Abstract
Osteosarcoma is a kind of high-risk sarcoma of the skeleton typically observed in people under 25 years old. Currently, radiotherapy is widely applied in cancer treatment. However, osteosarcoma is radioresistant and accordingly new, more effective radiosensitizers are needed. miRNAs have been reported to play an important role in osteosarcoma radiosensitivity. We examined the modulating effect of miR-328-3p in vivo and in vitro. miR-328-3p was downregulated in HOS-2R cells. The overexpression of miR-328-3p enhanced the radiosensitivity of osteosarcoma cells. miR-328-3p inhibited proliferation and promoted apoptosis in osteosarcoma cells under radiation conditions. In cells overexpressing miR-328-3p, H2AX expression was downregulated. We found that miR-328-3p targets H2AX and inhibits its expression. It was concluded, that miR-328-3p enhances the radiosensitization of osteosarcoma following X-ray irradiation, and determined that it directly targets H2AX to regulate radiosensitization.
Keywords: miR-328-3p, radiosensitivity, osteosarcoma, H2AX
Introduction
Osteosarcoma (OS) originates from the metaphyseal areas of the long bones and is a frequently occurring disease in children, adolescents, and young adults (1,2). The risk of developing OS is highest among people under 25 years old and second-highest in individuals aged 60–80 years (3,4). Before the 1970s, the mortality rate was ~80% because surgery was the only method for OS treatment. With advances in surgical techniques and the emergence of chemotherapy, the disease-free survival rate has improved substantially (5,6). In response to tumor metastasis, radiotherapy has emerged as a more efficient treatment (7,8). However, radiotherapy to treat OS is limited by radioresistance (9,10). Therefore, it is necessary to study the molecular mechanism of OS radioresistance to improve the efficiency of OS radiotherapy. Ionizing radiation can activate reactive oxygen species (ROS) and cause oxidative damage to DNA bases. DNA double-strand breaks (DSB) have a dual influence on tumor therapy; they can lead to cancer, but can also kill cancer cells. The phosphorylation of histone H2AX is a sensitive marker for DSB (11) and H2AX contributes to DSB repair (12). The expression level of phospho-H2AX (pH2AX) contributes to the detection of epithelial ovarian cancer (13). γH2AX plays an important role in the response to DSB and participates in the DSB repair process mediated by DNA repair enzymes (14).
MicroRNAs (miRNAs) are short (18–24 nucleotides) single-stranded non-coding RNAs; they cannot be translated into proteins or oligopeptides. In some cases, miRNAs can post-transcriptionally regulate gene expression by binding to the target site in mRNA (15,16). Some studies have found that miRNAs are involved in the process of tumor development. The overexpression of miR-187 can inhibit colorectal cancer progression by directly targeting CD276 (17). In OS, miR-489-3p is downregulated and the overexpression of miR-489-3p can suppress OS progression by targeting PAX3 (18). Recent research has indicated that miRNAs are also involved in tumor radioresistance. miR-185 can target ATR (rad3-related), which can respond to DNA damage and DNA replication stress, enhancing radiosensitivity in tumor cells (19). In esophageal squamous cell carcinoma, the expression level of miR-98 is positively correlated with radiosensitivity (20). In cervical cancer cells, miR-424 can target aprataxin and the overexpression of miR-424 can increase the cell sensitivity to radiation (21).
In our previous study, miR-328-3p expression was distinctly reduced in a radiation-tolerant HOS cell line (HOS-2R) (Fig. 1). We hypothesized that miR-328-3p plays a role in the development of OS cells. In the present study, we examined the correlation between miR-328-3p and radioresistance in OS cells in vivo and in vitro. We determined the regulatory role of miR-328-3p in OS radioresistance by overexpressing or knocking out miR-328-3p in OS cell lines, and we showed that miR-328-3p targets H2AX to enhance OS radiosensitivity.
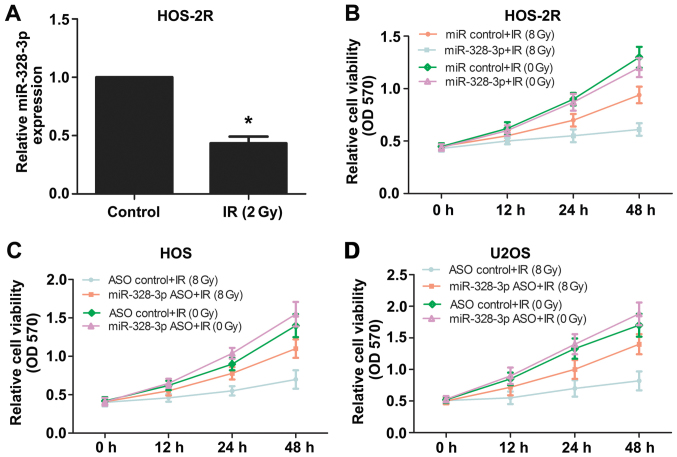
Figure 1.
The expression level of miR-328-3p affects the radiosensitivity of osteosarcoma cells. (A) miR-328-3p expression levels were detected by qPCR in HOS and HOS-2R cells. HOS acted as controls, and radiation-tolerant HOS cell lines (HOS-2R) acted as IR (2 Gy) group which were established by 2 Gy X-ray irradiation. (B) Survival rate of HOS-2R overexpressing miR-328-3p was determined by an MTT assay after 8 Gy X-ray irradiation. (C and D) Survival rates of HOS and U2OS cells after silencing miR-328-3p were determined by an MTT assay after irradiation at 8 Gy. *p<0.05.
