Abstract
Heart failure is a growing epidemic in the United States and throughout the world. The utilization of continuous-flow left ventricular assist devices (LVADs) has greatly increased over the last decade. In addition, a limited supply of organ donors has led to a rise in the use, and duration, of LVADs for destination therapy. The increased use of LVAD therapy has led to the observation of mechanical complications such as device thrombosis, de novo aortic insufficiency, and outflow graft stenosis, all of which are associated with prolonged LVAD support. Surgical repair for these complications remains the therapy of choice; however, surgery may be associated with high operative risk in some patients. The purpose of this article is to discuss mechanical complications associated with LVAD therapy and interventional transcatheter therapies that have been used to solve these increasingly complex problems.
Keywords: left ventricular assist device, LVAD, LVAD thrombosis, aortic insufficiency, outflow graft stenosis, HeartMate II, HeartWare
Heart failure has become a major public health epidemic, with approximately 5.1 million patients diagnosed with this condition in the United States alone; this is expected to increase by 25% by 2030.1 Left ventricular assist devices (LVADs) have become vital in the treatment of end-stage heart failure, with more than 12,000 implants between April 2008 and December 2014, and almost 2500 implants in 2014 alone.2 Given the relatively unchanged number of heart transplants over the last two decades, the number of patients receiving LVADs for treatment of end-stage heart failure is expected to continue to rise.3 Currently, there are two Food and Drug Administration (FDA)-approved continuous-flow LVADs (CF-LVADs) in the United States: HeartMate II (HMII; Thoratec), approved for bridge to transplant (BTT) and destination therapy (DT); and HVAD (HeartWare), approved for BTT only. 4 Since the FDA approval of HMII for DT in 2010, the proportion of patients receiving CF-LVADs for DT has risen significantly, peaking at 46% of all LVAD implantations in 2014. Importantly, the survival rates with CF-LVAD support remain excellent, with 1-year and 2-year survival rates at 80% and 70%, respectively, for both DT and BTT patients.2
The increasing use of CF-LVADs for DT, along with long transplant wait times,3,5 has led to a significant number of patients who are supported by CF-LVADs for an extended duration of time. The increased treatment time with CF-LVADs has resulted in a rise in complications associated with CF-LVAD support.2 The common complications associated with CF-LVAD therapy include gastrointestinal bleeding (event rates ranging from 0.27-0.67 gastrointestinal bleeds per patient-year of support,6-8 thromboembolic events (up to 0.17 events per patient-year of support despite anticoagulation, with up to 12% experiencing at least one event, 7,9 and infection (up to 12% over the life of the device).10 Mechanical complications, such as device thrombosis, de novo aortic insufficiency, and outflow graft stenosis, have also been encountered at increasing frequency. Surgery remains the therapy of choice for these mechanical complications; however, a subset of patients may have high operative risk, precluding them from undergoing surgical management. In this article, we will discuss the common mechanical complications associated with CF-LVAD support and interventional transcatheter therapies that have been used to solve these increasingly complex problems.
Aortic Insufficiency
Incidence
The development of aortic insufficiency (AI) has been recognized as an important complication of prolonged CF-LVAD therapy.11-13 AI while on CF-LVAD support reduces the efficiency of the LVAD as a portion of the forward flow from the LVAD outflow cannula enters back into the left ventricle via the regurgitant aortic valve, creating a circulatory loop with diminishing forward flow and, subsequently, decreasing end-organ perfusion.
The rates of mild or worse AI have been shown to be as high as 52% with CF-LVAD support, with numerous studies demonstrating that duration of CF-LVAD support and closed aortic valve are the strongest risk factors associated with development and progression of de novo AI.12-19 Pak et al showed that freedom from AI in patients with CF-LVADs was 75% at 12 months of support, with AI being more common in patients with aortic valves that failed to open.12 Similarly, in the study by Jorde et al, freedom from at least mild AI was 77% after 1 year of CF-LVAD support, with moderate AI developing in 38% of patients after 3 years of support.13 A persistently closed AV was again strongly associated with development of AI.13
Development of AI in patients with CF-LVADs often leads to clinical symptoms of heart failure and has been shown to reduce survival.13,17,18 Toda et al noted a statistically significant reduction in survival in patients with any degree of AI (2-year survival 93% vs 82%; P=.02).18 In their cohort, among patients who died with de novo AI after at least 2 years of LVAD support, 6 of the 7 deaths were secondary to development of heart failure.18 In the study by Jorde et al, 7 out of 21 patients that developed at least moderate AI had symptomatic heart failure and required surgical intervention.13 Similarly, in another study, patients with mild or greater AI had increased rates of cardiac events, defined as heart failure or arrhythmia requiring admission (33.3% vs 3.5%; P=.03).20 As is evident from these results, AI is a common and persistent issue with LVAD support, often with significant clinical implications.
Pathophysiology and diagnosis
The mechanical unloading of the left ventricle during LVAD support results in decreased aortic valve opening when compared with normal physiology. This leads to increased closure time, exposing the valve leaflets to extensive collagen deposition and subsequent commissural fusion.21,22 Mudd et al described a series of 9 patients with HMII, of which 8 patients had evidence of commissural fusion of the native aortic valve leaflets at the time of explant during heart transplantation.21 Additionally, increased shear forces from the flow through the outflow cannula (as well as retrograde flow from the outflow cannula) predispose the aortic root to dilation, further increasing risk of AI.12,22,23 Furthermore, increased transvalvular gradients during high LVAD support likely contribute to development of AI.24
Diagnosis of symptomatic AI should be suspected in a patient with LVAD and symptoms of heart failure, such as lower-extremity edema, dyspnea, fatigue. A transthoracic echocardiogram (TTE) should be obtained to quantify the degree of AI. Measuring AI in patients with CF-LVADs is challenging, as it tends to be present throughout the cardiac cycle compared to only in diastole during normal physiology. A recent study by Grinstein et al showed that AI using traditional TTE grading criteria may be underestimated and novel TTE parameters (peak systolic to diastolic velocity ratio and the diastolic acceleration of the LVAD outflow cannula) may more accurately measure AI in patients with continuous flow LVADs.25,26 LVAD parameters are often not helpful in diagnosis of AI, as the flows are preserved despite a low cardiac output and no significant change in the power requirement is observed.27 Therefore, right heart catheterization should be performed to assess true cardiac output (Figure 1).
FIGURE 1.
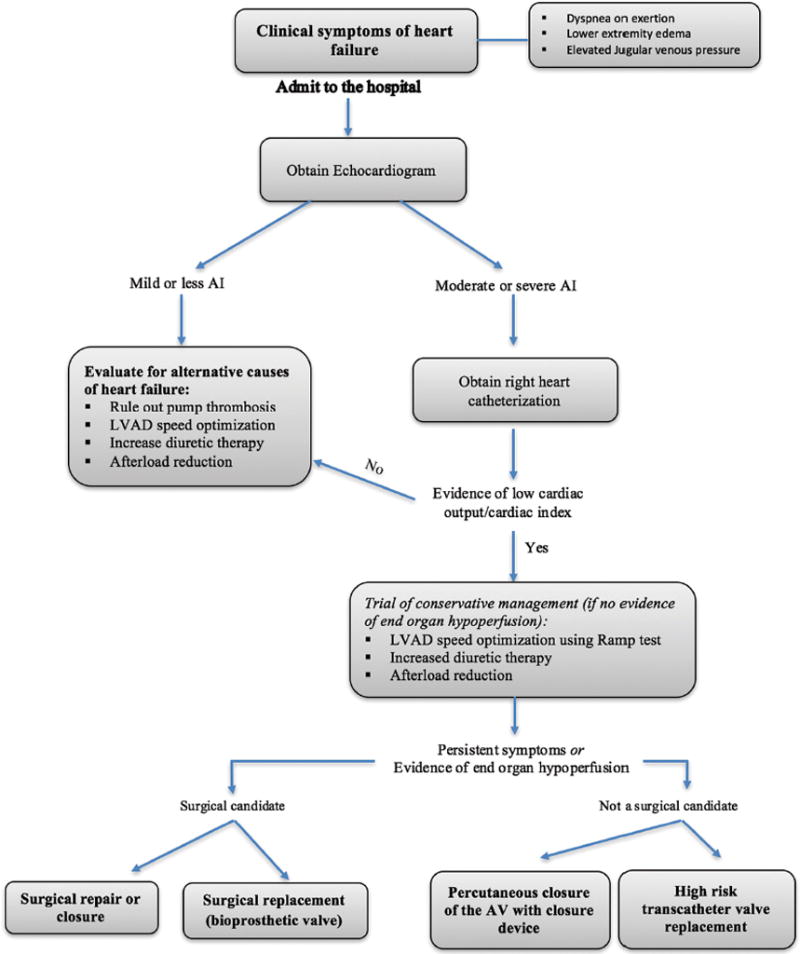
Diagnosis and management algorithm for de novo aortic insufficiency.
