Abstract
Background: Excess weight in adulthood is one of the few modifiable risk factors for pancreatic cancer, and height has associations as well. This leads to the question of whether body weight and height in childhood are associated with adult pancreatic cancer.
Objective: The aim of the study was to examine if childhood body mass index (BMI; kg/m2) and height are associated with pancreatic cancer in adulthood.
Methods: We linked 293,208 children born from 1930 to 1982 in the Copenhagen School Health Records Register who had measured values of weights and heights at ages 7–13 y with the Danish Cancer Registry to identify incident pancreatic cancer cases from 1968 to 2012. HRs and 95% CIs were estimated by using Cox proportional hazards regressions.
Results: During 8,207,015 person-years of follow-up, 1268 pancreatic cancer cases were diagnosed. Childhood BMI z scores at ages 7–13 y were positively and significantly associated with pancreatic cancer in men and women <70 y of age; ≥70 y of age, the associations diminished. The HRs of pancreatic cancer were 1.13 (95% CI: 1.05, 1.21) and 1.18 (95% CI: 1.09, 1.27) according to BMI z score at ages 7 and 13 y, respectively. A BMI z score of ≥1.5 at ages 7, 10, and 13 y was positively and significantly associated with pancreatic cancer; however, the effect did not differ from having a BMI z score ≥1.5 at only one of these ages. Positive, albeit nonsignificant, associations were identified with height.
Conclusions: BMI at all ages from 7 to 13 y was positively and linearly associated with adult pancreatic cancer: the higher the BMI, the higher the risk. Excess childhood BMI may be indicative of processes initiated early in life that lead to this cancer. The prevention of childhood adiposity may decrease the burden of pancreatic cancer in adults.
Keywords: cancer, children, growth, height, obesity, pancreas, weight
Introduction
Pancreatic cancer is the fourth leading cause of cancer death in the United States (1), and it ranks in the top 10 leading causes of cancer death in Denmark (2). Pancreatic cancer is highly fatal; 65% of patients die within 1 y of diagnosis and the current 5-y survival rate is 7.7% (3). Although overall cancer incidence and deaths are declining, incidence and death rates for pancreatic cancer have increased (1). One hypothesis that may explain these increasing rates is the current obesity and diabetes epidemic.
Adult obesity has emerged as a consistent risk factor for pancreatic cancer over the past decade (4–10). Current studies have shown overweight and obesity during late adolescence and early adulthood to be associated with an elevated risk of this disease (9–12). Evidence also suggests that there is a cumulative effect of adult overweight: the longer an adult is overweight or obese, the greater the risk of pancreatic cancer (12). Furthermore, as with many forms of cancer, a greater adult attained height is also associated with increased pancreatic cancer risk, but the exact mechanisms remain speculative (4, 13).
Childhood obesity is a global public health issue, with an estimated 155 million school-aged children currently classified as overweight or obese (14). Overweight and obesity in childhood are associated with a variety of diseases in adulthood (9, 11, 12, 15–18). The World Cancer Research Fund reported greater childhood growth as a probable risk factor for pancreatic cancer; however, this was based on attained height and BMI during early adulthood (4). The majority of the studies that examined body size during adolescence or early adulthood relied on recalled weight and height (9, 11, 12). Hence, we examined whether childhood BMI and height as determined from measures during childhood health examinations are associated with subsequent pancreatic cancer in adulthood in a Danish population.
Methods
Study population
In this prospective cohort study, 372,636 individuals from the Copenhagen School Health Records Register who were born from 1930 to 1989 underwent mandatory health examinations at public and private schools from ages 7 to 13 y annually through 1983, after which time the health examinations were performed only at school entry and exit unless a child had special health needs (19). During the examinations, school doctors and nurses measured the height and weight of the children by using equipment provided by the schools and standard procedures. Each child was assigned a health card with the child's name, year of birth, height, and weight. This information has been computerized. The Copenhagen School Health Records Register was built in collaboration between the Department of Clinical Epidemiology (formerly the Institute of Preventive Medicine) and the Copenhagen City Archives in Denmark.
A unique personal identification number was assigned to all Danish residents who were alive or born after April 1968, when the Central Person Register was established (20). Children who were in school at that time or later had their identification number recorded on their health cards, and children who attended school before this time had their identification numbers retrieved from the register on the basis of their name and date of birth (21). These numbers were identified for >88% of the children in the register. The process of retrieving the numbers was conservative (19), and the main reasons for not identifying the number were death, emigration before 1968, or misspellings in names that precluded the unique identification of the child.
