Abstract
The present study retrospectively analyzed 19 patients diagnosed with paraquat (PQ) poisoning with the aim to investigate the effect of activated charcoal hemoperfusion on renal function and PQ elimination. The results indicated that 7 patients died and 12 survived. Non-oliguric renal failure occurred in all of the 7 patients who died. Among the 12 surviving patients, 10 had normal renal function and 2 developed non-oliguric renal failure. There was a linear correlation between plasma and urine paraquat concentration prior to and during activated charcoal hemoperfusion. The equation parameters together with the correlation coefficient on admission were as follows: Y=0.5820+1.7348X (R2=0.678; F=35.768; P<0.0001). The equation parameters together with the correlation coefficient were as follows during activated charcoal hemoperfusion: Y=0.6827+1.2649X (R2=0.626; F=50.308; P<0.0001). Therefore, it was concluded that in patients with normal renal function, the elimination kinetics of PQ by the kidneys were only associated with the plasma PQ concentration. Activated charcoal hemoperfusion had little effect on avoiding acute kidney injury in patients with severe PQ poisoning.
Keywords: paraquat, poisoning, kidney, metabolism, activated charcoal hemoperfusion
Introduction
Paraquat (PQ) is an effective and commercially important herbicide widely used throughout the world. However, the mortality rate of PQ poisoning has been reported to range from 50 to 90% when the pesticide is ingested either accidentally or intentionally as a suicide attempt (1). A previous study suggested that absorbed PQ requires removal from the bloodstream in order to improve the survival rate of patients with severe PQ poisoning (2).
PQ is not metabolized to any extent and is rapidly excreted in the urine after PQ poisoning even at low doses. The kidneys are effective at eliminating PQ but are vulnerable to PQ injury (3,4). Therefore, elimination of PQ simply relying on the kidney is slow, and removal of PQ from the blood by activated charcoal hemoperfusion (HP) in the first 12–15 h following ingestion may be beneficial (5). Various studies have indicated that HP is more efficient in the clearance of plasma PQ than the kidneys (2). Therefore, activated charcoal HP is widely used in the treatment of PQ poisoning in China (6).
Although the kidneys and HP are the two major routes of eliminating PQ following ingestion (7,8), renal excretion is considered to be the major natural pathway of PQ elimination. Therefore, the initial renal function is an important factor for survival.
Application of HP has been reported to accelerate the removal of PQ from the blood. However, to the best of our knowledge, no previous study has focused on the effect of HP on renal function and PQ elimination by the kidneys. The present study determined the effect of HP on renal function and PQ elimination via the kidneys.
Materials and methods
Patients
A total of 19 patients with PQ poisoning were respectively observed in the present study. The subject selection criteria included the following: i) Oral PQ poisoning; ii) no acute kidney injury on admission; and iii) age of <65 years and <18 years. Potential participants were excluded if they were initially treated at a different hospital, or had any known cardiac, pulmonary or other chronic disease associated with a certain degree of renal failure as an underlying condition of PQ poisoning. Acute kidney injury was defined by a serum creatinine (SCr) level of >97 µmol/l and blood urea nitrogen (BUN) levels of >8.3 mmol/l.
Treatment of the patients
All patients received HP therapy (Braun Diapact CRRT machine; Braun GmbH, Kronberg im Taunus, Germany) until plasma PQ levels became undectable, and then received continuous veno-venous hemofiltration therapy for 12 h after HP. To prevent absorption of PQ via the gastrointestinal tract, gastric lavage was performed via a nasogastric tube using 1 g/kg activated charcoal in 500 ml 0.9% saline for one time every 4 h. Furthermore, Smecta (Beaufour Ipsen Pharmacy Co., Ltd., Tianjin, China) and magnesium sulfate (Huairen Pharmacy Co., Ltd., Tianjin, China) powder were placed into 20% mannitol (Shuanghe Pharmacy Co., Ltd., Tianjin, China) was administered via the anus.
Other treatments included intravenous infusion of cyclophosphamide (Guangdong Qingping Pharmacy Co., Ltd., Guangzhou, China), methylprednisolone sodium succinate injection (Pfizer, Inc., New York, NY, USA) and intravenous injections of dexamethasone (Jilin Extrawell Changbaishan Pharmaceutical Co., Ltd., Jilin, China). Vitamin E capsules (Xinyi Pharmaceutical Co., Ltd., Shanghai, China), metoprolol (Astra Zeneca, London, UK) and vitamin E injections (Zhongjing Biotechnology Co., Ltd., Harbin, China) were also administered.
