Abstract
Objective
In MS, Socio-Economic status (SES) may influence healthcare and access to disease-modifying treatments (DMTs). Optimising delays to switch patients to a second-line DMT may hamper disease progression most effectively and achieve long term disease control. The objective of this study is to identify the influence of SES on the delay between first and second line DMT in RRMS patients, in Western-Normandy, France.
Methods
The association between SES and the delay to access a second-line DMT were studied using data from the MS registry of Western-Normandy including 733 patients with a diagnosis of RRMS during the period in question [1982–2011]. We used the European Deprivation Index (EDI), a score with a rank level inversely related to SES. We performed multivariate adjusted Cox models for studying EDI effect on the delay between first and second line DMT.
Results
No significant influence of SES was observed on delay to access a second-line DMT if first-line DMT exposure time was less than 5 years. After 5 years from initiation of first-line treatment the risk of accessing a second-line DMT is 3 times higher for patients with lower deprivation indices (1st quintile of EDI) ([HR] 3.14 95%CI [1.72–5.72], p-value<0.001) compared to patients with higher values (EDI quintiles 2 to 5).
Interpretation
In RRMS, a high SES may facilitate access to a second-line DMT a few years after first-line DMT exposure. Greater consideration should also be given to the SES of MS patients as a risk factor in therapeutic healthcare issues throughout medical follow-up.
Introduction
Socio-economic factors have been shown to influence different health indicators (risk of occurrence, survival, clinical course and access to health care system) of many chronic diseases including cancer, cardiovascular diseases, HIV, diabetes and neurological diseases [1–9]. Regarding Multiple Sclerosis (MS), socioeconomic status (SES) tends to be associated with the risk of onset of MS although findings remain controversial and inconsistent [10].
The effect of SES can be investigated not only in terms of MS risk but also in terms of MS care system. Indeed, patients followed by a neurologist and/or a MS multidisciplinary care network may benefit from better global care. They are more likely to undergo diagnosis procedures and to receive disease-modifying treatments (DMTs) [11–13]. However, social inequalities may represent a barrier when it comes to accessing different medical specialists for MS care and can impact upon the quality of global MS care and access to DMTs.
In various chronic and systemic diseases, diagnosis and therapeutic delays are relevant health indicators at different stages in the medical process: from initial symptom presentation through treatments including time from first symptoms to diagnosis and time from diagnosis to treatment initiation. These delays may be influenced by many variables depending on the patient (behaviour when faced with a health problem, treatment acceptance), physicians (lengthy wait times, specialist referral times) and/or the medical process (availability of medical investigations and treatment). Many variables such as marital status, household income, level of education, living in rural or urban areas, presence of comorbidities and medical insurance cover may explain delayed diagnosis and treatment as demonstrated in cancers [14,15]. In MS, optimising treatment delays, including intervals to switch patients to second-line DMT may, if indicated, delay disease progression most effectively and achieve long-term disease control [16,17].
The aim of this study is to identify the influence of SES on the delay between first and second-line DMT in RRMS patients, in Western-Normandy, France.
Methods
Study population
Study data were extracted from the database of the Western-Normandy MS network, which is part of the French MS Observatory (OFSEP) [18]. All patients included in this MS database were diagnosed according to Poser and McDonald 2001 then 2005 criteria, between 1982 and 2011 in three French counties in Western-Normandy (Calvados, Manche, and Orne). The date of the first DMT treatment was known for these patients (n = 933) (Fig 1).
Fig 1. Flow chart of population of interest extracted from the Western-Normandy MS network database (n = 733).
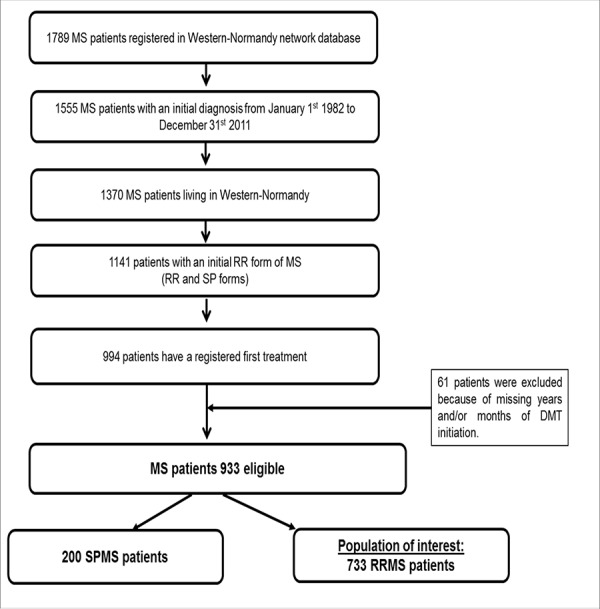
DMT: Disease Modifying Treatment. SPMS: Secondary Progressive Multiple Sclerosis. RRMS: Relapsing Remitting Multiple Sclerosis.