Materials and methods
Cell culture, irradiation, and transfection
The human osteosarcoma cell line HOS was purchased from the Cell Culture Center, Institute of Basic Medical Sciences (Beijing, China). U2-OS cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cultures of the cell lines were maintained in a humidified, 37°C, 5% CO2 incubator. McCoy's 5A Media (modified with Tricine) (Sigma, St. Louis, MO, USA) with 10% FBS (Gibco, Grand Island, NY, USA) was used for cell culture. The cells in exponential growth were exposed to X-ray radiation at 2, 4 or 8 Gy at room temperature to measure radioresistance in vitro. An X-ray machine (Faxitron RX-650; Tucson, AZ, USA) with 100 kVp (0.835 Gy/min) was used for irradiation.
Cells were transfected with miRNA mimics and a control, i.e., miR-328-3p mimics, miR-328-3p ASO (anti-miRNA oligos), or ASO control (RiboBio, Guangzhou, China). Additionally, siRNA-H2AX and siRNA control were purchased from GeneChem (Shanghai, China).
Establishment of radioresistant cell lines
HOS cells (2×106) were cultured in McCoy's 5A Media supplemented with 10% FBS. Ten minutes before irradiation, the cells were replaced with fresh medium, then cells were irradiated by an X-ray machine (0.835 Gy/min) with 2 Gy dose. To obtain radioresistant cell population, a total dose of 44 Gy was reached by 22 fractions of irradiation. Each time after irradiation, the culture medium was replaced with fresh complete medium. When the cell confluence reached >80%, the cells were subcultured. Additionally, the next irradiation was performed when the cell confluence reached 50%.
Quantitative real-time PCR
Total RNA was extracted from OS cell lines using TRIzol LS reagent (Invitrogen, Carlsbad, CA, USA) and cDNA was synthesized using the TaqMan miRNA Reverse Transcription kit (Applied Biosystems, Waltham, MA, USA). Then, the cDNA was amplified for 28 cycles in a PCR machine (Roche): 94°C for 30 sec; annealing for 30 sec, 72°C for 30 sec. Quantitative real-time PCR (qRT-PCR) analyses were performed with SYBR® Premix Ex Taq™ (Takara, Japan) using a StepOne-plus Real-Time PCR System (Applied Biosystems). The PCR cycling was as follows: 95°C for 2 min, 40 cycles of 95°C for 10 sec, 60°C for 20 sec, 72°C for 20 sec. The relative expression levels of miRNAs were calculated using the 2−∆∆Ct method. U6 snRNA was used as internal controls to normalize the expression levels of miRNAs. The qPCR primers for miR-328-3p were as follows: F-5′-TGCGGCTGGCCCTCTCTGCCC-3′; R-5′-CCAGTGCAGGGTCCGAGGT-3′. The qPCR primers for U6 snRNA were as follows: F-5′-TGCGGGTGCTCGCTTCGGCAGC-3′; R-5′-CCAGTGCAGGGTCCGAGGT-3′.
MTT assay
The 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) assay was performed to measure the viability of osteosarcoma cells using the Cell Proliferation Kit I (Sigma), following the procedure described in the kit manual. Cells were grown in 96-wells in a final volume of 100 µl of culture medium per well in a humidified 37°C incubator. MTT labeling reagent was added to each well to obtain a final concentration of 0.5 mg/ml. Samples were incubated for 4 h in a humidified atmosphere. Then, 100 µl of solubilization solution was added to each well, followed by incubation (37°C) overnight. Absorbance of the samples was measured spectrophotometrically using a microplate reader.
Western blot analysis
Cells were scraped from the wells after washing twice with cold PBS. Total proteins were extracted using ice-cold RIPA lysis buffer (Solarbio, Beijing, China) with a protease inhibitor. Then, 40 µg of total proteins from each sample was subjected to 12% SDS-PAGE and transferred to a nitrocellulose membrane (Bio-Rad, Hercules, CA, USA). After samples were washed and blocked with 5% non-fat milk, the membrane was incubated with the primary antibodies (Abcam, UK). After they were washed in TBS, the membrane was treated with a horseradish peroxidase-conjugated secondary antibody (ICLLAB, USA). Proteins were observed by chemiluminescence (Millipore, Billerica, MA, USA).
Survival colony formation assay
Cell suspensions were exposed to X-ray radiation; then, 1×106 cells were seeded in 6-well plates and cultured for 12 days. Detailed cultivation methods are described above (Cell culture and irradiation). The number of colonies that contained at least 50 cells was counted.
Apoptosis
Cells (1×107) were washed with cold PBS and fixed in cold 70% ethanol overnight at −20°C. The Annexin V-FITC Apoptosis Detection kit (Abcam) was used to perform apoptosis assays following the manufacturer's instructions. The samples were analyzed using the FACS III (BD Biosciences, Franklin Lakes, NJ, USA).
Tumor xenograft assay
Female nude mice (BALB/c, 5 weeks old) were purchased from Nanjing Biomedical Research Institute of Nanjing University (Nanjing, China) and housed under SPF conditions. All animal experiments were approved by the Institutional Animal Care and Use Committee of Guizhou Provincial People's Hospital (Guizhou, China).
The mouse tumor xenograft model was established; 1×107 HOS cells were suspended in 0.1 ml of PBS and subcutaneously injected into the right thigh of the nude mice. Ten days later, the mice were intratumorally injected with 5 nmol miR-328-3p agomir or 5 nmol miR-NC agomir at days 12, 16, 20, 24, 28 and 32. Mice were exposed to 4 Gy of X-ray radiation after each intratumoral injection. At day 34, the mice were sacrificed by cervical dislocation. The tumor weight was measured using electronic scales (Sartorius, Beijing, China), and the tumor volume was calculated as length × (width)2/2.
Statistical analysis
One-way ANOVA was performed to determine the statistical significance of differences between groups. The numerical results are presented as means ± standard deviation. Differences with a p-value of <0.05 were considered statistically significant.
