Abstract
Calcific aortic valve disease (CAVD) involves progressive valve leaflet thickening and severe calcification, impairing leaflet motion. The in vitro calcification of primary rat, human, porcine and bovine aortic valve interstitial cells (VICs) is commonly employed to investigate CAVD mechanisms. However, to date, no published studies have utilised cell lines to investigate this process. The present study has therefore generated and evaluated the calcification potential of immortalized cell lines derived from sheep and rat VICs. Immortalised sheep (SAVIC) and rat (RAVIC) cell lines were produced by transduction with a recombinant lentivirus encoding the Simian virus (SV40) large and small T antigens (sheep), or large T antigen only (rat), which expressed markers of VICs (vimentin and α-smooth muscle actin). Calcification was induced in the presence of calcium (Ca; 2.7 mM) in SAVICs (1.9 fold; P<0.001) and RAVICs (4.6 fold; P<0.01). Furthermore, a synergistic effect of calcium and phosphate was observed (2.7 mM Ca/2.0 mM Pi) on VIC calcification in the two cell lines (P<0.001). Analysis of SAVICs revealed significant increases in the mRNA expression of two key genes associated with vascular calcification in cells cultured under calcifying conditions, runt related transcription factor-2 (RUNX2;1.3 fold; P<0.05 in 4.5 mM Ca) and sodium-dependent phosphate transporter-1 (PiT1; 1.2 fold; P<0.05 in 5.4 mM Ca). A concomitant decrease in the expression of the calcification inhibitor matrix Gla protein (MGP) was noted at 3.6 mM Ca (1.3 fold; P<0.01). Assessment of RAVICs revealed alterations in Runx2, Pit1 and Mgp mRNA expression levels (P<0.01). Furthermore, a significant reduction in calcification was observed in SAVICs following treatment with established calcification inhibitors, pyrophosphate (1.8 fold; P<0.01) and etidronate (3.2 fold; P<0.01). Overall, the present study demonstrated that the use of immortalised sheep and rat VIC cell lines is a convenient and cost effective system to investigate CAVD in vitro, and will make a useful contribution to increasing current understanding of the pathophysiological process.
Keywords: calcification, valve interstitial cell, calcific aortic valve disease, in vitro model, large animal model
Introduction
Calcific aortic valve disease (CAVD) involves gradual thickening of the aortic valve leaflet (aortic sclerosis) and severe calcification, impairing leaflet motion (aortic stenosis) (1). CAVD is a prevalent heart valve disease, present in almost 30% of adults over 65 years, increasing to around 40–50% in those over 75 years (2–4). Dysfunctional heart valves frequently require surgical replacement using mechanical or bioprosthetic valves, however these are prone to failure over time due to structural or thrombosis-related problems (5).
Presently, CAVD is considered an actively regulated and progressive disease (6). The development of this disease is thought to be initiated by injury, inflammation and lipid deposition in the valve, followed by a propagation phase in which factors promoting calcification and osteogenesis drive disease progression (7,8). The increased mechanical stress and injury caused by this early calcification event may then elicit further calcification, leading to a continuous cycle of valve calcification (9).
Valve interstitial cells (VICs) are the predominant cell type in the aortic valves, and play a major role in CAVD progression (7,10). The underlying mechanisms of CAVD share many similarities with that of physiological bone formation (11). VICs are thought to acquire osteoblastic characteristics during the propagation phase of aortic stenosis, following inflammation (7,9). A number of studies have established the ability of VICs to undergo osteogenic trans-differentiation and calcification (12–14). Despite this knowledge, the pathways underlying the initiation and progression of CAVD remain unclear, and studies are needed to elucidate the mechanisms underpinning early disease pathogenesis.
The in vitro calcification of primary porcine (15–17), human (14,18,19), rat (20–22) and bovine (23,24) VICs is commonly used as models of aortic valve calcification. However, to date, the application of a cell line to interrogate VIC function has not been reported. Cell lines offer a valuable alternative to primary cells isolated directly from animals, reducing experimental variation and animal use. To our knowledge this is the first study reporting the generation and evaluation of the calcification potential of immortalised VIC lines derived from sheep (SAVIC) and rat (RAVIC).
Materials and methods
Ethics statement
All animal work was approved by The Roslin Institute's and the University of Edinburgh's Protocols and Ethics Committees. The animals were maintained in accordance with UK Home Office guidelines under the regulations of the Animal (Scientific Procedures) Act 1986.
Establishment of sheep and rat valve interstitial cell lines
Sheep primary aortic VICs were harvested from a 4-year-old Scottish mule sheep (generated from a Bluefaced Leicester sire and Scottish Blackface dam cross; Dryden Farm, Midlothian, UK). Rat aortic VICs were isolated from aortic valve leaflets dissected from the hearts of eight 5-week-old male Sprague Dawley rats as previously described (22). Sheep and rat valve leaflets were digested in 0.6 mg/ml collagenase Type II (Worthington, New Jersey USA) for 30 min and washed in Hanks' Balanced Salt Solution (HBSS; Life Technologies, Paisley, UK) to remove valve endothelial cells. The leaflets were subsequently digested with 0.6 mg/ml collagenase Type II for a further 1 h to release the VICs. Cells were pelleted at 300 × g for 5 min, before resuspension in growth medium consisting of Dulbecco's Modified Eagle Medium: Nutrient Mixture F-12 (DMEM/F12; Life Technologies) supplemented with 10% heat-inactivated foetal bovine serum (FBS; Life Technologies) and 1% gentamicin (Life Technologies), and cultured at 37°C in a humidified atmosphere of 95% air/5% CO2 and grown for four passages.