Data collection
The following data were collected: Demographical factors (age, sex and medical history), initial BUN, SCr, plasma and urine PQ concentration within 12 h of admission to the intensive care unit. Samples of plasma and urine were collected every 3 h while HP was performed. BUN and SCr were measured with a full automatic biochemical analyzer (AU5800; Beckman Coulter, Inc., Brea, CA, USA). Quantitative analysis of the plasma and urine PQ concentration was performed at the hospital laboratory by a gas chromatography method (9).
Statistical analysis
SPSS statistical software package 20.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism v4.0 (GraphPad Software, Inc., La Jolla, CA, USA) were used to perform statistical analysis. Values are expressed as the mean ± standard deviation. Categorical variables are expressed as numbers or percentages for each item. Statistically significant differences between the two groups were analyzed using the independent two-samples t-test or the Mann-Whitney U test. The Chi-square test was used to assess the association between treatment protocols and survival rate. P<0.05 was considered to indicate a statistically significant difference.
Results
Demographic and laboratory data
Demographic and laboratory results of the patients are summarized in Table I. A total of 19 patients were analyzed, including 8 females and 11 males (mean age, 40.37±12.55 years; range, 25–63 years). A total of 7 patients died from pulmonary fibrosis and 12 survived. There were no significant inter-group differences in age or time between poisoning and HP therapy.
Table I.
Clinical status of each of the 19 patients.
| SCr (µmol/l) | BUN (mmol/l) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Subject no. | Sex | Age (years) | Initial PPC (mg/l) | Initial UPC (mg/l) | Time between poisoning and HP therapy (h) | Duration of activated charcoal HP therapy (h) | Prior to HP | After HP | At discharge | Prior to HP | After HP | At discharge | Outcome |
| 1 | Male | 39 | 5.7 | 9.2 | 7 | 18 | 69 | 62 | 258 | 5.1 | 4.5 | 12.9 | Death |
| 2 | Male | 28 | 6.3 | 12.1 | 5 | 17 | 64 | 94 | 100 | 4.0 | 6.5 | 6.8 | Death |
| 3 | Female | 30 | 10.1 | 21.7 | 6 | 20 | 63 | 91 | 228 | 3.5 | 5.1 | 14.1 | Death |
| 4 | Female | 54 | 6.5 | 15.7 | 6 | 17 | 68 | 57 | 135 | 6.5 | 9.5 | 13.1 | Death |
| 5 | Male | 30 | 2.1 | 4.4 | 8 | 14 | 76 | 79 | 99 | 5.8 | 5.1 | 13.5 | Survival |
| 6 | Female | 39 | 4.6 | 11.2 | 10 | 16 | 32 | 112 | 53 | 4.2 | 8.5 | 7.0 | Survival |
| 7 | Female | 49 | 2.3 | 6.2 | 8.5 | 17 | 31 | 45 | 78 | 3.5 | 2.6 | 10.9 | Survival |
| 8 | Female | 56 | 1.6 | 2.8 | 9 | 16 | 49 | 86 | 84 | 6.3 | 7.8 | 9.4 | Survival |
| 9 | Male | 58 | 9.8 | 18.6 | 5.5 | 23 | 85 | 84 | 208 | 7.9 | 9.3 | 13.5 | Death |
| 10 | Male | 25 | 11.8 | 23.7 | 7 | 16 | 57 | 109 | 153 | 4.0 | 6.4 | 10.6 | Death |
| 11 | Female | 48 | 1.45 | 3.5 | 5 | 8 | 46 | 38 | 51 | 5.7 | 5.3 | 6.8 | Survival |
| 12 | Male | 47 | 4.1 | 6.1 | 10.5 | 17 | 70 | 101 | 188 | 5.1 | 8.9 | 11.2 | Death |
| 13 | Female | 25 | 0.1 | 0.7 | 4.5 | 6 | 45 | 38 | 49 | 3.7 | 7.0 | 3.5 | Survival |
| 14 | Female | 29 | 3.1 | 4.5 | 6.5 | 12 | 72 | 57 | 86 | 5.4 | 6.8 | 8.1 | Survival |
| 15 | Male | 33 | 2.2 | 3.5 | 8 | 14 | 45 | 46 | 46 | 2.9 | 2.4 | 6.3 | Survival |
| 16 | Male | 63 | 3.6 | 6.1 | 9 | 18 | 45 | 74 | 145 | 5.4 | 7.0 | 14.1 | Survival |
| 17 | Male | 36 | 3.5 | 6.9 | 5.5 | 14 | 78 | 75 | 82 | 4.6 | 4.7 | 6.3 | Survival |
| 18 | Male | 50 | 1.1 | 1.7 | 11 | 8 | 48 | 49 | 62 | 3.7 | 4.6 | 5.6 | Survival |
| 19 | Male | 26 | 4.3 | 16.7 | 6.5 | 6 | 69 | 92 | 88 | 4.5 | 3.5 | 7.6 | Survival |
BUN, blood urea nitrogen (normal range, 3.2–7.1 mmol/l); SCr, serum creatinine (normal range, 71–133 µmol/l); UPC, urine paraquat concentration; PPC, plasma paraquat concentration; HP, charcoal hemoperfusion.