The population of interest was limited to patients with an initial RR form of MS who did not convert to a secondary progressive form at the cut-off date (n = 733).
The end of inclusion date was established to ensure the completeness of the data. The cut-off date was set as the end of February 2015.
The MS registry and use of its data for clinical and epidemiological research were approved by the French National Ethics Committee Commission Nationale de l'Informatique et des Libertés (CNIL number 1011405/1) and written informed consent was obtained from all patients for collecting and analysing their data in medical research.
Variables
The main variable was defined as the delay (in years) between the introduction of first-line and second-line DMTs. The second-line DMTs selected for the analysis were cyclophosphamide, mitoxantrone, natalizumab and fingolimod. If the first treatment was a second-line DMT, delay was considered equal to half a day (n = 48; 5.1%). The event of interest for the entire survival analysis was the introduction of a second-line DMT. For patients who never received second-line DMT, delay was censored on the date of the last neurological consultation.
MS patient characteristics were as follows: date of birth, gender, years of disease onset, diagnosis years according to Poser and McDonald criteria and county of residence at diagnosis.
Social variables were based on each patient’s place of residence, geolocalised according to a Geographic Information System (ARCGIS 10.2) and assigned to an IRIS (Ilots Regroupés pour l’Information Statistique). IRIS is the smallest French geographic unit for which census data are available and an EDI score was calculated for each IRIS. In order to attribute a social deprivation score to the IRIS, we used the French EDI (European Deprivation Index) [19]. In our study we used EDI as a categorical variable in order to increase comparability with others studies. EDIs were therefore distributed into quintiles calculated according to the national distribution rate.
Statistical analysis
The Kaplan-Meier (KM) estimator was used in order to obtain graphical representation of the different categorical variables (gender, MS form, county of residence at diagnosis and period of diagnosis, age at disease onset, and EDI quintiles). For each stratified univariate, an analysis log-rank test was performed to detect any difference between the population groups in the probability of accessing a second-line DMT in the KM analysis. The KM univariate analysis comparing delay to access second-line DMT according to EDI quintiles wasn’t statistically significant. Based on an observation of the five curves of KM, the curve corresponding to EDI quintile 1 tends to be separate from the others four. Consequently, the analysis was carried out comparing the population of quintile1 to populations of quintiles 2 to 5 combined.
A series of adjusted multivariate cox proportional hazard models was performed with the delay of second-line DMT initiation as a dependent variable and EDI quintiles as independent variables. The following covariates gender, age at disease onset, county of residence at diagnosis, period of diagnosis and year of disease onset were used as other independent adjustment variables. The year of disease onset was taken as a continuous adjustment variable for all multivariate Cox regression models. All statistical analyses of this study were performed using SAS version 9.3 and STATA IC/SE 14.
Results
In the initial overall cohort, the majority of patients were females (74.4%; n = 695). The mean age at disease onset was 32.46 ±9.75 years, with age at disease onset ranging from 6 to 65 years. No patient received second-line DMT before the age of 16 or after the age of 64, with the mean age at onset of second-line DMT being 39.49 ±10.08 years. Most patients (62.3%; n = 581) were diagnosed between 2001 and 2011 in the county of Calvados (49.8%; n = 465).
The characteristics of the population of interest (RRMS patients; n = 733), are summarised in Table 1.
Table 1. Characteristics of patients diagnosed with RRMS during the period [1982–2011] in Western-Normandy.
