Abstract
Professional burnout syndrome has been described in association with insomnia and metabolic, inflammatory and immune correlates. We investigated the interest of exploring biological parameters and sleep disturbances in relation to burnout symptoms among white-collar workers. Fifty-four participants with burnout were compared to 86 healthy control participants in terms of professional rank level, sleep, job strain (Karasek questionnaire), social support, anxiety and depression (HAD scale). Fasting concentrations of glycaemia, glycosylated hemoglobin (HbA1C), total-cholesterol, triglycerides, C-reactive protein (CRP), thyroid stimulating hormone (TSH), 25-hydroxyvitamin D (25[OH]D), and white blood cell (WBC) counts were assessed. Analysis of variance and a forward Stepwise Multiple Logistic Regression were made to identify predictive factors of burnout. Besides reporting more job strain (in particular job control p = 0.02), higher levels of anxiety (p<0.001), and sleep disorders related to insomnia (OR = 21.5, 95%CI = 8.8–52.3), participants with burnout presented higher levels of HbA1C, glycaemia, CRP, lower levels of 25(OH)D, higher number of leukocytes, neutrophils and monocytes (P<0.001 for all) and higher total-cholesterol (P = 0.01). In particular, when HbA1c is > 3.5%, the prevalence of burnout increases from 16.6% to 60.0% (OR = 4.3, 95%CI = 2.8–6.9). Strong significant positive correlation existed between HbA1C and the two dimensions (emotional exhaustion and depersonalization (r = 0.79 and r = 0.71, p<0.01)) of burnout. Models including job strain, job satisfaction, anxiety and insomnia did not predict burnout (p = 0.30 and p = 0.50). However, when HbA1C levels is included, the prediction of burnout became significant (P = 0.03). Our findings demonstrated the interest of sleep and biological parameters, in particular HbA1C levels, in the characterization of professional burnout.
Introduction
The professional burnout is a psychological syndrome occurring in response to chronic job stress. The most widely used Maslach Burnout Inventory (MBI) measures burnout across the three dimensions, emotional exhaustion (EE), personal accomplishment (PA) and depersonalization (DP) [1,2]. It is an outcome of chronic depletion of the individual’s coping resources resulting from prolonged exposure to stress, particularly work-related stress [3]. Burnout in workplaces has been attributed to different factors, which can be classified as organizational and individual factors [3] and occurs in workers as a result of failure to cope with occupational stress [3]. Originally, the burnout symptoms were found to occur only in client-related occupations [4], but now include all types of work [5]. Most of the studies that estimate the prevalence of burnout focused on different occupational group primarily service—related professions with rates ranging from 25 to 60% [6–9]. With respect to stress measurement, the demand-control-support model of Karasek [10] is classically used and posits that job strain results from the joint effects of high job demand and low job control. This model of work stress has been widely used to examine adverse effects on health, such as type 2 diabetes and cardiovascular diseases, and mental illness [11]. There are now growing evidence supporting its negative implications for mental and physical health and recently accumulated evidence suggests that it may be considered a risk factor for severe injuries [1,3,12]. Thus, chronic job strain, has been found to predict cardiovascular disease [13], type 2 diabetes [13,14], and poor self-rated health [15]. High job strain increased the risk of mortality particularly in white-collar men [16]. Several epidemiological studies have implicated biological parameters such as level of glycosylated hemoglobin (HbA1C), which positively correlates with stress level regardless of the method of stress assessment: “job strain” with job demand-control model [17,18], or “job stress” evaluated by the effort-reward imbalance (ERI) model [[19].
Actually, burnout is not recognized as a disease and its definition remains uncertain, and there are no current standardized diagnostic criteria [20]. Some authors suggest that it is less a medical condition but more a phenomenon related to the match between a worker’s and his / her workplace’s expectations, and that this should be approached by management techniques rather than by treatment [21]. Fatigue, mood disorder, sleep problems and cognitive impairment seem to be the most common symptoms of burnout [22]. The fatigue is a dominant component in burnout but alternative or complementary factors could be disturbed sleep and mental disorders [23–25]. However, besides the mental health consequences of burnout, the negative physical health impact is important. In 2003, Grossi et al. [26], find burnout to be positively associated with HbA1C and TNF-α among women. The review of Melamed et al., (2006) [27] enlightened association between burnout and a subset of bio-clinical parameters (i.e. the metabolic syndrome, type 2 diabetes, systemic inflammation, impaired immunity functions) that may impact health more extensively than currently suggested. For them, allostatic load might be a mediating mechanism. However, the most recent systematic review on burnout, incorporating different biomarkers, has found no convincing relationship between biomarkers and burnout. For these authors, the “methodological inconsistencies” lead to the incomparability of studies [28].
We hypothesized that chronic exposure to job strain which leads to burnout has a specific biological signature, in particular metabolic and inflammatory responses. Our aim was to assess potential clinical and biological variables associated with burnout in a group of white collars workers using a cross-sectional multidimensional study. We focused on biological parameters, job strain and social support, and sleep and mental disorders. Symptomatic burned-out workers were compared to control healthy workers who were selected during the occupational medicine survey in a French financial company.
Materials and methods
Study design
We constructed a case-control study from a French working population. All subjects who were included were regular contract employees from a large financial French company. They were included during a three-year period study between September 2012 to September 2015. The analysis included white collar workers with symptomatic burnout (cases) and examined differences in clinical, professional and biological outcomes with non-burnout workers (healthy controls) from the same company. Study purposes and procedures were explained to the employees, and written informed consent was obtained from them prior the study. The procedures were in accordance with the Declaration of Helsinki and the study was approved by the Ethical Committee of the Hôtel Dieu Hospital, Paris, France.
Subjects
Cases were defined as subjects with no history of physical or mental disorder who consult as a matter of urgency for a psychological complaint or distress spontaneously related to work stress environment at the company’s occupational health center on site. All questionnaires, clinical interviews and examinations were conducted by the occupational physician and secondly addressed to a psychiatrist if necessary.
Subjects with a mood disorder, alexithymia, and other mental conditions such as anxiety disorders (generalized anxiety disorder, panic disorder, specific phobia, post-traumatic stress disorder…) that could overlap with burnout were excluded. During the period study, 9 subjects were addressed to the psychiatrist and were excluded because of a chronic severe psychiatric disorder (3 with major depressive disorders, 2 with personality disorders, 2 with a bipolar disorder, 1 panic disorder and 1 PTSD). Second, we limited enrollment to subjects without a history or previous diagnosis of metabolic and endocrinological problem, coronary heart disease (CHD), inflammatory diseases, allergies. Two subjects were excluded: 1 with hyperthyroidism and 1 with a type 2 diabetes.
After excluding other diagnosis and assessing the severity of burnout using the Maslach Burnout Inventory (MBI) questionnaire [2], each subject was included in the study.