The renal function of all patients was normal on admission, and that of 16 patients remained normal after HP. However, non-oliguric renal failure occurred in all of the 7 patients who died. Among the 12 surviving patients, 10 had a normal renal function, while 2 (subjects nos. 5 and 16) developed non-oliguric renal failure (Table I).
The group of patients who did not survive had a higher initial plasma PQ concentration and longer time of activated charcoal HP therapy than the group of survivors. All patients exhibited a progressive increase in SCr and BUN levels during hospitalization. However, there were no significant inter-group differences in BUN at baseline, as well as SCr and BUN after HP. However, the group of non-survivors had a higher SCr and BUN than the survivors at discharge (Table II).
Table II.
Demographic and laboratory data of patients stratified by outcome.
| SCr (µmol/l) | BUN (mmol/l) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Group | n | Age (years) | Initial PPC (mg/l) | Delay between poisoning and HP therapy (h) | Time of activated 1 charcoa HP therapy (h) | Prior to HP | After HP | At discharge | Prior to HP | After HP | At discharge |
| Death | 7 | 40.43±13.53 | 7.75±2.81 | 6.71±1.82 | 18.29±2.43 | 68.00±8.72 | 85.43±19.42 | 181.43±55.34 | 5.16±1.57 | 7.17±2.06 | 11.74±2.51 |
| Survival | 12 | 40.33±12.56 | 2.50±1.35 | 7.63±2.04 | 12.42±4.34 | 53.00±16.45 | 65.91±23.90 | 76.92±27.97 | 4.68±1.13 | 5.44±2.00 | 8.26±3.18 |
| P-value | 0.988 | <0.001 | 0.339 | 0.005 | 0.040 | 0.449 | 0.085 | 0.090 | <0.001 | 0.024 | |
BUN, blood urea nitrogen (normal range, 3.2–7.1 mmol/l); SCr, serum creatinine (normal range, 71–133 µmol/l); PPC, plasma paraquat concentration; HP, charcoal hemoperfusion.
Effect of activated charcoal HP on the kidney toxicokinetics of paraquat
Plasma and urine PQ concentrations prior to and during HP are presented in Fig. 1. The plasma PQ concentration decreased rapidly when HP was performed. The concentrations of PQ in urine were associated with the plasma PQ concentration. When the plasma PQ concentration was >0.5 mg/l, the urine PQ concentration decreased in parallel with the plasma PQ concentration. Fig. 1 also revealed that urinary PQ concentrations were almost 2 times greater than plasma PQ concentrations when the plasma PQ concentration was >0.5 mg/l (P<0.05). When the plasma PQ concentration was <0.5 mg/l, the urine PQ concentration tended to be equal to the plasma PQ concentration.
Figure 1.