Immortalised cell lines were established by Capital Biosciences (Gaithersburg, Maryland, USA) from the primary sheep and rat VICs through transduction with recombinant lentivirus encoding Simian virus (SV40) large and small T antigens (sheep), or large T antigen only (rat). The non-clonal cell lines were derived from multiple founder cells. Following continuous culture to 10 passages, transgene expression was confirmed by real-time quantitative PCR (RT-qPCR) for the expression of SV40 large T antigen (Capital Biosciences). The resulting cell lines were designated SAVIC (sheep; SVIC-SVTta) and RAVIC (rat; RVIC-SV40T).
SAVIC and RAVIC cell culture
SAVIC and RAVIC cells were seeded in growth media in multi-well plates at a density of 1.11×104 cells/cm2. Calcification was induced as described previously (22,25). Cells were grown to 80% confluence (Day 0), before treating with control (1.05 mM Ca/0.95 mM Pi) or test media: 1.5 to 3.6 mM calcium (Ca) and/or 1.5 to 2.5 mM phosphate (Pi). CaCl2 and Na2HPO4/NaH2PO4 (Sigma-Aldrich, Dorset, UK) were used to supplement ionic calcium and phosphate in the media. To study the effect of calcification inhibitors and bisphosphonates on VIC calcification, SAVICs were exposed to inorganic pyrophosphate (PPi) and etidronate (both 0.1 mM; Sigma-Aldrich). Cells were incubated for up to 7 days in a humidified atmosphere of 95% air/5% CO2, and the medium was changed every second/third day.
Detection of calcification
Calcium deposition was quantified based on a previously described method (26,27). Cells were washed twice with phosphate buffered saline (PBS) and decalcified with 0.6 M HCl at room temperature for 2 h. Free calcium was determined colorimetrically by a stable interaction with phenolsulphonethalein, using a commercially available kit (Randox Laboratories Ltd., County Antrim, UK), and corrected for total protein concentration (Bio-Rad Laboratories Ltd., Hemel Hempstead, UK) following solubilisation with 0.1 M NaOH/0.1% SDS. Absorbances were measured using a Synergy HT microplate reader (BioTek, Swindon UK) at 570 nm (calcium) and at 690 nm (protein).
Fluorescent immunocytochemical staining
To confirm retention of mesenchymal phenotype, cell monolayers cultured on glass coverslips were fixed with 4% paraformaldehyde (PFA) and washed with phosphate-buffered saline (PBS). Fixed cells were permeabilised with 0.3% Triton X-100 (Sigma-Aldrich) and incubated with rabbit polyclonal anti-α-smooth muscle actin (α-SMA; catalogue #ab5694; 1:100; Abcam, Cambridge, UK), mouse monoclonal anti-vimentin (catalogue #V6384; 1:900; Sigma-Aldrich) or rabbit polyclonal anti-cluster of differentiation 31 (CD31; 1:900; Abcam, Cambridge, UK) overnight at 4°C. After washing, cells were incubated with Alexa Fluor® 488 donkey-anti-rabbit antibody (catalogue #A-21,206; 1:250; ThermoFisher Scientific) or Alexa Fluor® 594 goat-anti-mouse antibody (catalogue #A-110,055; 1:250; ThermoFisher Scientific) for 1 h in the dark. Glass coverslips were mounted onto slides with Prolong Gold Anti-Fade Reagent containing DAPI (Life Technologies). Slides were examined using a Leica DMLB fluorescence microscope (Leica Geosystems, Milton Keynes, UK). In place of the primary antibody, control cells were incubated with non-immune mouse or rabbit IgG (2 µg IgG/ml, Sigma-Aldrich).
Real-time quantitative PCR
RNA extraction was performed using the RNeasy Mini kit (Qiagen, West Sussex, UK), according to the manufacturer's instructions. RNA abundance was quantified, RNA was reverse transcribed, and the expression of selected genes were quantified via RT-qPCR employing the SYBR green detection method (PrecisionPLUS mastermix, Primerdesign Ltd, Southampton, UK), measured on a Stratagene Mx3000P (Agilent Technologies, Stockport, UK), as previously reported (28,29). Sheep primers for Runt-related transcription factor 2 (RUNX2; Forward 5′-CTCCTCCATCCATCCACTCC-3′; Reverse 5′-CAGAGGCAGAAGTCAGAGGT-3′) and Matrix Gla protein (MGP; Forward 5′-ACAACAGAGATGGAGAGCGA-3′; Reverse 5′-CGGAAATAACGGTCGTAGGC-3′) were designed via Primer3 (http://primer3.ut.ee/) to span exon-exon junctions, and obtained from Invitrogen (Paisley, UK). Primers for sheep glyceraldehyde 3-phosphate dehydrogenase (GAPDH; sequences not disclosed), tyrosine 3-monooxygenase (YWHAZ; sequences not disclosed), and sodium-dependent phosphate transporter 1 (PiT1; also known as SLC20A1; Forward 5′-ACATCTTGAACGCCGCTA-3′; Reverse 5′-AGTAGCAGCAATAGCAGTGGTA-3′) were obtained from Primerdesign Ltd. Sheep expression data were normalised against the geometric mean of GAPDH and YWHAZ. Rat primers for Gapdh, Mgp, and Runx2 were obtained from Qiagen (sequences not disclosed; QuantiTect primers, Qiagen). Rat Pit1 primers were acquired from Primerdesign Ltd. (sequences not disclosed). Rat expression data were normalised against Gapdh. The ΔΔCq method was used to analyse relative gene expression (30).