Healthy controls came from the same company and were selected at random from other regular contract employees during the routine occupational health examination performed at the same day or week during which each case was included. They were defined as subjects who do not experience the outcome (i.e. burnout syndrome) during the period study. We matched every person with burnout as closely as possible by age, sex, BMI, and income category with healthy controls.
After an overnight fast, all participants were invited to a morning lab visit. This included the assessment of socio-demographic and work characteristics using self-report questionnaires; including age, gender, weight, body mass index (BMI), occupational rank, and subjective quality of life using a visual analogue scale (VAS).
During the study period, 54 burnout subjects have been included (30 men and 24 women) and matched with 86 healthy controls (41 men and 45 women).
Data collection
Burnout
The severity of burnout symptoms was assessed with the widely used Maslach Burnout Inventory (MBI). It is a questionnaire consisting of 22 items, distributed in three dimensions [2]: 9 items for Emotional Exhaustion (EE) (a drained, depleted feeling arising because of excessive psychological and emotional demands), 5 items for depersonalization (DP) (tendency to view others in an excessively detached, impersonal manner), and 8 items for Professional accomplishment (PA) (a sense of competence and accomplishment).
For the categorical definition of burnout, the cut-offs were chosen according to the scoring guidelines by MindGarden (Menlo Park [CA], USA). Burnout was identified at a high level (as opposed to low) based on the following scores: EE > 27, DP > 13 and PA < 30. A diagnosis of burnout (yes/no) was assigned if respondents presented high levels in at least two dimensions (either EE or/ and DP, associated or not with a low PA).
In this study, in order to analyze the internal consistency of the three domains of the Maslach Burnout Inventory (emotional distress, depersonalization and low professional fulfillment), the alpha Cronbach coefficient was respectively 0.86 for Emotional Exhaustion, 0.71 for depersonalization and 0.70 for personal achievement.
Psychosocial working conditions
They were evaluated from job strain levels which delineate job control and job demand scores from the validated French version of the Karasek job content questionnaire [11] completed during the occupational medical visit. Participants were asked to answer questions about the psychosocial aspects of their job. The response categories were a Likert-scale ranging from 1 (strongly disagree) to 4 (strongly agree). For each participant, mean response scores were calculated for job-demand items (i.e, questions about whether the participant had to work very hard, had excessive amounts of work, conflicting demands, or insufficient time) and job-control items (i.e., questions about decision freedom and learning new things at work). Scores on the job demands scale range from 12 to 48, with higher scores (> 24) representing higher demands. Scores on the job control scale range from 24 to 96, with higher scores representing higher control (> 72). Cut-off for each dimension was dichotomized to the median to define high and low score, from our previous study with a largest sample of the working population to which every included subject belongs [23]. In our study, the following Cronbach’s alpha coefficients were: job-demand (0.78), job control (0.76).
Sleep disturbances
Insomnia and other sleep disorders were assessed using a self-administered questionnaire, derived from the ‘Sleep Disorders Questionnaire—French version’ (SDQFV). The SDQFV is a 42-items questionnaire based on the “Stanford Sleep Questionnaire and Assessment of Wakefulness”. The French version has been validated in epidemiological studies [23,29].
In our study, we used part of the questionnaire that focused on sleep habits and insomnia. To assess insomnia symptoms, we add questions based on new reference documents: the “International Classification of Sleep Disorders” (ICSD-3) and the “Diagnostic and Statistical Manual of Mental Disorders”, 5th revision (DSM-5). Clinical reappraisal was carried out with all subjects by an occupational health and sleep medicine specialist. The clinical interviews were carried out to establish a diagnosis of insomnia with DSM-5. Insomnia was assessed using the following questions: “Do you have difficulty falling asleep at night?” (Difficulty in initiating sleep), “Do you wake up during the night after you have gone to sleep and have difficulty getting back to sleep?” (Difficulty in maintaining sleep), and “Do you wake up too early in the morning and have difficulty getting back to sleep?” (Early morning awakening). Each positive response was considered as “trouble” if occurring at least during “three nights per week”.
The DSM-5 criteria for insomnia include difficulty of initiating sleep, difficulty of maintaining sleep, and early morning awakening, for a period of ≥ 1 month. In addition, it is a prerequisite that sleep disturbance significantly impairs daily function. Severity of insomnia was divided in two groups: moderate insomnia (one trouble) and severe insomnia (at least two troubles). Non-restorative sleep disorder was assessed separately using the question “How frequently are you bothered because your sleep is not refreshing, you don’t feel rested even if the duration of your sleep is normal.” Non-restorative sleep was considered present when it was reported to occur 3 to 4 times a week or more and lasted for at least 1 month. Excessive sleepiness was assessed using the “Epworth Sleepiness Scale” (ESS) [30]. Based on the total score, patients were classified in two subgroups of sleepiness: no sleepiness = 0–10, excessive sleepiness > 10.
Mental health (anxiety and depression)
Symptom levels of depression and anxiety were assessed by the Hospital Anxiety and Depression Scale (HADS) [31] consisting of seven items for each dimension. Answers are coded on a 4-point Likert scale (0 = not at all, 3 = mostly), giving rise to a total depression and anxiety score ranging between 0 and 21 points. A > 10 score is considered as certainly symptomatic.
Biochemical parameters
Venous blood samples were collected in EDTA tubes between 8 and 10 a.m. in seated subjects who had fasted overnight. White blood cell (WBC) counts were determined with automated blood cell counters. Serum total-cholesterol, HDL-cholesterol, triglycerides, C-reactive protein (CRP) and glucose were measured using the automatic analyzer COBAS (Roche Diagnostics, Switzerland), all reagents were prepared according to the manufacturer’s instructions. LDL-cholesterol was calculated using the Friedewald formula. 25[OH]D3 was measured in one batch using the Roche Cobas E601 analyzer, according to the manufacturer’s instructions (vitamin D3 [25-OH] assay with polyclonal antibody; Roche Diagnostics, Burgess Hill, UK). Thyroid stimulating hormone (TSH) was measured by electrochemiluminescence immunoassay (Roche Cobas E 601, module immunology analyzer, Roche Diagnostics, Burgess Hill, UK). The glycosylated hemoglobin (HbA1C) test was performed using reagents, calibrators and control materials from Bio-Rad D 100 ion-exchange high-performance liquid chromatography (HPLC) (Bio-Rad Laboratories, Inc. USA) according to the manufacturer’s instructions.
