Abstract
Study objectives
Debate persists as to whether obstructive sleep apnea (OSA) is an independent risk factor for atherosclerosis. The purpose of this study was to compare carotid intima-media thickness (IMT), an early sign of atherosclerosis, in obese and nonobese adults with OSA before and following positive airway pressure (PAP) treatment.
Methods
A total of 206 adults newly diagnosed with OSA with an apnea-hypopnea index (AHI) of 15–75 events/hour and 53 controls with AHI <10 were studied. Waist circumference was used to classify participants as obese and nonobese. Bilateral common carotid artery B-mode ultrasound was performed at baseline to assess IMT, arterial diameter, arterial-wall mass, and circumferential wall stress. Measurements were repeated in 118 participants with OSA who completed a 4-month PAP treatment and had an average daily use over that period of ≥4 hours/day.
Results
No significant differences in carotid IMT, diameter, or arterial-wall mass were present at baseline between participants with OSA and controls stratified by waist circumference, after adjusting for other cardiovascular risk factors. In participants with OSA, who had adequate PAP adherence over the 4-month treatment, carotid artery diameter significantly increased (mean change [95% confidence interval] = 0.13 [0.06, 0.20] mm; p = .0004), but no significant changes in carotid IMT, arterial-wall mass, and circumferential stress were observed in obese and nonobese participants.
Conclusions
Regardless of obesity status, carotid IMT is not increased in adults with moderate to severe OSA versus controls and does not change following 4 months of PAP treatment.
Keywords: atherosclerosis, intima-media thickness, carotid artery diameter, circumferential wall stress, arterial-wall mass
Statement of Significance
Although previous studies show conflicting results about the independent effect of obstructive sleep apnea (OSA) on subclinical changes in the carotid artery, this study found no significant differences in intima-media thickness, diameter, arterial-wall mass, and circumferential wall stress between participants with moderate-to-severe OSA and controls at baseline, regardless of obesity status. The 4 months of positive airway pressure (PAP) treatment did not result in significant changes in carotid-artery measures but unexpectedly increased the carotid diameter. Further studies are needed to explore the potential effect of PAP on carotid arteries remodeling and differences between obese and nonobese participants.
INTRODUCTION
Obstructive sleep apnea (OSA), characterized by intermittent obstruction of the pharyngeal airway during sleep, is reported to be an independent and modifiable risk factor for cardiovascular disease.1,2 Carotid artery atherosclerosis, manifested as thickening of the vessel walls or plaque formation, is an early indicator of future cardiovascular events.3 Increasing evidence indicates that the recurrent deoxygenation and reoxygenation in OSA increases oxidative stress and inflammatory activity leading to tissue-cell damage that may cause arterial wall hypertrophy, stiffness, and plaque formation.4–6 In addition, recent studies suggest that vibration induced by snoring, a major symptom of OSA, may be transmitted to adjacent vessels or tissues (such as carotid arteries) and accelerate the atherosclerotic process by causing vessel damage or turbulent blood flow.7,8 Several studies have demonstrated that OSA is significantly associated with increased carotid intima-media thickness (IMT),9–13 while other studies found no difference between participants with OSA and controls.14,15 Controversy remains regarding whether the association is independent of other cardiovascular risk factors. Only a few studies have examined the effect of positive airway pressure (PAP) treatment on carotid IMT, and again the results are conflicting.6,16,17
Obesity is a major risk factor for OSA18 and a known risk factor for atherosclerosis.19 Furthermore, patients with OSA often have comorbid conditions including hypertension and hyperlipidemia, all of which accelerate atherosclerosis. The close association of OSA with these comorbid conditions has confounded attempts to determine if OSA is an independent risk factor of atherosclerosis. Some previous studies report that OSA increases the risk of atherosclerosis in carotid arteries, independent of obesity, by comparing results with those in obese individuals without OSA.6,20 Further studies are needed to confirm the effect of OSA on subclinical changes of carotid atherosclerosis and the possible role of obesity in mediating or modulating those effects.
The primary purpose of the present study was to examine carotid IMT in obese and nonobese adults with and without moderate to severe OSA and to further investigate whether changes in carotid IMT following PAP treatment were affected by the presence of obesity. The analysis was conducted using data from the Penn Iceland Sleep Apnea research project, a two-site, longitudinal study of functional, cardiovascular, and metabolic outcome measures following 4 months of PAP therapy in obese and nonobese patients with moderate to severe OSA. We hypothesized that carotid IMT in participants with OSA would be greater than that in controls and decrease after PAP therapy. We also hypothesized that the differences following PAP treatment would be greater in obese versus nonobese OSA participants.
