Abstract
Purpose
There is evidence from nonrandomized studies that a proportion of ipilimumab-treated patients with advanced melanoma experience long-term survival. To demonstrate a long-term survival benefit with ipilimumab, we evaluated the 5-year survival rates of patients treated in a randomized, controlled phase III trial.
Patients and Methods
A milestone survival analysis was conducted to capture the 5-year survival rate of treatment-naive patients with advanced melanoma who received ipilimumab in a phase III trial. Patients were randomly assigned 1:1 to receive ipilimumab at 10 mg/kg plus dacarbazine (n = 250) or placebo plus dacarbazine (n = 252) at weeks 1, 4, 7, and 10 followed by dacarbazine alone every 3 weeks through week 22. Eligible patients could receive maintenance ipilimumab or placebo every 12 weeks beginning at week 24. A safety analysis was conducted on patients who survived at least 5 years and continued to receive ipilimumab as maintenance therapy.
Results
The 5-year survival rate was 18.2% (95% CI, 13.6% to 23.4%) for patients treated with ipilimumab plus dacarbazine versus 8.8% (95% CI, 5.7% to 12.8%) for patients treated with placebo plus dacarbazine (P = .002). A plateau in the survival curve began at approximately 3 years. In patients who survived at least 5 years and continued to receive ipilimumab, grade 3 or 4 immune-related adverse events were observed exclusively in the skin.
Conclusion
The additional survival benefit of ipilimumab plus dacarbazine is maintained with twice as many patients alive at 5 years compared with those who initially received placebo plus dacarbazine. These results demonstrate a durable survival benefit with ipilimumab in advanced melanoma.
INTRODUCTION
Survival outcomes for patients with advanced melanoma have historically been poor, with the 5-year survival rate for patients with stage IV disease being less than 10%.1 Several new treatment options for advanced melanoma have become available in recent years, thus offering the potential to improve overall survival (OS) outcomes in these patients. The first therapy to improve OS in a phase III trial was ipilimumab,2,3 which was approved in 2011 for the treatment of unresectable or metastatic melanoma. Three targeted therapies have since been approved. Vemurafenib, based on an improvement in OS in a phase III trial, was approved in 2011 for the treatment of BRAF V600E–mutated unresectable or metastatic melanoma.4 More recently, the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib were approved for BRAF V600E–mutated and BRAF V600E- or BRAF V600K–mutated unresectable or metastatic melanoma, respectively, based on improvements in progression-free survival in phase III trials.4 The combination of the latter two agents is now approved by the US Food and Drug Administration for patients with BRAF V600E- or BRAF V600K–mutated unresectable or metastatic melanoma.
Ipilimumab is a fully human immunoglobulin G1 monoclonal antibody designed to promote antitumor immune responses through blockade of the immune checkpoint molecule cytotoxic T-lymphocyte antigen-4.5 Ipilimumab has demonstrated an improvement in OS in two randomized, controlled phase III clinical trials.2,3 In one phase III study, CA184-024 (Dacarbazine and Ipilimumab vs. Dacarbazine With Placebo in Untreated Unresectable Stage III or IV Melanoma), treatment-naive patients with advanced melanoma received ipilimumab at 10 mg/kg plus dacarbazine or placebo plus dacarbazine.3 Median OS was significantly longer in patients treated with ipilimumab plus dacarbazine than in patients treated with placebo plus dacarbazine: 11.2 versus 9.1 months (hazard ratio [HR], 0.72; 95% CI, 0.59 to 0.87; P < .001).3 Survival rates at 1, 2, and 3 years were also higher in ipilimumab-treated patients than in patients treated with placebo plus dacarbazine.3 The most common treatment-related adverse events were immune-related adverse events (irAEs), primarily affecting the skin (pruritus, rash), GI system (diarrhea, colitis), and liver (increased ALT and AST). Most irAEs could be reversed by using ipilimumab management algorithms.
