Abstract
Background
It has been reported that mutations in arginine vasopressin type 2 receptor (AVPR2) cause congenital X-linked nephrogenic diabetes insipidus (NDI). However, only a few cases of AVPR2 deletion have been documented in China.
Methods
An NDI pedigree was included in this study, including the proband and his mother. All NDI patients had polyuria, polydipsia, and growth retardation. PCR mapping, long range PCR and sanger sequencing were used to identify genetic causes of NDI.
Results
A novel 22,110 bp deletion comprising AVPR2 and ARH4GAP4 genes was identified by PCR mapping, long range PCR and sanger sequencing. The deletion happened perhaps due to the 4-bp homologous sequence (TTTT) at the junctions of both 5′ and 3′ breakpoints. The gross deletion co-segregates with NDI. After analyzing available data of putative clinical signs of AVPR2 and ARH4GAP4 deletion, we reconsider the potential role of AVPR2 deletion in short stature.
Conclusions
We identified a novel 22.1-kb deletion leading to X-linked NDI in a Chinese pedigree, which would increase the current knowledge in AVPR2 mutation.
Keywords: Nephrogenic diabetes insipidus, AVPR2, Deletion, Genetic diagnosis
Background
Nephrogenic diabetes insipidus (NDI) is a group of diseases characterized by inability to concentrate urine in response to arginine vasopressin (AVP) [1]. The main clinical manifestation of NDI is polyuria and/or polydipsia. X-linked NDI, caused by genetic defect in the arginine vasopressin V2 receptor (AVPR2), accounts for 90% of NDI cases. The remaining 10% of the NDI cases are mainly related to AQP2 gene mutations [2], which is autosomally inherited. For X-linked NDI, males with pathogenic mutations in AVPR2 are affected, while heterozygous females show various degrees of penetrance [3]. Furthermore, skewed X inactivation, which is preferential methylation of the normal allele of the AVPR2 gene, can cause NDI in female heterozygotes [4].
AVPR2 is located on Xq28 and centromeric to the adjacent ARHGAP4 gene that encodes rho GTPase activating protein 4. AVPR2 consists of three exons and encodes a 371-amino acid G protein-coupled receptor. To date, more than 277 AVPR2 putative disease-causing mutations have been reported (http://www.hgmd.cf.ac.uk/ac/index.php). Large deletions that lead to complete loss of AVPR2 and parts of the neighboring genes ARHGAP4 or L1 cell adhesion molecule (L1CAM) have also been reported [3, 5–12]. However, very few X-linked NDI cases caused by gross AVPR2 deletion have been reported in China.
In this study, we identified a novel gross deletion covering entire AVPR2 locus and approximately half of ARHGAP4 in a Chinese pedigree with NDI. In order to distinguish the putative clinical signs of an AVPR2 deletion, we reviewed all characterized AVPR2 deletions and found the potential role of AVPR2 in short stature.
Methods
Patients
The pedigree of the Chinese NDI family is shown in Fig. 1a. The proband had severe symptoms such as polyuria, polydipsia, and fatigue since infancy. He had persistent vomiting after feeding from birth to one year old. At 4 months of age he got varicella. The weight of the proband was 7 kg at 12 months. Unfortunately, NDI diagnosis was not confirmed until he was 6.5 years old, when his weight and height was 20.5 kg (about − 1 SD of WHO standards) and 1.24 m (about + 1 SD of WHO standards, Fig. 1b), respectively. The 24 h urine volume was 9 L, and specific gravity of urine was 1.003. Urine osmolality was 75 mOsmol/L and failed to rise after 24 h water deprivation (91 mOsmol/L). Diagnosis of NDI was made according to disease history and water deprivation test. His urine volume reduced 1/3 after hydrochlorothiazide treatment. However, treatment failed at the age of 13 year and his parents refused further treatment. His height growth curve was plotted in Fig. 1b. At 8–9 years, nosebleeds happened several times per month. He was very susceptible to catch a cold before the age of 10. X-ray assessment showed that his bone age was 9 years when he was 12 years old. Also, his serum uric acid level was 514 μmol/L, 25-hydroxy-vitamin D (25-OH-VD) level was 17.64 ng/ml. Serum concentration of glucose, electrolytes (Na+, K+, Ca2+, Cl−), urea nitrogen and creatinine, complete cell blood count as well as plasma osmolality were all in the reference range. Moreover, neither electrocardiogram nor brain magnetic resonance imaging showed anomaly. The two affected maternal male relatives (II:4, III:3) of the proband also had severe polyuria, polydipsia, fatigue, nocturia, failure to grow, and lower cognitive ability. All patients showed no clinical signs of immunodeficiency.