Statistical analysis
All statistical analyses were performed using Minitab 17 (Minitab Inc., Coventry, UK). General Linear Model (GLM) analysis incorporating pairwise comparisons and the Student's t-test were used to assess the data. Data are presented as mean ± standard error of the mean (SEM). P<0.05 was considered to be statistically significant, and P-values are represented as: *P<0.05; **P<0.01; ***P<0.001.
Results
SAVICs express VIC markers
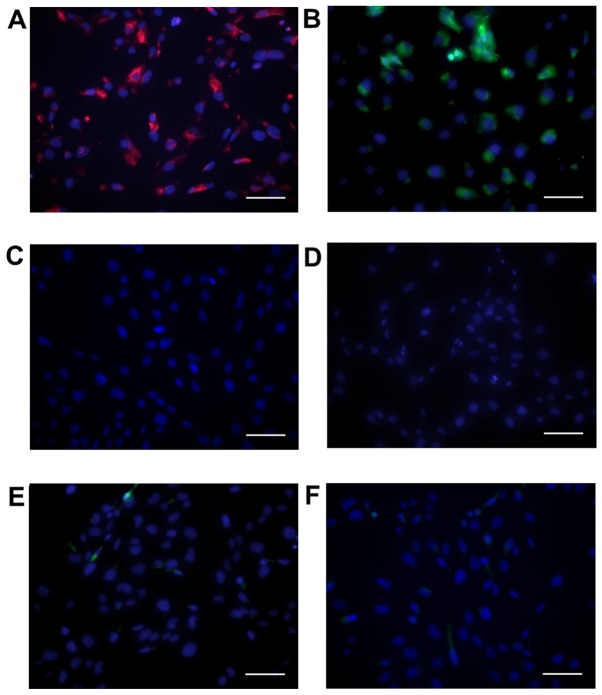
Cells showed positive immunohistochemical staining for vimentin (Fig. 1A) and α-SMA (Fig. 1B), in agreement with previous reports of primary VIC cultures (31,32). Cells were negative for the endothelial marker CD31 (Fig. 1C).
Figure 1.
Expression of valve interstitial cell markers in SAVICs. Positive fluorescent staining for (A) vimentin and (B) alpha-smooth muscle actin (α-SMA) in the sheep aortic valve interstitial cell line (SAVIC). (C) No expression of valve endothelial cell (VEC) marker, CD31, was observed. Representative images of (D) mouse IgG and (E and F) rabbit IgG negative controls. DAPI was used as a counterstain. Scale bar=100 µm.
Calcification of SAVICs
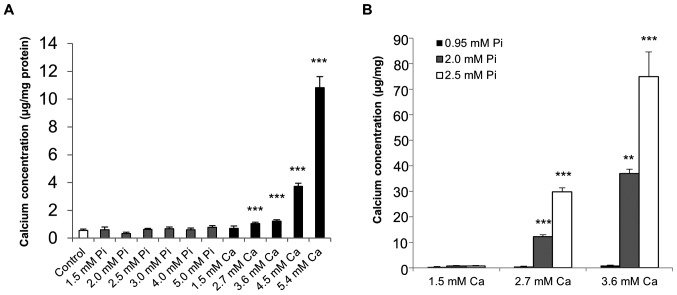
Initial studies were undertaken to determine whether the calcification of SAVICs could be induced when cultured in the presence of calcifying medium containing Ca and/or Pi (Fig. 2A). Ca potently induced the calcification of SAVICs from 2.7 mM (1.9 fold; P<0.001; n=6; Fig. 2A) whereas Pi treatment alone had no effect (Fig. 2A). The treatment of VICs with Ca and Pi together had a synergistic effect on VIC calcification from 2.7 mM Ca/2.0 mM Pi (22.2 fold; P<0.001; n=6; Fig. 2B).
Figure 2.
SAVICs undergo calcification in vitro. Calcium deposition determined by HCl leaching (µg/mg protein) in VICs treated with (A) calcium (Ca) alone (1.5–5.4 mM) and phosphate (Pi) alone (1.5–5.0 mM), and (B) calcium and phosphate in combination (1.5 to 3.6 mM Ca/0.95 to 2.5 mM Pi). Results are presented as mean ± SEM. **P<0.01; ***P<0.001 compared with control; n=6.
Gene expression in early calcification in vitro
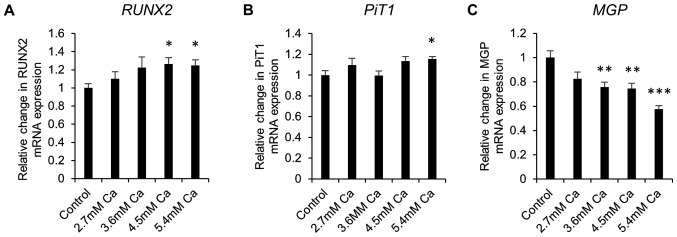
Next, gene expression studies were undertaken in calcifying SAVICs to investigate the expression profile of key genes associated with vascular calcification. Expression of the gene encoding the master osteoblastic transcription factor, RUNX2, was significantly increased from 4.5 mM Ca (1.3 fold; P<0.05; n=6; Fig. 3A) compared to control culture conditions. Expression of sodium-dependent phosphate transporter 1 gene (PiT1) was significantly increased at 5.4 mM Ca (1.2 fold; P<0.05; n=6; Fig. 3B). In contrast, a decrease in the expression of the calcification inhibitor Matrix Gla Protein gene (MGP) was noted from 3.6 mM Ca (1.3 fold; P<0.01; n=6; Fig. 3C).
