Abstract
Polyphenols including catechins from green tea (Camellia sinensis) have been reported to have anti-infective activities against a broad spectrum of viruses and other pathogens. During the last two decades, antiviral activities of catechins with different modes of action have been demonstrated on diverse families of viruses, such as human immunodeficiency virus, Herpes simplex virus, influenza virus, hepatitis B and C virus. In this study, we focused on the antiviral properties of catechins and their derivatives against viral hepatitis which have become a key public health issue due to their serious impact on human health with liver diseases.
Keywords: Catechin, Hepatitis, Antiviral agents
Introduction
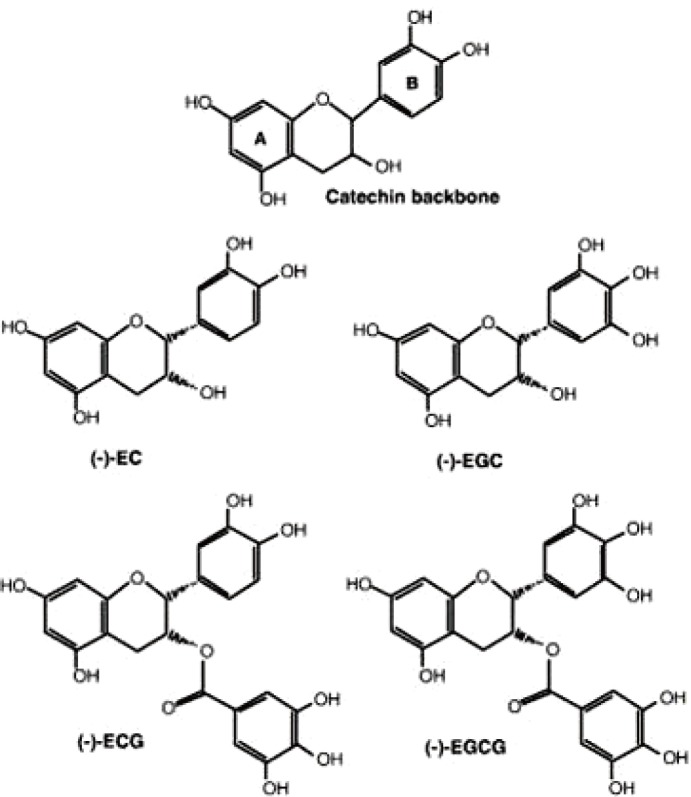
Green tea catechins (GTCs) are plant secondary metabolites that are believed to provide a variety of benefits for human health. Its major components include (−)-epigallocatechin 3-gallate (EGCG) which is the most abundant and well-studied catechins in green tea, and others, (−)-epigallocatechin (EGC), (−)-epicatechin gallate, and (−)-epicatechin (EC) (Fig. 1). So far, many studies demonstrated that their potential effects on anti-oxidative, anti-inflammatory, anti-infectious, and anti-cancerous properties in vitro and in vivo [1]. In 2006, Polyphenon E, a well-defined mixture of GTCs has been approved as a new drug for the treatment of genital warts by the U.S. Food and Drug Administration. To develop new treatments of polyphenon E, 30 clinical trials have been published with Polyphenon E alone or with other substances for treatment of cancer and cancer related diseases as of March 2017. Over the last two decades, scientists have discovered that GTCs have anti-infective properties with different modes of action on diverse families of viruses. GTCs inhibits the entry step of several viruses, such as influenza virus [2,3,4], human immunodeficiency virus (HIV) [5,6,7], and Chikungunya virus [8]. Few studies evaluated the effect of catechins and their derivatives on Herpes simplex virus by direct inhibition of viral particles [9,10,11]. Recently, several independent groups have identified GTCs as a potent inhibitor against hepatitis B virus (HBV) and hepatitis C virus (HCV). Hepatitis C is an infection caused by the HCV that causes liver inflammation and sometimes leading to serious liver damage [12]. The World Health Organization estimates that about 3% of the world's population has been infected with HCV and that there are more than 170 million chronic carriers who are at risk of developing liver cirrhosis, disease, and hepatocellular carcinoma [13,14]. No vaccine is available. A current standard therapy with pegylated interferon (IFN)-α and ribavirin has a limited efficacy and significant side effects [15]. Very recently, Interferon-free combination therapies, direct-acting antiviral drugs (DAAs) were approved for HCV treatment in the United States and Europe in 2011. However, the development of resistance has become a major concern for the DAA therapy and the longterm consequence of these treatment-emergent variants is still to be defined. The cost of DAAs also is significant and will give a large burden on health care expenses, thereby development of novel drugs with higher efficacy and more favorable side effect profiles to treat HCV infection is warranting. In this study, the potential of GTCs on anti-viral infection against hepatitis B and C viruses is elucidated in detail with an overview of research achievements.
