Abstract
Background
Schizophrenia is characterized by abnormalities in referential communication, which may be linked to more general deficits in proactive cognitive control. We used event-related potentials (ERPs) to probe the timing and nature of the neural mechanisms engaged as people with schizophrenia linked pronouns to their preceding referents during word-by-word sentence comprehension.
Methods
We measured ERPs to pronouns in two-clause sentences from 16 people with schizophrenia and 20 demographically-matched control participants. Our design crossed the number of potential referents (1-referent, 2-referent) with whether the pronoun matched the gender of its preceding referent(s) (matching, mismatching). This gave rise to four conditions: (1) 1-referent matching (“…Edward took courses in accounting but he…”), (2) 2-referent matching (“…Edward and Phillip took courses but he…”), (3) 1-referent mismatching (“…Edward took courses in accounting but she…”), and (4) 2-referent mismatching (“…Edward and Phillip took courses but she…”).
Results
Consistent with previous findings, healthy controls produced a larger left anteriorly-distributed negativity between 400-600ms to 2-referent matching than to 1-referent matching pronouns (the “Nref effect”). In contrast, people with schizophrenia produced a larger centro-posterior positivity effect between 600-800ms. Both patient and control groups produced a larger positivity between 400-800ms to mismatching than to matching pronouns.
Conclusions
These findings suggest that proactive mechanisms of referential processing, reflected by the Nref effect, are impaired in schizophrenia, while reactive mechanisms, reflected by the positivity effects, are relatively spared. Indeed, patients may compensate for proactive deficits by retro-actively engaging with context to influence the processing of inputs at a later stage of analysis.
Keywords: comprehension, discourse, ERPs, language, pronouns, reference
Introduction
In order to communicate effectively, producers and comprehenders must draw upon common sets of shared referents (1, 2). For example, to understand even a simple sentence like, “Edward took courses in accounting but he didn't learn much”, the comprehender must infer that the communicator intended the pronoun, “he” to refer to “Edward”. It is well established that people with schizophrenia struggle with referential processing and that this can impair social functioning. However, we know little about the mechanisms underlying such impairments. In healthy individuals, the ability to proactively hold potential referents (like “Edward”) over time, with the goal of linking them to subsequent words (like “he”), may be related to more general mechanisms of proactive cognitive control, which are known to be impaired in schizophrenia (3, 4). We used event-related potentials (ERPs) to ask whether people with schizophrenia are selectively impaired in their use of proactive mechanisms of linking referents with pronouns during word-by-word sentence comprehension.
In schizophrenia, problems of establishing referential cohesion have been well characterized in language production (5-11). They include an ambiguous use of pronouns (5-8), and an overall increase in the use of pronouns (9). These abnormalities tend to remain stable over time (12, 13), and they can predict measures of social cognition (9, 14). People with schizophrenia can also struggle with linking anaphors1 to their preceding referents during language comprehension (15, 16). This can contribute to social dysfunction, leading to difficulties following social conversations (17-19).
Healthy adult comprehenders are thought to engage two broad sets of mechanisms to achieve referential cohesion. The first is proactive in nature. Here, discourse context, along with stored real-world knowledge, is used to predict2 (or pre-activate) possible upcoming events or states, along with their associated referents (21-27). Upon encountering an anaphor in the bottom-up input, the comprehender must link it to one of the pre-activated referents, further enhancing its activation and suppressing any competing pre-activated alternative referents.
The process of maintaining predicted referents over time, and linking them to incoming anaphors, is associated with an ERP effect with a frontal, and sometimes left-lateralized, scalp distribution, which has been termed the “Nref effect” (see (28) for a review), where “N” refers to the more negative-going polarity of the waveform evoked by anaphors that are more difficult versus easier to link to their preceding referent(s) (“ref”). A smaller (less negative) Nref is evoked by anaphors that unambiguously match just one referent that has been predicted with high probability, whereas a larger Nref is evoked by anaphors that are more difficult to link to a specific preceding referent (29-35) (e.g. by “he” in the sentence, “Edward and Phillip took courses but he…” (32)), perhaps because of competition from pre-activated alternative referents.3
The second set of mechanisms used to achieve referential cohesion are ‘reactive’ in nature, and are triggered only when an anaphor is perceived as failing to match referents that have been pre-activated by the context. This initial perception of referential failure leads comprehenders to search outside their current focus of attention and engage in prolonged attempts to retrieve either a previously-mentioned referent (and the context in which it was introduced) (48), or a novel referent (49) from longer-term episodic memory. This, in turn, may lead comprehenders to update their current representation of context with these newly retrieved referents.
In the ERP waveform, reactive processes more generally are associated with a late positive-going ERP component with a widespread or central-posterior scalp distribution. Late posterior positivities (or P600s) are produced when incoming bottom-up information cannot be linked to any information that has been anticipated based on the context — that is, when new inputs are perceived as strongly violating contextual constraints (50-52). They may reflect continued attempts to establish coherence (52-55) by retrieving information from longer-term memory and updating the representation of context (or, more generally, the contents of working memory, see (56-58)). In the case of referential processing, larger late positivities are evoked by anaphors that mismatch versus match the gender of all referents in their immediate context (32, 59, 60), e.g. by the pronoun, “she” versus “he” in the sentence, “Edward took courses in accounting but she …”, particularly when the task encourages the assessment of coherence (32, 59). They may, in part, reflect retroactive attempts to establish coherence by retrieving novel referents from outside the current focus of attention (e.g. (61, 62)).
Although the mechanisms underlying referential impairments in schizophrenia remain unclear, there is evidence from other cognitive domains that proactive mechanisms, functioning to hold goal-relevant information within working memory (WM) (63), are impaired, while reactive mechanisms are relatively preserved (3, 4, 64). Moreover, patients' performance in tasks that tap into general proactive WM mechanisms can predict referential impairments during language production (65-68), as well as other aspects of language comprehension that rely on retaining contextual information over time (69), see (70, 71) for discussion of domain-general control mechanisms in relation to impairments in discourse processing in schizophrenia.
On the basis of this previous work, we hypothesized that, in schizophrenia, earlier proactive neural mechanisms engaged in referential processing would be impaired, but later reactive referential mechanisms would be relatively spared. ERPs are particularly well suited for testing this hypothesis because their temporal resolution provides a direct neural index of the sequence of processing each individual word as sentences unfold in real time. We therefore measured ERPs as people with schizophrenia and demographically-matched controls read four types of sentences, presented word by word, and then judged their coherence: (1) 1-referent matching, (2) 2-referent matching, (3) 1-referent mismatching (referentially violating), and (4) 2-referent mismatching (referentially ambiguous), see Table 1.
