Abstract
Extracellular vesicles (EVs), including exosomes, have a key role in the paracrine communication between organs and compartments. EVs shuttle virtually all types of biomolecules such as proteins, lipids, nucleic acids, metabolites and even pharmacological compounds. Their ability to transfer their biomolecular cargo into target cells enables EVs to play a key role in intercellular communication that can regulate cellular functions such as proliferation, apoptosis and migration. This has led to the emergence of EVs as a key player in tumor growth and metastasis through the formation of “tumor niches” in target organs. Recent data have also been shown that EVs may transform the microenvironment of primary tumors thus favoring the selection of cancer cells with a metastatic behavior. The release of EVs from resident non-malignant cells may contribute to the metastatic processes as well. However, cancer EVs may induce malignant transformation in resident mesenchymal stem cells, suggesting that the metastatic process is not exclusively due to circulating tumor cells. In this review, we outline and discuss evidence-based roles of EVs in actively regulating multiple steps of the metastatic process and how we can leverage EVs to impair metastasis.
Keywords: Extracellular vesicles, exosomes, metastasis, metastatic niche, tumor microenvironment, tumor metabolism
1. Introduction
Cancer metastasis is responsible for over 95% of cancer-related deaths [1]. It is a complex process where cancers spread from a primary tumor to different organs in the body [2,3]. Metastasis is not a cancer cell-autonomous function, rather a collective effort that requires the support of the tumor microenvironment. Rigorous scientific studies have identified that cancer cells modulate distinct non-malignant cells and components of the extracellular matrix (ECM) within the tumor microenvironment via several pathways. These pathways employ soluble factors such as growth factors, cytokines, proteins and metabolites that collectively create a microenvironment favorable for tumor growth and metastasis [4–12]. Recently, extracellular vesicles (EVs), especially, exosomes have emerged as an alternative mediator of communication within the tumor microenvironment (TME) and cancer metastasis.
EVs is the generic term that encompasses a variety of vesicles secreted by several types of cells into blood or by epithelial cells in the urinary tract into urine [196]. Exosomes are a subtype of EVs, which are derived from intraluminal endosomal vesicles. Inside their bilipid layer-enclosed bodies they contain miRNAs, mRNAs, proteins, lipids and free metabolites, which are released into the cytosol of cells that internalize EVs [13]. Therefore, these EVs play important and versatile roles in intercellular communication to maintain healthy cellular function. Cancer cell-derived EVs can induce transformation of non-malignant cells in TME to enhance tumor progression [14,15]. Importantly, cancers have been found to hijack EV-mediated communication to facilitate distinct components of the multi-step metastatic process. This review discusses the role EVs play in controlling cellular functions of cancer and stromal cells, transporting molecular cargo between cells, and facilitating bidirectional communication within the TME, with the purpose of supporting metastasis.
Unlike most signaling molecules, EVs have a certain level of specificity to target cells and in the circulatory system for longer periods than small molecules [197]. This property makes them ideal candidates for cancer cells to identify and condition/prime healthy tissue in distant organs before tumor cells arrive to form metastatic niches [16,17]. In addition to their role of long-distance messengers, EVs also have local effects within the tumor microenvironment (TME) that significantly influence metastasis by recruiting mesenchymal stem cells (MSCs). MSCs derived from bone marrow have long been thought be crucial for initiating metastatic process [18]. Only recently, EVs have found to be at the forefront of mechanisms involved in MSC-mediated metastasis [19,20]. Furthermore, the most recent discovery of EV-mediated regulation of tumor metabolism has sparked renewed interest in exploring alternative EV-mediated mechanisms of metastatic regulation. It has been posited that EVs can regulate nutrient availability in the TME as well as intracellular metabolism of cancer cells is also shifted favorably for cancer metastasis [21,22].
The versatile cargo carried by EVs is what allows them to play a multitude of roles within the TME. Studies are now focused on identifying specific mechanisms through which miRNA and proteins in EV cargo control crucial metastatic processes [23–26]. EV miRNA have been found to mediate nutrient shifts, enhance angiogenesis and propagate metastatic properties to poorly metastatic cancer cells, for supporting metastasis. EV proteins are also involved in processes such as modulating ECM, initiating epithelial-mesenchymal transformations and suppressing immune response that are critical for metastasis at the primary tumor site. Furthermore, EV proteins are directly involved in identifying suitable organs for secondary tumors and mediating mesenchymal-epithelial transformations to initiate their formation.
Given that EVs are a prominent medium of intercellular communication within the TME, there exists a bidirectional relationship between TME components and EVs. A striking example of this is the harsh conditions of hypoxia, acidity and nutrition deprivation in the TME that contribute to cancer metastasis by modulating of EVs release from cancer cells [27]. Furthermore, non-malignant cells in the TME also secrete EVs that can influence cancer metastasis by modulating proliferation, protrusion, invasion of cancer cells, transforming ECM and circumventing anti-tumor immune response [28–30].
As researchers discover the underlying mechanisms of EV-mediated metastasis, they have also identified ways to leverage EVs to hinder metastasis and treat cancers. The most promising aspect in this respect is utilizing EVs as diagnostic markers for disease progression. The high levels of circulating exosomes in cancer patients compared to healthy individuals can prove advantageous in discovering potential biomarkers [31]. Furthermore, profiling EV cargo for specific miRNAs and proteins can improve the diagnostic potential of EVs. More recently, the use of EVs for therapeutic application is being explored. The Fais group and others have leveraged special properties of EVs by incorporating drug compounds in EV cargo to create treatments with higher specificity as compared to conventional treatments [27,32,33].
2. Influence of tumor-derived EVs on processes essential to metastasis
2.1. EVs facilitate premetastatic niche (PMN) formation
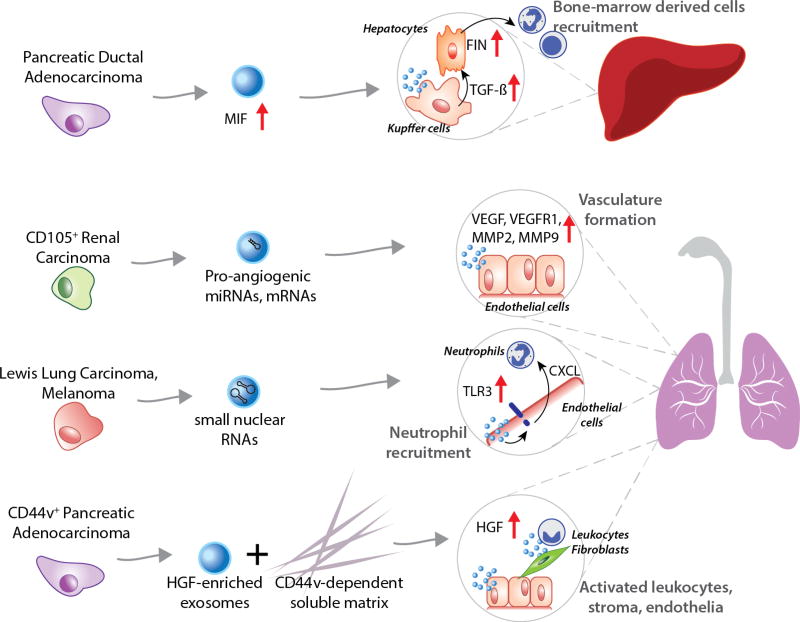
The concept of premetastatic niche formation is a derivative of Paget’s “seed and soil” theory, which tries to explain the affinity of cancers to metastasize to specific organs. A priming of the secondary site occurs before cancer cells from the primary site can colonize it, proliferate and form secondary tumors. This process is regulated by cytokines, growth factors and extracellular vesicles secreted by the tumor cells and usually involves the recruitment of bone-marrow derived cells [34]. EVs have emerged as important emissaries that trigger signaling pathways via proteins, lipids, RNAs, DNAs and provide metabolic support via free metabolites [35–38]. These properties make them ideal facilitators of premetastatic niche formation as they can condition recipient cells through the plethora of their cargo. More importantly, EVs seem to be tailor-made for organ-specific premetastatic niche (PMN) formation, given that they have organotropic properties dictated by integrin expression [16]. A recent study was able to track GFP-tagged EVs being transferred from breast cancer cells among themselves and to normal lung tissue in orthotopic nude-mouse models, thereby providing strong physical evidence of EVs acting as long-distance messengers [17]. Although, this study provided a visual confirmation of exosomal transfer to premetastatic organs, the earliest functional evidence was provided by researchers who discovered that EVs were essential to the PMN formation in lymph nodes [39]. Interestingly, they found that EVs from both poorly and highly metastatic rat pancreatic adenocarcinoma cells could induce PMN formation, but required a CD44v-dependent soluble matrix in vivo. Although, the mechanism of exosomal action on PMN formation was not discussed, the authors alluded to the hepatocyte growth factor (HGF) signaling axis being responsible (Figure 1). The higher abundance of HGF in EVs from highly-metastatic cells explained why they were more effective in forming the PMN as compared to EVs from poorly metastatic cells. The mechanism of PMN formation has only recently been described in a study [40]. The process is dependent on toll-like receptor (TLR3) interaction in normal lungs tissue. Tumor-derived RNAs, specifically small nuclear RNAs, are enriched in EVs, which activate TLR3 to induce chemokine secretion in lung tissue subsequently enhancing neutrophil recruitment (Figure 1). Another study implicated microvesicles and EVs from CD105-positive tumor-initiating renal cancer stem cells in activating angiogenic function in lung PMN [41]. The CD105-positive vesicles were found to contain proangiogenic mRNA and miRNA that enhanced VEGF, VEGFR1, MMP2, and MMP9 expression in lung endothelial cells (Figure 1). In the same vein, bioinformatics analysis of EVs from bulk and stem cells from human prostate cancer also revealed differentially expressed miRNAs, particularly miR-100-5p and miR-21-5p. These miRNAs are responsible for prostate tumorigenesis, fibroblast proliferation, differentiation and migration, angiogenesis, and are found to cooperatively contribute to local niche and PMN formation [42]. More recently, discoveries of EV-mediated PMN in lymph nodes as well as bone-marrow niches have been made. EVs from metastatic melanomas were found to condition bone marrow progenitor cells [19]. The EVs induced a pro-vasculogenic phenotype by upregulating the receptor tyrosine kinase, Met. Furthermore, the authors discovered that the molecular signature of melanoma-specific EVs isolated from stage 4 melanoma patients could serve as a prognostic marker. A similar observation was made in pancreatic cancers, where authors described the mechanism of PMN formation in the liver, mediated by EVs [43]. The process is initiated with Kupffer cells in the liver selectively internalizing EVs containing migration inhibitory factors (MIF) that triggered TGF-β secretion. TGF-β causes hepatocytic stellate cells to produce fibronectin that subsequently promotes the arrest of bone marrow cells and neutrophils (Figure 1). Remarkably, the authors observed circulating EVs with high MIF in mice with pancreatic ductal adenocarcinoma (PDAC) even before PDAC lesion formation. This incited the investigation into MIF expression in EVs isolated from plasma of PDAC patients. Remarkably, the authors found that high MIF expression could also be a clinically-relevant prognostic marker.
Figure 1. Premetastatic niche formation.
Extracellular act as emmisaries for primary tumors that metastasize to distant organs. PDAC cells secrete EVs enriched in Macrophage migration inhibitory factor (MIF) that trigger TGF-β secretion by Kupffer cells in the liver. TGF-β enhances fibronectin (FIN) expression by hepatocytes, which helps recruit bone-marrow derived cells and initiates PMN formation. CD105+ Renal carcinoma cells release EVs containing pro-angiogenic miRNAs and mRNAs that upregulate angiogenesis in healthy lung epithelial cells, thereby initiating blood vessel formation for secondary tumors. Melanoma cells secrete EVs that carry small nuclear RNAs (snRNAs) known to upregulate toll-like receptor (TLR3) expression on the surface of lung endothelial cells, leading to neutrophil recruitment. PDAC cells that are CD44v-positive secrete EVs that are enriched in hepatocyte growth factor (HGF). These EVs act as triggers for activation of leukocytes, fibroblasts and endothelial cells leading to PMN formation within the lungs. However, their action is dependent on CD44v-positive extracellular matrix.
Where most of the investigations of EV-mediated PMN formation have focused on cancer cell-derived EVs, one study also found that EVs from the non-malignant tumor microenvironment could also have an impact on metastatic niche [44]. Astrocyte-derived EVs carrying PTEN-inhibiting miRNA, targeted metastatic tumor cells to increase their chemokine secretion. This facilitated recruitment of bone-marrow derived myeloid cells that enhanced outgrowth of metastatic tumor cells. From these investigations, it is evident that the exosomal function in PMN formation can be targeted for therapeutic benefit. However, the more promising outcome is the ability to make prognostic decisions based on exosomal biomarkers during initial stages of the metastatic process. This will allow for accurate identification, and improved patients’ treatment and chance of survival before their tumor metastasize.
2.2. Tumor EVs induce a tumor-like transformation in mesenchymal stem cells
Mesenchymal stromal cells (MSCs) from the bone marrow have achieved notoriety in recent years for the role played in many steps of tumor progression, starting from the tumor promotion or inhibition ability, immunomodulation role and metastatic niche formation involvement [18]. However, whether MSCs promote or suppress the tumor progression remains controversial and still debated [18,45,46]. Evidence in the literature suggested that the cells of the bone marrow and the tumor microenvironment have a crucial role for the generation of a suitable microenvironment for the primary tumor and the development of metastasis. Cancer-derived EVs have been reported to play an active role in the disease evolution [19,47,48]. Growing evidence suggest that EVs derived from the primary tumor can act as potential mediators for priming the pre-metastatic niche. However, little is known about the ability of cancer-derived EVs to transform cells in their microenvironment. In prostate cancer, these nanosized EVs have shown to be able to reprogram adipose stem cells [49]. Furthermore, renal cancer EVs induce cancer promoting changes in their associated MSCs [20]. Even EVs isolated from breast cancer cell lines were highlighted as important signaling factors that contribute to adipose-derived stem cells desmoplastic reprogramming [50]. A study by Peinado and colleagues showed that melanoma-derived EVs can condition bone marrow progenitor cells, inducing a pro-vasculogenic phenotype by upregulating the receptor tyrosine kinase, Met [19].
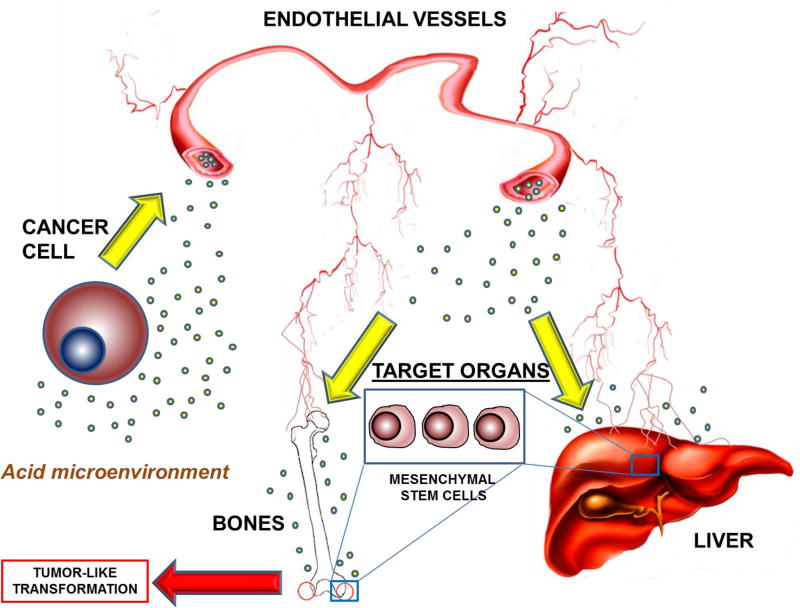
Very recently, the Fais group has provided evidence highlighting the key role played by EVs from colon cancer sources in promoting a tumor-like transformation in MSCs [51]. In particular, these cancer-derived EVs were able to induce morphological and functional changes in MSCs associated to the tumor-like phenotype, including atypical microvilli, pseudopods and vesicles release (Figure 2) [51]. The Fais group observed also aberrant functions and behaviors in MSCs, such as higher proliferation, migration and invasion rate [51], which may influence cancer progression [52,53]. These studies suggest that tumor-released EVs play a key role not only in inducing in target organs a niche where circulating tumor cells may settle and grow better; but also inducing locally a tumor-like transformation of cells that are present in target organs and have potential to differentiate depending on external stimuli (Figure 2).