BMI was calculated by dividing weight in kilograms by height in meters squared. BMI values were transformed to z scores on the basis of an internal age- and sex-specific reference that was computed by using the Lambda Median Sigma method (22). The reference population was chosen from a period when the prevalence of obesity was low and stable, and z scores were interpolated to the exact age if 2 measurements were available or extrapolated if only 1 measurement was available, but always within a ±12-mo time frame (15).
The identification number enabled linkage to the Danish Cancer Registry (23) from which information on pancreatic cancer was obtained. The cancer registry has collected information on all cancer diagnoses in Denmark since 1943. Pancreatic cancer was defined according to the modified Danish version of the International Classification of Diseases versions 7 (code 157.0) and 10 (codes C25.0–C25.4, C25.7–C25.9).
The first diagnosis of pancreatic cancer, regardless of a previous cancer diagnosis, was used in the analyses. Follow-up began in 1968, corresponding to when the computerized vital statistics register was established. We followed individuals from 30 y of age, corresponding to the birth years of 1930–1982. From the population of 348,974 individuals available for this study, 42,480 did not have a personal identification number; and from the remaining individuals with identification numbers, 2309 died, 6281 emigrated, and 196 were lost to follow-up before the age of 30 y (Supplemental Figure 1). Therefore, the eligible population for inclusion in the study consisted of 297,708 individuals who were alive and living in Denmark at 30 y of age. Exclusions were made for individuals who had pancreatic cancer before 30 y of age (n = 1) and individuals with missing (n = 4492) or outlying (z scores <−4.5 or >4.5) height or BMI values at all ages (n = 7) (Supplemental Figure 1). Of the 293,208 individuals included in the study, 6152 individuals emigrated, 166 individuals were lost to follow-up, and 56,147 died before the end of the study period. Follow-up ended on the date of a pancreatic cancer diagnosis, death, emigration, or 31 December 2013, whichever came first.
The analyses were conducted on anonymous data, and the study was approved by the Danish Data Protection agency. According to the Danish Act of Processing of Personal Data, informed consent is not required for register-based research of pre-existing personal data.
Statistical analyses
Associations between BMI and height z scores at each age from 7 to 13 y were estimated by using Cox proportional hazards regression models with age as the underlying time metric. We conducted the sensitivity analyses adjusting BMI for height, and vice versa, because they are correlated in childhood. To examine if there was a cumulative effect of excess BMI in childhood, we examined if children who were heavy, defined as having a BMI z score ≥1.5 (equivalent to the 93.3rd percentile), at ages 7, 10, and/or 13 y had a different risk of pancreatic cancer than did children who had a BMI z score <1.5 at these ages. Due to how the z scores were calculated, the ages of 7, 10, and 13 were chosen for analysis because they were calculated independently of each other. Children who never had a BMI z score ≥1.5 at any of these ages formed the reference group. Furthermore, we examined if children who were heavier at age 13 y irrespective of their body size history had a different risk of pancreatic cancer than children who were not large at age 13 y. For these analyses, the reference group was children with a BMI z score <1.5 at age 13 y only.
Because the cohort spans many years of birth, all of the analyses were stratified by birth cohort (5-y intervals from 1930–1975 to 1976–1982). Interactions of BMI and height z-score associations with pancreatic cancer by sex and birth cohort were also tested by using log likelihood ratio tests. No interactions were detected (all P > 0.40); however, because the sex-specific results may be of interest, these are presented as well. The linearity of the associations was assessed by restricted cubic splines (5 knots), and notable violations were not detected. The proportional hazards assumption was tested in each model by the inclusion of a time-varying effect of BMI and height, respectively, in the Cox regression models. Violations were detected such that the associations with BMI changed at ∼70 y of age (P-value range: 0.005–0.035). Therefore, we present analyses stratified by <70 and ≥70 y of age.
We calculated the unadjusted population attributable fraction (PAF) (24) to estimate the percentage of pancreatic cancer cases before age 70 y that would potentially be eliminated if children were not overweight or obese at age 13 y. Overweight and obesity were defined by using the CDC 2000 classifications (25). For these calculations, we used the HRs for boys and girls at age 13 y in our Danish study population and the proportions of overweight or obese girls and boys at age 12.5– 13.5 y in the US NHANES (all ethnicities) between 2003 and 2014 (26).
