Abstract
The easy accessibility of skin made dermal application, one of the approaches for local drug therapy. Effectiveness of topical drug application is depended on different parameters such as skin barrier properties, physicochemical properties of drug and vehicle, and interaction between drug and its vehicle with the skin layers. In this review, an overview of skin structure and feature of polymeric micelles as topical nanocarriers is provided. We also summarized the research studies dealing with the application of polymeric micelles for cutaneous delivery. In the past decades, numerous types of nanocarriers have been widely investigated as a novel delivery approach to improve skin penetration and localization of drugs in normal skin and dermatological diseases. Polymeric micelles are one of them, with their specific ability to encapsulate hydrophilic drugs. These carriers can enhance the therapeutic efficacy and minimize the systemic side effects of the drugs. Polymeric micelles could enhance the deposition of drugs in targeted sites of the skin in the normal and dermatological diseases such as psoriasis and acne. Nevertheless, still there is a need to investigate the mechanism of action of these carriers and the fate of polymeric micelles in skin.
Key words: Cutaneous drug delivery, nanocarriers, polymeric micelles, skin topical drug delivery
INTRODUCTION
Topical delivery of drugs for the treatment of dermatological disease is preferred by most patients and physicians for a local effect where it can reduce the need for systemically administration and their side effects. However, the skin barrier inhibits the drugs to penetrate easily, as the skin is naturally refractive to highly hydrophilic or highly lipophilic compounds. The various chemical and physical enhancers in the past decades have been investigated to overcome the skin barrier which most of them disrupted stratum corneum.[1] New strategy is application of small nanometer-sized carriers to improve skin penetration or localization without invasive manner.[2] Nanocarriers may facilitate drug delivery by encapsulation pharmaceutical active ingredients to form specific features such as penetrate hair follicle, interact with skin's lipid to transport, and create depot form to sustained release.[3] Nanocarriers increased surface area-to-volume ratio result in enhanced permeation through all routes across the skin including intracellular, intercellular, and finally, along the shafts of hair follicles in the trans-appendage pathway.[4]
In this review, we will discuss some of the investigation using polymeric micelles as nanocarriers for cutaneous deliveries in normal and dermatological disorder. In addition, selected examples from the literature have been included to demonstrate the mechanism of action of various drugs employing polymeric micellar-based drug delivery systems.
THE STRATUM CORNEUMMAIN BARRIER IN NORMAL SKIN AND DERMATOLOGICAL DISORDERS
Skin plays a protecting function of body from xenobiotics. Skin consists of a highly efficient, multilayered barrier that hampers the transdermal delivery of therapeutic agents. The stratum corneum (SC), the outermost skin layer (thickness 20 μm) composed of dead epidermal cells (corneocytes), is the main barrier of skin.[5,6] The corneocytes are surrounded by lipoprotein envelope, and they have less water-binding capacity. This general structure of the SC can be described as “brick and mortar” [Figure 1]. The “bricks” are composed of corneocytes and intercellular lipid matrix constituting the “mortar.”[7] The corneocytes are not only in horizontal layers but also in vertical columns. These columns are called “cluster” which correspond the wrinkle liens.[8] Intercluster regions (furrows) is one of the penetration pathways which we describe further in the text.
Figure 1.
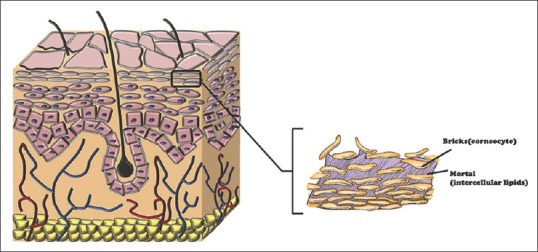
The brick-mortar structure of skin
The substance that can penetrate with a passive route through SC is limited to small molecules (<500 Da), with intermediate lipophilic and hydrophilic property (log 1–3).[9] To increase the range of active compounds with the lack of these specific properties, the use of nanocarriers has been interested.