Figure 2. Role of EVs released from cancer cells in tumor-like transformation of mesenchymal stem cells.
EVs released from cancer cells in acidic conditions enter the endothelial vessels, subsequently reaching target organs like liver and bones, where they act on mesenchymal stem cells to induce a tumor-like transformation.
2.3. Reprogrammed metabolism in metastasis relating to EV regulation
Metastatic transformations in cancer cells are typically coupled to alterations in their energy metabolism [54–56]. This occurs when the metabolic profile changes in cancer from a proliferative phenotype to a stem-like state. Furthermore, the nutrient availability in the TME is also shifted favorably for cancer metastasis [21,22]. EVs have been identified to play crucial roles in the regulation of metabolic change in cancer and its microenvironment. Both cancer cell-derived EVs and TME-derived EVs can enhance cancer metastasis. Cancer cell-derived EV miRNAs were transported into neighboring cells and shifted the metabolic activity in these cells from high-glycolytic to low-glycolytic profile; consequently, more glucose was made available to cancer to enhance its metastasis [57]. Furthermore, the change in nutrient availability in their local microenvironment as they detach from the primary site, invade into vasculature and colonize a foreign organ, has a major impact on their intracellular metabolism [58,59][1, 2]. It is well-known that EVs carry miRNA that can regulate metabolic pathways [60,61]. However, few examples exist that describe the direct role of EVs in regulating metabolism of cells going through metastasis. In one study, authors found that microvesicles from CD105-positive renal cancer stem cells carried miRNA that induced angiogenesis in lung tissue to help PMN formation. However, an unbiased target analysis showed that 25% of the miRNA targets were metabolic processes [41]. Although, the authors did not study metabolic changes in the lung cells, there was a strong indication that EV miRNA cargo can affect metabolism of recipient cells. Similarly, when validated target genes of miRNA found to be enriched in microvesicles from plasma of glioblastoma patients were analyzed, they revealed overrepresentation of genes related to DNA, RNA, and nitrogen metabolism.
Importantly, there now exists evidence that EVs have a two-pronged effect on the metabolism of recipient cells in the tumor microenvironment. Studies by the Nagrath group showed that cancer-associated fibroblast-derived EVs downregulated mitochondrial metabolism and enhanced glucose metabolism via their miRNA cargo [36]. Furthermore, EVs supplied free metabolites to fuel TCA cycle fluxes and support proliferative requirements of nutrient-deprived pancreatic cancer cells [62]. Since most EVs are bound by lipid membranes and are found to carry saturated fatty acids, it is possible that EVs internalization affects lipid homeostasis [13]. Although, this role of EVs has been overlooked, a similar phenomenon has been observed in cancer cells that internalize extracellular proteins to salvage amino acids to supplement increased nutritional demands [63]. The extracellular supply of lipids via EVs from stromal cells may provide free fatty acids as an alternative source of energy substrates to recipient cancer cells, thereby supporting energetic needs of metastatic transformation.
3. Role of EV cargo in metastatic processes
3.1. Cancer-derived EV microRNAs
MicroRNAs (miRNAs) present in EVs from cancer cells have been shown to enhance metastasis by conditioning the tumor microenvironment via multiple mechanisms. Exosomal miRNA are especially important to TME conditioning since the miRNA cargo can be tailor-made by cancer cells to recondition their environment [64]. Interestingly, EVs are also employed by cancer cells to selectively remove miRNAs that have tumor-suppressive properties. For example, the let-7 miRNA family, known to target oncogenes such as RAS and HMGA2, play tumor-suppressive roles in metastatic gastric cancer cell line AZ-P7a. EVs secreted by these highly metastatic cells are found to be enriched in let-7 miRNA [65,66]. However, selective enrichment of let-7 is not found in EVs from cancer cell lines with low metastatic properties [67]. Cancer cells also secrete EVs with miRNA cargo to regulate gene expression in recipient cells. A seminal study elucidated that miRNA, specifically miR-21 and miR-29a, found within EVs secreted by lung cancer lines can directly bind to Toll-like receptors (human TLR8) on immune cells. This mechanism of TLR activation initiated a protumorigenic inflammatory response that was a precursor to metastasis [195]. Another study discovered that miR-210, found in EVs secreted by cancer cells, was transferred to endothelial cells. The miR-210 was responsible for upregulation of genes involved in angiogenesis and subsequently promoted initiation of metastasis [68] (Figure 3). In another study, EVs secreted by breast cancer cells were observed to suppress nutrient utilization of stromal cells to increase nutrient availability for metastatic cells (Figure 3) [57]. These cancer-derived EVs, which were preferentially taken up by fibroblast cells, contained miR-122. miR-122 was found to target pyruvate kinase activity in fibroblasts, and subsequently suppress their glycolytic activity and glucose consumption, effectively increasing the glucose availability for cancer cells. This functional role of cancer-derived exosomal miR-122 was then verified in vivo to prove it was not an artefact of in vitro experiments [57].
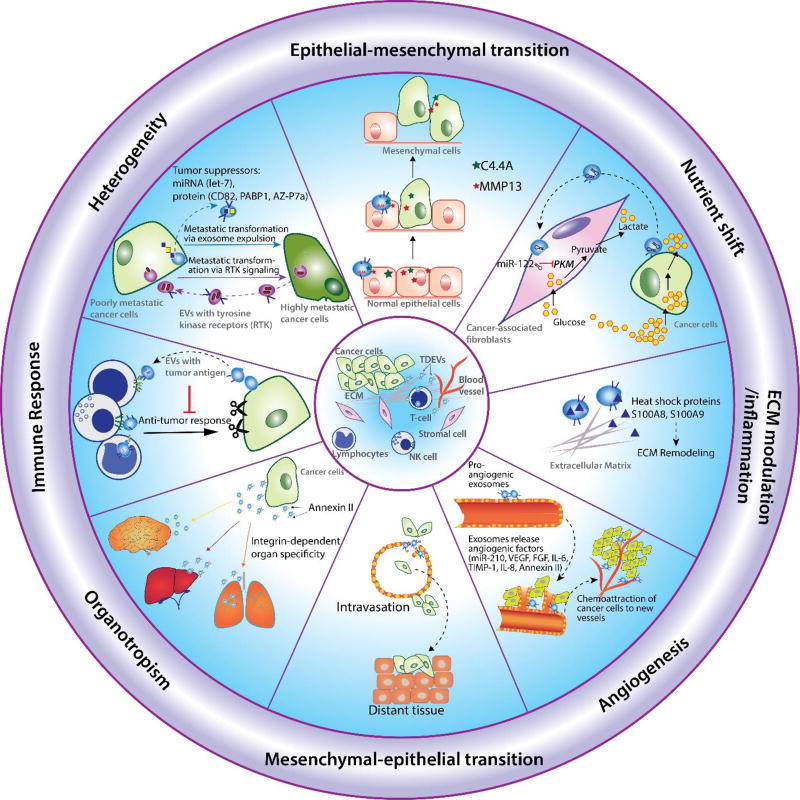
Figure 3. Cargo in cancer-derived EVs regulate metastasis.
Epithelial-mesenchymal transition (EMT): tumor derived exosomal proteins such as C4.4A, MMP13 are involved in EMT, a process known to initiate metastasis; Heterogeneity: highly metastatic melanoma cells enhance the metastatic propensity of primary tumor cells by transferring their exosomal tyrosine kinase receptor to less metastatic melanoma cells; Immune response: tumor derived EVs express tumor antigens that suppress the activation of immune cells; Organotropism: tumor derived EVs play critical roles in premetastatic niche formation of secondary tumors due to their organotropic properties; Mesenchymal-epithelial transition (MET): Tumor derived EVs carry proteins that modulate MET, a final step in formation of secondary tumors during the late stage of metastasis; Angiogenesis: tumor derived EVs induce blood vessel formation by secreting miRNAs, proteins such as VEGF, IL-6 at secondary tumor sites; Extracellular matrix (ECM) modulation: EVs secrete heat shock protein to recondition the ECM near tumor sites to support invasion and metastasis.
In addition to miRNAs, mRNAs also have been reported to be transported via EV cargo. EV mRNA from donor cells are translated into functional proteins in recipient cells. EV mRNA transport was tracked by transducing a lentivirus vector encoding a luciferase protein that is secreted by donor cells after internalization of EVs [69]. This study demonstrated that endothelial cells cocultured with microvesicles containing the Gaussia (Gluc) luciferase mRNA from glioblastoma cells, released Gluc protein into the medium increasingly over 24 hrs. Thus, verifying the translation of the Gluc mRNA within recipient cells. These results strongly indicate that mRNAs in EVs in the TME transferred to recipient cells could promote cancer metastasis.
3.2. Cancer cell-derived EV proteins
Cancer cells adjust intracellular levels of tumor-suppressing proteins by packaging them into EVs and secreting them. Metastatic duodenal cells, AZ-P7a, have been found to employ this mechanism to regulate the tumor suppressor protein, Polyadenylate-binding protein 1 (PABP1) [70]. Studies have shown that AZ-P7a cannot tolerate high intracellular PABP1 levels. They export the protein via EVs, as indicated by EVs that are more enriched in PABP1 as compared to EVs from normal AZ-521 cells. Colorectal cancer cells were also found to export, KAI1 (CD82), a suppressor of tumor metastasis, via EVs as a cell-autonomous mechanism to enhance metastasis [71–74]. However, further exploration of this mechanism is necessary to develop therapeutics that can inhibit EV-mediated secretion of tumor suppressors. Currently, no study has reported the comparison of the three mechanisms of protein regulation in cancers: EV-mediated secretion, lysosomal degradation and proteosomal degradation.
The proteins in cancer cell-derived EVs are either expressed on surface of EVs or found in their intraluminal cargo. The acidic TME has been shown to enhance lysis of extracellular EVs. EVs carrying protein cargo rich in angiogenic factors, such as VEGF, FGF, IL-6, and TIMP-1 were lysed and found the release their cargo into the TME. Upon interaction with cell surface receptors, the proteins released by EVs promoted angiogenesis and metastasis [75]. Another study has demonstrated that EVs secreted by invasive breast cancer cells contain heat-shock protein, hsp90α. Hsp90α promotes cancer cell invasiveness through the conversion of plasminogen to plasmin, leading to degradation of blood plasma proteins [76,77]. However, this process does not involve EV uptake, and therefore provides compelling evidence of the functional role of proteins expressed on EV surface. Unlike surface protein, intraluminal proteins contained within EVs need to be transported across the cell membrane for bioavailability and functional activity (Figure 3). One study observed that highly metastatic melanoma cells increased the metastatic capacity of non-metastatic tumor cells by transferring the tyrosine kinase receptor, MET, to bone marrow progenitor cells [78]. In contrast to this observation, another study found that EVs from poorly metastatic cells lacking the MET receptor, reduced the metastatic burden of highly metastatic primary tumors [19]. Furthermore, EVs from the highly metastatic melanoma cell line, B16-F10, were found to regulate genes related to extracellular matrix remodeling and inflammation (Figure 3), such as heat-shock proteins S100A8 and S100A9 [19,79]. Similarly, EVs from glioblastoma cancer cells were observed to be enriched in angiogenins [69], and pro-inflammatory cytokines, IL-6 and IL-8. Upregulation of these proteins are all associated with enhanced malignancy, vasculogenesis and ultimately, metastasis [80,81].
In addition to transporting oncoproteins and regulating ECM proteins to modulate metastasis, tumor-derived EVs also express tumor antigens [80, 82–84] (Figure 3). A study showed that EVs from ovarian cancer cells express 41kDa FasL that suppress immune response by inhibiting T-cell activation and inducing apoptosis in T-cells [85]. These observations coincide with a study showing that coculture of T-lymphocytes with FasL-expressing ovarian cancer cells leads to loss of CD3-ζ and apoptotic induction of lymphocytes [86]. Similarly, another study observed that FasL-bearing microvesicles induce apoptosis in lymphocytes [87]. Tumor-derived EVs were also found to inhibit T-cell proliferation. This inhibition was attributed to the ability of EVs to regulate the Fas/FasL pathway [88]. Furthermore, carcinoma-derived EVs have been found to play a role in the inhibitory function of perforin or other effector proteins that suppress natural (NK) cell activation. Interestingly, the increase in inhibitory function of EVs on NK cells was found to be associated directly with a decrease in metastasis-free survival period in patients [89–95].
Furthermore, cancer-derived EV proteins are also involved in modulating epithelial-mesenchymal transition (EMT) during the initiation of metastasis [96,97] and mesenchymal-epithelial transition (MET) during the final stages of metastasis [76] (Figure 3). Specifically, a study observed that the release of glycosyl-phosphatidyl-inositol–anchored molecule, C4.4A, from EVs derived from metastatic rat and human cancer cell-lines regulated the cell-adhesion phenotype of recipient cells. These recipient cells shifted from an adhesive phenotype to display motile behavior upon exposure to EVs, and this transformation subsequently contributed to metastasis [98]. Similarly, matrix metalloproteinase (MMP13) in EV cargo from Nasopharyngeal carcinoma (NPC) was also found to upregulate EMT in tumor cells to promote metastasis by increasing the levels of MMP13 in surrounding human umbilical vein endothelial cells and stromal human skin fibroblast cells [99].
Importantly, cancer cell-derived EVs have been shown to be master regulators of multiple metastatic processes [100–102] (Figure 3). For example, the EVs from “seed” melanoma cells were found to communicate with “soil” lymph nodes in three ways: a) EVs from distant melanoma cells recruited more melanoma cells; b) EVs tuned extracellular matrix near the secondary tumor sites to promote entrapment of melanoma cells within local niches; c) EVs present in the local microenvironment of secondary tumor sites increased the expression of vascular growth factors to promote the growth of trapped melanoma cells [103]. EVs from highly metastatic melanoma cells were also found to enhance the metastatic burden in the lungs of EV-treated mice [76]. In another study, annexin II found in EVs from aggressive breast cancer cells was shown to promote angiogenesis in a tPA (tissue plasminogen activator)-dependent manner to enhance breast cancer metastasis to the brain [104]. These breast cancer-secreted EVs were also shown to breach the blood-brain barrier to promote cancer metastasis.
4. Role of TME on EV-mediated metastasis
4.1. TME influence the EVs release by tumor cells
The microenvironment surrounding tumors is characterized by low nutrient supply, hypoxia and acidity [27], factors that contribute to the progression from benign to malignant phenotype and that normally are not permissive for the survival of normal cells. For those reasons, the role of the TME in determining tumor malignancy has received renewed interest in the past few decades; especially, with the suggestion that environmental conditions play a pivotal role in resistance to chemotherapy, proliferation and metastatic behavior [105–109]. Growing evidence suggests that cancer cells take up much more glucose than normal cells and mainly process it through aerobic glycolysis, the so-called “Warburg effect” [110,111]. Such an altered metabolic pattern associates with an increased production of lactate causing a drop in extracellular pH (pHe). Beside an acidic pHe, cancerous cells are characterized by a cytoplasmic pH (pHi) which generally remains neutral or even slightly more alkaline than in normal cells [112,113], leading to a pH gradient across cancer cell plasma membrane which is reversed with respect of normal cells [106,109,114–116]. More precisely, the pH of the TME has been shown to range between 6.0 and 6.8, with median values around 6.5, and the level of acidity was directly related with tumor malignancy [117–120]. Therefore, to survive in such an hostile microenvironment, tumor cells up-regulate the expression and activity of several proton extrusion mechanisms, which release protons into extracellular environment to avoid intracellular acidification, such as vacuolar H+ - ATPases (V-ATPases), Na+/H+ exchanger 1 (NHE1) and carbonic anhydrases IX (CAIX) [121,122]. Inhibition of these molecules always leads to potent anti-tumor effects [121].