Results
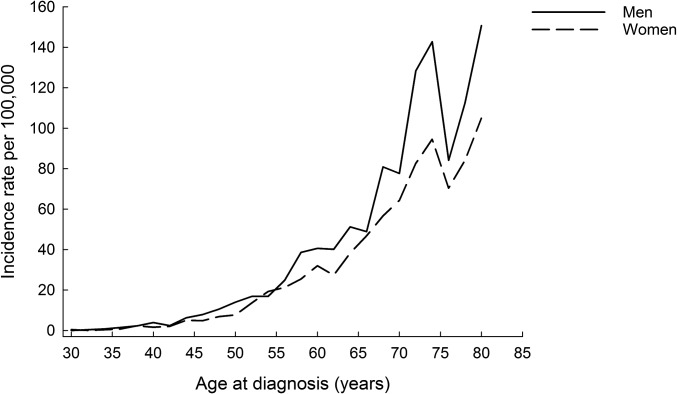
At least 1 BMI and height z score were available for 144,826 girls and 148,382 boys. As previously published and expected, height and BMI increased with age in this study population (18). The analyses included 8,207,015 person-years of follow-up after age 30 y. During follow-up there were 1268 cases of pancreatic cancer (699 in men and 569 in women). The incidence of pancreatic cancer increased with advancing age and was slightly higher in the men than in women (Figure 1).
FIGURE 1.
Incidence of pancreatic cancer among men and women from 30 to 83 y of age.
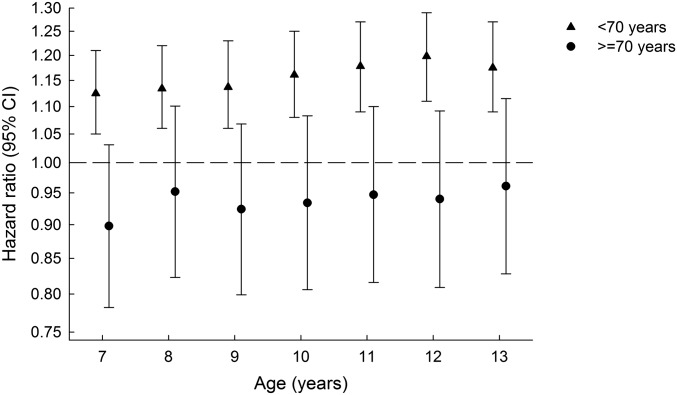
Positive and significant associations between childhood BMI z scores and adult pancreatic cancer were found at all ages (Supplemental Table 1, Figure 2). Childhood BMI z scores at each age from 7 to 13 y were significantly associated with pancreatic cancer risk in men and women up until the age of 70 y (Supplemental Table 1, Figure 2). Beyond 70 y of age, the associations were no longer significant. No notable violations of linearity were detected; the pancreatic cancer risk was greatest with the highest childhood BMI z scores. Adjustment for height in the model had no effect on HRs of pancreatic cancer (Supplemental Table 2). Height was positively, albeit nonsignificantly, associated with adult pancreatic cancer (Table 1).
FIGURE 2.
Childhood BMI z score and pancreatic cancer risk by diagnosis at ages <70 and ≥70 y. The dashed line represents an HR of 1. Analyses were stratified by sex and birth cohort.
TABLE 1.
Childhood height z scores at ages 7–13 y and pancreatic cancer risk in adulthood1
| Age at diagnosis | ||||||
|---|---|---|---|---|---|---|
| <70 y | ≥70 y | |||||
| Age, y | Individuals, n | Cases, n | HR (95% CI) | Individuals, n | Cases, n | HR (95% CI) |
| 7 | 275,525 | 922 | 1.03 (0.96, 1.10) | 58,475 | 264 | 1.10 (0.97, 1.24) |
| 8 | 279,796 | 928 | 1.04 (0.97, 1.11) | 59,813 | 270 | 1.10 (0.97, 1.24) |
| 9 | 273,388 | 930 | 1.04 (0.97, 1.11) | 59,989 | 275 | 1.11 (0.98, 1.25) |
| 10 | 267,867 | 930 | 1.03 (0.97, 1.10) | 60,015 | 275 | 1.10 (0.97, 1.24) |
| 11 | 266,091 | 922 | 1.04 (0.98, 1.11) | 60,128 | 278 | 1.09 (0.96, 1.22) |
| 12 | 263,328 | 929 | 1.04 (0.97, 1.11) | 59,898 | 271 | 1.07 (0.95, 1.21) |
| 13 | 259,342 | 927 | 1.05 (0.99, 1.13) | 59,043 | 264 | 1.09 (0.96, 1.23) |
Analyses were stratified by sex and birth cohort.