FOLLICULAR PENETRATION PATHWAYS
It had been assumed in the past years that substances penetrate often through the intercellular route, inside lipid surrounded corneocytes. In recent years, hair follicles indicate an efficient way to the application of substance topically across the skin and potential penetration pathway for nanocarriers.[10,11,12] Hair follicles represent an efficient reservoir for dermal delivery.[13] The size of particles has been reported to be an important factor to penetration into hair follicle.[14] Not only small molecules can penetrate through the follicular epithelium into the living tissue but also although molecules that are too large for penetration are entrapped within the hair follicles and make depot form.[11]
SKIN BARRIER PROPERTY IN DERMATOLOGICAL DISORDERS
Skin's structure undergoes alteration in some disease. Several skin diseases are characterized by a varying degree of skin barrier dysfunction. In psoriasis, the immune system is activated that leads to overproduction of skin cells and forming scaly patches on the skin surface. Barrier function of psoriatic skin is shown to be decreased. Transepidermal water loss (TWL) in psoriasis is significantly higher than normal skin.[15] Acne is chronic inflammation in pilosebaceous unit in the skin. Alterations in follicular keratinization occur in the subclinical stages of acne lesions formation. There is an increase in filaggrin expression in keratinocytes' lining follicle wall within acne. Filaggrin has a critical role in epidermal differentiation and structure and function of the SC.[16] Yamamoto et al. found that, in patient with acne exhibited significantly higher sebum secretion, greater TWL and lower skin hydration that lead to SC permeability barrier impairment.[17] Burn is another disease that skin undergoes trauma. In thermal burn, as temperature increases, protein disruption increases and protein denaturation progresses to coagulation. Cell necrosis usually begins at the skin surface where the heat energy is most intense. Due to this trauma, skin changes to eschar. Eschar represents an open wound without the properties and benefits of normal skin. Therefore, this wound does not provide a mechanical and immunological barrier against foreign objects such as drugs.[18] In fact, there are not adequate investigations on all substances which show more penetration rate in skin diseases,[19] but clinical studies show a moderate increase in penetration compared to intact skin.[20]
FUNDAMENTAL OF POLYMERIC MICELLES
Structure
It is well recognized that surfactants are able to self-assemble in water and above the critical micelle concentration (CMC), they spontaneously self-aggregate to form spherical micelles to decrease surface-free energy in an aqueous medium. Polymeric micelles have attracted enormous attention in drug delivery field because their CMC is several 1000-fold lower (<10 mg/l) than classic micelles[21] [Figure 2]. Polymeric micelles are a specific class of micelles which formed from copolymers consisting of both hydrophilic and hydrophobic monomer units (block copolymer). In aqueous media, hydrophobic portion forms the core, while hydrophilic portion forms the shell, also called corona.[22,23,24] One of the essential properties of the core is their ability to solubilize sparingly water-soluble drugs, whereas the shell provides a location for hydrophilic compounds.[25,26] Furthermore, shell generate required colloidal stability and plays an important role in limiting opsonization and results in longer blood circulation time and better accumulation in the target tissue.[22] Polymeric micelles can be subdivided into different classes: (1) di-block copolymers (hydrophilic-hydrophobic), (2) tri-block copolymers (hydrophilic-hydrophobic-hydrophilic), (3) graft copolymers (hydrophilic-hydrophobic), and (4) ionic copolymers (hydrophilic-ionic). The hydrophilic part of diblock copolymers is often poly(ethylene glycol) (PEG) which is highly biocompatible and fore hydrophobic part, polylactides (PLA), poly(ε-caprolactone), and poly (D, L-lactic-co-glycolic acid), is more common that have been approved by the Food and Drug Administration for biomedical applications in humans.[27,28]
Figure 2.
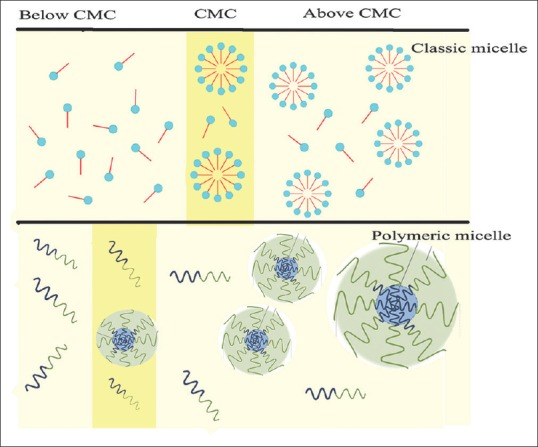
The micelle and polymeric micelle formation in the aqueous media
Size and morphology
The size of these micelles can vary from 10 to 200 nm but can be increased by drug incorporation.[29,30] Different types of micellar structures are reported such as worm-like, disc-like, hamburger, raspberry, and sheet.[22] There are some different methods which are used to study micelle dimensions including dynamic light scattering, static light scattering, atomic force microscopy (AFM), and transmission electron microscopy (TEM). AFM and TEM give direct images and insight into shape.
Techniques used for polymeric micelles preparation
In accordance to the physicochemical characteristics of the polymers and drug, several methods can be used.[31] The most common methods including direct dissolution, dialysis, solvent evaporation, oil-in-water emulsion, solid dispersion, and freeze-drying have been shown in Figure 3.