Results
Modulation of miR-328-3p alters the radiosensitivity of osteosarcoma cells
HOS and U2OS are human osteosarcoma cell lines, we used these two kinds of cells to explore the effect of miR-328-3p on the radiosensitivity of osteosarcoma cells. In order to investigate the relationship between miR-328-3p and radiosensitivity in OS cells, we also established radiation-tolerant HOS cell lines (HOS-2R) by X-ray (2 Gy). miR-328-3p levels in HOS-2R cells were detected by qPCR and the expression levels were downregulated compared with those in control cells (Fig. 1A). To explore whether the modulation of miR-328-3p affects the radiosensitivity of OS cells, we also performed an MTT assay to determine the survival rate of HOS-2R cells, HOS cells, and U2OS cells following irradiation at 0 or 8 Gy. The overexpression of miR-328-3p reduced HOS-2R cell viability following 8 Gy of X-ray, and there was no significant difference between miRNA control and miR-328-3p mimic groups following irradiation with 0 Gy (Fig. 1B). Then, we silenced miR-328-3p in HOS and U2OS cells using antisense oligonucleotides (ASO). The deletion of miR-328-3p increased the viability of HOS cells following 8-Gy irradiation (Fig. 1C), similar to the results obtained for U2OS cells (Fig. 1D). These results confirmed that miR-328-3p has a regulatory role in OS cell radiosensitivity.
miR-328-3p increases the rate of apoptosis and reduces the proliferation ability of osteosarcoma cells after ionizing radiation
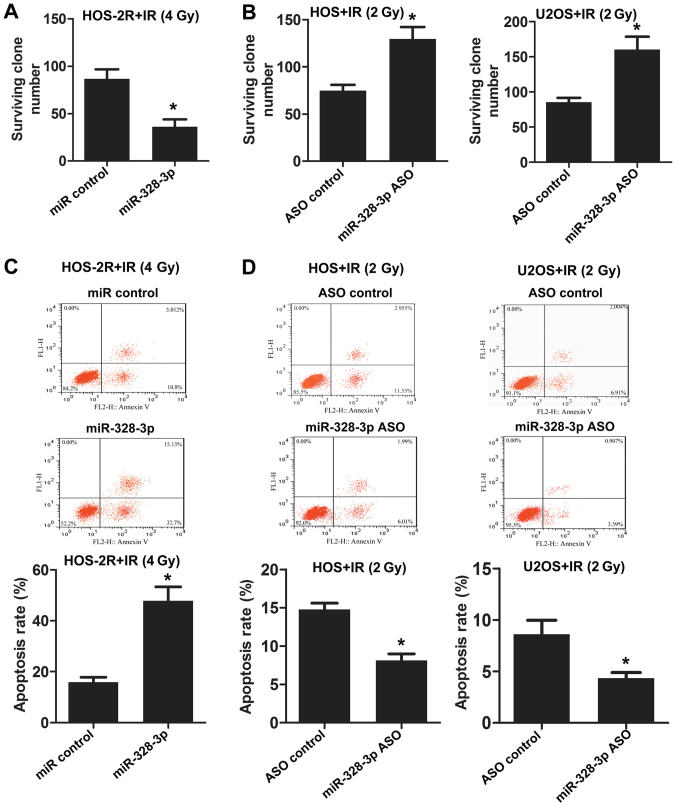
Based on the observation that the abnormal expression of miR-328-3p alters the radiosensitivity of OS cells, we examined whether miR-328-3p influences the apoptosis and proliferation of OS cells. Radioresistant cell lines were established under the condition of 2-Gy irradiation, so we need to increase the dose of X-ray to 4 Gy. Compared to HOS-2R, HOS and U2OS was more sensitive to irradiation. 2 Gy X-ray exposure was enough to investigate the biological characteristics of HOS and U2OS cells. miR-328-3p was overexpressed in HOS-2R cells, and the number of surviving clones after X-ray exposure at 0 or 4 Gy was counted. The surviving clone number for HOS-2R cells overexpressing miR-328-3p decreased following irradiation at 4 Gy (Fig. 2A). Then, we inhibited the expression of miR-328-3p in HOS and U2OS cells using miR-328-3p ASO. The deficiency of miR-328-3p increased the survival rate after X-ray exposure at 2 Gy, both in HOS and U2OS cells (Fig. 2B). Apoptosis was analyzed by flow cytometry following miR-328-3p overexpression or knockout in various cell types (including HOS-2R, HOS and U2OS cells). The overexpression of miR-328-3p enhanced HOS-2R cell apoptosis after exposure to 4 Gy (Fig. 2C). In HOS and U2OS cells in which miR-328-3p was knocked out, apoptosis decreased after exposure to 2 Gy (Fig. 2D). These results confirmed that miR-328-3p inhibits proliferation and promotes apoptosis in OS cells under radiation conditions, and miR-328-3p enhances the radiosensitivity of OS.