Follow-up of patients who received ipilimumab in phase II and phase III clinical trials suggested that a proportion of the patients achieve long-term survival. In three phase I/II trials of ipilimumab, with or without glycoprotein 100 peptides or interleukin-2, 5-year survival rates in patients with advanced melanoma were 13% to 25%.6 In four phase II trials of ipilimumab monotherapy (3 or 10 mg/kg) in advanced melanoma, 5-year survival rates were 21.4% to 49.5% for treatment-naive patients and were 16.5% to 28.4% for previously treated patients.7 Because these were nonrandomized studies, we conducted a milestone survival analysis of data from a randomized, controlled phase III trial to confirm the survival benefit of ipilimumab in patients with advanced melanoma. This is the first analysis of survival of 5 or more years in patients who received ipilimumab in a phase III trial (CA184-024). We further evaluated safety in long-term survivors that continued to receive ipilimumab as maintenance therapy.
PATIENTS AND METHODS
Eligibility Criteria
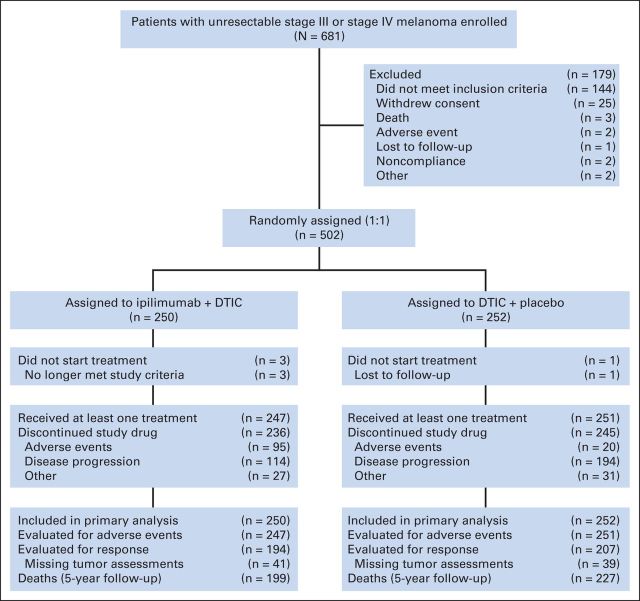
In the CA184-024 trial, eligible patients were at least 18 years of age and had previously untreated stage IIIc, N3 (unresectable), or stage IV melanoma, and an Eastern Cooperative Oncology Group performance status of 0 or 1 (Fig 1).3 Patients were eligible for enrollment regardless of baseline serum lactate dehydrogenase level or BRAF mutation status. Patients with brain metastasis (based on imaging), primary or ocular or mucosal melanoma, or symptomatic autoimmune disease were not eligible. All patients provided written informed consent to participate in the study.
Fig 1.
CONSORT diagram. DTIC, dacarbazine.
Study Design and Treatment
Patients were randomly assigned 1:1 to receive ipilimumab (10 mg/kg) plus dacarbazine (850 mg/m2; n = 250) or placebo plus dacarbazine (850 mg/m2; n = 252) administered at weeks 1, 4, 7, and 10 followed by dacarbazine alone every 3 weeks through week 22. Patients with stable disease or better from week 12 through week 24, without dose-limiting toxicities, could receive ipilimumab or placebo as maintenance therapy (every 12 weeks beginning at week 24). Dacarbazine was not given during the maintenance phase. The study design and treatment regimens for the CA184-024 trial have been described previously.3
Efficacy and Safety Assessments
We conducted a milestone survival analysis with a minimum follow-up of 5 years in all patients, current as of March 2013. As per protocol, OS was defined as the time from random assignment until death, with censoring on the last known alive date. Median estimates with 95% CIs were computed by using the Brookmeyer and Crowley method. Survival rates were estimated from Kaplan-Meier survival probabilities and the CIs were obtained by using log cumulative hazard transformation. A post hoc analysis was conducted to compare the 5-year OS rates between treatment groups.
In this trial, tumor assessments were performed at baseline and at week 12, with further assessments at weeks 16, 20, 24, 30, 36, 42, and 48 for patients without disease progression.3 Patients who remained on the study beyond week 48 and who did not progress had tumor assessments every 12 weeks. Best overall response was evaluated by using modified WHO criteria as previously described,3 in which a complete response was defined as the disappearance of all known lesions, partial response as a ≥ 50% decrease from baseline in index lesions, and stable disease as not having met the criteria for a partial response or progressive disease (defined as a 25% increase in an existing lesion or the development of a new lesion). We conducted an analysis of OS for responders versus nonresponders in both treatment arms. Specifically, a landmark analysis was conducted to exclude patients with OS of less than 6 months, which marks the maximal time to response observed in the study.
