Abstract
An anti-malarial transmission blocking vaccine (TBV) would be an important tool for disease control or elimination, though current candidates have failed to induce high efficacy in clinical studies. The ookinete surface protein P25 is a primary target for TBV development, but heterologous expression of P25 with appropriate conformation is problematic and a pre-requisite for achieving functional titers. A potential alternative to recombinant/sub-unit vaccine is immunization with a non-pathogenic, whole-parasite vaccine. This study examines the ability of a purified transgenic rodent-malaria parasite (PbPfs25DR3), expressing Plasmodium falciparum P25 in native conformation on the P. berghei ookinete surface, to act as a TBV. Vaccination with purified PbPfs25DR3 ookinetes produces a potent anti-Pfs25 response and high transmission-blocking efficacy in the laboratory, findings that are then translated to experimentation on natural field isolates of P. falciparum from infected individuals in Burkina Faso. Efficacy is demonstrated in the lab and the field (up to 93.3%/97.1% reductions in transmission intensity respectively), with both a homologous strategy with one and two boosts, and as part of a prime-boost regime, providing support for the future development of a whole-parasite TBV.
Introduction
To eradicate malaria, we must reduce disease within individuals, and inhibit transmission of Plasmodium through populations1. The dynamics of the malaria life-cycle indicate that transmission reduction will be most effective when targeting the parasite within the mosquito2,3. Anti-malarial transmission-blocking vaccines (TBVs) have shown promise as an effective means to reduce transmission. In this strategy, antibodies are ingested by the mosquito during a bloodmeal and prevent parasite development by binding to the surface proteins of gametocytes, gametes, zygotes/ookinetes4,5, or to mosquito-derived antigens expressed within the mosquito midgut (e.g. FREP16). A primary target for TBV development is the P25 protein, expressed on the surface of the zygote/ookinete7. The P25 family of proteins is characterized by the presence of EGF-like motifs, multiple cysteines, and a complex tertiary structure8, making them challenging to express with accurate native conformation. These proteins, whilst GPI anchored, are otherwise un-glycosylated in Plasmodium9, whereas eukaryotic expression systems typically glycosylate P25. Phase I trials of TBVs against Plasmodium falciparum P25 (Pfs25) have been performed previously10,11 and field trials are underway, with modest efficacy reported so far. This is widely hypothesised to be due to lack of correctly folded antigen with “correct” conformation/post-translational modification, and appropriate immunogenicity10–13.
To develop an effective TBV successfully, choosing a target antigen is only the first step. The need to express protein with native conformation and find a potent production/delivery system, complemented by appropriate formulation, adjuvant, dosages and immunization regimes is also vital. Various methods for P25 expression have been used in the past, including E. coli, L. lactis, Baculovirus, yeast, DNA administration and algae12–19. Delivery technologies such as virus-like/nano-particles have also been used20–23. Despite this, expression of recombinant P25, with appropriate glycosylation, conformation and antigenic presentation is problematic, and a potential pre-requisite for achieving high functional titers in humans. This is a clear technical bottleneck for successful vaccine development9,23–25.
An alternative to heterologous expression of recombinant/sub-unit based antigens is the use of whole-parasite vaccines, which naturally exhibit native expression of antigens. Typically, these vaccines use radiation or chemically-attenuated/killed parasites to induce an effective immune response. Although successfully demonstrated on a small scale in the 70 s26–29, this approach was initially deemed impractical and focus shifted to recombinant antigens. However, the success of researchers in recent years to manufacture/purify parasites to clinical grade and use them to develop vaccines that show protection in humans against infection has caused a re-evaluation30–35, generating clinical-grade, purified, irradiated or chemoattenuated sporozoites to act as potential pre-erythrocyte vaccines (PfSPZ). Trials of PfSPZ have demonstrated that whole-parasite vaccines, produced to GMP quality, can be well tolerated, immunogenic, and result in significant protection from infection in the lab36 and the field37. An alternative approach to induce P. falciparum sporozoite immunity is ongoing, using a transgenic rodent malaria lines (e.g. PbPfCS@UIS438,39). Whole-parasite vaccine approaches are also being extensively explored against the blood stages of the lifecycle, using chemically attenuated40, genetically attenuated41, irradiated42 or killed43 parasites.
Comparatively, efforts to generate a whole-parasite TBV have lagged. Previously, we have developed a transgenic rodent-malaria (Plasmodium berghei) line, PbPfs25DR3, expressing (human-malaria) P. falciparum Pfs25 in place of its rodent homologue (Pbs25). PbPfs25DR3 is phenotypically indistinguishable to WT P. berghei, expresses Pfs25 on the surface of the zygote/ookinete, and has been used previously to assay a range of TBVs16,17. Crucially, in this parasite, Pfs25 is expressed at high levels in native conformation, as demonstrated by functional complementation of its rodent homologue and the use of conformation dependent antibodies to assess expression16,17. The use of “humanized” rodent malaria parasites is recognized as a safe, cost-effective, and simple method to investigate the potency of anti-malarial vaccines16–19, however, their ability to induce a transmission-blocking response as part of a whole-organism vaccine has not been assessed. Immunization of these parasites will not act as a “traditional” anti-malarial whole parasite vaccine - ookinetes cannot establish infection in the vertebrate host. As a result, immunity due to the exploitation of natural infection of the host will not occur as with attenuated sporozoite or merozoite whole-parasite vaccines40–43. Conversely, the presentation of functionally competent Pfs25 antigen with proven native conformation is logically advantageous in terms of generation of a functional anti-parasitic immune response, and can potentially address concerns regarding the heterologous expression of malaria vaccine candidate antigens9,24,25.
Here, we have performed experiments to assess the ability of immunization with PbPfs25DR3 to initiate a transmission blocking response. A range of clinical adjuvants, doses and immunization regimes are examined; Alhydrogel, a common aluminium hydroxide wet-gel suspension is used as adjuvant, as is Matrix M, an adjuvant containing 40 nm nanoparticles composed of Quillaja saponins/cholesterol/phospholipid. Matrix M has previously demonstrated utility when used with Pfs25-based vaccine candidates23. Additionally, the ability of PbPfs25DR3 to act as “boost” within a “prime-boost” strategy was assessed, using a lead Pfs25 adenoviral vaccine (ChAd63-Pfs25) as the “prime” dose. Viral vectors have been shown to reliably induce functional antibodies against a range of malaria antigens in both pre-clinical and clinical studies in addition to being a reproducible means to induce cellular immunity. ChAd63 (chimpanzee adenovirus 63) has been used to express Pfs25 (and other TBVs previously), and when administered in a heterologous prime-boost regime, has elicited anti- parasitic moieties that are capable of blocking transmission in membrane-feeding assays16,17. Using these systems, we demonstrate that immunization with purified PbPfs25DR3 ookinetes elicits a potent anti-Pfs25 immune response, inducing antibodies that recognize native protein on the ookinete surface. We also demonstrate that PbPfs25DR3 immunization can initiate an anti-malarial transmission-blocking response with high efficacy, both in vivo in immunized mice, and by direct membrane feeding assay (DMFA), ex vivo at a range of infection densities, on blood samples from infected African blood donors. This data provides support for the future potential development of a whole-parasite TBV.