Figure 2.
miR-328-3p enhanced X-ray-induced apoptosis and decreased the proliferation ability of osteosarcoma cells after X-ray exposure. (A) The number of surviving clones of miR-328-3p-overexpressing HOS-2R cells was counted after 0 or 4 Gy X-ray irradiation. (B) Survival rates of miR-328-3p ASO HOS and U2OS cells were estimated after 2 Gy X-ray irradiation. The apoptosis of HOS-2R cells overexpressing miR-328-3p (C) and HOS and U2OS cells with silenced miR-328-3p (D) was analyzed by flow cytometry following irradiation. *p<0.05.
miR-328-3p targets H2AX and inhibits its expression
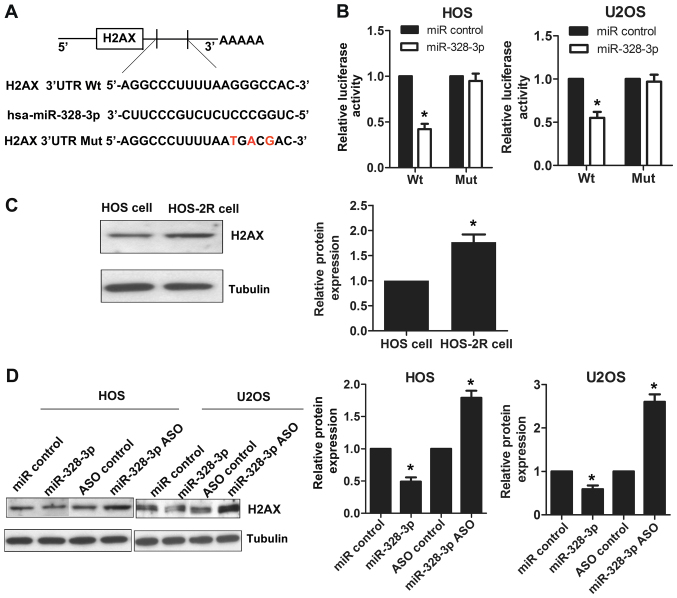
Using the bioinformatics tool TargetScan, we determined that H2AX is a predicted target for miR-328-3p. As shown in Fig. 3A, the 3′-UTR region of H2AX contains a sequence that matches the specific binding sequences of miR-328-3p. A luciferase reporter assay was used to further prove the targeting of miR-328-3p to the H2AX 3′-UTR. The cotransfection of miR-328-3p mimics with the wild-type vector reduced luciferase activity in HOS cells compared to that of control-treated cells, and a similar trend was found in U2OS cells (Fig. 3B). No statistically significant differences in luciferase activity were observed for cells cotransfected with miR-328-3p mimics and mutant vector compared to control-treated cells (Fig. 3B). We also detected the expression levels of H2AX in HOS-2R and HOS cells by western blotting, and HOS-2R cells had a higher level of H2AX expression compared to that of HOS cells (Fig. 3C), in contrast to the miR-328-3p expression patterns (Fig. 1A). To explore the miR-328-3p regulatory effect on H2AX, we used HOS and U2OS cell lines. We detected the expression levels of H2AX by western blotting after overexpressing or silencing miR-328-3p. We found that the expression of H2AX was suppressed after miR-328-3p mimic transfection in HOS cells. When miR-328-3p was knocked out using miR-328-3p ASO, the expression of H2AX increased. A similar phenomenon was observed in U2OS cells (Fig. 3D). These results showed that miR-328-3p targets H2AX, and the modulation of miR-328-3p affects the expression of H2AX.
Figure 3.
miR-208b targets H2AX and represses H2AX expression by binding to its 3′-UTR. (A) The 3′-UTR region of H2AX contains a sequence that matches the specific binding sequences of miR-328-3p. (B) The targeting effect of miR-328-3p on H2AX 3′-UTR was detected by a luciferase reporter assay in HOS and U2OS cells. (C) The expression levels of H2AX in HOS and HOS-2R cells were determined by western blotting. (D) The expression levels of H2AX in HOS and U2OS cells were determined by western blotting after overexpressing or silencing miR-328-3p. *p<0.05.
miR-328-3p mediates the radiosensitivity of osteosarcoma cells by targeting H2AX
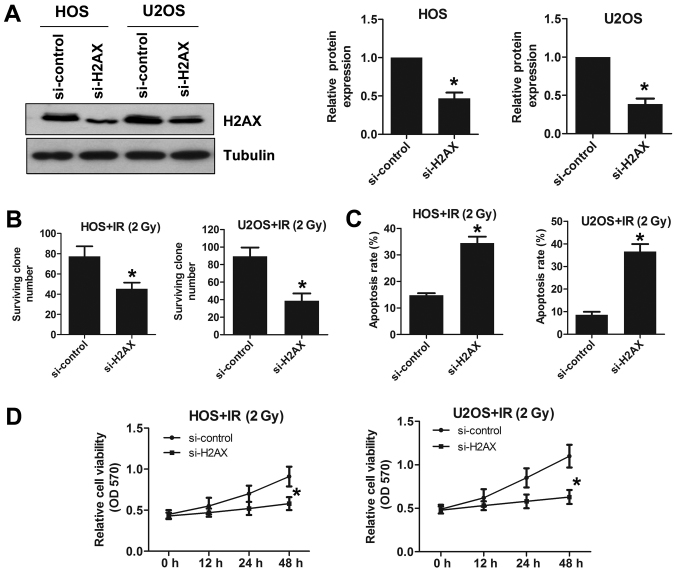
We knocked out H2AX using H2AX siRNA in the HOS and U2OS cell lines. We verified the knockout efficiency by western blotting (Fig. 4A). The survival rate of H2AX-deficient HOS cells was reduced following irradiation at 2 Gy compared to that of sham-treated cells (Fig. 4B). A similar trend was also found in U2OS cells (Fig. 4B). To explore the effect of H2AX expression changes on apoptosis in OS cell lines, we examined apoptosis using a flow cytometry assay. Under radiation conditions of 2 Gy, H2AX deletion increased apoptosis in both HOS and U2OS cells (Fig. 4C). We also found that deleting H2AX reduced cell viability following irradiation at 2 Gy in both HOS and U2OS cells (Fig. 4D). Taken together, these results showed that deleting H2AX increases the radiosensitivity of OS cell lines.
Figure 4.
