiencies.66
Because of these concerns, randomised efficacy trials of both RotaTeq and Rotarix were conducted in developing countries in Africa and Asia.67–69 These trials showed modest vaccine efficacy (50–64%) against severe rotavirus gastroenteritis. Despite this reduced efficacy, the public health benefits of vaccination in terms of number of severe rotavirus gastroenteritis episodes prevented per 100 infants was greater in developing than in industrialised countries. This is because of the substantially greater rate of severe rotavirus gastroenteritis in developing countries. These considerations led WHO to issue a global recommendation for vaccination in 2009 and have prompted several low income countries to include rotavirus vaccination in their immunization programs.
As rotavirus vaccines are introduced into immunization programs of low income countries globally, it will be important to assess the real world impact of vaccination to gain a better understanding of vaccine effectiveness and safety in a range of settings. Although both of the licensed rotavirus vaccines have shown good protection against a range of circulating rotavirus strains, including strains with either or both G and P types not contained in the vaccine, further monitoring of the long term impact of vaccination on strain ecology is vital. Also, given the moderate efficacy of rotavirus vaccines in low income countries, interventions to improve vaccine performance (such as additional vaccine doses or different vaccination schedules) should be considered and evaluated. Finally, to sustain global implementation of vaccination, an adequate supply of affordable rotavirus vaccines must be assured. It is therefore encouraging that several manufacturers in emerging markets, including India, China, Indonesia, and Brazil, are developing candidate rotavirus vaccines that may be available within the next five years.
Areas for future research.
Exploring the role of rotavirus in extraintestinal clinical syndromes (such as neurologic manifestations, necrotizing enterocolitis)
Investigating the value of clinical signs and symptoms in assessing dehydration, particularly in general practice
Defining the optimal dose of oral rehydration solution for each grade of dehydration, so that consistent recommendations can be made
Identifying treatments (including antiviral agents) with clinical efficacy against rotavirus gastroenteritis
Community based action research and sociocultural research on knowledge, attitudes, perceptions, cultural practices, and health seeking behaviours with regard to rotavirus infection
Understanding the benefit-risk profile of routine rotavirus vaccination in a diverse range of geographic and socioeconomic settings
Investigating the effectiveness and impact of rotavirus vaccination in low income settings
Identifying interventions (such as additional vaccine doses or alternate schedules, supplementation with micronutrients) or alternative approaches (such as parenteral vaccines) that might improve the performance of rotavirus vaccines in low income settings
Additional educational resources.
Resources for healthcare professionals
Centers for Disease Control and Prevention. Prevention of rotavirus gastroenteritis among infants and children—recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2009;58(RR02):1–25. www.cdc.gov/mmwr/preview/mmwrhtml/rr5802a1.htm (free access)
WHO. WHO position paper on rotavirus vaccines. Wkly Epidemiol Rec 2013;88:49–64. www.who.int/wer/2013/wer8805.pdf (free access)
Soares-Weiser K, MacLehose H, Bergman H, Ben-Aharon I, Nagpal S, Goldberg E, et al. Vaccines for preventing rotavirus diarrhoea: vaccines in use. Cochrane Database Syst Rev 2012;11:CD008521 (subscription required)
PATH (http://sites.path.org/rotavirusvaccine/rotavirus-advocacy-and-communications-toolkit/)—Free rotavirus disease and vaccine resources
PATH (http://sites.path.org/rotavirusvaccine/current-issue-of-rotaflash-jpg/)—Breaking news and updates on rotavirus vaccines
Resources for patients and parents
Centers for Disease Control and Prevention at (www.cdc.gov/rotavirus/index.html)—Basic information on the symptoms, treatment, and prevention of rotavirus gastroenteritis
Tips for non-specialists.
All children are infected with rotavirus in the first 3–5 years of life, regardless of hygiene and sanitation conditions
Vaccines are therefore the most effective way to prevent rotavirus diarrhea
Treatment of rotavirus diarrhea relies on hydration therapy
Rotavirus vaccines have performed well in countries where they are used routinely; in some settings, they have conferred additional benefits to unvaccinated children and adults through herd protection
Although rotavirus vaccines work less well in developing countries, the potential to prevent severe disease and deaths is greater because of the high disease burden in these settings
The documented health benefits of rotavirus vaccines far outweigh the small risk of intussusception that has been seen in some settings
Footnotes
Provenance and peer review: Commissioned; externally peer reviewed.