Results
Immunization with PbPfs25DR3 results in induction of anti-Pfs25 antibodies which recognize antigen in native conformation
In all regimes (Fig. 1), immunization with PbPfs25DR3 ookinetes results in induction of antibodies that recognize Pfs25 to varying degrees. To examine immunogenicity, end-point sera were examined by ELISA against recombinant (Pichia expressed) r-Pfs25. Sera were taken from individual mice (n = 5) and examined for anti-Pfs25 responses. Responses against r-Pfs25 were not detected when examining non-immunized serum. Immunization with purified WT ookinetes in all control regimes (7–8) resulted in no visible induction of anti-Pfs25 antibodies with any adjuvant/delivery systems tested here, suggesting a lack of cross-species induction of anti-P25 antibodies (against Pbs25 or Pbs28).
Figure 1.
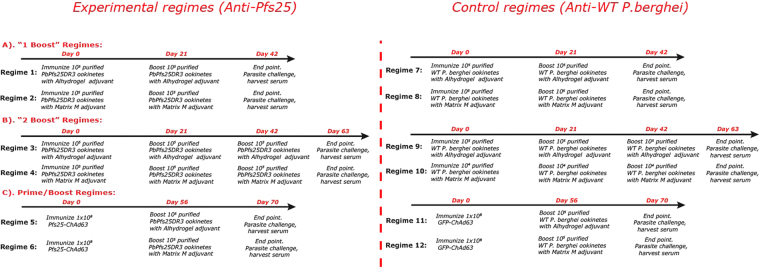
Anti-PbPfs25DR3 immunization regimes. Groups of 5 mice received each vaccine regime. For each individual experimental regime, the corresponding (negative) control regime is to its immediate right. In each regime, for DFA 5 mice were challenged with P. berghei PbPfs25DR3 to assess for transmission blockade. In regimes 1–6 mice were immunized to attempt to induce a Pfs25 response (‘experimental regimes’). In regimes 7–12 mice were immunized with carrier protein or empty vector controls (‘control regimes’). Regimes 1,2,7,8 use Matrix M as adjuvant, 3,4,9,10 use alhydrogel, 5,6,11,12 are prime/boost regimes, with ChAd63 prime and ookinete boost. All immunizations were performed i.m.
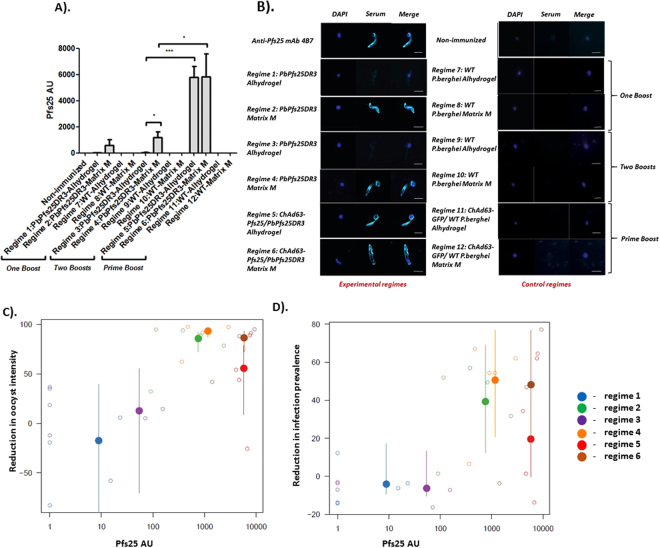
The lowest anti-Pfs25 antibody titers were observed when Alhydrogel was used as adjuvant (regimes 1 and 3) and higher titers were observed with Matrix M (regimes 2 and 4). When one boost was performed (1 and 2), no significant difference was observed in titer if either Alhydrogel or Matrix M was used. Conversely, if two boosts were performed (3 and 4), significantly higher titers were achieved with Matrix M (p = 0.0417) (Fig. 2A). For all regimes immunizing with ookinetes alone (in the absence of adenovirus), no significant difference in titer was observed between 1 and 2 boosts. Anti-Pfs25 responses rise dramatically when an adeno-prime/ookinete boost strategy is utilized. ChAd63-Pfs25-prime, PbPfs25DR3/Alhydrogel boost results in a significantly higher mean titer than the PbPfs25DR3/Alhydrogel 2 boost strategy (P = < 0.0001). A significant difference in titers is also observed when comparing PbPfs25DR3/Matrix M-2 boost and ChAd63-Pfs25-prime, PbPfs25DR3/Matrix M boost (p = 0.0241). Titers are equivalent to those previously observed with immunization with ChAd63-MVA-Pfs2517, but less than those observed with ChAd63-MVA-Pfs25-IMX31323. When a prime-boost strategy is performed, no significant difference in anti-Pfs25 titer is observed between Alhydrogel and Matrix M (regimes 5 and 6).
Figure 2.
Induction of antibody following immunization with PbPfs25DR3 ookinetes. The ability of each regime to generate Pfs25-specific antibody responses after administration was tested by ELISA against recombinant Pfs25 protein12,17 and IFA against P. falciparum (NF54) ookinete/retort stages within the mosquito midgut 26 hours post-feed. (A) End -point anti-Pfs25 titers in serum. Bars show mean titers from 5 mice. Pre-immune/non-immunized serum did not recognize recombinant Pfs25. Error bars represent SEM. Bars show mean titers from 5 mice. Error bars represent SEM. (B) IFA against P. falciparum (NF54) ookinete/retort stages. Ability of generated serum to recognize native Pfs25 on the surface of sexual stages of P. falciparum post-fertilization was assessed by immunofluorescence on fixed, non-permeabilized parasites probed with anti-serum from each regime. To control for non Pfs25-specific signal, IFA was performed using serum from control regimes (7–12). Each panel shows an overlay of anti-Pfs25 signal (turquoise) and DNA labelled with DAPI (blue). Scalebar = 5 µm. (C and D) The ability of induced titers to reduce both oocyst intensity (C) and infection prevalence (D) by DFA, and relationship with anti-Pfs25 titers. Small open circles denote estimates generated from mosquitoes fed on individual mice (Fig. 3) compared to the average from mosquitoes feeding on unimmunized mice) whilst the large filled dots show the average for the regimen as estimated using a generalized-linear mixed effect model. Vertical lines show 95% confidence interval on overall estimates. Point colours indicate the regimen tested, be it regime 1 (blue), 2 (green), 3 (purple), 4 (orange), 5 (red) or 6 (brown).