Fig. 1. Schematic diagram of catechins. EC, epicatechin; EGC, epigallocatechin; ECG, epicatechin-gallate; EGCG, epigallocatecin 3-gallate.
Anti-hepatic Viral Activities of Catechins and Their Derivatives
Experimental studies have reported that catechins and their derivative are potential candidates as a preventive and antiviral drug for HCV infection. Chen et al. [16] showed that HCV infection was significantly suppressed by EGCG in Huh7.5.1 cells using the JFH1 strain of HCV genotype 2a that produces infectious virus particles in cell culture. The inhibition of HCV RNA replication and protein expression were monitored by treatment of EGCG with a 50% effective concentration (EC50) of 17.9 µM. Both HCV entry and viral RNA replication were affected by EGCG but not translation directed by the viral internal ribosome entry site. Time-of-addition experiments suggested that the gallate moiety is essential for the anti-HCV activity of catechins. Fukazawa et al. [17] also discovered that the anti-HCV properties of EGCG and 7,8-benzoflavone using a cell-based, microplate colorimetric screen system. HCV entry step is the main target of EGCG inhibitory effect but 7,8-benzoflavone was assumed that the inhibitor for the post-entry stage. Interestingly, EGC which has been reported no obvious inhibitory activity on HCV infection showed anti-HCV activities similar to that of EGCG in this study. Catechin derivatives [18] that contain several additional galloyl moieties have been synthesized and evaluated for their in vitro inhibitory effects against HCV infection using HCV subgenomic replicon. Based on the structure-activity relationship between synthetic compounds, the 2,3,4-trihydroxybenzoyl moiety in tripodal compounds and 12 polymethylene in linear compounds provides better antiviral activities than 3,4,5-trihydroxybenzoyl moiety in natural green catechins. All these anti-HCV activities were observed at concentration below the cytotoxicity threshold.
A few studies have been carried out to investigate the potential anti-HBV activities of catechins. Xu et al. [19] explored the antiviral properties of natural green tea extract (GTE) in a stably expressed HBV cell line HepG2-N10 by analyzing level of viral antigens and DNA. Results showed that the expression of both HBV surface antigen (HbsAg) and HBV envelop antigen (HbeAg) were significantly inhibited followed by treatment of GTE in cell culture medium with an EC50 of 5.02 µg/mL and 5.68 µg/mL, respectively. Quantitative real-time polymerase chain reaction revealed that GTE inhibited the production of extracellular HBV DNA in the medium and intracellular replicative intermediates within the treated cells. Interestingly, these antiviral properties of GTE were more effective than EGCG. Another investigation showed similar results. Ye et al. [20] examined anti-hepatitis viral effect of tea catechin mixture on HBV-antigen secretion and DNA production in a stable HBV-transfected HepG2 cell line. The powder of tea polyphenols including EGCG and other catechins significantly inhibited the secretion of HbeAg and HBV DNA released into the supernatant in a dose-dependent with a 50% of inhibitory concentration (IC50) of 7.34 µg/mL and 2.54 µg/mL of respectively. In another investigation, He et al. [21] demonstrated somewhat different results. When HepG2.117 cells were grown in the presence of EGCG, the expression of HBeAg was suppressed, however, the expression of HBsAg was not affected. In HepG2.117, a newly reported cell line, HbsAg could be produced from both integral HBV genome and covalently closed circular DNA (cccDNA), but mostly from integral HBV. Therefore, EGCG does not target to chromosomeintegrated HBV genome but only replicative intermediates of DNA synthesis which resulted in less cccDNA production.
