Abstract
Purpose
BCR-ABL+ B-ALL leukemic cells are highly dependent on the expression of endogenous anti-apoptotic MCL-1 to promote viability and are resistant to BH3-mimetic agents such as navitoclax (ABT-263) that targets BCL-2, BCL-XL, and BCL-W. However, the survival of most normal blood cells and other cell types are also dependent on Mcl-1. Despite the requirement for MCL-1 in these cell types, initial reports of MCL-1-specific BH3-mimetics have not described any overt toxicities associated with single-agent use, but these agents are still early in clinical development. Therefore, we sought to identify FDA-approved drugs that could sensitize leukemic cells to ABT-263.
Experimental Design
A screen identified dihydroartemisinin (DHA), a water-soluble metabolite of the anti-malarial artemisinin. Using mouse and human leukemic cell lines, and primary patient-derived xenografts, the effect of DHA on survival was tested and mechanistic studies were carried out to discover how DHA functions. We further tested in vitro and in vivo whether combining DHA with ABT-263 could enhance the response of leukemic cells to combination therapy.
Results
DHA causes the down-modulation of MCL-1 expression by triggering a cellular stress response that represses translation. The repression of MCL-1 renders leukemic cells highly sensitive to synergistic cell death induced by ABT-263 in a mouse model of BCR-ABL+ B-ALL both in vitro and in vivo. Furthermore, DHA synergizes with ABT-263 in human Ph+ ALL cell lines, and primary patient derived xenografts of Ph+ ALL in culture.
Conclusions
Our findings suggest that combining DHA with ABT-263 can improve therapeutic response in BCR-ABL+ B-ALL.
Introduction
One in five pediatric patients with acute lymphoblastic leukemia (ALL) are diagnosed with poor prognosis disease (including BCR-ABL+ ALL) (1). Adults with ALL have even poorer survival rates (<40%) in part due to the dominant role of the BCR-ABL oncogene (2). In Philadelphia chromosome (Ph+) leukemia, BCR-ABL fusion proteins encode constitutively-active tyrosine kinases (e.g. p185 in B-ALL and p210 in chronic myelogenous leukemia, CML) essential for cell transformation (3, 4). CML therapy has been revolutionized by the BCR-ABL tyrosine kinase inhibitors (TKIs), which induce and maintain remission without serious side effects (5). In contrast, TKI treatment of Ph+ BALL results in short remissions and rapid outgrowth of TKI-resistant clones (6, 7). As a result, Ph+ ALL patients require intensive chemotherapy and stem cell transplants (1). Thus, new therapies are needed to improve patient outcomes.
Leukemic cells often feature the elevated expression of anti-apoptotic molecules that inhibit cell death by preventing BAX and BAK-dependent mitochondrial outer membrane permeabilization and caspase activation. Small molecule inhibitors, known as BH3-mimetics, including nativoclax (ABT-263) and venetoclax (ABT-199), have beendeveloped to inhibit the anti-apoptotic molecules BCL-2, BCL-XL, BCL-W or only BCL-2 respectively (8, 9). These agents are effective because malignant cells often become addicted to the function of anti-apoptotic BCL-2 members to permit their survival, thus making them more sensitive to inhibitors of this pathway than normal cells (10). ABT-199 is approved for treatment of some chronic lymphocytic leukemia with a 17p deletion. In contrast, ABT-263 is still in development with the caveat that it provokes a dose-limiting thrombocytopenia due to the on-target effect of inhibiting BCL-XL in platelets (11). While these BH3-mimetics show promise in treating malignancies, elevated anti-apoptotic MCL-1 expression represents a common resistance mechanism (12–15). MCL-1 levels are commonly elevated in cancer, but potent MCL-1 inhibitors are still in development (16, 17). MCL-1 is unique among pro-survival molecules in that it is essential for development and for the survival of many cellular lineages (18). Hence, it is still unclear whether effective MCL-1 inhibition during standard cancer therapy will result in toxic side effects in humans.
Here, we report that treatment of mouse BCR-ABL+ B-ALL with the orally-available, anti-malarial drug, dihydroartemisinin (DHA) reduces MCL-1 expression by inducing a CHOP-dependent cellular stress response. By repressing MCL-1, DHA sensitizes leukemia to synergistic treatment with BH3-mimetics such as ABT-263 both in vitro and in vivo. Similarly, in human Ph+ leukemic cells and primary patient samples, combining DHA with ABT-263 triggers synergistic leukemia killing, revealing a novel combinatorial therapy to treat poor prognosis BCR-ABL+ leukemia.
Materials and Methods
Plasmids, Expression Constructs, and Generation of Mutants
BCR-ABL (p185) plasmid was from Dr. Witte (UCLA, CA) and BCR-ABLT315I mutant was generated by site-directed mutagenesis. Murine Mcl-1 cDNA was stably expressed in leukemic cells by retroviral transduction.
Ecotropic Retroviral Production and Cell Transduction
Retroviruses were produced as previously described (19). Stable cells were generated by retroviral transduction and drug selection or by fluorescent activated cell sorting.
Cells and Cell Culture
Genetically-engineered mouse (GEM) p185+ or p185T315IAr−/− or p53−/− B-ALL were grown in RPMI with 10% fetal bovine serum, 55 μM 2-mercaptoethanol, 2 mM glutamine, penicillin, and streptomycin. SV40-transformed wild-type, Noxa-deficient and CHOP-deficient murine embryonic fibroblasts (MEFs) were cultured in DMEM with 10% fetal bovine serum, 55 μM 2-mercaptoethanol, 2 mM glutamine, and gentamycin (20, 21). ABT-263 was provided by AbbVie, IL. DHA was from AvaChem Scientific (San Antonio, TX). Human Ph+ leukemia cell lines, OP-1, TOM-1, SUP-B15, and BV-173 were grown in RPMI with 20% fetal bovine serum, 55 μM 2-mercaptoethanol, 2 mM glutamine, penicillin, and streptomycin.
Immunoblotting and Antibodies
Protein expression was assessed as previously described (19). Antibodies used were: anti-MCL-1 (Rockland Immunochemical, PA), anti-human MCL-1, anti-PARP, anti-CHOP, and anti-BCL-XL (Cell Signaling, MA). Anti-BCL-2 (BD Biosciences, CA), anti-NOXA and anti-PUMA (Sigma Aldrich, MO), and anti-Actin (Millipore, MA). Anti-rabbit or anti-mouse horseradish peroxidase-conjugated secondary antibodies were from Jackson Immunochemical, ME. Immunoblots were acquired on a LI-COR Odyssey (LI-COR, NB) and densitometry assessed for all blots in replicate for statistical analysis (Sup. Fig. 7).
Microarray Analysis and GSEA
Affymetrix Mouse Gene 2.0 ST arrays were used to assay RNA samples. Data was submitted to NIH GEO Accession number: GSE95809. The 4-hour DHA treated samples were statistically tested by unequal variance t test to the DMSO control (3 replicates) and the volcano plot was generated by converting the resultant pvalue to a score (−log10(pvalue)) using Spotfire (TIBCO, CA). Gene set enrichment analysis (GSEA) (version 2.2.3) was performed using 4-hour DHA samples vs. DMSO control samples and the c5.all.v5.2 symbol.gmt Gene Ontology gene sets. Gene set permutation was performed 1000 times using the weighted enrichment statistic and genes ranked by signal to noise metric.
Cell Death Experiments
Cells (6×104) were seeded in 96-well plates and DHA and ABT-263 (solubilized in DMSO or DMSO vehicle controls) added at indicated concentrations. After 24 hours, apoptotic cells were determined by staining with Annexin-V-APC and propidium iodide (BD Biosciences) and measured by flow cytometry.
Real-time PCR
RNA extracted using Ambion RNA Extraction Kit (Life Technologies, CA) was reverse transcribed with SuperScript III (Life Technologies). Real-time PCR was performed using primers and Fast SYBR-green (Thermo Fisher, MA). Data were analyzed by the ΔΔCt method in a Quantstudio7 Flex real-time PCR machine (Thermo Fisher) with housekeeping gene (Ubiquitin) and compared to unstimulated cells. Primer sequences are available by request.
Response Surface Modeling
Response surface modeling, implemented in Matlab version R2016a (Mathworks, MA), was used to determine changes in the response on viable cells (Annexin-V− and PI−) of two drugs given alone and in combination (22–24). A drug combination was considered either synergistic or antagonistic if the interaction term (α) describing the change in response relative to the additive model was either positive or negative, respectively.
CRISPR/Cas9 Mutagenesis of Mouse BCR-ABL+ B-ALL cells
Ddit3−/− murine BCR-ABL+ B-ALL cells were generated using CRISPR-Cas9 technology. Briefly, 4×105 BALL cells were transiently co-transfected with 1 μg of gRNA (5′ GACACCGTCTCCAAGGTGAA 3′) and 1 μg Cas9 expression plasmid via nucleofection (Lonza, 4D-NucleofectorTM X-unit) using solution SF, program CA-137 in small cuvettes according to the manufacturers recommended protocol. Cells were single cell sorted by flow cytometry, clonally selected and verified for disruption of the endogenous locus via targeted deep sequencing to identify frameshift mutations.