Figure 3.
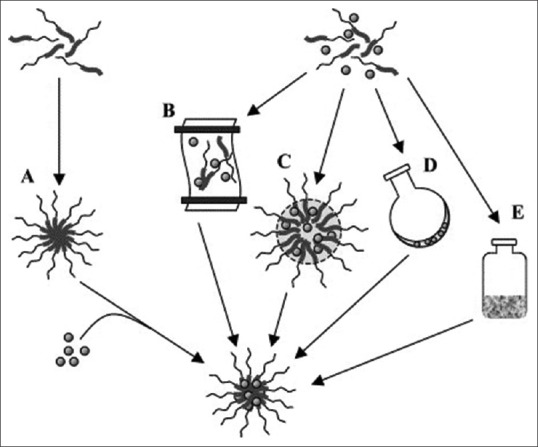
Different methods for preparation of polymeric micelles.[31] (a) Simple equilibrium, (b) dialysis, (c) oil-in-water emulsion, (d) solution casting, and (e) freeze-drying
Direct dissolution of the amphiphilic copolymer and drug in water, at or above CMC, results in self-assembling of the drug and copolymer to form polymeric micelles. This is the simplest approach to polymeric micelle preparation, although it is usually associated with low drug loading. Moreover, the low drug loading may be enhanced by increasing the temperature of the system.[32]
The dialysis method uses water-miscible organic solvents (such as N, N-dimethylformamide) along with a drug to co-solubilize the polymer and the drug. The organic solvent is replaced by water through a semipermeable membrane which induces self-association to form micelles.[33,34,35] This technique often requires >36 h for efficient loading which is a drawback in use of this approach.
In the oil-in-water emulsion method, drug along with the polymer dissolved in a water-immiscible organic solvent (such as chloroform) and added this organic under stirring in the aqueous phase. The organic solvent is then removed by evaporation and solvent-free solution containing micelles.[36,37]
The solvent evaporation method includes drug and polymer which are dissolved in a volatile organic solvent. This solution introduced to aqueous phase under sonication. After the evaporation of the organic solvent, a thin film of drug/polymer is formed at the bottom of the flask. This film is reconstituted by shaking in an aqueous phase.[38,39]
The copolymer and the drug are dissolved in an organic solvent in the solid dispersion method. Then, a polymeric drug matrix is obtained after the evaporation of solvent. Addition of hot water to this preheated matrix results in to form micelles.[40,41]
Freeze-drying method utilized freeze-dryable organic solvents such as tert-butanol. The polymer and the drug are both present in water/tert-butanol mixture, followed by the removal of the solvents using a freeze-drying process. The drug-loaded micelles assemble spontaneously upon reconstitution with water.[42]
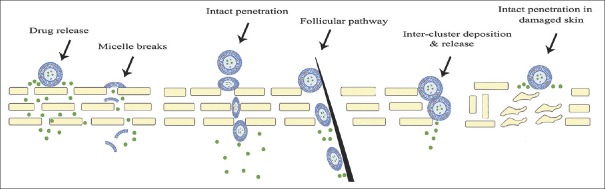
THE FATE OF POLYMERIC MICELLES IN NORMAL SKIN AND DERMATOLOGICAL DISORDER AFTER TOPICAL APPLICATION
What is the fate of polymeric micelles after topical application? For answer to this question, Smejkalova et al. designed a research and found that micelles with 200 nm particle size do not remain at the skin surface and penetrate into the skin. Confocal images showed the vehicle localized in the keratinocytes and fibroblasts. In the other hand, the micelles remained intact up to 10 mm in deep of skin, but images from deeper layers showed slow and progressive micelle disruption.[43] In another study, Lapteva et al. indicated that polymeric micelles made by methoxy-poly(ethylene glycol) di-(hexyl-substituted polylactide) substituted (MPEG-dihexPLA) polylactide copolymer was unable to cross the SC.[44] Bachhav et al., in another study, demonstrated higher skin delivery of Azole antifungal compounds by polymeric micelles accumulation in hair follicles. They showed by confocal laser scanning microscopy (CLSM) images that polymeric micelles reached into hair follicles intact.[45] However, Lapteva et al. showed that polymeric micelles were deposited between corneocytes and inter the clusters of corneocytes. Therefore, they suggested that intercluster penetration is the preferred transport pathway for polymeric micelles.[46] The fate of polymeric micelles in normal skin and dermatological diseases has been shown in Figure 4.
Figure 4.