Mechanistic Studies
Currently, mechanistic studies revealed that EGCG can interfere with HCV attachment on early phase of viral entry. Ciesek et al. [22] showed that EGCG inhibited HCV cell entry and cell-to-cell spread between neighboring cells but had no effect on HCV RNA replication, assembly, or release of progeny virions. In this study, the antiviral activity of EGCG but no other green tea cathchins such as EGC, EC, and epicatechin-gallate was confirmed both in cell-culture-derived HCV and HCV pseudoparticles (HCVpp) infection in primary human hepatocytes. Treatment of EGCG with Huh-7.5 cells before viral infection did not inhibit HCV infection whereas treatment with EGCG directly during inoculation strongly inhibited HCV infectivity. It means EGCG does not target on cell but HCV virion itself. Calland et al. [23] reported similar observations with the HCVpp expressing glycoprotein E1 and E2. EGCG treatment with different concentrations reduced HCV infectivity in a dose-dependent manner whatever the genotypes used. To confirm the inhibitory effect of EGCG on HCV attachment to target cells, 35S-HCV JFH-1 incubated with EGCG or control molecules was adsorbed onto Huh-7.5 cell and binding activity was evaluated by counts per minute bound to cells after washing with phosphate buffered saline. As a result, EGCG inhibited binding of HCV to cells but no other control molecules. EGCG also showed synergistic effects with boceprevir, a HCV protease (NS3/4A) and cyclosporine A which inhibit HCV replication. This group [24] also investigated another polyphenols which has anti-HCV infective potential. Delphinidin, an anthocyanidin present in plant pigment, was identified as a new HCV entry inhibitor with enhanced activity compared to that of EGCG. The mode of actions is similar with EGCG, leading to a loss of viral particles shape that might impair its attachment to the target cell surface. Another detailed mechanistic study was performed by Colpitts and Schang [25]. They showed that EGCG interacted with tryptophan residues in surface proteins in R18-labeled HCV virion by R-18 fluorescence emission spectra. They also found that EGCG competed with both heparan sulfates and sialic acid moieties in cellular glycans for virion binding. Since cellular glycans is a normal target molecule in a cell surface for attachment of most human viruses, this results supported broad-spectrum antiviral activities of EGCG targeting many virions attachment such HCV, HIV, and influenza virus. A recently study showed that the ability of EGCG to enhance innate immune responses against HCV in hepatocytes [26]. Although EGCG alone had little effect on the stimulation of HCV related innate immune responses, the combinatorial treatment of EGCG and HCV dsRNA (core, E1-P7, NS-3′NTR, and NS5A) significantly enhanced HCV dsRNA-induced intracellular innate immunity mediated by IFN-λ1, TLR3, RIG-I, and ISG in JFH-1‒infected Huh7 cells. Although extensive studies are required, this result suggested the potential anti-HCV property of EGCG in combination therapy with current HCV drugs.
Recent studies showed that autophagy has an important role in replication of many viruses including HBV and HCV [27,28,29]. Autophagy is an intracellular degradation system for unnecessary or dysfunctional components such as damaged organelles and foreign pathogens by the formation of doublemembraned vesicle known as an autophagosome. The formation of autophagosomes is regulated and controlled by genes such as Atg genes through Atg12 and LC3 complexes [30]. Zhong et al. [31] investigated the effect of EGCG on the relationship between replication of HBV and autophagosome formation in hepatocellular carcinoma. They showed that HBV transfection in HepG2 cells induced incomplete autophagy which was required for viral replication whereas EGCG induced a complete autophagy by increasing lysosomal acidification which did not facilitate HBV replication. Finally, EGCG treatment efficiently down-regulated HBV DNA level in HepG2.2.15 cells with dose-dependently. A previous study showed that resveratrol triggered an autophagic cell death via up-regulation of Atg5 and Atg12 expression in Huh-7 cell [32]. This anti-proliferative property of resveratrol in hepatocellular carcinoma could be an important role in HCV infection.
Alternative Approaches
Alternative approaches for the control of HCV infection have been studied in clinical and pre-clinical. Mekky et al. [33] suggested the potential role of EGCG in cellular during HCV entry via up-regulation of mir-194 expression followed by downregulation of CD81 expression which interacts with the HCV envelop protein 2 to facilitate endocytosis of the virus. Bioinformatics analysis was carried out to establish the miR-194 binding potential to CD81 receptor. Since miR-194 expression levels correlated to CD81 expression in liver biopsies of HCV patients in this study, this suggests that the miR-194 can act as a hepatocyte gate keeper hindering HCV entry through targeting CD81 expression and EGCG can enhance this inhibitory effect on HCV infection. Lee et al. [34] also showed cellular effects of (+)-catechin-containing natural extracts from San-Huang-Xie-Xin-Tang (SHXT), a transitional Chinese herbal formula, composed of Rhei rhizoma, Scutellaria radix and Coptidis rhizome. SHXT-frC, a catechin fraction extracted from SHXT, showed a concentration-dependent decrease of cyclooxygenase-2 (COX-2) expression. Recently studies showed that chronic inflammation and progress of hepatic carinogenesis is promoted by up-regulation of COX-2 expression [35] and COX-2 protein levels favor increased HCV replication [35,36,37]. Although the exact mechanism remains to be elucidated, SHXT-frC inhibits the improper induction of COX-2 by HCV nonstructural protein and may serve as a potential supplementary treatment for patients with chronic hepatitis C.