Generation of Patient-Derived Xenograft (PDX) Mice
Leukemia from adult patients with BCR-ABL1+ ALL obtained from the Eastern Cooperative Oncology Group E2993 study (ClinicalTrials.gov identifier NCT00002514) and from the University Health Network, Toronto, CA. were transplanted into un-irradiated immunodeficient NOD.Cg-PrkdcscidIl2rgtm1Wjl/SzJ (NSG) mice (Jackson Laboratories, ME) for 8–10 weeks prior to re-isolation (25–27). Mice were bred and utilized in accordance with St. Jude Children’s Research Hospital animal care and use committee (SJCRHACUC).
Treatment of Murine Leukemia in Recipient Mice
Mouse BCR-ABL+ Arf−/− Luciferase+ B-ALL cells were injected (2×105) into non-conditioned, 6–8 week-old, female C57BL/6 recipients (Jackson Laboratory). Five days after the transfer, recipients were treated with DHA and ABT-263 by oral gavage. ABT-263 was formulated in a mixture of 60% Phosal 50 PG, 30% PEG 400, and 10% EtOH and dosed at 100 mg/kg/day as previously described (28). DHA was formulated in 0.5% carboxymethylcellulose, 0.5% Tween-80, and 0.5% benzyl alcohol and dosed at 200 mg/kg/day. Treatment was given daily for 15 days (days 5–20) during and after which the mice were monitored. Mice were bred and utilized in accordance with SJCRHACUC. Bioluminescence imaging was assessed by Xenogen IVIS (Perkin Elmer, MA) after mice were injected with D-luciferin (Perkin Elmer) at 150 mg/kg. Images (photons/second) were quantified through application of a contour drawn around the target region and normalized to maximum luminescence activity. For ex vivo analysis of MCL-1 expression, recipient mice 10 days after transplant of 2×105 mouse BCR-ABL+ B-ALL cells were treated with vehicle or DHA (200 mg/kg) by gavage. Four or 8 hours after treatment, splenic blast cells were isolated and subjected to immunoblotting.
Results
Dihydroartemisinin (DHA) induces apoptosis in BCR-ABL+ B-ALL cells
Using a genetically-engineered mouse (GEM) model for BCR-ABL+ B-lineage acute lymphoblastic leukemia (hereafter referred to as BCR-ABL+ B-ALL) we previously demonstrated that endogenous MCL-1 is required to maintain leukemic cell survival (15). This is a powerful model to interrogate the biology of human, poor-prognosis BCR-ABL+ B-ALL (15, 29). While MCL-1 is clearly an important therapeutic target, potent and selective MCL-1 inhibitors are still in development and have only recently entered human Phase I trials (17). Therefore, we sought to identify alternative strategies by which MCL-1 function or expression can be attenuated to render BCR-ABL+ B-ALL cells susceptible to apoptosis induced by currently available BH3-mimetic small molecules.
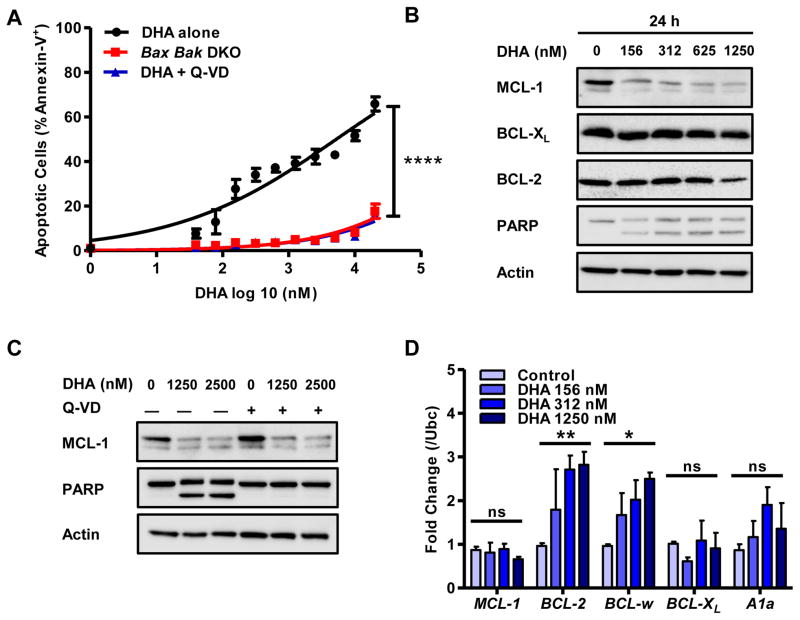
A library of approved drugs were screened to identify compounds that killed mouse BCR-ABL+ B-ALL leukemic cells (30). This screen identified members of the artemisinin class of anti-malarial agents including dihydroartemisinin (DHA), a widely-used, orally-delivered drug for malaria with favorable pharmacokinetics and bioavailability in humans (31). DHA posseses anti-cancer properties; however, the mechanism(s) by which DHA functions to kill cancer cells is unclear (32–36). Treatment of mouse BCR-ABL+ B-ALL cells with DHA induced apoptosis (Fig. 1A & Sup. Fig. 1A). Consistent with the induction of apoptosis, the leukemic cells responded to DHA treatment by cleaving poly ADP-ribose polymerase (PARP) (Fig. 1B & Sup. Fig. 1B). Caspase inhibitors (e.g. Q-VD) or treatment of Bax and Bak-deficient (hereafter DKO) BCR-ABL+ B-ALL mouse cells with DHA repressed the induction of leukemic apoptosis and cleavage of PARP (Fig. 1A,C & Sup. Fig. 1C).
Figure 1. Dihydroartemisinin (DHA) induces apoptosis and represses MCL-1 expression in Mouse BCR-ABL+ B-ALL Cells.
(A) BCR-ABL+ B-ALL wild-type and Bax and Bak-doubly deficient (DKO) cells were treated with indicated doses of DHA and the caspase inhibitor Q-VD (10 μM) for 24 hours. Apoptotic cells were determined by staining with Annexin-V and detection by flow cytometry. Data are average of 3 independent experiments (n=3) and error bars the S.E.M. One-way ANOVA with Bonferroni multiple comparison indicates significance p<0.001**** (B) BCR-ABL+ BALL cells were exposed to indicated doses of DHA for 24 hours and subjected to immunoblotting for expression of indicated proteins. Cleaved PARP detects the induction of apoptosis and Actin serves as loading control. (C) BCR-ABL+ B-ALL cells pretreated with Q-VD (10 μM) for 1 hour, followed by treatment with indicated doses of DHA for 24 hours and immunoblotted for indicated proteins. (D) BCR-ABL+ B-ALL were treated with indicated doses of DHA for 24 hours and the expression of anti-apoptotic BCL-2 family members was determined by quantitative PCR. Each measurement was performed from three biological replicates (in triplicate), normalized to Ubiquitin (Ubc), and compared to untreated cells. The average fold change is indicated and error bars the S.E.M. Two-way ANOVA with Bonferroni multiple comparison indicates significance p<0.05* and p<0.01**.
DHA represses MCL-1 expression in murine BCR-ABL+ B-ALL cells
Treatment of mouse BCR-ABL+ B-ALL cells with DHA, at significantly lower doses than those required for cytotoxicity, produced a loss of MCL-1 expression, but the expression levels of BCL-XL, BCL-2 were only marginally affected (Fig. 1B & Sup. Fig. 1B). The loss of MCL-1 expression was still observed in DHA-treated cultures when cell death was blocked by caspase inhibitors (Q-VD) or in mouse DKO BCR-ABL+ B-ALL cells (Fig. 1C & Sup. Fig. 1C). Therefore, the decline of MCL-1 expression is independent of caspase activation and BAX/BAK-dependent mitochondrial permeabilization. Diminished MCL-1 expression was detectable as early as 8 hours after DHA treatment, preceding evidence of apoptosis (Sup. Fig. 1B). While DHA treatment may affect a variety of cellular pathways at high concentration to induce single agent killing, overexpression of anti-apoptotic MCL-1 rendered mouse BCR-ABL+ B-ALL cells more resistant to DHA treatment as expected (Sup. Fig. 1D&E).