| Factors | Number of RRMS patients (%) | Number of second-line DMT initiation (n) | Median delay(a) [CI 95%] | p-value(b) |
|---|---|---|---|---|
| Gender | 733 | 199 | 0.069 | |
| Males | 186 (25.4%) | 60 | 5.34 [3.91–8.08] | |
| Females | 547 (74.6%) | 139 | 8.83 [6.58–9.58] | |
| Age at disease onset | 733 | 199 | 0.1881 | |
| [6–25 years] | 205 (28%) | 73 | 6.41 [4.5–9.58] | |
| [26–35 years] | 280 (38.2%) | 75 | 9.08 [6.83–12.75] | |
| [36–45 years] | 177 (24.1%) | 39 | 5.75 [4.59–9.58] | |
| >45 years | 71 (9.7%) | 12 | 5.33 [3.91-.] | |
| Periods of diagnosis | 733 | 199 | <0.001 | |
| [1982–2000] | 204 (27.8%) | 55 | 13.75 [10.33–14.25] | |
| [2001–2011] | 529 (72.2%) | 144 | 4.83 [3.83–5.66] | |
| County of residence at diagnosis | 733 | 199 | 0.0015 | |
| Manche | 247 (33.7%) | 76 | 5.58 [3.92–8] | |
| Calvados | 378 (51.6%) | 97 | 9.08 [6.83–10.33] | |
| Orne | 108 (14.7%) | 26 | 9.5 [5.33–15.83] | |
| EDI (quintiles) | 725 | 198 | 0.1923 | |
| Q1 | 108 (14.7%) | 35 | 5.83 [4.83–7.75] | |
| Q2 | 115 (15.7%) | 28 | 9.08 [8.34–13] | |
| Q3 | 155 (21.1%) | 41 | 9 [5.33–14.25] | |
| Q4 | 183 (25%) | 55 | 6.58 [5–12.25] | |
| Q5 | 164 (22.4%) | 39 | 9.5 [6.58–15.83] |
(a) Kaplan Meier survival analysis estimates median delay to access a second-line DMT.
(b) Log rank test used for calculation of significance.
DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. CI: Confidence Interval. EDI: European Deprivation Index.
As expected, the median delays to access a second-line DMT was significantly reduced by 9 years in patients diagnosed between 2001 and 2011 compared to patients diagnosed between 1982 and 2000 (p<0.001). A shorter median delay to access a second-line DMT was recorded for patients living in Manche at the time of diagnosis compared to their counterparts in Calvados and Orne (-3.5 years and -4 years, respectively) (p<0.001).
Median delays to access a second-line DMT did not differ statistically between the five EDI quintiles.
A Cox model performed with EDI separated into 2 groups, namely quintile 1 and quintiles 2 to 5 combined, and using this as a time-varying covariate shows a significant difference in favour of quintile 1 ([HR] 1.19; 95% CI [1.07–1.33], p-value<0.001).
After adjustment based on other independent variables (gender, age at disease onset, county of residence, period of diagnosis and year of disease onset) and taking into account the modifying effect of EDI quintiles over time, the risk to access a second-line DMT is significantly higher for patients with the lowest deprivation indices (the 1st quintile of EDI) ([HR]1.14 95% CI [1.06–1.22], p-value<0.001) compared to patients with a higher EDI. This effect is only relevant over a certain time period and does not apply to the overall period of analysis (Table 2).
Table 2. Association between SES and access to a second-line DMT for RRMS patients.
| Factors | HR [CI95%] | p-value |
|---|---|---|
| Gender | 0.4192 | |
| Males | 1.00 (Reference) | |
| Females | 0.88 [0.64–1.2] | |
| Age at disease onset | 0.1139 | |
| [6–25 years] | 1.00 (Reference) | |
| [26–35 years] | 0.7 [0.5–0.97] | |
| [36–45 years] | 0.71 [0.47–1.05] | |
| >45 years | 0.61 [0.32–1.14] | |
|
County of residence at diagnosis |
0.0005 | |
| Manche | 1.00 (Reference) | |
| Calvados | 0.56 [0.41–0.77] | |
| Orne | 0.52 [0.33–0.82] | |
| Period of diagnosis | 0.0076 | |
| 1982–2000 | 1.00 (Reference) | |
| 2001–2011 | 1.89 [1.18–3.02] | |
| Year of disease's onset | 1.1 [1.06–1.15] | < 0.0001 |
| EDI (a) (quintiles combined) | 0.0011 | |
| Q2-Q5 | 1.00 (Reference) | |
| Q1 | 1.14 [1.06–1.22] |
(a) Multivariate Cox model for studying EDI effect was performed using EDI as time varying covariate that allows taking into consideration the interaction between EDI and time. (tvc function of STATA ® software).
DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. HR: Hazard Ratio. CI: Confidence Interval. EDI: European Deprivation Index.