Fig. 1.
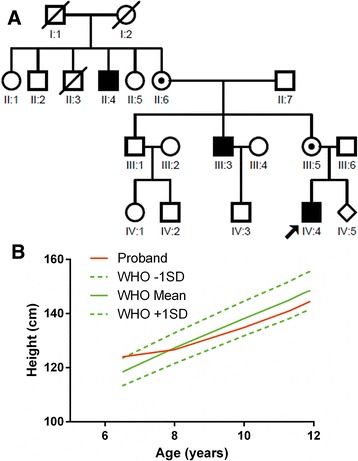
Clinical information about the Chinese NDI pedigree. a The pedigree of the Chinese family with nephrogenic diabetes insipidus. The affected subjects are indicated by black symbols, and the proband is indicated by an arrow; square and circle pedigree symbols indicate males and females respectively; b The growth curve of the proband’s height in comparison with the curves of WHO standards
DNA isolation and mutation detection
Genomic DNA was isolated from peripheral blood leucocytes using Tiangen Biotech (Beijing, China) Genomic DNA Purification Kit according to the manufacturer’s protocol. The entire coding sequence with the flanking intronic sequence of the AVPR2 gene was amplified using the polymerase chain reaction (PCR). To identify the deleted region around AVPR2 gene, primer pairs were designed at 2.5-kb intervals using Genetool software and listed in Table 1. PCR products were analyzed by electrophoresis in 1.5% agarose gel. Long range PCR was done with KOD FX Polymerase Kit (TOYOBO). Finally, a ~ 2.5 kb mutant genomic fragment was amplified using forward primer P3 and reverse primer P9 and sequenced for further analysis.
Table 1.
Primer sequence in this study
| Genes | Oligonucleotide | Sequence |
|---|---|---|
| AVPR2-E1 | Upper primer | 5′ gggggatcctgggttctgtgc 3’ |
| Lower primer | 5′ cccaggctcatgcagtccagaag 3’ | |
| AVPR2-E2A | Upper primer | 5′ ctgcatgagcctggggtgtgtatc 3’ |
| Lower primer | 5′ cgcaaagcaggcccagcagtc 3’ | |
| AVPR2-E2B | Upper primer | 5′ accgccaccgtgccatctg 3’ |
| Lower primer | 5′ ggccagcaacatgagtagcacaaag 3’ | |
| AVPR2-E3 | Upper primer | 5′ tggccaagactgtgaggatgac 3’ |
| Lower primer | 5′ cccctcctacacccagctcag 3’ | |
| P1 | Upper primer | 5′ gggcccttcctccagattcttc 3’ |
| Lower primer | 5′ gggcgaggaatccatgctaacc 3’ | |
| P2 | Upper primer | 5′ ctgccacacacccactctcac 3’ |
| Lower primer | 5′ tggcagatgaggacgtgacag 3’ | |
| P3 | Upper primer | 5′ tccccaaaccaaagatattacag 3’ |
| Lower primer | 5′ cggggtttcttcatgttgg 3’ | |
| P4 | Upper primer | 5′ cacgcataaccacatcactgaa 3’ |
| Lower primer | 5′ gggcgagatattgagagcttc 3’ | |
| P5 | Upper primer | 5′ cccaaacagcccactaacagcaact 3’ |
| Lower primer | 5′ cggggggtagaaggagggtgag 3’ | |
| P6 | Upper primer | 5′ cccgcactgtaggattccactc 3’ |
| Lower primer | 5′ ggattgcaggtgtgagccagtc 3’ | |
| P7 | Upper primer | 5′ ggcgcagaggagaaggttgac 3’ |
| Lower primer | 5′ cgcttccctgcatcttgttctc 3’ | |
| P8 | Upper primer | 5′ gcccctaggtgcgtgcttctc 3’ |
| Lower primer | 5′ ggtggggagcaggcagagc 3’ | |
| P9 | Upper primer | 5′ tggcccagtttaacattttttgata 3’ |
| Lower primer | 5′ cccggatctggactaggacatg 3’ | |
| qGAPDH | Upper primer | 5′ gcgctgagtacgtcgtggagtc 3’ |
| Lower primer | 5′ gagcctacagcagagaagcagacag 3’ | |
| qAVPR2 | Upper primer | 5′ gggccttctcgctccttct 3’ |
| Lower primer | 5′ agggcaatccaggtgacatag 3’ |
Quantitative PCR
Owing to the hemizygous state of the deletion in females, the qPCR analysis was performed using a FastStart Universal SYBR Green Master kit (Roche Applied Science, Germany) and on ABI 7900-HT system. An unrelated female control was also included to confirm successful amplification. The PCR mix was preheated at 94 °C for 5 min and then amplified in 40 cycles of 94 °C for 20 s and 60 °C for 15 s. Each PCR reaction was run in triplicate. To verify the specificity of PCR products, melting curve analysis was performed at the end of each PCR reaction. GAPDH was used as internal control. The sequences of all primers used are listed in Table 1. Data analysis was performed using the 2-△△Ct method as previously described [13].