DHA results in post-transcriptional repression of MCL-1 expression
MCL-1 is a labile protein regulated at many levels including transcription, translation, and protein degradation by the proteasome (37). To determine mechanistically how MCL-1 expression is repressed by DHA, RNA expression of anti-apoptotic BCL-2 family members was assessed by quantitative PCR in mouse BCR-ABL+ B-ALL cells treated with DHA in vitro. In response to DHA treatment, pro-survival BCL-2 family member expression was either unchanged or induced and notably MCL-1 mRNA expression was unaffected (Fig. 1D). When protein translation was assessed by pulsing mouse BCR-ABL+ B-ALL cells with 35S-containing media, the treatment of DHA substantially repressed new protein synthesis of many cellular proteins including MCL-1 (Sup. Fig. 2A). MCL-1 is a well-recognized labile protein and cellular signaling has been reported to modulate its turnover by the proteasome (19, 38). However, when mouse BCR-ABL+ B-ALL cells were pre-treated with DHA and then pulsed with cycloheximide to attenuate new MCL-1 protein synthesis, MCL-1 protein was eliminated at the same rate whether cultured with DHA or DMSO (Sup. Fig. 2B). Unsurprisingly, the loss of MCL-1 protein expression triggered by DHA treatment could be rescued by treatment with the proteasome inhibitor bortezomib (Sup. Fig. 2C). These data indicate that DHA treatment does not attenuate MCL-1’s transcription, but rather inhibits protein translation leading to MCL-1’s elimination by the proteasome in mouse BCR-ABL+ B-ALL cells.
DHA induces a cellular stress program that represses MCL-1 expression
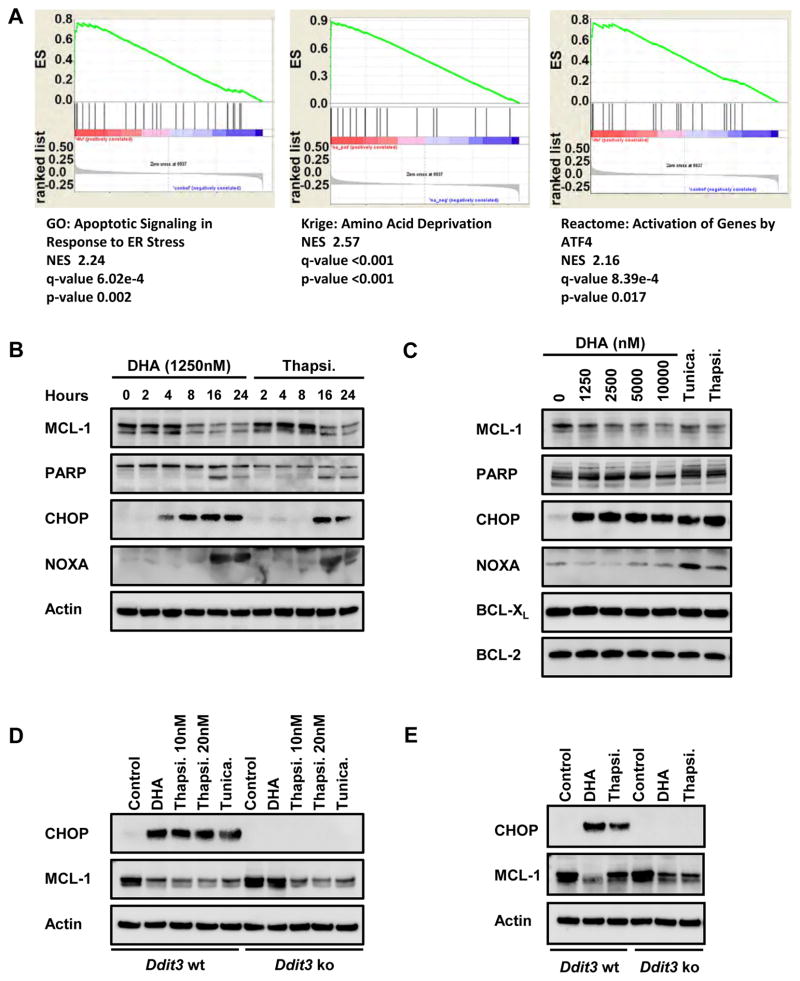
DHA has been proposed to induce killing of cancer cells through the induction of reactive oxygen species (ROS) (33, 34). Indeed, DHA treatment does induce ROS generation; however, multiple ROS scavengers that repressed the induced ROS were unable to block the attenuation of MCL-1 expression triggered by DHA (Sup. Fig. 2D&E). Therefore, we focused on identifying other mechanisms by which DHA represses MCL-1 expression. To decipher the cellular response to DHA, microarray analysis was performed to identify pathways of gene expression triggered by DHA. When compared to control treated mouse BCR-ABL+ B-ALL cells, cells treated with DHA for 4 hours revealed 18 genes that were significantly induced by DHA treatment (top induced genes were Ddit3, Chac1, Trib3, Atf3, Sesn2, Egr1, and Nupr1) (Sup. Fig. 3A). GSEA indicated that the gene set induced by DHA repression correlates significantly with known endoplasmic reticulum (ER) stress pathway signatures (Fig. 2A). The induction of these genes was confirmed by quantitative PCR and immunoblot analysis for CHOP, which is encoded by the Ddit3 gene (Sup. Fig. 3B and Fig. 2B). Even when the cells were treated with ROS scavengers, ER stress markers were induced by DHA treatment (Sup. Fig. 2E). Furthermore, the induction of the BH3-only gene Pmaip1, which encodes the pro-apoptotic protein NOXA, was detected in BCR-ABL+ B-ALL cells in response to DHA treatment (Sup Fig. 3B and Fig. 2B). Importantly, NOXA has been previously implicated in repressing MCL-1 protein expression by promoting its proteasome-mediated degradation, making it an attractive candidate (39, 40).
Figure 2. DHA treatment induces an ER stress response that represses MCL-1 expression.
(A) Gene-set enrichment analysis (GSEA, version 2.2.3) was performed using microarray data from mouse BCR-ABL+ B-ALL cells treated for 4 hours with vehicle (control) versus 312 nM DHA and the c5.all.v5.2 symbol.gmt Gene ontology gene sets. (B) Mouse BCR-ABL+ B-ALL cells were treated with 1250 nM DHA or 5 nM thapsigargin (Thapsi, positive control for ER stress). Cell lysates were analyzed by immunoblot to detect indicated proteins. This experiment is representative of 3 independently performed assays. (C) SV40-transformed wild-type mouse embryonic fibroblasts (MEFs) were treated with indicated doses of DHA. As a positive control for ER stress induction, MEFs were treated with either 25 ng/ml tunicamycin (Tunica) or 20 nM thapsigargin. After 24 hours, the MEFs were harvested and cell lysates analyzed by immunoblot analysis. This experiment is representative of 3 independently-performed assays. (D) Wild-type (Ddit3 wt) or Ddit3-deficient (Ddit3 ko) SV40-transformed MEFs were treated with 10 μM DHA and lysates prepared after 24 hours. To induce ER stress, the cells were treated with thapsigargin (10 or 20 nM) or tunicamycin 25ng/ml. The expression of CHOP (encoded for by Ddit3 gene), MCL-1 and Actin proteins were detected by immunoblot. This experiment is representative of 3 independently-performed assays. (E) Wild-type (Ddit3 wt) or Ddit3-deficient (Ddit3 ko) mouse BCR-ABL+ B-ALL cells were treated with 1250 nM DHA and lysates prepared after 24 hours. To induce ER stress, the cells were treated with thapsigargin (5 nM). The expression of CHOP (encoded for by Ddit3 gene), MCL-1 and Actin proteins were detected by immunoblot. This experiment is representative of 3 independently-performed assays.
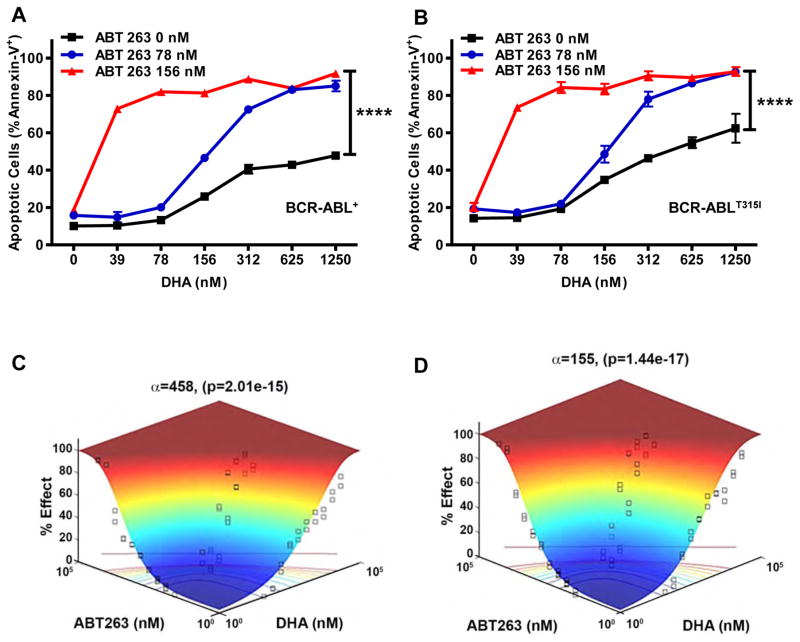
Figure 3. Synergism between DHA and ABT-263 in Mouse BCR-ABL+ B-ALL leukemia.