The occurrence of new irAEs during maintenance therapy was evaluated in patients still receiving ipilimumab at the last database lock in March 2013. Events are reported between the start date of maintenance phase and 70 days after the last maintenance dose. Primary safety data from the CA184-024 study has been previously reported.3
RESULTS
Patients and Treatment
Baseline demographics for patients alive at least 5 years are shown in Table 1. Most patients in the ipilimumab plus dacarbazine group were male (62.5%) and younger than age 65 years (65.0%); 45% had stage M1c disease and 20% had stage M1a disease. In the placebo plus dacarbazine group, nine (45%) of the 20 patients were male, most patients (60.0%) were younger than age 65 years, 25% had stage M1c disease, and 50% had stage M1a disease. In both groups, ≤ 7.5% of patients had unresectable stage III melanoma, and the majority had an Eastern Cooperative Oncology Group performance status of 0 and serum lactate dehydrogenase levels that were less than or equal to the upper limit of normal.
Table 1.
Baseline Demographics for Patients Alive at 5 Years and Beyond
| Characteristic | Ipilimumab + Dacarbazine (n = 40) |
Placebo + Dacarbazine (n = 20) |
||
|---|---|---|---|---|
| No. | % | No. | % | |
| Age, years | ||||
| Median | 57.5 | 61.0 | ||
| Range | 33-87 | 31-76 | ||
| < 65 | 26 | 65.0 | 12 | 60.0 |
| ≥ 65 | 14 | 35.0 | 8 | 40.0 |
| Sex | ||||
| Male | 25 | 62.5 | 9 | 45.0 |
| Female | 15 | 37.5 | 11 | 55.0 |
| M stage | ||||
| M0 | 3 | 7.5 | 1 | 5.0 |
| M1a | 8 | 20.0 | 10 | 50.0 |
| M1b | 11 | 27.5 | 4 | 20.0 |
| M1c | 18 | 45.0 | 5 | 25.0 |
| ECOG PS | ||||
| 0 | 35 | 87.5 | 19 | 95.0 |
| 1 | 5 | 12.5 | 1 | 5.0 |
| LDH | ||||
| ≤ Upper limit of normal | 34 | 85.0 | 17 | 85.0 |
| > Upper limit of normal | 6 | 15.0 | 3 | 15.0 |
Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase.
As delineated in Table 2, 247 of 250 patients in the ipilimumab plus dacarbazine group received at least one induction dose of ipilimumab and 43 (17.2%) received at least one maintenance dose. In the placebo plus dacarbazine group, 251 of 252 patients received at least one induction dose of placebo and 53 (21.0%) received at least one maintenance dose. Eleven patients in each group received maintenance therapy (ipilimumab or placebo) for more than 2 years. At a minimum follow-up of 5 years, 40 patients in the ipilimumab plus dacarbazine group and 20 patients in the placebo plus dacarbazine group were alive. Seven patients remained on ipilimumab maintenance therapy as of the last database lock in March 2013.
Table 2.
Patient Treatments and Outcomes
| Outcome | Ipilimumab + Dacarbazine | Placebo + Dacarbazine |
|---|---|---|
| Random assignment (ITT) | 250 | 252 |
| Received one or more induction doses | 247 | 251 |
| Received one or more maintenance doses | 43 | 53 |
| Still on maintenance after 2 years* | 11 | 11 |
| Patients alive at 5 years or beyond | 40 | 20 |
NOTE. Seven patients remained on ipilimumab therapy at the last database lock in March 2013.
Abbreviation: ITT, intention-to-treat.
Ipilimumab or placebo only (no dacarbazine).
Subsequent treatments received by patients who survived at least 5 years in both groups are detailed in Appendix Tables A1 and A2 (online only). Among these patients, 12 (30%) of 40 in the ipilimumab plus dacarbazine group and 11 (55%) of 20 in the placebo plus dacarbazine group received at least one subsequent treatment (Tables 1 and 2, respectively). The majority of patients received subsequent treatments as a result of disease progression, which in both groups included chemotherapy, immunotherapy, radiotherapy, and/or surgery.