H2AX downregulation reduces radioresistance in osteosarcoma cells. (A) To silence H2AX, si-H2AX was used, and the knockout efficiency of H2AX was detected by western blotting. (B) The survival rate of osteosarcoma cells with or without H2AX silencing after irradiation at 2 Gy. (C) The apoptosis assay was performed by flow cytometry following 2 Gy X-ray irradiation. (D) Cell viability assay was performed in si-control and si-H2AX cells after 2 Gy X-ray irradiation. *p<0.05.
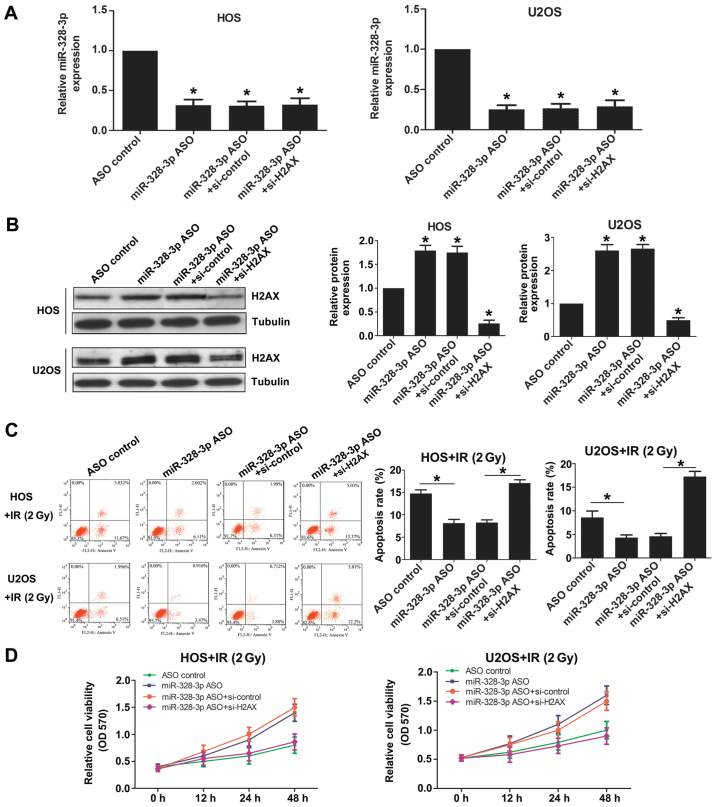
To further study the function of miR-328-3p targeted to H2AX, we detected the cell sensitivity to radiation by western blotting. Cells were transfected with ASO con (control), miR-328-3p ASO, miR-328-3p ASO+siRNA con or miR-328-3p ASO+H2AX siRNA, respectively. The expression levels of miR-328-3p were reduced in miR-328-3p ASO transfected, miR-328-3p ASO and siRNA con cotransfected, and miR-328-3p ASO and H2AX siRNA cotransfected HOS cells, similar to the results observed in U2OS cells (Fig. 5A). The expression levels of H2AX increased after silencing miR-328-3p in both HOS and U2OS cells (Fig. 5B). The deficiency of miR-328-3p decreased apoptosis after 2-Gy X-ray exposure in both HOS and U2OS cells (Fig. 5C). The effect of miR-328-3p knockout on reducing apoptosis was weakened after knocking out miR-328-3p and H2AX simultaneously (Fig. 5C). The deficiency of miR-328-3p increased the viability of both HOS and U2OS cells after 2-Gy X-ray irradiation (Fig. 5D). The enhanced cell viability after miR-328-3p knockout was weakened after knocking out miR-328-3p and H2AX simultaneously (Fig. 5D). These results showed that miR-328-3p targets H2AX and thereby miR-328-3p can participate in the regulation of OS radiosensitivity.
Figure 5.
miR-328-3p regulates the radiosensitivity of osteosarcoma cells by targeting H2AX. (A) The expression levels of miR-328-3p were detected by qPCR in HOS and H2OS cells cotransfected with ASO control, miR-328-3p ASO, miR-328-3p ASO+si-control, and miR-328-3p ASO+si-H2AX. (B) The expression levels of H2AX were detected by western blotting after silencing miR-328-3p in HOS and H2OS cells. (C) Apoptosis was detected by flow cytometry in HOS and H2OS cells. (D) Cell viability was examined by MTT assays in HOS and H2OS cells. *p<0.05.
Overexpression of miR-328-3p enhances the radiosensitivity of osteosarcoma in mice
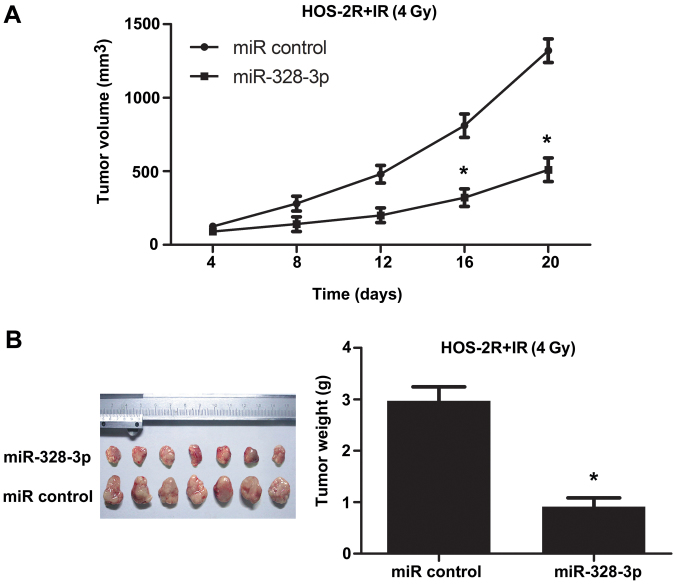
A xenograft tumor model was examined to further study the effect of miR-328-3p on the radiosensitivity of OS in vivo. To test tumor growth following irradiation at 4 Gy, we subcutaneously injected miRNA control transfected HOS-2R cells or miR-328-3p transfected HOS-2R cells into the right thigh of nude mice. In the miRNA con-transfected HOS-2R group (control group), tumor growth was checked. After irradiation at 4 Gy, tumors continued to grow in the control group. However, tumors in the miR-328-3p-transfected HOS-2R group grew substantially slower compared to tumor growth in the control group (Fig. 6). These data showed that miR-328-3p can promote growth restraint in OS after irradiation at 4 Gy in mice.