Data management and statistical methods
A database was developed using the software Sphinx Plus2 (V 5.1, Le Sphinx development, Chavanod, France) and statistics tests were made using R studio (Version 0.99.175–2009–2014 RStudio, Inc.) and significance (α risk) was fixed at p<0.05. Continuous variables were presented as mean ± standard deviation (SD) and means were compared (Burnout vs. Control) using a 2-tailed t test. Dichotomous variables were presented as occurrence and percentage (n (%)). A χ2 (or Fisher exact test) was used to test the relationship between the variable and Burnout. When significant, odd ratio and his 95% confidence interval (OR [95% CI]) was calculated. Dependences between quantitative variables was checked using a Pearson correlation test (r ≥ 0.6 and p<0.05). Dependence between quantitative variable and burnout was test using a one-way analysis of variance (ANOVA). A multiple logistic regression was made to identify factors with outcome and estimate the probability for burnout diagnostic. Variables were entered as independent variables and presence of burnout was entered as the dependent variable. A value of p < 0.05 for the Wald criterion was considered to denote regression coefficients significantly different from zero. The results are shown as odds ratio (OR) with 95% confidence intervals (95% CI) for ORs. The fit of the models was judged by the Likelihood Ratio Test Statistic.
A Forward Stepwise Multiple Logistic Regression was made to identify predictive factors (independent variables) of burnout (dependent variables). We included in the models the independent factors significantly associated with burnout, in the model 1: Job satisfaction, HADS anxiety, Job control and Job demand, in the model 2: + insomnia, in the model 3: + HbA1C and in the model 4 + insomnia and HbA1C in order to evaluated respective influence of HbA1C and insomnia in the diagnosis of burnout. A value of p < 0.05 for the Wald criterion was considered to denote regression coefficients significantly different from zero. The results are shown as odds ratio (OR) with 95% confidence intervals (95% CI) for ORs. The fit of the models was judged by the Likelihood Ratio Test Statistic.
Results
We compared 54 men and women aged 37.1 ± 7.4 y with symptomatic burnout to 86 healthy individuals matched for age, sex, BMI and professional rank. As expected, subjects with burnout scored higher on the two-related burnout dimensions, EE and DP, while lower on the PA score, compared to participants without burnout (Table 1).
Table 1. Sociodemographic and psychological characteristics of burnout and non-burnout (control) subjects.
| Control | Burnout | Anova p-value | χ2, OR (95% CI) | |
|---|---|---|---|---|
| Number, n | 86 | 54 | ||
| Women, n (%) | 45 (52.2%) | 24 (44.4%) | NS | |
| Age, yr | 30.8 ± 7.1 | 31.7 ± 7.4 | 0.51 | |
| Weight, Kg | 68.3 ± 13.4 | 67.0 ± 13.1 | 0.55 | |
| BMI, kg/m2 | 22.7 ± 3.2 | 22.4 ± 3.2 | 0.57 | |
| Rank /Level, n (%) | ||||
| Assistant | 6 (7.0%) | 4 (7.4%) | NS | |
| Associate | 2 (2.3%) | 2 (3.7%) | NS | |
| Director | 3 (3.4%) | 2 (3.7%) | NS | |
| Senior | 59 (68.6%) | 33 (61.1%) | NS | |
| Senior manager | 11 (12.8%) | 9 (16.6%) | NS | |
| Emotional exhaustion | 10.2 ± 3.5 | 37.9 ± 6.0 | <0.001 | |
| Depersonalization | 6.6 ± 2.7 | 17.8 ± 3.9 | <0.001 | |
| Accomplishment | 40.2 ± 2.0 | 29.6 ± 4.1 | <0.001 | |
| Job strain | ||||
| Job control | 64.2 ± 8.4 | 67.7 ± 9.3 | 0.02 | |
| Job demand | 27.1 ± 3.6 | 27.2 ± 3.5 | 0.5 | |
| Social support | 22.2 ± 4.3 | 22.1 ± 3.8 | 0.8 | |
| Job satisfaction | 7.1 ± 1.5 | 5.7 ±1.9 | 0.01 | |
| Quality of life (VAS) | 7.38 ± 1.29 | 7.07 ± 1.85 | 0.25 | |
| HAD anxiety | 7.1 ± 2.0 | 10.1 ± 3.4 | <0.001 | |
| Score HADa > 10 | 30 (34.8%) | 7 (12.9%) | 35.9*, 13.1 (5.5–36.1) | |
| HAD depression | 5.5 ± 2.4 | 6.7 ± 3.4 | 0.02 | |
| Score HADd > 10 | 0 | 5 (100%) |
Values are: Mean ± SD or occurrence (%).
* p<0.05.
Sleep and mental response to chronic work stress in burnout subjects
The group with burnout has significantly higher insomnia troubles, sleep fragmentation, and non-restorative sleep than the control group. In addition, burnout subjects have higher levels of anxiety and depression scores. About job strain characteristics, job control is significantly higher for burnout subjects. In addition, job satisfaction is lower for burnout subjects, and no significant difference was found between the two groups for social support (Tables 1 and 2).
Table 2. Sleep parameters in burnout and non-burnout [control] subject.
| Control | Burnout | ANOVA p-value | χ2, OR (95% CI) | |
|---|---|---|---|---|
| TST, h | 6.9 ± 0.7 | 6.7 ± 0.8 | 0.16 | |
| TST week, h | 6.7 ± 0.8 | 6.5 ± 0.9 | 0.23 | |
| TST week end, h | 8.9 ± 1.1 | 8.8 ± 1.1 | 0.37 | |
| Difference, h | 2.3 ± 1.2 | 2.3 ± 1.5 | 0.98 | |
| Epworth Sleepiness Scale (ESS) (/24) | 9.7 ± 4.0 | 9.4 ± 4.8 | 0.67 | |
| ESS >10, n (%) | 46 (53.5%) | 28 (51.9%) | NS | |
| ESS > 12, n (%) | 26 (30.2%) | 17 (31.5%) | NS | |
| ESS > 14, n (%) | 16 (18.6%) | 11(20.4%) | NS | |
| ESS > 16, n (%) | 10 (11.6%) | 6 (11.0%) | NS | |
| Insomnia n (%) | ||||
| Insomnia | 11 (12.8%) | 41 (75.9%) | 57*, 21.5 (8.8–52.3) | |
| Nb. troubles DSM 5 | 0.2 ± 0.7 | 1.6 ± 1.3 | <0.001 | |
| Sleep latency disorder | 3 (3.5%) | 11 (29.6%) | 19*, 11.6 (3.2–42.4) | |
| Nocturnal awaking | 7 (8.1%) | 21 (38.9%) | 20*, 7.2 (2.8–18.5) | |
| Early awaking | 3 (3.5%) | 19 (35.2%) | 25*,15.1(4.2–54.0) | |
| Non-restorative sleep | 7 (8.1%) | 27 (50.0%) | 32*,11.3 (4.4–28.9) | |
| Nap, n (%) | 11 (20.3%) | 18 (20.9%) | NS | |
| Snoring n (%) | 14 (25.9%) | 20 (28.3%) | NS |
Values are: Mean ± SD or occurrence (%),
* p<0.05.