Survival
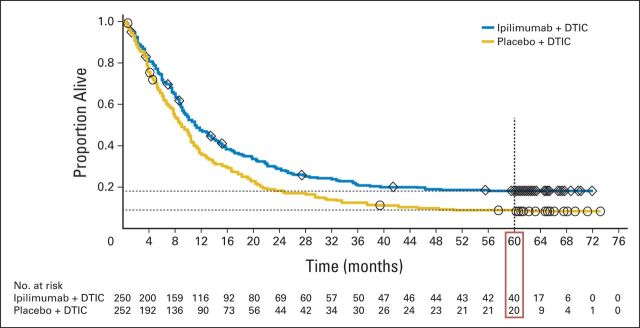
Median survival follow-up for the ipilimumab plus dacarbazine group (n = 250) was 11.0 months (range, 0.4 to 71.9 months) and was 8.9 months (range, 0.1 to 73.2 months) for the placebo plus dacarbazine group (n = 252). The updated median OS was 11.2 months (95% CI, 9.5 to 13.8 months) in the ipilimumab plus dacarbazine group and was 9.1 months (95% CI, 7.8 to 10.5 months) in the placebo plus dacarbazine group (HR, 0.69; 95% CI, 0.57 to 0.84). OS rates in the ipilimumab plus dacarbazine group were consistently higher than those in the placebo plus dacarbazine group (Table 3). At a minimum follow-up of 5 years, 18.2% of patients in the ipilimumab plus dacarbazine group were still alive compared with 8.8% of patients in the placebo plus dacarbazine group (P = .002). Kaplan-Meier plots of OS are shown in Figure 2.
Table 3.
Updated Median OS and Milestone Survival Rates
| Treatment Group | OS (months) |
OS Rate |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 Year |
2 Years |
3 Years |
4 Years |
5 Years |
||||||||
| Median | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | % | 95% CI | |
| Ipilimumab + dacarbazine (n = 250) | 11.2 | 9.5 to 13.8 | 47.6 | 41.2 to 53.7 | 28.9 | 23.3 to 34.7 | 21.3 | 16.3 to 26.6 | 19.1 | 14.4 to 24.3 | 18.2 | 13.6 to 23.4 |
| Placebo + dacarbazine (n = 252) | 9.1 | 7.8 to 10.5 | 36.4 | 30.4 to 42.4 | 17.8 | 13.3 to 22.8 | 12.1 | 8.4 to 16.5 | 9.7 | 6.4 to 13.7 | 8.8 | 5.7 to 12.8 |
Abbreviation: OS, overall survival.
Fig 2.
Kaplan-Meier estimates of overall survival in patients treated with ipilimumab plus dacarbazine (DTIC) or placebo plus DTIC in phase III CA184-024 study. Symbols indicate censored observations. Red box highlights updated 5-year survival data.
Best Overall Response
By using modified WHO criteria, best overall response was assessed in all patients who survived at least 5 years (Table 4). In the ipilimumab plus dacarbazine group (n = 40), three patients (7.5%) achieved a complete response and 17 (42.5%) achieved a partial response for an objective response rate of 50%. In the placebo plus dacarbazine group (n = 20), no patient achieved a complete response; however, a partial response was observed in seven patients for an objective response rate of 35%. Eleven patients (27.5%) and eight patients (40%) achieved stable disease in the ipilimumab plus dacarbazine and placebo plus dacarbazine groups, respectively. The proportion of patients with disease progression was similar between the two groups. In both treatment arms, responders had longer OS compared with nonresponders. At the data cutoff date, median OS had not been reached among responders versus 14.3 months (95% CI, 11.4 to 16.9 months) for nonresponders in the ipilimumab plus dacarbazine group (HR, 0.28; 95% CI, 0.16 to 0.47 for responders v nonresponders). In the placebo plus dacarbazine group, median OS for the responders and nonresponders was 20.2 months (95% CI, 14.6 to 45.3 months) and 12.3 months (95% CI, 10.9 to 15.4 months), respectively (HR, 0.51; 95% CI, 0.32 to 0.84 for responders v nonresponders).
Table 4.