Table 1. Sentence types and stimuli examples.
| Sentence Type | Example |
|---|---|
| 1) 1-referent gender matching | In night school Edward took courses in accounting but he didn't learn much. |
| 2) 2-referent gender matching | In night school Edward and Phillip took courses but he didn't learn much. |
| 3) 1-referent gender mismatching | In night school Edward took courses in accounting but she didn't learn much. |
| 4) 2-referent gender mismatching | In night school Edward and Phillip took courses but she didn't learn much. |
To probe proactive referential mechanisms, we contrasted ERPs to 1-referent matching pronouns (e.g. “he” in “…Edward took courses in accounting but he…”) and 2-referent matching pronouns (e.g. “he” in “Edward and Phillip took courses in accounting but he…”). Based on previous work (32), we expected that this contrast would produce an Nref effect in controls. We hypothesized that, if proactive referential mechanisms are impaired in people with schizophrenia, they would show a smaller Nref effect than in controls. To probe reactive referential mechanisms, we contrasted the 1-referent matching pronouns with both the 1-referent mismatching and the 2-referent mismatching pronouns (e.g. “she” versus “he” in “…Edward took courses in accounting but she/he…”). Based on previous work using similar stimuli and a similar task (59), we expected that this contrast would reveal a late positivity effect in healthy adults. Based on previous work suggesting that, despite proactive deficits, people with schizophrenia can still use high-level contextual information to influence later stages of processing (72, 73), we hypothesized that the schizophrenia group would also produce a late positivity effect to these referential violations.
Methods and Materials
Stimuli
120 two-clause sentences were constructed. The first clause introduced one character (1-referent) or two characters (2-referents, both of the same gender: 50% female, established using proper names). The second clause began with a pronoun that matched or mismatched the gender of these characters, giving rise to four sentence types, each with 30 items (Table 1). This fully-crossed design prevented participants from knowing on the basis of the context alone whether or not the pronoun would match or mismatch a preceding referent: a mismatching referent was just as likely to occur in a 1-referent as a 2-referent context. Because the 1-referent sentences were inherently shorter than the 2-referent sentences (due to the introduction of one rather than two characters), we added two additional words that did not alter sentence meaning to their first clause in order to match the total number of words prior to the pronoun across conditions.
Sentences were counterbalanced across four lists. To each list, we added 64 fillers without any pronouns. These introduced variety in the types of sentences viewed, and reduced the chance that participants would adopt an explicit strategy of anticipating that pronouns would always be encountered at particular points in the experimental sentences.
Participants
Individuals meeting DSM-IV criteria for schizophrenia, assessed using the SCID (74), were recruited from the Lindemann Mental Health Center, Boston. All but one were receiving stable doses of antipsychotic medication. Demographically-matched volunteers, who were not taking psychoactive medication and who had no histories of psychiatric disorders, were recruited by advertisement. All participants were native, primarily monolingual English speakers who had not learned any other language before the age of five. All were right-handed (75, 76), without histories of head trauma, neurological disorder, substance abuse within three months, or histories of substance dependence (as assessed using the DSM-IV). Written informed consent was obtained in all participants following the guidelines of the Massachusetts General Hospital and Tufts Medical Center Human Subjects Research Committees.
In the schizophrenia group, clinical assessments were carried out on the same day as the ERP study using the Scale for the Assessment of Positive Symptoms (77) and the Scale for the Assessment of Negative Symptoms (78). In all participants, working memory was assessed using a reading span task (79, 80).
Initially, 26 controls and 24 people with schizophrenia were recruited. However, the ERP datasets of 6 controls and 8 participants with schizophrenia were subsequently excluded from analysis because of low behavioral accuracy (3 controls; 5 patients), excessive artifact (2 controls) or both of these (1 control; 3 patients), see Methods for exclusion criteria. This left 20 controls and 16 people with schizophrenia whose data are included in the analyses reported. Demographic, psychopathological and reading span data are summarized in Table 2.
Table 2. Demographic, medication, working memory and symptom measures.
Means are shown with standard deviations in parentheses.
| Control Group | Schizophrenia Group | Comparison | |
|---|---|---|---|
| n | 20 [7 female] | 16 [3 female] | |
| Age | 45.6 (8.0) | 39.1 (12.7) | t(34)= 1.78, p=0.09 |
| Premorbid IQa | 107.7 (8.8) | 102.1 (9.6) | t(34)= 1.85, p=0.07 |
| Parental SESb | 3.2 (1.2) | 2.7 (1.3) | t(30)= 1.13, p=0.27 |
| Reading spanc | 45.7 (5.8) | 39.4 (5.0) | t(34) = 3.42, p<0.01 |
| CPZ Equivalentd | 432 (287.8) | ||
| SAPSe | 15.2 (15.8) | ||
| SANSf | 15.5 (14.2) |
Premorbid IQ was assessed using the North American Adult Reading Test: NAART (94).
Parental socio-economic status (SES) was calculated using the Hollingshead Index (95). Two controls and two patients did not provide parental occupation.
The reading span task constituted 60 one-clause, 5-word sentences. Reading span was operationalized as the total number of words recalled (following (32, 34)).
Chlorpromazine (CPZ) Equivalents were calculated following the International Consensus Study of Antipsychotic Dosing (96).
SAPS: Scale for the Assessment of Positive Symptoms (77). Summary scores (sum of the global ratings) are given.
SANS: Scale for the Assessment of Negative Symptoms (78). Summary scores (sum of the global ratings) are given.
Stimulus Presentation and Task
ERP data collection took place at Tufts University. Participants sat in a dimly-lit room while sentences were presented word-by-word (see Figure 1 and legend for details). Their task was to press either one of two buttons after seeing a “?” cue to indicate whether they judged each sentence to be acceptable or not. Participants were given examples of each type of sentence, and 10 practice trials.
Figure 1. Trial presentation.
The words were presented in white Arial font on a black background, with a viewing distance of 125cm and a visual angle of 2 degrees. Each trial began with a fixation cross followed by the sentence, presented word by word. The fixation cross and each word was presented for 400ms with an interstimulus interval of 150ms. The sentence-final word appeared with a period and was also presented for 400ms but was followed by a 700ms interstimulus interval, and then by a “?” which appeared in green font. This “?” cued participants to press either a “Yes” or “No” button, depending on whether they judged the sentence that they had just read was acceptable. After participants made their response, the word, “READY” in green font appeared on the screen. Participants then pressed a button to move them on to the next trial.
ERP Recording
Twenty-nine electrodes were secured on the scalp surface by an elastic cap (Electro-Cap International, Inc., Eaton, OH), see Figure 2. Electrodes were also attached below the left eye and at the outer canthus of the right eye, to monitor vertical and horizontal eye movements, and on the left and right mastoids. Impedances were kept below 10 kΏ for the eyes and below 2.5 kΏ at other sites. The EEG signal was amplified by an Isolated Bioelectric Amplifier System Model HandW-32/BA (SA Instrumentation Co., San Diego, CA) with a bandpass of 0.01–40 Hz, was continuously sampled at 200 Hz by an analogue-to-digital converter and was referenced online to the left mastoid.
Figure 2. Electrode montage with regions used for analysis.
For the purposes of statistical analyses, the scalp was divided into three-electrode regions. Regions in dark gray were part of the mid-regions omnibus ANOVA and regions in light gray were part of the peripheral regions omnibus ANOVA.
Behavioral Data Analysis
We considered accurate responses as being “Yes” following the matching sentences and “No” following the mismatching/violating sentences. A priori, we expected that these classifications would be more straightforward and less ambiguous for the 1-referent than the 2-referent sentences, and so we set a threshold such that if any participant responded with less than 60% accuracy in either of these two conditions, they were excluded. For included participants, the percentage of accurate responses was entered into a 4-way ANOVA with Sentence Type as a within-subjects factor and Group as a between-subjects factor. Significant effects were followed up with planned comparisons.