In Vitro Studies
A few in vitro studies have been carried out to establish the potential of EGCG against HCV. Roh and Jo [38] elucidated that recombinant NS5B which has HCV viral protease activity can be inhibited by EGCG interaction through a high throughput screening strategy using optical nanoparticle-based RNA oligonucleotide. Fatima et al. [39] performed the docking studies of Pakistani HCV NS3 helicase with six compounds, quercetin, β-carotene, resveratrol, catechins, lycopene and lutein, to investigate a possible antiviral drug target. Based on the computational docking analysis, quercetin and catechins can interact with NS3 helicase with best docking energy followed by resveratrol and lutein. Previously, Huang et al. [40] purposed a HBV infection system using an immortalized cell line derived from human primary hepatocytes, HuS-E/2, which becomes injectable after dimethyl sulfoxide (DMSO)-induced differentiation. In these studies, the authors showed that the HBV entry into DMSO-differentiated HuS-E/2 cells is mediated by the clathrin-dependent endocytosis pathway. In further investigation, Huang et al. [41] also showed that the presence of 50 µM EGCG decreased levels of HBV cccDNA and HBV mRNA during HBV infection. To evaluate the effects of EGCG on HBV entry step, transferrin uptake assay was performed to directly assess whether EGCG would interfere with clathrin-mediated endocytosis [41]. EGCG inhibited transferrin uptake and interfered with clathrin-mediated endocytosis of transferrin in DMSO-differentiated HuS-E/2 cells. EGCG interfered with HBV entry by blocking the endocytosis/ fusion step. Moreover, this in vitro anti-HBV activity of EGCG on HBV entry was observed using four different genotypes, A to D.
Conclusion
Despite that vaccines and several anti-viral hepatitis drugs are available for patients with HBV and HCV, hepatotropic viral infection is still a severe public health problem in the world. In this review, many natural compounds especially polyphenol and their derivatives are already available with diverse biological activities (Table 1) and these offer a large opportunity for finding novel antiviral lead compounds or good candidates with unique anti-hepatitis viral drugs. Although there is an increasing amount of evidence that polyphenol compounds have anti-hepatitis viral effects to date, but they are limited to cell-based or animal-based studies. So it needs to be carefully assessed whether data from animal experiments are relevant to human, and future efforts should be devoted to optimize and develop for clinical application. When some of these compounds become clinically available, a new path for chemoprevention of hepatotropic viral infection will be opened and control of liver diseases could be dramatically improved.
Table 1. Mechanism of HCV inhibition by catechins and their derivatives.