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention (CDC).
Contributors: UDP searched the literature and is guarantor. UDP, EASN, and GK each wrote different sections of the manuscript and revised the entire draft of the manuscript.
Competing interests: We have read and understood the BMJ Group policy on declaration of interests and declare the following interests: UDP and GK—none. EASN has participated in vaccine and disease surveillance studies funded by GlaxoSmithKline and Pfizer.
References
- 1.Parashar UD, Hummelman EG, Bresee JS, Miller MA, Glass RI. Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis. 2003;9:565–72. doi: 10.3201/eid0905.020562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tate JE, Burton AH, Boschi-Pinto C, Steele AD, Duque J, Parashar UD, et al. 2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: a systematic review and meta-analysis. Lancet Infect Dis. 2012;12:136–41. doi: 10.1016/S1473-3099(11)70253-5. [DOI] [PubMed] [Google Scholar]
- 3.Lanata CF, Fischer-Walker CL, Olascoaga AC, Torres CX, Aryee MJ, Black RE, et al. Global causes of diarrheal disease mortality in children <5 years of age: a systematic review. PLoS One. 2013;8:e72788. doi: 10.1371/journal.pone.0072788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dickey M, Jamison L, Michaud L, Care M, Bernstein DI, Staat MA. Rotavirus meningoencephalitis in a previously healthy child and a review of the literature. Pediatr Infect Dis J. 2009;28:318–21. doi: 10.1097/INF.0b013e31818ddbe9. [DOI] [PubMed] [Google Scholar]
- 5.Le Saux N, Bettinger JA, Halperin SA, Vaudry W, Scheifele DW. Substantial morbidity for hospitalized children with community-acquired rotavirus infections: 2005–2007 IMPACT surveillance in Canadian hospitals. Pediatr Infect Dis J. 2010;29:879–82. doi: 10.1097/INF.0b013e3181e20c94. [DOI] [PubMed] [Google Scholar]
- 6.Velazquez FR, Matson DO, Calva JJ, Guerrero L, Morrow AL, Carter-Campbell S, et al. Rotavirus infections in infants as protection against subsequent infections. N Engl J Med. 1996;335:1022–8. doi: 10.1056/NEJM199610033351404. [DOI] [PubMed] [Google Scholar]
- 7.Gladstone BP, Ramani S, Mukhopadhya I, Muliyil J, Sarkar R, Rehman AM, et al. Protective effect of natural rotavirus infection in an Indian birth cohort. N Engl J Med. 2011;365:337–46. doi: 10.1056/NEJMoa1006261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anderson EJ, Weber SG. Rotavirus infection in adults. Lancet Infect Dis. 2004;4:91–9. doi: 10.1016/S1473-3099(04)00928-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Newman RD, Grupp-Phelan J, Shay DK, Davis RL. Perinatal risk factors for infant hospitalization with viral gastroenteritis. Pediatrics. 1999;103:E3. doi: 10.1542/peds.103.1.e3. [DOI] [PubMed] [Google Scholar]
- 10.Dennehy PH, Cortese MM, Bégué RE, Jaeger JL, Roberts NE, Zhang R, et al. A case-control study to determine risk factors for hospitalization for rotavirus gastroenteritis in US children. Pediatr Infect Dis J. 2006;25:1123–31. doi: 10.1097/01.inf.0000243777.01375.5b. [DOI] [PubMed] [Google Scholar]
- 11.Patel MM, Pitzer VE, Alonso WJ, Vera D, Lopman B, Tate J, et al. Global seasonality of rotavirus disease. Pediatr Infect Dis J. 2013;32:e134–47. doi: 10.1097/INF.0b013e31827d3b68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ward RL, Bernstein DI, Young EC, Sherwood JR, Knowlton DR, Schiff GM. Human rotavirus studies in volunteers: determination of infectious dose and serological response to infection. J Infect Dis. 1986;154:871–80. doi: 10.1093/infdis/154.5.871. [DOI] [PubMed] [Google Scholar]