Polymeric micelle's pathway through normal skin and dermatological diseases
POLYMERIC MICELLES IN SKIN DRUG DELIVERY
Polymeric micelles have been investigated as alternative vehicles for parenteral,[47] oral,[23] ocular,[48] pulmonary,[49] and nasal routes.[50] However, investigation to targeted cutaneous delivery by application of polymeric micelles is rare, and the mechanism of their action is not clearly demonstrated. However, it has been observed that polymeric nanoparticles may penetrate through SC and have been accumulated in hair follicles.[51] A different application of polymeric micelles for cutaneous drug delivery has been demonstrated in Table 1.
Table 1.
Different application of polymeric micelles for cutaneous drug delivery
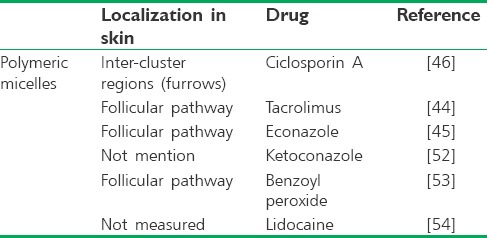
One of the studies shows polymeric micelles for skin delivery by Lapetva et al. they formulated ciclosporin A (CsA)-loaded polymeric micelles using MPEG-dihexPLA diblock copolymer and was tested on in vitro porcine ear skin. Micelle and drug penetration pathways were subsequently detected with CLSM using fluorescein-labeled CsA (Fluo-CsA) and Nile-Red (NR)-labeled copolymer. Micelles were prepared using the solvent evaporation method and demonstrated spherical morphology and small particle size (25–52 nm). These formulations increased the aqueous solubility of CsA by 518-folds. In spite of CsA delivery from the micelle formulation was 18-folds more than the control formulation, CsA permeation across porcine skin was extremely low and only very small amounts might reach the systemic circulation which is appropriate feature when the disease is limited to skin. Finally, it was observed that Fluo-CsA skin penetration was deeper into skin's layers by releasing drug from micelle in the intercluster region which is probably one of the penetration pathways for cutaneous drug delivery.[46]
The same researchers group formulated tacrolimus (TAC) using MPEG-dihexPLA to treatment psoriasis. Micelle diameters were 10–50 nm. Despite poor water solubility of TAC, micelle formulation incorporated a large amount of TAC and increases its solubility 518-folds in aqueous media. Optimized formulation showed 9-folds increase in delivery to porcine skin and 4-folds with human skin whereas the delivery of commercial ointment was low. They visualized by CLSM, NR-MPEG-dihexPLA micelles remained mainly on the skin surface and could not pass across the SC but localized into follicular ducts.[44]
In an expand study, different azoles' antifungal compounds (clotrimazole, fluconazole, and econazole nitrate) were loaded in polymeric micelles with different copolymers. The hydrodynamic diameters of the azoles-loaded micelles were between 70 and 165 nm with spherical morphology. The best formulation was provided by the MPEG-dihexPLA micelles loaded with econazole and incorporated with an efficiency of 98.3%. This micelle formulation showed significantly higher penetration than its commercial liposomal gel in both the porcine and human skins. The authors concluded that better skin delivery is due to the smaller size of formulation while the commercial formulation containing numerous penetration enhancers. SLCM study showed the MPEG-dihexPLA micelles could facilitate targeted follicular delivery.[45] In addition, the penetration of the ketoconazole-loaded methoxy poly (ethylene glycol)-b-poly (δ-valerolactone) (MPEG-PVL) micelles to skin delivery was investigated. The micelles were obtained with encapsulation efficiency of 86.39% and particle diameter of about 12 nm. Ketoconazole aqueous solubility increases to 86-fold. The fluorescein-loaded MPEG-PVL micelles confirmed that the micelles are able to deliver more amounts of the dye into deep skin layers compared to the control aqueous fluorescein solution.[52]
Furthermore, Kahraman et al.[53] optimized polymeric micelles carriers of the antiacne compound benzoyl peroxide (BPO). The polyethylene oxide–polypropylene oxide–polyethylene oxide (Pluronic® F127) copolymer was used to formulate micelles using the solid dispersion method. The optimized micelle formulation presented particle sizes of ~25 nm and spherical morphology and an efficiency of ~82%. The in vitro penetration of BPO into skin from an optimized micelle formulation and its commercial gel as a control was examined using porcine skin. The optimized micelle formulation was significantly more efficacious than the commercial gel because of BPO deposition in the porcine skin was 3-fold more. NR loaded Pluronic® F127 micelles is used to examine the localization of nanocarriers in the skin by CLSM. Deposition of NR loaded Pluronic® F127 micelles in the follicular pathway reported in comparison to skin samples treated with NR control solution.
