Abstract
Objective:
The present study was designed to investigate the effects of α-lipoic acid on the abdominal aorta and hypertension in a remnant kidney model histomorphometrically, immunohistochemically, and ultrastructurally.
Methods:
We surgically reduced the renal tissue mass to 5/6 by applying a remnant kidney model. The rats were divided into 4 groups: Group 1 control group, Group 2- lipoic acid group, Group 3- 5/6 nephrectomy group, and Group IV: 5/6 nephrectomy+lipoic acid-treated group. Lipoic acid solution 100 mg/kg was administered by oral gavage for 8 weeks to Groups II and IV. At the end of the experiment, systemic mean blood pressure was monitored. Then, aortic tissues were removed and fixed. After routine histological procedures, tissue sections were examined histochemically, immunohistochemically (type I angiotensin receptor, vascular endothelial growth factor, alpha-smooth muscle actin), and ultrastructurally.
Results:
The blood pressure measurements in 5/6 nephrectomy group were significantly higher compared to other groups. In the 5/6 nephrectomy+lipoic acid group, measured blood pressure values and tunica media thickness were significantly lower than in the 5/6 nephrectomy group. In the 5/6 nephrectomy+lipoic acid group, decreased aortic wall thickness, regularity in the structure of elastic fibrils, and more organized elastic lamellae were seen. The expression of type I angiotensin receptor, vascular endothelial growth factor, alpha-smooth muscle actin in the 5/6 nephrectomy+lipoic acid group was decreased compared to the 5/6 nephrectomy group.
Conclusion:
In the present study, we found that α-lipoic acid could be a favorable agent for the target organ effects of secondary hypertension.
Keywords: α-lipoic acid, hypertension, remnant kidney model, aorta, vascular endothelial growth factor, type-I angiotensin receptor, alpha-smooth muscle actin
Introduction
Hypertension is the most common cardiovascular disease, with a high incidence and lethal complications (1). Several animal models have been developed to reveal the mechanisms involved in the pathogenesis of hypertension. In experimental hypertension, genetic phenotype-driven (eg, SHR) or genotype- driven (eg, null mice) models or nongenetic (surgical intervention, as the endocrine, diet modification, partial or subtotal nephrectomy) models can be formed. Surgically reducing renal tissue to five-sixths (5/6) by renal mass ablation (remnant kidney model), helps to create secondary hypertension (1-3).
Hypertension has an impact on target organs, such as the heart, kidneys, and blood vessels. The vascular system is the main target of hypertension. One of the target organs in the vascular system is the aorta. In the aorta, not only the intima but also major structures, like the vascular smooth muscle cell (VSMC), collagen, elastic fibrils, and proteoglycans in the matrix, are affected by hypertension (4). As a result of hypertension, arterial elasticity is changed, and vessel wall injury occurs (5). The biological actions of the renin-angiotensin system (RAS) are mediated by the angiotensin II (Ang II) molecule in the blood vessels. Ang II, the main biological effector of RAS, has an important role in vascular structure and in hemodynamics. Ang II has vasoconstrictor effects on vessels and acts through the type I angiotensin receptor (AT1), which causes changes in hypertension (4, 5). AT1 receptors may also stimulate growth and hypertrophy of VSMCs (4, 6). Ang II is shown to upregulate vascular endothelial growth factor (VEGF). VEGF is a member of the platelet-derived growth factor superfamily (PDGF) and one of the most potent angiogenic factors. VEGF is thought to function as an endogenous regulator of endothelial integrity and takes part in endothelial cell proliferation, migration, and differentiation (6, 7). VEGF receptors are also expressed on VSMCs; this indicates that VEGF may act as a regulating molecule for the response of VSMCs. Smooth muscle actin (SMA) is a microfila- mentous contractile protein. There are different isoforms of this protein in the actin protein family. The alpha (α-SMA) form is present in skeletal muscle, smooth muscle, heart muscle, fibroblasts, and myoepithelial cells. It plays an important role in fibro-genesis and correlates with the activation of myofibroblasts (7).
Lipoic acid (LA) is a potent antioxidant and a free radical scavenger that is endogenously produced. It is known to inhibit lipid peroxidation both in vitro and in vivo. Its antioxidant properties are related to removing free radicals and chelating metal ions while being reduced to dihydrolipoic acid (8, 9). LA lowers blood lipids and protects against LDL oxidation. LA is thought to be a possible protective agent against the risk factors of cardiovascular diseases (9). Studies on hypertension and the protective effect of LA showed that in hypertensive rats, LA lowered the blood pressure (3).
Besides antihypertensive agents, such as diuretics and angiotensin-converting enzyme (ACE) inhibitors, recently, the protective and/or therapeutic effects of antioxidants in hypertension have been examined. In this content, the aim of this study was to investigate the cellular and ultrastructural changes occurring in the abdominal aorta due to hypertension in a remnant kidney model and evaluate the effects of lipoic acid on ameliorating the morphology of the aorta.