Statistical analysis
Relative DNA level was expressed by mean. DNA level between patients was compared by Cruskal-Wallis H test. All statistical analyses were performed using IBM SPSS Statistics 22.0 software, and P < 0.05 was considered statistically significant.
Results
According to the clinical manifestation and family history of the proband, we highly suspected the boy suffer from an X-linked NDI. Thus, we first carried out PCR to amplify and examine the exons of AVPR2 gene. However, none of the exons was amplified. Similarly, the two affected males of his maternal relatives (II:4 & III:3) showed no PCR product from either exon 1 or exon 3 in AVPR2 (Fig. 2a). These data strongly suggested a fragmental deletion covering AVPR2 gene. To narrowly mapping the deletion region, we performed long-range PCR at 2.5-kb consecutive intervals to amplify the upstream and downstream of AVPR2 gene of the proband. A total of nine pairs of primers (Table 1) were used and amplified region were schemed in Fig. 2b. After PCRs using different primer combinations, a product of ~ 2.5 kb was generated using the forward primer P3 and reverse primer P9. Subsequent sequencing and alignment revealed a deletion of 22,110 bp (chrX: 153,165,873–153,187,987; according to Human Feb. 2009 < GRCh37/hg19 > Assembly on UCSC Genome Browser). There were identical TTTT sequences between the two deletion ends (Fig. 2c). This indicates that the deletion may occur via the microhomology-mediated repair mechanism, consistent with findings in similar cases [8, 14]. The 5′ breakpoint was located within the intergenic region between L1CAM and AVPR2 genes, while the 3’breakpoint was located within the first intron of ARHGAP4 (NM_001164741). The deleted region contained the entire AVPR2 gene and all exons of the ARHGAP4 gene except exon 1 (Fig. 2b).
Fig. 2.
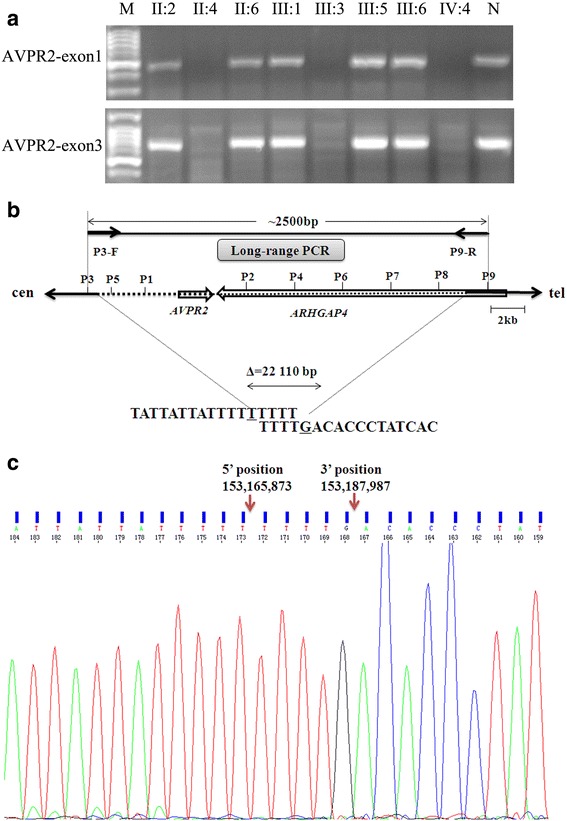
The large deletion of AVPR2 gene in the Chinese NDI pedigree. a The presentation of the deletion on agarose gel electrophoresis of the NDI family members. M, 50 bp marker; N, negative control. b Schematic presentation of the large deletion in the Chinese NDI family. Arrows indicate genes; Solid horizontal lines indicate retained regions; Broken horizontal lines indicate deleted regions; Multiple primers were designed to be spaced every ~ 2.5 kb around the AVPR2 gene. Long-range PCR with the forward primer of the P3 and the reverse primer of the P9 generates a PCR product of ~ 2.5 kb. Nucleotide sequences at the deletion end point, and the lowercase letters indicate deleted sequences. c Sequencing of deletion breakpoint in the Chinese NDI family. The possible breakpoints are indicated with arrows
Owing to the hemizygous state of the deletion in females, the qPCR analysis was performed to determine the carrier status in mother of the proband. Compared with unrelated female control, the proband’s mother showed a 50%-reduction in the copy number of AVPR2 gene, while the proband showed no amplification of AVPR2 (P = 0.027, Fig. 3).