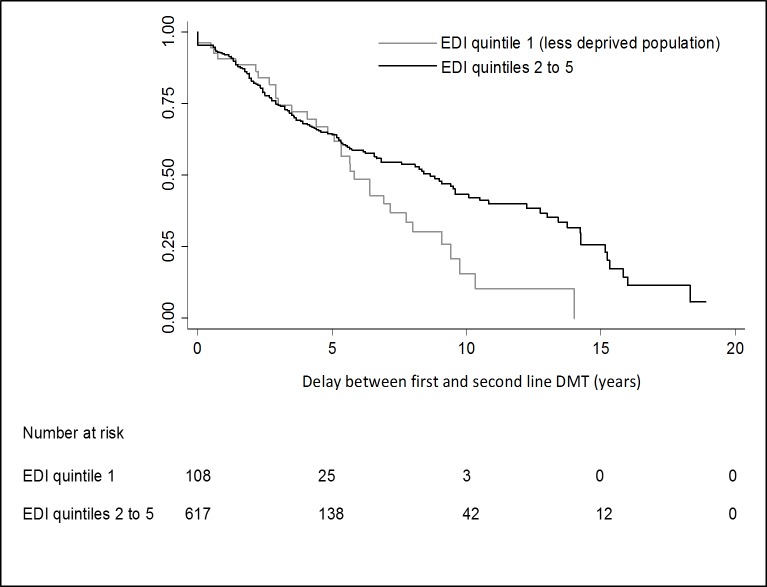
During the first 5 years of first-line treatment, SES is not significantly associated with the delay of accessing second-line DMT but was significantly associated after (Fig 2).
Fig 2. Influence of SES on delay between first and second line DMT in RRMS patients.
Kaplan-Meier estimates for delay to access a second-line DMT according to EDI quintiles used as SES indicator. A comparison was made between EDI quintile 1 and EDI quintiles 2 to 5 combined. SES: Socio-Economic Status. DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. EDI: European Deprivation Index.
Indeed, after 5 years delay from the initiation of first-line treatment initiation, risk to access a second-line DMT was 3 times higher for patients with lower deprivation indices (1st quintile of EDI) ([HR] 3.14 95% CI [1.72–5.72], p-value<0.001) compared to patients with higher values (EDI quintiles 2 to 5) (Table 3).
Table 3. Association between SES and access to a second-line DMT for RRMS patients after a 5-years delay between first and second line DMT.
| Factors | HR [CI 95%] | p-value |
|---|---|---|
| Gender | 0.4487 | |
| Males | 1.00 (Reference) | |
| Females | 0.89 [0.65–1.21] | |
| Age at disease onset | 0.1199 | |
| [15–25 years] | 1.00 (Reference) | |
| [26–35 years] | 0.7 [0.5–0.97] | |
| [36–45 years] | 0.71 [0.47–1.05] | |
| >45 years | 0.62 [0.33–1.16] | |
|
County of residence at diagnosis |
0.0006 | |
| Manche | 1.00 (Reference) | |
| Calvados | 0.57 [0.42–0.77] | |
| Orne | 0.51 [0.32–0.82] | |
| Period of diagnosis | 0.0075 | |
| 1982–2000 | 1.00 (Reference) | |
| 2001–2011 | 1.89 [1.18–3.02] | |
| Year of disease onset | 1.1 [1.06–1.15] | < 0.0001 |
| EDI (a) (quintiles combined) | 0.0005 | |
| Q2-Q5 | 1.00 (Reference) | |
| Q1 | 3.14 [1.72–5.72] |
(a) Multivariate Cox model for studying EDI effect was performed using EDI as time varying covariate that allows taking into consideration the interaction between EDI and time. (tvc function of STATA ® software).
DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. HR: Hazard Ratio. CI: Confidence Interval. EDI: European Deprivation Index.
Discussion
Different studies have already shown a significant association between high SES in childhood and adulthood, and a lower risk of MS [20–22]. Apart from influencing the MS risk, social inequalities also seem to impact upon access to treatments.
Our findings which confirmed that SES affects access to treatment is consistent with the study conducted by Owens and al. [23] in two different areas of the UK (Nottingham and Glasgow). They used the Index of Multiple Deprivation (IMD) as the social deprivation variable and found that being from a deprived area significantly reduced the probability of receiving a DMT prescription. The results were almost identical using another validated deprivation measure, namely the Carstairs’ score, and then ensuring the consistency of the link.
We went further, and we have shown that SES influences the initiation of second-line DMT in RRMS patients with a unique relevant difference between patients with a higher SES (first quintile of EDI) and the others. Indeed, patients with higher SES have faster access to second-line DMTs than patients with lower SES only 5 years after first-line DMT exposure.