Mouse (A) BCR-ABL+ and (B) BCR-ABLT315I B-ALL cells were cultured with ABT-263 (0 nM, 78 nM, or 156 nM concentrations) and DHA at indicated doses in culture. Following 24 hours of incubation, apoptosis was assessed (Annexin-V+). Each experiment was performed in triplicate 3 times and the average plotted with error bars indicating S.E.M. One-way ANOVA with Bonferroni multiple comparison indicates significance p<0.001**** between the DHA alone (0 nM ABT-263) and both combined treated arms (78 nM and 156 nM) at indicated doses. (C&D) Synergy was assessed by response surface modeling. The combination of DHA and ABT-263 showed a synergistic interaction in both the mouse (C) BCR-ABL+ (α=210; p=8.59×10−14) and (D) BCR-ABLT315I (α=61.4; p=3.44×10−15) B-ALL cells.
To mechanistically address how MCL-1 expression is attenuated by DHA treatment, we established that DHA could repress MCL-1 expression in murine embryonic fibroblast (MEF) cell lines without inducing apoptosis (Fig. 2C). As in BCR-ABL+ B-ALL cells, MEF lines treated with DHA also induced the expression of CHOP, a key regulator of the ER stress pathway, as well as pro-apoptotic NOXA (Fig. 2C). Therefore, we exploited genetic mutant MEFs to assess the necessity for these candidate genes in mediating DHA’s repression of MCL-1. Despite the induction of NOXA in wild-type MEFs, we still observed the repression of MCL-1 expression when Pmaip1-deficient MEF lines were treated with DHA, suggesting that NOXA induction is not required to repress MCL-1 expression (Sup. Fig. 3C). In support of this conclusion, when BCR-ABL+ p53-deficient B-ALL cells are treated with DHA, neither NOXA nor PUMA BH3-only proteins are induced (Sup. Fig. 3D). While the BCR-ABL+ p53-deficient B-ALL cells exhibit substantial resistance to cell death induced by DHA, MCL-1 expression is still diminished by DHA treatment (Sup. Fig. 3D&E). These data suggest that while NOXA and PUMA are clearly important in the induction of apoptosis in response to DHA, they are not essential for the repression of MCL-1 expression. In contrast, Ddit3-deficient MEFs and BCR-ABL+ B-ALL cells, which lack CHOP protein, exhibit much less repression of MCL-1 protein in response to DHA treatment (Fig. 2D&E) suggesting that CHOP induction contributes to repression of MCL-1 by DHA. Therefore, in both MEF lines and BCR-ABL+ B-ALL cells, a CHOP-dependent cellular stress pathway is triggered by DHA treatment and leads to MCL-1 repression.
DHA synergizes with ABT-263 in killing murine BCR-ABL+ B-ALL cells
Resistance to tyrosine kinase inhibitors (TKI) is a significant barrier to treatment of both Ph+ CML and Ph+ ALL (6, 7, 41, 42). Therefore, TKI-resistant mouse leukemic cell lines were generated using a common “gate keeper” mutation (BCR-ABLT315I) that is observed in TKI-resistant, adult Ph+ B-ALL patients. As expected, the BCR-ABLT315I BALL cells are resistant to TKI treatment (Sup. Fig. 4A). In contrast, treatment with DHA triggered apoptosis in both wild-type BCR-ABL+ and BCR-ABLT315I B-ALL cells to a similar extent at high concentrations (Sup. Fig. 4B). At lower concentrations of DHA, the expression of MCL-1 was similarly repressed in both wild-type BCR-ABL+ and BCR-ABLT315I B-ALL cells (Sup. Fig. 4C). These data indicate that DHA can repress MCL-1 expression in both BCR-ABL+ and TKI-resistant BCR-ABLT315I B-ALL cells, unlike first-generation TKIs.
Mouse BCR-ABL+ B-ALL cells depend upon the anti-apoptotic activity of MCL-1 to mediate their survival and, accordingly, are relatively insensitive to BH3-mimetic small molecules such as ABT-263 (15, 43). Therefore, we posited that the DHA-mediated MCL-1 repression would lead to increased response of the mouse BCR-ABL+ B-ALL cells to ABT-263. To test this hypothesis, both BCR-ABL+ and BCR-ABLT315I B-ALL cells were treated in vitro with sub-optimal doses of DHA and/or ABT-263 after which cell killing was assessed. While single agents induced modest cell death, when DHA treatment was combined with ABT-263 significantly more cell death was observed in both BCR-ABL+ and BCR-ABLT315I B-ALL cells (Fig. 3A&B respectively). Response surface modeling indicated synergy for both BCR-ABL+ and BCR-ABLT315I B-ALL cells when DHA and ABT-263 are combined (Fig. 3C&D respectively). These data support the hypothesis that DHA can repress MCL-1 expression to sensitize BCR-ABL+ B-ALL cells to BH3-mimetic drugs.
DHA represses MCL-1 expression in human leukemia cells
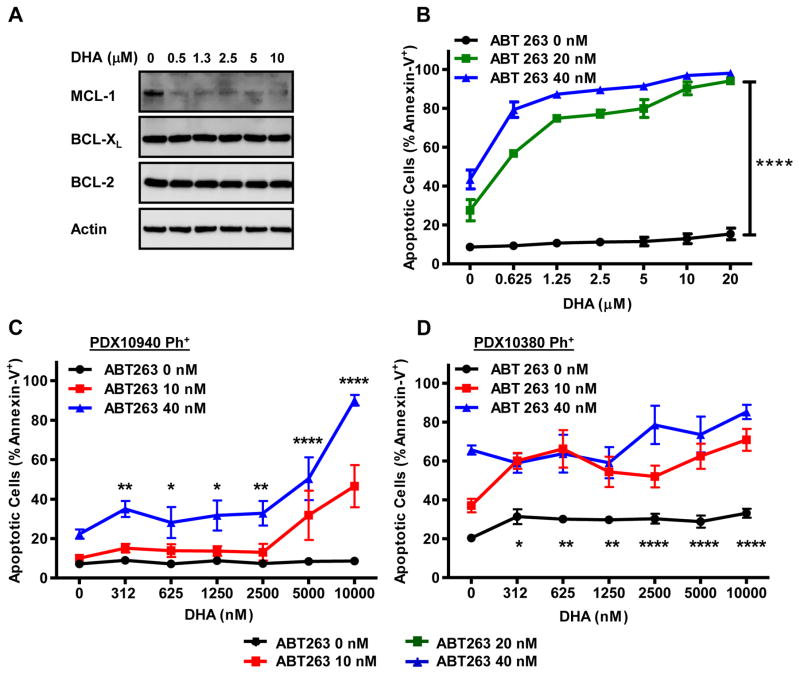
To establish whether we could potentiate the response of human Ph+ leukemia cell lines to ABT-263 treatment by co-treating with DHA, we assessed the response of TOM-1 (Ph+ B-ALL), SUP-B15 (Ph+ B-ALL), BV-173 (Ph+ CML blast crisis cell line) and OP-1 (Ph+ B-ALL) cells to treatment with DHA. Similar to our observations in murine BCR-ABL+ B-ALL cells, the treatment of these human Ph+ leukemia cell lines with DHA repressed MCL-1 expression, but did not affect the expression levels of BCL-XL or BCL-2 (Fig. 4A & Sup. Fig. 5A–C). In these human Ph+ cell lines, the repression of MCL-1 occurred even when cell death was inhibited by caspase inhibitors.
Figure 4. Synergistic effect of Ph+ human cell lines and primary patient samples to combined treatment with DHA and ABT-263.
(A) Human Ph+ ALL cell line TOM-1 was treated with indicated doses of DHA. After 24 hours, the cells were lysed and immunoblotted for expression of indicated proteins. (B) TOM-1 human Ph+ cells were cultured with ABT-263 (0 nM, 20 nM, or 40 nM concentrations) and combined with DHA at indicated doses. Following 24 hours of incubation, apoptotic cells (Annexin-V+) were assessed by flow cytometry. Each experiment was performed 3 times in triplicate and the average plotted with S.E.M. One-way ANOVA with Bonferroni multiple comparison indicates significance p<0.001**** between the DHA alone (0 nM ABT-263) and both combined treated arms (20 nM and 40 nM) at indicated doses. Human primary patient xenograft (PDX) Ph+ B-ALL cells from (C) PDX10940 and (D) PDX10380 were cultured with ABT-263 (0 nM, 10 nM, or 40 nM concentrations) and combined with DHA at indicated doses in culture. Following 24 hours of incubation, apoptotic cells were assessed. Each experiment was performed from at least 3 separate recipient mice and each assay was carried out in triplicate. The average are plotted with S.E.M. One-way ANOVA with Bonferroni multiple comparison indicates significance p<0.001**** between the DHA alone (0 nM ABT-263) and both combined treated arms (10 nM and 40 nM) at indicated doses. The combination of DHA and ABT-263 showed a synergistic interaction in (B) TOM-1 Ph+ cells (α=9.43; p=0.004) (C) primary patient xenograft PDX10940 and (α=118; p=2.64×10−10) and (D) PDX10380 (α=13667; p=2.67×10−5) by response surface modeling.