Best Overall Response for Patients Alive at Least 5 Years
| Best Overall Response | Ipilimumab + Dacarbazine (n = 40) |
Placebo + Dacarbazine (n = 20) |
||
|---|---|---|---|---|
| No. | % | No. | % | |
| Complete response | 3 | 7.5 | 0 | 0 |
| Partial response | 17 | 42.5 | 7 | 35 |
| Stable disease | 11 | 27.5 | 8 | 40 |
| Progressive disease | 7 | 17.5 | 3 | 15 |
| Unknown | 2 | 5 | 2 | 10 |
| Objective response rate | 50 | 35 | ||
Safety
Table 5 summarizes the irAEs with onset during ipilimumab maintenance therapy in patients who survived at least 5 years (n = 7). The most common irAEs of any grade affected the skin (rash, vitiligo, and pruritus). Low-grade irAEs that affected the GI tract, liver (increased ALT or AST), and endocrine system were also reported. Grade 3 to 4 irAEs were observed exclusively in the skin, and both of the reported grade 3 to 4 irAEs (rash, pruritus) occurred in the same patient. There were no grade 5 irAEs.
Table 5.
irAEs With Onset During Ipilimumab Maintenance Therapy in Patients Alive at 5 Years or Beyond (n = 7)
| irAE | Grade |
|
|---|---|---|
| Any | 3 to 4 | |
| Any irAE* | 5 | 1 |
| Skin | 5 | 1 |
| Rash | 2 | 1 |
| Vitiligo | 2 | 0 |
| Pruritus | 1 | 1 |
| GI | 2 | 0 |
| Diarrhea | 2 | 0 |
| Investigations | 2 | 0 |
| ALT increased | 1 | 0 |
| AST increased | 1 | 0 |
| Endocrine | 1 | 0 |
| Hypothyroidism | 1 | 0 |
Abbreviation: irAE, immune-related adverse event.
Patients may have had more than one event.
DISCUSSION
This is the first milestone analysis of 5-year survival for patients with advanced melanoma who received ipilimumab in a randomized, controlled phase III trial, which was conducted to confirm prior reports of long-term survival in a proportion of patients from nonrandomized phase II studies.6,7 This report also represents one of the longest follow-up analyses for survival from a phase III trial in advanced melanoma. The results demonstrate a long-term survival benefit for a proportion of patients with advanced melanoma who received ipilimumab plus dacarbazine, with a 5-year survival rate which is more than double that observed for patients who received placebo plus dacarbazine. A 25% survival rate in the placebo plus dacarbazine group occurred approximately 10 months earlier than in the ipilimumab plus dacarbazine group, further demonstrating the long-term benefit of this drug. Our results are consistent with the reported survival rates in phase I/II and phase II studies of ipilimumab, in which approximately 20% of previously treated patients survived to at least 5 years.6,7 Overall, the 5-year survival rates observed in ipilimumab clinical studies are approximately double those historically expected for patients with stage IV melanoma (approximately 10%).1
The Kaplan-Meier survival curve for the ipilimumab plus dacarbazine group revealed that a plateau begins at approximately year 3, which extends to year 5 and beyond (although heavy patient censoring may limit interpretation of survival data beyond 5 years). A plateau in the survival curve has consistently been observed in individual ipilimumab studies and has recently been demonstrated in a large analysis of pooled OS data from 12 clinical studies of ipilimumab in advanced melanoma (including our phase III trial). In this analysis of data from 1,861 patients, durable long-term survival was shown with a plateau in the survival curve beginning at approximately 3 years, which extended up to 10 years in some patients.8 In this study, it is interesting to note that a plateau in the survival curve was observed in both groups, indicating that a proportion of patients have long-term survival independent of treatment. However, ipilimumab treatment results in approximately 10% more patients who achieve long-term survival. This is reflected by the survival curves, because the survival curve for the placebo plus dacarbazine group is shifted toward lower survival rates at approximately year 1 and appears to reach the plateau at a later time point than the survival curve for ipilimumab plus dacarbazine (year 4 v year 3).