Fig. 3.
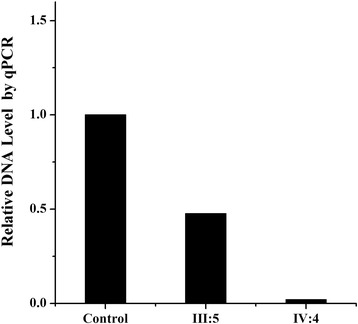
Carrier status detection of the 22-kb deletion using PCR analyses. The DNA samples were prepared from peripheral blood leucocytes. The extracted genomic DNA was spectrophotometrically quantified and diluted to 50 ng/μl. GAPDH was used as a control for normalization. Compared with unrelated female control, in III:5 DNA level of AVPR2 gene was decreased by half, while no PCR product was generated in IV:4 (P = 0.027)
Discussion
In the diagnosis of NDI, mutation screening for affected children by DNA sequencing has been accepted as a standard method. In this study, we identified a novel 22.1-kb deletion covering entire AVPR2 gene and most exons of ARHGAP4 gene in a Chinese family with NDI. All patients in this family had remarkably developmental retardation, including short stature and intellectual disability. It is particularly noteworthy that X-ray bone age was smaller than the actual age of the proband. To our knowledge, short stature is one common feature in some severe NDI cases, compared with twelve other NDI cases previously reported [3, 5–11, 15]. It is consistent with the hypothesis that AVPR2 is expressed in osteoblasts and osteoclasts and loss of AVPR2 function would affect bone remodeling [16]. The serum 25-hydroxy-vitamin D level in the proband was lower than normal. Vitamin D is a bone remodeling agent. Previous studies have shown that it plays a significant role in bone formation as well as bone resorption via direct regulation of gene expression [17, 18]. These data indicate that AVPR2 dysfunction in NDI patients may lead to the growth disturbance by both vitamin D-dependent and -independent pathways. However, its detailed mechanisms need to be further investigated.
In this study, we identified a 4 bp TTTT of identical sequence at both ends of the breakpoints using long range PCR. This indicates that deletion of AVPR2 may occur via the microhomology-mediated repair mechanism, as indicated by findings in previous studies [8, 14, 18]. The 3′ breakpoint of the 22.1 kb deletion was located with putative long range cis-regulatory elements in the ARHGAP4 introns [8]. All patients with the 22.1 kb deletion have the typical clinical features of cognitive impairment. This is consistent with the suggestion that these cis-regulatory elements within ARHGAP4 introns play a role in normal cognitive function [8]. The ARHGAP4 locus is highly conserved among species. It has been proposed that multiple long-range cis-regulatory elements in this region could function at long distances and regulate adjacent genes such as MECP2, SLC6A8 or L1CAM [19]. However, more studies will be needed to investigate how these cis-regulatory elements influence the normal cognitive function.
There are several limitations in our study. First, due to unavailability of previous clinical records, the developmental data of other patients from the NDI pedigree could not described in this study. Second, the impact of the gross deletion on cell functions of renal tubular epithelial cell was not examined. Therefore, further experimental studies on the biological significance of AVPR2 or ARHGAP4 deletion are needed.