Difference = (TST week end—TST week)
Biological response in burnout subjects
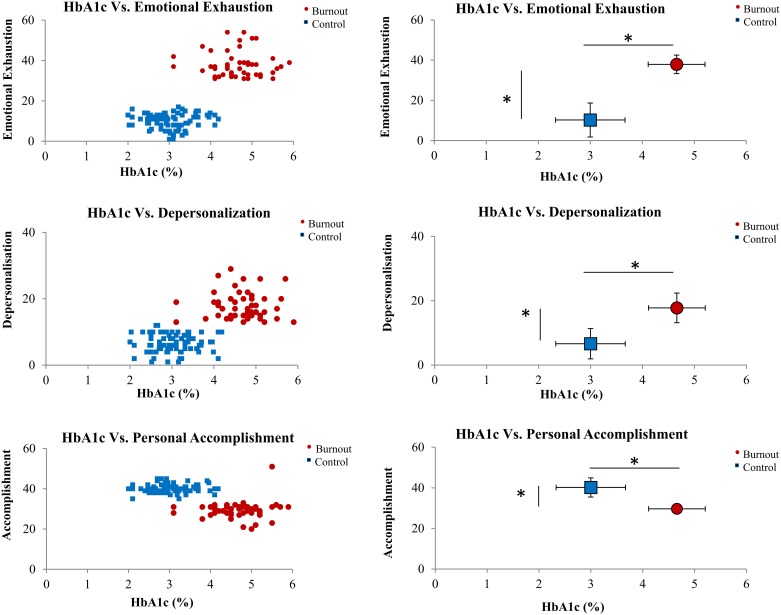
Differences were observed between the two groups of subjects for glycaemia, HbA1C, CRP, 25[OH]D, total-cholesterol, and leukocytes, neutrophils and monocytes cells counts (Table 3). Significant positive correlation (Table 4, Fig 1) existed between HbA1C and two dimensions (EE and DP) of burnout (r = 0.79 and r = 9.71, respectively) and negative correlation between 25[OH]D and EE (r = − 0.85). The third burnout dimension, PA, was negatively correlated with HbA1C (r = − 0.70), glycaemia, CRP and leukocytes and neutrophils counts, and positively with 25[OH]D (r = 0.60).
Table 3. Biological parameters in burnout and non-burnout (control) subjects.
| Control | Burnout | p | χ2, OR (95% CI) | |
|---|---|---|---|---|
| Glycaemia, g/L | 0.82 ± 0.13 | 0.89 ± 0.14 | 0.001 | |
| Glycaemia > 0.8 g/L | 41 (47.7%) | 40 (70.1%) | 8.4 *, 1.8 (1.2–2.0) | |
| HbA1C, (% | 3.0 ± 0.51 | 4.66 ± 0.57 | <0.001 | |
| HbA1C > 3.5% | 14 (16.6%) | 52 (60.0%) | 78.4*, 4.3 (2.8–6.9) | |
| CRP, mg/L | 1.2 ± 0.9 | 2.07 ± 1.8 | 0.001 | |
| CRP ≥ 3 mg/L | 7 (8.3%) | 14 (25.1%) | 6.9*, 2 (1.1–3.9) | |
| TSH, mUI/L | 1.95 ± 0.63 | 1.95 ± 0.80 | 0.999 | |
| 25(OH)D, ng/mL | 28.9 ± 4.9 | 17.7 ± 6.9 | <0.001 | |
| 25(OH)D<20 ng/mL | 4 (4.7%) | 33 (58.9%) | 51.3*, 4.4 (2.9–6.5) | |
| Total-cholesterol, mmol/L | 1.66 ± 0.33 | 1.81 ± 0.38 | 0.01 | |
| Triglycerides, mmol/L | 0.68 ± 0.35 | 0.74 ± 0.36 | 0.37 | |
| HDL, mmol/L | 0.62 ± 0.16 | 0.63 ± 0.15 | 0.48 | |
| LDL, mmol/L | 0.98 ± 0.32 | 1.03 ± 0.34 | 0.35 | |
| LDL/HDF | 0.70 ± 0.28 | 0.68 ± 0.28 | 0.78 | |
| Leukocytes, / mm3 | 5110 ± 535 | 6184 ± 1295 | <0.001 | |
| Neutrophils, / mm3 | 2460 ± 420 | 3418 ± 1248 | <0.001 | |
| Eosinophils, / mm3 | 155 ± 99 | 174 ± 126 | 0.32 | |
| Basophils, / mm3 | 39.5 ± 17.5 | 41.1± 19.8 | 0.61 | |
| Lymphocytes, / mm3 | 2002 ± 488 | 2068 ± 444 | 0.42 | |
| Monocytes, / mm3 | 463 ± 115 | 514 ± 133 | <0.001 | |
| Platelets, x 103 / mm3 | 253 ± 49 | 249 ± 44 | 0.68 |
Values are: Mean ± SD or occurrence (%),
*p<0.05
Table 4. The correlation analysis [Pearson coefficient correlation] between the three burnout dimensions [emotional exhaustion, depersonalization and personal accomplishment] and age, job strain [job demand-control-support], job satisfaction, sleep characteristics, anxiety and depression, and biological parameters.
| Emotional Exhaustion | Depersonalization | Personal accomplishment | |
|---|---|---|---|
| Age | 0.01 | 0.02 | -0.1 |
| Job demand | 0.08 | 0.04 | 0.02 |
| Job control | 0.17* | 0.18 | -0.12 |
| Job satisfaction | -0.04 | -0.31* | 0.33* |
| Social support | -0.05 | -0.12 | 0.09 |
| Nb. sleep troubles (DSM 5) | 0.65* | 0.45* | -0.51* |
| ESS | 0.01 | -0.02 | -0.02 |
| TST | -0.1 | -0.01 | 0.09 |
| HADS Anxiety | 0.52* | 0.45* | -0.50* |
| HADS Depression | 0.26* | 0.18 | -0.28* |
| Leucocytes | 0.46* | 0.44* | -0.44* |
| PNN | 0.45* | 0.47* | -0.39* |
| Glycaemia | 0.28* | 0.21 | -0.26* |
| HBA1C | 0.79* | 0.71* | -0.70* |
| Total-cholesterol | 0.22* | 0.19* | -0.17 |
| 25(OH)D | -0.67* | -0.59* | 0.60* |
| CRP | 0.28* | 0.15 | -0.20* |
Values are Pearson coefficient [R]
* is p<0.05
Fig 1. Comparison between HbA1C (individual and mean values (mean ± standard error) and the three burnout dimensions (emotional exhaustion, depersonalization and personal accomplishment) in control and burnout subjects.
*p<0.05.
The correlation analysis between biomarkers showed that HbA1C is negatively associated with 25[OH]D (r = − 0.73, p<0.01) and positively with total-cholesterol, glycaemia, CRP and the leukocytes and neutrophils counts (r = 0.56, p<0.01) (Table 5).