Figure 6.
Modulation of miR-328-3p regulates the radiosensitivity of osteosarcoma in mice. To explore the role of miR-328-3p in regulating osteosarcoma in vivo, a subcutaneously injected mouse model was established. Tumor volume and tumor weight were measured. *p<0.05.
Discussion
Some miRNAs can regulate cancer cell radiosensitivity to ionizing radiation, and downstream targets have been identified (20–26). In our radiation-tolerant HOS cell line (HOS-2R), miR-328-3p was downregulated. A previous study showed that miR-328 can restrain proliferation and migration of pulmonary arterial smooth muscle cells by targeting PIM-1 (27). In our study, we found a positive correlation between miR-328-3p and OS radiosensitivity. Overexpressed miR-328-3p in OS increases the rate of apoptosis and reduces the proliferation ability after irradiation. miRNAs cannot be translated into proteins or oligopeptides and they often combine with the target points in mRNA to regulate gene expression and further affect downstream pathways (15). miR-494-3p can inhibit the expression of Bmil, which leads to cell aging and thus to increased radiosensitivity of oral squamous cell carcinoma cells (28). In a study of radiation-resistant tumor glioblastoma multiforme, Guo et al found that miR-26a disturbs downstream pathways by inhibiting the expression of ATM via its 3′-UTR (29). In the SWI2/SNF2-family, helicase-like transcription factor (HLTF) is an ATP-dependent chromatin remodeling enzyme related to DNA damage repair. HLTF mRNA is a target of miR-145, by which miR-145 can enhance the radiosensitivity of cervical cancer cells (30). These studies show that miRNA can take part in the development of cancer. Also, the regulated role of miRNAs in OS has been revealed in previous studies, such as miR-542-5p, miR-217, miR-182, miR-199a-3p and miR-153 related to the OS progression (31–35).
These past results suggest that H2AX plays an important role in DSB (11–13); H2AX is involved in maintaining genome stability, and provides a platform for repair factors to function (14,36–38). Ionizing radiation can cause DSBs and, subsequently, H2AX phosphorylation at Ser139 via ATM, generating γH2AX foci (11,37). We found that H2AX was knocked out, the survival rate and the viability of OS cells were both reduced and the apoptosis was increased following irradiation at 2 Gy. The imbalance of H2AX probably perturbed the progress of chromatin remodeling and DNA repair, thus affecting the radiation tolerance of OS cells. The phosphorylation of H2AX can promote genome stability and accelerated DSB repair (12,36). γH2AX plays a critical role in the formation of chromatin-remodeling complexes, and γH2AX also helps DNA repair enzymes function effectively (14,38). Liao et al found that STAT3 can promote the phosphorylation of H2AX and enhance the expression of GADD45γ and MDC-1 after UV exposure, and STAT3 reduces DNA damage caused by UV (39). The phosphorylation of H2AX promotes NBS1 expression at damage sites, and the acetylation of histone H2AX at Lys 5 is crucially important for the damaged chromatin-specific binding of NBS1 (40).
We proved that miR-328-3p target to the H2AX 3′-UTR. Previous studies have shown that H2AX is regulated by upstream miRNAs. For example, miR-138 directly targets the histone H2AX to modulate the DNA damage response (41). Lin28 targets H2AX, and a deficiency of Lin28 increases radiosensitivity in breast cancer cells (42). H2AX has one more upstream regulatory element: miR-328-3p. In addition, a negative correlation was found between H2AX expression and miR-328-3p level in HOS-2R cells. This tendency provided an indirect proof that H2AX is a target of miR-328-3p.
One limitation of our study was the inability to obtain clinical specimens for further verification of the effect of miR-328-3p in humans. We tested xenograft tumor growth in mice. The results are consistent with the experiments in vivo. In general, miR-328-3p is a positive regulatory factor in OS radiosensitivity by H2AX, and it is worthwhile to study the function of miRNAs to treat cancer by regulating downstream elements. miR-328-3p shows promise as a new potential therapy target in OS.
Acknowledgements
This study was supported by The Youth Fund of Guizhou Provincial People's Hospital (GZSYQN[2015]06) and the National Natural Science Foundation of China (31660265).