Recent publications highlighted the key role played by cancer cells, TME, and EVs in the pathogenesis of cancers [36,123,124]. Tumor acidity has been pointed as a key factor in the regulation of EVs traffic within the tumor mass (Figure 2). The Fais group has provided evidence that microenvironmental acidity is involved in the regulation of some vesicle-mediated malignant tumor cell functions, such as autophagy and drug resistance [125,126]. For example, melanoma cells could survive under acidic conditions, thanks to hyper-functional proton pumps. In fact, a specific inhibition of proton release through proton pump inhibitors (PPIs) was able to induce acidification of the tumor cell cytosol [127] and acidic vesicle retention within tumor cells, with consequent increase in the antitumor activity of chemotherapeutic drugs [126]. The Fais group showed for the first time that the microenvironmental acidity not only increases the release of EVs by human melanoma cells but also induces relevant changes in the lipid composition of the released EVs [128]. Mechanisms inhibiting acidity, such as buffering the culture medium or treatment with anti-acidic molecules, were able to reduce the level of EVs release [27,128,129]. In pre-clinical in vivo settings, using xenograft models of human tumors, the levels of circulating EVs found in plasma increased related to tumor size [47]. Lastly, the levels of plasmatic EVs expressing either typical exosomal marker (e.g. CD63) or surrogate tumor marker (e.g. Cav-1) was significantly increased in tumor patients as compared to healthy individuals [47]. This suggests on one hand that the plasmatic levels of EVs may be considered as a tumor marker [130]; on the other the highest plasmatic levels of EVs may also reflect the acidity of tumor microenvironment that may be considered a phenotype of malignancy [27]. Furthermore, a recent study performed in human prostate cancer cell lines has provided clear evidence that microenvironmental acidity enhances EV release and changes their surface protein composition leading to production of PSA-positive particles [131]. This was analyzed by 3 different methods, such as NTA, nanoscale flow-cytometry and immunocapture-based ELISA, and all showed comparable results: an increased number of nanosized extracellular vesicles mostly expressing PSA. This condition was induced by low pH condition in vitro and was also observed in the plasma of prostate cancer patients [131]. There is now strong evidence now the TME influences the role of EVs in enhancing malignancy of primary tumors and in preparing secondary sites suitable for cancer cell survival [109]. Thus, the tight relationships between EVs release and the tumor microenvironment may well represent one of the most critical issues in the clinical use of EVs in both diagnostic and therapeutic fields [132].
4.2. TME-secreted EVs modulate cancer metastasis
The components within the TME and interactions among them are essential for tumor growth and progression. In addition to cancerous cells, it includes fibroblasts, immune cells, inflammatory cells, blood vessels, and the extracellular matrix. As tumor develops, malignant cells actively communicate with their microenvironment by sending out signals in the form of cytokines, signaling proteins, and extracellular vesicles. After sensing tumor-derived signals, the non-malignant cells in the microenvironment are reprogrammed to perform functions that support tumor growth. More recently, several non-malignant components of the TME have also been found to secrete soluble factors and extracellular vesicles. This contributes to the dynamic bidirectional communication between cancer and stromal cells. EVs are an essential mode of communication between components of the TME. Their versatile cargo carrying capacity, longer half-lives compared to other secreted factors, and ability to travel to distant regions in the tumors makes them ideal candidates for intercellular communication (Figure 4). CD81-positive EVs derived from fibroblasts have been shown to enhance the motility, protrusion, and invasion of breast cancer cells via the Wnt-PCP signaling pathway [45]. EVs derived from bone marrow mesenchymal stem cells (MSCs) have been shown to enhance tumor growth in vivo by enhancing VEGF and CXCR4 expression through ERK1/2 and p38 MAPK signaling pathways [28]. A recent study by Zhao et al., showed that EVs derived from cancer-associated fibroblasts (CAFs) enhanced growth of prostate cancer cells in nutrient-deprived environment by regulating metabolism in a K-ras independent manner [36]. These EVs inhibited mitochondrial metabolism, upregulated glycolysis and reductive glutamine carboxylation in PDAC cells. In a follow-up study by Achreja et al., it was shown that EVs could substantially supply free metabolite to the TCA cycle [62]. These discoveries are especially important in the context of metastasis, where EVs could regulate metabolism of metastatic or circulating tumor cells that can survive in nutrient-stressed environments.
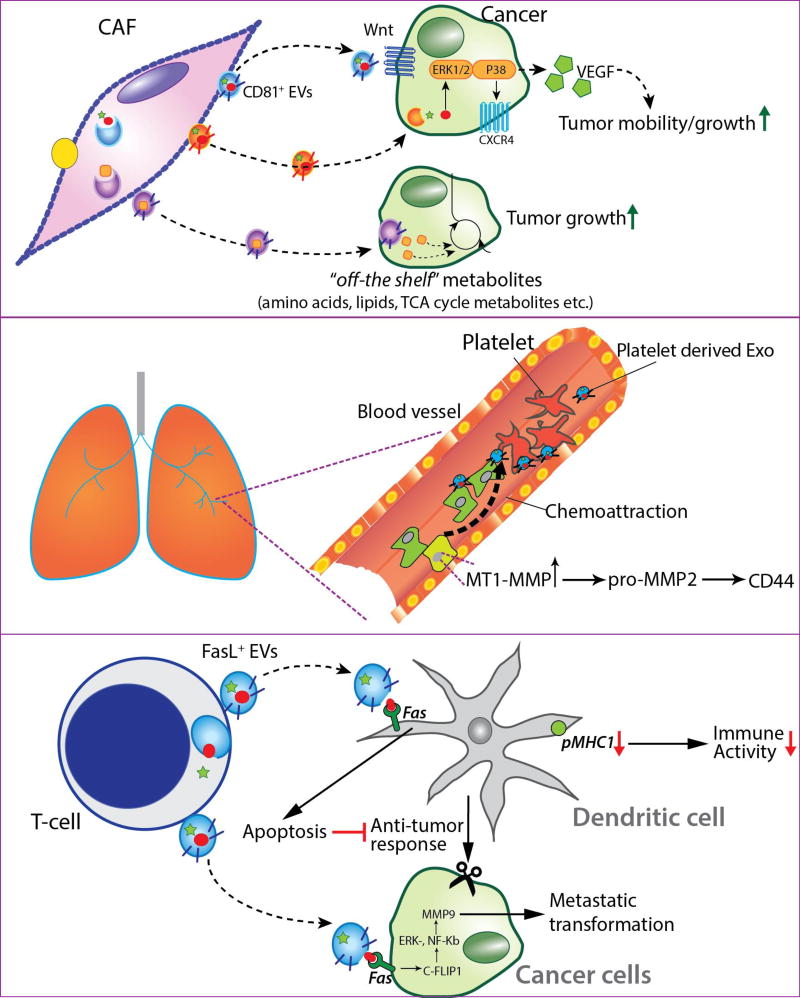
Figure 4. TME-secreted EVs modulate cancer cell metastasis.
CD81-positive EVs secreted by cancer-associated fibroblasts (CAFs) enhance motility, protrusion and invasion in breast cancer cells via the autocrine Wnt-PCP signaling pathway. EVs from mesenchymal stem cells (MSCs) enhance tumor growth in vivo and promote VEGF and CXCR4 expression in tumor cells via the ERK1/2 and p38 MAPK pathway. EVs derived from CAFs enhance viability of prostate cancer cells in nutrient-deprived condition by regulating their metabolism via miRNAs. Similarly, EVs from CAFs enhance growth in pancreatic cancer cells in nutrient-stressed microenvironment by supplying free metabolites that are incorporated into the central carbon metabolism. Platelets derived microvesicles chemoattract lung cancer cells by upregulating mRNA expression of MT1-MMP in cancer cells. MT1-MMP has is essential for reconditioning extracellular matrix components, activation of proMMP-2, cleaving a multifunctional adhesion molecule CD44, and cleaving integrin-binding adhesion receptor, subsequently enhancing cancer cell invasion. T-cells secrete bioactive EVs that induce cytotoxicity in dendritic cells (DCs) by activating the Fas/FasL pathway. These EVs also decrease pMHC1 expression on the surface of DCs, resulting in diminished antitumor immune response. FasL-positive EVs derived from CD8+ T-cell lymphocytes activate c-FLIPl, ERK, and NF-kB signaling pathways to increase the MMP9 expression in cancer cells, enhancing the invasive potential of tumors.
One study found that platelets secreted microvesicles that could chemoattract lung cancer cells by upregulating expression of MT1-MMP in the cells [133]. MT1-MMP has been shown to be essential for cancer cell invasion and acts via multiple mechanisms including catalysis of extracellular matrix components, activation of proMMP-2, cleavage of the multifunctional adhesion molecule CD44, and integrin-binding adhesion receptors (Figure 4) [134,135]. Notably, platelet-derived microvesicles (PMVs) showed the strongest chemoattraction to highly metastatic lung cancer cells. PMVs were also found to stimulate MAPK p42/44 and PI-3K-AKT signaling pathway to promote proliferation of lung cancer cells. Furthermore, mRNA for cyclin D2 was observed to be upregulated in A549 (lung cancer line) cells after internalizing PMV. Cyclin D2 is associated with enhanced invasiveness in breast cancer cells, and testicular germ tumors [136]. PMVs were also found to enhance the metastatic potential of lung cancer cells by increasing adhesion of cancer cells to endothelial cells and fibrinogen via surface expression of platelet-derived integrins like CD41. This phenomenon is also responsible for organotropic affinity of tumor cells to the bone marrow [133]. PMVs are also known to upregulate multiple genes involved in tumor vascularization, such as VEGF, IL-8, HGF, MMPs [133]. It is especially important to note that cells acquire functional proteins, which were not constitutively expressed, by internalizing extracellular microvesicles. PMVs have been demonstrated to transfer CXCR4 onto the surface of human lung cancer cell lines, which do natively express CXCR4, to bind to the SDF-1 domain in bone marrow cells [137–140].
Immune cells in the TME also play critical roles in tumor migration. FasL+ EVs derived from CD8+ T-cell lymphocytes were observed to activate c-FLIPl, ERK, and NF-kB signaling pathways to increase MMP9 expression, subsequently increasing tumor invasion potential (Figure 4) [141]. This mechanism potentially explained the phenomenon of diverting immune systems from tumor inhibition to tumor promotion. One study found that, T-cells secreted bioactive EVs were taken up by dendritic cells (DCs) and induced cytotoxicity by activating the Fas/FasL pathway, thereby weakening the overall immune activity of DCs against tumor growth and enhancing tumor metastasis. Notably, another study reported that T-cell-secreted EVs decreased pMHC I expression in DCs, resulting in a diminished antitumor immune response [142].
5. Biomarker discovery and therapeutic improvement with EVs
The level of circulating EVs in the body has been found to vary in response to diseased states and can potentially indicate progression of diseases, such as cancer. A much higher level of circulating EVs (2.85 mg/ml) was found in patients with lung adenocarcinoma compared to the control group of healthy individuals (0.77 mg/ml) [143]. Remarkably, the average concentration of EV-derived miRNA was significantly higher in the same group of patients with lung adenocarcinoma (158.6ng/ml) as compared to the control group (68.1 ng/ml) [143]. However, since the study only observed 27 patients in the diseased group and 9 in the control group it is necessary to validate this observation by performing similar studies with larger patient cohorts. These studies will be essential to justify the use of levels of circulating EVs or exosomal miRNAs as diagnostic marker for cancers.
Recent studies have indicated that profiling EV cargo could also help characterize or identify disease stages. For example, the circulating miR-122 levels, largely found enriched in cancer-derived EVs, were also associated with metastatic progression. Specifically, higher miR-122 levels were observed during later stages (Stages II-III) of breast cancer progression as compared to the earlier stages [144]. Furthermore, a related study has identified differential expression of proteins on membrane or lumen of EVs isolated from isogenic bladder cancer cells lines with increasing metastatic capacities. Particularly, vimentin, hepatoma derived growth factor, annexin 2, moesin and other EMT-related proteins were upregulated in EVs derived from cancer cells with highest metastatic capacities [145]. Similarly, ITGA3 and ITGB1 were identified by proteomics analysis to be highly expressed in EVs isolated from urine samples of patients with metastatic prostate cancer patients but not in samples of patients without metastasis [146]. Furthermore, systematic analysis of miRNA and protein profiles showed that miR-210 and fibronectin were preferentially enriched in EVs derived from breast cancer and melanoma cancer cells with the propensity to form brain metastases [147]. Recently, long non-coding RNAs (lncRNAs) have also been found to play important roles in gene regulation [148]. When the abundance of lncRNAs was measured in EVs derived from plasma of a cohort of 217 patients diagnosed with hepatocellular carcinoma (HCC), RP11-160H22.5, XLOC_014172 and LOC149086 were discovered to be enriched in cancer patients as compared to healthy subjects. Remarkably, the elevated levels of XLOC_014172 and LOC149086 were found to be indicators for HCC cell lines with higher metastatic potential [149]. These studies indicate that lncRNA in EVs could prove to be of diagnostic assistance in HCC patients. Similarly, double stranded DNA fragments observed in tumor-derived EVs were found to be a representation of the genome of the donor cancer cells. Therefore, DNA fragments were proposed to be employed as biomarkers to detect mutations in parental cells without performing tissue biopsies [36,148]. Specifically, mutations in KRAS and p53 were detected using genomic DNA from EVs isolated from serum of pancreatic cancer patients and pancreatic cancer cell lines [150].
In addition to be used as diagnostic markers, EVs can be leveraged to develop cancer therapy. In this regard, RAB27A was found to be positively correlated with EV production in a panel of 39 human-derived melanoma cell lines [19]. Similarly, the levels of nSMase2 also correlated with the rate of EV secretion. nSMase2 is, therefore, thought to affect EV secretion via ceramide signaling. However, the purview of studying these mechanisms needs to be expanded by exploring additional types of cancers to obtain actionable information for cancer therapeutics [68]. Despite these challenges, the therapeutic applications of EVs have been demonstrated in recent studies. EVs are triggering an interest in cancer therapeutics due to their property to deliver either in normal or disease states a great deal of molecules, including proteins, lipids, nucleic acids, but also chemical compounds. This shuttling ability may occur by interaction of EVs with target cells following its fusion with the target plasma membrane [128] or through ligand-receptor interaction [87,151]. The Fais group and others have reported that EVs may deliver fully active drugs, such as Cisplatin [27], Curcumin [152], Doxorubicin and Acridine Orange [153,32]. These studies clearly showed that EVs of human melanoma origin deliver a bioactive anti-tumor drug in its native form, and therefore totally functioning (i.e. Cisplatin), improving its cytotoxic activity at the disease site [27]. Moreover, very recently, it has been shown that an acidophilic dye with a strong tumoricidal action (i.e. Acridine Orange) may be delivered through the EVs [32], leading to an extended drug delivery time to cancer cells and improving the cytotoxicity of the dye [32][4], but also suggesting that EV may be the ideal delivery system for photodynamic molecules [33]. The tumor-released EVs may also participate in the tumor escape from the immune response, since they express the death ligands, FasL or TRAIL, that kill lymphocytes activating the Fas or TRAIL-mediated apoptotic pathway [87,151]. This property of EVs to deliver ligands for death receptors able to trigger death receptor-mediated apoptosis have also pointed to the possible future use of EVs in cancer immunotherapy [52,154]. Furthermore, EVs with anti-tumor antigen were reported to be more effective in activating T-cells than soluble antigens [155]. Most importantly, it was possible to obtain a large yield of EVs directly from tumor tissues without having to construct purified tumor cell lines. These nanovesicles were found to be a more effective source of tumor-specific antigen to trigger anti-tumor immune response compared antigens from whole cell lysate [83,156,157]. The tumor-derived NVs incorporated with the Toll-like receptor 3 agonist, polyI:C, showed increased efficacy in the treatment compared to NVs alone, indicating a promising strategy in cancer therapy [156]. Furthermore, it has been reported that DCs that were pre-loaded with tumor-derived EVs could be stimulated to gain anti-tumor effects. This suggested that DCs pre-treated with tumor-derived EVs can be injected into patients for improved therapeutic effect [158,159].