- 13.Greenberg HB, Estes MK. Rotaviruses: from pathogenesis to vaccination. Gastroenterology. 2009;136:1939–51. doi: 10.1053/j.gastro.2009.02.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Richardson S, Grimwood K, Gorrell R, Palombo E, Barnes G, Bishop R. Extended excretion of rotavirus after severe diarrhoea in young children. Lancet. 1998;351:1844–8. doi: 10.1016/S0140-6736(97)11257-0. [DOI] [PubMed] [Google Scholar]
- 15.Boshuizen JA, Reimerink JH, Korteland-van Male AM, van Ham VJ, Koopmans MP, Büller HA, et al. Changes in small intestinal homeostasis, morphology, and gene expression during rotavirus infection of infant mice. J Virol. 2003;77:13005–16. doi: 10.1128/JVI.77.24.13005-13016.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Blutt SE, Kirkwood CD, Parreño V, Warfield KL, Ciarlet M, Estes MK, et al. Rotavirus antigenaemia and viraemia: a common event? Lancet. 2003;362:1445–9. doi: 10.1016/S0140-6736(03)14687-9. [DOI] [PubMed] [Google Scholar]
- 17.Kang G, Iturriza-Gomara M, Wheeler JG, Crystal P, Monica B, Ramani S, et al. Quantitation of group A rotavirus by real-time reverse-transcription-polymerase chain reaction: correlation with clinical severity in children in South India. J Med Virol. 2004;73:118–22. doi: 10.1002/jmv.20053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Herring AJ, Inglis NF, Ojeh CK, Snodgrass DR, Menzies JD. Rapid diagnosis of rotavirus infection by direct detection of viral nucleic acid in silver-stained polyacrylamide gels. J Clin Microbiol. 1982;16:473–7. doi: 10.1128/jcm.16.3.473-477.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thomas EE, Puterman ML, Kawano E, Curran M. Evaluation of seven immunoassays for detection of rotavirus in pediatric stool samples. J Clin Microbiol. 1988;26:1189–93. doi: 10.1128/jcm.26.6.1189-1193.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iturriza Gómara M, Kang G, Gray JJ. Rotavirus genotyping: keeping up with an evolving population of human rotaviruses. J Clin Virol. 2004;31:259–65. doi: 10.1016/j.jcv.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 21.Lazzerini M, Ronfani L. Oral zinc for treating diarrhoea in children. Cochrane Database Syst Rev. 2013;1:CD005436. doi: 10.1002/14651858.CD005436.pub4. [DOI] [PubMed] [Google Scholar]
- 22.Gregorio GV, Dans LF, Silvestre MA. Early versus delayed refeeding for children with acute diarrhoea. Cochrane Database Syst Rev. 2011;7:CD007296. doi: 10.1002/14651858.CD007296.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fedorowicz Z, Jagannath VA, Carter B. Antiemetics for reducing vomiting related to acute gastroenteritis in children and adolescents. Cochrane Database Syst Rev. 2011;9:CD005506. doi: 10.1002/14651858.CD005506.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Allen SJ, Martinez EG, Gregorio GV, Dans LF. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst Rev. 2010;11:CD003048. doi: 10.1002/14651858.CD003048.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gregorio GV, Gonzales ML, Dans LF, Martinez EG. Polymer-based oral rehydration solution for treating acute watery diarrhoea. Cochrane Database Syst Rev. 2009;2:CD006519. doi: 10.1002/14651858.CD006519.pub2. [DOI] [PubMed] [Google Scholar]
- 26.Guideline Development Group. Diarrhoea and vomiting caused by gastroenteritis: diagnosis, assessment and management in children younger than 5 years. National Collaborating Centre for Women’s and Childrens Health; 2009. www.nice.org.uk/nicemedia/pdf/CG84FullGuideline.pdf. [PubMed] [Google Scholar]