To confirm the induction of anti-Pfs25 antibodies that recognize native protein on the surface of the parasite, pooled end-point serum was used to perform IFA on mosquito-derived P. falciparum ookinetes/retorts. Parasite-specific surface staining corresponding to the established expression of Pfs25, and identical to that observed with anti-Pfs25 mAb 4B7, was observed using sera from regimes 2, 4, 5 and 6 (Fig. 2B), containing Matrix M as adjuvant. Conversely, only very low intensity signal was observed with regimes utilizing Alhydrogel. Control immunizations (7–12) resulted in no observable fluorescence.
Immunization with PbPfs25DR3 results in a potent transmission-blocking effect in vivo
To examine induced transmission blocking efficacy following immunization, 5 mice per regime were mechanically infected with P. berghei PbPfs25DR3, following which, direct feeding on individual mice was performed (Fig. 3). Adverse effects on rodent health were not observed following immunization with any regime. Specifically, pallor, piloerection, reduced mobility, lethargy/weakness, weight loss compared to cage mates, or respiratory distress was not observed in any immunized mice. Mean reductions in intensity and prevalence are reported in Table 1. In the control regimes (regimes 7–12), no significant transmission blocking efficacy was observed. The exception to this was observed with regime 10 (WT 2.34 Matrix M – 2 boosts), potentially due to the induction of non-Pfs25, uncharacterized P. berghei ookinete-specific transmission-blocking moieties under these conditions.
Figure 3.
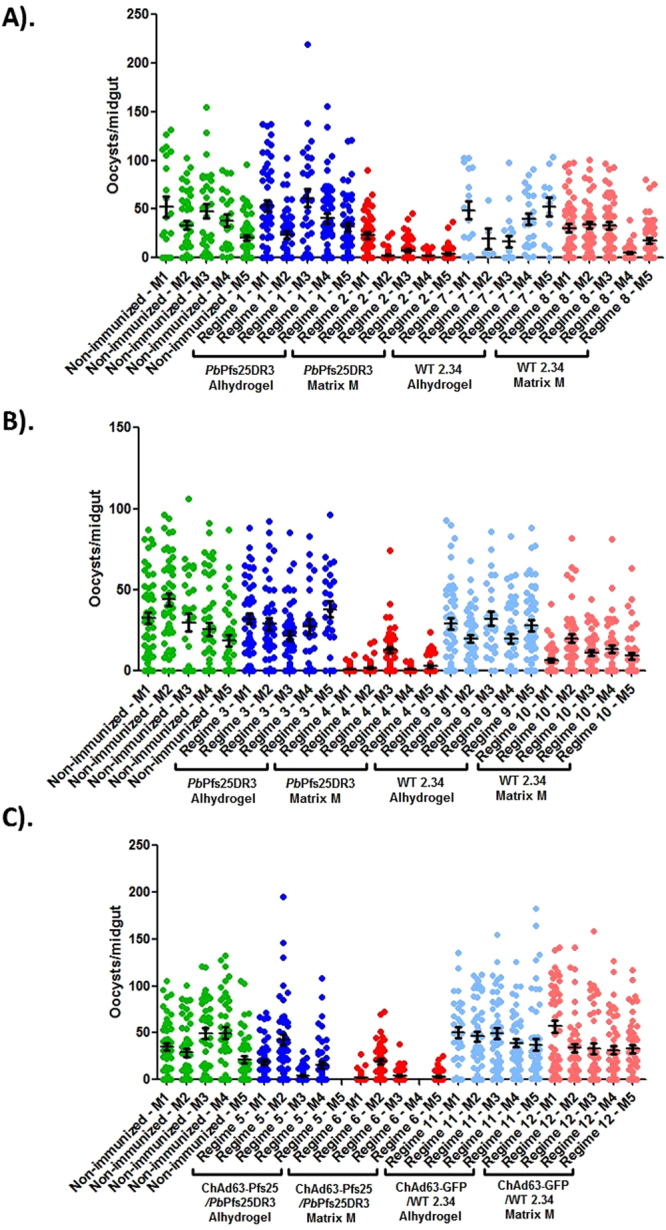
Assessment of in vivo transmission blockade following immunization with PbPfs25DR3 ookinetes by DFA. To assess transmission-blocking activity following immunization, 5 mice per regime (experimental and control) were infected/challenged with P. berghei PbPfs25DR3 and three days later, mosquitoes were exposed to individual mice to perform DFA. DFAs were performed in three individual tranches to account for varying experimental timings within multiple regimes (A) regimes 1,2,7,8; (B) regimes 3,4,9,10; (C) regimes 5,6,11,12). Transmission blockade was assessed as mean reduction in oocyst intensity/ prevalence with respect to cohorts of non-immunized challenged mice within each DFA. Regimes 5 and 6 contain four mice due to death before challenge. Individual data points represent the number of oocysts found in individual mosquitoes 12 days post feed. Horizontal bars indicate mean intensity, whilst error bars indicate S.E.M. For each regime, mean reductions in intensity and prevalence for each group are reported in Table 1.
Table 1.
Overall in vivo transmission-blockade following immunization with PbPfs25DR3 ookinetes by DFA.