METHODS
Study Participants
Participants were recruited from the sleep centers at the University of Pennsylvania and Landspitali—The National University Hospital of Iceland. Consecutive patients referred to the sleep centers for evaluation of suspected OSA were informed of the study and asked to participate. Inclusion criteria included age 40–65 years; postmenopausal if female (no menstruation in the past 12 months and not on female hormone replacement therapy), stable medical history and medications, including antihypertensive, thyroid replacement, and lipid-lowering medications, in the previous 2 months; and no regular use (≥3 times/week) of sedative or hypnotic medications. Individuals were excluded from the study for the following reasons: body mass index (BMI) >40 kg/m2; diagnosis of another sleep disorder in addition to OSA based on clinical information and polysomnography (eg, periodic limb movement disorder, central sleep apnea, insomnia, obesity hypoventilation syndrome, or narcolepsy); previous treatment of OSA with PAP, home oxygen therapy, or surgery; requiring oxygen or bilevel PAP for treatment of OSA; active infection, malignancy, or chronic inflammatory disorders such as autoimmune diseases that can alter inflammatory activity; night-shift workers or occupations where individuals regularly experience jet lag, or had irregular work schedules by history over the last 6 months; unable to perform tests due to inability to communicate verbally or inability to write and read in English/Icelandic, depending on the site of enrollment; arm circumference >50 cm; routine consumption of more than two alcoholic beverages per day, excessive caffeine use (>10 caffeinated beverages per day); current illicit drug use and positive urine toxicology; metal parts in the body excluding magnetic resonance imaging (MRI) (as part of this protocol, individuals had MRIs; results not reported here). The protocol was approved by the Institutional Review Board at both clinical sites, and written informed consent was obtained from all participants.
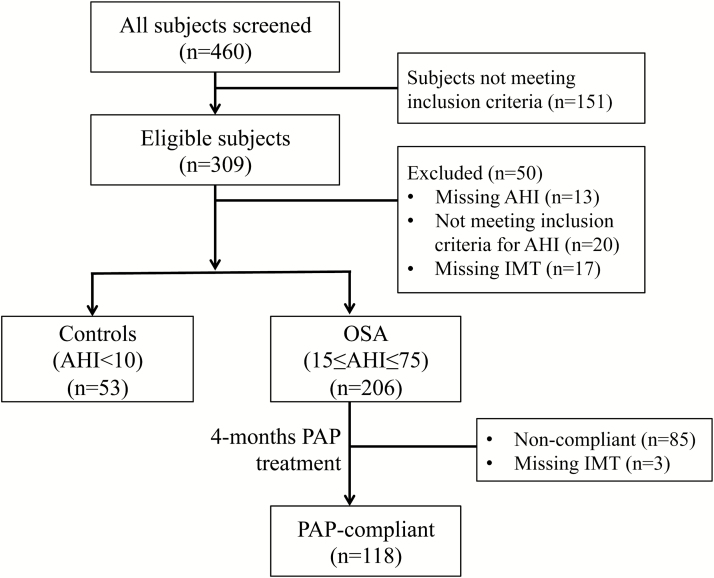
A full-night diagnostic polysomnogram (PSG) was performed in all participants. OSA case or control status was defined based on the apnea–hypopnea index (AHI). Participants with OSA were required to have an AHI between 15 and 75 events/hour, and controls were individuals with an AHI < 10 events/hour; otherwise they were excluded from the study. Other baseline assessments included bilateral carotid ultrasound; 24-hour ambulatory blood pressure monitoring (SpaceLabs Model 90217-1A, Spacelabs Healthcare, Snoqualmie, WA); and completed questionnaires on demographic information, medical conditions, and current medications, and smoking and drinking habits. Blood samples for glucose and cholesterols including triglycerides and high density lipoprotein (HDL) and low density lipoprotein cholesterols were collected in a fasting state and analyzed at the University of Pennsylvania for all participants. A total of 206 OSA cases and 53 controls had available baseline carotid IMT measurements (see Figure 1 and Table 1). Assessments were repeated in the participants with OSA who completed 16-week PAP treatment and had an average daily PAP use over the follow-up (144 ± 36 days) period of at least 4 hours/day. A total of 118 OSA participants adherent to treatment had available carotid IMT data at baseline and follow-up. Individuals not adherent with PAP were not assessed at follow-up. Figure 1 shows the flow of study participants from screening to follow-up; final numbers of participants included in analyses may vary slightly due to a small amount of missing data on covariates included as adjustments in statistical models.
Figure 1.
Flow diagram of participant participation. AHI = apnea-hypopnea index; IMT = intima-media thickness; OSA = obstructive sleep apnea; PAP = positive airway pressure.
Table 1.