Since DHA repressed MCL-1 protein levels in human Ph+ leukemia cell lines, we tested whether we could potentiate the apoptosis induced by ABT-263 by combining with DHA in culture. While DHA alone did not provoke substantial apoptosis in these human cell lines, when DHA was combined with ABT-263 significant synergy was observed by response surface modeling (TOM-1: α=9.43; p=0.004; BV-173: α=75; p=7.11×10−7; OP-1: α=902; p=2.82×10−15; and SUP-B15: α=23602; p=1.91×10−44) (Fig. 4B & Sup. Fig. 5D–F respectively). These data demonstrate that, in all of the human Ph+ leukemia cell lines tested, apoptosis is potentiated when ABT-263 is combined with DHA.
To extend these studies beyond established human Ph+ leukemia cell lines, we took advantage of primary patient-derived xenografts (PDX) that were established by direct injection of patient leukemia into immunodeficient (NSG) recipients. Of the 5 Ph+ B-ALL PDX leukemia assessed, most were extremely sensitive to apoptosis induced by ABT-263 treatment alone; however, two Ph+ leukemia (PDX 10940 and 10380) were further tested for in vitro synergy. Leukemic cells were isolated from the recipient mice and subjected to treatment with DHA and/or ABT-263 in culture. While both PDX samples were sensitive to single agent ABT-263, they were quite resistant to cell death induced by DHA alone. However, the killing by low dose ABT-263 synergized with DHA in both PDX models (Fig. 4C&D). Response surface modeling indicated synergy of both primary patient xenografts PDX10940 and (α=118; p=2.64×10−10) and PDX10380 (α=13667; p=2.67×10−5). These data indicate that combining DHA with ABT-263 can potentiate the response of primary patient leukemia when treated in culture. Future studies will be necessary to determine whether the combination therapy can be similarly efficacious when delivered in vivo to NSG-mice bearing luciferase-labeled human PDX Ph+ leukemia.
DHA synergizes with ABT-263 to repress mouse BCR-ABL+ B-ALL leukemic progression in vivo
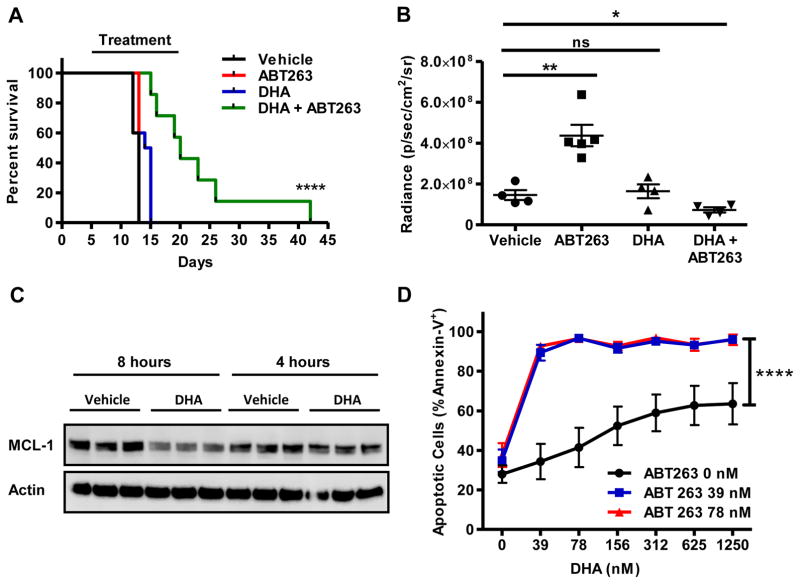
The ability of DHA to sensitize cultured BCR-ABL+ B-ALL cells to apoptosis induced by ABT-263 suggests that combining the two agents together would lead to improved responses of BCR-ABL+ B-ALL in animals. To test this concept, murine luciferase-expressing BCR-ABL+ B-ALL cells were transplanted into non-conditioned C57BL/6 recipient mice. Recipient mice received either vehicle controls, DHA (200 mg/kg), ABT-263 (100 mg/kg), or DHA (200 mg/kg) and ABT-263 (100 mg/kg) daily from day 5 after transplant to day 20 by oral gavage. Mice receiving either vehicle or ABT-263 alone rapidly succumbed to a fatal leukemia indicating no survival extension from mice treated daily with the BH3-mimetic agent (Fig. 5A). Treatment with DHA alone marginally extended survival indicating a small, but significant, effect. In contrast, combining DHA and ABT-263 together for 15 days extended survival when compared to the other treatment groups (Fig. 5A). Similar to overall survival data, mice receiving the combination treatment had reduced bioluminescence when compared to the mice treated with single agents or control (Fig. 5B and Sup. Fig. 6A). Complete blood counts from mice also showed a repression in circulating leukemic cells in combination treated animals when compared to mice only receiving single agents (Sup. Fig. 6B). Ex vivo analysis of mouse BCR-ABL+ B-ALL cells isolated from syngeneic recipient mice after in vivo DHA treatment revealed that MCL-1 protein expression was decreased when compared to vehicle treated animals at 8, but not 4, hours after DHA treatment (Fig. 5C). It remains to be established whether human leukemia cells will similarly repress MCL-1 expression when treated in vivo. The mice receiving combined treatment with both DHA and ABT-263 harvested on day 13 did not exhibit any overt toxicities (data not shown). The leukemia resulting from the transplanted cells was highly aggressive, replacing normal hematopoiesis in the bone marrow. Furthermore, the leukemic cells invaded the central nervous system and lymphatics and often resulted in hind-limb paralysis (Sup. Fig. 6C). The resultant leukemic cells expressed B220+ cell surface marker and were Pax5+ (Sup. Fig. 6D).
Figure 5. Potentiated Response of BCR-ABL+ B-ALL cells treated in vivo with DHA and ABT-263.
Mouse BCR-ABL+ B-ALL cells were adoptively transferred into syngeneic, non-irradiated C57BL/6 recipients. Five days after the transfer the recipients were divided into 4 treatment arms: vehicle controls, ABT-263 alone, DHA alone, and ABT-263 and DHA. Mice received the treatment daily (indicated by “Treatment”) for 15 days. (A) Kaplan-Meier survival curve of the cohorts of mice (n=10 per group). Log-rank test shows p<0.001**** for combined treatment group. (B) Quantification of bioluminescence measurements for the indicated mouse cohorts from analysis on day 13 after leukemia transplant. Each symbol represents one animal, the lines indicate the average radiance, and the error bars are the S.E.M. One-way ANOVA followed by t test with post-hoc correction for multiple comparisons indicate significance between indicated groups p<0.05* and p<0.01**. (C) Splenic blast cells harvested from mouse BCR-ABL+ B-ALL recipient mice, 4 or 8 hours after treatment with vehicle or DHA (200 mg/kg) were lysed and immunoblotted for indicated proteins. Significant repression was observed only at 8 hour time point. Each lane represents a recipient mouse. (D) Mouse BCR-ABL+ BALL cells from moribund recipients that received the combined treatment with ABT-263 and DHA (on day 42) were re-cultured with ABT-263 (0 nM, 39 nM, or 78 nM concentrations) and then combined with DHA at indicated doses in culture. Following 24 hours of incubation, cell viability was assessed. Each experiment was performed 3 times in triplicate and the average apoptotic cells (Annexin-V+) are plotted with S.E.M. Two-way ANOVA indicates significance p<0.001**** between the DHA alone (0 nM ABT-263)and both combined treated arms at all doses. The combination of DHA and ABT-263 still showed a synergistic interaction in ex vivo mouse BCR-ABL+ B-ALL cells (α=1871; p=3.24×10−3) by response surface modeling.
Despite the extension of survival over the 15 days of treatment, all mice, including those in the combination treated group, eventually succumbed to leukemia (Fig. 5A). To assess whether the leukemia from the combination treated mice became resistant, leukemic cells were isolated from the bone marrow of moribund mice (sacrificed on day 42) that received combined DHA and ABT-263 in vivo. The ex vivo leukemia cells were treated in culture with DHA and/or ABT-263. Similarly to the response of parental BCR-ABL+ BALL cells, synergy was still observed when DHA and ABT-263 were combined in vitro (Fig. 5D). These data indicate that the leukemic cells from the combination treated mice did not display any inherent resistance to combined DHA and ABT-263 treatment.