Figure 3.
Upregulation of osteogenic gene marker expression in calcifying SAVICs. Fold change in the mRNA expression of (A) RUNX2, (B) PIT1, and (C) MGP in sheep aortic valve interstitial cell line (SAVIC) treated with Ca (2.7–5.4 mM) for 48 h. Results are presented as mean ± SEM. *P<0.05, **P<0.01; ***P<0.001 compared to control; n=6.
Inhibition of SAVICs calcification by pyrophosphate and etidronate
Further experiments investigated whether the calcification of SAVICs could be reduced using recognised inhibitors of calcification.
As CAVD is a consequence of hydroxyapatite formation and deposition in the aortic valve, we examined the effects of PPi (0.01-1 mM; Fig. 4A) and the bisphosphonate etidronate (0.01-1 mM; Fig. 4B), both established inhibitors of hydroxyapatite formation (33–35) on SAVIC calcification. A significant decrease in calcium deposition was observed at 1 mM PPi (1.8 fold; P<0.01; n=6; Fig. 4A). Additionally, following exposure to 1 mM etidronate, a significant reduction in calcium deposition (3.2 fold; n=6; P<0.01) was observed, confirming the inhibitory effect of this bisphosphonate on valve calcification in vitro (Fig. 4B).
Figure 4.
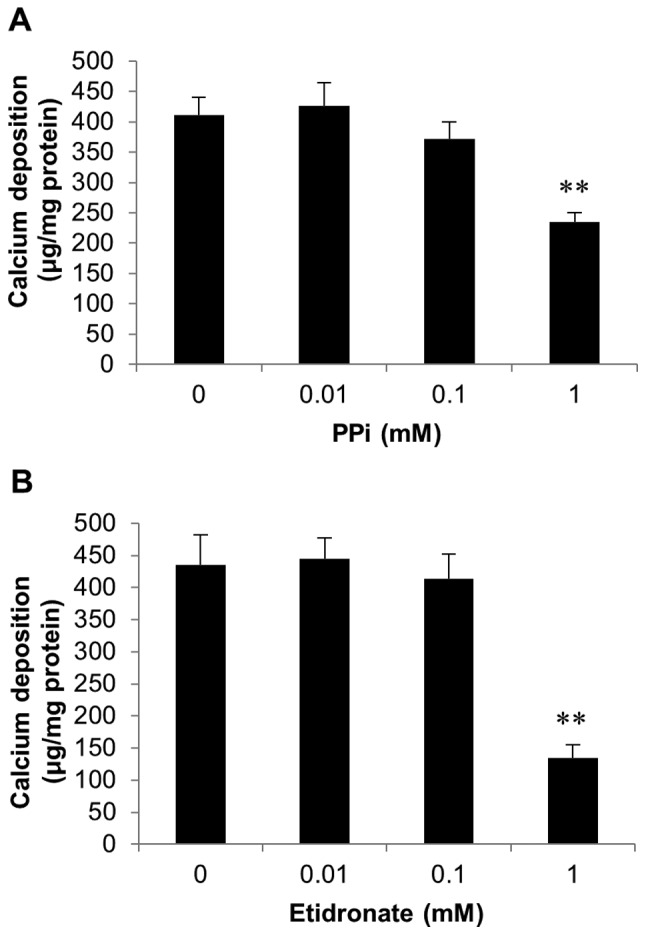
Modulation of SAVIC calcification with established calcification inhibitors. Effect of (A) pyrophosphate (PPi) and (B) etidronate exposure on SAVIC calcification in vitro, following culture for 7 days under calcifying conditions (2.7 mM Ca/2.0 mM Pi). Results are presented as mean ± SEM; **P<0.01; n=6.
Additional RAVIC studies
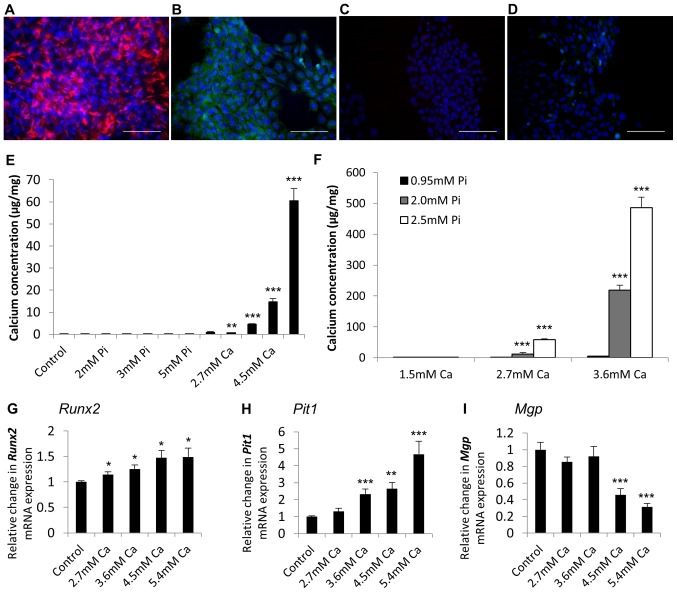
We further corroborated the application of immortalised VICs as an in vitro model of aortic valve calcification through the generation of a rat immortalised VIC cell line, RAVIC. A presented in Fig. 5, RAVICs also showed positive immunohistochemical staining for vimentin (Fig. 5A) and α-SMA (Fig. 5B), and were negative for the endothelial marker CD31 (data not shown).