In summary, identification of exosomal biomarkers to non-invasively characterize cancer progression can greatly improve diagnostic accuracy and reduce the physical and economic burden on patients as compared to traditional biopsies. However, their use in cancer therapies has both advantages and limitations. EVs derived from specific cells, tissue, or body fluid are resistant to immune rejection because of they are sourced from the same patient. EVs can be tailor-made to carry a multitude of cargo including siRNA, miRNA, protein, catalase, therapeutic compounds, and intracellular metabolites by (1) chemical-based transfection; (2) incubation; (3) electroporation; (4) transfection of EV-producing cells [160,161]. In addition to the diversity of cargo content, therapeutic delivery of EVs can be improved by binding tumor-specific peptides onto their surface, thereby enhancing selectivity and reducing side-effects [160]. However, there are limitations that challenge the widespread use of EVs for anti-tumor treatment. First, the low yield of EVs from cells is a bottleneck for scaling up EV production. Second, there are no studies documenting the potential side-effects of using EVs in a clinical setting. EVs, therefore, have strong potential to be diagnostic markers, but will require significant effort until they can be used for treatment.
6. Conclusion
From the very beginning of the work in experimental oncology, it appeared reasonable that the metastatic process was due to the circulation of cancer cells through either lymphatic or blood vessels. These circulating cells would ultimately metastasize to the target organs by seeding themselves in these organs and forming secondary tumors. However, this simple concept raises many questions. The first question is: Why does metastasis occur in a selected few organs, regardless of the primary tumor site? The answer can’t be simply because the target organs are filters of the venous blood. In fact, while liver, lungs, brain, surrenal gland and bones are common metastatic organs, kidneys are never observed as the place of blood metastasis, and the kidney is a filtration organ by definition. The other question is: Why does the same primary tumor give rise to metastatic tumors that differ in nature and behavior depending on the target organs: e.g. prostate and breast cancer give liver and bone metastasis but while liver metastasis are solid neoplasms, the bone metastases are holes called “osteolytic lesions”. Furthermore, an interesting observation is that we know that a tumor cells with average diameter of 12 microns, which means that only 50 cells can form a 1 mm solid formation. However, this would suggest that a lung embolism may occur even before metastatic formation. Moreover, metastatic lesions, with an exception of few common features, are histologically different from the primary tumors. Based on these thoughts it is difficult to accept that metastatic lesions are entirely due to circulating tumor cells. Investigation of extracellular vesicles, including those of a nanosize (i.e. exosomes) is still on the tip of the visible part of the “iceberg”. However, recent evidence suggests that EVs may play a key role in the metastatic process not only in the early steps, such as the formation of a niche, but most conceivably in the entire process. This may or may not involve the participation of circulating tumor cells that form the tumor after an initial seeding into the target organs. In fact, EVs per se have transforming capacity on resident cells with a high differentiation potential, such as mesenchymal stem cells. Of course, EVs have shown to participate to the invasion of the tumor cells into the surrounding tissues as well, but the ability of EVs to transform MSC into tumor-like cells would be irrefutable evidence that proves a central role of these nanovesicles in defining the metastatic process.
This review focused on a series of groundbreaking findings, most importantly, the role of EVs in shuttling cargo of biomolecules that control the metastatic process. For example, a series of mRNAs encoding for tumor-related or expressed factors that may well have a role in the tumor-niche formation; however, tumor EVs may deliver nucleic acid of various origins, including virus-related mi- and m-RNAs [162], but have also shown to be able to transfer their content into target cells, favored by the different electrostatic charges [128]. Moreover, tumor EVs are able to transfer reporter genes to the germline in in vivo setting [163], indicating the high capacity of these nanovesicles to transfer any kind of cargo into target cells, including the tumor-related factors. There is also compelling evidence that the microenvironment plays a key role in the participation of EVs in the metastatic process. For example, extracellular acidity that has shown to induce both an increase of EV release [27,128] and changing composition of lipids and proteins in EV membranes. EVs seem to exert a prominent role in both these cancer phenotypes; in as much as they have all the potentiality to transfer malignant factors in a paracrine way through a cell-to-cell communication within the primary tumors, but also to transfer their contents in the metastatic organs with participations in multiple activities, including the setting of the pre-metastatic and metastatic niche, but also in inducing a tumor-like transformation in normal cells with high differentiating potential, such as stem cells. In supporting this, that is to date more than a hypothesis, there is the clinical evidence that cancer patients of different histology show a paramount EV cargo in their blood as compared to non-tumor conditions or healthy individuals [47,131]. This has recently suggested that the high plasmatic levels of EV in cancer patients may represent a sort of “circulating tumor mass” [130].
Despite these discoveries, there still lies a gap in our knowledge of the dynamics of EV-mediated metastatic processes: from the formation of the pre-metastatic niches, to the metastatic niches to the actual formation of the metastatic lesion. This is due to the challenges involved in monitoring the niche formation as there are no reliable biomarkers specific to this phenomenon. We also lack pre-clinical or clinical validation of the phenomenon, such as the observation of vascular leakiness or stromal tumor-like modification in metastatic sites. Furthermore, observation of molecular targets such as stromal fibronectin, LOX, S100 proteins, MIF; exosomal targets such as CD81; or cellular targets such as VEGFR1+ myeloid cells, MDSC, activated fibroblasts and EPC, has not been reliable substitute. Unfortunately, the pre-metastatic modifications are very difficult to identify by immunohistochemistry as well. This is partly due to the concept of tumor dormancy, which is a huge challenge in clinical oncology. The pre-metastatic niche formation is often a sudden phenomenon that can be missed during a long-term follow-up of tumor patients. The complexity of pre-metastatic and metastatic niches with respect to its architecture, in addition to the unknown mechanisms of interplay between secreted factors and cellular components present a serious challenge in recapitulating the dynamics of metastatic niche formation, both, in vitro and in vivo models. Consequently, making it hard to define the real role of EVs in this process.
In summary, EVs either from cancer cell or TME play multiple roles in cancer metastasis through the interactions between different cells (Table 1). However, all the involved mechanisms remain to be elaborated. As EVs contain miRNAs, proteins, free metabolites and other molecules, the study of any single component is insufficient to justify the overarching function of EVs in metastasis. If clear mechanisms of the functional components of EVs are understood, EVs can be engineered to pharmacologically modify the effects of their cargo, or modify their cargo altogether. However, methods to engineer EVs remains challenging, as new discoveries uncover novel functions of EVs. EVs are now not only known to carry metabolites as cells but also active enzymes responsible for biochemical reactions [164,165]. Furthermore, functions such as the maturation of miRNAs are also possible within EVs [166]. Further exploration of functionality of EVs could lead to notion of EVs as independent units capable of functions that typically occur in cells. As of now EVs have proven to be useful as biomarkers because of their diverse cargo and stability in body fluids. These features can be leveraged to profile cancer patients at distinct stages of the disease and to identify the possibility of metastatic occurrences. With regards to therapeutic application, EVs have been used in clinical trials in a very limited manner because of the lack of understanding of side-effects. However, there remains a prominent level of enthusiasm about the clinical use of EVs for anti-tumor therapies [132]. Most importantly, we have to carefully consider the role of EVs in favoring the development of cancer while developing non-mainstream strategies [167] aimed at controlling the release and circulation of EVs as means to impair cancer metastasis.
Table 1.
Summary of papers demonstrating exosomes/EVs role in tumour metastasis
| Cancer Type | Target | Molecules involved |
Type of study | References |
|---|---|---|---|---|
| Bladder Cancer | Urothelial cells | --------- | In vitro | [95] Franzen et al., 2015. Oncogenesis |
|
| ||||
| Breast Cancer | CAFs | Wnt11 | In vivo | [5] Hoffman et al., 2013. Breast Cancer Res |
|
| ||||
| Breast | hsp90α | In vitro | [75] McCready et al., 2010. BMC Cancer | |
|
| ||||
| Lung | --------- | In vitro and in vivo | [17] Suetsugu et al., 2013. Adv Drug Deliv Rev | |
|
| ||||
| Lung | Annexin II | In vitro and in vivo | [102] Maji et al., 2017. Mol Cancer Res | |
|
| ||||
| Lung | miRNAs | In vitro and in vivo | [66] Kosaka et al., 2013. J. Biol Chem | |
| In vitro | [6] Zhou et al., 2014. Cancer Cell | |||
|
| ||||
| Lung | cytokine | Ex-vivo | [7] Gorczynski et al., 2016 Cancer Med | |
| Liver | ||||
|
| ||||
| Lymph nodes | mRNAs | In vitro and ex-vivo | [171] Rodríguez et al., 2015. Oncotarget | |
| Lung | ||||
| Brain | ||||
|
| ||||
| Colorectal Cancer | Liver | CXCR4 | In vitro and in vivo | [172] Wang et al., 2015. Oncol Rep |
|
| ||||
| Endothelial cells | mRNA | In vitro | [173] Hong et al., 2009. BMC Genomics | |
|
| ||||
| --------- | proteins | In vitro | [174] Choi et al., 2012. J Extracell Vesicles | |
|
| ||||
| Duodenal Cancer | --------- | PABP1 | In vitro | [68] Ohshima et al., 2014. Proteomics |
|
| ||||
| Esophageal Carcinoma | esophagus | --------- | In vitro | [175] Min et al., 2017. Pathol Oncol Res |
|
| ||||
| Gastric Cancer | --------- | let-7 miRNA family | In vitro | [8][65] Ohshima et al., 2010. PLoS ONE |
|
| ||||
| Liver | EGFR | In vivo and ex-vivo | [176] Zhang et al., 2017. Nat Commun | |
|
| ||||
| Lymph node | CD97 | In vitro | [177] Liu et al., 2016. Gastric Cancer | |
|
| ||||
| Glioma | -------- | miR-21 | Ex-vivo | [178] Shi et al., 2015. Oncotarget |
|
| ||||
| Hepatocellular Carcinoma | Hepatocyte | Proteins RNAs | In vitro | [179] He et al., 2015. Carcinogenesis |
|
| ||||
| Lung | Bone | miRNAs | In vitro and in vivo | [180] Valencia et al., 2014. Mol Oncol. |
|
| ||||
| Lung | Small nuclear RNA | Ex-vivo | [181] Liu et al., 2016. Cancer Cell | |
|
| ||||
| Lymph node | periostin | In vitro | [182] Vardaki et al., 2016. Oncotarget | |
|
| ||||
| Melanoma | --------- | In vitro | [126] Parolini et al., 2009. J Biol Chem. | |
|
| ||||
| --------- | miRNAs | Ex-vivo | [183] Pfeffer et al., 2015. J Clin Med | |
|
| ||||
| Bone marrow | --------- | Ex-vivo | [19] Peinado et al., 2012. Nat Med | |
|
| ||||
| Lung | Met 72 | In vitro and ex-vivo | [184] Hao et al., 2006. Exp Oncol | |
| Met | In vitro and in vivo | [185] Adachi et al., 2016. Oncotarget | ||
|
| ||||
| Lymph nodes | miRNAs | In vitro and ex-vivo | [101] Hood et al., 2011. Cancer Res | |
| HIF-1α | Ex-vivo | [186] Hood et al., 2016. Med Hypotheses | ||
|
| ||||
| Nasopharyngeal Carcinoma | -------- | HIF1α | In vitro | [187] Aga et al., 2014. Oncogene |
|
| ||||
| Stromal cells | MMP13 | In vitro | [97] You et al., 2015. Cancer Sci | |
|
| ||||
| Non-small cell lung cancer | bone | AREG | In vitro and ex-vivo | [188] Taverna et al., 2017. Sci Rep |
|
| ||||
| Oral Squamous | Lymph node | miR-21 | In vitro and in vivo | [189] Li et al., 2016. Cancer Res |
| Cell Carcinoma | ||||
|
| ||||
| Pancreatic ductal adenocarcinoma | Liver | MIF | In vivo and in vivo | [190] Costa-Silva et al., 2015. Nat Cell Biol |
|
| ||||
| Prostate Cancer | Prostate | miRNAs | In vitro | [191] Sánchez et al., 2016. Oncotarget |
|
| ||||
| Prostate | αvβ6 | In vitro | [192] Fedele et al., 2015. J Biol Chem | |
|
| ||||
| Rat pancreatic adenocarcinoma | Lymph nodes | CD44v6 | In vitro and in vivo | [37] Jung et al., 2009. Neoplasia |
| Lung | In vitro and in vivo | [193] Wang et al., 2013. Int J Cancer | ||
|
| ||||
| Lymph nodes | mRNAs | In vitro and in vivo | [194] Rana et al., 2013. Neoplasia N.Y. | |
| miRNAs | ||||
|
| ||||
| Renal Carcinoma | Lung | CD105 | In vitro and in vivo | [39] Grange et al., 2011. Cancer Res |
Acknowledgments
This work was supported by National Institutes of Health (Grant R01CA204969) and by Filas (National Institute of Health internal classification: J9L). We thank Luciana Pinheiro for critical reading of this manuscript.
Footnotes
Conflicts of interest
The authors declare no conflicts of interest.