In the subanalysis of 119,799 men and 119,080 women who had BMI values at 7, 10, and 13 y of age, we examined if there was an effect of repeatedly high BMI or if it was only size at 13 y that mattered for later pancreatic cancer. Compared with the reference group of children who had BMI z scores <1.5 at all ages of 7, 10, and 13 y, having a BMI z score of ≥1.5 once, twice, or 3 times at any of these ages (7, 10, or 13 y) was associated with an increased risk of pancreatic cancer until age 70 y (Table 2). Even though the HRs for children who had a BMI z score ≥1.5 at all 3 ages were higher than for other combinations of BMI z scores, these differences were not significant compared with having a z score >1.5 at only 1 age (P = 0.88) or at 2 ages (P = 0.67). Compared with the reference group of 225,606 children with a BMI z score <1.5 at age 13 y (823 pancreatic cancer cases), the 13,273 children who had a BMI z score ≥1.5 at the same age (61 pancreatic cancer cases) had an HR for pancreatic cancer <70 y of 1.51 (95% CI: 1.16, 1.96).
TABLE 2.
Patterns of repeatedly high BMI values (≥1.5 z score) at ages 7, 10, and 13 y and pancreatic cancer risk in adulthood by age at diagnosis1
| Age at diagnosis | ||||||
|---|---|---|---|---|---|---|
| BMI z score ≥1.5 | <70 y | ≥70 y | ||||
| Individuals , n | Cases, n | HR (95% CI) | Individuals, n | Cases, n | HR (95% CI) | |
| Never | 217,721 | 789 | Reference | 50,105 | 224 | Reference |
| Age 7 y only | 3839 | 17 | 1.32 (0.82, 2.13) | 753 | 3 | 0.93 (0.30, 2.91) |
| Age 10 y only | 2131 | 11 | 1.51 (0.83, 2.75) | 369 | 2 | 1.52 (0.38, 6.12) |
| Age 13 y only | 4243 | 21 | 1.54 (1.00, 2.37) | 703 | 4 | 1.50 (0.56, 4.03) |
| Ages 7 and 10 y | 1915 | 6 | 1.04 (0.46, 2.32) | 255 | 1 | 1.05 (0.15, 7.46) |
| Ages 7 and 13 y | 830 | 3 | 1.20 (0.39, 3.72) | 131 | 0 | — |
| Ages 10 and 13 y | 2996 | 13 | 1.34 (0.78, 2.32) | 481 | 0 | — |
| Ages 7, 10, and 13 y | 5204 | 24 | 1.70 (1.13, 2.56) | 566 | 1 | 0.47 (0.67, 3.33) |
Analyses were stratified by sex and birth cohort. Compared with a child with a BMI z score ≥1.5 at 7, 10, and 13 y, the HR did not significantly differ from children who had a BMI z score ≥1.5 at only 1 age (P = 0.88) or at 2 ages (P = 0.67).
With the use of the pooled data from NHANES and the CDC criteria, among 13-y-old US children, 14.8% of boys and 17.2% of girls are classified as overweight and 21.7% of boys and 21.5% of girls are classified as obese. On the basis of these US prevalence data and the pancreatic cancer HRs for BMI at age 13 y from the continuous model from our Danish study, we estimated that the PAFs of pancreatic cancer before age 70 y will be 12.7% and 11.4% for men and women, respectively.
Discussion
In this large, prospective, population-based study that used measured heights and weights in childhood, we found that BMI at each age from 7 to 13 y was significantly and positively associated with pancreatic cancer in adulthood diagnosed up until age 70 y; beyond this age, there were no associations. Repeatedly having a high BMI at ages 7, 10, and 13 y was not significantly different from having a high BMI 1 time. Associations were similar for men and women and consistent for cancer diagnosis from 30 to 70 y of age. Associations between child height and pancreatic cancer, although positive, were not significant. We think our study is novel because it examines child body size in relation to the risk of pancreatic cancer in adulthood and other investigations focused on body size at older ages.