Figure 5.
Characterisation of RAVICs confirms the use of immortalised VICs as an in vitro CAVD model. Positive staining for (A) vimentin and (B) α-SMA in the rat aortic valve interstitial cell line (RAVIC). Representative images of (C) mouse and (D) rabbit IgG negative control. DAPI was used as a counterstain. Scale bar=100 µm. Calcium deposition determined by HCl leaching (µg/mg protein) in VICs treated with (E) calcium (Ca) alone (1.5–5.4 mM) and phosphate (Pi) alone (1.5–5.0 mM) and (F) calcium and phosphate in combination (1.5 to 3.6 mM Ca/0.95 to 2.5 mM Pi). Fold change in the mRNA expression of (G) Runx2 (H) PiT1 and (I) Mgp in VICs treated with Ca (2.7–5.4 mM) for 48 h. Results are presented as mean ± SEM. *P<0.05, **P<0.01; ***P<0.001 compared to control; n=6.
Analogous to our findings with the SAVICs, Ca markedly induced the calcification of RAVICs from 2.7 mM (4.6 fold; P<0.01; n=6; Fig. 5E) and Pi treatment alone had no effect (Fig. 5E). Furthermore, the treatment of RAVICs with Ca and Pi together had a synergistic effect on cell calcification (2.7 mM Ca/2.0 mM Pi; 82.2 fold; P<0.001; n=6; Fig. 5F). 5.4 mM Ca treatment induced a significant increase in the mRNA expression of Runx2 and Pit1 (1.5 fold; P<0.01, and 4.7 fold; P<0.001, respectively; Fig. 5G and H), with a concomitant reduction in Mgp expression (3.2 fold; P<0.001; Fig. 5I).
Discussion
The calcification of primary porcine (15–17), human (14,18,19), rat (20–22) and bovine (23,24) VICs in 10% serum, inducing an activated phenotype, is frequently employed to produce in vitro models of aortic valve calcification. Compared to cell lines, primary cultures exhibit slow growth and cannot be used beyond a limited number of passages due to senescence and phenotypic changes that occur during culture. Furthermore, the use of primary cells requires animal sacrifice and is labour intensive, costly, and time consuming (36). A number of studies have employed the mouse vascular smooth muscle cell line, MOVAS-1, to investigate arterial calcification in vitro (28,37,38). As yet, however, no published studies have employed immortalized VIC cell lines to investigate aortic valve calcification. In the present study, we have characterized the in vitro calcification potential of immortalised sheep (SAVIC) and rat (RAVIC) VIC cell lines.
Our data have established that calcification of SAVICs and RAVICs can be induced in the presence of calcifying medium containing high concentrations of calcium and phosphate. This was demonstrated through standard assays of in vitro vascular calcification (28,39,40), verifying these cell lines as a feasible and relevant in vitro model of aortic valve calcification.
Calcified SAVICs and RAVICs showed increased gene expression of RUNX2 and PiT1, recognized markers of vascular calcification, with a concomitant reduction in the expression of MGP, an established calcification inhibitor. RUNX2 is an early marker of vascular calcification, initiating osteoblastic differentiation via the upregulation of mineralization proteins, including osteopontin and osteocalcin (10,41). Increased PiT1 expression leads to elevated intracellular phosphate and induces the osteogenic conversion of vascular smooth muscle cells (VSMCs) (42). Conversely, down-regulation of PiT1 gene expression by siRNA silencing has been shown to reduce phosphate uptake and inhibit phosphate-induced VSMC phenotypic transition and calcification (42). MGP is a γ-carboxyglutamic acid-rich and vitamin K-dependent protein, and is proposed to block calcification by antagonising bone morphogenetic protein signaling (10,43). Mouse models that lack MGP develop arterial calcification that results in blood vessel rupture, as well as ectopic cartilage calcification (44). Additionally, circulating MGP levels have been shown to be reduced in aortic valve disease patients (45). Taken together, our data suggests that the culture of SAVICs and RAVICs in calcifying medium is an appropriate in vitro model with which to study the processes leading to aortic valve calcification.
Using known molecular inhibitors, we have also shown that functional studies can be performed in SAVICs. Pyrophosphate is a potent inhibitor of the calcification of primary VSMCs (46) and VICs (23). The bisphosphonate etidronate, an inhibitor of hydroxyapatite formation and a non-hydrolysable analogue of PPi, has also been reported to inhibit the calcification of MOVAS-1 cells and NIH3T3 cells (28,47). The reduction in calcification observed in SAVICs following treatment with PPi and etidronate therefore establishes this cell line as highly appropriate for modeling aortic valve calcification.
The severe clinical implications of CAVD are widely recognized. Nonetheless, the underlying mechanisms have not been fully determined and effective therapeutic strategies that may prevent or possibly reverse aortic valve calcification have yet to be developed. The SAVIC and RAVIC lines are convenient and inexpensive models in which to investigate aortic valve calcification in vitro, and will make a valuable contribution to expanding our knowledge of this pathological process.