As in previous ipilimumab studies,9 long-term survivors in the CA184-024 trial included those with a complete or partial response, stable disease, or disease progression as their best overall response. Higher objective response rates and more durable objective responses were observed in the ipilimumab plus dacarbazine group than in the placebo plus dacarbazine group.3 In the original analyses of data from the CA184-024 trial, median duration of response was 19.2 months in the ipilimumab plus dacarbazine group compared with 8.1 months in the placebo plus dacarbazine group (P = .03).3 Moreover, in both the CA184-024 study and the prior phase III trial of ipilimumab in previously treated advanced melanoma (MDX010-20; A Randomized, Double-blind, Multicenter Study Comparing MDX-010 Monotherapy, MDX-010 in Combination with a Melanoma Peptide Vaccine, and Melanoma Vaccine Monotherapy in HLA-A*0201 Positive Patients with Previously Treated Unresectable Stage III or Stage IV Melanoma), responses improved over time in some patients without subsequent ipilimumab treatment.2,3 The unique kinetic profile of ipilimumab is such that some patients experience a slow and steady decrease in total tumor burden following a period of durable stable disease.10,11 Collectively, the durability of responses with ipilimumab and the potential for improved responses over time may contribute to long-term survival in some patients. These response patterns may explain, in part, why a smaller proportion of long-term survivors in the ipilimumab plus dacarbazine group received subsequent therapy compared with the placebo plus dacarbazine group. However, the reasons for the latter outcome remain unclear.
Among the subset of patients eligible for maintenance therapy (ie, those who achieved stable disease or better through week 24 and who met core safety criteria), we evaluated the safety in those alive at 5 years and beyond who had continued to receive ipilimumab maintenance therapy. Despite the small number of patients (n = 7), these data provide some insight into the safety profile of ipilimumab during extended treatment (ie, beyond the four induction doses). In the primary analysis of safety data from the CA184-024 study, the most common grade 3 to 4 irAEs were shown to occur in the liver (increased AST or ALT levels).3 Although increased ALT or AST levels were reported in the long-term survivors who continued to receive ipilimumab maintenance, grade 3 to 4 irAEs occurred exclusively in the skin. This observation is consistent with previous safety reports for patients who survived more than 2 years from random assignment in the CA184-024 study, in which low rates of irAEs were observed, and grade 3 to 4 irAEs occurred only in the skin.12 In those patients who continued to receive ipilimumab maintenance therapy for more than 2 years (11 in each group), grade 3 to 4 irAEs were pruritus (n = 1; 9.1%) and rash (n = 1; 9.1%).
This article describes the first and only 5-year survival follow-up for patients who received ipilimumab in a randomized, controlled phase III trial and represents one of the longest follow-up analyses for survival among approved treatments for advanced melanoma. Because the data are derived from a randomized, controlled phase III trial, they provide one of the strongest pieces of evidence for a long-term survival benefit with ipilimumab and also lend support to the results of prior analyses in nonrandomized studies.6,7 In summary, this analysis demonstrates that a proportion of treatment-naive patients with advanced melanoma can achieve durable, long-term survival with ipilimumab therapy.
Acknowledgment
We thank the patients and investigators who participated in the CA184-024 (Dacarbazine and Ipilimumab vs. Dacarbazine With Placebo in Untreated Unresectable Stage III or IV Melanoma) trial and Haolan Lu (Bristol-Myers Squibb) for her contributions to the survival analyses reported herein. Professional medical writing assistance was provided by Ward A. Pedersen and professional editing assistance was provided by Matthew Dougherty at StemScientific, funded by Bristol-Myers Squibb.
Appendix
Table A1.
Subsequent Treatments for Patients Who Initially Received Ipilimumab Plus Dacarbazine and Who Survived 5 Years or Longer
| Patient | Days From Random Assignment | Subsequent Treatment | Reason for Treatment |
|---|---|---|---|
| 1 | 365 | Dacarbazine | Disease progression |
| 519 | Autolymphocyte therapy | Disease progression | |
| 904 | Investigational antineoplastic | Disease progression | |
| 1,142 | Fotemustine | Disease progression | |
| 2 | 85 | Dacarbazine | Maintenance therapy without progression |
| 3 | 158 | Surgery | Disease progression |
| 4 | 442 | Surgery | Disease progression |
| 922 | Dacarbazine | Disease progression | |
| 1,021 | Surgery | Disease progression | |
| 5 | 72 | Dacarbazine | Physician's choice |
| 6 | 88 | Dacarbazine | Toxicity |
| 506 | Dacarbazine | Disease progression | |
| 992 | Paclitaxel | Disease progression | |
| 1,156 | Radiotherapy (hepatic lesion) | Disease progression | |
| 1,191 | Radiotherapy (lung lesion) | Disease progression | |
| 7 | 91 | Dacarbazine | Toxicity |
| 216 | Dacarbazine | Maintenance therapy without progression | |
| 8 | 232 | Lomustine | Disease progression |
| 232 | Vincristine | Disease progression | |
| 234 | Cisplatin | Disease progression | |
| 314 | Cisplatin | Disease progression | |
| 314 | Vincristine | Disease progression | |
| 314 | Lomustine | Disease progression | |
| 9 | 569 | Surgery | Clinical deterioration without progression |
| 625 | Radiotherapy (lymph node) | Palliation without documented progression | |
| 10 | 86 | Cisplatin | Disease progression |
| 11 | 273 | Radiotherapy (clavicle) | Disease progression |
| 880 | Surgery | Local recurrence: right axilla skin subcutaneous tissue with metastatic malignant melanoma | |
| 12 | 372 | Surgery | Resection of residual pulmonary lesion |
Table A2.