Table 5. The correlation analysis (Pearson coefficient correlation) between biological parameters.
| PNN | Glycemia | HbA1C | Cholesterol | 25(OH)D | CRP | |
| Leukocytes | 0.86* | 0.14 | 0.54* | 0.11 | -0.47 | 0.48 |
| PNN | 0.14 | 0.56* | 0.17 | -0.43 | 0.56* | |
| Glycaemia | 0.27* | 0.10 | -0.30* | 0.08 | ||
| HbA1C | 0.36* | -0.73* | 0.35* | |||
| Cholesterol | -0.25 | 0.12 | ||||
| 25(OH)D | -0.13 |
Values are Pearson coefficient (R)
* is p<0.05
In the Model 1 multiple logistic regression (Table 6), job control, job satisfaction and anxiety are significantly associated with burnout. These significant associations persist when insomnia is added in the Model 2 (Table 7). However, these models did not significantly predict burnout occurrence (Chi-square = 142 and 131, p = 0.30 and p = 0.50). When HbA1C is included in the Model 1, HbA1C became highly associated with burnout (p<0.001) while job control and job satisfaction were not significantly associated (Model 3, Table 8). When HbA1C and insomnia are included in the Model 1 (Model 4, Table 9), HbA1C remained highly associated with burnout (p<0.001), and insomnia also (p<0.04) (Model 4). In the models 3 and 4, HbA1C significantly predict burnout occurrence (Chi-square = 164 and 166, p = 0.03 for the two). HbA1C has been chosen because its association with burnout was the higher among the biological parameters. Moreover, there are significant correlations between biological parameters associated with burnout.
Table 6. Model 1 multiple logistic regression analysis for burnout diagnosis.
| Ind. variable | Coefficient | Standard Error | Wald Statistic | p | OR | 95%CI |
|---|---|---|---|---|---|---|
| Job satisfaction | -0.46 | 0.14 | 9.745 | 0.002 | 0.62 | (0.41–0.84) |
| HADS anxiety | 0.41 | 0.11 | 13.506 | <0.001 | 1.51 | (1.21–1.88) |
| Job control | 0.08 | 0.02 | 7.657 | 0.006 | 1.08 | (1.02–1.15) |
| Job demand | -0.02 | 0.06 | 0.143 | 0.70 | 0.98 | (0.86–1.11) |
Pearson Chi-square = 142 (p = 0.30), Likelihood Ratio Test Statistic: 54.0 (P = <0.001)
Table 7. Model 2 multiple logistic regression analysis for burnout diagnosis.
| Ind. variable | Coefficient | Standard Error | Wald Statistic | p | OR | 95%CI |
|---|---|---|---|---|---|---|
| Job satisfaction | -0.41 | 0.17 | 5.93 | 0.02 | 0.67 | (0.48–0.92) |
| HADS anxiety | 0.38 | 0.13 | 7.28 | 0.007 | 1.40 | (1.02–1.79) |
| Job control | 0.08 | 0.03 | 6.30 | 0.01 | 1.09 | (1.02–1.16) |
| Job demand | -0.05 | 0.08 | 0.40 | 0.53 | 0.95 | (0.82–1.11) |
| Insomnia | 1.26 | 0.29 | 19.02 | <0.001 | 3.52 | (1.21–6.2) |
Model 2 = model 1 + Insomnia. Pearson Chi-square = 131 (p = 0.50), Likelihood Ratio Test Statistic: 83.0 (P = <0.001)
Table 8. Model 3 multiple logistic regression analysis for burnout diagnosis.
| Ind. variable | Coefficient | Standard Error | Wald Statistic | p | OR | 95%CI |
|---|---|---|---|---|---|---|
| Job satisfaction | -0.17 | 0.31 | 0.31 | 0.58 | 0.84 | (0.46–1.54) |
| HADS anxiety | 0.42 | 0.21 | 4.00 | 0.05 | 1.52 | (1.01–2.30) |
| Job control | 0.04 | 0.05 | 0.62 | 0.42 | 1.04 | (0.94–1.15) |
| Job demand | -0.12 | 0.12 | 0.89 | 0.34 | 0.89 | (0.69–1.13) |
| HbA1C | 5.05 | 1.08 | 22.0 | <0.001 | 156 | (19.2–1293) |
Model 3 = model 1 + HbA1C. Pearson Chi-square = 164 (P = 0.03), Likelihood Ratio Test Statistic: 148.2 (P = <0.001)
Table 9. Model 3 multiple logistic regression analysis for burnout diagnosis.
| Ind. variable | Coefficient | Standard Error | Wald Statistic | p | OR | 95%CI |
|---|---|---|---|---|---|---|
| Job satisfaction | -0.02 | 0.34 | 0.03 | 0.95 | 0.98 | (0.50–1.91) |
| HADS anxiety | 0.34 | 0.23 | 2.22 | 0.13 | 1.41 | (0.89–2.20) |
| Job control | 0.052 | 0.05 | 1.06 | 0.30 | 1.05 | (0.95–1.16) |
| Job demand | -0.19 | 0.14 | 1.84 | 0.17 | 0.83 | (0.69–1.13) |
| Insomnia | 0.96 | 0.47 | 4.17 | 0.04 | 0.2.6 | (1.11–6.5) |
| HbA1C | 5.42 | 1.40 | 14.9 | <0.001 | 226 | (14.4–3325) |
Model 4 = model 1 + Insomnia + HbA1C. Pearson Chi-square = 166 (P = 0.03), Likelihood Ratio Test Statistic: 153.9 (P = <0.001)
Discussion
Our results show that blood concentrations of HbA1C, 25[OH]D and insomnia (particularly the number of troubles) are significantly associated with burnout in our population of white-collar workers. The emotional exhaustion dimension of burnout is particularly associated with the three parameters. In addition, as described previously in the large population (1,300 subjects), insomnia may be a risk factor of burnout, and job control is the only one component of job strain that is associated with the emotional dimension of burnout [32].
A growing base of research has linked disturbances of sleep duration to diabetes, obesity, and cardiovascular disorders, and work stress has been also evidenced to represent a possible risk factor for these diseases [19]. With respect to work stress measurement, job strain, the demand-control-support model of Karasek, is a risk factor of diabetes [33], and high concentration of HbA1C, an index of glucose homeostasis and an indicator of insulin resistance, is associated with high job strain and low social support [18,34]. Work social support is even offered as protective against diabetes diagnosed by HbA1C, while both underload and overload work may increase the risk [35]. To our knowledge, the study of Grossi et al. (2003) [26] evidence that high burnout in women is associated with high levels of HbA1C, independently of confounders including depression. In this study, participants with high scores for burnout reported less control and poorer social support at work, more emotional distress, and greater sleep impairments. According to the literature, we are the first to document that burnout, and particularly emotional exhaustion, the core burnout dimension, is associated with insomnia and HbA1C. We also found higher glycaemia levels in the burnout group but the measurement of HbA1C is recognized for greater monitoring of blood glucose and is more representative of microvascular complications; it has become the gold standard in the assessment of average blood glucose levels over time [36]. At last, subjects with burnout were also significantly higher than controls for the inflammatory biomarker CRP and total-cholesterol, and lower for 25[OH]D concentrations. The leukocyte number and particularly neutrophils and monocytes were also significantly higher. The identification of higher levels of HbA1C and inflammatory biomarkers in burnout together with low levels of 25[OH]D adds information on the latest systematic review which concluded that there are no potential biomarkers for burnout [28]. This review has analyzed 31 burnout studies incorporating 38 different biomarkers and has found any convincing relationship between biomarkers and burnout, due to methodological inconsistencies and vast heterogeneity of this ill-defined syndrome.