Acknowledgements
This study was supported by funding from the Biotechnology and Biological Sciences Research Council (BBSRC) in the form of an Institute Strategic Programme Grant (BB/J004316/1; BBS/E/D/20,221,657) (VEM, KMS and CF), a CASE Studentship BB/K011618/1 (LC) and an East of Scotland Bioscience Doctoral Training Partnership (EASTBIO DTP) Studentship BB/J01446X/1 (HGT).
Glossary
Abbreviations
- CAVD
calcific aortic valve disease
- VIC
valve interstitial cell
- SAVIC
sheep aortic valve interstitial cell line
- RAVIC
rat aortic valve interstitial cell line
- RUNX2/Runx2
Runt-related transcription factor 2
- PiT1/Pit1
sodium-dependent phosphate transporter 1
- MGP/Mgp
matrix Gla protein
- PPi
pyrophosphate
- α-SMA
alpha-smooth muscle actin
- CD31
cluster of differentiation 31
- VEC
valve endothelial cell
- DMEM
Dulbecco's modified eagles media
- FBS
foetal bovine serum
- PFA
paraformaldehyde
- Ca
calcium
- Pi
phosphate
References
- 1.Freeman RV, Otto CM. Spectrum of calcific aortic valve disease: Pathogenesis, disease progression and treatment strategies. Circulation. 2005;111:3316–3326. doi: 10.1161/CIRCULATIONAHA.104.486738. [DOI] [PubMed] [Google Scholar]
- 2.Stewart BF, Siscovick D, Lind BK, Gardin JM, Gottdiener JS, Smith VE, Kitzman DW, Otto CM. Clinical factors associated with calcific aortic valve disease. Cardiovascular Health Study. J Am Coll Cardiol. 1997;29:630–634. doi: 10.1016/S0735-1097(96)00563-3. [DOI] [PubMed] [Google Scholar]
- 3.Coffey S, Cox B, Williams MJ. The prevalence, incidence, progression and risks of aortic valve sclerosis: A systematic review and meta-analysis. J Am Coll Cardiol. 2014;63:2852–2861. doi: 10.1016/j.jacc.2014.04.018. [DOI] [PubMed] [Google Scholar]
- 4.Lindman BR, Clavel MA, Mathieu P, Gardin JM, Gottdiener JS, Smith VE, Kitzman DW, Otto CM. Calcific aortic stenosis. Nat Rev Dis Primers. 2016;2:16006. doi: 10.1038/nrdp.2016.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Balaoing LR, Post AD, Liu H, Minn KT, Grande-Allen KJ. Age-related changes in aortic valve hemostatic protein regulation. Arterioscler Thromb Vasc Biol. 2014;34:72–80. doi: 10.1161/ATVBAHA.113.301936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yutzey KE, Demer LL, Body SC, Huggins GS, Towler DA, Giachelli CM, Hofmann-Bowman MA, Mortlock DP, Rogers MB, Sadeghi MM, Aikawa E. Calcific aortic valve disease: A consensus summary from the alliance of investigators on calcific aortic valve disease. Arterioscler Thromb Vasc Biol. 2014;34:2387–2393. doi: 10.1161/ATVBAHA.114.302523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pawade TA, Newby DE, Dweck MR. Calcification in aortic stenosis: The skeleton key. J Am Coll Cardiol. 2015;66:561–577. doi: 10.1016/j.jacc.2015.05.066. [DOI] [PubMed] [Google Scholar]
- 8.New SE, Aikawa E. Molecular imaging insights into early inflammatory stages of arterial and aortic valve calcification. Circ Res. 2011;108:1381–1391. doi: 10.1161/CIRCRESAHA.110.234146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dweck MR, Pawade TA, Newby DE. Aortic stenosis begets aortic stenosis: Between a rock and a hard place? Heart. 2015;101:919–920. doi: 10.1136/heartjnl-2015-307519. [DOI] [PubMed] [Google Scholar]
- 10.Leopold JA. Cellular mechanisms of aortic valve calcification. Circ Cardiovasc Interv. 2012;5:605–614. doi: 10.1161/CIRCINTERVENTIONS.112.971028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mohler ER, III, Gannon F, Reynolds C, Zimmerman R, Keane MG, Kaplan FS. Bone formation and inflammation in cardiac valves. Circulation. 2001;103:1522–1528. doi: 10.1161/01.CIR.103.11.1522. [DOI] [PubMed] [Google Scholar]
- 12.Poggio P, Sainger R, Branchetti E, Grau JB, Lai EK, Gorman RC, Sacks MS, Parolari A, Bavaria JE, Ferrari G. Noggin attenuates the osteogenic activation of human valve interstitial cells in aortic valve sclerosis. Cardiovasc Res. 2013;98:402–410. doi: 10.1093/cvr/cvt055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Monzack EL, Masters KS. Can valvular interstitial cells become true osteoblasts? A side-by-side comparison. J Heart Valve Dis. 2011;20:449–463. [PMC free article] [PubMed] [Google Scholar]
- 14.Osman L, Yacoub MH, Latif N, Amrani M, Chester AH. Role of human valve interstitial cells in valve calcification and their response to atorvastatin. Circulation. 2006;114:I547–I552. doi: 10.1161/CIRCULATIONAHA.105.001115. [DOI] [PubMed] [Google Scholar]
- 15.Yip CY, Chen JH, Zhao R, Simmons CA. Calcification by valve interstitial cells is regulated by the stiffness of the extracellular matrix. Arterioscler Thromb Vasc Biol. 2009;29:936–942. doi: 10.1161/ATVBAHA.108.182394. [DOI] [PubMed] [Google Scholar]