ERP Data analysis
ERPs were averaged offline at each electrode site in each experimental condition. Trials contaminated by eye artifact or amplifier blockage were excluded. If, after artifact rejection, a participant had fewer than 18 remaining items in any one of the four conditions, their dataset was excluded. Of the participants included in the final analysis, artifact contamination led to the rejection of 18.7% of trials in the schizophrenia group and 18.3% of trials in the control group (no between-group difference, p>.10).
Analyses were conducted on the mean amplitudes of ERPs evoked by the pronouns, regardless of participants' judgments. In the schizophrenia group, but not the control group, examination of the waveforms using pre-stimulus (pre-pronoun) baselines of -200-0ms and -100-0ms revealed a divergence between ERPs evoked by the 1-referent matching pronouns and the pronouns in the other three conditions within the first 100ms after pronoun onset. We attribute this to artifact in the pre-stimulus baseline period.4 To reduce the chance that these early differences in the patient group would confound our planned between-group analyses of later effects, we chose to correct for them by carrying out all planned analyses using a post-stimulus baseline of 0-100ms in all four conditions in both groups. However, to check that that this did not lead us to falsely infer significant late effects, we carried out selected post-hoc analyses to determine whether effects remained significant when using a -200-0ms pre-stimulus baseline.
Planned ANOVAs focused on two time windows: 400-600ms and 600-800ms. Previous studies show that the Nref effect in healthy controls consistently falls within the first time window, sometimes extending to the second (30-34). The 600-800ms window was selected on the basis on previous studies showing that it captures the late positivity/P600 effect, while minimizing component overlap from earlier negativity effects (e.g. (81)).
To gain comprehensive coverage of the scalp, we included Region and Hemisphere as spatial factors in omnibus ANOVAs that contrasted the 1-referent matching sentences with each of the three other sentence types, between the two groups.5 For each contrast, we carried out two omnibus ANOVAs, covering mid-regions (Figure 2, dark gray) and peripheral regions (Figure 2, light gray). In mid-regions analyses, the within-subject factors were Sentence Type and Region (prefrontal, frontal, central, parietal, and occipital), and the between-subject factor was Group (control, schizophrenia). In peripheral regions analyses, we included an additional within-subject factor, Hemisphere. In these omnibus analyses, we report only main effects and interactions involving Sentence Type, as we were primarily interested in group differences in how ERPs were modulated across conditions. Alpha was set to .05 for planned comparisons.6 A Greenhouse-Geisser correction was applied to analyses with more than one degree of freedom in the numerator (original degrees of freedom with corrected p values are reported).
Exploratory correlations between ERP effects of interest and (a) WM scores within both the control and patient groups, and (b) selected clinical measures within the schizophrenia group are reported in Supplementary Material.
Results
Behavioral data
People with schizophrenia performed less accurately overall than controls (controls: 92%, SD: 7.4; patients: 83%, SD: 12.3; main effect of Group, F(1,34)=7.33, p<.01), but the pattern of accuracy across the four types of sentences did not differ between the two groups (no interactions between Group and Sentence Type, Fs<0.6, ps >.4). Across both groups, there was a significant effect of Sentence Type, F(3, 102) = 7.77, p < .0001, due to lower accuracies in classifying the 1-referent mismatching sentences (84.8%, SD: 12.2) than the 1-referent matching sentences (91.5%, SD 9.8), F(1,34) = 16.20, p<.0003. There were no significant differences in accuracy between the 2-referent matching sentences (85.8%, SD: 13.5) and the 2-referent mismatching sentences (88.9%, SD: 12.4), F(1,34) = 3.20, p > .08.
ERP data
2-referent matching versus 1-referent matching pronouns (Figure 3)
Figure 3. Contrast between 1-referent matching and 2-referent matching pronouns.
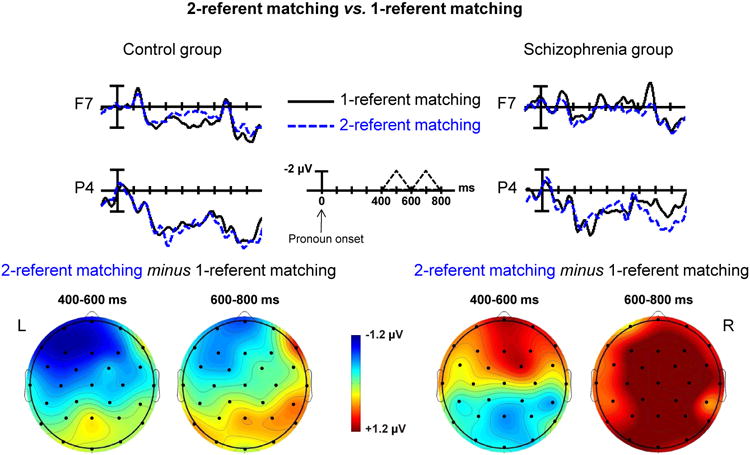
Top: Grand-averaged waveforms evoked by pronouns in the control and schizophrenia groups at a left frontal site (F7) and a right parietal site (P4). Solid black lines indicate ERPs to the 1-referent matching pronouns; dotted blue lines indicate ERPs to the 2-referent matching pronouns. Negative voltage is plotted upwards for all ERP plots.
Bottom: Voltage maps show differences between ERPs evoked by the pronouns in these two conditions between 400-600ms and between 600-800ms. In the 400-600ms time window, an Nref effect was seen in the control group but not in the schizophrenia group. In the 600-800ms time window, a late positivity effect was seen in the schizophrenia group but not in the control group.
In healthy controls, but not people with schizophrenia, the waveforms evoked by the 2-referent matching and the 1-referent matching pronouns appeared to diverge between 400-600ms, particularly at left-lateralized frontal sites. In keeping with previous studies in healthy controls (e.g. (29, 30, 32, 34)), we interpret this as an Nref effect.
Group differences in this effect were reflected by three-way interactions between Group, Sentence Type and Region (mid-regions: F(4, 136) = 4.26, p < .05; peripheral: F(1, 34) = 6.05, p < .05). In controls, follow-ups revealed a three-way interaction between Sentence Type, Region and Hemisphere in the peripheral regions ANOVA (F(1, 19) = 4.78, p < .05), due to a significant interaction between Region and Sentence Type over the left hemisphere (F(1,19) = 5.36, p<.05), but no effects over the right hemisphere (p > .7), reflecting the left lateralized distribution of the Nref effect. Post-hoc tests within the left frontal region using a -200-0ms baseline revealed significant effects at all sites. In the schizophrenia group, there were no main effects or interactions involving Sentence Type (all ps > .10).