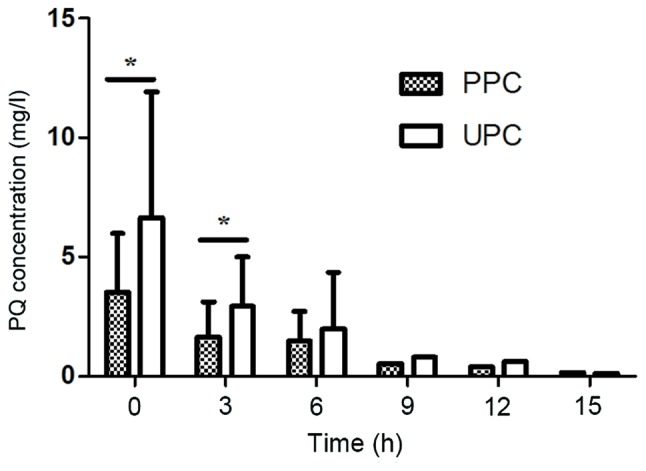
PPC and UPC at various time-points during HP therapy. Prior to HP therapy (0 h): PPC, 3.50±2.493 mg/l; UPC, 6.66±5.25 mg/l (n=19)-; 3 h after HP therapy: PPC, 1.65±1.477 mg/l; UPC, 2.94±2.085 mg/l (n=17); 6 h after HP therapy: PPC, 1.48±1.228 mg/l; UPC, 1.98±2.362 mg/l (n=10); 9 h after HP therapy: PPC, 0.50 mg/l; UPC, 0.80 mg/l (n=3); 12 h after HP therapy: PPC, 0.40 mg/l; UPC, 0.60 mg/l (n=1); 15 h after HP therapy: PPC, 0.15 mg/l; UPC, 0.1 mg/l (n=1). *P<0.05 as indicated. UPC, urine paraquat concentration; PPC, plasma paraquat concentration; HP, charcoal hemoperfusion.
The association between the plasma and urine PQ concentration prior to and during HP is presented in Fig. 2. At baseline, there was a linear association between plasma and urine PQ concentration. The equation parameters together with the correlation coefficient on admission were as follows: Y=0.5820+1.7348X (R2=0.678; F=35.768; P<0.0001). However, during HP, there was also a linear correlation between blood and urine PQ concentration. The equation parameters together with the correlation coefficient were as follows: Y=0.6827+1.2649X (R2=0.626; F=50.308; P<0.0001).
Figure 2.
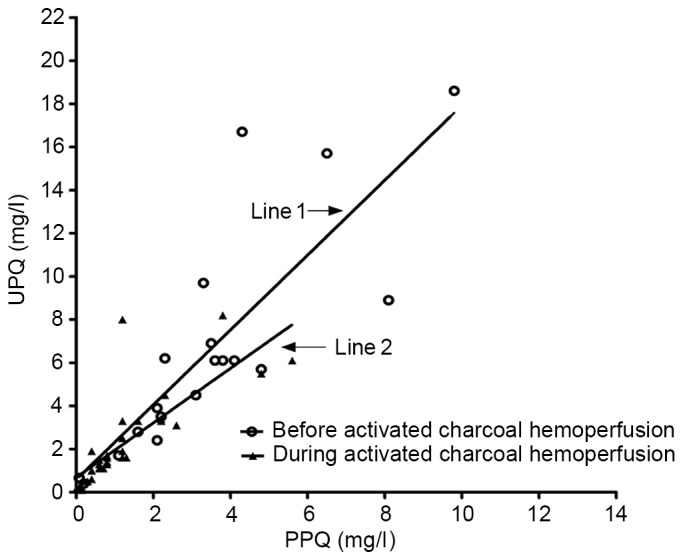
Line 1, correlation between PPC and UPC prior to HP therapy. Regression line, Y=0.5820+1.7348X (R2=0.678; F=35.768; P<0.0001); Line 2, correlation between PPQ and UPQ during HP therapy. Regression line, Y=0.6827+1.2649X (R2=0.626; F=50.308; P<0.0001). PPC, plasma paraquat concentration; UPC, urine paraquat concentration; HP, charcoal hemoperfusion.
Discussion
In the present study, HP was demonstrated to effectively eliminate PQ. The kidney toxicokinetics of PQ during HP were assessed, which were previously not well assessed in humans. In the present study, urinary and plasma PQ concentrations were continuously monitored while HP was performed. A linear correlation was identified between the plasma and urine PQ concentration while HP was performed. When the plasma PQ concentration was >0.5 mg/l, urinary concentrations of PQ were nearly 2 times greater than the plasma concentrations. It was suggested that the kidney toxicokinetics of PQ at a given time may be interpreted along with the plasma levels of PQ, and are not associated with HP.