| Regime | Total mice | Total mosquitoes | Mean oocyst intensity (±SEM) | Mean infection prevalence | Mean reduction in intensity | Mean reduction in prevalence |
|---|---|---|---|---|---|---|
| 1: PbPfs25DR3 Alhydrogel – 1 boost | 5 | 250 | 41.96 (5.6) | 83.2% | −17.4% | −4.1% |
| 2: PbPfs25DR3 Matrix M – 1 boost | 5 | 250 | 7.56 (1.1) | 48.4% | 85.7%*** | 39.3%*** |
| 3: PbPfs25DR3 Alhydrogel – 2 boost | 5 | 199 | 29.53 (3.8) | 85% | 12.8% | −6.3% |
| 4: PbPfs25DR3 Matrix M – 2 boost | 5 | 250 | 3.82 (0.7) | 50% | 93.3%*** | 50.6%*** |
| 5: ChAd63-Pfs25/PbPfs25DR3 Alhydrogel | 4 | 200 | 20.14 (3.3) | 62.5% | 55.7%*** | 19.6%*** |
| 6: ChAd63-Pfs25/PbPfs25DR3 Matrix M | 4 | 200 | 7.1 (1.3) | 42.5% | 86.5%*** | 48.2%** |
| 7: WT 2.34 Alhydrogel – 1 boost | 5 | 77 | 35.41 (8.3) | 87.6% | −5.3% | −5.4% |
| 8: WT 2.34 Matrix M – 1 boost | 5 | 250 | 24 (3.1) | 79.2% | 38.9% | −0.2% |
| 9: WT 2.34 Alhydrogel – 2 boost | 5 | 227 | 25.8 (3.3) | 80.1% | 24.1% | −0.2% |
| 10: WT 2.34 Matrix M – 2 boost | 5 | 250 | 12.01 (1.9) | 75.6% | 66.1%* | 4.2% |
| 11: ChAd63-GFP/ WT 2.34 Alhydrogel | 5 | 230 | 44.5 (5.5) | 91.6% | −33.2% | −14.9% |
| 12: ChAd63-GFP/ WT 2.34 Matrix M | 5 | 250 | 37.6 (5.1) | 83.6% | −10.6% | −5.2% |
Immunization with PbPfs25DR3/Alhydrogel results in no significant efficacy in terms of reduction in intensity or prevalence. Conversely, immunization with PbPfs25DR3/Matrix M preparations results in significant and potent levels of blockade, with an 85.7% reduction in intensity/39.3% reduction in prevalence observed with a single boost (regime 2). When the number of boosts is increased, reduction in intensity/prevalence of 93.3% and 50.6% respectively are achieved (regime 4). No significant difference between performing one boost or two boosts was observed when immunizing with PbPfs25DR3/Matrix M, in terms of inhibition of intensity (p = 0.072) or prevalence (p = 0.431). When immunizing PbPfs25DR3 with Alhydrogel or Matrix M as adjuvant as part of a “prime-boost” regime, significant efficacy is also observed. For regime 5 (ChAd63-Pfs25-prime/PbPfs25DR3 Alhydrogel-boost), reductions in intensity/prevalence of 55.7%/19.6% were achieved. This efficacy was considerably higher when using ChAd63-Pfs25-prime/PbPfs25DR3 Matrix M-boost (regime 6), with an 86.5%/48.2% reduction in intensity/prevalence observed. Significant differences between regime 2 (PbPfs25DR3/Matrix M 1 boost) or regime 4 (PbPfs25DR3/Matrix M 2 boosts) and regime 6 (ChAd63-Pfs25-prime/PbPfs25DR3 Matrix M-boost) were not observed, in terms of inhibition in intensity or prevalence. When considering blockade observed in individual immunized mice, transmission blocking efficacy is linked to induced anti-Pfs25 titer (Fig. 2C,D), with a significant relationship between reduction in intensity (p = 8.99 × 10−8) and prevalence (p = 2.56 × 10−6) and titer.
Serum derived from immunization with PbPfs25DR3 results in transmission-blocking efficacy against field isolates of P. falciparum
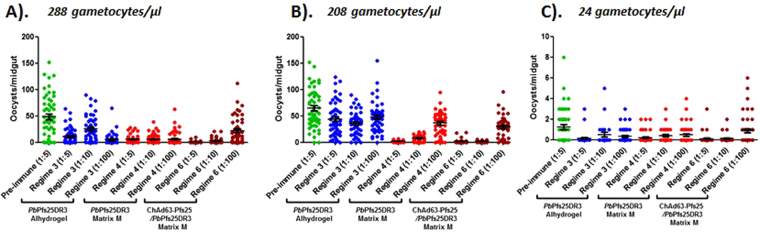
To assess ability to block transmission of field isolates, P. falciparum gametocytes were collected from naturally infected volunteers, and DMFA performed (Fig. 4, Table 2). Multiple gametocyte densities were assessed. Serum from regimes 3, 4 and 6 were examined at dilutions of 1:5, 1:10 and 1:100.
Figure 4.
Transmission blocking efficacy of serum derived from immunization with PbPfs25DR3 against field isolates of P. falciparum. To assess the ability of serum generated from regimes 3 (PbPfs25DR3/Alhydrogel 2 boosts), 4 (PbPfs25DR3/Matrix M 2 boosts) and 6 (ChAd63-Pfs25-prime/PbPfs25DR3 Matrix M-boost) to block transmission of malarial field isolates, P. falciparum gametocytes were collected from naturally infected volunteers recruited in malaria endemic localities, and DMFA subsequently performed. A range of naturally occurring gametocyte densities were assessed (A,B,C) were examined at dilutions of 1:5, 1:10 and 1:100, and control (pre-immune) serum at 1:5. Individual data points represent the number of oocysts found in individual mosquitoes 12 days post feed. Horizontal bars indicate mean intensity of infection, whilst error bars indicate S.E.M. within individual samples. For each regime, the mean reductions in intensity and prevalence at each dilution are reported in Table 2.
Table 2.
Overall transmission-blocking efficacy of serum derived from immunization with PbPfs25DR3 against field isolates of P. falciparum.
| PbPfs25DR3 Alhydrogel – 2 boost | PbPfs25DR3 Matrix M – 2 boost | ChAd63-Pfs25/PbPfs25DR3 Matrix M | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 in 5 | 1 in 10 | 1 in 100 | 1 in 5 | 1 in 10 | 1 in 100 | 1 in 5 | 1 in 10 | 1 in 100 | |
| Expt. A: | |||||||||
| Inhibition in intensity (%) | 77.3%*** | 47.8%** | 90.3%*** | 89.1%*** | 87.7%*** | 87.3%*** | 98.3%*** | 94.5%*** | 54.8%** |
| Inhibition in prevalence (%) | 9.5% | 0% | 38.1%*** | 19.1% | 26.2% | 42.0%* | 59.5%*** | 40.5%** | 4.7% |
| Expt. B: | |||||||||
| Inhibition in intensity (%) | 32.5%** | 45.6%*** | 27.0%* | 96.8%*** | 87.3%*** | 64.7%*** | 97.3%*** | 96.1%*** | 46.6%*** |
| Inhibition in prevalence (%) | 0% | 2.0% | −1.9% | 22.0% * | 13.7% | 4.7% | 46.0%*** | 26.1%** | 5.9% |
| Expt. C: | |||||||||
| Inhibition in intensity (%) | 89.0%*** | 59.4%** | 71.7%* | 83.1%*** | 68.5%* | 62.3%* | 91.5%*** | 90.8%*** | 29.7% |
| Inhibition in prevalence (%) | 89.1%*** | 50%** | 52.1%* | 70.8%** | 41.9% | 47.6%* | 87.5%*** | 83.3%*** | 7.7% |
| Overall inhibition: | |||||||||
| Inhibition in intensity (%) | 62.5%** | 46.9%* | 65.2%*** | 90.7%*** | 85.6%*** | 69.1%*** | 97.7%*** | 94.7%*** | 46.6%*** |
| Inhibition in prevalence (%) | 22.7%* | 9.3% | 32.0%*** | 35.6%*** | 28.9%** | 32.7%*** | 71.2%*** | 51.0%*** | 5.48% |
Intensity and prevalence were profoundly reduced by addition of immune serum, in comparison with feeds supplemented with pre-immune (control) serum. The highest reductions in intensity/prevalence were observed with regime 6, where a 97.7%/71.2% reduction was observed at a serum dilution of 1:5. Significant inhibition was demonstrated at lower dilutions. Feeds supplemented with serum from regime 4 also resulted in high levels of inhibition (90.7%/35.6%) at a dilution of 1:5, again, with significant inhibition demonstrated at lower dilutions. Across all dilutions, no significant difference between reduction in intensity or prevalence was observed when comparing regimes 4 or 6. Lower, but significant, levels of inhibition were seen using serum from regime 3 at dilutions of 1:5 and 1:100. Efficacy from regime 3 was lower than that observed with regimes 4 and 6, but higher than that observed with in vivo immunization/challenge in the DFA (Fig. 3). This is potentially due to higher than physiological concentrations of anti-Pfs25 moieties or reduced parasitic densities in the ex vivo DMFA, compared to the in vivo DFA.