In the 600-800ms window, healthy controls did not appear to show any ERP modulation. However, people with schizophrenia appeared to produce a larger positivity to the 2-referent matching than the 1-referent matching pronouns. This group difference was reflected by interactions between Group and Sentence Type (mid-regions: F(1,34)=5.78, p<.05; peripheral regions, marginal: F(1,34)=2.72, p<.10). In the schizophrenia group, simple effects ANOVAs revealed a larger widespread positivity to the 2-referent matching than the 1-referent matching pronouns (main effect of Sentence Type at mid-regions: F(1, 15) = 5.43, p < .05; peripheral regions, marginal: F(1, 15) =3.82, p=.07), see also Supplementary Materials for results of a Mass Univariate Analysis. Post-hoc tests within parietal and occipital mid-regions using a -200-0ms baseline revealed significant effects at all sites. No such differences were observed in the controls (no main effects of Sentence Type in either the mid-regions or peripheral regions analyses (F(1,19) = 0.14, p > .7 and F(1,19) = 0.25, p > .6, respectively).
1-referent mismatching versus 1-referent matching pronouns (Figure 4A)
Figure 4. Contrasts between 1-referent matching and the two types of mismatching pronouns.

A. 1-referent mismatching vs. 1-referent matching. Grand-averaged waveforms evoked by pronouns in the control and schizophrenia groups at Cz and Pz. Solid black lines indicate ERPs to the 1-referent matching pronouns; dotted red lines indicate ERPs to the 1-referent mismatching pronouns. Negative voltage is plotted upwards for all ERP plots. Voltage maps show differences between ERPs evoked by the 1-referent mismatching and 1-referent matching pronouns between 400-600ms and 600-800mec. B. 2-referent mismatching vs. 1-referent matching. Grand-averaged waveforms evoked by pronouns in the control and schizophrenia groups at Cz and Pz. Solid black lines indicate ERPs to the 1-referent matching pronouns; dashed green lines indicate ERPs to the 2-referent mismatching pronouns. Negative voltage is plotted upwards for all ERP plots. Voltage maps show differences between ERPs evoked by the 2-referent mismatching and 1-referent matching pronouns between 400-600ms and 600-800ms.
Between 400-600ms, there was a main effect of Sentence Type across both groups at mid-regions, F(1, 34) = 6.08, p < .05, and in the later 600-800ms later time window, this effect approached significance at mid-regions: F(1, 34) = 3.24, p = .08. As shown in Figure 4A, this contrast was associated with a positivity effect in both groups, although the scalp distribution of these positivity effects differed between the two groups (for full report, see Supplementary Materials).
2-referent mismatching versus 1-referent matching pronouns (Figure 4B)
Between 400-600ms and 600-800ms, there was a main effect of Sentence Type across both groups (400-600ms: mid-regions: F(1, 34) = 5.34, p < .05; peripheral regions: F(1, 34) = 4.35, p < .05, 600-800ms: mid-regions, F(1, 34) = 4.14, p < .05, and peripheral regions, F(1, 34) = 6.23, p < .05). As shown in Figure 4A, this contrast was associated with a positivity effect in both groups, although again the scalp distribution of these positivity effects differed between the two groups. Moreover, in the control but not the schizophrenia group, it was accompanied by an anteriorly distributed negativity effect (see Supplementary Materials for full report and discussion).
Discussion
We exploited the excellent temporal resolution of ERPs to investigate the timing and nature of proactive and reactive mechanisms of establishing referential cohesion during language comprehension in healthy adults and people with schizophrenia. Controls produced a larger left anteriorly-distributed negativity between 400-600ms to 2-referent matching than to 1-referent matching pronouns (the “Nref effect”). In contrast, people with schizophrenia failed to produce this effect, but instead produced a later positivity effect between 600-800ms. Both groups produced a larger positivity between 400-800ms to pronouns that mismatched versus matched the gender of their referent(s).
In controls, modulation of the Nref is thought to reflect the difficulty of linking an anaphor to a preceding referent. Upon encountering a pronoun that is consistent with a referent that has been pre-activated/predicted with high probability, it is easier to establish referential cohesion and a smaller (less negative) Nref is produced. If additional competing referents have also been pre-activated, however, it is harder to select the appropriate referent and establish referential cohesion, and a larger Nref is produced (for a related interpretation of other late negativity effects, see (39, 40, 81), and Supplement for further discussion). In schizophrenia, the reduced Nref modulation suggests that these proactive mechanisms of establishing referential cohesion are impaired. This may be because, in the 2-referent matching sentences, patients failed to use context to pre-activate or proactively maintain both referents over a delay, leading to less competition upon encountering the anaphor. Alternatively, they may have found it relatively difficult to process the 1-referent matching pronouns, either because they failed to pre-activate the single referent with high probability or because they failed to use the incoming pronoun to select this referent.7
In contrast, reactive referential mechanisms appeared to be relatively spared in schizophrenia. Like controls, people with schizophrenia produced a larger late positivity to gender mismatching than matching pronouns (although with different scalp distributions, see Supplementary Materials for full report and discussion). We suggest that, upon encountering the pronoun, patients engaged in initial attempts to link it to a referent within the immediate context, and, like controls, they perceived the mismatch in gender between the pronoun and its referent(s) (32, 59, 60), reflected by the positivity between 400-600ms. This then led to prolonged retroactive attempts to establish coherence (52, 53), perhaps through attempts to retrieve novel referents (61, 62) from episodic memory, outside the focus of attention, thereby updating the current representation of context (56, 57), as reflected by the positivity between 600-800ms.
In addition to producing a positivity effect to the gender mismatching (versus matching) pronouns, people with schizophrenia, unlike the controls, also produced a positivity effect between 600-800ms to the 2-referent (versus 1-referent) pronouns. In other words, they produced this later positivity effect instead of the earlier Nref effect (400-600ms). This suggests not only that reactive mechanisms of establishing referential coherence were relatively preserved, but that patients employed these later mechanisms to compensate for their proactive deficits. This finding is strikingly consistent with the results of a previous ERP study of discourse comprehension in schizophrenia where, instead of influencing modulation of an earlier ERP component (in that case, the N400), the discourse context influenced modulation of a late positivity between 700-1000ms (72). It is also in line with other findings suggesting that, although a predictive use of context is impaired in schizophrenia, given enough time, patients can still extract information within a discourse context to inform their final interpretations (73). Finally, it provides evidence against the argument that the absent Nref effect in the schizophrenia group simply reflected a failure to engage with the task or understand instructions.
Our findings are consistent with the theory that schizophrenia is characterized by deficits of proactive cognitive control and WM (3, 4). As shown in Table 2, verbal WM span scores in the schizophrenia group were significantly smaller than in the controls, although, unlike in previous studies in healthy adults (32, 34), there were no significant correlations between the Nref and WM spans in either group, likely due to restricted individual variability (see Supplementary Materials). More generally, they are consistent with the idea that predictive impairments in schizophrenia can explain and unify multiple abnormalities of language comprehension and production (86). Of course, it will be important to replicate these findings with a larger sample size, and to determine whether referential impairments vary across subpopulations within schizophrenia.