References
- 1.Longhi A, Errani C, De Paolis M, Mercuri M, Bacci G. Primary bone osteosarcoma in the pediatric age: State of the art. Cancer Treat Rev. 2006;32:423–436. doi: 10.1016/j.ctrv.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 2.Picci P. Osteosarcoma (osteogenic sarcoma) Orphanet J Rare Dis. 2007;2:6. doi: 10.1186/1750-1172-2-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Meyers PA, Gorlick R. Osteosarcoma. Pediatr Clin North Am. 1997;44:973–989. doi: 10.1016/S0031-3955(05)70540-X. [DOI] [PubMed] [Google Scholar]
- 4.Dorfman HD, Czerniak B. Bone cancers. Cancer. 1995;75(Suppl):203–210. doi: 10.1002/1097-0142(19950101)75:1+<203::AID-CNCR2820751308>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 5.Wittig JC, Bickels J, Priebat D, Jelinek J, Kellar-Graney K, Shmookler B, Malawer MM. Osteosarcoma: A multidisciplinary approach to diagnosis and treatment. Am Fam Physician. 2002;65:1123–1132. [PubMed] [Google Scholar]
- 6.Ferguson WS, Goorin AM. Current treatment of osteosarcoma. Cancer Invest. 2001;19:292–315. doi: 10.1081/CNV-100102557. [DOI] [PubMed] [Google Scholar]
- 7.Ta HT, Dass CR, Choong PF, Dunstan DE. Osteosarcoma treatment: State of the art. Cancer Metastasis Rev. 2009;28:247–263. doi: 10.1007/s10555-009-9186-7. [DOI] [PubMed] [Google Scholar]
- 8.Marina N, Gebhardt M, Teot L, Gorlick R. Biology and therapeutic advances for pediatric osteosarcoma. Oncologist. 2004;9:422–441. doi: 10.1634/theoncologist.9-4-422. [DOI] [PubMed] [Google Scholar]
- 9.Schwarz R, Bruland O, Cassoni A, Schomberg P, Bielack S. The role of radiotherapy in oseosarcoma. Cancer Treat Res. 2009;152:147–164. doi: 10.1007/978-1-4419-0284-9_7. [DOI] [PubMed] [Google Scholar]
- 10.Anderson PM, Wiseman GA, Erlandson L, Rodriguez V, Trotz B, Dubansky SA, Albritton K. Gemcitabine radiosensitization after high-dose samarium for osteoblastic osteosarcoma. Clin Cancer Res. 2005;11:6895–6900. doi: 10.1158/1078-0432.CCR-05-0628. [DOI] [PubMed] [Google Scholar]
- 11.Bonner WM, Redon CE, Dickey JS, Nakamura AJ, Sedelnikova OA, Solier S, Pommier Y. GammaH2AX and cancer. Nat Rev Cancer. 2008;8:957–967. doi: 10.1038/nrc2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bassing CH, Alt FW. H2AX may function as an anchor to hold broken chromosomal DNA ends in close proximity. Cell Cycle. 2004;3:149–153. doi: 10.4161/cc.3.2.684. [DOI] [PubMed] [Google Scholar]
- 13.Mei L, Hu Q, Peng J, Ruan J, Zou J, Huang Q, Liu S, Wang H. Phospho-histone H2AX is a diagnostic and prognostic marker for epithelial ovarian cancer. Int J Clin Exp Pathol. 2015;8:5597–5602. [PMC free article] [PubMed] [Google Scholar]
- 14.van Attikum H, Gasser SM. Crosstalk between histone modifications during the DNA damage response. Trends Cell Biol. 2009;19:207–217. doi: 10.1016/j.tcb.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 15.Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–355. doi: 10.1038/nature02871. [DOI] [PubMed] [Google Scholar]
- 16.Penna E, Orso F, Cimino D, Vercellino I, Grassi E, Quaglino E, Turco E, Taverna D. miR-214 coordinates melanoma progression by upregulating ALCAM through TFAP2 and miR-148b downmodulation. Cancer Res. 2013;73:4098–4111. doi: 10.1158/0008-5472.CAN-12-3686. [DOI] [PubMed] [Google Scholar]
- 17.Wang ZS, Zhong M, Bian YH, Mu YF, Qin SL, Yu MH, Qin J. MicroRNA-187 inhibits tumor growth and invasion by directly targeting CD276 in colorectal cancer. Oncotarget. 2016;7:44266–44276. doi: 10.18632/oncotarget.10023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu Q, Yang G, Qian Y. Loss of MicroRNA-489-3p promotes osteosarcoma metastasis by activating PAX3-MET pathway. Mol Carcinog. 2017;56:1312–1321. doi: 10.1002/mc.22593. [DOI] [PubMed] [Google Scholar]
- 19.Wang J, He J, Su F, Ding N, Hu W, Yao B, Wang W, Zhou G. Repression of ATR pathway by miR-185 enhances radiation-induced apoptosis and proliferation inhibition. Cell Death Dis. 2013;4:e699. doi: 10.1038/cddis.2013.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jin YY, Chen QJ, Wei Y, Wang YL, Wang ZW, Xu K, He Y, Ma HB. Upregulation of microRNA-98 increases radiosensitivity in esophageal squamous cell carcinoma. J Radiat Res (Tokyo) 2016;57:468–476. doi: 10.1093/jrr/rrw068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang X, Li Q, Jin H, Zou H, Xia W, Dai N, Dai XY, Wang D, Xu CX, Qing Y. miR-424 acts as a tumor radiosensitizer by targeting aprataxin in cervical cancer. Oncotarget. 2016;7:77508–77515. doi: 10.18632/oncotarget.12716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang YH, Wang QQ, Li H, Ye T, Gao F, Liu YC. miR-124 radiosensitizes human esophageal cancer cell TE-1 by targeting CDK4. Genet Mol Res. 2016;15 doi: 10.4238/gmr.15027893. [DOI] [PubMed] [Google Scholar]
- 23.Song L, Liu S, Zhang L, Yao H, Gao F, Xu D, Li Q. MiR-21 modulates radiosensitivity of cervical cancer through inhibiting autophagy via the PTEN/Akt/HIF-1alpha feedback loop and the Akt-mTOR signaling pathway. Tumour Biol. 2016;37:12161–12168. doi: 10.1007/s13277-016-5073-3. [DOI] [PubMed] [Google Scholar]