Management
Initial management of symptomatic heart failure in the setting of moderate or severe AI should involve diuresis and afterload reduction. If symptoms fail to improve, ramp study for LVAD speed optimization should be considered.13 If symptoms persist despite speed optimization, definitive therapy with surgical or percutaneous means may be required.
The International Heart and Lung Transplantation guidelines recommend surgical intervention with either aortic valve replacement or repair during LVAD implantation in patients who have greater than mild AI at baseline.28-30 In patients who develop AI after LVAD, so called de novo AI (Table 1), surgical treatment includes replacement with bioprosthetic valve31 (mechanical valves should be avoided due to high risk of thrombosis given low-flow state,24,32 valve repair using a coaptation stitch [“Park’s stitch”],33,34 or closure). While surgical management may be appropriate for many patients, re-do surgery is complex and high risk. Therefore, in select patients, less invasive, non-surgical transcatheter options must be explored. Transcatheter options for treatment of de novo AI include application of a septal occluder device to close the aortic valve or percutaneous transcatheter aortic valve replacement (TAVR).35-39 Grohmann et al first reported the use of an Amplatzer post-infarct muscular ventricular septal defect (VSD) occluder (St. Jude Medical) in a patient presenting with severe AI and symptoms consistent with congestive heart failure.35 The aortic valve was percutaneously closed via left subclavian approach under transesophageal echocardiographic (TEE) guidance using a 24 mm VSD occluder, sized using a preoperative computed tomography (CT) scan. While there was an improvement in the hemodynamics, the patient experienced significant transfusion-requiring hemolysis before ultimately being successfully discharged home.37 Since then, numerous studies have been published using the Amplatzer Cribriform device (St. Jude Medical). Russo et al described a patient who underwent surgical aortic valve repair 8 months post LVAD implantation and subsequently presented in cardiogenic shock 3 months later due to recurrent severe AI.36 Using transfemoral approach with a 7 Fr sheath, the left ventricular side of the Amplatzer occluder device was first exposed followed by the deployment of the aortic side under TEE guidance. The device was successfully deployed with no further AI and significant hemodynamic improvement. Subsequently, the group has published further results of their experience with the Cribriform device (Figure 2).35 In 5 patients with AI and excessively high surgical risk, percutaneous closure successfully improved AI (from severe to mild or absent; P=.04) with concurrent improvement in the hemodynamics. There were no changes in the LVAD parameters after the AV closure. In 1 of these patients, the device embolized to the aortic arch on day 4 and was successfully retrieved percutaneously. In this sick cohort, 2 patients were alive at 30 days with well-positioned devices and no residual AI. In a larger retrospective cohort of 10 LVAD patients with high surgical risk (STS mortality risk, 22%) and severe AI who underwent Cribriform septal occluder implantation, the 6-month survival was 30%.39 In this cohort, device-to-annulus ratio of less than 0.9 and lack of significant preoperative right heart dysfunction was associated with improved survival. Smaller devices may apply less pressure on the interventricular septum and hence may not affect the RV filling and outflow, leading to improved survival. It is important to note that with the Cribriform device deployed, the patient is fully reliant on the LVAD and a device malfunction can quickly become fatal.
Table 1.
Percutaneous therapies for de novo aortic insufficiency (AI) with continuous flow left ventricular assist devices.
| Author | Year | Case Series | Indication/Diagnosis | Intervention | Outcome | Long-Term Outcome |
|---|---|---|---|---|---|---|
| Russo et al36 | 2012 | 1 patient with HMII | Cardiogenic shock due to severe AI 2 months after surgical repair of the AI | Amplatzer Cribriform septal occluder |
|
|
| Parikh et al35 | 2013 | 5 patients: 4 with HMII, 1 with HVAD | Patients with symptomatic AI, deemed to have high surgical risk | Amplatzer Cribriform septal occluder |
|
|
| Retzer et al39 | 2015 | 10 patients, 6 HMII, 4 HVAD | Patient with severe AI and high surgical risk (STS mortality 22%) | Amplatzer Cribriform septal occluder |
|
|
| D’Ancona et al41 | 2012 | 1 patient with LVAD | Severe AI | Edwards SAPIEN 29 mm oversized valve via left anterior thoracotomy under fem-fem bypass |
|
|
| Santini et al42 | 2012 | 1 patient with HMII | Refractory symptomatic AI and no surgical options | 29 mm CoreValve implantation via femoral approach (required 2 valves due to paravalvular leak after first CoreValve) |
|
|
| Khan et al40 | 2013 | 1 patient with HMII | Medically refractory symptomatic AI | Melody transcatheter pulmonary valve in aortic position (femoral approach) |
|
|
FIGURE 2.
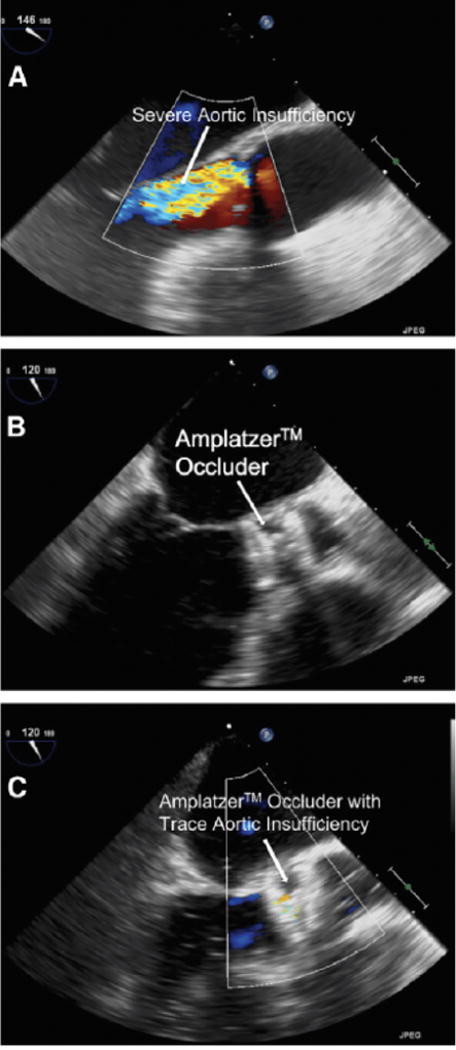
Treatment of aortic insufficiency using the Amplatzer occluder device. (A) Transesophageal echocardiogram showing severe aortic insufficiency. (B) Deployment of the occluder. (C) Transesophageal echocardiogram demonstrating trace residual aortic insufficiency after device deployment. Reprinted with permission from Parikh et al.35
Another strategy to percutaneously treat AI involves TAVR (Figure 3). A case report by Khan et al described a patient with history of aortic valve replacement and homograft conduit who underwent LVAD implantation 10 years later, and subsequently developed medically refractory severe symptomatic AI.40 The patient was deemed to not be a surgical candidate and he successfully underwent a Melody transcatheter pulmonary valve placement in the aortic position via femoral approach. There was trace AI after valve deployment, with significant improvement in hemodynamics. The patient was discharged on day 6 and survived 10 additional months. Another case by D’Ancona used an oversized 29 mm Edwards Sapien valve (Edwards Lifesciences) via left anterior thoracotomy under femo-femoral bypass.41 The valve was successfully deployed without immediate complications, but no information regarding longer-term follow-up is available. In another case report by Santini et al, a patient with refractory symptomatic AI and no surgical options underwent 29 mm CoreValve (Medtronic) implantation via femoral approach.42 A second valve was implanted immediately due to periprosthetic regurgitation with minimal residual leak after deployment of the second valve. The patient recovered and was successfully discharged. While these case reports are encouraging, TAVR in AI without valvular calcification should be approached with extreme caution due to concerns regarding anchoring of the valve and risk of embolization. Additionally, there is high likelihood of fusion of the leaflets of the newly implanted bioprosthetic valve.43
FIGURE 3.