Subsequent Treatments for Patients Who Initially Received Placebo Plus Dacarbazine and Who Survived 5 Years or Longer
| Patient | Days From Random Assignment | Subsequent Treatment | Reason for Treatment |
|---|---|---|---|
| 1 | 800 | Dacarbazine | Disease progression |
| 1,012 | Surgery | Complete resection (liver) | |
| 2 | 94 | Imiquimod | Topical treatment |
| 196 | Interferon-α2b | Not specified | |
| 450 | Radiotherapy | Disease progression | |
| 681 | Surgery | Disease progression | |
| 895 | Surgery | Ulceration of melanoma metastasis | |
| 3 | 197 | Surgery | Remove index lesion (to achieve a complete response) |
| 4 | 273 | Surgery | Disease progression |
| 5 | 126 | Dacarbazine | Disease progression |
| 6 | 637 | Surgery | Resection of metastasis |
| 843 | Surgery | Resection of stable lesion | |
| 7 | 584 | Temozolomide | Disease progression |
| 8 | 134 | Radiotherapy | Disease progression |
| 793 | Surgery | Excision of lesion | |
| 9 | 104 | Interferon | Disease progression |
| 10 | 746 | Surgery | Disease progression |
| 806 | Cyclophosphamide | Disease progression | |
| 809 | Cancer vaccine | Disease progression | |
| 11 | 513 | Surgery | Disease progression |
Footnotes
Supported by Bristol-Myers Squibb.
Presented at the European Cancer Congress, Amsterdam, the Netherlands, September 27-October 1, 2013.
Authors' disclosures of potential conflicts of interest are found in the article online at www.jco.org. Author contributions are found at the end of this article.
Clinical trial information: NCT00324155.
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
Disclosures provided by the authors are available with this article at www.jco.org.
AUTHOR CONTRIBUTIONS
Conception and design: Caroline Robert, Alessandro Testori, Jedd D. Wolchok
Provision of study materials or patients: Jean-Jacques Grob, Igor Bondarenko, Luc Thomas, Vanna Chiarion-Sileni, Alessandro Testori
Collection and assembly of data: Michele Maio, Jean-Jacques Grob, Steinar Aamdal, Igor Bondarenko, Luc Thomas, Claus Garbe, Vanna Chiarion-Sileni
Data analysis and interpretation: Michele Maio, Igor Bondarenko, Tai-Tsang Chen, Marina Tschaika, Jedd D. Wolchok
Manuscript writing: All authors
Final approval of manuscript: All authors
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
Five-Year Survival Rates for Treatment-Naive Patients With Advanced Melanoma Who Received Ipilimumab Plus Dacarbazine in a Phase III Trial
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to www.asco.org/rwc or jco.ascopubs.org/site/ifc.