References
- 1.Chambers AF, Groom AC, MacDonald IC. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer. 2002;2:563–572. doi: 10.1038/nrc865. [DOI] [PubMed] [Google Scholar]
- 2.Fidler IJ. The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat. Rev. Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 3.Poste G, Fidler IJ. The pathogenesis of cancer metastasis. Nature. 1980;283:139–146. doi: 10.1038/283139a0. [DOI] [PubMed] [Google Scholar]
- 4.Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411:375–379. doi: 10.1038/35077241. [DOI] [PubMed] [Google Scholar]
- 5.Lin WW, Karin M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Invest. 2007;117:1175–1183. doi: 10.1172/JCI31537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat. Rev. Cancer. 2004;4:71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 7.Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- 8.Takebe N, Warren RQ, Ivy SP. Breast cancer growth and metastasis: interplay between cancer stem cells, embryonic signaling pathways and epithelial-to-mesenchymal transition. Breast Cancer Res. 2011;13:211. doi: 10.1186/bcr2876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Salimian Rizi B, Caneba C, Nowicka A, Nabiyar AW, Liu X, Chen K, Klopp A, Nagrath D. Nitric oxide mediates metabolic coupling of omentum-derived adipose stroma to ovarian and endometrial cancer cells. Cancer Res. 2015;75:456–471. doi: 10.1158/0008-5472.CAN-14-1337. [DOI] [PubMed] [Google Scholar]
- 10.Yang L, Achreja A, Yeung TL, Mangala LS, Jiang D, Han C, Baddour J, Marini JC, Ni J, Nakahara R, Wahlig S, Chiba L, Kim SH, Morse J, Pradeep S, Nagaraja AS, Haemmerle M, Kyunghee N, Derichsweiler M, Plackemeier T, Mercado-Uribe I, Lopez-Berestein G, Moss T, Ram PT, Liu J, Lu X, Mok SC, Sood AK, Nagrath D D. Targeting Stromal Glutamine Synthetase in Tumors Disrupts Tumor Microenvironment-Regulated Cancer Cell Growth. Cell. Metab. 2016;24:685–700. doi: 10.1016/j.cmet.2016.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Caneba CA, Yang L, Baddour J, Curtis R, Win J, Hartig S, Marini J, Nagrath D. Nitric oxide is a positive regulator of the Warburg effect in ovarian cancer cells. Cell Death Dis. 2014;5:e1302. doi: 10.1038/cddis.2014.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Salimian Rizi B, Achreja A, Nagrath D. Nitric Oxide: The Forgotten Child of Tumor Metabolism. Trends Cancer. 2017;3:659–672. doi: 10.1016/j.trecan.2017.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Record M, Carayon K, Poirot M, Silvente-Poirot S. Exosomes as new vesicular lipid transporters involved in cell–cell communication and various pathophysiologies. Biochim. Biophys. Acta. 2014;1841:108–120. doi: 10.1016/j.bbalip.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 14.Cho JA, Park H, Lim EH, Lee KW. Exosomes from breast cancer cells can convert adipose tissue-derived mesenchymal stem cells into myofibroblast-like cells. Int. J. Oncol. 2012;40:130–138. doi: 10.3892/ijo.2011.1193. [DOI] [PubMed] [Google Scholar]
- 15.Paggetti J, Haderk F, Seiffert M, Janji B, Distler U, Ammerlaan W, Kim YJ, Adam J, Lichter P, Solary E, Berchem G, Moussay E. Exosomes released by chronic lymphocytic leukemia cells induce the transition of stromal cells into cancer-associated fibroblasts. Blood. 2015;126:1106–1117. doi: 10.1182/blood-2014-12-618025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, Uryu K, Pharmer L, King T, Bojmar L, Davies AE, Ararso Y, Zhang T, Zhang H, Hernandez J, Weiss JM, Dumont-Cole VD, Kramer K, Wexler LH, Narendran A, Schwartz GK, Healey JH, Sandstrom P, Labori KJ, Kure EH, Grandgenett PM, Hollingsworth MA, de Sousa M, Kaur S, Jain M, Mallya K, Batra SK, Jarnagin WR, Brady MS, Fodstad O, Muller V, Pantel K, Minn AJ, Bissell MJ, Garcia BA, Kang Y, Rajasekhar VK, Ghajar CM, Matei I, Peinado H, Bromberg J, Lyden D. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–335. doi: 10.1038/nature15756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Suetsugu A, Honma K, Saji S, Moriwaki H, Ochiya RM T. Imaging exosome transfer from breast cancer cells to stroma at metastatic sites in orthotopic nude-mouse models. Adv. Drug. Deliv. Rev. 2013;65:383–390. doi: 10.1016/j.addr.2012.08.007. [DOI] [PubMed] [Google Scholar]
- 18.Barcellos-de-Souza P, Gori V, Bambi F, Chiarugi P. Tumor microenvironment: bone marrow-mesenchymal stem cells as key players. Biochim. Biophys. Acta. 2013;1836:321–335. doi: 10.1016/j.bbcan.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 19.Peinado H, Alečković M, Lavotshkin S, Matei I, Costa-Silva B, Moreno-Bueno G, Hergueta-Redondo M, Williams C, García-Santos G, Ghajar C, Nitadori-Hoshino A, Hoffman C, Badal K, Garcia BA, Callahan MK, Yuan J, Martins VR, Skog J, Kaplan RN, Brady MS, Wolchok JD, Chapman PB, Kang Y, Bromberg J, Lyden D. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 2012;18:883–891. doi: 10.1038/nm.2753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lindoso RS, Collino F, Camussi G. Extracellular vesicles derived from renal cancer stem cells induce a pro-tumorigenic phenotype in mesenchymal stromal cells. Oncotarget. 2015;6:7959–7969. doi: 10.18632/oncotarget.3503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nieman KM, Kenny HA, Penicka CV, Ladanyi A, Buell-Gutbrod R, Zillhardt MR, Romero IL, Carey MS, Mills GB, Hotamisligil GS, Yamada SD, Peter ME, Gwin K, Lengyel E. Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nat. Med. 2011;17:1498–1503. doi: 10.1038/nm.2492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen EI, Hewel J, Krueger JS, Tiraby C, Weber MR, Kralli A, Becker K, Yates JR, 3rd, Felding-Habermann B. Adaptation of energy metabolism in breast cancer brain metastases. Cancer Res. 2007;67:1472–1486. doi: 10.1158/0008-5472.CAN-06-3137. [DOI] [PubMed] [Google Scholar]
- 23.Singh R, Pochampally R, Watabe K, Lu Z, Mo YY. Exosome-mediated transfer of miR-10b promotes cell invasion in breast cancer. Mol. Cancer. 2014;13:256. doi: 10.1186/1476-4598-13-256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hood JL, Pan H, Lanza GM, Wickline SA. Consortium for Translational Research in Advanced Imaging and Nanomedicine (C-TRAIN), Paracrine induction of endothelium by tumor exosomes. Lab. Invest. 2009;89:1317–1328. doi: 10.1038/labinvest.2009.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Whiteside TL. Immune modulation of T-cell and NK (natural killer) cell activities by TEXs (tumour-derived exosomes) Biochem. Soc. Trans. 2013;41:245–251. doi: 10.1042/BST20120265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Taylor DD, Gercel-Taylor C. Exosomes/microvesicles: mediators of cancer-associated immunosuppressive microenvironments. Semin. Immunopathol. 2011;33:441–454. doi: 10.1007/s00281-010-0234-8. [DOI] [PubMed] [Google Scholar]
- 27.Federici C, Petrucci F, Caimi S, Cesolini A, Logozzi M, Borghi M, D'Ilio S, Lugini L, Violante N, Azzarito T, Majorani C, Brambilla D, Fais S. Exosome release and low pH belong to a framework of resistance of human melanoma cells to cisplatin. PLoS One. 2014;9:e88193. doi: 10.1371/journal.pone.0088193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhu W, Huang L, Li Y, Zhang X, Gu J, Yan Y, Xu X, Wang M, Qian H, Xu W. Exosomes derived from human bone marrow mesenchymal stem cells promote tumor growth in vivo. Cancer Lett. 2012;315:28–37. doi: 10.1016/j.canlet.2011.10.002. [DOI] [PubMed] [Google Scholar]
- 29.Lin R, Wang S, Zhao RC. Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Mol. Cell. Biochem. 2013;383:13–20. doi: 10.1007/s11010-013-1746-z. [DOI] [PubMed] [Google Scholar]
- 30.Roccaro AM, Sacco A, Maiso P, Azab AK, Tai YT, Reagan M, Azab F, Flores LM, Campigotto F, Weller E, Anderson KC, Scadden DT, Ghobrial IM. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J. Clin. Invest. 2013;123:1542–1555. doi: 10.1172/JCI66517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alvarez ML, Khosroheidari M, Kanchi Ravi R, DiStefano JK. Comparison of protein, microRNA, and mRNA yields using different methods of urinary exosome isolation for the discovery of kidney disease biomarkers. Kidney Int. 2012;82:1024–1032. doi: 10.1038/ki.2012.256. [DOI] [PubMed] [Google Scholar]
- 32.Iessi E, Logozzi M, Lugini L, Azzarito T, Federici C, Spugnini EP, Mizzoni D, Di Raimo R, Angelini DF, Battistini L, Cecchetti S, Fais S. Acridine Orange/exosomes increase the delivery and the effectiveness of Acridine Orange in human melanoma cells: A new prototype for theranostics of tumors. J. Enzyme Inhib. Med. Chem. 2017;32:648–657. doi: 10.1080/14756366.2017.1292263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kusuzaki K, Matsubara T, Murata H, Logozzi M, Iessi E, Di Raimo R, Carta F, Supuran CT, Fais S. Natural extracellular nanovesicles and photodynamic molecules: is there a future for drug delivery? J. Enzyme Inhib. Med. Chem. 2017;32:908–916. doi: 10.1080/14756366.2017.1335310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sceneay J, Smyth MJ, Möller A. The pre-metastatic niche: finding common ground. Cancer Metastasis Rev. 2013;32:449–464. doi: 10.1007/s10555-013-9420-1. [DOI] [PubMed] [Google Scholar]
- 35.Song X, Ding Y, Liu G, Yang X, Zhao R, Zhang Y, Zhao X, Anderson GJ, Nie G. Cancer Cell-derived Exosomes Induce Mitogen-activated Protein Kinase-dependent Monocyte Survival by Transport of Functional Receptor Tyrosine Kinase. J. Biol. Chem. 2016;291:8453–8464. doi: 10.1074/jbc.M116.716316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhao H, Yang L, Baddour J, Achreja A, Bernard V, Moss T, Marini JC, Tudawe T, Seviour EG, San Lucas FA, Alvarez H, Gupta S, Maiti SN, Cooper L, Peehl D, Ram PT, Maitra A, Nagrath D. Tumor microenvironment derived exosomes pleiotropically modulate cancer cell metabolism. Elife. 2016;5:e10250. doi: 10.7554/eLife.10250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Thind A, Wilson C. Exosomal miRNAs as cancer biomarkers and therapeutic targets. J. Extracell. Vesicles. 2016;5:31292. doi: 10.3402/jev.v5.31292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J, Williams C, Rodriguez-Barrueco R, Silva JM, Zhang W, Hearn S, Elemento O, Paknejad N, Manova-Todorova K, Welte K, Bromberg J, Peinado H, Lyden D. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell. Res. 2014;24:766–769. doi: 10.1038/cr.2014.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jung T, Castellana D, Klingbeil P, Cuesta Hernández I, Vitacolonna M, Orlicky DJ, Roffler SR, Brodt P, Zöller M M. CD44v6 Dependence of Premetastatic Niche Preparation by Exosomes. Neoplasia. 2009;11:1093–1105. doi: 10.1593/neo.09822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu Y, Gu Y, Han Y, Zhang Q, Jiang Z, Zhang X, Huang B, Xu X, Zheng J, Cao X. Tumor Exosomal RNAs Promote Lung Pre-metastatic Niche Formation by Activating Alveolar Epithelial TLR3 to Recruit Neutrophils. Cancer Cell. 2016;30:243–256. doi: 10.1016/j.ccell.2016.06.021. [DOI] [PubMed] [Google Scholar]
- 41.Grange C, Tapparo M, Collino F, Vitillo L, Damasco C, Deregibus MC, Tetta C, Bussolati B, Camussi G. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 2011;71:5346–5356. doi: 10.1158/0008-5472.CAN-11-0241. [DOI] [PubMed] [Google Scholar]
- 42.Sánchez CA, Andahur EI, Valenzuela R, Castellón EA, Fullá JA, Ramos CG, Triviño JC. Exosomes from bulk and stem cells from human prostate cancer have a differential microRNA content that contributes cooperatively over local and pre-metastatic niche. Oncotarget. 2016;26:3993–4008. doi: 10.18632/oncotarget.6540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, Xiang J, Zhang T, Theilen TM, García-Santos G, Williams C, Ararso Y, Huang Y, Rodrigues G, Shen TL, Labori KJ, Lothe IM, Kure EH, Hernandez J, Doussot A, Ebbesen SH, Grandgenett PM, Hollingsworth MA, Jain M, Mallya K, Batra SK, Jarnagin WR, Schwartz RE, Matei I, Peinado H, Stanger BZ, Bromberg J, Lyden D. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nature Cell. Biol. 2015;17:816–826. doi: 10.1038/ncb3169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang L, Zhang S, Yao J, Lowery FJ, Zhang Q, Huang WC, Li P, Li M, Wang X, Zhang C, Wang H, Ellis K, Cheerathodi M, McCarty JH, Palmieri D, Saunus J, Lakhani S, Huang S, Sahin AA, Aldape KD, Steeg PS, Yu D. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature. 2015;527:100–104. doi: 10.1038/nature15376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Luga V, Zhang L, Viloria-Petit AM, Ogunjimi AA, Inanlou MR, Chiu E, Buchanan M, Hosein AN, M Basik JL. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell. 2012;151:1542–1556. doi: 10.1016/j.cell.2012.11.024. [DOI] [PubMed] [Google Scholar]
- 46.Klopp AH, Gupta A, Spaeth E, Andreeff M, Marini F., 3rd Concise review: Dissecting a discrepancy in the literature: do mesenchymal stem cells support or suppress tumor growth? Stem. Cells. 2011;29:11–19. doi: 10.1002/stem.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Logozzi M, De Milito A, Lugini L, Borghi M, Calabrò L, Spada M, Perdicchio M, Marino ML, Federici C, Iessi E, Brambilla D, Venturi G, Lozupone F, Santinami M, Huber V, Maio M, Rivoltini L, Fais S. High levels of exosomes expressing CD63 and caveolin-1 in plasma of melanoma patients. PLoS One. 2009;4:e5219. doi: 10.1371/journal.pone.0005219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang HG, Grizzle WE. Exosomes: a novel pathway of local and distant intercellular communication that facilitates the growth and metastasis of neoplastic lesions. Am. J. Pathol. 2014;184:28–41. doi: 10.1016/j.ajpath.2013.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Abd Elmageed ZY, Yang Y, Thomas R, Ranjan M, Mondal D, Moroz K, Fang Z, Rezk BM, Moparty K, Sikka SC, Sartor O, Abdel-Mageed AB. Neoplastic reprogramming of patient-derived adipose stem cells by prostate cancer cell-associated exosomes. Stem. Cells. 2014;32:983–997. doi: 10.1002/stem.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Song YH, Warncke C, Choi SJ, Choi S, Chiou AE, Ling L, Liu HY, Daniel S, Antonyak MA, Cerione RA, Fischbach C. Breast cancer-derived extracellular vesicles stimulate myofibroblast differentiation and pro-angiogenic behavior of adipose stem cells. Matrix Biol. 2017;60-61:190–205. doi: 10.1016/j.matbio.2016.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lugini L, Valtieri M, Federici C, Cecchetti S, Meschini S, Condello M, Signore M, Fais S. Exosomes from human colorectal cancer induce a tumor-like behavior in colonic mesenchymal stromal cells. Oncotarget. 2016;7:50086–50098. doi: 10.18632/oncotarget.10574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lugini L, Cecchetti S, Huber V, Luciani F, Macchia G, Spadaro F, Paris L, Abalsamo L, Colone M, Molinari A, Podo F, Rivoltini L, Ramoni C, Fais S. Immune surveillance properties of human NK cell-derived exosomes. J. Immunol. 2012;189:2833–2842. doi: 10.4049/jimmunol.1101988. [DOI] [PubMed] [Google Scholar]