Discussion
We have identified that the anti-malarial drug DHA can repress MCL-1 protein expression in both mouse and human BCR-ABL+ B-ALL cells. While treatment with DHA alone can trigger mouse BCR-ABL+ B-ALL killing by a variety of mechanisms, including induction of NOXA and PUMA, DHA treatment represses MCL-1 protein expression by triggering a cellular stress response that blocks protein translation. When MCL-1 is repressed in mouse and human BCR-ABL+ leukemic cells by DHA treatment, they are significantly more sensitive to ABT-263 induced cell death. Importantly, sensitization was observed not only in wild-type BCR-ABL+ B-ALL cells, but also in TKI-resistant BCR-ABLT315I mouse leukemia indicating that DHA can repress MCL-1 in a mechanism distinct from the action of TKIs. These data suggest that combining DHA and ABT-263 together may lead to improved therapeutic responses of poor prognosis BCR-ABL+ B-ALL.
MCL-1 is a pro-survival molecule that is frequently amplified in human cancer (16). Furthermore, elevated MCL-1 expression in malignant cells is a chief mediator of resistance to both ABT-263 and the recently FDA-approved ABT-199 (12–14). As a result, laboratories in industry and academia have expended significant efforts to identify and develop specific and potent MCL-1 inhibitors (18). Many of these inhibitors suffer from a lack of specificity and potency for MCL-1 inhibition (44). Recently, the first highly-potent MCL-1 inhibitor, S63845, was reported to be well-tolerated in mice and efficacious in a models of mouse and human cancer (17). The lack of toxicity associated with S63845 treatment stands in stark contrast to mouse Mcl-1-deletion experiments, indicating that transient inhibition of MCL-1 may be better tolerated than complete and permanent genetic deletion. While promising, specific MCL-1 inhibitors like S63845 will require substantial development and testing before impacting human disease. Furthermore, it is unclear whether MCL-1 inhibition will exacerbate toxicities to normal cell types, especially when delivered with conventional chemotherapy in humans. Therefore, we focused on identifying available drugs that can attenuate MCL-1 expression and enhance the sensitivity of cancer cells to BH3-mimetic drugs that are currently being tested in humans. The dose limiting thrombocytopenia associated with ABT-263 represents a barrier to the combined use with DHA; however, this proof-of-principle study suggests that DHA can synergize with BH3-mimetic agents.
DHA has previously been reported to have anti-cancer properties; however, the specificity of activity and the concentrations needed for efficacy have remained unclear (32–35, 45). Furthermore, how DHA kills cancer cells has been linked to a number of mechanisms (32–36, 45). The treatment of mouse BCR-ABL+ B-ALL cells and MEF cells with DHA treatment induces the activation of a genetic program that is similar to the ER stress pathway as well as the pro-apoptotic protein NOXA. While NOXA has been reported to destabilize MCL-1 protein levels, we were unable to abrogate MCL-1 repression by DHA in MEF cells lacking the NOXA-encoding Pmaip1 gene. Importantly, the loss of p53 function in mouse BCR-ABL+ B-ALL cells rendered the cells quite resistant to DHA-induced apoptosis suggesting that NOXA and PUMA induction are critical apoptotic regulators.
We observed that DHA treatment of mouse BCR-ABL+ B-ALL cells triggered the potent induction of a genetic program consistent with an ER stress response. One of the key features of an ER stress response is the inhibition of protein translation (46). As MCL-1 is a very labile protein, blocking protein synthesis rapidly induces its clearance by the proteasome (47). Consistent with this hypothesis, it has been observed in other systems that the induction of ER stress can rapidly eliminate MCL-1 expression (48–51). When we genetically ablated the CHOP-encoding cellular stress gene Ddit3 in MEFs or mouse BCR-ABL+ B-ALL cells, we observed a significant decrease in MCL-1 repression triggered by DHA. These data suggest that the induction of cellular stress by DHA induces CHOP expression and represses MCL-1 protein levels. The fact that DHA treatment does not inhibit MCL-1 mRNA expression or induce enhanced degradation suggests that MCL-1 protein translation is repressed. How MCL-1 protein translation is being inhibited in a CHOP-dependent manner is still unclear, but these data suggest that CHOP-mediates the expression of additional target genes that regulate MCL-1 translation. Indeed, a myriad of gene targets are CHOP-dependent and involved in many cellular pathways including transcription and translation (52). Additionally, some non-coding microRNAs have been found to be CHOP-dependent and regulate translation of protein targets (53). Studies are ongoing to identify how DHA triggers the cellular stress pathway as identifying these cellular pathway(s) may help to identify additional therapeutic targets by which MCL-1 expression can be even more specifically antagonized.
The labile nature of MCL-1 and its response to cellular signaling pathways makes it a good candidate for combinatorial therapy. Indeed, other strategies have been reported for repressing MCL-1 expression and leading to improved responses in cancer cells (14, 15, 54–60). Our identification of DHA represents an additional therapeutic strategy in which the induction of a cellular stress pathway triggers the repression of MCL-1 protein expression in mouse BCR-ABL+ B-ALL cells. We look forward to conducting future experiments testing whether combined DHA and ABT-263 treatment of NSG recipient mice bearing Ph+ primary patient leukemia will reveal similarly synergistic responses and MCL-1 repression.. Furthermore, we are also investigating whether this mechanism might be common to other tumor and leukemia types. Importantly, in leukemic cells DHA may have the added benefit that it bypasses resistance to kinase inhibitors that would be expected to occur in the clinic. Therefore, our data indicate that DHA represents an additional strategic pathway by which MCL-1 expression can be repressed leading to enhanced response in poor prognosis BCR-ABL+ B-ALL.
Supplementary Material
Translational Relevance.
The BH3-mimetic agents venetoclax (ABT-199) and ABT-263 are promising drug candidates, particularly for hematological malignancies that are dependent on the BCL-2and BCL-XL survival genes respectively. Despite their promise, experimental evidence has indicated that elevated expression of other pro-survival molecules, such as MCL-1, represents a common resistance mechanism to these BH3-mimetics. Furthermore, in MCL-1-dependent malignancies, ABT-199 and ABT-263 resistance remains a significant barrier. We sought to identify agents that could render ABT-263-resistant BCR-ABL+ BALL cells lines sensitive to ABT-263. Our efforts identified dihydroartemisinin, an anti-malarial drug, as a repressor of MCL-1 protein expression. We demonstrate in mouse and human BCR-ABL+ B-lineage leukemia that combining DHA-treatment with ABT-263 induces significant synergistic responses in cell culture, patient-derived xenografts, and in animal models. These findings suggest that combinatorial therapy of DHA with BH3-mimetic agents can significantly improve leukemic response.
Acknowledgments
We thank the Opferman laboratory, Drs. Hendershot, Singh, and Rehg for helpful discussions. The Ddit3-deficient MEFs were from Dr. Ron (Cambridge Institute for Medical Research) and Pmaip1-deficient MEFs were from Dr. Li (University of Louisville). We thank Mr. S.T. Peters and Dr. S.M. Pruett-Miller of St. Jude’s Center for Advanced Genome Engineering for generating the mouse Ddit3-deficient BCR-ABL+ BALL cells.
Grant Support
J.T. Opferman is supported by the NIH R01HL102175 and R01CA201069; the American Cancer Society 119130-RSG-10-255-01-LIB; P30CA021765; and the American Lebanese Syrian Associated Charities.
Footnotes
Conflict of Interest
The authors declare no conflict of interest.
Authors’ Contributions
Concept and design: A. Budhraja, J.T. Opferman
Development of methodology: A. Budhraja, M.E. Turnis., A. Kothari, X. Yang.
Acquisition of data (provided animals, etc.): A. Budhraja, M.E. Turnis, M. Churchman, C.G. Mullighan, J.T. Opferman
Analysis and interpretation of data: A. Budhraja, M.E. Turnis, J.C. Panetta, D. Finkelstein, J.T. Opferman.