- 27.Hartling L, Bellemare S, Wiebe N, Russell K, Klassen TP, Craig W. Oral versus intravenous rehydration for treating dehydration due to gastroenteritis in children. Cochrane Database Syst Rev. 2006;3:CD004390. doi: 10.1002/14651858.CD004390.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hahn S, Kim S, Garner P. Reduced osmolarity oral rehydration solution for treating dehydration caused by acute diarrhoea in children. Cochrane Database Syst Rev. 2002;1:CD002847. doi: 10.1002/14651858.CD002847. [DOI] [PubMed] [Google Scholar]
- 29.Nelson EA, Ko WK, Kwan E, Leung SF, Poon KH, Chow CB, et al. Guidelines for the management of acute diarrhoea in young children. Hong Kong J Paediatr. 2003;8:203–36. [Google Scholar]
- 30.Armon K, Stephenson T, MacFaul R, Eccleston P, Werneke U. An evidence and consensus based guideline for acute diarrhoea management. Arch Dis Child. 2001;85:132–42. doi: 10.1136/adc.85.2.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Murphy MS. Guidelines for managing acute gastroenteritis based on a systematic review of published research. Arch Dis Child. 1998;79:279–84. doi: 10.1136/adc.79.3.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Goldman RD, Friedman JN, Parkin PC. Validation of the clinical dehydration scale for children with acute gastroenteritis. Pediatrics. 2008;122:545–9. doi: 10.1542/peds.2007-3141. [DOI] [PubMed] [Google Scholar]
- 33.Gorelick MH, Shaw KN, Murphy KO. Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics. 1997;99:E6. doi: 10.1542/peds.99.5.e6. [DOI] [PubMed] [Google Scholar]
- 34.Alberta Health Service. Alberta Health Services acute childhood vomiting and diarrhea pathway. 2011 http://pert.ucalgary.ca/pathways/reference/Revised%20ED-UCC%20pathway%20Oct%2025%202011.pdf.
- 35.Lewis KDC, Dallas MJ, Victor JC, Ciarlet M, Mast TC, Ji M, et al. Comparison of two clinical severity scoring systems in two multi-center, developing country rotavirus vaccine trials in Africa and Asia. Vaccine. 2012;30(suppl 1):A159–66. doi: 10.1016/j.vaccine.2011.07.126. [DOI] [PubMed] [Google Scholar]
- 36.Freedman SB, Eltorky M, Gorelick M Pediatric Emergency Research Canada Gastroenteritis Study Group. Evaluation of a gastroenteritis severity score for use in outpatient settings. Pediatrics. 2010;125:e1278–85. doi: 10.1542/peds.2009-3270. [DOI] [PubMed] [Google Scholar]
- 37.Van den Berg J, Berger MY. Guidelines on acute gastroenteritis in children: a critical appraisal of their quality and applicability in primary care. BMC Fam Pract. 2011;12:134. doi: 10.1186/1471-2296-12-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lo Vecchio A, Giannattasio A, Duggan C, De Masi S, Ortisi MT, Parola L, et al. Evaluation of the quality of guidelines for acute gastroenteritis in children with the AGREE instrument. J Pediatr Gastroenterol Nutr. 2011;52:183–9. doi: 10.1097/MPG.0b013e3181e233ac. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pringle K, Shah SP, Umulisa I, Munyaneza RBM, Dushimiyimana JM, Stegmann K, et al. Comparing the accuracy of the three popular clinical dehydration scales in children with diarrhea. Int J Emerg Med. 2011;4:58. doi: 10.1186/1865-1380-4-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.WHO. The treatment of diarrhoea: a manual for physicians and other senior health workers. (4) 2005 http://whqlibdoc.who.int/publications/2005/9241593180.pdf.
- 41.Lawson AL. Acute gastroenteritis and dehydration (child) Li STT, Ebell MH, editors. Essential Evidence. 2012 www.essentialevidenceplus.com/content/eee?class=GI.