- 53.Scadden DT. Nice neighborhood: emerging concepts of the stem cell niche. Cell. 2014;157:41–50. doi: 10.1016/j.cell.2014.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yang L, Moss T, Mangala LS, Marini J, Zhao H, Wahlig S, Armaiz-Pena G, Jiang D, Achreja A, Win J, Roopaimoole R, Rodriguez-Aguayo C, Mercado-Uribe I, Lopez-Berestein G, Liu J, Tsukamoto T, Sood AK, Ram PT, Nagrath D. Metabolic shifts toward glutamine regulate tumor growth, invasion and bioenergetics in ovarian cancer. Mol. Syst. Biol. 2014;10:728. doi: 10.1002/msb.20134892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Caneba CA, Bellance N, Yang L, Pabst L, Nagrath D. Pyruvate uptake is increased in highly invasive ovarian cancer cells under anoikis conditions for anaplerosis, mitochondrial function, and migration. Am. J. Physiol. Endocrinol. Metab. 2012;303:E1036–1052. doi: 10.1152/ajpendo.00151.2012. [DOI] [PubMed] [Google Scholar]
- 56.Yang L, Venneti S, Nagrath D. Glutaminolysis: A Hallmark of Cancer Metabolism. Annu. Rev. Biomed. Eng. 2017;19:163–194. doi: 10.1146/annurev-bioeng-071516-044546. [DOI] [PubMed] [Google Scholar]
- 57.Fong MY, Zhou W, Liu L, Alontaga AY, Chandra M, Ashby J, Chow A, O'Connor ST, Li S, Chin AR, Somlo G, Palomares M, Li Z, Tremblay JR, Tsuyada A, Sun G, Reid MA, Wu X, Swiderski P, Ren X, Shi Y, Kong M, Zhong W, Chen Y, Wang SE. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat. Cell. Biol. 2015;17:183–194. doi: 10.1038/ncb3094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Payen VL, Porporato PE, Baselet B, Sonveaux P. Metabolic changes associated with tumor metastasis, part 1: tumor pH, glycolysis and the pentose phosphate pathway. Cell. Mol. Life Sci. 2016;73:1333–1348. doi: 10.1007/s00018-015-2098-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dong C, Yuan T, Wu Y, Wang Y, Fan TW, Miriyala S, Lin Y, Yao J, Shi J, Kang T, Lorkiewicz P, St Clair D, Hung MC, Evers BM, Zhou BP. Loss of FBP1 by Snail-Mediated Repression Provides Metabolic Advantages in Basal-like Breast Cancer. Cancer Cell. 2013;23:316–331. doi: 10.1016/j.ccr.2013.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhu H, Shyh-Chang N, Segrè AV, Shinoda G, Shah SP, Einhorn WS, Takeuchi A, Engreitz JM, Hagan JP, Kharas MG, Urbach A, Thornton JE, Triboulet R, Gregory RI, DIAGRAM Consortium; MAGIC Investigators. Altshuler D, Daley GQ. The Lin28/let-7 Axis Regulates Glucose Metabolism. Cell. 2011;147:81–94. doi: 10.1016/j.cell.2011.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chau BN, Xin C, Hartner J, Ren S, Castano AP, Linn G, Li J, Tran PT, Kaimal V, Huang X, Chang AN, Li S, Kalra A, Grafals M, Portilla D, MacKenna DA, Orkin SH, Duffield JS. MicroRNA-21 Promotes Fibrosis of the Kidney by Silencing Metabolic Pathways. Sci. Transl. Med. 2012;4:121ra18. doi: 10.1126/scitranslmed.3003205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Achreja A, Zhao H, Yang L, Yun TH, Marini J, Nagrath D. Exo-MFA - A 13C metabolic flux analysis framework to dissect tumor microenvironment-secreted exosome contributions towards cancer cell metabolism. Metab. Eng. 2017 doi: 10.1016/j.ymben.2017.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Commisso C, Davidson SM, Soydaner-Azeloglu RG, Parker SJ, Kamphorst JJ, Hackett S, Grabocka E, Nofal M, Drebin JA, Thompson CB, Rabinowitz JD, Metallo CM, Vander Heiden MG, Bar-Sagi D. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature. 2013;497:633–637. doi: 10.1038/nature12138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Guduric-Fuchs J, O'Connor A, Camp B, O'Neill CL, Medina RJ, Simpson DA. Selective extracellular vesicle-mediated export of an overlapping set of microRNAs from multiple cell types. BMC Genomics. 2012;13:357. doi: 10.1186/1471-2164-13-357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lee YS, Dutta A. The tumor suppressor microRNA let-7 represses the HMGA2 oncogene. Genes Dev. 2007;21:1025–1030. doi: 10.1101/gad.1540407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A, Labourier E, Reinert KL, Brown D, Slack FJ. RAS is regulated by the let-7 microRNA family. Cell. 2005;120:635–647. doi: 10.1016/j.cell.2005.01.014. [DOI] [PubMed] [Google Scholar]
- 67.Ohshima K, Inoue K, Fujiwara A, Hatakeyama K, Kanto K, Watanabe Y, Muramatsu K, Fukuda Y, Ogura S, Yamaguchi K, Mochizuki T. Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PLoS One. 2010;5:e13247. doi: 10.1371/journal.pone.0013247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kosaka N, Iguchi H, Hagiwara K, Yoshioka Y, Takeshita F, Ochiya T. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J. Biol. Chem. 2013;288:10849–10859. doi: 10.1074/jbc.M112.446831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry WT, Jr, Carter BS, Krichevsky AM, Breakefield XO. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell. Biol. 2008;10:1470–1476. doi: 10.1038/ncb1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ohshima K, Kanto K, Hatakeyama K, Ide T, Wakabayashi-Nakao K, Watanabe Y, Sakura N, Terashima M, Yamaguchi K, Mochizuki T. Exosome-mediated extracellular release of polyadenylate-binding protein 1 in human metastatic duodenal cancer cells. Proteomics. 2014;14:2297–2306. doi: 10.1002/pmic.201300477. [DOI] [PubMed] [Google Scholar]
- 71.Lise M, Belluco C, Perera SP, Patel R, Thomas P, Ganguly A. Clinical correlations of alpha2,6-sialyltransferase expression in colorectal cancer patients. Hybridoma. 2000;19:281–286. doi: 10.1089/027245700429828. [DOI] [PubMed] [Google Scholar]
- 72.Sawada M, Moriya S, Saito S, Shineha R, Satomi S, Yamori T, Tsuruo T, Kannagi R, Miyagi T. Reduced sialidase expression in highly metastatic variants of mouse colon adenocarcinoma 26 and retardation of their metastatic ability by sialidase overexpression. Int. J. Cancer. 2002;97:180–185. doi: 10.1002/ijc.1598. [DOI] [PubMed] [Google Scholar]
- 73.Seales EC, Jurado GA, Brunson BA, Wakefield JK, Frost AR, Bellis SL. Hypersialylation of beta1 integrins, observed in colon adenocarcinoma, may contribute to cancer progression by up-regulating cell motility. Cancer Res. 2005;65:4645–4652. doi: 10.1158/0008-5472.CAN-04-3117. [DOI] [PubMed] [Google Scholar]
- 74.Jung YR, Park JJ, Jin YB, Cao YJ, Park MJ, Kim EJ, Lee M. Silencing of ST6Gal I enhances colorectal cancer metastasis by down-regulating KAI1 via exosome-mediated exportation and thereby rescues integrin signaling. Carcinogenesis. 2016:pii. doi: 10.1093/carcin/bgw091. bgw091. [DOI] [PubMed] [Google Scholar]
- 75.Taraboletti G, D'Ascenzo S, Giusti I, Marchetti D, Borsotti P, Millimaggi D, Giavazzi R, Pavan A, Dolo V. Bioavailability of VEGF in tumor-shed vesicles depends on vesicle burst induced by acidic pH. Neoplasia. 2006;8:96–103. doi: 10.1593/neo.05583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Vassalli JD, Sappino AP, Belin D. The plasminogen activator/plasmin system. J. Clin. Invest. 1991;88:1067–1072. doi: 10.1172/JCI115405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.McCready J, Sims JD, Chan D, Jay DG. Secretion of extracellular hsp90alpha via exosomes increases cancer cell motility: a role for plasminogen activation. BMC Cancer. 2010;10:294. doi: 10.1186/1471-2407-10-294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gentile A, Trusolino L, Comoglio PM. The Met tyrosine kinase receptor in development and cancer. Cancer. Metastasis Rev. 2008;27:85–94. doi: 10.1007/s10555-007-9107-6. [DOI] [PubMed] [Google Scholar]
- 79.Hermani A, De Servi B, Medunjanin S, Tessier PA, Mayer D. S100A8 and S100A9 activate MAP kinase and NF-kappaB signaling pathways and trigger translocation of RAGE in human prostate cancer cells. Exp. Cell. Res. 2006;312:184–197. doi: 10.1016/j.yexcr.2005.10.013. [DOI] [PubMed] [Google Scholar]
- 80.Brat DJ, Bellail AC, Van Meir EG. The role of interleukin-8 and its receptors in gliomagenesis and tumoral angiogenesis. Neuro. Oncol. 2005;7:122–133. doi: 10.1215/S1152851704001061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Eberle K, Oberpichler A, Trantakis C, Krupp W, Knüpfer M, Tschesche H, Seifert V. The expression of angiogenin in tissue samples of different brain tumours and cultured glioma cells. Anticancer Res. 2000;20:1679–1684. [PubMed] [Google Scholar]
- 82.Andre F, Schartz NE, Chaput N, Flament C, Raposo G, Amigorena S, Angevin E, Zitvogel L. Tumor-derived exosomes: a new source of tumor rejection antigens. Vaccine. 2002;20:A28–31. doi: 10.1016/s0264-410x(02)00384-5. [DOI] [PubMed] [Google Scholar]
- 83.Wolfers J, Lozier A, Raposo G, Regnault A, Théry C, Masurier C, Flament C, Pouzieux S, Faure F, Tursz T, Angevin E, Amigorena S, Zitvogel L. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat. Med. 2001;7:297–303. doi: 10.1038/85438. [DOI] [PubMed] [Google Scholar]
- 84.Poutsiaka DD, Schroder EW, Taylor DD, Levy EM, Black PH. Membrane vesicles shed by murine melanoma cells selectively inhibit the expression of Ia antigen by macrophages. J. Immunol. 1985;134:138–144. [PubMed] [Google Scholar]
- 85.Taylor DD, Gercel-Taylor C. Tumour-derived exosomes and their role in cancer-associated T-cell signalling defects. Br. J. Cancer. 2005;92:305–311. doi: 10.1038/sj.bjc.6602316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Whiteside TL. Tumor-induced death of immune cells: its mechanisms and consequences. Semin. Cancer. Biol. 2002;12:43–50. doi: 10.1006/scbi.2001.0402. [DOI] [PubMed] [Google Scholar]
- 87.Andreola G, Rivoltini L, Castelli C, Huber V, Perego P, Deho P, Squarcina P, Accornero P, Lozupone F, Lugini L, Stringaro A, Molinari A, Arancia G, Gentile M, Parmiani G, Fais S. Induction of lymphocyte apoptosis by tumor cell secretion of FasL-bearing microvesicles. J. Exp. Med. 2002;195:1303–1316. doi: 10.1084/jem.20011624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Abusamra AJ, Zhong Z, Zheng X, Li M, Ichim TE, Chin JL, Min WP. Tumor exosomes expressing Fas ligand mediate CD8+ T-cell apoptosis. Blood. Cells. Mol. Dis. 2005;35:169–173. doi: 10.1016/j.bcmd.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 89.Esartia PT, Deichman GI, Kluchareva TE, Matveeva VA, Uvarova EN, Trapesnikov NN. Allogenic bone-marrow transfusion suppresses development of lung metastases in osteogenic sarcoma patients after radical surgery. Int. J. Cancer. 1993;54:907–910. doi: 10.1002/ijc.2910540606. [DOI] [PubMed] [Google Scholar]
- 90.Pross HF, Lotzova E. Role of natural killer cells in cancer. Nat. Immun. 1993;12:279–292. [PubMed] [Google Scholar]
- 91.Jovic V, Konjević G, Radulović S, Jelić S, Spuzić I. Impaired perforin-dependent NK cell cytotoxicity and proliferative activity of peripheral blood T cells is associated with metastatic melanoma. Tumori. 2001;87:324–329. doi: 10.1177/030089160108700509. [DOI] [PubMed] [Google Scholar]
- 92.Kodama T, Takeda K, Shimozato O, Hayakawa Y, Atsuta M, Kobayashi K, Ito M, Yagita H, Okumura K. Perforin-dependent NK cell cytotoxicity is sufficient for anti-metastatic effect of IL-12. Eur. J. Immunol. 1999;29:1390–1396. doi: 10.1002/(SICI)1521-4141(199904)29:04<1390::AID-IMMU1390>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- 93.Smyth MJ, Thia KY, Cretney E, Kelly JM, Snook MB, Forbes CA, Scalzo AA. Perforin is a major contributor to NK cell control of tumor metastasis. J. Immunol. 1999;162:6658–6662. [PubMed] [Google Scholar]
- 94.Liu C, Yu S, Zinn K, Wang J, Zhang L, Jia Y, Kappes JC, Barnes S, Kimberly RP, Grizzle WE, Zhang HG. Murine mammary carcinoma exosomes promote tumor growth by suppression of NK cell function. J. Immunol. 2006;176:1375–1385. doi: 10.4049/jimmunol.176.3.1375. [DOI] [PubMed] [Google Scholar]
- 95.Zhang HG, Kim H, Liu C, Yu S, Wang J, Grizzle WE, Kimberly RP, Barnes S. Curcumin reverses breast tumor exosomes mediated immune suppression of NK cell tumor cytotoxicity. Biochim. Biophys. Acta. 2007;1773:1116–1123. doi: 10.1016/j.bbamcr.2007.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Greening DW, Gopal SK, Mathias RA, Liu L, Sheng J, Zhu HJ, Simpson RJ. Emerging roles of exosomes during epithelial-mesenchymal transition and cancer progression. Semin. Cell. Dev. Biol. 2015;40:60–71. doi: 10.1016/j.semcdb.2015.02.008. [DOI] [PubMed] [Google Scholar]
- 97.Franzen CA, Blackwell RH, Todorovic V, Greco KA, Foreman KE, Flanigan RC, Kuo PC, Gupta GN. Urothelial cells undergo epithelial-to-mesenchymal transition after exposure to muscle invasive bladder cancer exosomes. Oncogenesis. 2015;4:e163. doi: 10.1038/oncsis.2015.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ngora H, Galli UM, Miyazaki K, Zöller M. Membrane-bound and exosomal metastasis-associated C4.4A promotes migration by associating with the alpha(6)beta(4) integrin and MT1-MMP. Neoplasia. 2012;14:95–107. doi: 10.1593/neo.111450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.You Y, Shan Y, Chen J, Yue H, You B, Shi S, Li X, Cao X. Matrix metalloproteinase 13-containing exosomes promote nasopharyngeal carcinoma metastasis. Cancer Sci. 2015;106:1669–1677. doi: 10.1111/cas.12818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, Uryu K, Pharmer L, King T, Bojmar L, Davies AE, Ararso Y, Zhang T, Zhang H, Hernandez J, Weiss JM, Dumont-Cole VD, Kramer K, Wexler LH, Narendran A, Schwartz GK, Healey JH, Sandstrom P, Labori KJ, Kure EH, Grandgenett PM, Hollingsworth MA, de Sousa M, Kaur S, Jain M, Mallya K, Batra SK, Jarnagin WR, Brady MS, Fodstad O, Muller V, Pantel K, Minn AJ, Bissell MJ, Garcia BA, Kang Y, Rajasekhar VK, Ghajar CM, Matei I, Peinado H, Bromberg J, Lyden D. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–335. doi: 10.1038/nature15756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu Y, Cao X. Organotropic metastasis: role of tumor exosomes. Cell. Res. 2016;26:149–150. doi: 10.1038/cr.2015.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Choy C, Jandial R. Breast Cancer Exosomes Breach the Blood-Brain Barrier. Neurosurgery. 2016;78:N10–11. doi: 10.1227/NEU.0000000000001242. [DOI] [PubMed] [Google Scholar]