Our findings also have social implications. Some researchers have discussed referential impairments in schizophrenia as stemming from a social communicative failure — a failure to take the communicator's assumptions into account (7, 8, 87), see also (88, 89). In practice, however, given the speed of everyday communication, establishing and drawing upon common referents in real time will depend largely on the speed at which both the comprehender and producer can access relevant information from memory (see (90-93)). In healthy individuals, this relies heavily upon the ability to predictively mobilize contextual information to constrain processing of new inputs (see (20) for a review). Although it is encouraging that people with schizophrenia can engage reactive mechanisms to compensate for proactive deficits, reactive mechanisms tend to be slower and less efficient than predictive processing, and so in most situations, there may not be enough time to engage them and still keep up with the fast pace of everyday conversation. Thus, our findings may, at least in part, help explain why patients struggle with social communicative interactions.
Supplementary Material
Table S1. Between-group analysis. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type and Group, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows.
Table S2. Control group. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows. Follow-ups of contrasts are also shown in individual regions.
Table S3. Schizophrenia group. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows. Follow-ups of contrasts are also shown in individual regions.
Acknowledgments
This work was funded by the National Institute of Mental Health (R01MH071635 to G.R.K.), the Brain and Behavior Research Foundation (Young Investigator NARSAD to T. D.), as well as the Sidney R. Baer, Jr. Foundation who supported undergraduate students, including Gena Gorlin and Marissa Lipton, who contributed to data collection and other aspects of the project. We thank Marianna Eddy and Kana Okano who contributed to data collection. We are grateful to Donald Goff for his support in patient recruitment, and to Ted Alexander for help with analysis. We also thank Meredith Brown and Lotte Schoot for their insightful comments on the manuscript.
Footnotes
An anaphor is an expression whose interpretation depends upon another expression in the context. For example, in the sentence, “Edward took courses in accounting but he didn't learn much”, “he” is an anaphor that refers to the preceding referent, “Edward”. If the comprehender links an anaphor to the particular referent(s) that was intended by the producer, “referential cohesion” is said to be established.
By prediction, we refer to an implicit, probabilistic process of pre-activating upcoming information, at multiple levels of representation, in a graded fashion and at least partially in parallel, rather than to a strategic or all-or-nothing mechanism (20).
Note that, in this example, “he” is referentially ambiguous. While the Nref is classically associated with referential ambiguities, it can also be evoked by non-ambiguous anaphors (e.g. (34, 35)). Note also that similar anteriorly-distributed negativities are also associated with maintaining and/or attempting to select other types of predicted entities from competing pre-activated alternatives, including individual lexical items (36, 37), syntactic structures (38), event structures (39-44), and types of interpretation ((45-47), Experiment 1), see Supplementary Materials for further discussion of the functional significance of this effect.
This differential activity between 0-100ms in the schizophrenia group is unlikely to have reflected differences across conditions in perceptual processing of the pronouns, as these were identical across conditions. Similarly, differential activity within the pre-stimulus period is unlikely to have reflected true differences in anticipatory activity across these conditions prior to the onset of the critical pronoun because, by design, the words that preceded the pronouns were identical between the matching and mismatching 1-referent sentences, and between the matching and mismatching 2-referent sentences.
An alternative approach for comparing effects between patients and controls would have been to carry out analyses only within spatial regions of interest where we expected to see maximal effects in the control group. This, however, would have potentially introduced Type II error. For example, while we had strong a priori hypotheses that the healthy controls would produce an Nref effect in the left frontal region, we were reluctant to compare controls and patients only within this region. This is because any group differences in the effect identified here might lead us to infer that the Nref effect is reduced in patients compared to controls. However, given that the Nref effect is not necessarily or always confined just to left frontal regions, this approach would exclude the possibility that patients did in fact produce an Nref effect over other frontal regions that were not examined at all. An omnibus ANOVA that includes spatial factors reduces this possibility. On the other hand, it comes at a cost: the number of tests and comparisons entailed means that any unpredicted effects at p < 0.05 within the schizophrenia group may be due to Type I error (82). See footnote 6 and Supplementary Materials for our approach to guard against this possibility.
In the control group, we had strong a priori hypotheses about the location and nature of the ERP effects: we expected to see an Nref effect in comparing the 1-referent matching and the 2-referent matching pronouns within the left frontal region, and we expected to see a widespread late positivity effect in comparing the 1-referent matching pronouns with each of the two types of mismatching pronouns. Thus, for planned comparisons within these regions in the control group, an alpha of p < 0.05 is appropriate (as explained in footnote 3, if we had just considered the controls, an omnibus test was not strictly necessary: we could have proceeded straight to these planned comparisons to test these effects). An alpha level, however, is less appropriate for testing the significance of less well characterized effects in the schizophrenia group, which were only resolved through hierarchical follow-up of our omnibus ANOVAs. This is because, although our approach allowed for full coverage of the scalp, it created multiple opportunities to detect effects, which increased the probability of Type I error (82). This is particularly problematic for interpreting less well-characterized ERP effects. To address this issue, we supplemented our approach by carrying out a mass univariate analysis, in combination with a cluster-based permutation test (83, 84) to account for multiple comparisons, in the schizophrenia group. Details of these methods and results are reported in the Supplementary Material.
Another way of conceptualizing this is to posit that, in controls, the Nref effect was primarily driven by a left anterior positive-going waveform that was evoked by the 1-referent matching pronouns when reference was successfully established. On this account, the absent Nref effect in the schizophrenia group resulted from a failure to produce this positivity to the 1-referent matching pronouns, due to failure to quickly establish reference. This account is consistent with the pattern of waveforms across conditions, which does indeed suggest a smaller positivity to the 1-referent matching pronouns in the schizophrenia than the control group (see Figure 3). On the other hand, given the difficulty of interpreting the absolute polarity of any ERP response (in relation to baseline) to individual conditions, particularly in comparing patients and controls (see (85), pages 73-74 for discussion), this observation should be interpreted with caution.