- 24.Mao A, Zhao Q, Zhou X, Sun C, Si J, Zhou R, Gan L, Zhang H. MicroRNA-449a enhances radiosensitivity by downregulation of c-Myc in prostate cancer cells. Sci Rep. 2016;6:27346. doi: 10.1038/srep27346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lin SM, Xia Q, Zhang YQ, Sun AM, Shi YS, Zheng L, Chen LH. miR-124 regulates radiosensitivity of colorectal cancer cells by targeting PRRX1. Nan Fang Yi Ke Da Xue Xue Bao. 2016;36:1110–1116. (In Chinese) [PubMed] [Google Scholar]
- 26.Chen S, Wang Y, Ni C, Meng G, Sheng X. HLF/miR-132/TTK axis regulates cell proliferation, metastasis and radiosensitivity of glioma cells. Biomed Pharmacother. 2016;83:898–904. doi: 10.1016/j.biopha.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 27.Qian Z, Zhang L, Chen J, Li Y, Kang K, Qu J, Wang Z, Zhai Y, Li L, Gou D. MiR-328 targeting PIM-1 inhibits proliferation and migration of pulmonary arterial smooth muscle cells in PDGFBB signaling pathway. Oncotarget. 2016;7:54998–55011. doi: 10.18632/oncotarget.10714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Weng JH, Yu CC, Lee YC, Lin CW, Chang WW, Kuo YL. miR-494-3p induces cellular senescence and enhances radiosensitivity in human oral squamous carcinoma cells. Int J Mol Sci. 2016;17:E1092. doi: 10.3390/ijms17071092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Guo P, Lan J, Ge J, Nie Q, Guo L, Qiu Y, Mao Q. MiR-26a enhances the radiosensitivity of glioblastoma multiforme cells through targeting of ataxia-telangiectasia mutated. Exp Cell Res. 2014;320:200–208. doi: 10.1016/j.yexcr.2013.10.020. [DOI] [PubMed] [Google Scholar]
- 30.Ye C, Sun NX, Ma Y, Zhao Q, Zhang Q, Xu C, Wang SB, Sun SH, Wang F, Li W. MicroRNA-145 contributes to enhancing radiosensitivity of cervical cancer cells. FEBS Lett. 2015;589:702–709. doi: 10.1016/j.febslet.2015.01.037. [DOI] [PubMed] [Google Scholar]
- 31.Cheng DD, Yu T, Hu T, Yao M, Fan CY, Yang QC. MiR-542-5p is a negative prognostic factor and promotes osteosarcoma tumorigenesis by targeting HUWE1. Oncotarget. 2015;6:42761–42772. doi: 10.18632/oncotarget.6199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen L, Wang P, Yang J, Li X. MicroRNA-217 regulates WASF3 expression and suppresses tumor growth and metastasis in osteosarcoma. PLoS One. 2014;9:e109138. doi: 10.1371/journal.pone.0109138. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 33.Hu J, Lv G, Zhou S, Zhou Y, Nie B, Duan H, Zhang Y, Yuan X. The downregulation of miR-182 is associated with the growth and invasion of osteosarcoma cells through the regulation of TIAM1 expression. PLoS One. 2015;10:e0121175. doi: 10.1371/journal.pone.0121175. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 34.Gao Y, Feng Y, Shen JK, Lin M, Choy E, Cote GM, Harmon DC, Mankin HJ, Hornicek FJ, Duan Z. CD44 is a direct target of miR-199a-3p and contributes to aggressive progression in osteosarcoma. Sci Rep. 2015;5:11365. doi: 10.1038/srep11365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Niu G, Li B, Sun L, An C. MicroRNA-153 inhibits osteosarcoma cells proliferation and invasion by targeting TGF-β2. PLoS One. 2015;10:e0119225. doi: 10.1371/journal.pone.0119225. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 36.Fernandez-Capetillo O, Lee A, Nussenzweig M, Nussenzweig A. H2AX: The histone guardian of the genome. DNA Repair (Amst) 2004;3:959–967. doi: 10.1016/j.dnarep.2004.03.024. [DOI] [PubMed] [Google Scholar]
- 37.Stucki M, Jackson SP. gammaH2AX and MDC1: Anchoring the DNA-damage-response machinery to broken chromosomes. DNA Repair (Amst) 2006;5:534–543. doi: 10.1016/j.dnarep.2006.01.012. [DOI] [PubMed] [Google Scholar]
- 38.Pinder JB, Attwood KM, Dellaire G. Reading, writing, and repair: The role of ubiquitin and the ubiquitin-like proteins in DNA damage signaling and repair. Front Genet. 2013;4:45. doi: 10.3389/fgene.2013.00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liao XH, Zheng L, He HP, Zheng DL, Wei ZQ, Wang N, Dong J, Ma WJ, Zhang TC. STAT3 regulated ATR via microRNA-383 to control DNA damage to affect apoptosis in A431 cells. Cell Signal. 2015;27:2285–2295. doi: 10.1016/j.cellsig.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 40.Ikura M, Furuya K, Matsuda S, Matsuda R, Shima H, Adachi J, Matsuda T, Shiraki T, Ikura T. Acetylation of histone H2AX at Lys 5 by the TIP60 histone acetyltransferase complex is essential for the dynamic binding of NBS1 to damaged chromatin. Mol Cell Biol. 2015;35:4147–4157. doi: 10.1128/MCB.00757-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang Y, Huang JW, Li M, Cavenee WK, Mitchell PS, Zhou X, Tewari M, Furnari FB, Taniguchi T. MicroRNA-138 modulates DNA damage response by repressing histone H2AX expression. Mol Cancer Res. 2011;9:1100–1111. doi: 10.1158/1541-7786.MCR-11-0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang L, Yuan C, Lv K, Xie S, Fu P, Liu X, Chen Y, Qin C, Deng W, Hu W. Lin28 mediates radiation resistance of breast cancer cells via regulation of caspase, H2A.X and Let-7 signaling. PLoS One. 2013;8:e67373. doi: 10.1371/journal.pone.0067373. [DOI] [PMC free article] [PubMed] [Google Scholar]