This time-related result may initially be explained by the financial difficulties encountered by lower SES patients who may be discouraged from receiving appropriate medical follow-up for MS. Previous studies conducted in the USA have highlighted differences in accessing medical specialists for MS care based on insurance status and SES (education, incomes and employment status) [24]. Patients with no health insurance and a lower household income are less likely to consult a neurologist [25]. In France, some neurologists may charge additional fees that may be refunded either in full or in part by complementary health insurance schemes. Some patients may be out-of-pocket in terms of medical expenditure after each neurologist consultation. Approximately 5% of the French population is estimated to be without complementary health insurance [26]. Patients with lower SES are more likely to fall into this category. According to an INSEE survey, 21% of patients belonging to the lower 20% SES category are reported not to have supplementary health cover compared to 4% of the higher quintile population [27]. This survey assumes that 17% of patients with overall health insurance cover for chronic disease do not have supplementary health cover. Out-of-pocket medical expenditure can be significant in MS patients with a low SES and can discourage them from having regular follow-up and consultations. However, in France, second-line DMT initiation is strictly reserved to public or private hospital neurological care. For these reasons, patients receive second-line DMT only in hospital, at least regarding initiation, and are mainly followed up in MS centres where health costs directly or indirectly related to MS are fully covered by public health insurance. Thus, if financial criteria were to impact upon neurological follow-up, the effect on second-line treatment initiation would be fairly weak.
A clearer explanation is that patients with a high SES pay closer attention to and are more involved throughout their medical follow-up compared to patients with a lower SES. The former is more likely to have better attendance at medical appointments and constructive dialogue with physicians. As second-line DMTs have greater efficacy on MS disease but present a significant risk of serious side effects, patients with a high SES may have a better understanding of the benefit-risk ratio of second-line DMT and may accept treatment change more readily.
Indeed, neurologists may be more reluctant to propose second-line DMT for patients with a lower SES, as these ones have generally a low level of education that makes it difficult to understand health-related information and limits their participation in treatment decisions [28]. Unfortunately, we were not able to assess this criterion because of the excessive amount of missing data for our population.
In addition, neurologists may find it difficult to identify breakthrough disease in these patients and/or to decide to prescribe second-line DMT. In a complementary study, we performed similar survival analyses and adjusted multivariate Cox analyses selecting only RRMS patients who had at least one relapse under first-line DMT (i.e. as a proxy of a potential indication of second-line DMT switch) (n = 382).
The risk of accessing second-line DMT remains significant after 5 years of first-line treatment exposure in favour of socially less deprived patients ([HR] 2.13 95%CI [1.07–4.22], p-value = 0.039) (Supporting information S1 Table), supporting our main result. This complementary result refutes hypothesis of disease stability as a possible explanation for MS patients not accessing a second-line treatment.
Apart from social inequalities, geographical disparities can pose another barrier to accessing medical MS care. In a survey study conducted in 50 US states, significant differences were apparent between urban/rural places of residence in terms of accessing specialist MS care providers. For instance, most of the geographically isolated patients chose not to use MS-related care because of excessively long travel times [29,30].
In our study we showed that patients living in Manche had earlier access to second-line DMT that those living in Orne and Calvados. We ensured that this result was not biased by the effect of the diagnosis period. Moreover, this difference is not consistent with all the means implemented to avoid MS care disparities between counties. In these three counties, a common MS health network and regular regional medical team meetings were set up to help neurologists with therapeutic decisions and to harmonise the healthcare system. One possible explanation is that, in the Manche county, there were no private neurologists particularly involved in MS during a large part of the study period. MS patients had their medical follow-up at hospital and second-line DMT decisions were not influenced by the discussion time between private and hospital neurologists.
The period of diagnosis and years to disease onset, used as adjustment covariates in the multivariate analysis, have a strong influence on access to second-line DMTs. These results were expected and can be explained by the gradual availability of second-line DMT between 2000 and 2011. In addition, the effect of disease onset is obviously explained by improved diagnostic investigations.
Our study has some limitations. Data are collected from a population-based observatory and not from a registry. In the Western-Normandy regional observatory patient inclusion is completely dependent on the reporting neurologist. Most patients suffering from MS in Western-Normandy can be expected to be registered in the observatory. This represents a statistical recruitment bias. However, MS-diagnosed patients had necessarily consulted a neurologist and neurologists are advised to record each diagnosed patient in the observatory. However, this study is based on high-resolution data, because a great deal of information was collected and recorded as exhaustively as possible for each patient thanks to subsequent neurological consultations or via medical records. Data are steadily updated and validated by a clinical research assistant. The Western-Normandy registry forms part of the National Multiple Sclerosis Observatory (OFSEP), which provides detailed guidelines for collecting data and assessing the quality of these data.