| Category | Test compound | Propagation and target cells | Genotype | Mode of action | Molecular target of inhibition | Reference |
|---|---|---|---|---|---|---|
| Catechin and derivatives | EGCG | HCVcc (JFH1) and Huh7.5.1 | 2a | Suppression of both the HCV entry and RNA replication steps | ND | [16] |
| EGCG, 7,8-benzoflavone | HCVcc (JFH1) and Huh7.5.1 | 2a, 1b | Early step (EGCG) and later stage (7,8-benzoflavone) | ND | [17] | |
| 63 Synthetic derivatives | Huh5.2 | - | In vitro inhibitory effect of catechin derivatives (linear and branched alkyl-esters, amides of gallic acid and other (mono-, di-, and tri-) hydroxy benzoyl derivatives) | ND | [18] | |
| Mechanistic studies | EGCG | HCVcc, HCVpp and Huh-7.5 | 1a, 1b, 2a | Inhibition of virion attachment | E1/E2 | [22] |
| EGCG | HCVcc, HCVpp and Huh-7 | 1a, 1b, 2a, 2b, 3a, 4, 5, 6 | EGCG possesses antiviral activity and impairs cellular lipid metabolism | E1/E2 | [23] | |
| EGCG, delphinidin | HCVcc, HCVpp and Huh-7 | 2a | E1, E2 interaction mediated deformation of viral membrane | E1/E2 | [24] | |
| EGCG | HCVcc (JFH1), Huh-7.5 | 2a | Competition with heparan sulfate for binding of HCV | Heparan sulfate | [25] | |
| Alternative approaches | EGCG | HCVcc (JFH1), Huh-7 | 2a | Regulation of mir-194 and CD81 expression levels | miR-194, CD81 | [33] |
| SHXT | HCVcc (JFH1), Huh-7 | 2a | Modulation of the activation of NF-κB and COX-2 | COX-2 | [34] | |
| In vitro studies | EGCG | In vitro | - | QDs-RNA oligonucleotide | NS5B | [38] |
| EGCG | In silico | - | Binding Interaction of HCV NS3 helicase active pocket | GT3a NS3 helicase | [39] |
HCV, hepatitis C virus; EGCG, (−)-epigallocatechin 3-gallate; HCVcc, cell-culture-derived HCV; ND, not determined; HCVpp, HCV pseudoparticles; SHXT, San-Huang-Xie-Xin-Tang; NF-κB, nuclear factor κB; COX-2, cyclooxygenase-2; QD, quantum dots.
No potential conflict of interest relevant to this article was reported.
This work was supported by the Sungshin University Research Grant of 2015-2-11-075.
References
- 1.Cao J, Han J, Xiao H, Qiao J, Han M. Effect of tea polyphenol compounds on anticancer drugs in terms of anti-tumor activity, toxicology, and pharmacokinetics. Nutrients. 2016;8:E762. doi: 10.3390/nu8120762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nakayama M, Suzuki K, Toda M, Okubo S, Hara Y, Shimamura T. Inhibition of the infectivity of influenza virus by tea polyphenols. Antiviral Res. 1993;21:289–299. doi: 10.1016/0166-3542(93)90008-7. [DOI] [PubMed] [Google Scholar]
- 3.Kim M, Kim SY, Lee HW, et al. Inhibition of influenza virus internalization by (−)-epigallocatechin-3-gallate. Antiviral Res. 2013;100:460–472. doi: 10.1016/j.antiviral.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 4.Song JM, Lee KH, Seong BL. Antiviral effect of catechins in green tea on influenza virus. Antiviral Res. 2005;68:66–66. doi: 10.1016/j.antiviral.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 5.Yamaguchi K, Honda M, Ikigai H, Hara Y, Shimamura T. Inhibitory effects of (−)-epigallocatechin gallate on the life cycle of human immunodeficiency virus type 1 (HIV-1) Antiviral Res. 2002;53:19–34. doi: 10.1016/s0166-3542(01)00189-9. [DOI] [PubMed] [Google Scholar]
- 6.Fassina G, Buffa A, Benelli R, Varnier OE, Noonan DM, Albini A. Polyphenolic antioxidant (−)-epigallocatechin-3-gallate from green tea as a candidate anti-HIV agent. AIDS. 2002;16:939–941. doi: 10.1097/00002030-200204120-00020. [DOI] [PubMed] [Google Scholar]
- 7.Williamson MP, McCormick TG, Nance CL, Shearer WT. Epigallocatechin gallate, the main polyphenol in green tea, binds to the T-cell receptor, CD4: potential for HIV-1 therapy. J Allergy Clin Immunol. 2006;118:1369–1374. doi: 10.1016/j.jaci.2006.08.016. [DOI] [PubMed] [Google Scholar]
- 8.Weber C, Sliva K, von Rhein C, Kummerer BM, Schnierle BS. The green tea catechin, epigallocatechin gallate inhibits chikungunya virus infection. Antiviral Res. 2015;113:1–3. doi: 10.1016/j.antiviral.2014.11.001. [DOI] [PubMed] [Google Scholar]