Methods
The experiment was performed on 28 male Wistar albino rats (230-260 g) in accordance with the guidelines provided by the experimental animal laboratory. The study was approved by the ethic committee of the research of laboratory animals, and all procedures were performed according to the “Principles of Laboratory Animal Care” by the National Institute of Health. The animals were maintained on a constant 12-h light/dark cycle at constant room temperature (23±2°C) and humidity (60%), and food and tap water were provided ad libitum throughout the experiments.
Experimental groups
The rats were divided into 4 group: Group 1- control (8 week), Group 2- lipoic acid (LA group; rats administered LA until being sacrificed at Week 8), Group 3- 5/6 nephrectomy (Nx) group (8 week), and Group 4- 5/6 nephrectomy+lipoic acid-treated (8 week) (5/6 Nx+LAT) group.
The constitution of the experimental 5/6 Nx model
This model was performed by right nephrectomy followed by partial infarction of approximately two-thirds of the left kidney by selective ligation of two to three of three to four extrarenal branches of the left renal artery (3, 10).
Lipoic acid administration
Lipoic acid (Sigma, St Louis, USA) 100 mg/kg was mixed with sterile saline in a dark bottle, and 1 M NaOH was added until the suspension dissolved. The pH was then brought to 7.4 with 1 M HCl. Fresh LA solutions were administered by oral gavage for 8 weeks (3).
Systolic blood pressure measurements
The animals were anesthetized by an intraperitoneal dose of urethane 1.5 g/kg. A tracheal cannula was inserted via tracheotomy and sutured. A catheter was inserted into the right carotid artery to monitor systemic mean blood pressure with an MP30 BPT300 pressure transducer connected to a computerized system for data acquisition (MP Biopac system Inc., California, USA). The arterial catheter was filled with heparinized saline (250 U/mL). Blood samples were measured (3, 11).
Light microscopic examination
At the end of the experiment, abdominal aorta tissues were removed and fixed in 10% formalin. After routine histological procedures, specimens were blocked, and 4-μm sections were taken with a rotary microtome (RM 2255, Leica Instruments, Germany). The hematoxylin and eosin (H&E)-stained sections were used for evaluating general histomorphology; Masson's trichrome (MTKR) stained (GBL cat no:5022) sections were used for connective tissue and collagen content, and orceinlight green-stained (Merck, Millipore Cat no:107100) sections were used for elastic fibril structure. The images obtained from the selected areas were analyzed by using a computer-assisted image analyzer system consisting of a microscope (Olympus BX-51, Japan) equipped with a high-resolution video camera (Olympus, DP70, Japan). At least four areas from each section were analyzed and evaluated for each rat. Measurements were made using the UTHSCA Image Tool (software version 3.0, University of Texas Health Science Center, USA).
Immunohistochemistry
After deparaffinization and rehydration, sections were treated with 10 mM citrate buffer (Cat No.AP-9003-125 Labvision). Then, sections were incubated in a solution of 3% H2O2 to inhibit endogenous peroxidase activity and then incubated with normal serum blocking solution. Next, sections were incubated in a humid chamber for 18 h at +4°C overnight with antibodies to AT1 (ab9391-1, Abcam), VEGF (sc-7269, Santa Cruz), and α-SMA (ab7817, Abcam) and then with biotinylated IgG and streptavidin, conjugated to horseradish peroxidase, prepared according to kit instructions (Invitrogen, Histostain-Plus Kit Broad Spectrum, 85-9043). Finally, the sections were stained with 3, 3’-diamino-benzidine (DAB) substrate (1718096, Roche), counter-stained with Mayer's hematoxylin, and analyzed using a BH-2 microscope (Olympus, Tokyo, Japan).
Semi-quantification of immunostaining data
A grading system was used to score the quantity of immunostaining as follows: 1, very little positive staining was observed in an image and the staining was mild; 2, positive staining was moderate and between grade 1 and grade 3; 3, strong positive staining was evenly distributed in the whole image; and 0, no immunoreactivity. To maintain the consistency of scoring, each section was graded by two persons blinded to the treatments, and the average was taken. The average of the scores was used to represent the grade of immunostaining for each aorta section (12).
Electron microscopic examination
Tissue samples for EM were fixed in 2.5% glutaraldehyde. The specimens were washed in phosphate buffer and subsequently submerged in osmium tetroxide. This procedure provided both staining and fixation. Then, they were passed through a series of graded alcohol solutions and left in propylene oxide for final fixation. Specimens were embedded in Araldite-CY 212 and dodecenyl succinic anhydride (DDSA) for one night. Then, the specimens were placed in a mixture of Araldite-CY 212, DDSA and benzyl dimethylamine (BDMD) and incubated in an autoclave. The tissues were left in it to cool so that polymerization was attained. The specimen lumps were cut into sections with a Reicheld LK (Vien, Austria) ultra-microtome and stained with toluidine blue. The marked sections were cut into thin slices and were stained with uranyl acetate and then with lead citrate. Finally, the tissue samples were examined on a Libra120 (Carl- Zeiss, Germany) transmission electron microscope.