Lalatsa et al.[54] characterized three different lidocaine-loaded nanocarriers include polymeric micelles, solid lipid nanoparticles (SLNs), and self-nanoemulsifying drug delivery systems (SNEDDSs) as transcutaneous drug delivery systems. All particles had a size below 150 nm, demonstrated good colloidal stability with a negative zeta-potential and a spherical morphology. They evaluated skin permeability of nanocarriers using artificial membranes in conjunction with Franz cell. Cumulative lidocaine concentration after 6 h was significant for both polymeric micelles (345.7 ± 23.8 mg/cm2/h) and SNEDDS (224.8 ± 118.2 mg/cm2/h) compared with SLNs (127.3 ± 25.4 mg/cm2/h). Therefore, in this case, better polymeric micelles' penetration can because of lack a formidable lipidic barrier in cellulose acetate.
More recently, Šmejkalová et al. made polymeric micelles from hyaluronan and loaded with NR by solvent evaporation method. Micelle diameters were 21–230 nm. They suggested using CLSM that the penetration route is transcellular. In this article, it was not reported that accumulation in follicular hair observed by CLSM or not.[43]
CONCLUSION
Topical application of drugs would be useful in the treatment of most skin diseases. Achievement to effective drug concentration is mostly dependent to skin barrier property. Traditional dosage forms such as cream, gel, and ointment are not efficient for topical treatment because of poor penetration of drugs into targeted layers of skin. Polymeric micelles as novel drug carrier provide perfect drug delivery through skin. In this review, several studies have been shown improved cutaneous targeted delivery of several drugs. Benefits of polymeric micelles as cutaneous nanocarrier that have been reported are including improve drug solubilization into the skin, increase partitioning of hydrophilic drug into the SC, drug localization into the hair follicles and keratinocytes in different layers of epidermis, and providing depot into the skin by slow and sustained drug release from intact polymeric micelles. Even after skin barrier alteration in dermatological disorders such as psoriasis, acne, and burn, it seems that polymeric micelles show efficient carrier for targeted drug delivery.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol. 2008;26:1261–8. doi: 10.1038/nbt.1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fireman S, Toledano O, Neimann K, Loboda N, Dayan N. A look at emerging delivery systems for topical drug products. Dermatol Ther. 2011;24:477–88. doi: 10.1111/j.1529-8019.2012.01464.x. [DOI] [PubMed] [Google Scholar]
- 3.Vogt A, Wischke C, Neffe AT, Ma N, Alexiev U, Lendlein A, et al. Nanocarriers for drug delivery into and through the skin – Do existing technologies match clinical challenges? J Control Release. 2016;242:3–15. doi: 10.1016/j.jconrel.2016.07.027. [DOI] [PubMed] [Google Scholar]
- 4.Lee RW, Shenoy DB, Sheel R. Handbook of Non-Invasive Drug Delivery Systems. Burlington, MA: Elsevier Inc; 2010. Micellar nanoparticles: Applications for topical and passive transdermal drug delivery; p. 37.p. 58. [Google Scholar]
- 5.Pham QD, Björklund S, Engblom J, Topgaard D, Sparr E. Chemical penetration enhancers in stratum corneum - relation between molecular effects and barrier function. J Control Release. 2016;232:175–87. doi: 10.1016/j.jconrel.2016.04.030. [DOI] [PubMed] [Google Scholar]
- 6.Bhatnagar S, Dave K, Venuganti VVK. Microneedles in the clinic. J Control Release. 2017;260:164–82. doi: 10.1016/j.jconrel.2017.05.029. [DOI] [PubMed] [Google Scholar]
- 7.Baumann LS, Baumann L. Cosmetic dermatology. McGraw-Hill Professional Publishing; 2009. [Google Scholar]
- 8.Cevc G, Vierl U. Nanotechnology and the transdermal route: A state of the art review and critical appraisal. J Control Release. 2010;141:277–99. doi: 10.1016/j.jconrel.2009.10.016. [DOI] [PubMed] [Google Scholar]