Conclusions
In conclusion, this study identified a novel 22.1-kb deletion that is associated with polyuria, polydipsia, short stature and lower cognitive ability. Then the proband’s mother was confirmed as a carrier of the deletion by qPCR analysis. Further studies are necessary to elucidate the role of AVPR2 gene in short stature. The results provide insights into the molecular pathogenic mechanism of NDI. Genetic analysis of the AVPR2 gene for affected children and 3rd-generation in vitro fertilization can prevent the birth of affected children.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81501851) and Henan Province Medical Science and Technique Foundation (No. 201303015). None of the funding bodies participated in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Availability of data and materials
The sequence accession numbers for AVPR2 and ARHGAP4 are NG_008687.1 and NG_013220.1, respectively. The raw sequences are saved in the private human mutation database of the First Affiliated Hospital, Zhengzhou University. Data used in the current study are available from the corresponding author on reasonable request.
Abbreviations
- ARHGAP4
Rho GTPase activating protein 4
- AVP
Arginine vasopressin
- AVPR2
Arginine vasopressin V2 receptor
- GAPDH
Glyceraldehyde-3-phosphate dehydrogenase
- L1CAM
L1 cell adhesion molecule
- NDI
Nephrogenic diabetes insipidus
- WHO
World Health Organization
Author contributions
Research idea and study design: XK; experiment and data analysis: YB; manuscript drafting and revision: YC. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Zhengzhou University and written informed consents were obtained from all participants. All experimental procedures were performed according to the Helsinki Declaration.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ying Bai, Email: baiying198611@126.com.
Yibing Chen, Phone: +86-371-66913236, Email: chen.yibing@qq.com.
Xiangdong Kong, Phone: +86-371-66913236, Email: kongxd@263.net.
References
- 1.van den Ouweland AM, Dreesen JC, Verdijk M, Knoers NV, Monnens LA, Rocchi M, et al. Mutations in the vasopressin type 2 receptor gene (AVPR2) associated with nephrogenic diabetes insipidus. Nat Genet. 1992;2:99–102. doi: 10.1038/ng1092-99. [DOI] [PubMed] [Google Scholar]
- 2.Bichet DG. V2R mutations and nephrogenic diabetes insipidus. Prog Mol Biol Transl Sci. 2009;89:15–29. doi: 10.1016/S1877-1173(09)89002-9. [DOI] [PubMed] [Google Scholar]
- 3.Fujimoto M, Imai K, Hirata K, Kashiwagi R, Morinishi Y, Kitazawa K, et al. Immunological profile in a family with nephrogenic diabetes insipidus with a novel 11 kb deletion in AVPR2 and ARHGAP4 genes. BMC Med Genet. 2008;9:42. doi: 10.1186/1471-2350-9-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nomura Y, Onigata K, Nagashima T, Yutani S, Mochizuki H, Nagashima K, et al. Detection of skewed X-inactivation in two female carriers of vasopressin type 2 receptor gene mutation. J Clin Endocrinol Metab. 1997;82:3434–3437. doi: 10.1210/jcem.82.10.4312. [DOI] [PubMed] [Google Scholar]
- 5.Broides A, Ault BH, Arthus MF, Bichet DG, Conley ME. Severe combined immunodeficiency associated with nephrogenic diabetes insipidus and a deletion in the Xq28 region. Clin Immunol. 2006;120:147–155. doi: 10.1016/j.clim.2006.05.001. [DOI] [PubMed] [Google Scholar]
- 6.Demura M, Takeda Y, Yoneda T, Furukawa K, Usukura M, Itoh Y, et al. Two novel types of contiguous gene deletion of the AVPR2 and ARHGAP4 genes in unrelated Japanese kindreds with nephrogenic diabetes insipidus. Hum Mutat. 2002;19:23–29. doi: 10.1002/humu.10011. [DOI] [PubMed] [Google Scholar]