Statistical analysis
Initial and final body weight, blood pressure, tunica media thickness values, and numeric values belonging to the correlation between the blood pressure measurements and tunica media thickness were analyzed with Kolmogorov-Smirnov test and tested for compliance for normal distribution. These values were not accordant with a normal distribution. That is why initial and final body weights were analyzed with nonparametric Wilcoxon signed-rank test. The blood pressure and tunica media thickness values were analyzed with no parametric Mann-Whitney U test, and the correlation was evaluated with Spearman correlation test. Nonparametric Mann-Whitney U test was used for semiquantitative immune scoring in the comparison of the two groups.
Initial and final body weights, blood pressure, and tunica media thickness values were numerical values, and they were expressed as the median±standard deviation. Semiquantitative immune scoring values were nominal values, and they were expressed as the mean±standard deviation. All statistical procedures were performed with SPSS software for Windows, Version 15.0 (SPSS, Chicago, IL, USA). A value of p<0.05 was considered significant.
Results
Body weight, blood pressure, and tunica media thickness (TMT)
The final body weights of rats in all groups were significantly increased (Table 1). At end of the experiment, blood pressure values were measured. The blood pressure measurements from the carotid artery in the 5/6Nx group were significantly higher compared to the control, LA, and 5/6Nx+LAT groups (p=0.002). In the 5/6Nx+LAT group, measured blood pressure values were significantly lower than in the 5/6Nx group (p=0.002) (Table 1).
Table 1.
Initial and final body weight (median±SD), blood pressure, and tunica media thickness in all groups
| Groups | Initial/final body weight (g) | Blood pressure (mm Hg) | Tunica media thickness (μm) |
|---|---|---|---|
| Control (C) | 241.0±4.52/251.0±6.87a | 100.0±2.2 | 122.3±3.3 |
| LA | 241.0±5.64/250.0±4.96a | 101.3±5.7 | 126.9±17.8 |
| 5/6 Nx | 245.0±6.91/254.0±3.59a | 144.3±6.5b | 181.0±11.1d |
| 5/6Nx+LAT | 246.0±5.25/256.0±4.49a | 127.0±9.5c | 138.0±11.2e |
P<0.05 compared to initial body weight (nonparametric Wilcoxon signed-rank test)
P<0.05 in 5/6 Nx group compared to other groups.
P<0.05 in 5/6 Nx+LAT group compared to 5/6 sNx group.
P<0.05 in 5/6 Nx group compared to other groups.
P<0.05 in 5/6 Nx+LAT group compared to 5/6 Nx group (nonparametric Mann-Whiney U test). LA- lipoic acid, 5/6 Nx- 5/6 nephrectomy, 5/6Nx+LAT- 5/6 nephrectomy+lipoic acid-treated group
In the 5/6Nx group, TMT was higher than in the control (p<0.001), LA (p=0.002), and 5/6Nx+LAT groups (p=0.002). In the 5/6Nx+LAT group, TMT was significantly lower than in the 5/6Nx group (p<0.001) (Table 1).
When we compare the blood pressures and TMT values of the groups, there was a significant positive correlation between groups (r=0.71; p<0.001) (Fig. 1) (Spearman correlation test).
Figure 1.
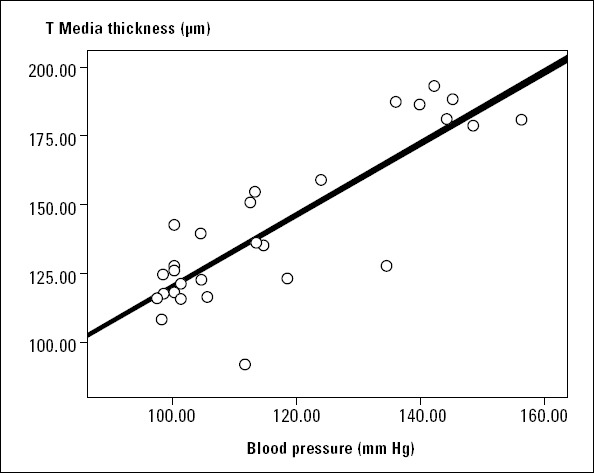
Blood pressure and tunica media thickness correlation
Histochemical and histomorphological evaluation
In the control group, the tunica intima, tunica media, and tunica adventitia of the aorta had a normal histomorphological structure in all stains (Fig. 2-1A-C). The LA group showed a nearly normal structure, like the control group (Fig. 2-2A-C). In the 5/6 Nx group, increased aortic wall thickness, irregularity in the structure of the elastic fibrils in the tunica media (arrows), and deterioration in the formation of elastic lamellae (arrows) were observed (Fig. 2-3A-C). When we observed the 5/6 Nx+LAT group, decreased aortic wall thickness, regularity in the structure of the fibrils in the tunica media, and more organized formation of elastic lamellae were seen (Fig.2-4A-C). When we compared the 5/6 Nx+LAT group with the 5/6 Nx group, the elastic fibril lamellae were organized and regular, and there was a decrease in the connective tissue content.
Figure 2.