Our findings are consistent with previous studies that examined adolescent or early adulthood BMI and pancreatic cancer (11, 27). Within a cohort of 720,000 Israeli men with BMI measured at ages 16–19 y, adolescent overweight as defined by a BMI z score >1, compared with a z score ≥−1 to ≤1, showed a significant 2-fold increase in pancreatic cancer risk (27). One case-control study found a significant increase in risk associated with a 5-unit increase in recalled BMI during adolescence and early adulthood (11). The American NIH-AARP Diet and Health Study Cohort (NIH-AARP) and 2 pooled analyses of cohort studies showed adolescent or early adulthood body size obtained by recall to be positively associated with pancreatic cancer (8, 9, 11). The associations were independent of BMI at an older age, suggesting that the risk was established to a certain extent in earlier life. In a study in American retired adults (50–71 y of age) in the NIH-AARP, adolescent body size obtained by recall had a positive association with pancreatic cancer (12). The effect was not attenuated by including adult size, thus suggesting that the risk was established to a certain extent in earlier life. Because adult body size was not available in the current study, we could not conduct a fully comparable analysis. When BMI was analyzed as a continuous variable by using recalled height and weight at age 50 y from participants in the NIH-AARP study, the HRs for the association between BMI and pancreatic cancer were 1.18 (95% CI: 1.07, 1.32) for men and 1.02 (95% CI: 0.94, 1.12) for women per z score (R Stolzenberg-Solomon, M Gamborg, unpublished data, 2016). Interestingly, these associations were of a smaller magnitude than those observed in the current study per BMI z score, which suggests that tracking of childhood BMI whereby heavy children become heavy adults is not the only explanation for the observed results.
Prospective studies have also shown that BMI gain during adulthood and after age 50 y is associated with an elevated risk of pancreatic cancer (9, 10, 12). Given that we did not observe an association between childhood BMI and pancreatic cancer diagnosed after age 70 y, it is possible that childhood adiposity contributes to pancreatic cancer diagnosed at younger ages, whereas adiposity related to weight gain during adulthood contributes more to occurrence of cancer diagnosis later at older ages.
Our results suggest that there is not a cumulative effect of excess adiposity during childhood associated with adult pancreatic cancer. In our study, the association between having a high BMI value >1 time from the ages of 7 to 13 y and subsequent pancreatic cancer did not differ significantly from the association observed for having a high BMI only 1 time (Table 2). In contrast, the NIH-AARP study showed that longer absolute and cumulative years of being overweight or obese [BMI (kg/m2) ≥25.0] were significantly associated with pancreatic cancer (12). It is possible that we examined too few years during childhood to detect a cumulative effect, that we had too few cases to distinguish such patterns, or that it is body size change across many years during mid- to late adult life that is important for pancreatic cancer development.
Despite evidence from several studies that suggests that adult height has a positive association with pancreatic cancer (4, 13), there is great heterogeneity in the literature, with later reports pointing toward the association being very modest in magnitude (28). In our study, the HRs for the associations between childhood height and pancreatic cancer were positive, but weak and not significant. Given that the CIs around the estimates were small, it is unlikely that our study was underpowered to detect an effect, but this cannot be ruled out. Nonetheless, our findings are in accord with the majority of the adult literature.
BMI is not a perfect indicator of adiposity in either adults or children. In childhood, it is correlated with height, and this reflects that the body composition of healthy tall children has a greater amount of lean and adipose tissue (29). However, even when we accounted for the effects of height beyond what is captured by BMI in the models, the associations between childhood BMI and pancreatic cancer remained virtually unchanged. These results suggest that some aspect of body composition, presumably the adipose tissue component, is contributing to our observed associations. Adipose tissue is an endocrinologically active tissue that is known to be linked to pancreatic inflammation, β cell dysfunction, insulin resistance, and diabetes (30). These processes have been suggested to play an etiologic role in adulthood pancreatic cancer (31) and may likewise contribute to our association between childhood BMI and pancreatic cancer. Excess childhood weight may also be linked to later pancreatic cancer through correlations between child and adult size or through independent processes that stimulate early steps of carcinogenesis. Genes involved in the embryonic development of pancreas have been associated with BMI, diabetes, as well as pancreatic adenocarcinoma (32–34), which lends support to early-life exposures that affect the pancreas having a role in the pathways that lead to the development of pancreatic cancer later in life. Future research is warranted to determine whether the association between childhood excess weight and risk of pancreatic cancer later in life is mediated through adult adiposity and to understand the biological mechanisms that explain the associations that we observed.