It has been evidenced that chronic sleep loss and behavioral or sleep-disorder related represent novel risk factors for weight gain, insulin resistance, and Type 2 diabetes [37]. A recent prospective cohort study demonstrated that long-term excessive daytime napping is associated with the development of elevated HbA1C levels and high HOMA-IR index (homeostasis model assessment of insulin resistance) over an average of 4.5 years of follow-up [38]. Our bivariate analys is showed a significant association between burnout and insomnia, sleep initiation and maintenance disorders or non-restorative sleep. We also found an association with levels of HbA1C when higher than 3.5%, glycaemia (> 0.80 g/L), and CRP (> 3 mg/L). In addition, significant correlations were found between (i) the three dimensions of burnout and HbA1C, 25[OH]D, troubles of insomnia, anxiety and depression, (ii) depersonalization and personal accomplishment and job satisfaction, and (iii) emotional exhaustion and job control. Finally, after adjusting for all potential factors influencing burnout, HbA1c levels and insomnia remain independent statistical risk factors. Our results confirm the Kachi et al [39] study showing that insomnia symptoms (specifically difficulty in maintaining sleep and early morning awakening) have a close association with high HbA1c in a dose-response relationship, in full-time white-collar workers. The association between insomnia and HbA1c level is not clearly elucidated in this study, and authors did not assess the relationship with work stress. Interestingly we confirmed an association between insomnia symptoms and HbA1c levels in the burned-out workers chronically exposed to work stress. In a sense, these two factors could be considered as a bio-clinical signature of chronic stress at workplace. Future prospective research should be conducted to demonstrate the causal pathway. Our findings could also reflect a stress-related dysregulation through multiple neuroendocrine systems. Indeed, Hirotsu et al. [40] has recently underlined the association between sleep disturbance and glucocorticoid dysregulation in response to chronic psychosocial stress. In addition, autonomic and glucocorticoid dysregulations are implicated in metabolic disorders such as the central fat deposition and the metabolic syndrome [41,42].
Another interesting finding of our study is the significant lower level of vitamin D in the burned-out subjects. Vitamin D level seems to be a potent risk factor when lower than 20 ng/mL. In addition, 25[OH]D are in significant negative correlation with HbA1C and glycaemia, and leukocyte count. Recently, low levels of serum 25[OH]D were found associated with short sleep duration and poorer sleep efficiency in a large study of older men [43], and independent inverse associations were found between 25(OH)D and sleepiness (as well as insomnia) [44]. The hypothesis that sleep disorders have become epidemic because of widespread vitamin D deficiency has been proposed, based on the hypothesis of the presence of vitamin D receptors in cerebral areas that play a role in the initiation and maintenance of sleep [45]. On the other hand, available data indicated that both type 1 and type 2 diabetes patients had lower levels of 25[OH]D than controls overall [46]. The mechanism of action of vitamin D in type 2 diabetes is thought to be mediated not only through regulation of plasma calcium levels which regulate insulin synthesis and secretion, but also through a direct action on pancreatic beta-cell function, and its role in inflammation, immunity and gene transcription [47]. In our study, the relationship between burnout and lower levels of 25[OH]D may be inherent to higher HbA1C and glycaemia, that themselves could signed presence of metabolic disorder related to insomnia troubles.
In a broad comprehensive approach encompassing our main findings, we suggest that association between insomnia symptoms, higher HbA1C and lower 25[OH] D levels in a burned-out group could be a specific physiological response to a high level of chronic job strain. In other words, it could also mean that burned employees, all of whom expressed psychological distress at the time of measurement, were close to the "allostatic overload" phase described by McEwen [47]. In our burned-out group, physiological effects could be delayed compared to the psychological effects of burnout. Moreover, insomnia symptoms might be either a protective mechanism for maintaining physical health or a step before irreversible damaging effects of stress. It has been suggested in some other studies cited by Melamed et al. (2006) [27] that allostatic load could represent a possible mechanism explaining the physical health consequences of burnout. A high allostatic load disrupts dynamic biological responses, resulting notably in inadequate biological responses, and higher risk for chronic diseases such as hypertension, obesity, cardiovascular disease, and diabetes [47,48]. The chronic allostatic load can be a mechanism by which stress exposure contributes to the risk of diabetes and is also implicated in the adverse health consequences of diabetes [34].
This study has several limitations, and caution should be taken when interpreting the results. First, this study was a descriptive, cross-sectional research design, therefore it was not possible to analyze the causal relationships between the variables. A prospective study is required to examine the link between burnout and HbA1C and 25[OH]D serum levels in our population of white-collar workers [19]. Secondly, the use of self-reporting questionnaires may have influenced the sample and the results. For example, burned-out subjects may have respond more negatively because of their negative felling at the time they filled out the questionnaire. In the future, it would be interesting for example to objectively investigate inter-relationship between elevated levels of HbA1C and sleep characteristics (e.g., fragmentation) using ultra-miniaturized PSG as previously used by us [49]. Moreover, we did not record the length of burnout experience that could be pertinent to study the causal link with the development of chronic inflammatory or metabolic diseases and/or the sleep disorders.
In conclusion, this study evidenced that serum levels of HbA1C higher than 3.5% and 25[OH]D lower than 20 ng/mL and insomnia symptoms are closely associated with burnout in white collar workers. Biological parameters and sleep disorders should be an interesting path to explore health consequences of burnout at work place. Thus, for better characterization of work-related burnout it would be interesting to develop an allostatic approach including subjective and objective sleep parameters that could open up new perspectives for research. Future studies should be prospective and focused on high job strain workers such as physicians or the large group of medical occupations who are more prone to burnout prevalence. At the end, our results confirm the need for enhancing the primary prevention in the workplace through social-behavioral approaches and management techniques.
Acknowledgments
We are immensely grateful to Mrs Y. Balika and Mrs S. Berot for their technical and administrative support. We also thankful to Mrs A. Rommel, Mrs J. Spinelli and Mr B. Gouache who assisted the research.
Data Availability
The study data set cannot be made publicly available due to ethical restrictions imposed by the Occupational Safety and Health Ethics Committee (CHSCT). However, a de-identified dataset can be made available on request from the corresponding author at arnaud.metlaine@aphp.fr. Additionally, data requests may also be sent to the following institutional address: Centre du Sommeil et de la vigilance de l’Hôtel-Dieu. APHP. 1 place de Parvis de Notre Dame-75181 Paris cedex 4 (France).