- 9.Lyu SY, Rhim JY, Park WB. Antiherpetic activities of flavonoids against herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) in vitro. Arch Pharm Res. 2005;28:1293–1301. doi: 10.1007/BF02978215. [DOI] [PubMed] [Google Scholar]
- 10.Isaacs CE, Wen GY, Xu W, et al. Epigallocatechin gallate inactivates clinical isolates of herpes simplex virus. Antimicrob Agents Chemother. 2008;52:962–970. doi: 10.1128/AAC.00825-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Isaacs CE, Xu W, Merz G, Hillier S, Rohan L, Wen GY. Digallate dimers of (−)-epigallocatechin gallate inactivate herpes simplex virus. Antimicrob Agents Chemother. 2011;55:5646–5653. doi: 10.1128/AAC.05531-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shepard CW, Finelli L, Alter MJ. Global epidemiology of hepatitis C virus infection. Lancet Infect Dis. 2005;5:558–558. doi: 10.1016/S1473-3099(05)70216-4. [DOI] [PubMed] [Google Scholar]
- 13.Kenny-Walsh E. The natural history of hepatitis C virus infection. Clin Liver Dis. 2001;5:969–977. doi: 10.1016/s1089-3261(05)70204-x. [DOI] [PubMed] [Google Scholar]
- 14.Alter MJ. Epidemiology of hepatitis C. Hepatology. 1997;26(3 Suppl 1):62S–65S. doi: 10.1002/hep.510260711. [DOI] [PubMed] [Google Scholar]
- 15.Manns MP, Wedemeyer H, Cornberg M. Treating viral hepatitis C: efficacy, side effects, and complications. Gut. 2006;55:1350–1359. doi: 10.1136/gut.2005.076646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen C, Qiu H, Gong J, et al. (−)-Epigallocatechin-3-gallate inhibits the replication cycle of hepatitis C virus. Arch Virol. 2012;157:1301–1312. doi: 10.1007/s00705-012-1304-0. [DOI] [PubMed] [Google Scholar]
- 17.Fukazawa H, Suzuki T, Wakita T, Murakami Y. A cell-based, microplate colorimetric screen identifies 7,8-benzoflavone and green tea gallate catechins as inhibitors of the hepatitis C virus. Biol Pharm Bull. 2012;35:1320–1327. doi: 10.1248/bpb.b12-00251. [DOI] [PubMed] [Google Scholar]
- 18.Rivero-Buceta E, Carrero P, Doyaguez EG, et al. Linear and branched alkyl-esters and amides of gallic acid and other (mono-, di- and tri-) hydroxy benzoyl derivatives as promising anti-HCV inhibitors. Eur J Med Chem. 2015;92:656–671. doi: 10.1016/j.ejmech.2015.01.033. [DOI] [PubMed] [Google Scholar]
- 19.Xu J, Wang J, Deng F, Hu Z, Wang H. Green tea extract and its major component epigallocatechin gallate inhibits hepatitis B virus in vitro. Antiviral Res. 2008;78:242–249. doi: 10.1016/j.antiviral.2007.11.011. [DOI] [PubMed] [Google Scholar]
- 20.Ye P, Zhang S, Zhao L, et al. Tea polyphenols exerts antihepatitis B virus effects in a stably HBV-transfected cell line. J Huazhong Univ Sci Technolog Med Sci. 2009;29:169–169. doi: 10.1007/s11596-009-0206-1. [DOI] [PubMed] [Google Scholar]
- 21.He W, Li LX, Liao QJ, Liu CL, Chen XL. Epigallocatechin gallate inhibits HBV DNA synthesis in a viral replicationinducible cell line. World J Gastroenterol. 2011;17:1507–1514. doi: 10.3748/wjg.v17.i11.1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ciesek S, von Hahn T, Colpitts CC, et al. The green tea polyphenol, epigallocatechin-3-gallate, inhibits hepatitis C virus entry. Hepatology. 2011;54:1947–1955. doi: 10.1002/hep.24610. [DOI] [PubMed] [Google Scholar]
- 23.Calland N, Albecka A, Belouzard S, et al. (−)-Epigallocatechin-3-gallate is a new inhibitor of hepatitis C virus entry. Hepatology. 2012;55:720–729. doi: 10.1002/hep.24803. [DOI] [PubMed] [Google Scholar]