Several strengths of our study should be noted, including the prospective design and the availability of measured, instead of recalled, height and weight during childhood. Furthermore, selection bias is unlikely to be present in our study, because it included virtually all children born from 1930 to 1982 who attended school in Copenhagen and we were able to follow these individuals from 1977 onward with the Danish registers. Pancreatic cancer diagnoses were obtained from the Danish Cancer Registry, which has a high coverage due to the mandatory reporting of all malignant neoplasms. In Denmark, health care is universal and paid for by taxation, so it is unlikely that this study is affected by access bias. Furthermore, there was minimal loss to follow-up because all subjects could be efficiently tracked through the Danish registry system. Information on family history of pancreatic cancer, childhood diet and second-hand smoke exposure, adult body size, and other adult health conditions (e.g., diabetes and pancreatitis) and exposures (e.g., adult smoking history) was not available. Exposures in adulthood, however, are unlikely confounders of measured childhood height and weight. Furthermore, some of these factors may be mediating factors on the causal pathway from obesity and pancreatic cancer. There may also be other unknown exogenous exposures as well as genetic susceptibility (35) contributing to the pancreatic cancer associations that we observed that cannot be controlled for in our present analysis. Our Danish population was primarily white, and our results might not be generalizable to other ethnicities. Nonetheless, our results along with those of others suggest that childhood BMI is associated with future pancreatic cancer and that body size across the life span contributes to this disease (8, 9, 11).
Childhood overweight and obesity in children have increased in Denmark (36) and across the globe (14). Our results show that risks are significantly elevated even at BMI z scores of 1.0 (84.1 percentile) in our study population. For example, when compared with the contemporary US reference for 13-y-old children, our BMI z scores of 1.0 (BMI of 20.9 in girls and 20.3 in boys) are equivalent to BMI values that fall just below the 75th percentiles. Thus, elevated risks are being observed at BMI values far below those that are currently classified as “overweight” by the CDC reference. To place our results in perspective, we calculated PAFs by using the prevalence of overweight and obesity in contemporary American children (>36% among 13-y-old boys and girls). If contemporary children moved from being overweight (including obesity) to normal weight at age 13 y, 12.7% and 11.4% of cases of pancreatic cancer before age 70 y might be avoided among men and women, respectively. These estimations, even with the limitations of their assumptions of causality, underscore that there is cause for concern about the future risk of pancreatic cancer in children of today.
In conclusion, we found that childhood BMI has a strong and positive association with pancreatic cancer in adulthood before age 70 y. The association between having a high childhood BMI repeatedly during the ages 7–13 y and pancreatic cancer did not significantly differ from having a high BMI at only one age during this age period. Associations between child height and pancreatic cancer were not significant. These results support the hypothesis that excess BMI in childhood contributes to the future risk of pancreatic cancer before 70 y of age.
Supplementary Material
Acknowledgments
The authors' responsibilities were as follows—TIAS and JLB: acquired the data; LN, RS-S, MG, TIAS, and JLB: designed the research; LN, RS-S, MG, and JLB: conducted the research; MG: analyzed the data; LN, RS-S, and JLB: wrote the manuscript; JLB: had primary responsibility for final content; and all authors: read and approved the final manuscript.
Footnotes
Supported by the European Research Council Starting Grant childgrowth2cancer FP7/2007-2013 (grant agreement 281419; JLB) and by the Intramural Research Program of the NIH, Division of Cancer Epidemiology and Genetics, National Cancer Institute (RS-S).
References
- 1. Ryerson AB, Eheman CR, Altekruse SF, Ward JW, Jemal A, Sherman RL, Henley SJ, Holtzman D, Lake A, Noone AM, et al. . Annual report to the nation on the status of cancer, 1975-2012, featuring the increasing incidence of liver cancer. Cancer 2016;122:1312–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Engholm G, Ferlay J, Christensen N, Kejs AMT, Hertzum-Larsen R, Johannesen TB, Khan S, Leinonen MK, Ólafsdóttir E, Petersen T, et al. NORDCAN: cancer incidence, mortality, prevalence and survival in the Nordic countries, version 7.3 (08.07.2016) [Internet]. Association of the Nordic Cancer Registries. Danish Cancer Society. [cited 2017 May 21]. Available from: http://www-dep.iarc.fr/NORDCAN/english/frame.asp.