Writing and review: A. Budhraja, M.E. Turnis., J.T. Opferman
Administrative, technical, or material support: X. Yang, H. Xu, E. Kaminska
Study Supervision: A. Budhraja, J.T. Opferman
References
- 1.Pui CH, Jeha S. New therapeutic strategies for the treatment of acute lymphoblastic leukaemia. Nat Rev Drug Discov. 2007;6(2):149–65. doi: 10.1038/nrd2240. [DOI] [PubMed] [Google Scholar]
- 2.Burmeister T, Schwartz S, Bartram CR, Gokbuget N, Hoelzer D, Thiel E. Patients’ age and BCR-ABL frequency in adult B-precursor ALL: a retrospective analysis from the GMALL study group. Blood. 2008;112(3):918–9. doi: 10.1182/blood-2008-04-149286. [DOI] [PubMed] [Google Scholar]
- 3.Rowley JD. Letter: A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining. Nature. 1973;243(5405):290–3. doi: 10.1038/243290a0. [DOI] [PubMed] [Google Scholar]
- 4.Witte ON, Dasgupta A, Baltimore D. Abelson murine leukaemia virus protein is phosphorylated in vitro to form phosphotyrosine. Nature. 1980;283(5750):826–31. doi: 10.1038/283826a0. [DOI] [PubMed] [Google Scholar]
- 5.Deininger M, Buchdunger E, Druker BJ. The development of imatinib as a therapeutic agent for chronic myeloid leukemia. Blood. 2005;105(7):2640–53. doi: 10.1182/blood-2004-08-3097. [DOI] [PubMed] [Google Scholar]
- 6.Lilly MB, Ottmann OG, Shah NP, Larson RA, Reiffers JJ, Ehninger G, et al. Dasatinib 140 mg once daily versus 70 mg twice daily in patients with Ph-positive acute lymphoblastic leukemia who failed imatinib: Results from a phase 3 study. American journal of hematology. 2010;85(3):164–70. doi: 10.1002/ajh.21615. [DOI] [PubMed] [Google Scholar]
- 7.Nicolini FE, Mauro MJ, Martinelli G, Kim DW, Soverini S, Muller MC, et al. Epidemiologic study on survival of chronic myeloid leukemia and Ph(+) acute lymphoblastic leukemia patients with BCR-ABL T315I mutation. Blood. 2009;114(26):5271–8. doi: 10.1182/blood-2009-04-219410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA, et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature. 2005;435(7042):677–81. doi: 10.1038/nature03579. [DOI] [PubMed] [Google Scholar]
- 9.Souers AJ, Leverson JD, Boghaert ER, Ackler SL, Catron ND, Chen J, et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med. 2013;19(2):202–8. doi: 10.1038/nm.3048. [DOI] [PubMed] [Google Scholar]
- 10.Sarosiek KA, Letai A. Directly targeting the mitochondrial pathway of apoptosis for cancer therapy using BH3 mimetics - recent successes, current challenges and future promise. The FEBS journal. 2016;283(19):3523–33. doi: 10.1111/febs.13714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mason KD, Carpinelli MR, Fletcher JI, Collinge JE, Hilton AA, Ellis S, et al. Programmed anuclear cell death delimits platelet life span. Cell. 2007;128(6):1173–86. doi: 10.1016/j.cell.2007.01.037. [DOI] [PubMed] [Google Scholar]
- 12.Konopleva M, Contractor R, Tsao T, Samudio I, Ruvolo PP, Kitada S, et al. Mechanisms of apoptosis sensitivity and resistance to the BH3 mimetic ABT-737 in acute myeloid leukemia. Cancer Cell. 2006;10(5):375–88. doi: 10.1016/j.ccr.2006.10.006. [DOI] [PubMed] [Google Scholar]
- 13.van Delft MF, Wei AH, Mason KD, Vandenberg CJ, Chen L, Czabotar PE, et al. The BH3 mimetic ABT-737 targets selective Bcl-2 proteins and efficiently induces apoptosis via Bak/Bax if Mcl-1 is neutralized. Cancer Cell. 2006;10(5):389–99. doi: 10.1016/j.ccr.2006.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Choudhary GS, Al-Harbi S, Mazumder S, Hill BT, Smith MR, Bodo J, et al. MCL-1 and BCL-xL-dependent resistance to the BCL-2 inhibitor ABT-199 can be overcome by preventing PI3K/AKT/mTOR activation in lymphoid malignancies. Cell death & disease. 2015;6:e1593. doi: 10.1038/cddis.2014.525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Koss B, Morrison J, Perciavalle RM, Singh H, Rehg JE, Williams RT, et al. Requirement for antiapoptotic MCL-1 in the survival of BCR-ABL B-lineage acute lymphoblastic leukemia. Blood. 2013;122(9):1587–98. doi: 10.1182/blood-2012-06-440230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J, et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463(7283):899–905. doi: 10.1038/nature08822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kotschy A, Szlavik Z, Murray J, Davidson J, Maragno AL, Le Toumelin-Braizat G, et al. The MCL1 inhibitor S63845 is tolerable and effective in diverse cancer models. Nature. 2016;538(7626):477–82. doi: 10.1038/nature19830. [DOI] [PubMed] [Google Scholar]
- 18.Opferman JT. Attacking cancer’s Achilles heel: antagonism of anti-apoptotic BCL-2 family members. The FEBS journal. 2016;283(14):2661–75. doi: 10.1111/febs.13472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stewart DP, Koss B, Bathina M, Perciavalle RM, Bisanz K, Opferman JT. Ubiquitin-independent degradation of antiapoptotic MCL-1. Mol Cell Biol. 2010;30(12):3099–110. doi: 10.1128/MCB.01266-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Villunger A, Michalak EM, Coultas L, Mullauer F, Bock G, Ausserlechner MJ, et al. p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science. 2003;302(5647):1036–8. doi: 10.1126/science.1090072. [DOI] [PubMed] [Google Scholar]
- 21.Zinszner H, Kuroda M, Wang X, Batchvarova N, Lightfoot RT, Remotti H, et al. CHOP is implicated in programmed cell death in response to impaired function of the endoplasmic reticulum. Genes Dev. 1998;12(7):982–95. doi: 10.1101/gad.12.7.982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Greco WR, Park HS, Rustum YM. Application of a new approach for the quantitation of drug synergism to the combination of cis-diamminedichloroplatinum and 1-beta-D-arabinofuranosylcytosine. Cancer Res. 1990;50(17):5318–27. [PubMed] [Google Scholar]
- 23.Minto CF, Schnider TW, Short TG, Gregg KM, Gentilini A, Shafer SL. Response surface model for anesthetic drug interactions. Anesthesiology. 2000;92(6):1603–16. doi: 10.1097/00000542-200006000-00017. [DOI] [PubMed] [Google Scholar]
- 24.Jonker DM, Visser SA, van der Graaf PH, Voskuyl RA, Danhof M. Towards a mechanism-based analysis of pharmacodynamic drug-drug interactions in vivo. Pharmacology & therapeutics. 2005;106(1):1–18. doi: 10.1016/j.pharmthera.2004.10.014. [DOI] [PubMed] [Google Scholar]
- 25.Rowe JM, Buck G, Burnett AK, Chopra R, Wiernik PH, Richards SM, et al. Induction therapy for adults with acute lymphoblastic leukemia: results of more than 1500 patients from the international ALL trial: MRC UKALL XII/ECOG E2993. Blood. 2005;106(12):3760–7. doi: 10.1182/blood-2005-04-1623. [DOI] [PubMed] [Google Scholar]
- 26.Notta F, Mullighan CG, Wang JC, Poeppl A, Doulatov S, Phillips LA, et al. Evolution of human BCR-ABL1 lymphoblastic leukaemia-initiating cells. Nature. 2011;469(7330):362–7. doi: 10.1038/nature09733. [DOI] [PubMed] [Google Scholar]
- 27.Churchman ML, Low J, Qu C, Paietta EM, Kasper LH, Chang Y, et al. Efficacy of Retinoids in IKZF1-Mutated BCR-ABL1 Acute Lymphoblastic Leukemia. Cancer Cell. 2015;28(3):343–56. doi: 10.1016/j.ccell.2015.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tse C, Shoemaker AR, Adickes J, Anderson MG, Chen J, Jin S, et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. 2008;68(9):3421–8. doi: 10.1158/0008-5472.CAN-07-5836. [DOI] [PubMed] [Google Scholar]