- 24.Calland N, Sahuc ME, Belouzard S, et al. Polyphenols inhibit hepatitis C virus entry by a new mechanism of action. J Virol. 2015;89:10053–10063. doi: 10.1128/JVI.01473-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Colpitts CC, Schang LM. A small molecule inhibits virion attachment to heparan sulfate- or sialic acid-containing glycans. J Virol. 2014;88:7806–7817. doi: 10.1128/JVI.00896-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang Y, Li J, Wang X, et al. (−)-Epigallocatechin-3-gallate enhances hepatitis C virus double-stranded RNA intermediates-triggered innate immune responses in hepatocytes. Sci Rep. 2016;6:21595. doi: 10.1038/srep21595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wong J, Zhang J, Si X, et al. Autophagosome supports coxsackievirus B3 replication in host cells. J Virol. 2008;82:9143–9143. doi: 10.1128/JVI.00641-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li J, Liu Y, Wang Z, et al. Subversion of cellular autophagy machinery by hepatitis B virus for viral envelopment. J Virol. 2011;85:6319–6333. doi: 10.1128/JVI.02627-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sir D, Kuo CF, Tian Y, et al. Replication of hepatitis C virus RNA on autophagosomal membranes. J Biol Chem. 2012;287:18036–18043. doi: 10.1074/jbc.M111.320085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tanida I. Autophagy basics. Microbiol Immunol. 2011;55:1–11. doi: 10.1111/j.1348-0421.2010.00271.x. [DOI] [PubMed] [Google Scholar]
- 31.Zhong L, Hu J, Shu W, Gao B, Xiong S. Epigallocatechin-3-gallate opposes HBV-induced incomplete autophagy by enhancing lysosomal acidification, which is unfavorable for HBV replication. Cell Death Dis. 2015;6:e1770. doi: 10.1038/cddis.2015.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liao PC, Ng LT, Lin LT, Richardson CD, Wang GH, Lin CC. Resveratrol arrests cell cycle and induces apoptosis in human hepatocellular carcinoma Huh-7 cells. J Med Food. 2010;13:1415–1423. doi: 10.1089/jmf.2010.1126. [DOI] [PubMed] [Google Scholar]
- 33.Mekky RY, El-Ekiaby NM, Hamza MT, et al. Mir-194 is a hepatocyte gate keeper hindering HCV entry through targeting CD81 receptor. J Infect. 2015;70:78–87. doi: 10.1016/j.jinf.2014.08.013. [DOI] [PubMed] [Google Scholar]
- 34.Lee JC, Tseng CK, Wu SF, Chang FR, Chiu CC, Wu YC. San-Huang-Xie-Xin-Tang extract suppresses hepatitis C virus replication and virus-induced cyclooxygenase-2 expression. J Viral Hepat. 2011;18:e315–e324. doi: 10.1111/j.1365-2893.2010.01424.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lu L, Wei L, Peng G, et al. NS3 protein of hepatitis C virus regulates cyclooxygenase-2 expression through multiple signaling pathways. Virology. 2008;371:61–70. doi: 10.1016/j.virol.2007.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nunez O, Fernandez-Martinez A, Majano PL, et al. Increased intrahepatic cyclooxygenase 2, matrix metalloproteinase 2, and matrix metalloproteinase 9 expression is associated with progressive liver disease in chronic hepatitis C virus infection: role of viral core and NS5A proteins. Gut. 2004;53:1665–1672. doi: 10.1136/gut.2003.038364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Giannitrapani L, Ingrao S, Soresi M, et al. Cyclooxygenase-2 expression in chronic liver diseases and hepatocellular carcinoma: an immunohistochemical study. Ann N Y Acad Sci. 2009;1155:293–299. doi: 10.1111/j.1749-6632.2009.03698.x. [DOI] [PubMed] [Google Scholar]
- 38.Roh C, Jo SK. (−)-Epigallocatechin gallate inhibits hepatitis C virus (HCV) viral protein NS5B. Talanta. 2011;85:2639–2639. doi: 10.1016/j.talanta.2011.08.035. [DOI] [PubMed] [Google Scholar]
- 39.Fatima K, Mathew S, Suhail M, et al. Docking studies of Pakistani HCV NS3 helicase: a possible antiviral drug target. PLoS One. 2014;9:e106339. doi: 10.1371/journal.pone.0106339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huang HC, Chen CC, Chang WC, Tao MH, Huang C. Entry of hepatitis B virus into immortalized human primary hepatocytes by clathrin-dependent endocytosis. J Virol. 2012;86:9443–9453. doi: 10.1128/JVI.00873-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Huang HC, Tao MH, Hung TM, Chen JC, Lin ZJ, Huang C. (−)-Epigallocatechin-3-gallate inhibits entry of hepatitis B virus into hepatocytes. Antiviral Res. 2014;111:100–111. doi: 10.1016/j.antiviral.2014.09.009. [DOI] [PubMed] [Google Scholar]