- 3. Howlader N, Noone AM, Krapcho M, Miller D, Bishop K, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z, et al. , editors. SEER Cancer Statistics Review, 1975-2013 [Internet] Bethesda (MD): National Cancer Institute; 2016. [cited 2017 May 21]. Available from: http://seer.cancer.gov/csr/1975_2013/. [Google Scholar]
- 4. World Cancer Research Fund/American Institute for Cancer Research Continuous update project report: food, nutrition, physical activity, and the prevention of pancreatic cancer. 2012. [cited 2016 Sep 8]. Available from: http://www.dietandcancerreport.org.
- 5. Larsson SC, Orsini N, Wolk A.. Body mass index and pancreatic cancer risk: a meta-analysis of prospective studies. Int J Cancer 2007;120:1993–8. [DOI] [PubMed] [Google Scholar]
- 6. Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M.. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 2008;371:569–78. [DOI] [PubMed] [Google Scholar]
- 7. Arslan AA, Helzlsouer KJ, Kooperberg C, Shu XO, Steplowski E, Bueno-de-Mesquita HB, Fuchs CS, Gross MD, Jacobs EJ, LaCroix AZ, et al. . Anthropometric measures, body mass index, and pancreatic cancer: a pooled analysis from the Pancreatic Cancer Cohort Consortium (PanScan). Arch Intern Med 2010;170:791–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Jiao L, de Gonzalez AB, Hartge P, Pfeiffer RM, Park Y, Freedman DM, Gail MH, Alavanja MCR, Albanes D, Freeman LEB, et al. . Body mass index, effect modifiers, and risk of pancreatic cancer: a pooled study of seven prospective cohorts. Cancer Causes Control 2010;21:1305–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Genkinger JM, Spiegelman D, Anderson KE, Bernstein L, van den Brandt PA, Calle EE, English DR, Folsom AR, Freudenheim JL, Fuchs CS, et al. . A pooled analysis of 14 cohort studies of anthropometric factors and pancreatic cancer risk. Int J Cancer 2011;129:1708–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Genkinger JM, Kitahara CM, Bernstein L, Berrington de Gonzalez A, Brotzman M, Elena JW, Giles GG, Hartge P, Singh PN, Stolzenberg-Solomon RZ, et al. . Central adiposity, obesity during early adulthood, and pancreatic cancer mortality in a pooled analysis of cohort studies. Ann Oncol 2015;26:2257–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Li D, Morris J, Liu J.. Body mass index and risk, age of onset, and survival in patients with pancreatic cancer. JAMA 2009;301:2553–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stolzenberg-Solomon RZ, Schairer C, Moore S, Hollenbeck A, Silverman DT.. Lifetime adiposity and risk of pancreatic cancer in the NIH-AARP Diet and Health Study cohort. Am J Clin Nutr 2013;98:1057–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Aune D, Vieira A, Chan D, Navarro Rosenblatt D, Vieira R, Greenwood D, Cade J, Burley V, Norat T.. Height and pancreatic cancer risk: a systematic review and meta-analysis of cohort studies. Cancer Causes Control 2012;23:1213–22. [DOI] [PubMed] [Google Scholar]
- 14. Wang Y, Lim H.. The global childhood obesity epidemic and the association between socio-economic status and childhood obesity. Int Rev Psychiatry 2012;24:176–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Baker JL, Olsen LW, Sørensen TIA.. Childhood body-mass index and the risk of coronary heart disease in adulthood. N Engl J Med 2007;357:2329–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Reilly JJ, Kelly J.. Long-term impact of overweight and obesity in childhood and adolescence on morbidity and premature mortality in adulthood: systematic review. Int J Obes (Lond) 2011;35:891–8. [DOI] [PubMed] [Google Scholar]
- 17. Park MH, Falconer C, Viner RM, Kinra S.. The impact of childhood obesity on morbidity and mortality in adulthood: a systematic review. Obes Rev 2012;13:985–1000. [DOI] [PubMed] [Google Scholar]