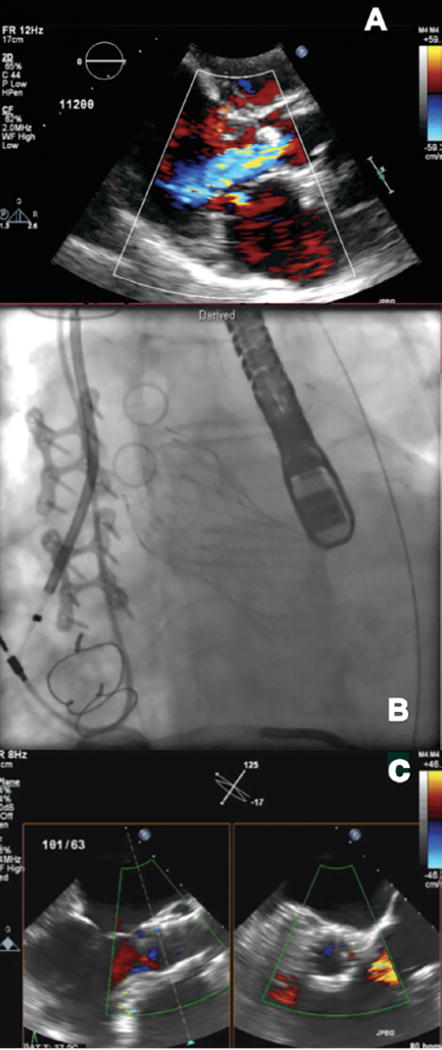
Transcatheter aortic valve replacement in a patient with left ventricular assist device (LVAD) and severe symptomatic aortic insufficiency (AI). (A) Parasternal long-axis view on the transthoracic echocardiogram demonstrating severe AI. (B) Fluoroscopic image demonstrating a CoreValve deployed in the aortic position. (C) Transesophageal echocardiogram demonstrating significant improvement in AI after CoreValve deployment.
In summary, percutaneous treatment of LVAD-acquired and clinically significant moderate-to-severe AI, not amenable to medical management (use of optimal LVAD speed and lower mean arterial pressure goal), has been documented. Percutaneous interventional therapies are important options for patients who are not deemed to be surgical candidates or have high surgical risk. These therapies should be approached with caution since associated outcomes are not ideal and long-term data are lacking.
LVAD Thrombosis
Incidence
Pump thrombosis, defined as a clot located either in the inflow cannula, the central rotary component, or in the outflow graft, is the leading cause of LVAD failure.44 The most common location of thrombosis is typically within the central rotary component and will be the focus of this section. In patients implanted with HMII, rates of pump thrombosis increased dramatically in 2011 and were reported between 8%-12%, with median time to thrombosis decreasing from 18.6 months prior to 2011 to just 3 months since then.45-48 Recently, a reduction of device thrombosis rate was reported in the PREVENT study as a result of improved implantation techniques and focus on heparin bridging and appropriate long-term anticoagulation.49 Similar to HMII, the rate of thrombosis with HVAD has been high, up to 8% at 1 year, with median time to thrombosis event of 245 days.50 The presence of pump thrombosis is associated with significant morbidity and mortality, with data from the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) suggesting a drop in 12-month survival from 82% for the overall cohort to 70% for patients who have first pump exchange due to thrombosis (P<.001).48 Najjar et al also noted decreased 1-year survival in those with pump thrombosis compared to those without evidence of pump thrombosis (69.4% vs 85.5%; P=.21).50 Additionally, many of these patients require pump exchange, which can be associated with significant morbidity as well as economic burden.51
Pathophysiology and diagnosis
Several mechanisms responsible for LVAD thrombosis have been proposed, but the pathophysiology remains unclear. The underlying etiology is likely multifactorial, with activation of the coagulation cascade52 along with shear-stress induced platelet dysfunc tion53 playing a major role. Several pump-related (ie, intrinsic heat generated by the pump, malposition of the inflow cannula, regions of flow stasis within the circuit), patient-related (ie, atrial fibrillation, non-compliance, intrinsic hyper coagulable state, etc), and management-related risk factors (inappropriate antithrombotic and antiplatelet agents) may increase risk of pump thrombosis. In the study by Najjar et al, mean arterial pressure greater than 90 mm Hg, aspirin dose <81 mg, and international normalized ratio (INR) <2 were associated with increased risk of pump thrombosis.50
Pump thrombosis should be suspected in patients with symptoms of left heart failure and/or isolated elevation of lactate dehydrogenase (LDH) levels (2.5× greater than the upper limit of normal), evidence of hemolysis (elevated LDH, plasma free hemoglobin, dark urine), sustained power elevation (>24 hours or power increase of 2W above base-line), or decreasing LVAD flows.54-59 If any of these findings are present, a TTE is the preferred next step. Echocardiographic parameters such as rightward deviation of septum, increased left ventricular end-diastolic diameter, or aortic valve opening with each cardiac cycle are suggestive of pump thrombosis and further evaluation with a “ramp” test may be warranted. Uriel et al have demonstrated that failure of the left ventricular end-diastolic dimension (LVEDD) to change with increasing LVAD speeds is a strong predictor of device thrombosis in HMII patients.57 Ramp test should not be used to diagnose device thrombosis in HVAD patients as they are less informative.58 In patients with HVADs, analysis of the log files may allow for earlier detection of LVAD thrombosis.60 Importantly, CT of the chest should be ordered in patients with suspected pump thrombosis to rule out mechanical issues such as malpositioned inflow cannula or to identify thrombus within the inflow or outflow cannula.61-63 An algorithm for diagnosis and management of LVAD thrombosis is shown in Figure 4.
FIGURE 4.

Algorithm for diagnosis and management of pump thrombosis.
Management
Pump thrombosis is treated by either medical therapy alone or in combination with surgical management via either pump exchange or transplantation. Regardless of the route chosen, heparin and antiplatelet agents are requisite therapy for suspected or confirmed pump thrombosis and more aggressive medical therapy may involve the addition of intra-venous tissue plasminogen activator (tPA). ISHLT provides a consensus document with an algorithm for the diagnosis and treatment of device thrombosis in a setting of HMII.55
Thrombus in an HVAD (which tends to be laminar fibrin deposition) is generally felt to be more amenable to thrombolytic therapy than thrombus in an HMII (which tends to be globular clot and therefore less amenable to thrombolytics at the time of detection).54,60,64 Stulak et al reported results in a multi-center cohort of 175 patients who underwent HVAD implantation.65 In this retrospective study, there were 36 pump thromboses, of which 29 were initially treated medically (majority with tPA) and 7 were initially treated surgically (device exchange). Significant morbidity and mortality were associated with medical management (hemorrhagic stroke in 21%, need for urgent device exchange or transplant in 21%, and death in 10%) compared with no early complications in those initially treated surgically. Similarly, Levin et al reported significant improvement in overall survival in patients treated with early device exchange compared with medical management in patients with suspected pump thrombosis.66
While surgical management is the treatment of choice for pump thrombosis,45,53,65,66 some patients may not be candidates due to significant morbidity and mortality, especially in the setting of an acutely unstable patient.50,67,68 As such, thrombolysis may be necessary in a select few patients. While there have been case reports of successful use of glycoprotein IIb/IIIa inhibitors for pump thrombosis,69,70 in larger studies it has been associated with poor therapeutic response and significant morbidity.71 Given these results, the risks of eptifibatide outweigh the benefits in treatment of pump thrombosis and they were not included in the algorithm proposed by Goldstein et al.55
Compared with glycoprotein IIb/IIIa inhibitors, thrombolytics have demonstrated higher success and lower complication rates. In the trial of HVAD patients by Najjar et al, success rate with thrombolytics was 63% (12/19), with similar success rates when thrombolytics were administered peripherally (6/8) or centrally, within the LV cavity (4/7).50 The route of administration was unknown in 4 patients. The complications included 5 bleeding events, with 2 hemorrhagic cerebrovascular accidents, 2 gastrointestinal bleeds, and 1 pocket implantable cardioverter defibrillator hematoma, and the study did not indicate if there were differences in the incidences of adverse events between peripheral and central tPA. In a case series of 8 patients who failed therapy with heparin and eptifibatide for LVAD throm bosis, intraventricular alteplase was administered via pigtail catheter in the left ventricle at 1 mg/min over 30-50 minutes with concomitant heparin administration.72 Of the 8 patients, 3 patients were successfully treated, 3 patients died, and 1 patient underwent emergent LVAD exchange while another under went heart transplantation. Similarly, in a small case series of 2 patients by Thenappan et al, patients received intraventricular alteplase via a pigtail catheter in the left ventricle (Figure 5).73 Unlike the previously described case series, these patients received lower bolus dose of alteplase but with longer duration (alteplase 25 mg bolus and 1 mg/hour for 30 hours in 1 patient and 25 mg bolus followed by 1 mg/hour for 96 hours in 1 patient). In each case, the markers of thrombosis improved significantly over the course of thrombolytic therapy. There have been many other case reports of successful treatment of LVAD thrombosis with intraventricular thrombolytics as well.74,75
FIGURE 5.