- 7.Dong Y, Sheng H, Chen X, Yin J, Su Q. Deletion of the V2 vasopressin receptor gene in two Chinese patients with nephrogenic diabetes insipidus. BMC Genet. 2006;7:53. doi: 10.1186/1471-2156-7-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang L, Poke G, Gecz J, Gibson K. A novel contiguous gene deletion of AVPR2 and ARHGAP4 genes in male dizygotic twins with nephrogenic diabetes insipidus and intellectual disability. Am J Med Genet A. 2012;158A:2511–2518. doi: 10.1002/ajmg.a.35591. [DOI] [PubMed] [Google Scholar]
- 9.Knops NB, Bos KK, Kerstjens M, van Dael K, Vos YJ. Nephrogenic diabetes insipidus in a patient with L1 syndrome: a new report of a contiguous gene deletion syndrome including L1CAM and AVPR2. Am J Med Genet A. 2008;146A:1853–1858. doi: 10.1002/ajmg.a.32386. [DOI] [PubMed] [Google Scholar]
- 10.Schoneberg T, Pasel K, von Baehr V, Schulz A, Volk HD, Gudermann T, et al. Compound deletion of the rhoGAP C1 and V2 vasopressin receptor genes in a patient with nephrogenic diabetes insipidus. Hum Mutat. 1999;14:163–174. doi: 10.1002/(SICI)1098-1004(1999)14:2<163::AID-HUMU8>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 11.Schulz A, Sangkuhl K, Lennert T, Wigger M, Price DA, Nuuja A, et al. Aminoglycoside pretreatment partially restores the function of truncated V(2) vasopressin receptors found in patients with nephrogenic diabetes insipidus. J Clin Endocrinol Metab. 2002;87:5247–5257. doi: 10.1210/jc.2002-020286. [DOI] [PubMed] [Google Scholar]
- 12.Cho SY, Law CY, Ng KL, Lam CW. Novel large deletion in AVPR2 gene causing copy number variation in a patient with X-linked nephrogenic diabetes insipidus. Clin Chim Acta. 2016;455:84–86. doi: 10.1016/j.cca.2016.01.032. [DOI] [PubMed] [Google Scholar]
- 13.Bai Y, Nie S, Jiang G, Zhou Y, Zhou M, Zhao Y, et al. Regulation of CARD8 expression by ANRIL and association of CARD8 single nucleotide polymorphism rs2043211 (p.C10X) with ischemic stroke. Stroke. 2014;45:383–388. doi: 10.1161/STROKEAHA.113.003393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang F, Seeman P, Liu P, Weterman MA, Gonzaga-Jauregui C, Towne CF, et al. Mechanisms for nonrecurrent genomic rearrangements associated with CMT1A or HNPP: rare CNVs as a cause for missing heritability. Am J Hum Genet. 2010;86:892–903. doi: 10.1016/j.ajhg.2010.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anesi L, de Gemmis P, Galla D, Hladnik U. Two new large deletions of the AVPR2 gene causing nephrogenic diabetes insipidus and a review of previously published deletions. Nephrol Dial Transplant. 2012;27:3705–3712. doi: 10.1093/ndt/gfs359. [DOI] [PubMed] [Google Scholar]
- 16.Tamma R, Sun L, Cuscito C, Lu P, Corcelli M, Li J, et al. Regulation of bone remodeling by vasopressin explains the bone loss in hyponatremia. Proc Natl Acad Sci U S A. 2013;110:18644–18649. doi: 10.1073/pnas.1318257110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fretz JA, Zella LA, Kim S, Shevde NK, Pike JW. 1,25-Dihydroxyvitamin D3 induces expression of the Wnt signaling co-regulator LRP5 via regulatory elements located significantly downstream of the gene's transcriptional start site. J Steroid Biochem Mol Biol. 2007;103:440–445. doi: 10.1016/j.jsbmb.2006.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ishida H, Bellows CG, Aubin JE, Heersche JN. Characterization of the 1,25-(OH)2D3-induced inhibition of bone nodule formation in long-term cultures of fetal rat calvaria cells. Endocrinology. 1993;132:61–66. doi: 10.1210/endo.132.1.8419147. [DOI] [PubMed] [Google Scholar]
- 19.Liu J, Francke U. Identification of cis-regulatory elements for MECP2 expression. Hum Mol Genet. 2006;15:1769–1782. doi: 10.1093/hmg/ddl099. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The sequence accession numbers for AVPR2 and ARHGAP4 are NG_008687.1 and NG_013220.1, respectively. The raw sequences are saved in the private human mutation database of the First Affiliated Hospital, Zhengzhou University. Data used in the current study are available from the corresponding author on reasonable request.