Michele Maio
Honoraria: Bristol-Myers Squibb, GlaxoSmithKline, Roche
Consulting or Advisory Role: Bristol-Myers Squibb, GlaxoSmithKline, Roche
Research Funding: Bristol-Myers Squibb (Inst)
Travel, Accommodations, Expenses: Bristol-Myers Squibb, GlaxoSmithKline, Roche
Jean-Jacques Grob
Honoraria: Bristol-Myers Squibb
Consulting or Advisory Role: Almirall, Amgen, Bristol-Myers Squibb, Celgene, GlaxoSmithKline, Meda Pharmaceuticals, Merck, Roche
Speakers' Bureau: Bristol-Myers Squibb, GlaxoSmithKline, Roche
Travel, Accommodations, Expenses: Bristol-Myers Squibb, GlaxoSmithKline, Merck, Roche
Steinar Aamdal
No relationship to disclose
Igor Bondarenko
No relationship to disclose
Caroline Robert
Consulting or Advisory Role: Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, Roche, Amgen
Luc Thomas
No relationship to disclose
Claus Garbe
Honoraria: Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, Philogen, Roche
Consulting or Advisory Role: Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, Roche
Research Funding: Bristol-Myers Squibb (Inst), GlaxoSmithKline (Inst), Roche (Inst)
Travel, Accommodations, Expenses: Roche, Bristol-Myers Squibb
Vanna Chiarion-Sileni
Consulting or Advisory Role: Bristol-Myers Squibb, GlaxoSmithKline, Roche, MSD Oncology
Travel, Accommodations, Expenses: Bristol-Myers Squibb, Roche, MSD Oncology
Alessandro Testori
Honoraria: Amgen, Bristol-Myers Squibb, GlaxoSmithKline, IGEA, Merck, Roche
Consulting or Advisory Role: Amgen, Bristol-Myers Squibb, GlaxoSmithKline, IGEA, Merck, Roche
Travel, Accommodations, Expenses: IGEA, Oncovision
Tai-Tsang Chen
Employment: Bristol-Myers Squibb
Stock or Other Ownership: Bristol-Myers Squibb
Marina Tschaika
Employment: Bristol-Myers Squibb
Jedd D. Wolchok
Honoraria: EMD Serono, Janssen Oncology
Consulting or Advisory Role: Bristol-Myers Squibb, Merck, MedImmune, ZIOPHARM Oncology, Polynoma, Polaris Pharmaceuticals, Jounce Therapeutics, GlaxoSmithKline
Research Funding: Bristol-Myers Squibb (Inst), MedImmune (Inst), GlaxoSmithKline (Inst), Merck (Inst)
Patents, Royalties, Other Intellectual Property: I am a co-inventor on an issued patent for DNA vaccines for treatment of cancer in companion animals
Travel, Accommodations, Expenses: Bristol-Myers Squibb
REFERENCES
- 1. Garbe C Eigentler TK Keilholz U , etal: Systematic review of medical treatment in melanoma: Current status and future prospects Oncologist 16: 5– 24,2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Hodi FS O'Day SJ McDermott DF , etal: Improved survival with ipilimumab in patients with metastatic melanoma N Engl J Med 363: 711– 723,2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Robert C Thomas L Bondarenko I , etal: Ipilimumab plus dacarbazine for previously untreated metastatic melanoma N Engl J Med 364: 2517– 2526,2011. [DOI] [PubMed] [Google Scholar]
- 4. Luke JJ, Hodi FS: Ipilimumab, vemurafenib, dabrafenib, and trametinib: Synergistic competitors in the clinical management of BRAF mutant malignant melanoma Oncologist 18: 717– 725,2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hoos A Ibrahim R Korman A , etal: Development of ipilimumab: Contribution to a new paradigm for cancer immunotherapy Semin Oncol 37: 533– 546,2010. [DOI] [PubMed] [Google Scholar]
- 6. Prieto PA Yang JC Sherry RM , etal: CTLA-4 blockade with ipilimumab: Long-term follow-up of 177 patients with metastatic melanoma Clin Cancer Res 18: 2039– 2047,2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lebbé C Weber JS Maio M , etal: Long-term survival in patients with metastatic melanoma who received ipilimumab in four phase II trials J Clin Oncol 31,2013. suppl 15s abstr 9053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Schadendorf D Hodi FS Robert C , etal: Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma J Clin Oncol doi: 10.1200/JCO.2014.56.2736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wolchok JD Weber JS Maio M , etal: Four-year survival rates for patients with metastatic melanoma who received ipilimumab in phase II clinical trials Ann Oncol 24: 2174– 2180,2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wolchok JD Hoos A O'Day S , etal: Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria Clin Cancer Res 15: 7412– 7420,2009. [DOI] [PubMed] [Google Scholar]
- 11. Hoos A: Evolution of end points for cancer immunotherapy trials Ann Oncol 23: viii47– viii52,2012. [DOI] [PubMed] [Google Scholar]
- 12. Thomas L Wolchok JD Garbe C , etal: Safety of ipilimumab in patients (pts) with untreated, advanced melanoma alive beyond 2 years: Results from a phase III study J Clin Oncol 30: 543s,2012. suppl abstr 8512 [Google Scholar]