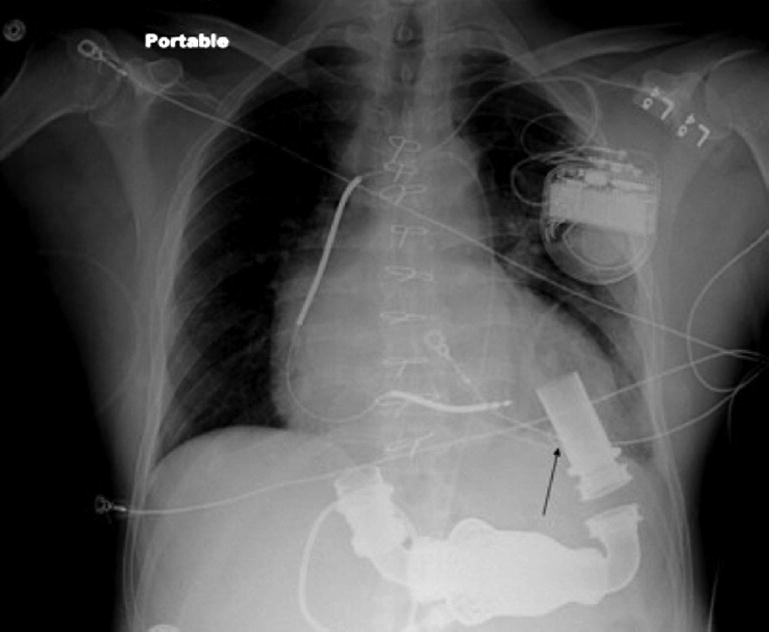
Chest x-ray demonstrating the location of pigtail catheter (arrow) in the left ventricle. Reprinted with permission from Thenappan et al.73
As is evidenced from the results above, thrombolysis with tPA may successfully treat pump thrombosis in some HVAD patients, albeit with significant risk of bleeding, specifically hemorrhagic cerebrovascular accident. Jorde et al have shown that while overall success rate of tPA therapy for HVAD pump thrombosis was only 57%, when using log file parameters, the success of medical therapy increases to 77%.60 Using this algorithm, those with lower measures of expected power and gradual power increase are more likely to benefit from thrombolysis compared with those who suffer sudden power increase.
In summary, treatment for pump thrombosis varies on the type of device, with HMII patients experiencing significantly higher morbidity and mortality with thrombolysis than HVAD patients. Therefore, thrombolysis should be avoided in HMII patients and pump exchange should be the treatment of choice. In those with HVADs, recent data suggest that initial surgical management is preferred, but medical management may be selected in those with appropriate log file parameters. In these patients, the data for peripheral vs central administration of tPA are lacking. Intraventricular thrombolytics to maximize the dose administered directly to the LVAD rotor may be an effective technique to treat thrombosis in patients with HVADs. Using either radial or femoral approach, a pigtail catheter in the left ventricle can be used to deliver tPA as a bolus followed by either short or long course of tPA while carefully monitoring LVAD and laboratory parameters for signs of improvement.
Outflow Graft Stenosis
Incidence
Outflow graft stenosis is a very rare complication of LVAD therapy (Table 2).2 The rates of this complication are significantly lower than the more common structural complications such as LVAD thrombosis or de novo AI described in this paper. Clinical implications of outflow graft stenosis or thrombosis are similar to the LVAD thrombosis described above, with decreased LVAD flows, increased power requirements, and clinical symptoms of heart failure.
Table 2.
Percutaneous therapies for outflow graft stenosis.
| Author | Year | Case Series | Indication/Diagnosis | Intervention | Outcome | Long-Term Outcome |
|---|---|---|---|---|---|---|
| Ganapathi et al79 | 2013 | 1 patient with HMII |
|
|
|
|
| Retzer et al78 | 2015 | 1 patient with HVAD |
|
|
|
|
| Abraham et al82 | 2015 | 1 patient with HMII |
|
|
|
|
| Hanke et al 83 | 2015 | 1 patient with HVAD (history of 2 exchanges previously) |
|
|
|
|
| Hubbert et al 84 | 2016 | 1 patient with HMII |
|
|
|
|
| Wiedermann et al80 | 2016 | 3 patients with HVAD |
|
|
|
|
TTE = transthoracic echocardiogram; HVAD = HeartWare ventricular assist device; HMII = HeartMate II; CT = computed tomography; PTA = percutaneous transluminal angioplasty; mo. = months.
Pathophysiology and diagnosis
The outflow graft is prone to various mechanical as well as non-mechanical issues. Mechanical obstruction can be caused by kinking of the out-flow graft or due to bend relief disconnect, which is the most common cause for outflow graft stenosis.76 On the other hand, non-mechanical causes of outflow graft obstruction include aortic atherosclerosis resulting in suboptimal surgical landing site, fibrotic changes of the aorta, or thrombosis of the outflow graft.62,77,78 The mechanisms of these complications are not fully understood. If thrombosis is diagnosed, low flow state within the outflow graft may be the culprit.79
Patients with outflow graft stenosis present in the same manner as those with LVAD thrombosis, with LVAD parameters showing decreasing flows and increasing power requirements, laboratory evidence of hemolysis, and clinical symptoms of heart failure. As part of the algorithm, Goldstein et al recommend contrast CT scan of the chest to assess the inflow cannula position and the outflow graft.55
Treatment
Management of outflow graft is often surgical, especially if a mechanical issue such as kinking of the outflow graft is diagnosed. Acharya et al describe a case where significant outflow graft stenosis near the aortic anastomosis was identified with a CT scan and subsequently treated with surgical graft revision.63
Percutaneous treatment of outflow graft stenosis or thrombosis has been described in multiple case reports in the last few years. In a case report by Retzer et al, a patient with HVAD was diagnosed with outflow graft stenosis (90%) resulting in a substantial gradient of 120 mm Hg across the lesion.78 Serial balloon dilations followed by placement of 10 × 38 mm, stainless-steel, balloon-expandable Atrium iCAST stent (Maquet Gettinge Group) resulted in immediate improvement in LVAD parameters (flow increased from 2.5 L/min to 9.0 L/min at 2900 rpm, necessitating drop of the speed to 2350 rpm, with flow of 4 L/min), with reduction of the gradient to 30 mm Hg (Figure 6). Similarly, in a case series of 3 patients with HVADs presenting with heart failure symptoms and diagnosed with graft outflow stenosis, all patients were treated with percutaneous transcatheter dilation of the outflow graft and implantation of the Smart Control 12 × 40 mm stent.80 Another case report by Ganapathi et al showed percutaneous treatment of outflow graft pseudoaneurysm in a patient with prohibitive surgical risk for open repair.79 A Gore Excluder iliac limb stent graft (W.L. Gore) was successfully deployed with interval follow-up imaging showing well-positioned stent-graft without evidence of endoleak and complete resolution of the pseudoaneurysm. Another case described the use of a 20 × 55 mm AneuRx iliac limb stent-graft (Medtronic) to treat an arterio-bronchial fistula of the outflow graft.81
FIGURE 6.
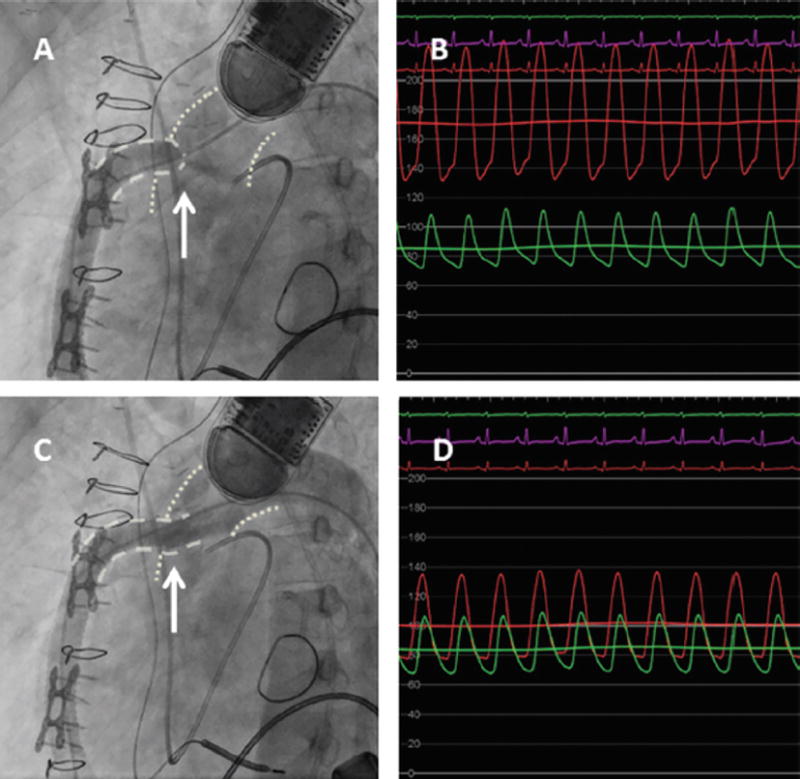
Outflow graft stenosis in a patient with left ventricular assist device, treated with an Atrium 10 × 38 mm covered stent. (A) Significant stenosis at the aortic-outflow graft anastomosis seen on the angiogram with the arrow pointing to the tightest area of stenosis with narrow contrast jet extravasation into the aorta. (B) Hemodynamic assessment demonstrating a significant gradient across the lesion with green aortic waveform and the red waveform measured directly in the outflow graft. (C) Angiography post deployment of the Atrium 10 × 38 mm covered stent. (D) Significant improvement of the hemodynamics, from 120 mm Hg gradient to 30 mm Hg gradient. Reprinted with permission from Retzer et al.78
In a case report by Abraham et al, an outflow graft thrombosis of HMII was treated via subclavian approach and retrograde access to the outflow graft.82 A 9 × 59 mm Atrium stent was deployed in the outflow graft followed by deployment of an additional stent, resulting in satisfactory improvement in angiographic and clinical findings. The patient was discharged 4 days later on anticoagulation. Similarly, an HVAD patient with history of two prior LVAD exchanges was diagnosed with LVAD outflow graft thrombosis.83 This was successfully treated via brachial arterial approach along with embolic protection in the carotid artery via femoral approach. A 12.0 × 61 mm covered Atrium stent was deployed and postdilated with a balloon with significant improvement in the degree of stenosis. The patient did well and was discharged on postoperative day 8. The patient did well until 8 months later, at which time the patient had signs of hemolysis and ultimately underwent heart transplantation. It should be noted that when stenting the outflow graft, risk of embolism is present, especially when thrombosis of the outflow graft is present, and it is reasonable to protect supra-aortic branches with dilated balloons or carotid filters.82-84
Percutaneous therapies for outflow graft stenosis, a rare complication of LVAD therapy, have been described in the literature. While long-term follow-up data are not available, percutaneous outflow graft stenting is a viable option for many patients with LVAD outflow graft stenosis and its clinical application is expected to increase due to the rising number of LVAD implantations and longer duration of LVAD therapy. However, more long-term follow-up data are needed regarding the durability of outflow graft stenting compared with surgical replacement. It is evident that outflow graft stenting is a viable alternative and should be explored prior to proceeding with surgical replacement.