Beside the new relapse under first-line DMT, which is the main predictive variable for the switch to second-line DMT, some other clinical variables could influence the switch time to a second-line DMT that are not considered because of missing or unreliable data. One of them, radiological disease activity which is obviously an important criterion for switching to a second-line DMT, is not, nowadays in France, done and/or collected in a standardized way in real life setting (various MRI scanning protocols depending on the different MRI machines) and, thus, could not be included in a population study. Moreover, contrary to clinical trials, even high-resolution population-based study do no afford the possibility to collect other clinical data such as comorbidities or patient’s adherence, and the major strength of population based studies is to analyse data collected on a long term period and to include patients which receive sub-optimal treatments.
The use of EDI, aggregated data at a small area population level, as an indicator of SES, can infer an ecological bias [31]. Taking into account the proximal social environment of a place of residence is a pragmatic solution used in numerous countries. Since the 1990s, many deprivation indices (e.g. Townsend, Carstairs, IMD, Quebec index of material and social deprivation and EDI) linked to small areas and based on aggregated data respond favourably to various requirements for validity, reliability, responsiveness and use in public health [32–34].
Finally, as patients with a secondary progressive MS were withdrawn from the analysis, the time-dependent influence of SES may not be biased by the clinical course of the disease towards a progressive form. There is an independent effect on the time dependant association between high SES and an opportunity to access second-line DMT, even after accounting for gender, age and year of disease onset and period of diagnosis.
Conclusion
We have shown that, in RR-MS patients, SES may influence the access to second-line DMT after a few years of exposure to first-line DMT. This result does not appear to be explained by the MS care system in Western-Normandy where the effective involvement of the regional MS network and regular regional medical team meetings for treatment care may not be sufficient to prevent social inequalities in terms of access to treatment.
On account of different geographical contexts and healthcare systems these results cannot be generalized to other parts of Europe, and of the world. Our results should also be confirmed with a more extended cohort of patients in France. We plan to extend this work to other French regional MS databases for this purpose and to investigate whether social inequalities play a role in other healthcare delays.
Furthermore, the collection of new data, for instance level of education and associated comorbidities, will be necessary to ensure a better understanding of the mechanisms that govern social inequalities in terms of access to DMTs.
Greater consideration should also be given to the SES of MS patients as a risk factor in therapeutic healthcare issues throughout medical follow-up. National health authorities should make every effort to reduce health-related inequalities regarding treatment access and to ensure more effective long-term medical follow-up for patients with a low SES.
Supporting information
Multivariate Cox model for studying EDI effect was performed using EDI as time varying covariate that allows taking into consideration the interaction between EDI and time. (tvc function of STATA ® software). DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. HR: Hazard Ratio. CI: Confidence Interval. EDI: European Deprivation Index.
(PDF)
Acknowledgments
We would like to thank the French Observatory of Multiple Sclerosis (OFSEP), which is supported by a grant provided by the French State and handled by the “Agence Nationale de la Recherche” within the framework of the “Investments for the Future” program, under the reference ANR-10-COHO-002. We also thank the geographical plateform MAPinMED and “Cancers and Preventions” group for performing geolocalization of our study population of MS patients, and especially to Ludivine Launay for her large contribution in this task.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
Floriane Calocer received a fellowship from the "Réseau Bas-Normand pour la SEP" and from the Regional Council of Normandy for carrying out this study. The Regional Council of Normandy had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. The "Réseau Bas Normand pour la SEP" had only a role in data collection.