Discussion
This study describes the establishment and assessment of a novel whole-parasite transmission blocking vaccine, using a transgenic rodent malaria parasite, PbPfs25DR316 expressing the human malaria antigen Pfs25, on the surface of the ookinete. PbPfs25DR3 expresses biologically competent Pfs25 with native conformation, evidenced by its ability to enable transmission in the absence of Pbs25 and Pbs28 (both P25 and P28 are mutually redundant, and loss of both proteins without appropriate complementation results in absence of transmission ex vivo and in vitro44). We demonstrate that following immunization with PbPfs25DR3, anti-Pfs25 responses are initiated, and that induced antibodies have Pfs25-approriate conformation, evidenced by their ability to recognize the ookinete/retort surface of P. falciparum. We show that immunization with purified PbPfs25DR3 ookinetes confers significant transmission-blocking immunity, assessed directly in vivo, and ex vivo, on field isolates of P. falciparum. As described in previous studies, transmission-blocking efficacy is broadly related to anti-Pfs25 titer45.
To assess the induction of transmission-blocking responses, we utilized a range of regimes, doses and adjuvants. Two clinically relevant adjuvants were assessed, Alhydrogel, and Matrix M. Additionally, the ability of PbPfs25DR3 to act as the “boost” for a “prime-boost” strategy was assessed, using ChAd63-Pfs25 as “prime”. Our DFA results indicate that regimes using Alhydrogel as adjuvant do not result in high efficacy, whereas Matrix M results in a potent effect, whether used with a single boost (regime 2), two boosts (regime 4) or as part of a prime-boost (regime 6). Maximal efficacy was achieved with regime 4, with a 93.3% reduction in intensity and 50.6% reduction in prevalence, but significant differences in efficacy between regimes 2, 4 and 6 were not detected. This study used inbred Balb/C mice for immunization. Whereas the inbred nature of this rodent model allows for a more direct, head-to-head comparison between immunizations with different antigenic preparations (variation due to outbred effects is reduced), this may impact on the predictive nature of these studies to human studies. Further studies examining effects in outbred models maybe advantageous. High efficacy was also seen by DMFA on field isolates of P. falciparum with Matrix M-derived serum. Within the DMFA, dose-responsive inhibition of transmission was observed with serum derived from regimes 4 and 6, but not with regime 5 (PbPfs25DR3 Alhydrogel – 2 boost).
The potential uses and applications of whole-parasite vaccines against malaria have been well discussed on multiple occasions24,25,46. In the case of PbPfs25DR3, there are numerous advantages to its potential development as a TBV. Pfs25 is expressed in this format in proven native conformation, resulting in induction of antibody that recognizes native protein on the surface of ookinetes. Given that Plasmodium lacks many common post-translational modifications, previous attempts to express Pfs25 in a heterologous system have led to potential issues with unwanted glycosylation, impacting on functional transmission blockade9,47. The ability of antigen to form appropriate disulphide bonds is also key. An as yet uncharacterized number of disulphide bonds are essential for the function of Pfs25 as an immunogen9. Using a transgenic rodent Plasmodium parasite to express Pfs25 circumvents these issues, while maintaining high titers and production of functional antibody.
There remain multiple technical and safety challenges relating to the use of whole-parasite vaccines in a clinical setting. A general concern is the possibility of underattenuation, or reversion to wild type46. The use of PbPfs25DR3 makes this impossible; firstly, the ookinete form of the parasite is mosquito exclusive, not transmitted, and is unable to establish/propagate bloodstage infection. Secondly, PbPfs25DR3 was constructed using double homologous recombination16, making spontaneous reversion impossible. Thirdly, even if reversion was to occur, P. berghei is not a human pathogen, and cannot establish bloodstage infection in humans, and finally, ookinetes are dead when immunized. The use of mouse-derived blood products in the manufacturing process is an issue when considering immunization with purified P. berghei parasites, potentially leading to alloimmunization. This must be thoroughly assessed prior to clinical trials. Here, ookinetes were prepared by CF11 purification to remove white blood cells, and lysis of erythrocytes (by NH4Cl), followed by magnetic purification to remove contaminants48. These methods lead to ookinete isolation with high purity (~99%)49. Previous MALDI-TOF analysis of these preparations show low levels of contamination50–52, typically by mouse haemoglobin-β52. It is unknown whether purification at this level is appropriate to avoid alloimmunization, however, if necessary, multiple purification methods could be combined, including density-gradients48,52, antibody-conjugation49 or enhanced chromatography53. Further examination is clearly required to minimize an inappropriate immune response, but murine-derived proteins (e.g. Zevalin, Bexxar) have been successfully utilized as therapeutics for decades. Production/purification of P. berghei ookinetes is cheap, robust, and routinely performed at HTP scale54, with yields of 12 × 106/ml, and proven scalability52. At the dose examined within this study (105 ookinetes immunized), a single ml of blood will provide ~120 individual immunizing doses, however, this scalability is only currently proven for experimental purposes. Clearly, further investigation would be needed to explore feasibility to scale production to support a vaccine pipeline, and to manufacture a viable vaccine to GMP standard. Experiments to assess the long-term persistence of functional antibody post-immunization would be desirable in the future. Generally, as described in multiple previous studies36–43, it is widely accepted that the manufacture of whole-parasite vaccines will require innovative approaches that are unconventional in “traditional” vaccinology25.
Within this study, to expand the repertoire of effective whole-parasite anti-malarial vaccines, we performed experiments using a safe, cheap, and easily cultured transgenic rodent-malaria parasite expressing a clinically-relevant TBV antigen (Pfs25) on its surface in native conformation. Vaccination produces an anti-Pfs25 immune response, and induces a potent response in vivo and ex vivo, in the lab and against field isolates of P. falciparum. High efficacy is demonstrated with a single boost, and without implementation of a prime-boost strategy. This data provides strong support for the further development and assessment of a whole-parasite TBV in the future.