- 9.Roberts MS, Cross SE, Pellett MA. Skin transport. Drugs and The Pharmaceutical Sciences. 2002;119:89–196. [Google Scholar]
- 10.Lauterbach A, Müller-Goymann CC. Comparison of rheological properties, follicular penetration, drug release, and permeation behavior of a novel topical drug delivery system and a conventional cream. Eur J Pharm Biopharm. 2014;88:614–24. doi: 10.1016/j.ejpb.2014.10.001. [DOI] [PubMed] [Google Scholar]
- 11.Patzelt A, Richter H, Knorr F, Schäfer U, Lehr CM, Dähne L, et al. Selective follicular targeting by modification of the particle sizes. J Control Release. 2011;150:45–8. doi: 10.1016/j.jconrel.2010.11.015. [DOI] [PubMed] [Google Scholar]
- 12.Lademann J, Richter H, Meinke MC, Lange-Asschenfeldt B, Antoniou C, Mak WC, et al. Drug delivery with topically applied nanoparticles: Science fiction or reality. Skin Pharmacol Physiol. 2013;26:227–33. doi: 10.1159/000351940. [DOI] [PubMed] [Google Scholar]
- 13.Lademann J, Richter H, Teichmann A, Otberg N, Blume-Peytavi U, Luengo J, et al. Nanoparticles – An efficient carrier for drug delivery into the hair follicles. Eur J Pharm Biopharm. 2007;66:159–64. doi: 10.1016/j.ejpb.2006.10.019. [DOI] [PubMed] [Google Scholar]
- 14.Blume-Peytavi U, Vogt A. Human hair follicle: Reservoir function and selective targeting. Br J Dermatol. 2011;165(Suppl 2):13–7. doi: 10.1111/j.1365-2133.2011.10572.x. [DOI] [PubMed] [Google Scholar]
- 15.Takahashi H, Tsuji H, Minami-Hori M, Miyauchi Y, Iizuka H. Defective barrier function accompanied by structural changes of psoriatic stratum corneum. J Dermatol. 2014;41:144–8. doi: 10.1111/1346-8138.12393. [DOI] [PubMed] [Google Scholar]
- 16.Thiboutot D, Del Rosso JQ. Acne vulgaris and the epidermal barrier: Is acne vulgaris associated with inherent epidermal abnormalities that cause impairment of barrier functions? Do any topical acne therapies alter the structural and/or functional integrity of the epidermal barrier. J Clin Aesthet Dermatol. 2013;6:18–24. [PMC free article] [PubMed] [Google Scholar]
- 17.Yamamoto A, Takenouchi K, Ito M. Impaired water barrier function in acne vulgaris. Arch Dermatol Res. 1995;287:214–8. doi: 10.1007/BF01262335. [DOI] [PubMed] [Google Scholar]
- 18.Zadeh BS, Moghimi H, Santos P, Hadgraft J, Lane ME. A comparative study of the in vitro permeation characteristic of sulphadiazine across synthetic membranes and eschar tissue. Int Wound J. 2008;5:633–8. doi: 10.1111/j.1742-481X.2008.00539.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yoshiike T, Aikawa Y, Sindhvananda J, Suto H, Nishimura K, Kawamoto T, et al. Skin barrier defect in atopic dermatitis: Increased permeability of the stratum corneum using dimethyl sulfoxide and theophylline. J Dermatol Sci. 1993;5:92–6. doi: 10.1016/0923-1811(93)90076-2. [DOI] [PubMed] [Google Scholar]
- 20.Gattu S, Maibach HI. Modest but increased penetration through damaged skin: An overview of the in vivo human model. Skin Pharmacol Physiol. 2011;24:2–9. doi: 10.1159/000314995. [DOI] [PubMed] [Google Scholar]
- 21.Abolmaali SS, Tamaddon AM, Salmanpour M, Mohammadi S, Dinarvand R. Block ionomer micellar nanoparticles from double hydrophilic copolymers, classifications and promises for delivery of cancer chemotherapeutics. Eur J Pharm Sci. 2017;104:393–405. doi: 10.1016/j.ejps.2017.04.009. [DOI] [PubMed] [Google Scholar]
- 22.Deshmukh AS, Chauhan PN, Noolvi MN, Chaturvedi K, Ganguly K, Shukla SS, et al. Polymeric micelles: Basic research to clinical practice. Int J Pharm. 2017;532:249–68. doi: 10.1016/j.ijpharm.2017.09.005. [DOI] [PubMed] [Google Scholar]
- 23.Makhmal Zadeh BS, Soror Khodarahmpour AS. Preparation and evaluation of the polymeric micellar formulation for oral delivery of griseofulvin. Int J Pharm. 2014;4:92–7. [Google Scholar]