- 103.Hood JL, San RS, Wickline SA. Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res. 2011;71:3792–3801. doi: 10.1158/0008-5472.CAN-10-4455. [DOI] [PubMed] [Google Scholar]
- 104.Maji S, Chaudhary P, Akopova I, Nguyen PM, Hare RJ, Gryczynski I, Vishwanatha JK. Exosomal Annexin II Promotes Angiogenesis and Breast Cancer Metastasis. Mol. Cancer Res. 2017;15:93–105. doi: 10.1158/1541-7786.MCR-16-0163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer. 2004;4:891–899. doi: 10.1038/nrc1478. [DOI] [PubMed] [Google Scholar]
- 106.Tredan O, Galmarini CM, Patel K, Tannock IF. Drug resistance and the solid tumor microenvironment. J. Natl. Cancer Inst. 2007;99:1441–1454. doi: 10.1093/jnci/djm135. [DOI] [PubMed] [Google Scholar]
- 107.Hammond EM, Asselin MC, Forster D, O'Connor JP, Senra JM, Williams KJ. The meaning, measurement and modification of hypoxia in the laboratory and the clinic. Clin. Oncol. (R. Coll. Radiol.) 2014;26:277–288. doi: 10.1016/j.clon.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 108.Cosse JP, Michiels C. Tumour hypoxia affects the responsiveness of cancer cells to chemotherapy and promotes cancer progression. Anticancer Agents Med. Chem. 2008;8:790–797. doi: 10.2174/187152008785914798. [DOI] [PubMed] [Google Scholar]
- 109.Fais S, Venturi G, Gatenby B. Microenvironmental acidosis in carcinogenesis and metastases: new strategies in prevention and therapy. Cancer Metastasis Rev. 2015;34:165. [Google Scholar]
- 110.Warburg O. On the origin of cancer cells. Science. 1956;123:309–314. doi: 10.1126/science.123.3191.309. [DOI] [PubMed] [Google Scholar]
- 111.Cairns RA, Harris IS, Mak TW. Regulation of cancer cell metabolism. Nat. Rev. Cancer. 2011;11:85–95. doi: 10.1038/nrc2981. [DOI] [PubMed] [Google Scholar]
- 112.Huber V, De Milito A, Harguindey S, Reshkin SJ, Wahl ML, Rauch C, Chiesi A, Pouysségur J, Gatenby RA, Rivoltini L, Fais S. Proton dynamics in cancer. J. Transl. Med. 2010;8:57. doi: 10.1186/1479-5876-8-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Webb BA, Chimenti M, Jacobson MP, Barber DL. Dysregulated pH: a perfect storm for cancer progression. Nat. Rev. Cancer. 2011;11:671–677. doi: 10.1038/nrc3110. [DOI] [PubMed] [Google Scholar]
- 114.Barar J, Omidi Y. Dysregulated pH in Tumor Microenvironment Checkmates Cancer Therapy. Bioimpacts. 2013;3:149–162. doi: 10.5681/bi.2013.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Daniel C, Bell C, Burton C, Harguindey S, Reshkin SJ, Rauch C. The role of proton dynamics in the development and maintenance of multidrug resistance in cancer. Biochim. Biophys. Acta. 2013;1832:606–617. doi: 10.1016/j.bbadis.2013.01.020. [DOI] [PubMed] [Google Scholar]
- 116.Gillies RJ, Raghunand N, Karczmar GS, Bhujwalla ZM. MRI of the tumor microenvironment. J. Magn. Reson. Imaging. 2002;16:430–450. doi: 10.1002/jmri.10181. [DOI] [PubMed] [Google Scholar]
- 117.Gallagher FA, Kettunen MI, Day SE, Hu DE, Ardenkjaer-Larsen JH, Ri Zandt, Jensen PR, Karlsson M, Golman K, Lerche MH, Brindle KM. Magnetic resonance imaging of pH in vivo using hyperpolarized 13C-labelled bicarbonate. Nature. 2008;453:940–943. doi: 10.1038/nature07017. [DOI] [PubMed] [Google Scholar]
- 118.van Sluis R, Bhujwalla ZM, Raghunand N, Ballesteros P, Alvarez J, Cerdán S, Galons JP, Gillies RJ. In vivo imaging of extracellular pH using 1H MRSI. Magn. Reson. Med. 1999;41:743–750. doi: 10.1002/(sici)1522-2594(199904)41:4<743::aid-mrm13>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 119.De Milito A, Canese R, Marino ML, Borghi M, Iero M, Villa A, Venturi G, Lozupone F, Iessi E, Logozzi M, Della Mina P, Santinami M, Rodolfo M, Podo F, Rivoltini L, Fais S. pH-dependent antitumor activity of proton pump inhibitors against human melanoma is mediated by inhibition of tumor acidity. Int. J. Cancer. 2010;127:207–219. doi: 10.1002/ijc.25009. [DOI] [PubMed] [Google Scholar]
- 120.Azzarito T, Venturi G, Cesolini A, Fais S. Lansoprazole induces sensitivity to suboptimal doses of paclitaxel in human melanoma. Cancer Lett. 2015;256:697–703. doi: 10.1016/j.canlet.2014.10.017. [DOI] [PubMed] [Google Scholar]
- 121.Spugnini EP, Sonveaux P, Stock C, Perez-Sayans M, De Milito A, Avnet S, Garcìa AG, Harguindey S, Fais S. Proton channels and exchangers in cancer. Biochim. Biophys. Acta. 2015;1848:2715–2726. doi: 10.1016/j.bbamem.2014.10.015. [DOI] [PubMed] [Google Scholar]
- 122.Izumi H, Torigoe T, Ishiguchi H, Uramoto H, Yoshida Y, Tanabe M, Ise T, Murakami T, Yoshida T, Nomoto M, Kohno K. Cellular pH regulators: potentially promising molecular targets for cancer chemotherapy. Cancer Treat. Rev. 2003;29:541–549. doi: 10.1016/s0305-7372(03)00106-3. [DOI] [PubMed] [Google Scholar]
- 123.Rabinowitz JD, Coller HA. Partners in the Warburg effect. Elife. 2016;5:e15938. doi: 10.7554/eLife.15938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Milane L, Singh A, Mattheolabakis G, Suresh M, Amiji MM. Exosome mediated communication within the tumor microenvironment. J. Control. Release. 2015;219:278–294. doi: 10.1016/j.jconrel.2015.06.029. [DOI] [PubMed] [Google Scholar]
- 125.Lugini L, Matarrese P, Tinari A, Lozupone F, Federici C, Iessi E, Gentile M, Luciani F, Parmiani G, Rivoltini L, Malorni W, Fais S. Cannibalism of live lymphocytes by human metastatic but not primary melanoma cells. Cancer Res. 2006;66:3629–3638. doi: 10.1158/0008-5472.CAN-05-3204. [DOI] [PubMed] [Google Scholar]
- 126.Luciani F, Spada M, De Milito A, Molinari A, Rivoltini L, Montinaro A, Marra M, Lugini L, Logozzi M, Lozupone F, Federici C, Iessi E, Parmiani G, Arancia G, Belardelli F, Fais S. Effect of proton pump inhibitor pretreatment on resistance of solid tumors to cytotoxic drugs. J. Natl. Cancer Inst. 2004;96:1702–1713. doi: 10.1093/jnci/djh305. [DOI] [PubMed] [Google Scholar]
- 127.De Milito A, Iessi E, Logozzi M, Lozupone F, Spada M, Marino ML, Federici C, Perdicchio M, Matarrese P, Lugini L, Nilsson A, Fais S. Proton pump inhibitors induce apoptosis of human B-cell tumors through a caspase-independent mechanism involving reactive oxygen species. Cancer Res. 2007;67:5408–5417. doi: 10.1158/0008-5472.CAN-06-4095. [DOI] [PubMed] [Google Scholar]
- 128.Parolini I, Federici C, Raggi C, Lugini L, Palleschi S, De Milito A, Coscia C, Iessi E, Logozzi M, Molinari A, Colone M, Tatti M, Sargiacomo M, Fais S. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J. Biol. Chem. 2009;284:34211–34222. doi: 10.1074/jbc.M109.041152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Properzi F, Logozzi M, Fais S. Exosomes: the future of biomarkers in medicine. Biomark. Med. 2013;7:769–778. doi: 10.2217/bmm.13.63. [DOI] [PubMed] [Google Scholar]
- 130.Cappello F, Logozzi M, Campanella C, Bavisotto CC, Marcilla A, Properzi F, Fais S. Exosome levels in human body fluids: A tumor marker by themselves? Eur. J. Pharm. Sci. 2017;96:93–98. doi: 10.1016/j.ejps.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 131.Logozzi M, Angelini DF, Iessi E, Mizzoni D, Di Raimo R, Federici C, Lugini L, Borsellino G, Gentilucci A, Pierella F, Marzio V, Sciarra A, Battistini L, Fais S. Increased PSA expression on prostate cancer exosomes In In vitro condition and in cancer patients. Cancer Lett. 2017;403:318–329. doi: 10.1016/j.canlet.2017.06.036. [DOI] [PubMed] [Google Scholar]
- 132.Fais S, O'Driscoll L, Borras FE, Buzas E, Camussi G, Cappello F, Carvalho J, Cordeiro da Silva A, Del Portillo H, El Andaloussi S, Ficko Trček T, Furlan R, Hendrix A, Gursel I, Kralj-Iglic V, Kaeffer B, Kosanovic M, Lekka ME, Lipps G, Logozzi M, Marcilla A, Sammar M, Llorente A, Nazarenko I, Oliveira C, Pocsfalvi G, Rajendran L, Raposo G, Rohde E, Siljander P, van Niel G, Vasconcelos MH, Yáñez-Mó M, Yliperttula ML, Zarovni N, Zavec AB, Giebel B. Evidence-Based Clinical Use of Nanoscale Extracellular Vesicles in Nanomedicine. ACS Nano. 2016;10:3886–3899. doi: 10.1021/acsnano.5b08015. [DOI] [PubMed] [Google Scholar]
- 133.Janowska-Wieczorek A, Wysoczynski M, Kijowski J, Marquez-Curtis L, Machalinski B, Ratajczak J, Ratajczak MZ. Microvesicles derived from activated platelets induce metastasis and angiogenesis in lung cancer. Int. J. Cancer. 2005;113:752–760. doi: 10.1002/ijc.20657. [DOI] [PubMed] [Google Scholar]
- 134.Kajita M, Itoh Y, Chiba T, Mori H, Okada A, Kinoh H, Seiki M. Membrane-type 1 matrix metalloproteinase cleaves CD44 and promotes cell migration. J. Cell. Biol. 2001;153:893–904. doi: 10.1083/jcb.153.5.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Belkin AM, Akimov SS, Zaritskaya LS, Ratnikov BI, DeryuginaI EI, Strongin AY. Matrix-dependent proteolysis of surface transglutaminase by membrane-type metalloproteinase regulates cancer cell adhesion and locomotion. J. Biol. Chem. 2001;276:18415–18422. doi: 10.1074/jbc.M010135200. [DOI] [PubMed] [Google Scholar]
- 136.Liu SC, Bassi DE, Zhang SY, Holoran D, Conti CJ, Klein-Szanto AJ. Overexpression of cyclin D2 is associated with increased in vivo invasiveness of human squamous carcinoma cells. Mol. Carcinog. 2002;34:131–139. doi: 10.1002/mc.10057. [DOI] [PubMed] [Google Scholar]
- 137.Janowska-Wieczorek A, Majka M, Kijowski J, Baj-Krzyworzeka M, Reca R, Turner AR, Ratajczak J, Emerson SG, Kowalska MA, Ratajczak MZ. Platelet-derived microparticles bind to hematopoietic stem/progenitor cells and enhance their engraftment. Blood. 2001;98:3143–3149. doi: 10.1182/blood.v98.10.3143. [DOI] [PubMed] [Google Scholar]
- 138.Baj-Krzyworzeka M, Majka M, Pratico D, Ratajczak J, Vilaire G, Kijowski J, Reca R, Janowska-Wieczorek A, Ratajczak MZ. Platelet-derived microparticles stimulate proliferation, survival, adhesion, and chemotaxis of hematopoietic cells. Exp. Hematol. 2002;30:450–459. doi: 10.1016/s0301-472x(02)00791-9. [DOI] [PubMed] [Google Scholar]
- 139.Rozmyslowicz T, Majka M, Kijowski J, Murphy SL, Conover DO, Poncz M, Ratajczak J, Gaulton GN, Ratajczak MZ. Platelet- and megakaryocyte-derived microparticles transfer CXCR4 receptor to CXCR4-null cells and make them susceptible to infection by X4-HIV. AIDS. 2003;17:33–42. doi: 10.1097/00002030-200301030-00006. [DOI] [PubMed] [Google Scholar]
- 140.Libura J, Drukala J, Majka M, Tomescu O, Navenot JM, Kucia M, Marquez L, Peiper SC, Barr FG, Janowska-Wieczorek A, Ratajczak MZ. CXCR4-SDF-1 signaling is active in rhabdomyosarcoma cells and regulates locomotion, chemotaxis, and adhesion. Blood. 2002;100:2597–2606. doi: 10.1182/blood-2002-01-0031. [DOI] [PubMed] [Google Scholar]
- 141.Cai Z, Yang F, Yu L, Yu Z, Jiang L, Wang Q, Yang Y, Wang L, Cao X, Wang J. Activated T cell exosomes promote tumor invasion via Fas signaling pathway. J. Immunol. 2012;188:5954–5961. doi: 10.4049/jimmunol.1103466. [DOI] [PubMed] [Google Scholar]
- 142.Xie Y, Zhang H, Li W, Deng Y, Munegowda MA, Chibbar R, Qureshi M, Xiang J. Dendritic cells recruit T cell exosomes via exosomal LFA-1 leading to inhibition of CD8+ CTL responses through downregulation of peptide/MHC class I and Fas ligand-mediated cytotoxicity. J. Immunol. 2010;185:5268–5278. doi: 10.4049/jimmunol.1000386. [DOI] [PubMed] [Google Scholar]
- 143.Rabinowits G, Gerçel-Taylor C, Day JM, Taylor DD, Kloecker GH. Exosomal microRNA: a diagnostic marker for lung cancer. Clin. Lung Cancer. 2009;10:42–46. doi: 10.3816/CLC.2009.n.006. [DOI] [PubMed] [Google Scholar]
- 144.Wu X, Somlo G, Yu Y, Palomares MR, Li AX, Zhou W, Chow A, Yen Y, Rossi JJ, Gao H, Wang J, Yuan YC, Frankel P, Li S, Ashing-Giwa KT, Sun G, Wang Y, Smith R, Robinson K, Ren X, Wang SE. De novo sequencing of circulating miRNAs identifies novel markers predicting clinical outcome of locally advanced breast cancer. J. Transl. Med. 2012;10:42. doi: 10.1186/1479-5876-10-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Jeppesen DK, Nawrocki A, Jensen SG, Thorsen K, Whitehead B, Howard KA, Dyrskjøt L, Ørntoft TF, Larsen MR, Ostenfeld MS. Quantitative proteomics of fractionated membrane and lumen exosome proteins from isogenic metastatic and nonmetastatic bladder cancer cells reveal differential expression of EMT factors. Proteomics. 2014;14:699–712. doi: 10.1002/pmic.201300452. [DOI] [PubMed] [Google Scholar]
- 146.Bijnsdorp IV, Geldof AA, Lavaei M, Piersma SR, van Moorselaar RJ, Jimenez CR. Exosomal ITGA3 interferes with non-cancerous prostate cell functions and is increased in urine exosomes of metastatic prostate cancer patients. J. Extracell. Vesicles. 2013;2 doi: 10.3402/jev.v2i0.22097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Camacho L, Guerrero P, Marchetti D. MicroRNA and protein profiling of brain metastasis competent cell-derived exosomes. PLoS One. 2013;8:e73790. doi: 10.1371/journal.pone.0073790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Prensner JR, Chinnaiyan AM. The emergence of lncRNAs in cancer biology. Cancer Discov. 2011;1:391–407. doi: 10.1158/2159-8290.CD-11-0209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Tang J, Jiang R, Deng L, Zhang X, Wang K, Sun B. Circulation long non-coding RNAs act as biomarkers for predicting tumorigenesis and metastasis in hepatocellular carcinoma. Oncotarget. 2015;6:4505–4515. doi: 10.18632/oncotarget.2934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kahlert C, Melo SA, Protopopov A, Tang J, Seth S, Koch M, Zhang J, Weitz J, Chin L, Futreal A, Kalluri R. Identification of double-stranded genomic DNA spanning all chromosomes with mutated KRAS and p53 DNA in the serum exosomes of patients with pancreatic cancer. J. Biol. Chem. 2014;289:3869–3875. doi: 10.1074/jbc.C113.532267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Huber V, Fais S, Iero M, Lugini L, Canese P, Squarcina P, Zaccheddu A, Colone M, Arancia G, Gentile M, Seregni E, Valenti R, Ballabio G, Belli F, Leo E, Parmiani G, Rivoltini L. Human colorectal cancer cells induce T-cell death through release of proapoptotic microvesicles: role in immune escape. Gastroenterology. 2005;128:1796–1804. doi: 10.1053/j.gastro.2005.03.045. [DOI] [PubMed] [Google Scholar]