- 42.Dalby-Payne J, Elliott E. Gastroenteritis in children. Clin Evid. 2011;07:314. [Google Scholar]
- 43.Rossignol JF, Abu-Zekry M, Hussein A, Santoro MG. Effect of nitazoxanide for treatment of severe rotavirus diarrhoea: randomised double-blind placebo-controlled trial. Lancet. 2006;368:124–9. doi: 10.1016/S0140-6736(06)68852-1. [DOI] [PubMed] [Google Scholar]
- 44.Sarker SA, Jäkel M, Sultana S, Alam NH, Bardhan PK, Chisti MJ, et al. Anti-rotavirus protein reduces stool output in infants with diarrhea: a randomized placebo-controlled trial. Gastroenterology. 2013;145:740–8. e8. doi: 10.1053/j.gastro.2013.06.053. [DOI] [PubMed] [Google Scholar]
- 45.Sarker SA, Casswall TH, Juneja LR, Hoq E, Hossain I, Fuchs GJ, et al. Randomized, placebo-controlled, clinical trial of hyperimmunized chicken egg yolk immunoglobulin in children with rotavirus diarrhea. J Pediatr Gastroenterol Nutr. 2001;32:19–25. doi: 10.1097/00005176-200101000-00009. [DOI] [PubMed] [Google Scholar]
- 46.Clemens J, Rao M, Ahmed F, Ward R, Huda S, Chakraborty J, et al. Breast-feeding and the risk of life-threatening rotavirus diarrhea: prevention or postponement? Pediatrics. 1993;92:680–5. [PubMed] [Google Scholar]
- 47.Centers for Disease Control and Prevention. Rotavirus vaccine for the prevention of rotavirus gastroenteritis among children. Morb Mortal Wkly Rep. 1999;48(RR-2) [Google Scholar]
- 48.Centers for Disease Control and Prevention. Intussusception among recipients of rotavirus vaccine—United States, 1998–1999. Morb Mortal Wkly Rep. 1999;48:577–81. [PubMed] [Google Scholar]
- 49.Murphy TV, Gargiullo PM, Massoudi MS, Nelson DB, Jumaan AO, Okoro CA, et al. Intussusception among infants given an oral rotavirus vaccine. N Engl J Med. 2001;344:564–72. doi: 10.1056/NEJM200102223440804. [DOI] [PubMed] [Google Scholar]
- 50.Ruiz-Palacios GM, Perez-Schael I, Velazquez FR, Abate H, Breuer T, Clemens SC, et al. Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med. 2006;354:11–22. doi: 10.1056/NEJMoa052434. [DOI] [PubMed] [Google Scholar]
- 51.Vesikari T, Matson DO, Dennehy P, Van Damme P, Santosham M, Rodriguez Z, et al. Safety and efficacy of a pentavalent human-bovine (WC3) reassortant rotavirus vaccine. N Engl J Med. 2006;354:23–33. doi: 10.1056/NEJMoa052664. [DOI] [PubMed] [Google Scholar]
- 52.WHO. Meeting of the strategic advisory group of experts on immunization, October 2009—conclusions and recommendations. Wkly Epidemiol Rec. 2009;84:518. [PubMed] [Google Scholar]
- 53.Patel MM, Glass R, Desai R, Tate JE, Parashar UD. Fulfilling the promise of rotavirus vaccines: how far have we come since licensure? Lancet Infect Dis. 2012;12:561–70. doi: 10.1016/S1473-3099(12)70029-4. [DOI] [PubMed] [Google Scholar]
- 54.Gastañaduy PA, Curns AT, Parashar UD, Lopman BA. Gastroenteritis hospitalizations in older children and adults in the United States before and after implementation of infant rotavirus vaccination. JAMA. 2013;310:851–3. doi: 10.1001/jama.2013.170800. [DOI] [PubMed] [Google Scholar]
- 55.Zlamy M, Kofler S, Orth D, Würzner R, Heinz-Erian P, Streng A, et al. The impact of rotavirus mass vaccination on hospitalization rates, nosocomial rotavirus gastroenteritis and secondary blood stream infections. BMC Infect Dis. 2013;13:112. doi: 10.1186/1471-2334-13-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.De Carmo GM, Yen C, Cortes J, Siqueira AA, de Oliveira WK, Cortez-Escalante JJ, et al. Decline in diarrhea mortality and admissions after routine childhood rotavirus immunization in Brazil: a time-series analysis. PLoS Med. 2011;8:e1001024. doi: 10.1371/journal.pmed.1001024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Richardson V, Hernandez-Pichardo J, Quintanar-Solares M, Esparza-Aguilar M, Johnson B, Gomez-Altamirano CM, et al. Effect of rotavirus vaccination on death from childhood diarrhea in Mexico. N Engl J Med. 2010;362:299–305. doi: 10.1056/NEJMoa0905211. [DOI] [PubMed] [Google Scholar]