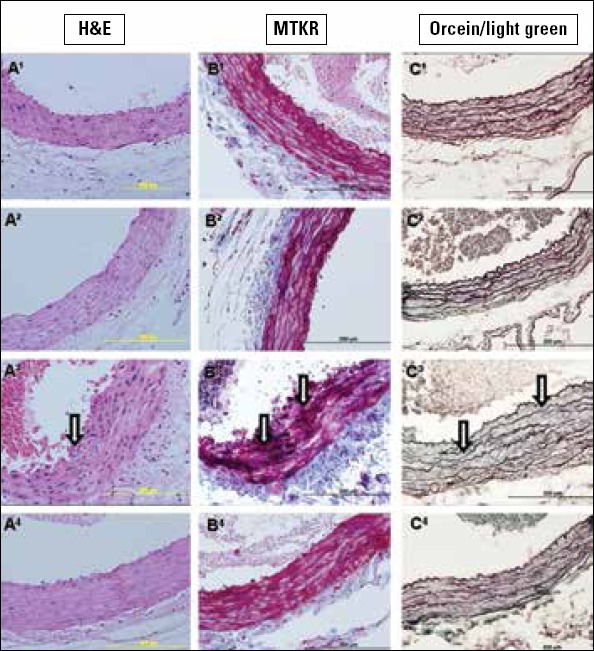
1A-C. Sections belonging to the control group, 2A-C. belonging to the LA group, 3A-C. belonging to the 5/6 Nx group, and 4A-C. belonging to the 5/6 Nx+LAT group. Arrows show irregular and increased collagen content and irregular elastic fibril bundles. A1-4. Stained with H&E, B1-4. stained with MTKR, and C1-4. sections stained with orcein/light green. Scale bar 200 μm
Immunohistochemistry
The expression of AT-1, VEGF, and α-SMA was determined by immunohistochemistry. Results of the semiquantitative scoring and statistical analysis details are given in Table 2. In the control (Fig. 3-1A-C) and LA groups (Fig. 3-2A-C), the tunica intima, media, and adventitia showed very little positive staining. In the 5/6 Nx group, the tunica media and tunica adventitia especially showed mild, even moderate, immunopositive staining of all three antibodies compared to the other groups (Fig. 3-3A-C). Immunopositive staining in the 5/6 Nx+LAT group was decreased of all three antibodies compared to the 5/6 Nx group (Fig. 3-4A-C).
Table 2.
Semiquantitative scoring of immunohistochemical staining
| Groups | T intima | T media | T adventitia | |
|---|---|---|---|---|
| AT-1 | Control | 0.71±0.48 | 0.57±0.53 | 0.85±0.37 |
| LA | 0.71±0.48 | 0.71±0.48 | 0.71±0.48 | |
| 5/6Nx | 1.14±0.37 | 2.14±0.37a, b | 2.00±0.00a,b | |
| 5/6Nx+LAT | 1.00±0.57 | 1.14±0.37c | 1.00±0.00c | |
| VEGF | Control | 0.85±0.37 | 1.14±0.37 | 1.0±0.00 |
| LA | 1.00±0.00 | 1.14±0.37 | 1.28±0.48 | |
| 5/6Nx | 1.14±0.37 | 2.00±0.57a, b | 2.14±0.37a, b | |
| 5/6Nx+LAT | 1.14±0.37 | 1.14±0.37c | 1.28±0.48c | |
| a-SMA | Control | 0.5±0.37 | 0.71±0.48 | 0.71±0.48 |
| LA | 1.00±0.00 | 0.85±0.37 | 0.85±0.37 | |
| 5/6Nx | 1.14±0.37 | 1.85±0.37a, b | 1.71±0.48a, b | |
| 5/6Nx+LAT | 1.00±0.00 | 1.14±0.37c | 1.14±0.69c |
Semiquantitative scoring of immunohistochemical staining of the groups.
Significantly increased vs. control and LA group.
AT-1; p=0.001, P<0.001, VEGF; p=0.01, P<0.001, α-SMA; P=0.002, P=0.006, respectively. (b): Significantly increased vs. LA group. AT-1; P=0.001, P=0.001, VEGF; P=0.01, P=0.007, α-SMA; P=0.002, P=0.007, respectively.
Significantly decreased vs. 5/6Nx group AT-1; P=0.002, P<0.001, VEGF; P=0.01, P=0.007. α-SMA; P=0.01, P=0.01 respectively (nonparametric Mann-Whiney U test).
AT1: type I angiotensin receptor, LA - lipoic acid, 5/6Nx-5/6 nephrectomy, 5/6Nx+LAT-5/6 nephrectomy+lipoic acid-treated, VEGF - vascular endothelial growth factor, α-SMA - alpha-smooth muscle actin
Figure 3.