References
- 1.Ben-Shlomo Y, Kuh D. A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol. 2002;31: 285–293. [PubMed] [Google Scholar]
- 2.Vohra J, Marmot MG, Bauld L, Hiatt RA. Socioeconomic position in childhood and cancer in adulthood: a rapid-review. J Epidemiol Community Health. 2016;70: 629–634. doi: 10.1136/jech-2015-206274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Agardh E, Ahlbom A, Andersson T, Efendic S, Grill V, Hallqvist J, et al. Socio-economic position at three points in life in association with type 2 diabetes and impaired glucose tolerance in middle-aged Swedish men and women. Int J Epidemiol. 2007;36: 84–92. doi: 10.1093/ije/dyl269 [DOI] [PubMed] [Google Scholar]
- 4.Kamphuis CB, Turrell G, Giskes K, Mackenbach JP, van Lenthe FJ. Socioeconomic inequalities in cardiovascular mortality and the role of childhood socioeconomic conditions and adulthood risk factors: a prospective cohort study with 17-years of follow up. BMC Public Health. 2012;12: 1 doi: 10.1186/1471-2458-12-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beltran VM, Harrison KM, Hall HI, Dean HD. Collection of social determinant of health measures in U.S. national surveillance systems for HIV, viral hepatitis, STDs, and TB. Public Health Rep. 2011;126 Suppl 3: 41–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Marshall IJ, Wang Y, Crichton S, McKevitt C, Rudd AG, Wolfe CDA. The effects of socioeconomic status on stroke risk and outcomes. Lancet Neurol. 2015;14: 1206–1218. doi: 10.1016/S1474-4422(15)00200-8 [DOI] [PubMed] [Google Scholar]
- 7.Szaflarski M. Social determinants of health in epilepsy. Epilepsy Behav. 2014;41: 283–289. doi: 10.1016/j.yebeh.2014.06.013 [DOI] [PubMed] [Google Scholar]
- 8.Qian W, Schweizer TA, Fischer CE. Impact of socioeconomic status on initial clinical presentation to a memory disorders clinic. Int Psychogeriatr. 2014;26: 597–603. doi: 10.1017/S1041610213002299 [DOI] [PubMed] [Google Scholar]
- 9.Frier A, Barnett F, Devine S. The relationship between social determinants of health, and rehabilitation of neurological conditions: a systematic literature review. Disabil Rehabil. 2016;0: 1–8. doi: 10.3109/09638288.2016.1172672 [DOI] [PubMed] [Google Scholar]
- 10.Goulden R, Ibrahim T, Wolfson C. Is high socioeconomic status a risk factor for multiple sclerosis? A systematic review. Eur J Neurol. 2015;22: 899–911. doi: 10.1111/ene.12586 [DOI] [PubMed] [Google Scholar]
- 11.Halper J, Burks JS. Care patterns in multiple sclerosis: principal care, comprehensive team care, consortium care. NeuroRehabilitation. 1994;4: 67–75. doi: 10.3233/NRE-1994-4203 [DOI] [PubMed] [Google Scholar]
- 12.Burks J. Multiple sclerosis care: an integrated disease-management model. J Spinal Cord Med. 1998;21: 113–116. [DOI] [PubMed] [Google Scholar]
- 13.Creange A, Debouverie M, Jaillon-Riviere V, Taithe F, Liban D, Moutereau A, et al. Home administration of intravenous methylprednisolone for multiple sclerosis relapses: the experience of French multiple sclerosis networks. Mult Scler. 2009;15: 1085–1091. doi: 10.1177/1352458509106710 [DOI] [PubMed] [Google Scholar]
- 14.Neal RD, Tharmanathan P, France B, Din NU, Cotton S, Fallon-Ferguson J, et al. Is increased time to diagnosis and treatment in symptomatic cancer associated with poorer outcomes? Systematic review. Br J Cancer. 2015;112: S92–S107. doi: 10.1038/bjc.2015.48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Allgar VL, Neal RD. Delays in the diagnosis of six cancers: analysis of data from the National Survey of NHS Patients: Cancer. Br J Cancer. 2005;92: 1959–70. doi: 10.1038/sj.bjc.6602587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ziemssen T, De Stefano N, Pia Sormani M, Van Wijmeersch B, Wiendl H, Kieseier BC. Optimizing therapy early in multiple sclerosis: An evidence-based view. Mult Scler Relat Disord. 2015;4: 460–469. doi: 10.1016/j.msard.2015.07.007 [DOI] [PubMed] [Google Scholar]
- 17.Noyes K, Weinstock-Guttman B. Impact of diagnosis and early treatment on the course of multiple sclerosis. Am J Manag Care. 2013;19: s321–331. [PubMed] [Google Scholar]
- 18.Cotton F, Kremer S, Hannoun S, Vukusic S, Dousset V, Imaging Working Group of the Observatoire Français de la Sclérose en Plaques. OFSEP, a nationwide cohort of people with multiple sclerosis: Consensus minimal MRI protocol. J Neuroradiol J Neuroradiol. 2015;42: 133–140. doi: 10.1016/j.neurad.2014.12.001 [DOI] [PubMed] [Google Scholar]