LVAD Decommissioning
A small subset of patients with LVADs may require decommissioning of the LVAD either due to myocardial recovery or due to pump failure. During cases of acute pump failure, valveless CF-LVADs allow retrograde flow from the aorta resulting in acute elevation of left-sided filling pressures akin to acute AI.85 In addition, stasis of flow in the outflow graft leads to thrombus formation, which can result in distal embolization with potentially devastating complications. While the outflow graft is expected to thrombose over time due to the low-flow state, the risk of embolization persists until it is completely thrombosed. Surgical explanation of CF-LVADs often necessitates sternotomy with cardiopulmonary bypass, which carries an inherent risk in this patient population. Minimally invasive surgical techniques that reduce morbidity associated with explantation have been recently described.86-88
Percutaneous LVAD decommissioning has the ability to quickly stabilize patients who experience sudden pump failure resulting in clinical deterioration. Acute tamponade of the outflow graft using 14-16 mm peripheral balloons has been described by Chrysant et al.89 In this case series, both patients presented with acute decompensation in the setting of pump failure and each underwent balloon tamponade of the outflow graft with eventual surgical pump exchange after clinical stability. A catheter-based outflow graft occlusion using 22 mm Amplatzer Vascular Plug II (St. Jude Medical) has been described in a patient with HMII who did not wish to undergo repeat sternotomy for device explantation after myocardial recovery.90 Recently, percutaneous decommissioning of HVAD has been described with a 14 mm Amplatzer septal occluder successfully deployed in the outflow graft in a patient with pump thrombosis who was not a candidate for device exchange (Figure 7).91
FIGURE 7.
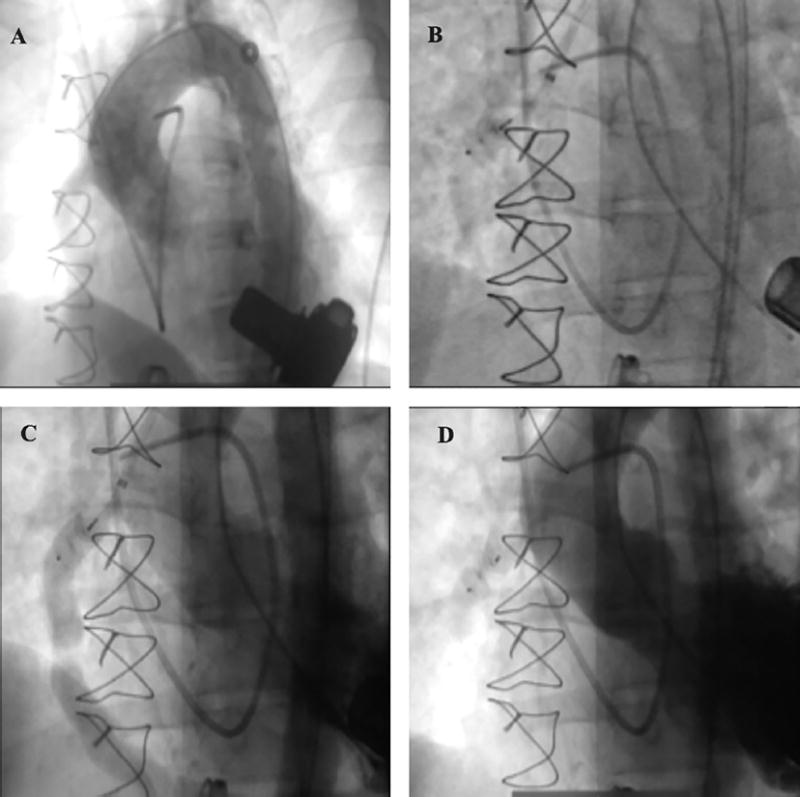
Percutaneous left ventricular assist device (HVAD; HeartWare) decommissioning with a 12 mm Amplatzer Vascular Plug II (St. Jude Medical) deployed in the left ventricular assist device outflow cannula via femoral approach in a patient whose cardiac function recovered. (A) HVAD and outflow graft are visible with anterograde flow from the graft. (B, C) Amplatzer Vascular Plug II positioned in the outflow graft. (D) Fully deployed Vascular Plug II with completely occluded outflow graft and no anterograde or retrograde flow.
Percutaneous LVAD decommissioning of the LVAD outflow graft is an intriguing option for a select group of patients as it allows for rapid correction of retrograde flow within the outflow graft. It can also exclude the outflow graft in patients with myocardial recovery in whom device explantation is not an option.
Conclusion
LVADs have become critical in the treatment of end-stage heart failure with significant rise in device implantations and duration of LVAD therapy. The increasing use of LVADs has led to the observation of increasing complications. In some patients, surgical therapy may be associated with prohibitively high risk, thereby leading to the development of minimally invasive transcatheter interventional therapies to treat LVAD-associated AI, LVAD thrombosis, outflow graft stenosis, and pump failure.
Acknowledgments
Dr Uriel reports consultant/grant fees from Medtronic, Abbott Vascular, and Novartis. Dr Shah reports consulting and honoraria from St. Jude Medical.
Footnotes
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. The remaining authors report no conflicts of interest regarding the content herein.
References
- 1.Go AS, Mozaffarian D, Roger VL, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics–2013 update: a report from the American Heart Association. Circulation. 2013;127:e6–e245. doi: 10.1161/CIR.0b013e31828124ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kirklin JK, Naftel DC, Pagani FD, et al. Seventh INTERMACS annual report: 15,000 patients and counting. J Heart Lung Transplant. 2015;34:1495–1504. doi: 10.1016/j.healun.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 3.Lund LH, Edwards LB, Kucheryavaya AY, et al. The registry of the international society for heart and lung transplantation: thirty-first official adult heart transplant report–2014; focus theme: retransplantation. J Heart Lung Transplant. 2014;33:996–1008. doi: 10.1016/j.healun.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 4.U.S. Food and Drug Administration. Serious adverse events with implantable left ventricular assist devices (LVADs): FDA safety communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm457327.htm Accessed August 22, 2016.