Materials and Methods
General Parasite Maintenance
General maintenance of P. berghei ANKA 2.34 (WT) and PbPfs25DR3 parasites was carried out as described previously49,55. The generation, genotyping and phenotypic analysis of PbPfs25DR3, where endogenous Pbs25 (and Pbs28) was replaced with Pfs25, was previously described16.
Ookinete culture and purification
P. berghei ANKA 2.34 or PbPfs25DR3 ookinete culture and purification was performed as described in16. Purification of ookinetes was performed by passing cultures through a CF11 (Whatman) column to remove white blood cells, and erythrocytes were lysed by NH4Cl lysis16. Ookinetes were then purified on a VarioMACS Magnetic Cell Separator48,49 to remove contaminating cell debris. Purified ookinete preparations were aliquoted into individual doses and frozen to kill live parasites and preserve until immunization.
Immunization regimes
Sixty mice (Female BALB/c, 6–8 weeks of age (Harlan, UK)) were divided into twelve groups of 5 mice (Fig. 1). Groups 1–6 were experimental whereas groups 7–12 were immunized (i.m.) with control immunogens (either non-transgenic, WT 2.34 ookinetes to control for immunization with PbPfs25DR3 ookinetes, or ChAd63-GFP to control for immunization with ChAd63-Pfs2517. Transmission blockade observed following immunization with WT 2.34 ookinetes or ChAd63-GFP was non-specific, and not due to immunization with Pfs25.
For Alhydrogel vaccination, doses were prepared by combining 50 µL of Alhydrogel with 1 × 105 purified ookinetes. For Matrix M (Novavax), 1 × 105 ookinetes were mixed with adjuvant, vortexed and injected (12 µg Matrix M dose per mouse in total volume). Adenoviral vaccines were prepared in sterile, endotoxin-free PBS with doses of 1 × 108 viral particles of ChAd63 vaccines at day 0. Endpoint serum was collected for ELISA at day 42 for regimes 1, 2, 7 and 8; at day 63 for regimes 3, 4, 9 and 10; and at day 70 for regimes 5, 6, 11 and 12 (adeno-prime, ookinete-boost).
Enzyme-linked Immunosorbent Assay (ELISA)
Nunc-Immuno Maxisorp 96-well plates (NUNC) were coated with 100 ng recombinant protein per well (r-Pfs25, described in12,17). After blocking with 5% skimmed-milk, sera were incubated for 2 hours followed by goat anti-mouse IgG-AP (Sigma), 1:5000. Plates were developed using pNPP and read at OD405 until set end-point detection. Control regime titers were subtracted from mean end-point titers. Endpoint titer was defined as the x-axis intercept of the dilution curve at an absorbance value three standard deviations greater than the mean OD of pre-immune sera.
Immunofluorescence Assay (IFA)
The ability of sera to recognize native Pfs25 on the surface of the P. falciparum ookinete/retort was assessed by IFA on bloodmeal extracts from parasite-fed Anopheles gambiae mosquitoes 26 hrs post-feed. Briefly, P. falciparum (NF54) gametocytes were cultured as described previously, and fed to pots of >50 An. gambiae mosquitoes16. 26 hrs post feed, fed mosquitoes were anesthetized, and engorged midguts containing semi-digested blood removed. These preparations were washed to remove excess blood, macerated, smeared onto glass slides, and were fixed in 4% PFA in PBS. IFA was performed as described in17. Control experiments from pre-immune mouse serum, and mice immunized with WT 2.34 ookinetes in each respective regime were performed.
Direct Feeding Assay (DFA)
DFA was performed on immunized mice as described in55, with >50 Anopheles stephensi mosquitoes allowed to feed on each mouse. DFAs were performed in three tranches to account for varying experimental timings within regimes (Fig. 3A,B,C). Twelve days post-feed, midguts were dissected, and prevalence and intensity recorded. These values were compared to intensity and prevalence in non-immunized mice to assess inhibition of transmission.
DMFA
To determine efficacy against field isolates of P. falciparum, children aged between 5–11 years in Bobo-Dioulasso, Burkina Faso, were screened for the presence of P. falciparum by thick blood smears. Gametocytemias ranging from 288-24 per µl blood were harvested to examine wide range of parasitic densities. 10 mL blood was drawn in heparinized tubes to obtain gametocytes. The plasma from the gametocyte positive donor blood was replaced by AB+ serum from a European donor, test (or control) serum was added at the stated dilution, and fed to An. colluzi mosquitoes. Mosquitoes were dissected day 7 post-feeding. Intensity, prevalence, and reduction in both were calculated as described previously. Mean observed control intensity/prevalence values were 48.3/80.8% (experiment 1), 65.1/96.1% (experiment 2), 1.2/50% (experiment 3).
Ethical statement
All methods were carried out in accordance with relevant guidelines and regulations and were approved by the Imperial College Local Ethical Review Committee. Specifically, for animal studies, all procedures were performed in accordance with the UK Animals (Scientific Procedures) Act (UK Home Office License PPL 70/8788) and approved by Imperial College Animal Welfare and Ethical Review Body. For field/human studies, informed consent from all parents or guardians was obtained for children positive for gametocytes prior to blood sampling/DMFA (Protocol 003-2009/CE-CM, Centre Muraz institutional ethical committee).
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Statistical analysis
A regimen’s ability to reduce prevalence or intensity was assessed using generalized linear mixed effect models, GLMM56. GLMMs with and without antibody titre as a continuous linear effect were compared to investigate whether there was an association between titre and efficacy. Comparison between parametric ELISA tests were assessed using t-test. Statistical analysis was performed using R and Graphpad Prism. P values < 0.05 were considered statistically significant (*** = < 0.001, ** = 0.001–0.01, * = 0.01–0.05).
One Sentence Summary
A novel transgenic rodent malarial parasite induces potent transmission-blocking efficacy upon immunization.
Acknowledgements
We gratefully acknowledge Mark Tunnicliff for mosquito production, and Sumi Biswas for help with adenoviral constructs. This work was funded by a Confidence in Concept grant from the UK MRC (award number MC-PC_15028). A.M.B. thanks the MRC (New Investigator Research Grant; award number MR/N00227X/1) and PATH-MVI for funding. Funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Author Contributions
A.M.B. conceived the work. A.M.B. and F.A. acquired funding. Investigation performed by K.A.S., F.A., D.F.D., I.J.T. Data analysis by A.M.B. and T.S.C. Writing – original draft by A.M.B. Writing – review and editing by K.A.S., F.A., D.F.D., I.J.T., T.S.C., A.M.B.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.WHO World Malaria report. World Health Organisation, http://www.who.int/malaria/publications/world_malaria_report/en/ (2017).