- 152.Sun D, Zhuang X, Xiang X, Liu Y, Zhang S, Liu C, Barnes S, Grizzle W, Miller D, Zhang HG. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol. Ther. 2010;18:1606–1614. doi: 10.1038/mt.2010.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Smyth T, Kullberg M, Malik N, Smith-Jones P, Graner MW, Anchordoquy TJ. Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J. Control. Release. 2015;199:145–155. doi: 10.1016/j.jconrel.2014.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Rivoltini L, Chiodoni C, Squarcina P, Tortoreto M, Villa A, Vergani B, Bürdek M, Botti L, Arioli I, Cova A, Mauri G, Vergani E, Bianchi B, Della Mina P, Cantone L, Bollati V, Zaffaroni N, Gianni AM, Colombo MP, Huber V. TNF-Related Apoptosis-Inducing Ligand (TRAIL)-Armed Exosomes Deliver Proapoptotic Signals to Tumor Site. Clin. Cancer Res. 2016;22:3499–3512. doi: 10.1158/1078-0432.CCR-15-2170. [DOI] [PubMed] [Google Scholar]
- 155.Zeelenberg IS, Ostrowski M, Krumeich S, Bobrie A, Jancic C, Boissonnas A, Delcayre A, Le Pecq JB, Combadière B, Amigorena S, Théry C. Targeting tumor antigens to secreted membrane vesicles in vivo induces efficient antitumor immune responses. Cancer Res. 2008;68:1228–1235. doi: 10.1158/0008-5472.CAN-07-3163. [DOI] [PubMed] [Google Scholar]
- 156.Lee EY, Park KS, Yoon YJ, Lee J, Moon HG, Jang SC, Choi KH, Kim YK, Gho YS. Therapeutic effects of autologous tumor-derived nanovesicles on melanoma growth and metastasis. PLoS One. 2012;7:e33330. doi: 10.1371/journal.pone.0033330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Hwang I, Shen X, Sprent J. Direct stimulation of naive T cells by membrane vesicles from antigen-presenting cells: distinct roles for CD54 and B7 molecules. Proc. Natl. Acad. Sci. U. S. A. 2003;100:6670–6675. doi: 10.1073/pnas.1131852100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, Ricciardi-Castagnoli P, Raposo G, Amigorena S. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 1998;4:594–600. doi: 10.1038/nm0598-594. [DOI] [PubMed] [Google Scholar]
- 159.Steinman RM, Pope M. Exploiting dendritic cells to improve vaccine efficacy. J. Clin. Invest. 2002;109:1519–1526. doi: 10.1172/JCI15962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Johnsen KB, Gudbergsson JM, Skov MN, Pilgaard L, Moos T, Duroux M. A comprehensive overview of exosomes as drug delivery vehicles - endogenous nanocarriers for targeted cancer therapy. Biochim. Biophys. Acta. 2014;1846:75–87. doi: 10.1016/j.bbcan.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 161.Haney MJ, Klyachko NL, Zhao Y, Gupta R, Plotnikova EG, He Z, Patel T, Piroyan A, Sokolsky M, Kabanov AV, Batrakova EV. Exosomes as drug delivery vehicles for Parkinson's disease therapy. J. Control. Release. 2015;207:18–30. doi: 10.1016/j.jconrel.2015.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Canitano A, Venturi G, Borghi M, Ammendolia MG, Fais S. Exosomes released in vitro from Epstein-Barr virus (EBV)-infected cells contain EBV-encoded latent phase mRNAs. Cancer Lett. 2013;337:193–199. doi: 10.1016/j.canlet.2013.05.012. [DOI] [PubMed] [Google Scholar]
- 163.Cossetti C, Lugini L, Astrologo L, Saggio I, Fais S, Spadafora C. Soma-to-germline transmission of RNA in mice xenografted with human tumour cells: possible transport by exosomes. PLoS One. 2014;9:e101629. doi: 10.1371/journal.pone.0101629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Iraci N, Gaude E, Leonardi T, Costa ASH, Cossetti C, Peruzzotti-Jametti L, Bernstock JD, Saini3 HK, Gelati M, Vescovi AL, Bastos C, Faria N, Occhipinti LG, Enright AJ, Frezza C, Pluchino S. Extracellular vesicles are independent metabolic units with asparaginase activity. Nat. Chem. Biol. 2017;13:951–955. doi: 10.1038/nchembio.2422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Burke M, Choksawangkarn W, Edwards N, Ostrand-Rosenberg S, Fenselau C. Exosomes from myeloid-derived suppressor cells carry biologically active proteins. J. Proteome Res. 2014;13:836–843. doi: 10.1021/pr400879c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Melo SA, Sugimoto H, O'Connell JT, Kato N, Villanueva A, Vidal A, Qiu L, Vitkin E, Perelman LT, Melo CA, Lucci A, Ivan C, Calin GA, Kalluri R. Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell. 2014;26:707–721. doi: 10.1016/j.ccell.2014.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Fais S. A nonmainstream approach against cancer. J. Enzyme Inhib. Med. Chem. 2016;31:882–889. doi: 10.3109/14756366.2016.1156105. [DOI] [PubMed] [Google Scholar]
- 168.Hoffman RM. Stromal-cell and cancer-cell exosomes leading the metastatic exodus for the promised niche. Breast Cancer Res. 2013;15:310. doi: 10.1186/bcr3426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Zhou W, Fong MY, Min Y, Somlo G, Liu L, Palomares MR, Yu Y, Chow A, O'Connor ST, Chin AR, Yen Y, Wang Y, Marcusson EG, Chu P, Wu J, Wu X, Li AX, Li Z, Gao H, Ren X, Boldin MP, Lin PC, Wang SE. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell. 2014;25:501–515. doi: 10.1016/j.ccr.2014.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Gorczynski RM, Erin N, Zhu F. Serum-derived exosomes from mice with highly metastatic breast cancer transfer increased metastatic capacity to a poorly metastatic tumor. Cancer Med. 2016;5:325–336. doi: 10.1002/cam4.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Rodríguez M, Silva J, Herrera A, Herrera M, Peña C, Martín P, Gil-Calderón B, Larriba MJ, Coronado MJ, Soldevilla B, Turrión VS, Provencio M, Sánchez A, Bonilla F, García-Barberán V. Exosomes enriched in stemness/metastatic-related mRNAS promote oncogenic potential in breast cancer. Oncotarget. 2015;6:40575–40587. doi: 10.18632/oncotarget.5818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Wang X, Ding X, Nan L, Wang Y, Wang J, Yan Z, Zhang W, Sun J, Zhu W, Ni B, Dong S, Yu L. Investigation of the roles of exosomes in colorectal cancer liver metastasis. Oncol. Rep. 2015;33:2445–2453. doi: 10.3892/or.2015.3843. [DOI] [PubMed] [Google Scholar]
- 173.Hong BS, Cho JH, Kim H, Choi EJ, Rho S, Kim J, Kim JH, Choi DS, Kim YK, Hwang D, Gho YS. Colorectal cancer cell-derived microvesicles are enriched in cell cycle-related mRNAs that promote proliferation of endothelial cells. BMC Genomics. 2009;10:556. doi: 10.1186/1471-2164-10-556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Choi DS, Choi DY, Hong BS, Jang SC, Kim DK, Lee J, Kim YK, Kim KP, Gho YS. Quantitative proteomics of extracellular vesicles derived from human primary and metastatic colorectal cancer cells. J. Extracell. Vesicles. 2012:1. doi: 10.3402/jev.v1i0.18704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Min H, Sun X, Yang X, Zhu H, Liu J, Wang Y, Chen G, Sun X. Exosomes Derived from Irradiated Esophageal Carcinoma-Infiltrating T Cells Promote Metastasis by Inducing the Epithelial-Mesenchymal Transition in Esophageal Cancer Cells. Pathol. Oncol. Res. 2017 Jan 28; doi: 10.1007/s12253-016-0185-z. [DOI] [PubMed] [Google Scholar]
- 176.Zhang H, Deng T, Liu R, Bai M, Zhou L, Wang X, Li S, Wang X, Yang H, Li J, Ning T, Huang D, Li H, Zhang L, Ying G, Ba Y. Exosome-delivered EGFR regulates liver microenvironment to promote gastric cancer liver metastasis. Nat. Commun. 2017;8:15016. doi: 10.1038/ncomms15016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Liu D, Li C, Trojanowicz B, Li X, Shi D, Zhan C, Wang Z, Chen L. CD97 promotion of gastric carcinoma lymphatic metastasis is exosome dependent. Gastric Cancer. 2016;19:754–766. doi: 10.1007/s10120-015-0523-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Shi R, Wang PY, Li XY, Chen JX, Li Y, Zhang XZ, Zhang CG, Jiang T, Li WB, Ding W, Cheng SJ. Exosomal levels of miRNA-21 from cerebrospinal fluids associated with poor prognosis and tumor recurrence of glioma patients. Oncotarget. 2015;6:26971–26981. doi: 10.18632/oncotarget.4699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.He M, Qin H, Poon TC, Sze SC, Ding X, Co NN, Ngai SM, Chan TF, Wong N. Hepatocellular carcinoma-derived exosomes promote motility of immortalized hepatocyte through transfer of oncogenic proteins and RNAs. Carcinogenesis. 2015;36:1008–1018. doi: 10.1093/carcin/bgv081. [DOI] [PubMed] [Google Scholar]
- 180.Valencia K, Luis-Ravelo D, Bovy N, Antón I, Martínez-Canarias S, Zandueta C, Ormazábal C, Struman I, Tabruyn S, Rebmann V, De Las Rivas J, Guruceaga E, Bandrés E, Lecanda F. MiRNA cargo within exosome-like vesicle transfer influences metastatic bone colonization. Mol. Oncol. 2014;8:689–703. doi: 10.1016/j.molonc.2014.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Liu Y, Gu Y, Han Y, Zhang Q, Jiang Z, Zhang X, Huang B, Xu X, Zheng J, Cao X. Tumor Exosomal RNAs Promote Lung Pre-metastatic Niche Formation by Activating Alveolar Epithelial TLR3 to Recruit Neutrophils. Cancer Cell. 2016;30:243–256. doi: 10.1016/j.ccell.2016.06.021. [DOI] [PubMed] [Google Scholar]
- 182.Vardaki I, Ceder S, Rutishauser D, Baltatzis G, Foukakis T, Panaretakis T. Periostin is identified as a putative metastatic marker in breast cancer-derived exosomes. Oncotarget. 2016;7:74966–74978. doi: 10.18632/oncotarget.11663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Pfeffer SR, Grossmann KF, Cassidy PB, Yang CH, Fan M, Kopelovich L, Leachman SA, Pfeffer LM. Detection of Exosomal miRNAs in the Plasma of Melanoma Patients. J. Clin. Med. 2015;4:2012–2027. doi: 10.3390/jcm4121957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Hao S, Ye Z, Li F, Meng Q, Qureshi M, Yang J, Xiang J. Epigenetic transfer of metastatic activity by uptake of highly metastatic B16 melanoma cell-released exosomes. Exp. Oncol. 2006;28:126–131. [PubMed] [Google Scholar]
- 185.Adachi E, Sakai K, Nishiuchi T, Imamura R, Sato H, Matsumoto K. Different growth and metastatic phenotypes associated with a cell-intrinsic change of Met in metastatic melanoma. Oncotarget. 2016;7:70779–70793. doi: 10.18632/oncotarget.12221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Hood JL. Melanoma exosome induction of endothelial cell GM-CSF in pre-metastatic lymph nodes may result in different M1 and M2 macrophage mediated angiogenic processes. Med. Hypotheses. 2016;94:118–122. doi: 10.1016/j.mehy.2016.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Aga M, Bentz GL, Raffa S, Torrisi MR, Kondo S, Wakisaka N, Yoshizaki T, Pagano JS, Shackelford J. Exosomal HIF1α supports invasive potential of nasopharyngeal carcinoma-associated LMP1-positive exosomes. Oncogene. 2014;33:4613–4622. doi: 10.1038/onc.2014.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Taverna S, Pucci M, Giallombardo M, Di Bella MA, Santarpia M, Reclusa P, Gil-Bazo I, Rolfo C, Alessandro R. Amphiregulin contained in NSCLC-exosomes induces osteoclast differentiation through the activation of EGFR pathway. Sci. Rep. 2017;7:3170. doi: 10.1038/s41598-017-03460-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Li L, Li C, Wang S, Wang Z, Jiang J, Wang W, Li X, Chen J, Liu K, Li C, Zhu G. Exosomes Derived from Hypoxic Oral Squamous Cell Carcinoma Cells Deliver miR-21 to Normoxic Cells to Elicit a Prometastatic Phenotype. Cancer Res. 2016;76:1770–1780. doi: 10.1158/0008-5472.CAN-15-1625. [DOI] [PubMed] [Google Scholar]
- 190.Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, Xiang J, Zhang T, Theilen TM, García-Santos G, Williams C, Ararso Y, Huang Y, Rodrigues G, Shen TL, Labori KJ, Lothe IM, Kure EH, Hernandez J, Doussot A, Ebbesen SH, Grandgenett PM, Hollingsworth MA, Jain M, Mallya K, Batra SK, Jarnagin WR, Schwartz RE, Matei I, Peinado H, Stanger BZ, Bromberg J, Lyden D. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat. Cell. Biol. 2015;17:816–826. doi: 10.1038/ncb3169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Sánchez CA, Andahur EI, Valenzuela R, Castellón EA, Fullá JA, Ramos CG, Triviño JC. Exosomes from bulk and stem cells from human prostate cancer have a differential microRNA content that contributes cooperatively over local and pre-metastatic niche. Oncotarget. 2016;7:3993–4008. doi: 10.18632/oncotarget.6540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Fedele C, Singh A, Zerlanko BJ, Iozzo RV, Languino LR. The αvβ6 integrin is transferred intercellularly via exosomes. J. Biol. Chem. 2015;290:4545–4551. doi: 10.1074/jbc.C114.617662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Wang H, Rana S, Giese N, Büchler MW, Zöller M. Tspan8, CD44v6 and alpha6beta4 are biomarkers of migrating pancreatic cancer-initiating cells. Int. J. Cancer. 2013;133:416–426. doi: 10.1002/ijc.28044. [DOI] [PubMed] [Google Scholar]
- 194.Rana S, Malinowska K, Zöller M. Exosomal tumor microRNA modulates premetastatic organ cells. Neoplasia N.Y.N. 2013;15:281–295. doi: 10.1593/neo.122010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Fabbri M, et al. MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response. Proc Natl Acad Sci U S A. 2012;109(31):E2110–6. doi: 10.1073/pnas.1209414109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Huebner AR, et al. Exosomes in urine biomarker discovery. Adv Exp Med Biol. 2015;845:43–58. doi: 10.1007/978-94-017-9523-4_5. [DOI] [PubMed] [Google Scholar]
- 197.Sarko DK, McKinney CE. Exosomes: Origins and Therapeutic Potential for Neurodegenerative Disease. Frontiers in Neuroscience. 2017;11:82. doi: 10.3389/fnins.2017.00082. [DOI] [PMC free article] [PubMed] [Google Scholar]