- 5.Colvin M, Smith JM, Skeans MA, et al. Heart. Am J Transplant. 2016;16(Suppl 2):115–140. doi: 10.1111/ajt.13670. [DOI] [PubMed] [Google Scholar]
- 6.Stulak JM, Lee D, Haft JW, et al. Gastrointestinal bleeding and subsequent risk of thromboembolic events during support with a left ventricular assist device. J Heart Lung Transplant. 2014;33:60–64. doi: 10.1016/j.healun.2013.07.020. [DOI] [PubMed] [Google Scholar]
- 7.Boyle AJ, Jorde UP, Sun B, et al. HeartMate II Clinical Investigators Pre-operative risk factors of bleeding and stroke during left ventricular assist device support: an analysis of more than 900 HeartMate II outpatients. J Am Coll Cardiol. 2014;63:880–888. doi: 10.1016/j.jacc.2013.08.1656. [DOI] [PubMed] [Google Scholar]
- 8.Goldstein DJ, Aaronson KD, Tatooles AJ, et al. ADVANCE Investigators Gastrointestinal bleeding in recipients of the HeartWare ventricular assist system. JACC Heart Fail. 2015;3:303–313. doi: 10.1016/j.jchf.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 9.Backes D, van den Bergh WM, van Duijn AL, Lahpor JR, van Dijk D, Slooter AJ. Cerebrovascular complications of left ventricular assist devices. Eur J Cardiothorac Surg. 2012;42:612–620. doi: 10.1093/ejcts/ezs320. [DOI] [PubMed] [Google Scholar]
- 10.Sharma V, Deo SV, Stulak JM, et al. Driveline infections in left ventricular assist devices: implications for destination therapy. Ann Thorac Surg. 2012;94:1381–1386. doi: 10.1016/j.athoracsur.2012.05.074. [DOI] [PubMed] [Google Scholar]
- 11.Cowger JA, Aaronson KD, Romano MA, Haft J, Pagani FD. Consequences of aortic insufficiency during long-term axial continuous-flow left ventricular assist device support. J Heart Lung Transplant. 2014;33:1233–1240. doi: 10.1016/j.healun.2014.06.008. [DOI] [PubMed] [Google Scholar]
- 12.Pak SW, Uriel N, Takayama H, et al. Prevalence of de novo aortic insufficiency during long-term support with left ventricular assist devices. J Heart Lung Transplant. 2010;29:1172–1176. doi: 10.1016/j.healun.2010.05.018. [DOI] [PubMed] [Google Scholar]
- 13.Jorde UP, Uriel N, Nahumi N, et al. Prevalence, significance, and management of aortic insufficiency in continuous flow left ventricular assist device recipients. Circ Heart Fail. 2014;7:310–319. doi: 10.1161/CIRCHEARTFAILURE.113.000878. [DOI] [PubMed] [Google Scholar]
- 14.Aggarwal A, Raghuvir R, Eryazici P, et al. The development of aortic insufficiency in continuous-flow left ventricular assist device-supported patients. Ann Thorac Surg. 2013;95:493–498. doi: 10.1016/j.athoracsur.2012.09.020. [DOI] [PubMed] [Google Scholar]
- 15.Deo SV, Sharma V, Cho YH, Shah IK, Park SJ. De novo aortic insufficiency during long-term support on a left ventricular assist device: a systematic review and meta-analysis. ASAIO J. 2014;60:183–188. doi: 10.1097/MAT.0000000000000042. [DOI] [PubMed] [Google Scholar]
- 16.Patil NP, Sabashnikov A, Mohite PN, et al. De novo aortic regurgitation after continuous-flow left ventricular assist device implantation. Ann Thorac Surg. 2014;98:850–857. doi: 10.1016/j.athoracsur.2014.05.030. [DOI] [PubMed] [Google Scholar]
- 17.Rajagopal K, Daneshmand MA, Patel CB, et al. Natural history and clinical effect of aortic valve regurgitation after left ventricular assist device implantation. J Thorac Cardiovasc Surg. 2013;145:1373–1379. doi: 10.1016/j.jtcvs.2012.11.066. [DOI] [PubMed] [Google Scholar]
- 18.Toda K, Fujita T, Domae K, Shimahara Y, Kobayashi J, Nakatani T. Late aortic insufficiency related to poor prognosis during left ventricular assist device support. Ann Thorac Surg. 2011;92:929–934. doi: 10.1016/j.athoracsur.2011.04.115. [DOI] [PubMed] [Google Scholar]
- 19.Cowger J, Pagani FD, Haft JW, Romano MA, Aaronson KD, Kolias TJ. The development of aortic insufficiency in left ventricular assist device-supported patients. Circ Heart Fail. 2010;3:668–674. doi: 10.1161/CIRCHEARTFAILURE.109.917765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mano A, Gorcsan J, Teuteberg J, et al. 38 Incidence and impact of de novo aortic insufficiency following continuous flow LVADs implantation. J Heart Lung Transplant. 2012;31:S22. [Google Scholar]
- 21.Mudd JO, Cuda JD, Halushka M, Soderlund KA, Conte JV, Russell SD. Fusion of aortic valve commissures in patients supported by a continuous axial flow left ventricular assist device. J Heart Lung Transplant. 2008;27:1269–1274. doi: 10.1016/j.healun.2008.05.029. [DOI] [PubMed] [Google Scholar]
- 22.Holtz J, Teuteberg J. Management of aortic insufficiency in the contiuous flow left ventricular assist device population. Curr Heart Fail Rep. 2014;11:103–110. doi: 10.1007/s11897-013-0172-6. [DOI] [PubMed] [Google Scholar]
- 23.John R, Panch S, Hrabe J, et al. Activation of endothelial and coagulation systems in left ventricular assist device recipients. Ann Thorac Surg. 2009;88:1171–1179. doi: 10.1016/j.athoracsur.2009.06.095. [DOI] [PubMed] [Google Scholar]
- 24.Cowger J, Rao V, Massey T, et al. Comprehensive review and suggested strategies for the detection and management of aortic insufficiency in patients with a continuous-flow left ventricular assist device. J Heart Lung Transplant. 2015;34:149–157. doi: 10.1016/j.healun.2014.09.045. [DOI] [PubMed] [Google Scholar]
- 25.Grinstein J, Kruse E, Sayer G, et al. Accurate quantification methods for aortic insufficiency severity in patients with LVAD: role of diastolic flow acceleration and systolic-to-diastolic peak velocity ratio of outflow cannula. JACC Cardiovasc Imaging. 2016;9:641–651. doi: 10.1016/j.jcmg.2015.06.020. [DOI] [PubMed] [Google Scholar]
- 26.Grinstein J, Kruse E, Sayer G, et al. Improved prognostic performance of novel parameters for grading aortic insufficiency severity in patients with left ventricular assist devices. Circulation. 2015;132:A16373. [Google Scholar]
- 27.Morgan JA, Brewer RJ, Nemeh HW, et al. Management of aortic valve insufficiency in patients supported by long-term continuous flow left ventricular assist devices. Ann Thorac Surg. 2012;94:1710–1712. doi: 10.1016/j.athoracsur.2012.03.105. [DOI] [PubMed] [Google Scholar]
- 28.Feldman D, Pamboukian SV, Teuteberg JJ, et al. The 2013 International Society for Heart and Lung Transplantation guidelines for mechanical circulatory support: executive summary. J Heart Lung Transplant. 2013;32:157–187. doi: 10.1016/j.healun.2012.09.013. [DOI] [PubMed] [Google Scholar]
- 29.Rao V, Slater JP, Edwards NM, Naka Y, Oz MC. Surgical management of valvular disease in patients requiring left ventricular assist device support. Ann Thorac Surg. 2001;71:1448–1453. doi: 10.1016/s0003-4975(01)02479-1. [DOI] [PubMed] [Google Scholar]
- 30.Stringham JC, Bull DA, Karwande SV. Patch closure of the aortic annulus in a recipient of a ventricular assist device. J Thorac Cardiovasc Surg. 2000;119:1293–1294. doi: 10.1067/mtc.2000.106035. [DOI] [PubMed] [Google Scholar]
- 31.Feldman CM, Silver MA, Sobieski MA, Slaughter MS. Management of aortic insufficiency with continuous flow left ventricular assist devices: bioprosthetic valve replacement. J Heart Lung Transplant. 2006;25:1410–1412. doi: 10.1016/j.healun.2006.10.004. [DOI] [PubMed] [Google Scholar]
- 32.Robertson JO, Naftel DC, Myers SL, et al. Concomitant aortic valve procedures in patients undergoing implantation of continuous-flow left ventricular assist devices: an INTERMACS database analysis. J Heart Lung Transplant. 2015;34:797–805. doi: 10.1016/j.healun.2014.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Park SJ, Liao KK, Segurola R, Madhu K, Miller LW. Management of aortic insufficiency in patients with left ventricular assist devices: a simple coaptation stitch method (Park’s stitch) J Thorac Cardiovasc Surg. 2004;127:264–266. doi: 10.1016/s0022-5223(03)01301-1. [DOI] [PubMed] [Google Scholar]