- 19.Pornet C, Delpierre C, Dejardin O, Grosclaude P, Launay L, Guittet L, et al. Construction of an adaptable European transnational ecological deprivation index: the French version. J Epidemiol Community Health. 2012;66: 982–9. doi: 10.1136/jech-2011-200311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bjørnevik K, Riise T, Cortese M, Holmøy T, Kampman MT, Magalhaes S, et al. Level of education and multiple sclerosis risk after adjustment for known risk factors: The EnvIMS study. Mult Scler J. 2016;22: 104–111. doi: 10.1177/1352458515579444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nielsen NM, Jørgensen KT, Bager P, Stenager E, Pedersen BV, Hjalgrim H, et al. Socioeconomic Factors in Childhood and the Risk of Multiple Sclerosis. Am J Epidemiol. 2013;177: 1289–1295. doi: 10.1093/aje/kws350 [DOI] [PubMed] [Google Scholar]
- 22.Riise T, Kirkeleit J, Aarseth JH, Farbu E, Midgard R, Mygland Å, et al. Risk of MS is not associated with exposure to crude oil, but increases with low level of education. Mult Scler J. 2011;17: 780–787. doi: 10.1177/1352458510397686 [DOI] [PubMed] [Google Scholar]
- 23.Owens T, Evangelou N, Whynes DK. Rationing and deprivation: disease-modifying therapies for multiple sclerosis in the United Kingdom. Eur J Health Econ. 2013;14: 315–21. doi: 10.1007/s10198-012-0378-7 [DOI] [PubMed] [Google Scholar]
- 24.Minden SL, Frankel D, Hadden L, Hoaglin DC. Access to health care for people with multiple sclerosis. Mult Scler. 2007;13: 547–558. doi: 10.1177/1352458506071306 [DOI] [PubMed] [Google Scholar]
- 25.Minden SL, Hoaglin DC, Hadden L, Frankel D, Robbins T, Perloff J. Access to and utilization of neurologists by people with multiple sclerosis. Neurology. 2008;70: 1141–1149. doi: 10.1212/01.wnl.0000306411.46934.ef [DOI] [PubMed] [Google Scholar]
- 26.IRDES. La couverture complémentaire santé en France dans ESPS: Données générales. IRDES. Institut de Recherche et Documentation en Economie de la Santé [Internet]. Sep 2013. Available: http://www.irdes.fr/EspaceEnseignement/ChiffresGraphiques/CouvertureComplementaire/DonneesGnles.html
- 27.De Saint Pol T, Marical F. La complémentaire santé: une généralisation qui n’efface pas les inégalités. Insee Prem. 2007; 4.
- 28.Willems S, De Maesschalck S, Deveugele M, Derese A, De Maeseneer J. Socio-economic status of the patient and doctor–patient communication: does it make a difference? Patient Educ Couns. 2005;56: 139–146. doi: 10.1016/j.pec.2004.02.011 [DOI] [PubMed] [Google Scholar]
- 29.Buchanan RJ, Wang S, Stuifbergen A, Chakravorty BJ, Zhu L, Kim M. Urban/rural differences in the use of physician services by people with multiple sclerosis. NeuroRehabilitation. 2006;21: 177–187. [PubMed] [Google Scholar]
- 30.Buchanan RJ, Stuifbergen A, Chakravorty BJ, Wang S, Zhu L, Kim M. Urban/rural differences in access and barriers to health care for people with multiple sclerosis. J Health Hum Serv Adm. 2006;29: 360–375. [PubMed] [Google Scholar]
- 31.Greenland S, Morgenstern H. Ecological Bias, Confounding, and Effect Modification. Int J Epidemiol. 1989;18: 269–274. doi: 10.1093/ije/18.1.269 [DOI] [PubMed] [Google Scholar]
- 32.Krieger N. Overcoming the absence of socioeconomic data in medical records: validation and application of a census-based methodology. Am J Public Health. 1992;82: 703–710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mustard CA, Derksen S, Berthelot J-M, Wolfson M. Assessing ecologic proxies for household income: a comparison of household and neighbourhood level income measures in the study of population health status. Health Place. 1999;5: 157–171. doi: 10.1016/S1353-8292(99)00008-8 [DOI] [PubMed] [Google Scholar]
- 34.Pampalon R, Hamel D, Gamache P, Simpson A, Philibert MD. Validation of a deprivation index for public health: a complex exercise illustrated by the Quebec index. Chronic Dis Inj Can. 2014;34: 12–22. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Multivariate Cox model for studying EDI effect was performed using EDI as time varying covariate that allows taking into consideration the interaction between EDI and time. (tvc function of STATA ® software). DMT: Disease Modifying Treatment. RRMS: Relapsing Remitting Multiple Sclerosis. HR: Hazard Ratio. CI: Confidence Interval. EDI: European Deprivation Index.
(PDF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.