- 29.Boulos N, Mulder HL, Calabrese CR, Morrison JB, Rehg JE, Relling MV, et al. Chemotherapeutic agents circumvent emergence of dasatinib-resistant BCR-ABL kinase mutations in a precise mouse model of Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood. 2011;117(13):3585–95. doi: 10.1182/blood-2010-08-301267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Singh H, Shelat AA, Singh A, Boulos N, Williams RT, Guy RK. A screening-based approach to circumvent tumor microenvironment-driven intrinsic resistance to BCR-ABL+ inhibitors in Ph+ acute lymphoblastic leukemia. Journal of biomolecular screening. 2014;19(1):158–67. doi: 10.1177/1087057113501081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Newton PN, van Vugt M, Teja-Isavadharm P, Siriyanonda D, Rasameesoroj M, Teerapong P, et al. Comparison of oral artesunate and dihydroartemisinin antimalarial bioavailabilities in acute falciparum malaria. Antimicrobial agents and chemotherapy. 2002;46(4):1125–7. doi: 10.1128/AAC.46.4.1125-1127.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang CZ, Zhang H, Yun J, Chen GG, Lai PB. Dihydroartemisinin exhibits antitumor activity toward hepatocellular carcinoma in vitro and in vivo. Biochemical pharmacology. 2012;83(9):1278–89. doi: 10.1016/j.bcp.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 33.Cabello CM, Lamore SD, Bair WB, 3rd, Qiao S, Azimian S, Lesson JL, et al. The redox antimalarial dihydroartemisinin targets human metastatic melanoma cells but not primary melanocytes with induction of NOXA-dependent apoptosis. Investigational new drugs. 2012;30(4):1289–301. doi: 10.1007/s10637-011-9676-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang Z, Hu W, Zhang JL, Wu XH, Zhou HJ. Dihydroartemisinin induces autophagy and inhibits the growth of iron-loaded human myeloid leukemia K562 cells via ROS toxicity. FEBS open bio. 2012;2:103–12. doi: 10.1016/j.fob.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Handrick R, Ontikatze T, Bauer KD, Freier F, Rubel A, Durig J, et al. Dihydroartemisinin induces apoptosis by a Bak-dependent intrinsic pathway. Molecular cancer therapeutics. 2010;9(9):2497–510. doi: 10.1158/1535-7163.MCT-10-0051. [DOI] [PubMed] [Google Scholar]
- 36.Hu CJ, Zhou L, Cai Y. Dihydroartemisinin induces apoptosis of cervical cancer cells via upregulation of RKIP and downregulation of bcl-2. Cancer biology & therapy. 2014;15(3):279–88. doi: 10.4161/cbt.27223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Perciavalle RM, Opferman JT. Delving deeper: MCL-1’s contributions to normal and cancer biology. Trends Cell Biol. 2013;23(1):22–9. doi: 10.1016/j.tcb.2012.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Maurer U, Charvet C, Wagman AS, Dejardin E, Green DR. Glycogen synthase kinase-3 regulates mitochondrial outer membrane permeabilization and apoptosis by destabilization of MCL-1. Mol Cell. 2006;21(6):749–60. doi: 10.1016/j.molcel.2006.02.009. [DOI] [PubMed] [Google Scholar]
- 39.Czabotar PE, Lee EF, van Delft MF, Day CL, Smith BJ, Huang DC, et al. Structural insights into the degradation of Mcl-1 induced by BH3 domains. Proc Natl Acad Sci U S A. 2007;104(15):6217–22. doi: 10.1073/pnas.0701297104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Day CL, Smits C, Fan FC, Lee EF, Fairlie WD, Hinds MG. Structure of the BH3 domains from the p53-inducible BH3-only proteins Noxa and Puma in complex with Mcl-1. J Mol Biol. 2008;380(5):958–71. doi: 10.1016/j.jmb.2008.05.071. [DOI] [PubMed] [Google Scholar]
- 41.Druker BJ. Circumventing resistance to kinase-inhibitor therapy. N Engl J Med. 2006;354(24):2594–6. doi: 10.1056/NEJMe068073. [DOI] [PubMed] [Google Scholar]
- 42.Ravandi F, O’Brien S, Thomas D, Faderl S, Jones D, Garris R, et al. First report of phase 2 study of dasatinib with hyper-CVAD for the frontline treatment of patients with Philadelphia chromosome-positive (Ph+) acute lymphoblastic leukemia. Blood. 2010;116(12):2070–7. doi: 10.1182/blood-2009-12-261586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Koss B, Ryan J, Budhraja A, Szarama K, Yang X, Bathina M, et al. Defining specificity and on-target activity of BH3-mimetics using engineered B-ALL cell lines. Oncotarget. 2016;7(10):11500–11. doi: 10.18632/oncotarget.7204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Varadarajan S, Vogler M, Butterworth M, Dinsdale D, Walensky LD, Cohen GM. Evaluation and critical assessment of putative MCL-1 inhibitors. Cell Death Differ. 2013;20(11):1475–84. doi: 10.1038/cdd.2013.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lu M, Sun L, Zhou J, Yang J. Dihydroartemisinin induces apoptosis in colorectal cancer cells through the mitochondria-dependent pathway. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2014;35(6):5307–14. doi: 10.1007/s13277-014-1691-9. [DOI] [PubMed] [Google Scholar]
- 46.Wang M, Kaufman RJ. The impact of the endoplasmic reticulum protein-folding environment on cancer development. Nat Rev Cancer. 2014;14(9):581–97. doi: 10.1038/nrc3800. [DOI] [PubMed] [Google Scholar]
- 47.Nijhawan D, Fang M, Traer E, Zhong Q, Gao W, Du F, et al. Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev. 2003;17(12):1475–86. doi: 10.1101/gad.1093903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Martin-Perez R, Niwa M, Lopez-Rivas A. ER stress sensitizes cells to TRAIL through down-regulation of FLIP and Mcl-1 and PERK-dependent up-regulation of TRAIL-R2. Apoptosis. 2012;17(4):349–63. doi: 10.1007/s10495-011-0673-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gomez-Bougie P, Halliez M, Moreau P, Pellat-Deceunynck C, Amiot M. Repression of Mcl-1 and disruption of the Mcl-1/Bak interaction in myeloma cells couple ER stress to mitochondrial apoptosis. Cancer Lett. 2016;383(2):204–11. doi: 10.1016/j.canlet.2016.09.030. [DOI] [PubMed] [Google Scholar]
- 50.Liu X, Lv Z, Zou J, Liu X, Ma J, Wang J, et al. Afatinib down-regulates MCL-1 expression through the PERK-eIF2alpha-ATF4 axis and leads to apoptosis in head and neck squamous cell carcinoma. Am J Cancer Res. 2016;6(8):1708–19. [PMC free article] [PubMed] [Google Scholar]
- 51.Li X, Li M, Ruan H, Qiu W, Xu X, Zhang L, et al. Co-targeting translation and proteasome rapidly kills colon cancer cells with mutant RAS/RAF via ER stress. Oncotarget. 2016;8(6):9280–92. doi: 10.18632/oncotarget.14063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Teske BF, Fusakio ME, Zhou D, Shan J, McClintick JN, Kilberg MS, et al. CHOP induces activating transcription factor 5 (ATF5) to trigger apoptosis in response to perturbations in protein homeostasis. Mol Biol Cell. 2013;24(15):2477–90. doi: 10.1091/mbc.E13-01-0067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Behrman S, Acosta-Alvear D, Walter P. A CHOP-regulated microRNA controls rhodopsin expression. J Cell Biol. 2011;192(6):919–27. doi: 10.1083/jcb.201010055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Essafi A, Fernandez de Mattos S, Hassen YA, Soeiro I, Mufti GJ, Thomas NS, et al. Direct transcriptional regulation of Bim by FoxO3a mediates STI571-induced apoptosis in Bcr-Abl-expressing cells. Oncogene. 2005;24(14):2317–29. doi: 10.1038/sj.onc.1208421. [DOI] [PubMed] [Google Scholar]
- 55.Cragg MS, Kuroda J, Puthalakath H, Huang DC, Strasser A. Gefitinib-induced killing of NSCLC cell lines expressing mutant EGFR requires BIM and can be enhanced by BH3 mimetics. PLoS medicine. 2007;4(10):1681–89. doi: 10.1371/journal.pmed.0040316. discussion 90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Reynoso D, Nolden LK, Yang D, Dumont SN, Conley AP, Dumont AG, et al. Synergistic induction of apoptosis by the Bcl-2 inhibitor ABT-737 and imatinib mesylate in gastrointestinal stromal tumor cells. Molecular oncology. 2011;5(1):93–104. doi: 10.1016/j.molonc.2010.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Narita T, Ishida T, Ito A, Masaki A, Kinoshita S, Suzuki S, et al. Cyclin-dependent kinase 9 is a novel specific molecular target in adult T-cell leukemia/lymphoma. Blood. 2017 doi: 10.1182/blood-2016-09-741983. [DOI] [PubMed] [Google Scholar]
- 58.Rahmani M, Davis EM, Bauer C, Dent P, Grant S. Apoptosis induced by the kinase inhibitor BAY 43-9006 in human leukemia cells involves down-regulation of Mcl-1 through inhibition of translation. J Biol Chem. 2005;280(42):35217–27. doi: 10.1074/jbc.M506551200. [DOI] [PubMed] [Google Scholar]
- 59.Rahmani M, Nguyen TK, Dent P, Grant S. The multikinase inhibitor sorafenib induces apoptosis in highly imatinib mesylate-resistant bcr/abl+ human leukemia cells in association with signal transducer and activator of transcription 5 inhibition and myeloid cell leukemia-1 down-regulation. Mol Pharmacol. 2007;72(3):788–95. doi: 10.1124/mol.106.033308. [DOI] [PubMed] [Google Scholar]
- 60.Nguyen TK, Jordan N, Friedberg J, Fisher RI, Dent P, Grant S. Inhibition of MEK/ERK1/2 sensitizes lymphoma cells to sorafenib-induced apoptosis. Leuk Res. 2010;34(3):379–86. doi: 10.1016/j.leukres.2009.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.