However, these results did not agree with those of Ikebuchi (10), who reported that urinary concentrations of PQ were 3.3–4.5 times greater than the plasma concentrations if renal function was normal. This discrepancy is likely due to differences in test methods and study populations. Another reason may be that HP eliminates BUN and SCr, which affects the judgment of renal function.
Theoretically, accelerated removal of PQ from the blood should have a protective effect on the kidney. However, SCr and BUN of all patients displayed a progressive increase during hospitalization. Non-oliguric acute renal failure still occurred in 9 patients in spite of a rapid decrease in the PQ concentrations being observed. Not only the 7 patients who died, but also two of the surviving patients developed non-oliguric renal failure. However, no severe renal failure occurred in the survival group. These results suggested that HP has positive effects on avoiding kidney damage in patients with mild-to-moderate PQ-poisoning, and is ineffective for patients with severe PQ poisoning. It is likely that the potentially damaging concentration of PQ had already been attained in the kidney when HP was performed.
Finally, it should be noted that, even though renal function is normal in the early stage of PQ poisoning, and the toxin was removed effectively, this did not affect the clinical outcome in patients who had ingested a potentially lethal dose of PQ.
In conclusion, the present study suggested that the kidney toxicokinetics of PQ were only associated with the plasma PQ concentration in patients with normal renal function. Activated charcoal HP had little effect on avoiding acute kidney injury in patients with severe PQ poisoning.
Acknowledgements
The authors are grateful to the Basic Research Project of the Logistics University of Chinese People's Armed Police Force (grant no. WHJ2015020) for financially supporting the present study.
References
- 1.Jones GM, Vale JA. Mechanisms of toxicity, clinical features, and management of diquat poisoning: A review. J Toxicol Clin Toxicol. 2000;38:123–128. doi: 10.1081/CLT-100100926. [DOI] [PubMed] [Google Scholar]
- 2.Hsu CW, Lin JL, Lin-Tan DT, Chen KH, Yen TH, Wu MS, Lin SC. Early hemoperfusion may improve survival of severely paraquat-poisoned patients. PLoS One. 2012;7:e48397. doi: 10.1371/journal.pone.0048397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kang MS, Gil HW, Yang JO, Lee EY, Hong SY. Comparison between kidney and hemoperfusion for paraquat elimination. J Korean Med Sci. 2009;24(Suppl):S156–S160. doi: 10.3346/jkms.2009.24.S1.S156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pavan M. Acute kidney injury following paraquat poisoning in india. Iran J Kidney Dis. 2013;7:64–66. [PubMed] [Google Scholar]
- 5.Hawksworth GM, Bennett PN, Davies DS. Kinetics of paraquat elimination in the dog. Toxicol Appl Pharmacol. 1981;57:139–145. doi: 10.1016/0041-008X(81)90273-8. [DOI] [PubMed] [Google Scholar]
- 6.Hu L, Hong G, Ma J, Wang X, Lin G, Zhang X, Lu Z. Clearance rate and BP-ANN model in paraquat poisoned patients treated with hemoperfusion. Biomed Res Int. 2015;2015:298253. doi: 10.1155/2015/298253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pond SM, Rivory LP, Hampson EC, Roberts MS. Kinetics of toxic doses of paraquat and the effects of hemoperfusion in the dog. J Toxicol Clin Toxicol. 1993;31:229–246. doi: 10.3109/15563659309000391. [DOI] [PubMed] [Google Scholar]
- 8.Hong SY, Yang JO, Lee EY, Kim SH. Effect of haemoperfusion on plasma paraquat concentration in vitro and in vivo. Toxicol Ind Health. 2003;19:17–23. doi: 10.1191/0748233703th171oa. [DOI] [PubMed] [Google Scholar]
- 9.Posecion NC, Ostrea EM, Bielawski DM. Quantitative determination of paraquat in meconium by sodium borohydride-nickel chloride chemical reduction and gas chromatography/mass spectrometry (GC/MS) J Chromatogr B Analyt Technol Biomed Life Sci. 2008;862:93–99. doi: 10.1016/j.jchromb.2007.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ikebuchi J. Evaluation of paraquat concentrations in paraquat poisoning. Arch Toxicol. 1987;60:304–310. doi: 10.1007/BF01234670. [DOI] [PubMed] [Google Scholar]