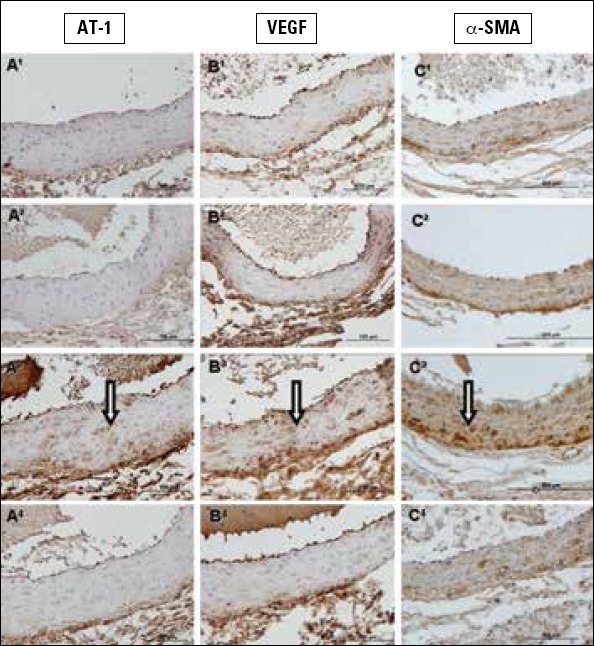
1A-C. Sections belonging to the control group, 2A-C. sections belonging to the LA group, 3A-C. sections belonging to the 5/6 Nx group, and 4A-C. 5/6 Nx + LAT groups. Arrows show immunopositive aorta tissue. A1-4. Stained with type I angiotensin (AT1) receptor, B1-4. stained with vascular endothelial growth factor, and C1-4. stained with alpha-smooth muscle actin immunohistochemically. Scale bar 200 μm
Electron microscopic evaluation
Electron microscopic examination showed that endothelial cells and nuclei were regular with no ultrastructural changes in the control group (Fig. 4-1A, 2A). The elastic membrane was beginning immediately under the basement of the endothelial cells and continued with muscle cells and elastic fibers. The LA group had a similar appearance to the control group's ultrastructure (Fig. 4-1B, 2B). In the 5/6Nx group, irregularity was observed in endothelial cells. Elastic bundles were disorganized (Fig. 4-2C), and subintimal thickening and collagen deposits were observed between endothelial cells and the membrana elastica interna (Fig. 4-1C, 2C). In the 5/6 Nx+LAT group, membrane elastic bundles were more organized, and endothelial cells were more regular when compared to the 5/6 Nx group. The deposits and the collagen between the endothelium decreased (Fig. 4-1D, 2D).
Figure 4.
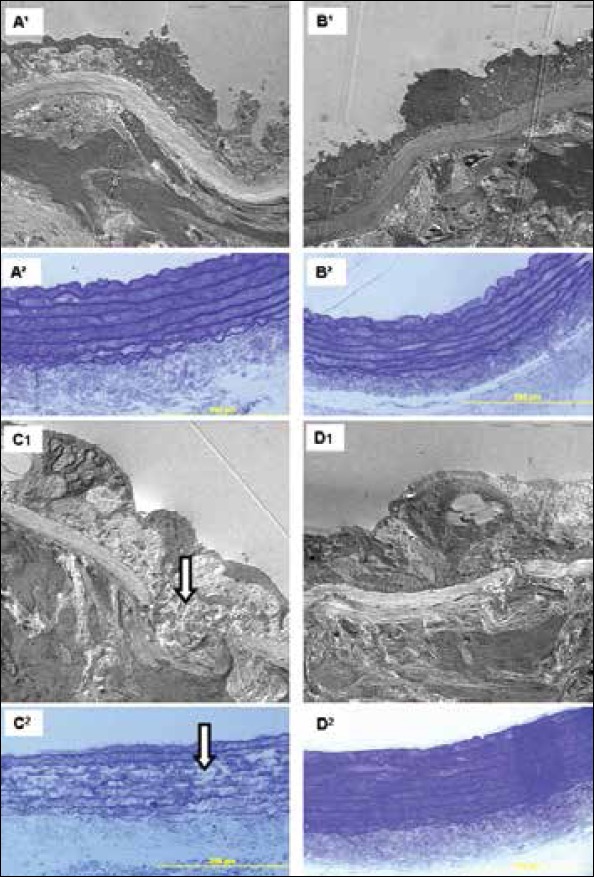
A1-2. Control group, B1-2. LA group, C1-2. 5/6 Nx group, and D1-2. 5/6 Nx+LAT. Arrows show irregularity in elastic bundles (C2). 1A-D. Electron microscopic micrographs, stained with lead citrateuranyl acetate (x 4000). 2A-D. Semithin sections stained with toluidine blue. Scale bar 200 μm
Discussion
The present study demonstrated that LA attenuates hypertension-induced abdominal aorta injuries in a model of rat rem nant kidney by histomorphological, immunohistological, and ultrastructural examinations.
Hypertension, together with renal insufficiency, is a common problem observed in patients with chronic renal failure. Experimental models of nephrectomy have been implemented as models that reflect chronic renal failure (1). Experimentally induced nephrectomy models result in increased blood pressure, and they are used to try to clarify the mechanisms of target organ damage similar to those in humans (13). A few days after the experimental 5/6 nephrectomy model, it is reported that there is an increase in blood pressure (1, 14). We surgically reduced renal tissue to five-sixths (5/6) by renal mass ablation (remnant kidney model) in order to create secondary hypertension (1). When secondary hypertension has settled, target organ damage starts. The aorta in the vascular system is one of the targets in secondary hypertension (15).