Funding Statement
This study was supported by the occupational health and safety programs of the subject’s employers. It did not receive specific founds. Indeed, in France, according to the regulatory framework for occupational health and safety, employers are required to take measures to ensure workers’ safety and protect their physical and mental health. In regards of French Labour Legislation, the occupational health and safety is exclusively financed by employers’ contributions in every company and the system is managed by the social partners. Occupational medical services are provided by occupational health officers whose exclusively preventive role lies in ensuring there is no deterioration in the health of workers owing to their work. The data collection including clinical examinations and biological analysis were done during the required occupational health survey of workers’ company. So, no additional financial support was necessary. The biological analyses were done by a private laboratory which usually collaborates with the company from which participants of the study were selected. Doctor Thierry Boucher (TB) is employed in this private laboratory which belongs to a commercial company Bio Paris Ouest. We declare this commercial affiliation. But this company was not a funder but provided support in the form of salaries for author Doctor Thierry Boucher (TB). He did not have any additional role in the study design, data collection and analysis, decision to publish or preparation manuscript. The specific roles of this author are articulated in the author contribution section.
References
- 1.Maslach C. What have we learned about burnout and health? Psychol Health. 2001;16: 607–611. doi: 10.1080/08870440108405530 [DOI] [PubMed] [Google Scholar]
- 2.Maslach C, Jackson SE, Leiter MP, others. Maslach burnout inventory Eval Stress Book Resour. 1997;3: 191–218. [Google Scholar]
- 3.Carod-Artal FJ, Vázquez-Cabrera C. Burnout syndrome in an international setting Burnout for experts. Springer; 2013. pp. 15–35. [Google Scholar]
- 4.Maslach C, Jackson SE. The measurement of experienced burnout. J Organ Behav. 1981;2: 99–113. [Google Scholar]
- 5.Kalimo R. Knowledge jobs–how to manage without burnout? Scand J Work Environ Health. 1999;25: 605–609. [DOI] [PubMed] [Google Scholar]
- 6.Mojsa-Kaja J, Golonka K, Marek T. Job burnout and engagement among teachers–worklife areas and personality traits as predictors of relationships with work. Int J Occup Med Environ Health. 2015;28: 102–119. doi: 10.13075/ijomeh.1896.00238 [DOI] [PubMed] [Google Scholar]
- 7.Finney C, Stergiopoulos E, Hensel J, Bonato S, Dewa CS. Organizational stressors associated with job stress and burnout in correctional officers: a systematic review. BMC Public Health. 2013;13: 82 doi: 10.1186/1471-2458-13-82 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kim HJ, Shin KH, Umbreit WT. Hotel job burnout: The role of personality characteristics. Int J Hosp Manag. 2007;26: 421–434. [Google Scholar]
- 9.de O Costa EF, Santos SA, de A Santos ATR, de Melo EV, de Andrade TM. Burnout Syndrome and associated factors among medical students: a cross-sectional study. Clinics. 2012;67: 573–580. doi: 10.6061/clinics/2012(06)05 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Karasek R, Theorell T. Healthy work: stress, productivity, and the reconstruction of working life. Basic books; 1992. [Google Scholar]
- 11.Nieuwenhuijsen K, Bruinvels D, Frings-Dresen M. Psychosocial work environment and stress-related disorders, a systematic review. Occup Med. 2010;60: 277–286. [DOI] [PubMed] [Google Scholar]
- 12.Ahola K, Salminen S, Toppinen-Tanner S, Koskinen A, Väänänen A. Occupational burnout and severe injuries: An eight-year prospective cohort study among Finnish forest industry workers. J Occup Health. 2013;55: 450–457. [DOI] [PubMed] [Google Scholar]
- 13.Nyberg ST, Fransson EI, Heikkilä K, Alfredsson L, Casini A, Clays E, et al. Job strain and cardiovascular disease risk factors: meta-analysis of individual-participant data from 47,000 men and women. PloS One. 2013;8: e67323 doi: 10.1371/journal.pone.0067323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huth C, Thorand B, Baumert J, Kruse J, Emeny RT, Schneider A, et al. Job strain as a risk factor for the onset of type 2 diabetes mellitus: findings from the MONICA/KORA Augsburg cohort study. Psychosom Med. 2014;76: 562–568. [DOI] [PubMed] [Google Scholar]
- 15.Theme Filha MM, de S Costa MA, Guilam MCR. Occupational stress and self-rated health among nurses. Rev Lat Am Enfermagem. 2013;21: 475–483. [DOI] [PubMed] [Google Scholar]
- 16.von Bonsdorff MB, Seitsamo J, von Bonsdorff ME, Ilmarinen J, Nyg\a ard C-H akan, Rantanen T. Job strain among blue-collar and white-collar employees as a determinant of total mortality: a 28-year population-based follow-up. BMJ Open. 2012;2: e000860 doi: 10.1136/bmjopen-2012-000860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Netterstrøm B, Kristensen TS, Damsgaard MT, Olsen O, Sjøl A. Job strain and cardiovascular risk factors: a cross sectional study of employed Danish men and women. Br J Ind Med. 1991;48: 684–689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kawakami N, Akachi K, Shimizu H, Haratani T, Kobayashi F, Ishizaki M, et al. Job strain, social support in the workplace, and haemoglobin A1c in Japanese men. Occup Environ Med. 2000;57: 805–809. doi: 10.1136/oem.57.12.805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li J, Jarczok MN, Loerbroks A, Schöllgen I, Siegrist J, Bosch JA, et al. Work stress is associated with diabetes and prediabetes: cross-sectional results from the MIPH Industrial Cohort Studies. Int J Behav Med. 2013;20: 495–503. doi: 10.1007/s12529-012-9255-0 [DOI] [PubMed] [Google Scholar]
- 20.Seidler A, Thinschmidt M, Deckert S, Then F, Hegewald J, Nieuwenhuijsen K, et al. The role of psychosocial working conditions on burnout and its core component emotional exhaustion–a systematic review. J Occup Med Toxicol. 2014;9: 10 doi: 10.1186/1745-6673-9-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bianchi R, Schonfeld IS, Laurent E. Is it time to consider the “burnout syndrome” a distinct illness? Front Public Health. 2015;3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schaufeli WB, Buunk BP. Burnout: An overview of 25 years of research and theorizing. Handb Work Health Psychol. 2003;2: 282–424. [Google Scholar]