- 16.Cloyd KL, El-Hamamsy I, Boonrungsiman S, Grau JB, Lai EK, Gorman RC, Sacks MS, Parolari A, Bavaria JE, Ferrari G. Characterization of porcine aortic valvular interstitial cell ‘calcified’ nodules. PLoS One. 2012;7:e48154. doi: 10.1371/journal.pone.0048154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gomez-Stallons MV, Wirrig-Schwendeman EE, Hassel KR, Conway SJ, Yutzey KE. Bone morphogenetic protein signaling is required for aortic valve calcification. Arterioscler Thromb Vasc Biol. 2016;36:1398–1405. doi: 10.1161/ATVBAHA.116.307526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Côté N, El Husseini D, Pépin A, Guauque-Olarte S, Ducharme V, Bouchard-Cannon P, Audet A, Fournier D, Gaudreault N, Derbali H, et al. ATP acts as a survival signal and prevents the mineralization of aortic valve. J Mol Cell Cardiol. 2012;52:1191–1202. doi: 10.1016/j.yjmcc.2012.02.003. [DOI] [PubMed] [Google Scholar]
- 19.El Husseini D, Boulanger MC, Fournier D, Mahmut A, Bossé Y, Pibarot P, Mathieu P. High expression of the Pi-transporter SLC20A1/Pit1 in calcific aortic valve disease promotes mineralization through regulation of Akt-1. PLoS One. 2013;8:e53393. doi: 10.1371/journal.pone.0053393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Seya K, Yu Z, Kanemaru K, Daitoku K, Akemoto Y, Shibuya H, Fukuda I, Okumura K, Motomura S, Furukawa K. Contribution of bone morphogenetic protein-2 to aortic valve calcification in aged rat. J Pharmacol Sci. 2011;115:8–14. doi: 10.1254/jphs.10198FP. [DOI] [PubMed] [Google Scholar]
- 21.Acharya A, Hans CP, Koenig SN, Nichols HA, Galindo CL, Garner HR, Merrill WH, Hinton RB, Garg V. Inhibitory role of Notch1 in calcific aortic valve disease. PLoS One. 2011;6:e27743. doi: 10.1371/journal.pone.0027743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cui L, Rashdan NA, Zhu D, Milne EM, Ajuh P, Milne G, Helfrich MH, Lim K, Prasad S, Lerman DA, et al. End stage renal disease-induced hypercalcemia may promotes aortic valve calcification via Annexin VI enrichment of valve interstitial cell-derived matrix vesicles. J Cell Physiol. 2017;232:2985–2995. doi: 10.1002/jcp.25935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rattazzi M, Bertacco E, Iop L, D'Andrea S, Puato M, Buso G, Causin V, Gerosa G, Faggin E, Pauletto P. Extracellular pyrophosphate is reduced in aortic interstitial valve cells acquiring a calcifying profile: Implications for aortic valve calcification. Atherosclerosis. 2014;237:568–576. doi: 10.1016/j.atherosclerosis.2014.10.027. [DOI] [PubMed] [Google Scholar]
- 24.Rattazzi M, Iop L, Faggin E, Bertacco E, Zoppellaro G, Baesso I, Puato M, Torregrossa G, Fadini GP, Agostini C, et al. Clones of interstitial cells from bovine aortic valve exhibit different calcifying potential when exposed to endotoxin and phosphate. Arterioscler Thromb Vasc Biol. 2008;28:2165–2172. doi: 10.1161/ATVBAHA.108.174342. [DOI] [PubMed] [Google Scholar]
- 25.Reynolds JL, Joannides AJ, Skepper JN, McNair R, Schurgers LJ, Proudfoot D, Jahnen-Dechent W, Weissberg PL, Shanahan CM. Human vascular smooth muscle cells undergo vesicle-mediated calcification in response to changes in extracellular calcium and phosphate concentrations: A potential mechanism for accelerated vascular calcification in ESRD. J Am Soc Nephrol. 2004;15:2857–2867. doi: 10.1097/01.ASN.0000141960.01035.28. [DOI] [PubMed] [Google Scholar]
- 26.Zhu D, Mackenzie NC, Millan JL, Farquharson C, Macrae VE. Upregulation of IGF2 expression during vascular calcification. J Mol Endocrinol. 2014;52:77–85. doi: 10.1530/JME-13-0136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhu D, Mackenzie NC, Shanahan CM, Shroff RC, Farquharson C, MacRae VE. BMP-9 regulates the osteoblastic differentiation and calcification of vascular smooth muscle cells through an ALK1 mediated pathway. J Cell Mol Med. 2015;19:165–174. doi: 10.1111/jcmm.12373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mackenzie NC, Zhu D, Longley L, Patterson CS, Kommareddy S, MacRae VE. MOVAS-1 cell line: A new in vitro model of vascular calcification. Int J Mol Med. 2011;27:663–668. doi: 10.3892/ijmm.2011.631. [DOI] [PubMed] [Google Scholar]
- 29.Staines KA, Zhu D, Farquharson C, MacRae VE. Identification of novel regulators of osteoblast matrix mineralization by time series transcriptional profiling. J Bone Miner Metab. 2014;32:240–251. doi: 10.1007/s00774-013-0493-2. [DOI] [PubMed] [Google Scholar]