Financial Disclosures: All authors report no biomedical financial interests or potential conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Clark HH, Marshall CR. Definite reference and mutual knowledge. In: JA K, WB L, SI A, editors. Elements of Discourse Understanding. Cambridge: Cambridge University Press; 1981. [Google Scholar]
- 2.Clark HH, Murphy GL. Audience design in meaning and reference. Advances in psychology. 1982;9:287–299. [Google Scholar]
- 3.Barch DM, Smith E. The cognitive neuroscience of working memory: Relevance to CNTRICS and schizophrenia. Biological Psychiatry. 2008;64:11–17. doi: 10.1016/j.biopsych.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barch DM, Ceaser A. Cognition in schizophrenia: core psychological and neural mechanisms. Trends in Cognitive Sciences. 2012;16:27–34. doi: 10.1016/j.tics.2011.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rochester S, Martin JR. Crazy Talk: A Study of the Discourse of Schizophrenic Speakers. New York: Plenum Press; 1979. [Google Scholar]
- 6.Docherty NM, DeRosa M, Andreasen NC. Communication disturbances in schizophrenia and mania. Archives of General Psychiatry. 1996;53:358–364. doi: 10.1001/archpsyc.1996.01830040094014. [DOI] [PubMed] [Google Scholar]
- 7.Hoffman RE, Kirstein L, Stopek S, Cicchetti DV. Apprehending schizophrenic discourse: A structural analysis of the listener's task. Brain and Language. 1982;15:207–233. doi: 10.1016/0093-934x(82)90057-8. [DOI] [PubMed] [Google Scholar]
- 8.Hoffman RE. Tree structures, the work of listening, and schizophrenic discourse: A reply to Beveridge and Brown. Brain and Language. 1986;27:385–392. doi: 10.1016/0093-934x(86)90027-1. [DOI] [PubMed] [Google Scholar]
- 9.Buck B, Penn DL. Lexical characteristics of emotional narratives in schizophrenia: Relationships with symptoms, functioning, and social cognition. The Journal of Nervous and Mental Disease. 2015;203:702–708. doi: 10.1097/NMD.0000000000000354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saavedra J. Quantitative criteria of narrative coherence and complexity in persons with paranoid schizophrenia. The Journal of Nervous and Mental Disease. 2010;198:349–355. doi: 10.1097/NMD.0b013e3181da47f5. [DOI] [PubMed] [Google Scholar]
- 11.Noel-Jorand MC, Reinert M, Giudicelli S, Dassa D. A new approach to discourse analysis in psychiatry, applied to a schizophrenic patient's speech. Schizophrenia Research. 1997;25:183–198. doi: 10.1016/s0920-9964(97)00022-4. [DOI] [PubMed] [Google Scholar]
- 12.Docherty NM, Cohen AS, Nienow TM, Dinzeo TJ, Dangelmaier RE. Stability of formal thought disorder and referential communication disturbances in schizophrenia. Journal of Abnormal Psychology. 2003;112:469–475. doi: 10.1037/0021-843X.112.3.469. [DOI] [PubMed] [Google Scholar]
- 13.Docherty N, Schnur M, Harvey PD. Reference performance and positive and negative thought disorder: A follow-up study of manics and schizophrenics. Journal of Abnormal Psychology. 1988;97:437–442. doi: 10.1037//0021-843x.97.4.437. [DOI] [PubMed] [Google Scholar]
- 14.Buck B, Minor KS, Lysaker PH. Differential lexical correlates of social cognition and metacognition in schizophrenia; a study of spontaneously-generated life narratives. Comprehensive Psychiatry. 2015;58:138–145. doi: 10.1016/j.comppsych.2014.12.015. [DOI] [PubMed] [Google Scholar]
- 15.Ditman T, Kuperberg GR. Building coherence: A framework for exploring the breakdown of links across clause boundaries in schizophrenia. Journal of Neurolinguistics. 2010:254–269. doi: 10.1016/j.jneuroling.2009.03.003. “Language in Schizophrenia” Special Issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ditman T, Goff D, Kuperberg GR. Slow and steady: sustained effects of lexicosemantic associations can mediate referential impairments in schizophrenia. Cogn Affect Behav Neurosci. 2011;11:245–258. doi: 10.3758/s13415-011-0020-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hogarty GE, Flesher S. Practice principles of cognitive enhancement therapy for schizophrenia. Schizophrenia Bulletin. 1999;25:693–708. doi: 10.1093/oxfordjournals.schbul.a033411. [DOI] [PubMed] [Google Scholar]
- 18.Patterson TL, Moscona S, McKibbin CL, Davidson K, Jeste DV. Social skills performance assessment among older patients with schizophrenia. Schizophrenia Research. 2001;48:351–360. doi: 10.1016/s0920-9964(00)00109-2. [DOI] [PubMed] [Google Scholar]
- 19.Roberts DL, Penn DL. Social cognition and interaction training (SCIT) for outpatients with schizophrenia: a preliminary study. Psychiatry Research. 2009;166:141–147. doi: 10.1016/j.psychres.2008.02.007. [DOI] [PubMed] [Google Scholar]
- 20.Kuperberg GR, Jaeger TF. What do we mean by prediction in language comprehension? Lang Cogn Neurosci. 2016;31:32–59. doi: 10.1080/23273798.2015.1102299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kehler A. Coherence, reference, and the theory of grammar. CSLI publications; Stanford: 2002. [Google Scholar]
- 22.Kehler A, Kertz L, Rohde H, Elman JL. Coherence and coreference revisited. Journal of Semantics. 2008;25:1–44. doi: 10.1093/jos/ffm018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hartshorne JK, O'Donnell TJ, Tenenbaum JB. The causes and consequences explicit in verbs. Language, Cognition and Neuroscience. 2015;30:716–734. doi: 10.1080/23273798.2015.1008524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kuperberg GR. Separate streams or probabilistic inference? What the N400 can tell us about the comprehension of events. Language, Cognition and Neuroscience. 2016;31:602–616. doi: 10.1080/23273798.2015.1130233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arnold JE. The effect of thematic roles on pronoun use and frequency of reference continuation. Discourse Processes. 2001;31:137–162. [Google Scholar]
- 26.Arnold JE, Brown-Schmidt S, Trueswell J. Children's use of gender and order-of-mention during pronoun comprehension. Language and Cognitive Processes. 2007;22:527–565. [Google Scholar]
- 27.Hobbs JR. Coherence and coreference. Cognitive Science. 1979;3:67–90. [Google Scholar]
- 28.Nieuwland MS, Van Berkum JJA. The neurocognition of referential ambiguity in language comprehension. Language and Linguistics Compass. 2008;2:603–630. [Google Scholar]
- 29.Van Berkum JJA, Brown CM, Hagoort P. Early referential context effects in sentence processing: Evidence from event-related brain potentials. Journal of Memory and Language. 1999;41:147–182. [Google Scholar]
- 30.Van Berkum JJA, Brown CM, Hagoort P, Zwitserlood P. Event-related brain potentials reflect discourse-referential ambiguity in spoken language comprehension. Psychophysiology. 2003;40:235–248. doi: 10.1111/1469-8986.00025. [DOI] [PubMed] [Google Scholar]
- 31.Anderson JE, Holcomb PJ. An electrophysiological investigation of the effects of coreference on word repetition and synonymy. Brain and Language. 2005;94:200–216. doi: 10.1016/j.bandl.2005.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nieuwland MS, Van Berkum JJA. Individual differences and contextual bias in pronoun resolution: Evidence from ERPs. Brain Research. 2006;1118:155–167. doi: 10.1016/j.brainres.2006.08.022. [DOI] [PubMed] [Google Scholar]