- 24.Lee SM, Jang WD. Polyion complex micelle formed from tetraphenylethene containing block copolymer. Biomater Res. 2017;21:17. doi: 10.1186/s40824-017-0103-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Torchilin VP. Structure and design of polymeric surfactant-based drug delivery systems. J Control Release. 2001;73:137–72. doi: 10.1016/s0168-3659(01)00299-1. [DOI] [PubMed] [Google Scholar]
- 26.Mahmud A, Xiong XB, Aliabadi HM, Lavasanifar A. Polymeric micelles for drug targeting. J Drug Target. 2007;15:553–84. doi: 10.1080/10611860701538586. [DOI] [PubMed] [Google Scholar]
- 27.Cagel M, Tesan FC, Bernabeu E, Salgueiro MJ, Zubillaga MB, Moretton MA, et al. Polymeric mixed micelles as nanomedicines: Achievements and perspectives. Eur J Pharm Biopharm. 2017;113:211–28. doi: 10.1016/j.ejpb.2016.12.019. [DOI] [PubMed] [Google Scholar]
- 28.Mandal A, Bisht R, Rupenthal ID, Mitra AK. Polymeric micelles for ocular drug delivery: From structural frameworks to recent preclinical studies. J Control Release. 2017;248:96–116. doi: 10.1016/j.jconrel.2017.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu Y, Park K. Polymeric micelles and alternative nanonized delivery vehicles for poorly soluble drugs. Int J Pharm. 2013;453:198–214. doi: 10.1016/j.ijpharm.2012.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Torchilin VP. Micellar nanocarriers: Pharmaceutical perspectives. Pharm Res. 2007;24:1–6. doi: 10.1007/s11095-006-9132-0. [DOI] [PubMed] [Google Scholar]
- 31.Gaucher G, Dufresne MH, Sant VP, Kang N, Maysinger D, Leroux JC, et al. Block copolymer micelles: Preparation, characterization and application in drug delivery. J Control Release. 2005;109:169–88. doi: 10.1016/j.jconrel.2005.09.034. [DOI] [PubMed] [Google Scholar]
- 32.Yokoyama M. Polymeric micelles as a new drug carrier system and their required considerations for clinical trials. Expert Opin Drug Deliv. 2010;7:145–58. doi: 10.1517/17425240903436479. [DOI] [PubMed] [Google Scholar]
- 33.Yu BG, Okano T, Kataoka K, Kwon G. Polymeric micelles for drug delivery: Solubilization and haemolytic activity of amphotericin B. J Control Release. 1998;53:131–6. doi: 10.1016/s0168-3659(97)00245-9. [DOI] [PubMed] [Google Scholar]
- 34.Rapoport NY, Herron JN, Pitt WG, Pitina L. Micellar delivery of doxorubicin and its paramagnetic analog, ruboxyl, to HL-60 cells: Effect of micelle structure and ultrasound on the intracellular drug uptake. J Control Release. 1999;58:153–62. doi: 10.1016/s0168-3659(98)00149-7. [DOI] [PubMed] [Google Scholar]
- 35.Kedar U, Phutane P, Shidhaye S, Kadam V. Advances in polymeric micelles for drug delivery and tumor targeting. Nanomedicine. 2010;6:714–29. doi: 10.1016/j.nano.2010.05.005. [DOI] [PubMed] [Google Scholar]
- 36.Djordjevic J, Michniak B, Uhrich KE. Amphiphilic star-like macromolecules as novel carriers for topical delivery of nonsteroidal anti-inflammatory drugs. AAPS PharmSci. 2003;5:E26. doi: 10.1208/ps050426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Patil YB, Toti US, Khdair A, Ma L, Panyam J. Single-step surface functionalization of polymeric nanoparticles for targeted drug delivery. Biomaterials. 2009;30:859–66. doi: 10.1016/j.biomaterials.2008.09.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lavasanifar A, Samuel J, Kwon GS. Micelles self-assembled from poly(ethylene oxide)-block-poly(N-hexyl stearate L-aspartamide) by a solvent evaporation method: Effect on the solubilization and haemolytic activity of amphotericin B. J Control Release. 2001;77:155–60. doi: 10.1016/s0168-3659(01)00477-1. [DOI] [PubMed] [Google Scholar]
- 39.Singh PK, Pawar VK, Jaiswal AK, Singh Y, Srikanth CH, Chaurasia M, et al. Chitosan coated pluronicF127 micelles for effective delivery of amphotericin B in experimental visceral leishmaniasis. Int J Biol Macromol. 2017;105:1220–31. doi: 10.1016/j.ijbiomac.2017.07.161. [DOI] [PubMed] [Google Scholar]
- 40.Zhang X, Jackson JK, Burt HM. Development of amphiphilic diblock copolymers as micellar carriers of taxol. Int J Pharm. 1996;132:195–206. [Google Scholar]