- 18. Kitahara CM, Gamborg M, Berrington de González A, Sørensen TIA, Baker JL.. Childhood height and body mass index were associated with risk of adult thyroid cancer in a large cohort study. Cancer Res 2014;74:235–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Baker JL, Olsen LW, Andersen I, Pearson S, Hansen B, Sørensen TIA.. Cohort profile: the Copenhagen School Health Records Register. Int J Epidemiol 2009;38:656–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pedersen CB, Gotzsche H, Moller JO, Mortensen PB.. The Danish Civil Registration System—a cohort of eight million persons. Dan Med Bull 2006;53:441–9. [PubMed] [Google Scholar]
- 21. Baker JL, Sørensen TIA.. Obesity research based on the Copenhagen School Health Records Register. Scand J Public Health 2011;39(7Suppl):196–200. [DOI] [PubMed] [Google Scholar]
- 22. Cole TJ.. The LMS method for constructing normalized growth standards. Eur J Clin Nutr 1990;44:45–60. [PubMed] [Google Scholar]
- 23. Gjerstorff ML.. The Danish Cancer Registry. Scand J Public Health 2011;39:42–5. [DOI] [PubMed] [Google Scholar]
- 24. Rockhill B, Newman B, Weinberg C.. Use and misuse of population attributable fractions. Am J Public Health 1998;88:15–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kuczmarski RJ, Odgen CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R, Mei Z, Curtin LR, Roche AF, Johnson CL.. CDC growth charts: United States. Hyattsville (MD): National Center for Health Statistics; 2000. [PubMed] [Google Scholar]
- 26. CDC, National Center for Health Statistics National Health and Nutrition Examination Survey: questionnaires, datasets, and related documentation [Internet]. [cited 2016 Apr 25]. Available from: http://www.cdc.gov/nchs/nhanes/nhanes_questionnaires.htm.
- 27. Levi Z, Kark JD, Afek A, Derazne E, Tzur D, Furman M, Gordon B, Barchana M, Liphshitz I, Niv Y, et al. . Measured body mass index in adolescence and the incidence of pancreatic cancer in a cohort of 720,000 Jewish men. Cancer Causes Control 2012;23:371–8. [DOI] [PubMed] [Google Scholar]
- 28. Wirén S, Häggström C, Ulmer H, Manjer J, Bjørge T, Nagel G, Johansen D, Hallmans G, Engeland A, Concin H, et al. . Pooled cohort study on height and risk of cancer and cancer death. Cancer Causes Control 2014;25:151–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Freedman DS, Khan LK, Serdula MK, Dietz WH, Srinivasan SR, Berenson GS.. Inter-relationships among childhood BMI, childhood height, and adult obesity: the Bogalusa Heart Study. Int J Obes Relat Metab Disord 2004;28:10–6. [DOI] [PubMed] [Google Scholar]
- 30. Alempijevic T, Dragasevic S, Zec S, Popovic D, Milosavljevic T.. Non-alcoholic fatty pancreas disease. Postgrad Med J 2017;93:226–30. [DOI] [PubMed] [Google Scholar]
- 31. Stolzenberg-Solomon RZ, Amundadottir LT.. Epidemiology and inherited predisposition for sporadic pancreatic adenocarcinoma. Hematol Oncol Clin North Am 2015;29:619–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Li D, Duell EJ, Yu K, Risch HA, Olson SH, Kooperberg C, Wolpin BM, Jiao L, Dong X, Wheeler B, et al. . Pathway analysis of genome-wide association study data highlights pancreatic development genes as susceptibility factors for pancreatic cancer. Carcinogenesis 2012;33:1384–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wolpin BM, Rizzato C, Kraft P, Kooperberg C, Petersen GM, Wang Z, Arslan AA, Beane-Freeman L, Bracci PM, Buring J, et al. . Genome-wide association study identifies multiple susceptibility loci for pancreatic cancer. Nat Genet 2014;46:994–1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Pierce BL, Austin MA, Ahsan H.. Association study of type 2 diabetes genetic susceptibility variants and risk of pancreatic cancer: an analysis of PanScan-I data. Cancer Causes Control 2011;22:877–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Carrera S, Sancho A, Azkona E, Azkuna J, Lopez-Vivanco G.. Hereditary pancreatic cancer: related syndromes and clinical perspective. Hered Cancer Clin Pract 2017;15:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Bua J, Olsen LW, Sørensen TIA.. Secular trends in childhood obesity in Denmark during 50 years in relation to economic growth. Obesity (Silver Spring) 2007;15:977–85. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.