- 58.Carlin JB, Macartney K, Lee KJ, Quinn HE, Buttery J, Lopert R, et al. Intussusception risk and disease prevention associated with rotavirus vaccines in Australia’s national immunisation program. Clin Infect Dis. 2013 doi: 10.1093/cid/cit520. published online 30 Aug. [DOI] [PubMed] [Google Scholar]
- 59.Buttery JP, Danchin MH, Lee KJ, Carlin JB, McIntyre PB, Elliott EJ, et al. Intussusception following rotavirus vaccine administration: post-marketing surveillance in the National Immunization Program in Australia. Vaccine. 2011;29:3061–6. doi: 10.1016/j.vaccine.2011.01.088. [DOI] [PubMed] [Google Scholar]
- 60.Patel M, López-Collada V, Bulhões M, De Oliveira LH, Bautista Márquez A, Flannery B, et al. Intussusception risk and health benefits of rotavirus vaccination in Mexico and Brazil. N Engl J Med. 2011;364:2283–92. doi: 10.1056/NEJMoa1012952. [DOI] [PubMed] [Google Scholar]
- 61.Velazquez FR, Colindres RE, Grajales C, Hernández MT, Mercadillo MG, Torres FJ, et al. Postmarketing surveillance of intussusception following mass introduction of the attenuated human rotavirus vaccine in Mexico. Pediatr Infect Dis J. 2012;31:736–44. doi: 10.1097/INF.0b013e318253add3. [DOI] [PubMed] [Google Scholar]
- 62.Haber P, Patel M, Pan Y, Baggs J, Haber M, Museru O, et al. Intussusception after rotavirus vaccines reported to US VAERS, 2006–2012. Pediatrics. 2013;131:1042–9. doi: 10.1542/peds.2012-2554. [DOI] [PubMed] [Google Scholar]
- 63.Shui I, Baggs J, Patel M, Parashar UD, Rett M, Belongia EA, et al. Risk of intussusception following administration of a pentavalent rotavirus vaccine in US Infants. JAMA. 2012;307:598–604. doi: 10.1001/jama.2012.97. [DOI] [PubMed] [Google Scholar]
- 64.Cortese M. Estimates of benefits and potential risks of rotavirus vaccination in the US presented at the Advisory Committee on Immunization Practices meeting; Atlanta, GA, USA. 20 June 2013; www.cdc.gov/vaccines/acip/meetings/downloads/slides-jun-2013/06-Rotavirus-Cortese.pdf. [Google Scholar]
- 65.Food and Drug Administration. Final study results of a mini-sentinel postlicensure observational study of rotavirus vaccines and intussusception. 2013 www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/ucm356758.htm.
- 66.Glass RI, Parashar UD, Bresee JS, Turcios R, Fischer TK, Widdowson MA, et al. Rotavirus vaccines: current prospects and future challenges. Lancet. 2006;368:323–32. doi: 10.1016/S0140-6736(06)68815-6. [DOI] [PubMed] [Google Scholar]
- 67.Armah GE, Sow SO, Breiman RF, Dallas MJ, Tapia MD, Feikin DR, et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-Saharan Africa: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;376:606–14. doi: 10.1016/S0140-6736(10)60889-6. [DOI] [PubMed] [Google Scholar]
- 68.Zaman K, Dang DA, Victor JC, Shin S, Yunus M, Dallas MJ, et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in Asia: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;376:615–23. doi: 10.1016/S0140-6736(10)60755-6. [DOI] [PubMed] [Google Scholar]
- 69.Madhi SA, Cunliffe NA, Steele D, Witte D, Kirsten M, Louw C, et al. Effect of human rotavirus vaccine on severe diarrhea in African infants. N Engl J Med. 2010;362:289–98. doi: 10.1056/NEJMoa0904797. [DOI] [PubMed] [Google Scholar]