- 23.Metlaine A, Leger D, Choudat D. Socioeconomic impact of insomnia in working populations. Ind Health. 2005;43: 11–19. [DOI] [PubMed] [Google Scholar]
- 24.Schonfeld IS, Bianchi R. Burnout and depression: two entities or one? J Clin Psychol. 2016;72: 22–37. doi: 10.1002/jclp.22229 [DOI] [PubMed] [Google Scholar]
- 25.Söderström M, Jeding K, Ekstedt M, Perski A, AAkerstedt T. Insufficient sleep predicts clinical burnout. J Occup Health Psychol. 2012;17: 175 doi: 10.1037/a0027518 [DOI] [PubMed] [Google Scholar]
- 26.Grossi G, Perski A, Eveng\a ard B, Blomkvist V, Orth-Gomér K. Physiological correlates of burnout among women. J Psychosom Res. 2003;55: 309–316. [DOI] [PubMed] [Google Scholar]
- 27.Melamed S, Shirom A, Toker S, Berliner S, Shapira I. Burnout and risk of cardiovascular disease: evidence, possible causal paths, and promising research directions. Psychol Bull. 2006;132: 327 doi: 10.1037/0033-2909.132.3.327 [DOI] [PubMed] [Google Scholar]
- 28.Danhof-Pont MB, van Veen T, Zitman FG. Biomarkers in burnout: a systematic review. J Psychosom Res. 2011;70: 505–524. [DOI] [PubMed] [Google Scholar]
- 29.Leger D, Guilleminault C, Dreyfus JP, Delahaye C, Paillard M. Prevalence of insomnia in a survey of 12 778 adults in France. J Sleep Res. 2000;9: 35–42. [DOI] [PubMed] [Google Scholar]
- 30.Johns MW. Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep. 1992;15: 376–381. [DOI] [PubMed] [Google Scholar]
- 31.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67: 361–370. [DOI] [PubMed] [Google Scholar]
- 32.Metlaine A, Sauvet F, Gomez-Merino D, Elbaz M, Delafosse JY, Leger D, et al. Association between insomnia symptoms, job strain and burnout syndrome: a cross-sectional survey of 1300 financial workers. BMJ Open. 2017;7: e012816 doi: 10.1136/bmjopen-2016-012816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nyberg ST, Fransson EI, Heikkilä K, Ahola K, Alfredsson L, Bjorner JB, et al. Job strain as a risk factor for type 2 diabetes: a pooled analysis of 124,808 men and women. Diabetes Care. 2014;37: 2268–2275. doi: 10.2337/dc13-2936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Steptoe A, Hackett RA, Lazzarino AI, Bostock S, La Marca R, Carvalho LA, et al. Disruption of multisystem responses to stress in type 2 diabetes: investigating the dynamics of allostatic load. Proc Natl Acad Sci. 2014;111: 15693–15698. doi: 10.1073/pnas.1410401111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Toker S, Shirom A, Melamed S, Armon G. Work characteristics as predictors of diabetes incidence among apparently healthy employees. J Occup Health Psychol. 2012;17: 259 doi: 10.1037/a0028401 [DOI] [PubMed] [Google Scholar]
- 36.Higgins T. HbA1c for screening and diagnosis of diabetes mellitus. Endocrine. 2013;43: 266–273. doi: 10.1007/s12020-012-9768-y [DOI] [PubMed] [Google Scholar]
- 37.Spiegel K, Knutson K, Leproult R, Tasali E, Van Cauter E. Sleep loss: a novel risk factor for insulin resistance and Type 2 diabetes. J Appl Physiol. 2005;99: 2008–2019. doi: 10.1152/japplphysiol.00660.2005 [DOI] [PubMed] [Google Scholar]
- 38.Li Y, Gao X, Winkelman JW, Cespedes EM, Jackson CL, Walters AS, et al. Association between sleeping difficulty and type 2 diabetes in women. Diabetologia. 2016;59: 719–727. doi: 10.1007/s00125-015-3860-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kachi Y, Nakao M, Takeuchi T, Yano E. Association between Insomnia Symptoms and Hemoglobin A 1c Level in Japanese Men. PloS One. 2011;6: e21420 doi: 10.1371/journal.pone.0021420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hirotsu C, Tufik S, Andersen ML. Interactions between sleep, stress, and metabolism: from physiological to pathological conditions. Sleep Sci. 2015;8: 143–152. doi: 10.1016/j.slsci.2015.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Asensio C, Muzzin P, Rohner-Jeanrenaud F. Role of glucocorticoids in the physiopathology of excessive fat deposition and insulin resistance. Int J Obes. 2004;28: S45–S52. [DOI] [PubMed] [Google Scholar]
- 42.Licht CM, Vreeburg SA, van Reedt Dortland AK, Giltay EJ, Hoogendijk WJ, DeRijk RH, et al. Increased sympathetic and decreased parasympathetic activity rather than changes in hypothalamic-pituitary-adrenal axis activity is associated with metabolic abnormalities. J Clin Endocrinol Metab. 2010;95: 2458–2466. doi: 10.1210/jc.2009-2801 [DOI] [PubMed] [Google Scholar]
- 43.Massa J, Stone KL, Wei EK, Harrison SL, Barrett-Connor E, Lane NE, et al. Vitamin D and actigraphic sleep outcomes in older community-dwelling men: the MrOS sleep study. Sleep. 2015;38: 251–257. doi: 10.5665/sleep.4408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Beydoun MA, Gamaldo AA, Canas JA, Beydoun HA, Shah MT, McNeely JM, et al. Serum nutritional biomarkers and their associations with sleep among US adults in recent national surveys. PloS One. 2014;9: e103490 doi: 10.1371/journal.pone.0103490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gominak S, Stumpf W. The world epidemic of sleep disorders is linked to vitamin D deficiency. Med Hypotheses. 2012;79: 132–135. doi: 10.1016/j.mehy.2012.03.031 [DOI] [PubMed] [Google Scholar]
- 46.Papandreou D, Hamid Z-T-N. The role of vitamin D in diabetes and cardiovascular disease: an updated review of the literature. Dis Markers. 2015;2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.McEwen BS. Protective and damaging effects of stress mediators. N Engl J Med. 1998;338: 171–179. doi: 10.1056/NEJM199801153380307 [DOI] [PubMed] [Google Scholar]
- 48.McEwen BS. Protection and damage from acute and chronic stress: allostasis and allostatic overload and relevance to the pathophysiology of psychiatric disorders. Ann N Y Acad Sci. 2004;1032: 1–7. doi: 10.1196/annals.1314.001 [DOI] [PubMed] [Google Scholar]
- 49.Arnal PJ, Sauvet F, Leger D, Van Beers P, Bayon V, Bougard C, et al. Benefits of Sleep Extension on Sustained Attention and Sleep Pressure Before and During Total Sleep Deprivation and Recovery. Sleep. 2014;38: 1935–1943. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The study data set cannot be made publicly available due to ethical restrictions imposed by the Occupational Safety and Health Ethics Committee (CHSCT). However, a de-identified dataset can be made available on request from the corresponding author at arnaud.metlaine@aphp.fr. Additionally, data requests may also be sent to the following institutional address: Centre du Sommeil et de la vigilance de l’Hôtel-Dieu. APHP. 1 place de Parvis de Notre Dame-75181 Paris cedex 4 (France).