- 2.Sinden RE. A biologist’s perspective on malaria vaccine development. Hum. Vaccin. 2010;6:3–11. doi: 10.4161/hv.6.1.9604. [DOI] [PubMed] [Google Scholar]
- 3.Rosenberg R. Malaria: some considerations regarding parasite productivity. Trends Paras. 2008;24:487–491. doi: 10.1016/j.pt.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 4.The malERA Refresh Consultative Panel on Tools for Malaria Elimination. An updated research agenda for diagnostics, drugs, vaccines, and vector control in malaria elimination and eradication. PLoS Med. 10.1371/journal.pmed.1002455. [DOI] [PMC free article] [PubMed]
- 5.Kaslow DC, Bathurst IC, Barr PJ. Malaria transmission-blocking vaccines. Trends Biotechnol. 1992;10(11):388–91. doi: 10.1016/0167-7799(92)90280-9. [DOI] [PubMed] [Google Scholar]
- 6.Niu G, et al. The fibrinogen-like domain of FREP1 protein is a broad-spectrum malaria transmission-blocking vaccine antigen. J Biol Chem. 2017;14(28):11960–11969. doi: 10.1074/jbc.M116.773564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vermeulen AN, et al. Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med. 1985;162:1460–76. doi: 10.1084/jem.162.5.1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kaslow DC, et al. A vaccine candidate from the sexual stage of human malaria that contains EGF-like domains. Nature. 1988;333:74. doi: 10.1038/333074a0. [DOI] [PubMed] [Google Scholar]
- 9.Lee SM, et al. Assessment of Pfs25 expressed from multiple soluble expression platforms for use as transmission-blocking vaccine candidates. Malaria Journal. 2016;15:405. doi: 10.1186/s12936-016-1464-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu Y, et al. Phase 1 trial of malaria transmission-blocking vaccine candidates Pfs25 and Pvs25 formulated with montanide ISA 51. PLoS ONE. 2008;3:e2636. doi: 10.1371/journal.pone.0002636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Talaat KR, et al. Safety and Immunogenicity of Pfs25-EPA/Alhydrogel, a Transmission Blocking Vaccine against Plasmodium falciparum: An Open Label Study in Malaria Naïve Adults. PLoS One. 2016;11(10):e0163144. doi: 10.1371/journal.pone.0163144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zou L, Miles AP, Wang J, Stowers AW. Expression of malaria transmission-blocking vaccine antigen Pfs25 in Pichia pastoris for use in human clinical trials. Vaccine. 2003;21(15):1650–7. doi: 10.1016/S0264-410X(02)00701-6. [DOI] [PubMed] [Google Scholar]
- 13.Kumar R, Angov E, Kumar N. Potent malaria transmission blocking antibody responses elicited by Plasmodium falciparum Pfs25 expressed in E.coli after successful protein refolding. Infect Immun. 2014;82(4):1453–1459. doi: 10.1128/IAI.01438-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Farrance CE, et al. Antibodies to plant-produced Plasmodium falciparum sexual stage protein Pfs25 exhibit transmission blocking activity. Hum Vaccin. 2011;7(Suppl):191–198. doi: 10.4161/hv.7.0.14588. [DOI] [PubMed] [Google Scholar]
- 15.Jones Chichester JA, et al. A Plant-Produced Pfs25 VLP Malaria Vaccine Candidate Induces Persistent Transmission Blocking Antibodies against Plasmodium falciparum in Immunized Mice. PLoS ONE. 2013;8(11):e79538. doi: 10.1371/journal.pone.0079538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Goodman AL, et al. A viral vectored prime-boost immunization regime targeting the malaria Pfs25 antigen induces transmission-blocking activity. PLoS One.. 2011;6(12):e29428. doi: 10.1371/journal.pone.0029428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kapulu MC, et al. Comparative assessment of transmission-blocking vaccine candidates against Plasmodium falciparum. Sci Rep. 2015;11(5):11193. doi: 10.1038/srep11193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blagborough AM, Yoshida S, Sattabongkot J, Tsuboi T, Sinden RE. Intranasal and intramuscular immunization with Baculovirus Dual Expression System-based Pvs25 vaccine substantially blocks Plasmodium vivax transmission. Vaccine. 2010;23(37):6014–20. doi: 10.1016/j.vaccine.2010.06.100. [DOI] [PubMed] [Google Scholar]
- 19.Blagborough AM, et al. Transmission blocking potency and immunogenicity of a plant-produced Pvs25-based subunit vaccine against Plasmodium vivax. Vaccine. 2016;14(28):3252–9. doi: 10.1016/j.vaccine.2016.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu Y, et al. Sustained high-titer antibody responses induced by conjugating a malarial vaccine candidate to outer-membrane protein complex. Proc Natl Acad Sci USA. 2006;103:18243–18248. doi: 10.1073/pnas.0608545103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shimp RL, Jr., et al. Development of a Pfs25-EPA malaria transmission blocking vaccine as a chemically conjugated nanoparticle. Vaccine. 2013;31:2954–2962. doi: 10.1016/j.vaccine.2013.04.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kumar R, et al. Nanovaccines for malaria using Plasmodium falciparum antigen Pfs25 attached gold nanoparticles. Vaccine. 2015;33(29):5064–5071. doi: 10.1016/j.vaccine.2015.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li, Y. et al. Enhancing immunogenicity and transmission-blocking activity of malaria vaccines by fusing Pfs25 to IMX313 multimerization technology. Sci Rep. 18848 (2016). [DOI] [PMC free article] [PubMed]
- 24.Hoffman SL, Vekemans J, Richie TL, Duffy PE. The March Toward Malaria Vaccines. Vaccine. 2015;27(33 Suppl 4):D13–23. doi: 10.1016/j.vaccine.2015.07.091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sauerwein RW, Richie TL. Malaria vaccines getting close to clinical reality. Vaccine. 2015;2(52):7423–4. doi: 10.1016/j.vaccine.2015.10.092. [DOI] [PubMed] [Google Scholar]
- 26.Clyde DF, Most H, McCarthy VC, Vanderberg JP. Immunization of man against sporozoite-induced falciparum malaria. Am J Med Sci. 1973;266:169–77. doi: 10.1097/00000441-197309000-00002. [DOI] [PubMed] [Google Scholar]
- 27.Rieckmann KH, Carson PE, Beaudoin RL, Cassells JS, Sell KW. Sporozoite induced immunity in man against an Ethiopian strain of Plasmodium falciparum. Trans R Soc Trop Med Hyg. 1974;68:258–9. doi: 10.1016/0035-9203(74)90129-1. [DOI] [PubMed] [Google Scholar]
- 28.Gwadz RW. Successful immunization against the sexual stages of Plasmodium gallinaceum. Science. 1976;193:1150–1. doi: 10.1126/science.959832. [DOI] [PubMed] [Google Scholar]