- 34.McKellar SH, Deo S, Daly RC, et al. Durability of central aortic valve closure in patients with continuous flow left ventricular assist devices. J Thorac Cardiovasc Surg. 2014;147:344–348. doi: 10.1016/j.jtcvs.2012.09.098. [DOI] [PubMed] [Google Scholar]
- 35.Parikh KS, Mehrotra AK, Russo MJ, et al. Percutaneous transcatheter aortic valve closure successfully treats left ventricular assist device-associated aortic insufficiency and improves cardiac hemodynamics. JACC Cardiovasc Interv. 2013;6:84–89. doi: 10.1016/j.jcin.2012.08.021. [DOI] [PubMed] [Google Scholar]
- 36.Russo MJ, Freed BH, Jeevanandam V, et al. Percutaneous transcatheter closure of the aortic valve to treat cardiogenic shock in a left ventricular assist device patient with severe aortic insufficiency. Ann Thorac Surg. 2012;94:985–988. doi: 10.1016/j.athoracsur.2012.01.089. [DOI] [PubMed] [Google Scholar]
- 37.Grohmann J, Blanke P, Benk C, Schlensak C. Trans-catheter closure of the native aortic valve with an Amplatzer occluder to treat progressive aortic regurgitation after implantation of a left-ventricular assist device. Eur J Cardiothorac Surg. 2011;39:e181–e183. doi: 10.1016/j.ejcts.2011.01.036. [DOI] [PubMed] [Google Scholar]
- 38.Freed BH, Paul JD, Bhave NM, et al. Percutaneous transcatheter closure of the native aortic valve to treat de novo aortic insufficiency after implantation of a left ventricular assist device. JACC Cardiovasc Interv. 2012;5:358–359. doi: 10.1016/j.jcin.2011.11.012. [DOI] [PubMed] [Google Scholar]
- 39.Retzer EM, Sayer GT, Fedson SE, et al. Predictors of survival following trans-catheter aortic valve closure for left ventricular assist device associated aortic insufficiency. Catheter Cardiovasc Interv. 2016;87:971–979. doi: 10.1002/ccd.26280. Epub 2015 Nov 3. [DOI] [PubMed] [Google Scholar]
- 40.Khan S, Koerner MM, Pae W, et al. Successful percutaneous transcatheter aortic valve replacement in multi-organ failure due to aortic bioprosthesis regurgitation in a patient with continuous-flow LVAD. J Heart Lung Transplant. 2013;32:659–663. doi: 10.1016/j.healun.2013.03.007. [DOI] [PubMed] [Google Scholar]
- 41.D’Ancona G, Pasic M, Buz S, et al. TAVI for pure aortic valve insufficiency in a patient with a left ventricular assist device. Ann Thorac Surg. 2012;93:e89–e91. doi: 10.1016/j.athoracsur.2011.11.019. [DOI] [PubMed] [Google Scholar]
- 42.Santini F, Forni A, Dandale R, et al. First successful management of aortic valve insufficiency associated with HeartMate II left ventricular assist device support by transfemoral CoreValve implantation: the Columbus’s egg? JACC Cardiovasc Interv. 2012;5:114–115. doi: 10.1016/j.jcin.2011.10.009. [DOI] [PubMed] [Google Scholar]
- 43.Parry D, Rao V, Butany J, et al. Transcatheter aortic valve implantation and left ventricular assist device: a word of caution. Ann Thorac Surg. 2014;97:e41–e42. doi: 10.1016/j.athoracsur.2013.09.071. [DOI] [PubMed] [Google Scholar]
- 44.Xie A, Phan K, Yan TD. Durability of continuous-flow left ventricular assist devices: a systematic review. Ann Cardiothorac Surg. 2014;3:547–556. doi: 10.3978/j.issn.2225-319X.2014.11.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Starling RC, Moazami N, Silvestry SC, et al. Unexpected abrupt increase in left ventricular assist device thrombosis. N Engl J Med. 2014;370:33–40. doi: 10.1056/NEJMoa1313385. [DOI] [PubMed] [Google Scholar]
- 46.Slaughter MS, Rogers JG, Milano CA, et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361:2241–2251. doi: 10.1056/NEJMoa0909938. [DOI] [PubMed] [Google Scholar]
- 47.Miller LW, Pagani FD, Russell SD, et al. Use of a continuous-flow device in patients awaiting heart transplantation. N Engl J Med. 2007;357:885–896. doi: 10.1056/NEJMoa067758. [DOI] [PubMed] [Google Scholar]
- 48.Kirklin JK, Naftel DC, Pagani FD, et al. Pump thrombosis in the Thoratec Heart-Mate II device: an update analysis of the INTERMACS registry. J Heart Lung Transplant. 2015;34:1515–1526. doi: 10.1016/j.healun.2015.10.024. [DOI] [PubMed] [Google Scholar]
- 49.Maltais S, Kilic A, Nathan S, et al. Prevention of HeartMate II pump thrombosis through clinical management (PREVENT); International Society for Heart and Lung Transplantation 2016 Scientific Sessions; April 30, 2016; Washington, D.C.. [DOI] [PubMed] [Google Scholar]
- 50.Najjar SS, Slaughter MS, Pagani FD, et al. HVAD Bridge to Transplant ADVANCE Trial Investigators An analysis of pump thrombus events in patients in the HeartWare ADVANCE bridge to transplant and continued access protocol trial. J Heart Lung Transplant. 2014;33:23–34. doi: 10.1016/j.healun.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 51.Moreno SG, Novielli N, Cooper NJ. Cost-effectiveness of the implantable HeartMate II left ventricular assist device for patients awaiting heart transplantation. J Heart Lung Transplant. 2012;31:450–458. doi: 10.1016/j.healun.2011.10.017. [DOI] [PubMed] [Google Scholar]
- 52.John R, Panch S, Hrabe J, et al. Activation of endothelial and coagulation systems in left ventricular assist device recipients. Ann Thorac Surg. 2009;88:1171–1179. doi: 10.1016/j.athoracsur.2009.06.095. [DOI] [PubMed] [Google Scholar]
- 53.Steinlechner B, Dworschak M, Birkenberg B, et al. Platelet dysfunction in out-patients with left ventricular assist devices. Ann Thorac Surg. 2009;87:131–137. doi: 10.1016/j.athoracsur.2008.10.027. [DOI] [PubMed] [Google Scholar]
- 54.Uriel N, Han J, Morrison KA, et al. Device thrombosis in HeartMate II continuous-flow left ventricular assist devices: a multifactorial phenomenon. J Heart Lung Transplant. 2014;33:51–59. doi: 10.1016/j.healun.2013.10.005. [DOI] [PubMed] [Google Scholar]
- 55.Goldstein DJ, John R, Salerno C, et al. Algorithm for the diagnosis and management of suspected pump thrombus. J Heart Lung Transplant. 2013;32:667–670. doi: 10.1016/j.healun.2013.05.002. [DOI] [PubMed] [Google Scholar]
- 56.Cowger JA, Romano MA, Shah P, et al. Hemolysis: a harbinger of adverse outcome after left ventricular assist device implant. J Heart Lung Transplant. 2014;33:35–43. doi: 10.1016/j.healun.2013.08.021. [DOI] [PubMed] [Google Scholar]
- 57.Uriel N, Morrison KA, Garan AR, et al. Development of a novel echocardiography ramp test for speed optimization and diagnosis of device thrombosis in continuous-flow left ventricular assist devices: the Columbia ramp study. J Am Coll Cardiol. 2012;60:1764–1775. doi: 10.1016/j.jacc.2012.07.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Uriel N, Levin AP, Sayer GT, et al. Left ventricular decompression during speed optimization ramps in patients supported by continuous-flow left ventricular assist devices: device-specific performance characteristics and impact on diagnostic algorithms. J Card Fail. 2015;21:785–791. doi: 10.1016/j.cardfail.2015.06.010. Epub 2015 Jun 25. [DOI] [PubMed] [Google Scholar]
- 59.Estep JD, Vivo RP, Cordero-Reyes AM, et al. A simplified echocardiographic technique for detecting continuous-flow left ventricular assist device mal-function due to pump thrombosis. J Heart Lung Transplant. 2014;33:575–586. doi: 10.1016/j.healun.2014.01.865. [DOI] [PubMed] [Google Scholar]
- 60.Jorde UP, Aaronson KD, Najjar SS, et al. Identification and management of pump thrombus in the HeartWare left ventricular assist device system: a novel approach using log file analysis. JACC Heart Fail. 2015;3:849–856. doi: 10.1016/j.jchf.2015.06.015. [DOI] [PubMed] [Google Scholar]
- 61.Krishnan B, Yarmohammadi H, Eckman P, Adatya S. Outflow thrombus in a left ventricular-assist device: visualization by CT angiography. J Cardiovasc Comput Tomogr. 2013;8:473–474. doi: 10.1016/j.jcct.2014.08.002. [DOI] [PubMed] [Google Scholar]