- 33.Nieuwland MS, Otten M, Van Berkum JJA. Who are you talking about? Tracking discourse-level referential processes with ERPs. Journal of Cognitive Neuroscience. 2007;19:1–9. doi: 10.1162/jocn.2007.19.2.228. [DOI] [PubMed] [Google Scholar]
- 34.Nieuwland M. “Who's he?” Event-related brain potentials and unbound pronouns. Journal of Memory and Language. 2014;76:1–28. [Google Scholar]
- 35.Almor A, Nair VA, Boiteau TW, Vendemia JM. The N400 in processing repeated name and pronoun anaphors in sentences and discourse. doi: 10.1016/j.bandl.2017.06.003. Under review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wlotko EW, Federmeier KD. So that's what you meant! Event-related potentials reveal multiple aspects of context use during construction of message-level meaning. NeuroImage. 2012;62:356–366. doi: 10.1016/j.neuroimage.2012.04.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wlotko EW, Federmeier KD. Age-related changes in the impact of contextual strength on multiple aspects of sentence comprehension. Psychophysiology. 2012;49:770–785. doi: 10.1111/j.1469-8986.2012.01366.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kluender R, Kutas M. Bridging the gap: Evidence from ERPs on the processing of unbounded dependencies. Journal of Cognitive Neuroscience. 1993;5:196–214. doi: 10.1162/jocn.1993.5.2.196. [DOI] [PubMed] [Google Scholar]
- 39.Paczynski M, Jackendoff R, Kuperberg GR. When events change their nature: the neurocognitive mechanisms underlying aspectual coercion. Journal of Cognitive Neuroscience. 2014;26:1905–1917. doi: 10.1162/jocn_a_00638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wittenberg E, Paczynski M, Wiese H, Jackendoff R, Kuperberg GR. The difference between “giving a rose” and “giving a kiss”: A sustained anterior negativity to the light verb construction. Journal of Memory and Language. 2014;73:31–42. doi: 10.1016/j.jml.2014.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Baggio G, van Lambalgen M, Hagoort P. Computing and recomputing discourse models: An ERP study. Journal of Memory and Language. 2008;59:36–53. [Google Scholar]
- 42.Bott O. The Processing of Events. Philadelphia, USA: John Benjamins Publishing; 2010. [Google Scholar]
- 43.Ferretti TR, McRae K, Hatherell A. Integrating verbs, situation schemas, and thematic role concepts. Journal of Memory and Language. 2001;44:516–547. [Google Scholar]
- 44.Lee CL, Federmeier KD. Wave-ering: An ERP study of syntactic and semantic context effects on ambiguity resolution for noun/verb homographs. Journal of Memory and Language. 2009;61:538–555. doi: 10.1016/j.jml.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Coulson S, Van Petten C. A special role for the right hemisphere in metaphor comprehension? ERP evidence from hemifield presentation. Brain Research. 2007;1146:128–145. doi: 10.1016/j.brainres.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 46.Coulson S. Semantic Leaps: Frame Shifting and Conceptual Blending in Meaning Construction. New York: Cambridge University Press; 2001. [Google Scholar]
- 47.Nieuwland MS, Van Berkum JJA. When peanuts fall in love: N400 evidence for the power of discourse. Journal of Cognitive Neuroscience. 2006;18:1098–1111. doi: 10.1162/jocn.2006.18.7.1098. [DOI] [PubMed] [Google Scholar]
- 48.Gerrig RJ, Horton WS, Stent A. Production and comprehension of unheralded pronouns: A corpus analysis. Discourse Processes. 2011;48:161–182. [Google Scholar]
- 49.Gordon P, Hendrick R. The representation and processing of coreference in discourse. Cognitive science. 1998;22:389–424. [Google Scholar]
- 50.Osterhout L, Holcomb PJ. Event-related potentials elicited by syntactic anomaly. Journal of Memory and Language. 1992;31:785–806. [Google Scholar]
- 51.Kuperberg GR, Sitnikova T, Caplan D, Holcomb PJ. Electrophysiological distinctions in processing conceptual relationships within simple sentences. Cognitive Brain Research. 2003;17:117–129. doi: 10.1016/s0926-6410(03)00086-7. [DOI] [PubMed] [Google Scholar]
- 52.Kuperberg GR. Neural mechanisms of language comprehension: Challenges to syntax. Brain Research. 2007;1146:23–49. doi: 10.1016/j.brainres.2006.12.063. [DOI] [PubMed] [Google Scholar]
- 53.Osterhout L, Holcomb PJ, Swinney DA. Brain potentials elicited by garden-path sentences: Evidence of the application of verb information during parsing. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1994;20:786–803. doi: 10.1037//0278-7393.20.4.786. [DOI] [PubMed] [Google Scholar]
- 54.Osterhout L, Bersick M, McLaughlin J. Brain potentials reflect violations of gender stereotypes. Memory and Cognition. 1997;25:273–285. doi: 10.3758/bf03211283. [DOI] [PubMed] [Google Scholar]
- 55.Paczynski M, Kuperberg GR. Multiple influences of semantic memory on sentence processing: Distinct effects of semantic relatedness on violations of real-world event/state knowledge and animacy selection restrictions. Journal of Memory and Language. 2012;67:426–448. doi: 10.1016/j.jml.2012.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kuperberg GR. The proactive comprehender: What event-related potentials tell us about the dynamics of reading comprehension. In: Miller B, Cutting L, McCardle P, editors. Unraveling Reading Comprehension: Behavioral, Neurobiological, and Genetic Components. Baltimore, MD: Paul Brookes Publishing; 2013. pp. 176–192. [Google Scholar]
- 57.Kaan E, Dallas AC, Barkley CM. Processing bare quantifiers in discourse. Brain Research. 2007;1146:199–209. doi: 10.1016/j.brainres.2006.09.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee J, Park S. Working memory impairments in schizophrenia: A meta-analysis. Journal of Abnormal Psychology. 2005;114:599–611. doi: 10.1037/0021-843X.114.4.599. [DOI] [PubMed] [Google Scholar]
- 59.Osterhout L, Mobley LA. Event-related brain potentials elicited by failure to agree. Journal of Memory and Language. 1995;34:739–773. [Google Scholar]
- 60.Van Berkum JJA, Koornneef AW, Otten M, Nieuwland MS. Establishing reference in language comprehension: An electrophysiological perspective. Brain Research. 2007;1146:158–171. doi: 10.1016/j.brainres.2006.06.091. [DOI] [PubMed] [Google Scholar]
- 61.Burkhardt P. Inferential bridging relations reveal distinct neural mechanisms: Evidence from event-related brain potentials. Brain and Language. 2006;98:159–168. doi: 10.1016/j.bandl.2006.04.005. [DOI] [PubMed] [Google Scholar]
- 62.Burkhardt P. The P 600 reflects cost of new information in discourse memory. NeuroReport. 2007;18:1851–1854. doi: 10.1097/WNR.0b013e3282f1a999. [DOI] [PubMed] [Google Scholar]
- 63.Braver TS. The variable nature of cognitive control: a dual mechanisms framework. Trends Cogn Sci. 2012;16:106–113. doi: 10.1016/j.tics.2011.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lesh TA, Westphal AJ, Niendam TA, Yoon JH, Minzenberg MJ, Ragland JD, et al. Proactive and reactive cognitive control and dorsolateral prefrontal cortex dysfunction in first episode schizophrenia. NeuroImage: Clinical. 2013;2:590–599. doi: 10.1016/j.nicl.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Harvey PD, Earle-Boyer EA, Levinson JC. Distractibility and discourse failure their association in mania and schizophrenia. The Journal of Nervous and Mental Disease. 1986;174:274–279. doi: 10.1097/00005053-198605000-00003. [DOI] [PubMed] [Google Scholar]