- 41.Montanha MC, Silva LL, Pangoni FB, Cesar GB, Gonçalves RS, Caetano W, et al. Response surface method optimization of a novel Hypericin formulation in P123 micelles for colorectal cancer and antimicrobial photodynamic therapy. J Photochem Photobiol B Biol. 2017;170:247–55. doi: 10.1016/j.jphotobiol.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 42.Fournier E, Dufresne MH, Smith DC, Ranger M, Leroux JC. A novel one-step drug-loading procedure for water-soluble amphiphilic nanocarriers. Pharm Res. 2004;21:962–8. doi: 10.1023/b:pham.0000029284.40637.69. [DOI] [PubMed] [Google Scholar]
- 43.Šmejkalová D, Muthný T, Nešporová K, Hermannová M, Achbergerová E, Huerta-Angeles G, et al. Hyaluronan polymeric micelles for topical drug delivery. Carbohydr Polym. 2017;156:86–96. doi: 10.1016/j.carbpol.2016.09.013. [DOI] [PubMed] [Google Scholar]
- 44.Lapteva M, Mondon K, Möller M, Gurny R, Kalia YN. Polymeric micelle nanocarriers for the cutaneous delivery of tacrolimus: A targeted approach for the treatment of psoriasis. Mol Pharm. 2014;11:2989–3001. doi: 10.1021/mp400639e. [DOI] [PubMed] [Google Scholar]
- 45.Bachhav YG, Mondon K, Kalia YN, Gurny R, Möller M. Novel micelle formulations to increase cutaneous bioavailability of azole antifungals. J Control Release. 2011;153:126–32. doi: 10.1016/j.jconrel.2011.03.003. [DOI] [PubMed] [Google Scholar]
- 46.Lapteva M, Santer V, Mondon K, Patmanidis I, Chiriano G, Scapozza L, et al. Targeted cutaneous delivery of ciclosporin A using micellar nanocarriers and the possible role of inter-cluster regions as molecular transport pathways. J Control Release. 2014;196:9–18. doi: 10.1016/j.jconrel.2014.09.021. [DOI] [PubMed] [Google Scholar]
- 47.Richter A, Olbrich C, Krause M, Kissel T. Solubilization of sagopilone, a poorly water-soluble anticancer drug, using polymeric micelles for parenteral delivery. Int J Pharm. 2010;389:244–53. doi: 10.1016/j.ijpharm.2010.01.032. [DOI] [PubMed] [Google Scholar]
- 48.Ribeiro A, Sandez-Macho I, Casas M, Alvarez-Pérez S, Alvarez-Lorenzo C, Concheiro A, et al. Poloxamine micellar solubilization of α-tocopherol for topical ocular treatment. Colloids Surf B Biointerfaces. 2013;103:550–7. doi: 10.1016/j.colsurfb.2012.10.055. [DOI] [PubMed] [Google Scholar]
- 49.Andrade F, das Neves J, Gener P, Schwartz S, Jr, Ferreira D, Oliva M, et al. Biological assessment of self-assembled polymeric micelles for pulmonary administration of insulin. Nanomedicine. 2015;11:1621–31. doi: 10.1016/j.nano.2015.05.006. [DOI] [PubMed] [Google Scholar]
- 50.Abdelbary GA, Tadros MI. Brain targeting of olanzapine via intranasal delivery of core-shell difunctional block copolymer mixed nanomicellar carriers:In vitro characterization, ex vivo estimation of nasal toxicity and in vivo biodistribution studies. Int J Pharm. 2013;452:300–10. doi: 10.1016/j.ijpharm.2013.04.084. [DOI] [PubMed] [Google Scholar]
- 51.Alvarez-Román R, Naik A, Kalia YN, Guy RH, Fessi H. Skin penetration and distribution of polymeric nanoparticles. J Control Release. 2004;99:53–62. doi: 10.1016/j.jconrel.2004.06.015. [DOI] [PubMed] [Google Scholar]
- 52.Deng P, Teng F, Zhou F, Song Z, Meng N, Feng R, et al. Methoxy poly (ethylene glycol)-b-poly (δ-valerolactone) copolymeric micelles for improved skin delivery of ketoconazole. J Biomater Sci Polym Ed. 2017;28:63–78. doi: 10.1080/09205063.2016.1244371. [DOI] [PubMed] [Google Scholar]
- 53.Kahraman E, Özhan G, Özsoy Y, Güngör S. Polymeric micellar nanocarriers of benzoyl peroxide as potential follicular targeting approach for acne treatment. Colloids Surf B Biointerfaces. 2016;146:692–9. doi: 10.1016/j.colsurfb.2016.07.029. [DOI] [PubMed] [Google Scholar]
- 54.Lalatsa A, Emeriewen K, Protopsalti V, Skelton G, Saleh GM. Developing transcutaneous nanoenabled anaesthetics for eyelid surgery. Br J Ophthalmol. 2016;100:871–6. doi: 10.1136/bjophthalmol-2015-308250. [DOI] [PubMed] [Google Scholar]