- 62.Mishkin JD, Enriquez JR, Meyer DM, et al. Utilization of cardiac computed tomography angiography for the diagnosis of left ventricular assist device thrombosis. Circ Heart Failure. 2012;5:e27–e29. doi: 10.1161/CIRCHEARTFAILURE.111.966119. [DOI] [PubMed] [Google Scholar]
- 63.Acharya D, Singh S, Tallaj JA, et al. Use of gated cardiac computed tomography angiography in the assessment of left ventricular assist device dysfunction. ASAIO J. 2011;57:32–37. doi: 10.1097/MAT.0b013e3181fd3405. [DOI] [PubMed] [Google Scholar]
- 64.Kirklin JK, Naftel DC, Kormos RL, et al. INTERMACS analysis of pump thrombosis in the HeartMate II left ventricular assist device. J Heart Lung Transplant. 2014;33:12–22. doi: 10.1016/j.healun.2013.11.001. [DOI] [PubMed] [Google Scholar]
- 65.Stulak JM, Dunlay SM, Sharma S, et al. Treatment of device thrombus in the HeartWare HVAD: success and outcomes depend significantly on the initial treatment strategy. J Heart Lung Transplant. 2015;34:1535–1541. doi: 10.1016/j.healun.2015.10.020. [DOI] [PubMed] [Google Scholar]
- 66.Levin AP, Saeed O, Willey JZ, et al. Watchful waiting in continuous-flow left ventricular assist device patients with ongoing hemolysis is associated with an increased risk for cerebrovascular accident or death. Circ Heart Fail. 2016;9 doi: 10.1161/CIRCHEARTFAILURE.115.002896. [DOI] [PubMed] [Google Scholar]
- 67.Ota T, Yerebakan H, Akashi H, et al. Continuous-flow left ventricular assist device exchange: clinical outcomes. J Heart Lung Transplant. 2014;33:65–70. doi: 10.1016/j.healun.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 68.Moazami N, Milano CA, John R, et al. Pump replacement for left ventricular assist device failure can be done safely and is associated with low mortality. Ann Thorac Surg. 2013;95:500–505. doi: 10.1016/j.athoracsur.2012.09.011. [DOI] [PubMed] [Google Scholar]
- 69.Thomas MD, Wood C, Lovett M, Dembo L, O’Driscoll G. Successful treatment of rotary pump thrombus with the glycoprotein iib/iiia inhibitor tirofiban. J Heart Lung Transplant. 2008;27:925–927. doi: 10.1016/j.healun.2008.05.015. [DOI] [PubMed] [Google Scholar]
- 70.Jennings DL, Cabrera R, Wang DD, Tita C. Successful treatment of a continuous flow left ventricular assist device thrombosis with eptifibatide. ASAIO J. 2012;58:633–635. doi: 10.1097/MAT.0b013e318271bde2. [DOI] [PubMed] [Google Scholar]
- 71.Tellor BR, Smith JR, Prasad SM, Joseph SM, Silvestry SC. The use of eptifibatide for suspected pump thrombus or thrombosis in patients with left ventricular assist devices. J Heart Lung Transplant. 2014;33:94–101. doi: 10.1016/j.healun.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 72.Schlendorf K, Patel CB, Gehrig T, et al. Thrombolytic therapy for thrombosis of continuous flow ventricular assist devices. J Card Fail. 2014;20:91–97. doi: 10.1016/j.cardfail.2013.12.011. [DOI] [PubMed] [Google Scholar]
- 73.Thenappan T, Anderson AS, Jeevanadham V, Rich JD, Shah AP. Treatment of left ventricular assist device thrombosis with extended catheter-directed intraventricular thrombolytic therapy. Circ Heart Fail. 2013;6:e27–e29. doi: 10.1161/CIRCHEARTFAILURE.113.000013. [DOI] [PubMed] [Google Scholar]
- 74.Kamouh A, John R, Eckman P. Successful treatment of early thrombosis of HeartWare left ventricular assist device with intraventricular thrombolytics. Ann Thorac Surg. 2012;94:281–283. doi: 10.1016/j.athoracsur.2011.12.024. [DOI] [PubMed] [Google Scholar]
- 75.Kiernan MS, Pham DT, DeNofrio D, Kapur NK. Management of HeartWare left ventricular assist device thrombosis using intracavitary thrombolytics. J Thorac Cardiovasc Surg. 2011;142:712–714. doi: 10.1016/j.jtcvs.2010.11.022. [DOI] [PubMed] [Google Scholar]
- 76.Yuzefpolskaya M, Uriel N, Chow DS, et al. Prevalence and timing of bend relief disconnection in patients supported by the late version HeartMate II left ventricular assist device. J Heart Lung Transplant. 2013;32:320–325. doi: 10.1016/j.healun.2012.11.016. [DOI] [PubMed] [Google Scholar]
- 77.Raman SV, Sahu A, Merchant AZ, Louis LB, 4th, Firstenberg MS, Sun B. Non-invasive assessment of left ventricular assist devices with cardiovascular computed tomography and impact on management. J Heart Lung Transplant. 2010;29:79–85. doi: 10.1016/j.healun.2009.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Retzer EM, Tannenbaum SA, Fedson SE, et al. Successful percutaneous transcatheter treatment of left ventricular assist device outflow graft stenosis with a covered stent. ESC Heart Failure. 2015;2:100–102. doi: 10.1002/ehf2.12030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ganapathi AM, Andersen ND, Prastein DJ, et al. Endovascular stent grafting of a left ventricular assist device outflow graft pseudoaneurysm. Circ Heart Fail. 2013;6:e16–e18. doi: 10.1161/CIRCHEARTFAILURE.112.971861. [DOI] [PubMed] [Google Scholar]
- 80.Wiedemann D, Schlöglhofer T, Haberl T, et al. Interventional treatment of LVAD outflow graft stenosis by introduction of bare metal stents. Thorac Cardiovasc Surg. 2016;64:OP121. doi: 10.1097/MAT.0000000000000547. [DOI] [PubMed] [Google Scholar]
- 81.Yanagida R, Kass R, Czer L, Khoynezhad A. Endovascular repair of arterio-bronchial fistula of the outflow graft of HeartMate II left ventricular assist device. J Thorac Cardiovasc Surg. 2011;142:710–711. doi: 10.1016/j.jtcvs.2010.11.021. [DOI] [PubMed] [Google Scholar]
- 82.Abraham J, Remick JD, Caulfield T, et al. Left ventricular assist device outflow cannula obstruction treated with percutaneous endovascular stenting. Circ Heart Fail. 2015;8:229–230. doi: 10.1161/CIRCHEARTFAILURE.114.001891. [DOI] [PubMed] [Google Scholar]
- 83.Hanke JS, El Sherbini A, Rojas SV, Avsar M, Shrestha M, Schmitto JD. Aortic outflow graft stenting in patient with left ventricular assist device outflow graft thrombosis. Artif Organs. 2016;40:414–416. doi: 10.1111/aor.12569. [DOI] [PubMed] [Google Scholar]
- 84.Hubbert L, Forssell C, Baranowski J, et al. Endovascular stenting of an outflow graft thrombosis in a continuous-flow left ventricular assist device. ASAIO J. 2017;63:e3–e5. doi: 10.1097/MAT.0000000000000371. [DOI] [PubMed] [Google Scholar]
- 85.Giridharan GA, Koenig SC, Soucy KG, et al. Hemodynamic changes and retrograde flow in lvad failure. ASAIO J. 2015;61:282–291. doi: 10.1097/MAT.0000000000000200. [DOI] [PubMed] [Google Scholar]
- 86.Haj-Yahia S, Birks EJ, Dreyfus G, Khaghani A. Limited surgical approach for explanting the HeartMate II left ventricular assist device after myocardial recovery. J Thorac Cardiovasc Surg. 2008;135:453–454. doi: 10.1016/j.jtcvs.2007.10.012. [DOI] [PubMed] [Google Scholar]
- 87.Rojas SV, Avsar M, Hanke JS, et al. Minimally invasive ventricular assist device surgery. Artif Organs. 2015;39:473–479. doi: 10.1111/aor.12422. [DOI] [PubMed] [Google Scholar]
- 88.Tansley P, Yacoub M. Minimally invasive explantation of implantable left ventricular assist devices. J Thorac Cardiovasc Surg. 2002;124:189–191. doi: 10.1067/mtc.2002.122786. [DOI] [PubMed] [Google Scholar]
- 89.Chrysant GS, Horstmanshof DA, Snyder T, et al. Successful percutaneous management of acute left ventricular assist device stoppage. ASAIO J. 2010;56:483–485. doi: 10.1097/MAT.0b013e3181ed937f. [DOI] [PubMed] [Google Scholar]
- 90.Zeigler SM, Sheikh AY, Lee PH, et al. A novel, catheter-based approach to left ventricular assist device deactivation after myocardial recovery. Ann Thorac Surg. 2014;98:710–713. doi: 10.1016/j.athoracsur.2013.09.073. [DOI] [PubMed] [Google Scholar]
- 91.Grinstein J, Estrada J, Sayer G, et al. Left ventricular assist device deactivation via percutaneous closure of the outflow graft. J Card Fail. 2016;22:653–655. doi: 10.1016/j.cardfail.2016.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]