- 66.Docherty NM, Hawkins KA, Hoffman RE, Quinlan DM, Rakfeldt J, Sledge WH. Working memory, attention, and communication disturbances in schizophrenia. Journal of Abnormal Psychology. 1996;105:212–219. doi: 10.1037//0021-843x.105.2.212. [DOI] [PubMed] [Google Scholar]
- 67.Docherty NM. Cognitive impairments and disordered speech in schizophrenia: Thought disorder, disorganization, and communication failure perspectives. Journal of Abnormal Psychology. 2005;114:269–278. doi: 10.1037/0021-843X.114.2.269. [DOI] [PubMed] [Google Scholar]
- 68.Docherty NM, Hall MJ, Gordinier SW, Cutting LP. Conceptual sequencing and disordered speech in schizophrenia. Schizophrenia Bulletin. 2000;26:723–735. doi: 10.1093/oxfordjournals.schbul.a033489. [DOI] [PubMed] [Google Scholar]
- 69.Bagner DM, Melinder MR, Barch DM. Language comprehension and working memory deficits in patients with schizophrenia. Schizophrenia Research. 2003;60:299–309. doi: 10.1016/s0920-9964(02)00280-3. [DOI] [PubMed] [Google Scholar]
- 70.Boudewyn MA, Carter CS, Swaab TY. Cognitive control and discourse comprehension in schizophrenia. Schizophr Res Treatment. 2012;2012:484–502. doi: 10.1155/2012/484502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Swaab TY, Boudewyn MA, Long DL, Luck SJ, Kring AM, Ragland JD, et al. Spared and impaired spoken discourse processing in schizophrenia: effects of local and global language context. Journal of Neuroscience. 2013;33:15578–15587. doi: 10.1523/JNEUROSCI.0965-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ditman T, Kuperberg GR. The time course of building discourse coherence in schizophrenia: An ERP investigation. Psychophysiology. 2007;44:991–1001. doi: 10.1111/j.1469-8986.2007.00565.x. [DOI] [PubMed] [Google Scholar]
- 73.Rabagliati H, Delaney-Busch N, Snedeker J, Kuperberg G. Spared bottom-up but impaired top-down interactive effects during language processing in schizophrenia. doi: 10.1017/S0033291718001952. Submitted. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.First M, Spitzer R, Miriam G, Williams J. Biometrics Research. New York: New York State Psychiatric Institute; 2002. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition (SCID-I/P) [Google Scholar]
- 75.Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9:97–113. doi: 10.1016/0028-3932(71)90067-4. [DOI] [PubMed] [Google Scholar]
- 76.White K, Ashton R. Handedness assessment inventory. Neuropsychologia. 1976;14:261–264. doi: 10.1016/0028-3932(76)90058-0. [DOI] [PubMed] [Google Scholar]
- 77.Andreasen NC. The Scale for the Assessment of Positive Symptoms (SAPS) Iowa City, IA: The University of Iowa; 1984. [Google Scholar]
- 78.Andreasen NC. Scale for the Assessment of Negative Symptoms (SANS) British Journal of Psychiatry. 1989;155:53–58. [PubMed] [Google Scholar]
- 79.Daneman M, Carpenter PA. Individual differences in working memory and reading. Journal of Verbal Learning and Verbal Behavior. 1980;19:450–466. [Google Scholar]
- 80.Just MA, Carpenter PA. A capacity theory of comprehension: individual differences in working memory. Psychological Review. 1992;99:122–149. doi: 10.1037/0033-295x.99.1.122. [DOI] [PubMed] [Google Scholar]
- 81.Xiang M, Kuperberg G. Reversing expectations during discourse comprehension. Language, Cognition, & Neuroscience. 2015;30:648–672. doi: 10.1080/23273798.2014.995679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Luck SJ, Gaspelin N. How to get statistically significant effects in any ERP experiment (and why you shouldn't) Psychophysiology. 2017;54:146–157. doi: 10.1111/psyp.12639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Maris E, Oostenveld R. Nonparametric statistical testing of EEG- and MEG-data. J Neurosci Meth. 2007;164:177–190. doi: 10.1016/j.jneumeth.2007.03.024. [DOI] [PubMed] [Google Scholar]
- 84.Groppe DM, Urbach TP, Kutas M. Mass univariate analysis of event-related brain potentials/fields I: a critical tutorial review. Psychophysiology. 2011;48:1711–1725. doi: 10.1111/j.1469-8986.2011.01273.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kuperberg GR, Kreher DA, Ditman T. What can event-related potentials tell us about language, and perhaps even thought, in schizophrenia? International Journal of Psychophysiology: Special Issue on Language and Psychophysiology. 2010;75:66–76. doi: 10.1016/j.ijpsycho.2009.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Brown M, Kuperberg GR. A hierarchical generative framework of language processing: Linking language perception, interpretation, and production abnormalities in schizophrenia. Frontiers in Human Neuroscience. 2015;9:643. doi: 10.3389/fnhum.2015.00643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rochester S, Martin JR, Thurston S. Thought-process disorder in schizophrenia: The listener's task. Brain and Language. 1977;4:95–114. doi: 10.1016/0093-934x(77)90009-8. [DOI] [PubMed] [Google Scholar]
- 88.Rutter DR. Language in schizophrenia. The structure of monologues and conversations. The British Journal of Psychiatry. 1985;146:399–404. doi: 10.1192/bjp.146.4.399. [DOI] [PubMed] [Google Scholar]
- 89.Harrow M, Lanin-Kettering I, Miller JG. Impaired perspective and thought pathology in schizophrenic and psychotic disorders. Schizophrenia Bulletin. 1989;15:605–623. doi: 10.1093/schbul/15.4.605. [DOI] [PubMed] [Google Scholar]
- 90.Horton WS, Gerrig RJ. The impact of memory demands on audience design during language production. Cognition. 2005;96:127–142. doi: 10.1016/j.cognition.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 91.Horton WS. Conversational common ground and memory processes in language production. Discourse Processes. 2005;40:1–35. [Google Scholar]
- 92.Metzing C, Brennan SE. When conceptual pacts are broken: Partner-specific effects on the comprehension of referring expressions. Journal of Memory and Language. 2003;49:201–213. [Google Scholar]
- 93.Hanna JE, Tanenhaus MK, Trueswell JC. The effects of common ground and perspective on domains of referential interpretation. Journal of Memory and Language. 2003;49:43–61. [Google Scholar]
- 94.Blair JR, Spreen O. Predicting premorbid IQ: A revision of the National Adult Reading Test. Clinical Neuropsychologist. 1989;3:129–136. [Google Scholar]
- 95.Hollingshead AB. Two factor index of social position. New Haven, CT: Yale University Press; 1965. [Google Scholar]
- 96.Gardner DM, Murphy AL, O'Donnell H, Centorrino F, Baldessarini RJ. International consensus study of antipsychotic dosing. American Journal of Psychiatry. 2010;167:686–693. doi: 10.1176/appi.ajp.2009.09060802. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Between-group analysis. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type and Group, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows.
Table S2. Control group. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows. Follow-ups of contrasts are also shown in individual regions.
Table S3. Schizophrenia group. Results of the mid-regions and peripheral regions omnibus ANOVAs showing effects of Sentence Type as well as interactions between Sentence Type, Region, and/or Hemisphere in contrasting the 1-referent matching pronouns with the 1-referent and 2-referent mismatching pronouns in the 400-600ms and 600-800ms time windows. Follow-ups of contrasts are also shown in individual regions.