- 30.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 31.Liu AC, Joag VR, Gotlieb AI. The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am J Pathol. 2007;171:1407–1418. doi: 10.2353/ajpath.2007.070251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Latif N, Quillon A, Sarathchandra P, McCormack A, Lozanoski A, Yacoub MH, Chester AH. Modulation of human valve interstitial cell phenotype and function using a fibroblast growth factor 2 formulation. PLoS One. 2015;10:e0127844. doi: 10.1371/journal.pone.0127844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Register TC, Wuthier RE. Effect of pyrophosphate and two diphosphonates on 45Ca and 32Pi uptake and mineralization by matrix vesicle-enriched fractions and by hydroxyapatite. Bone. 1985;6:307–312. doi: 10.1016/8756-3282(85)90320-5. [DOI] [PubMed] [Google Scholar]
- 34.Lomashvili KA, Garg P, Narisawa S, Millan JL, O'Neill WC. Upregulation of alkaline phosphatase and pyrophosphate hydrolysis: Potential mechanism for uremic vascular calcification. Kidney Int. 2008;73:1024–1030. doi: 10.1038/ki.2008.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lomashvili KA, Monier-Faugere MC, Wang X, Malluche HH, O'Neill WC. Effect of bisphosphonates on vascular calcification and bone metabolism in experimental renal failure. Kidney Int. 2009;75:617–625. doi: 10.1038/ki.2008.646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Afroze T, Yang LL, Wang C, Gros R, Kalair W, Hoque AN, Mungrue IN, Zhu Z, Husain M. Calcineurin-independent regulation of plasma membrane Ca2+ ATPase-4 in the vascular smooth muscle cell cycle. Am J Physiol Cell Physiol. 2003;285:C88–C95. doi: 10.1152/ajpcell.00518.2002. [DOI] [PubMed] [Google Scholar]
- 37.Idelevich A, Rais Y, Monsonego-Ornan E. Bone Gla protein increases HIF-1alpha-dependent glucose metabolism and induces cartilage and vascular calcification. Arterioscler Thromb Vasc Biol. 2011;31:e55–e71. doi: 10.1161/ATVBAHA.111.230904. [DOI] [PubMed] [Google Scholar]
- 38.Kelynack KJ, Holt SG. An in vitro murine model of vascular smooth muscle cell mineralization. Methods Mol Biol. 2016;1397:209–220. doi: 10.1007/978-1-4939-3353-2_14. [DOI] [PubMed] [Google Scholar]
- 39.Zhu D, Rashdan NA, Chapman KE, Hadoke PW, MacRae VE. A novel role for the mineralocorticoid receptor in glucocorticoid driven vascular calcification. Vascul Pharmacol. 2016;86:87–93. doi: 10.1016/j.vph.2016.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhu D, Hadoke PW, Wu J, Vesey AT, Lerman DA, Dweck MR, Newby DE, Smith LB, MacRae VE. Ablation of the androgen receptor from vascular smooth muscle cells demonstrates a role for testosterone in vascular calcification. Sci Rep. 2016;6:24807. doi: 10.1038/srep24807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Johnson RC, Leopold JA, Loscalzo J. Vascular calcification: Pathobiological mechanisms and clinical implications. Circ Res. 2006;99:1044–1059. doi: 10.1161/01.RES.0000249379.55535.21. [DOI] [PubMed] [Google Scholar]
- 42.Li X, Yang HY, Giachelli CM. Role of the sodium-dependent phosphate cotransporter, Pit-1, in vascular smooth muscle cell calcification. Circ Res. 2006;98:905–912. doi: 10.1161/01.RES.0000216409.20863.e7. [DOI] [PubMed] [Google Scholar]
- 43.Yao Y, Bennett BJ, Wang X, Rosenfeld ME, Giachelli C, Lusis AJ, Boström KI. Inhibition of bone morphogenetic proteins protects against atherosclerosis and vascular calcification. Circ Res. 2010;107:485–494. doi: 10.1161/CIRCRESAHA.110.219071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, Behringer RR, Karsenty G. Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature. 1997;386:78–81. doi: 10.1038/386078a0. [DOI] [PubMed] [Google Scholar]
- 45.Koos R, Krueger T, Westenfeld R, Kühl HP, Brandenburg V, Mahnken AH, Stanzel S, Vermeer C, Cranenburg EC, Floege J, et al. Relation of circulating Matrix Gla-Protein and anticoagulation status in patients with aortic valve calcification. Thromb Haemost. 2009;101:706–713. [PubMed] [Google Scholar]
- 46.Villa-Bellosta R, Wang X, Millan JL, Dubyak GR, O'Neill WC. Extracellular pyrophosphate metabolism and calcification in vascular smooth muscle. Am J Physiol Heart Circ Physiol. 2011;301:H61–H68. doi: 10.1152/ajpheart.01020.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Li Q, Kingman J, Sundberg JP, Levine MA, Uitto J. Dual effects of bisphosphonates on ectopic skin and vascular soft tissue mineralization versus bone microarchitecture in a mouse model of generalized arterial calcification of infancy. J Invest Dermatol. 2016;136:275–283. doi: 10.1038/JID.2015.377. [DOI] [PMC free article] [PubMed] [Google Scholar]