As a result of high blood pressure, arterial elasticity is reduced, and VSMCs and collagen fibrils are affected (5, 16, 17). In our study, we also found changes in aortic wall thickness, irregularity in the structure of the tunica media, and deterioration in the formation of elastic lamellae in the 5/6 Nx group. Besides, in the 5/6 Nx group, the collagen content and smooth muscle cells in the tunica media were increased. On the other hand, in the 5/6 Nx+LAT group showed less histomorphological degenerative changes (Fig. 2-4). These data confirmed that LA attenuates the target organ effects of hypertension in rat abdominal aorta. In our study, we evaluated the correlation between blood pressure and TMT. We found a positive correlation (r: 0.71) between them. As shown in another study, low antioxidant intake leads to severe ultrastructural changes in the vessel walls in hypertensive rats, whereas antioxidant supplementation slows down the elastin degradation and degenerative changes of the vessel walls (18). We observed an irregular and thick basal membrane and subendothelial deposits in the 5/6Nx group in our study. It was seen that ultrastructural changes in endothelial and elastic bundle regularity and thickness significantly decreased in the 5/6 Nx+LAT group (Fig. 3).
It has been well known that activation of the RAS plays an important role in the physiology and pathophysiology of hypertension. Ang II regulates blood pressure and electrolyte homeostasis and takes part in the inflammatory response in the vascular wall (12). Cardiovascular diseases induce complex structural and functional changes in the vasculature. Increased pressure precedes and elicits vascular damage (5). Activation of the RAS as a result of impaired endothelial function plays an important role in the initiation and progression of arterial damage. Ang II is implicated in the process of angiogenesis. Ang II is shown to upregulate VEGF. VEGF is one of the most potent angiogenic factors known to date and is thought to function as an endogenous regulator of endothelial integrity (6, 7). VEGF was discovered as a factor capable of inducing endothelial cell permeability and angiogenesis. So, VEGF-targeted therapies were introduced into clinical use for treatment of malignant diseases (19). It was noted that hypertension and proteinuria were major toxicities. Hypertension occurs in up to 80% of patients receiving VEGF- targeted therapy (20-22).
Previous animal studies have reported that local delivery of VEGF after endothelial injury promotes endothelial regeneration, accelerates the recovery of endothelium-dependent relaxation, and reduces neointimal formation (6, 23). Zhao et al. (6) showed that Ang II-mediated expression of VEGF receptors has a biological effect in inducing vascular inflammation (monocyte infiltration) and proliferation, as well as in causing vascular structural changes. As shown in Table 2, we found that VEGF immunopositivity in the 5/6Nx group was higher than in the other groups. VEGF immunopositivity was observed mainly in the tunica media and adventitia. On the other hand, the LA-treated group showed statistically significantly reduced VEGF immunopositivity.
In addition, AT1 receptors in the vasculature, including the aorta and pulmonary and mesenteric arteries, are present in high levels on smooth muscle cells and low levels in the adventitia. Ang II stimulation of AT1 receptors in blood vessels causes vasoconstriction, leading to an increase in peripheral vascular tone and systemic blood pressure (4, 12, 16, 24). In our study, in the 5/6 Nx group, the AT1 immunopositivity was statistically significantly increased compared to the control, LA, and 5/6Nx+LAT groups. The 5/6Nx+LAT group showed statistically significantly decreased AT1 immunopositivity. With the help of this finding, we think that decreased AT1 affects vasoconstriction and leads to the regulation of blood pressure.
Ang II is also known to mediate cell growth and proliferation in cardiac myocytes and fibroblasts, as well as in VSMCs. The vasculopathy is leading to increased expression of ECM and VSMC proliferation (5). SMA is a marker for fibrogenesis and myofibroblast generation. SMA takes part in fibrogenesis and myofibroblast generation. α-SMA correlates with the activation of myofibroblasts (7). Medial thickening with hypertension is attributable primarily to increased smooth muscle cell mass, although enhanced deposition of collagen and elastin plays a contributory role. Recent data would indicate that smooth muscle cell hypertrophy, rather than hyperplasia, is primarily responsible for the greater smooth muscle mass with hypertension (25). In a remnant kidney model, investigators revealed that kidneys from 5/6 Nx hypertensive rats showed a 10-fold increase in α-SMA staining compared to controls (26). We found that there was a significant increase in α-SMA in the 5/6Nx group immu-nohistochemically. α-SMA immunopositivity was observed mainly in the tunica media and adventitia. Besides, in the 5/6Nx+LAT group, α-SMA immunopositivity was decreased. It is possible to think that LA ameliorates VSMC proliferation in abdominal aorta.
The antihypertensive effect of LA was associated with an attenuation of oxidative stress in the aortic artery and with the preservation of glutathione peroxidase activity in the plasma of rats (27). Vasdev et al. (28) observed that LA decreased the blood pressure in spontaneous hypertensive rats. El Midaouri et al. (29) administered 500 mg/kg LA for 3 weeks to Sprague- Dawley rats that were fed a glucose diet and found out that LA caused a decrease in arterial pressure and aortic basal oxygen production. In a study, dietary supplement of LA for 3 weeks prevented the rise of systolic blood pressure (14). Midaoui et al. (30) sorted out whether the effects of dietary supplementation with LA could prevent the increase in mito-chondrial superoxide production in the heart, as well as the enhanced formation of end-products that are associated with the development of hypertension. They found that LA supplementation prevents the development of hypertension and hyperglycemia, presumably through its antioxidative properties (30). Also, researchers found that LA attenuated blood pressure increases in a remnant kidney model (3, 14). In another study, they suggested that the development of hypertension could be either totally prevented or markedly attenuated by chronic treatment with potent antioxidative therapies, such as alpha-lipoic acid (31).