- 29.Carter R, Chen DH. Malaria transmission blocked by immunisation with gametes of the malaria parasite. Nature. 1976;263:57–60. doi: 10.1038/263057a0. [DOI] [PubMed] [Google Scholar]
- 30.Hoffman SL, et al. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J Infect Dis. 2002;185:1155–64. doi: 10.1086/339409. [DOI] [PubMed] [Google Scholar]
- 31.Luke TC, Hoffman SL. Rationale and plans for developing a non-replicating, metabolically active, radiation-attenuated Plasmodium falciparum sporozoite vaccine. J Exp Biol. 2003;206:3803–8. doi: 10.1242/jeb.00644. [DOI] [PubMed] [Google Scholar]
- 32.Luke TC, Hoffman SL. Rationale and plans for developing a non-replicating, metabolically active, radiation-attenuated Plasmodium falciparum sporozoite vaccine. J Exp Biol. 2003;206:3803–8. doi: 10.1242/jeb.00644. [DOI] [PubMed] [Google Scholar]
- 33.Seder RA, et al. Protection against malaria by intravenous immunization with a nonreplicating sporozoite vaccine. Science. 2013;341:1359–65. doi: 10.1126/science.1241800. [DOI] [PubMed] [Google Scholar]
- 34.Roestenberg M, et al. Protection against a malaria challenge by sporozoite inoculation. N Engl J Med. 2009;361:468–77. doi: 10.1056/NEJMoa0805832. [DOI] [PubMed] [Google Scholar]
- 35.Roestenberg M, et al. Long-term protection against malaria after experimental sporozoite inoculation: an open-label follow-up study. Lancet. 2011;377:1770–6. doi: 10.1016/S0140-6736(11)60360-7. [DOI] [PubMed] [Google Scholar]
- 36.Mordmüller B, et al. Sterile protection against human malaria by chemoattenuated PfSPZ vaccine. Nature. 2017;542(7642):445–449. doi: 10.1038/nature21060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sissoko MS, et al. Safety and efficacy of PfSPZ Vaccine against Plasmodium falciparum via direct venous inoculation in healthy malaria-exposed adults in Mali: a randomised, double-blind phase 1 trial. Lancet Infect Dispii. 2017;S1473-3099(17):30104–4. doi: 10.1016/S1473-3099(17)30104-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lin JW, et al. A novel 'gene insertion/marker out' (GIMO) method for transgene expression and gene complementation in rodent malaria parasites. PLoS One. 2011;6(12):e29289. doi: 10.1371/journal.pone.0029289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.MVI-PATH portfolio; http://www.malariavaccine.org/projects/vaccine-projects/transgenic-p-berghei (2016).
- 40.Good MF, et al. Cross-species malaria immunity induced by chemically attenuated parasites. J Clin Invest. 2013;123:3353–62. doi: 10.1172/JCI66634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ting LM, Gissot M, Coppi A, Sinnis P, Kim K. Attenuated Plasmodium yoelii lacking purine nucleoside phosphorylase confer protective immunity. Nat Med. 2008;14:954–8. doi: 10.1038/nm.1867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.McCarthy JS, Good MF. Whole parasite blood stage malaria vaccines: a convergence of evidence. Hum Vaccin. 2010;6:114–23. doi: 10.4161/hv.6.1.10394. [DOI] [PubMed] [Google Scholar]
- 43.Pinzon-Charry A, et al. Low doses of killed parasite in CpG elicit vigorous CD4+ T cell responses against blood-stage malaria in mice. J Clin Invest. 2010;120:2967–78. doi: 10.1172/JCI39222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tomas AM, et al. P25 and P28 proteins of the malaria ookinete surface have multiple and partially redundant functions. EMBO J. 2001;20(15):3975–83. doi: 10.1093/emboj/20.15.3975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Miura K, et al. Transmission-blocking activity induced by malaria vaccine candidates Pfs25/Pvs25 is a direct and predictable function of antibody titer. Malar J. 2007;6:107. doi: 10.1186/1475-2875-6-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stanisic DI, Good MF. Whole organism blood stage vaccines against malaria. Vaccine. 2015;33(52):7469–7475. doi: 10.1016/j.vaccine.2015.09.057. [DOI] [PubMed] [Google Scholar]
- 47.Tsai CW, Duggan PF, Shimp RL, Jr, Miller LH, Narum DL. Overproduction of Pichia pastoris or Plasmodium falciparum protein disulfide isomerase affects expression, folding and O-linked glycosylation of a malaria vaccine candidate expressed in P. pastoris. J Biotechnol. 2006;121(4):458–7. doi: 10.1016/j.jbiotec.2005.08.025. [DOI] [PubMed] [Google Scholar]
- 48.Carter V, Cable HC, Underhill AB, Jackie Williams J, Hurd H. Isolation of Plasmodium berghei ookinetes in culture using Nycodenz density gradient columns and magnetic isolation. Malaria Journal. 2003;2:35. doi: 10.1186/1475-2875-2-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ramakrishnan C, et al. Laboratory maintenance of rodent malaria parasites. Methods Mol Biol. 2013;923:51–72. doi: 10.1007/978-1-62703-026-7_5. [DOI] [PubMed] [Google Scholar]
- 50.Florens L, et al. A proteomic view of the Plasmodium falciparum life cycle. Nature. Oct. 2002;3(6906):520–6. doi: 10.1038/nature01107. [DOI] [PubMed] [Google Scholar]
- 51.Hall N, et al. A comprehensive survey of the Plasmodium life cycle by genomic, transcriptomic, and proteomic analyses. Science. 2005;7(5706):82–6. doi: 10.1126/science.1103717. [DOI] [PubMed] [Google Scholar]
- 52.Lal K, et al. Characterisation of Plasmodium invasive organelles; an ookinete microneme proteome. Proteomics. 2009;9(5):1142–51. doi: 10.1002/pmic.200800404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Heidrich HG, Mrema JE, Vander Jagt DL, Reyes P, Rieckmann KH. Isolation of intracellular parasites (Plasmodium falciparum) from culture using free-flow electrophoresis: separation of the free parasites according to stages. J Parasitol. Jun. 1982;68(3):443–50. doi: 10.2307/3280956. [DOI] [PubMed] [Google Scholar]
- 54.Delves MJ, et al. A high-throughput assay for the identification of malarial transmission-blocking drugs and vaccines. Int J Parasitol. Oct. 2012;42(11):999–1006. doi: 10.1016/j.ijpara.2012.08.009. [DOI] [PubMed] [Google Scholar]
- 55.Blagborough AM, et al. Assessing transmission blockade in Plasmodium spp. Methods Mol Biol. 2013;923:577–600. doi: 10.1007/978-1-62703-026-7_40. [DOI] [PubMed] [Google Scholar]
- 56.Churcher TS, et al. Measuring the blockade of malaria transmission - An analysis of the Standard Membrane Feeding Assay. Int J Parasitol. 2012;42(11):1037–44. doi: 10.1016/j.ijpara.2012.09.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.