We used 100 mg/kg LA for 8 weeks in a remnant kidney model. We noticed that LA caused a decrease in blood pressure and ameliorated the histomorphological changes seen in abdominal aorta. Besides these changes, we evaluated blood pressure and TMT in the experimental groups and found a positive correlation between these parameters.
Study limitations
The most important limitation of our study was the lack of oxidative stress parameters. We plan to perform our experiments on a higher budget by adding biochemical and molecular data. This study was designed primarily to investigate whether or not lipoid acid could ameliorate histomorphological alterations due to hypertension in aorta tissue. Further studies should be done to expose the effects of hypertension on oxidative stress parameters and damaged cellular mechanisms in aorta tissue.
Conclusion
In conclusion, according to the findings of our study, chronic treatment with appropriate antioxidative therapies could prevent the development of harmful effects seen in target organs in hypertension. LA seems to be a favorable agent because of its antioxidant properties. In our opinion, there is still a need for comparative studies in order to determine the effects of LA on target organ functions in hypertension and complications in various experimental models of hypertension.
Footnotes
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept - B.U.E.; Design - B.U.E.; Supervision - B.U.E.; Resource - B.U.E., S.Ç.M., P.A., O.Y.; Materials - S.Ç.M.; Data collection &/or processing - B.U.E., S.Ç.M., P.A., O.Y.; Analysis &or interpretation - B.U.E.; Literature search - B.U.E.; Writing - B.U.E.; Critical review - B.U.E., S.Ç.M., P.A., O.Y.; Other - B.U.E.
References
- 1.Badyal DK, Lata H, Dadhich AP. Animal models of hypertension and effects of drugs. Indian J Pharmacol. 2003;35:349–62. [Google Scholar]
- 2.Lerman LO, Chade AR, Sica V, Napoli C. Animal models of hypertension: an overview. J Lab Clin Med. 2005;146:160–73. doi: 10.1016/j.lab.2005.05.005. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 3.Mıcılı SÇ, Ergür BU, Özoğul C, Sarıoğlu S, Bağrıyanık HA, Tuğyan K, et al. Effects of lipoic acid in an experimentally induced hypertensive and diabetic rat model. Clin Exp Hypertens. 2013;35:373–81. doi: 10.3109/10641963.2012.732647. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 4.Sparks MA, Parsons KK, Stegbauer J, Gürley SB, Vivekanandan-Giri A, Fortner CN, et al. Angiotension 2 type 1a receptors in vascular smooth muscle cells do not influence aortic remodeling in hypertension. Hypertension. 2011;57:577–85. doi: 10.1161/HYPERTENSIONAHA.110.165274. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cohuet G, Struijker-Boudier H. Mechanisms of target organ damage caused by hypertension: therapeutic potential. Pharmacol Ther. 2006;111:81–98. doi: 10.1016/j.pharmthera.2005.09.002. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 6.Zhao Q, Ishibashi M, Hiasa K, Tan C, Takeshita A, Egashira K. Essential role of vascular endothelial growth factor in angiotensin II-induced vascular inflammation and remodeling. Hypertension. 2004;44:264–70. doi: 10.1161/01.HYP.0000138688.78906.6b. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 7.Mıcılı SC, Göker A, Sayın O, Akokay P, Ergür BU. The effect of lipoic acid on wound healing in a full thickness uterine injury model in rats. J Mol Hist. 2013;44:339–45. doi: 10.1007/s10735-013-9485-8. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 8.Packer L, Witt EH, Tritschler HJ. Alpha-lipoic acid as a biological antioxidant. Free Radic Biol Med. 1995;19:227–50. doi: 10.1016/0891-5849(95)00017-r. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 9.Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM. Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta. 2009;1790:1149–60. doi: 10.1016/j.bbagen.2009.07.026. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Güray M, Sarıoğlu S, Türkmen M, Yılmaz O, Ellidokuz H, Gelal A, et al. Cyclosporine A toxicity in association with reduced renal mass. Transplant Proc. 2003;35:3128–33. doi: 10.1016/j.transproceed.2003.10.029. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 11.Bal E, Murat N, Demir Ö, Soner BC, Can E, Gidener S, et al. Restraint stress impairs erectile responses in rats. Tohoku J Exp Med. 2009;217:239–42. doi: 10.1620/tjem.217.239. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 12.Tüzün F, Gençpınar P, Özbal S, Dilek M, Ergür BU, Duman N, et al. Neuroprotective effect of neotrofin in a neonatal rat model of periventricular leukomalacia. Neurosci Lett. 2012;520:6–10. doi: 10.1016/j.neulet.2012.04.076. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 13.Kosaka S, Pelisch N, Rahman M, Nakano D, Hitomi H, Kobori H, et al. Effects of angiotensin II at1-receptor blockade on high fat diet–induced vascular oxidative stress and endothelial dysfunction in Dahl salt-sensitive rats. J Pharmacol Sci. 2013;121:95–102. doi: 10.1254/jphs.12169fp. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yu X, Liu H, Zou J, Zhu J, Xu X, Ding X. Oxidative stress in 5/6 nephrectomized rat model: effect of alpha-lipoic acid. Ren Fail. 2012;34:907–14. doi: 10.3109/0886022X.2012.691012. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 15.Ghibu S, Richard C, Vergely C, Zeller M, Cottin Y, Rochette L. Antioxidant properties of an endogenous thiol: Alpha-lipoic acid, Useful in the prevention of cardiovascular diseases. J Cardiovasc Pharmacol. 2009;54:391–8. doi: 10.1097/fjc.0b013e3181be7554. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 16.Plante GE. Predisease biological markers: early diagnosis and prevention of arterial hypertension. Metabolism. 2008;57:S36–9. doi: 10.1016/j.metabol.2008.07.014. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 17.Sakuta T, Morita Y, Satoh M, Fox DA, Kashihara N. Involvement of the renin-angiotensin system in the development of vascular damage in a rat model of arthritis. Arthritis Rheum. 2010;62:1319–28. doi: 10.1002/art.27384. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 18.Ruseva B, Atanasova M, Georgieva M, Shumkov N, Laleva P. Effects of selenium on the vessel walls and anti-elastin antibodies in spontaneously hypertensive rats. Exp Biol Med. 2012;237:160–6. doi: 10.1258/ebm.2011.011212. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 19.Yang JC, Haworth L, Sherry RM, Hwu P, Schwartzentruber DJ, Topalian SL, et al. A randomized trial of bevacizumab an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med. 2003;349:427–34. doi: 10.1056/NEJMoa021491. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ferrara N. Vascular endothelial growth factor: basic science and clinical progress. Endocr Rev. 2004;25:581–611. doi: 10.1210/er.2003-0027. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 21.Ho QT, Kuo CJ. Vascular endothelial growth factor: biology and therapeutic applications. Int J Biochem Cell Biol. 2007;39:1349–57. doi: 10.1016/j.biocel.2007.04.010. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Editorial. Is VEGF a new therapeutic target for hypertension in chronic kidney disease? Kidney Res Clin Pract. 2013;32:49–51. doi: 10.1016/j.krcp.2013.04.008. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ishida A, Murray J, Saito Y, Kanthou C, Benzakour O, Shibuya M, et al. Expression of vascular endothelial growth factor receptors in smooth muscle cells. J Cell Physiol. 2001;188:359–68. doi: 10.1002/jcp.1121. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 24.Walter A, Etienne-Selloum N, Sarr M, Kane MO, Beretz A, Schini-Kerth VB. Angiotensin II induces the vascular expression of VEGF and MMP-2 in vivo: preventive effect of red wine polyphenols. J Vasc Res. 2008;45:386–94. doi: 10.1159/000121408. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 25.Chobanian AV, Prescott MF, Haudenschild CC. Recent advances in molecular pathology. The effects of hypertension on the arterial wall. Exp Mol Pathol. 1984;41:153–69. doi: 10.1016/0014-4800(84)90015-7. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 26.Nutter F, Khwaja A, Haylor J. Seliciclib inhibits renal hypertrophy but not fibrosis in the rat following subtotal nephrectomy. Nephron Exp Nephrol. 2012;122:114–22. doi: 10.1159/000350248. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 27.Queiroz TM, Guimarães DD, Mendes-Junior LG, Braga VA. α-Lipoic acid reduces hypertension and increases baroreflex sensitivity in renovascular hypertensive rats. Molecules. 2012;17:13357–67. doi: 10.3390/molecules171113357. [CrossRef] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vasdev S, Gill V, Parai S, Gadag V. Dietary lipoic acid supplementation attenuates hypertension in Dahl salt sensitive rats. Mol Cell Biochem. 2005;275:135–41. doi: 10.1007/s11010-005-1095-7. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 29.El Midaoui A, de Champlain J. Prevention of hypertension, insulin resistance and oxidative stress by a-lipoic acid. Hypertension. 2002;39:303–7. doi: 10.1161/hy0202.104345. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 30.Midaoui AE, Elimadi A, Wu L, Haddad PS, de Champlain J. Lipoic acid prevents hypertension, hyperglycemia, and the increase in heart mitochondrial superoxide production. Am J Hypertens. 2003;16:173–9. doi: 10.1016/s0895-7061(02)03253-3. [CrossRef] [DOI] [PubMed] [Google Scholar]
- 31.de Champlain J, Wu R, Girouard H, Karas M, EL Midaoui A, Laplante MA, et al. Oxidative stress in hypertension. Clin Exp Hypertens. 2004;26:593–601. doi: 10.1081/ceh-200031904. [CrossRef] [DOI] [PubMed] [Google Scholar]