Summary of Participant Characteristics at Baseline.
| Variable | Overall | Controls (AHI < 10) | OSA (15 ≤ AHI ≤ 75) | p † | |||
|---|---|---|---|---|---|---|---|
| N* | Estimate | N* | Estimate | N* | Estimate | ||
| Age, years | 259 | 53.4 ± 6.8 | 53 | 53.1 ± 6.7 | 206 | 53.4 ± 6.8 | .722 |
| Caucasian, % | 256 | 74.6% | 53 | 66.0% | 203 | 76.8% | .107 |
| Male, % | 259 | 81.9% | 53 | 67.9% | 206 | 85.4% | .003 |
| BMI, kg/m2 | 257 | 30.9 ± 4.4 | 53 | 28.1 ± 4.2 | 204 | 31.6 ± 4.2 | <.0001 |
| Waist circumference, cm | 257 | 106.9 ± 12.4 | 53 | 98.5 ± 12.5 | 204 | 109.1 ± 11.3 | <.0001 |
| Hypertension, % | 251 | 45.4% | 51 | 27.5% | 200 | 50.0% | .004 |
| CAD, % | 246 | 4.1% | 50 | 0.0% | 196 | 5.1% | .103 |
| Heart failure, % | 253 | 1.6% | 52 | 1.9% | 201 | 1.5% | .824 |
| Diabetes, % | 254 | 11.0% | 52 | 7.7% | 202 | 11.9% | .390 |
| Glucose | 245 | 114.5 ± 31.9 | 51 | 104.4 ± 15.7 | 194 | 117.1 ± 34.4 | .011 |
| Cholesterol measures | |||||||
| HDL | 245 | 49.9 ± 17.4 | 51 | 56.9 ± 19.2 | 194 | 48.1 ± 16.5 | .001 |
| Triglycerides | 245 | 131.1 ± 65.6 | 51 | 113.3 ± 51.1 | 194 | 135.8 ± 68.2 | .029 |
| CLDL | 244 | 122.6 ± 32.1 | 51 | 118.7 ± 30.7 | 193 | 123.6 ± 32.4 | .336 |
| Total cholesterol | 245 | 198.9 ± 37.4 | 51 | 198.3 ± 31 | 194 | 199.1 ± 39 | .886 |
| Current smoker, % | 250 | 19.2% | 49 | 8.2% | 201 | 21.9% | .029 |
| Current alcohol, % | 259 | 75.7% | 53 | 66.0% | 206 | 78.2% | .067 |
| AHI, events/hour | 259 | 28.9 ± 18.6 | 53 | 4.1 ± 2.7 | 206 | 35.3 ± 15.3 | <.0001 |
| ODI 4%, events/hour | 222 | 18.7 ± 15.4 | 47 | 2 ± 2.2 | 175 | 23.2 ± 14.3 | <.0001 |
| % time SpO2 < 90% | 233 | 5.4 ± 9.5 | 46 | 0.7 ± 2.6 | 187 | 6.6 ± 10.2 | .0001 |
| Minimum SpO2 (%) | 252 | 80.9 ± 8.2 | 53 | 87.9 ± 4.4 | 199 | 79.1 ± 8 | <.0001 |
| Medication use | |||||||
| Antihypertensive | 258 | 37.2% | 52 | 21.2% | 206 | 41.3% | .007 |
| Lipid-lowering | 258 | 39.9% | 52 | 40.4% | 206 | 39.8% | .939 |
| Hypoglycemic | 258 | 10.9% | 52 | 9.6% | 206 | 11.2% | .748 |
AHI = apnea−hypopnea index; BMI = body mass index; CAD = coronary artery disease; CLDL = calculated low-density lipoprotein; HDL = high density lipoprotein; ODI = oxygen desaturation index; OSA = obstructive sleep apnea; SpO2 = pulse oximeter oxygen desaturation.
*Sample size with available data;
† p-value from t test or chi-square test comparing obese versus nonobese.
Diagnostic PSG
Overnight PSGs were performed using standard techniques according to American Academy of Sleep Medicine (AASM) 2007 clinical practice guidelines.21 Standard operating procedures were implemented at the two clinical sites to ensure uniform data collection. Briefly, the following signals were recorded: electroencephalograms (C3-M2, C4-M1, O1-M2, O2-M1), bilateral electroculograms, electromyograms of the chin and right and left anterior tibialis, movement of the rib cage and abdomen (respiratory inductance plethysmography or piezoelectric belts), oxygen saturation (SpO2) by pulse oximetry, electrocardiogram, and body position. Airflow was assessed by nasal airway pressure. All recordings were scored manually with the aid of computer software (Natus Medical Inc, Pleasanton, CA) at the University of Pennsylvania Clinical Research Center for Sleep. Apneas were defined as an absence of airflow (≥90% reduction from baseline) on the nasal pressure cannula for at least 10 seconds. Hypopneas were defined as a greater than 50% reduction from baseline in airflow for at least 10 seconds associated with at least a 3% oxygen desaturation and/or an arousal. AHI was calculated as the mean number of apneas and hypopneas per hour of sleep. The oxygen desaturation index (ODI) was calculated as the number of oxygen desaturations of at least 4% per hour of sleep.
PAP Treatment
Following education about OSA and its treatment options, participants were initiated on either continuous or auto-adjusting PAP (S9 PAP apparatus, ResMed, Inc.). Participants started on continuous PAP performed an overnight PSG according to current clinical practice guidelines to determine the appropriate pressure setting. The PAP device download was used to monitor the participants’ average daily adherence to PAP treatment and the treatment’s efficacy. There were no restrictions regarding type of mask interface, and participants were allowed to change the size and model of their mask interface throughout the study.
Carotid Artery Measurements
Common carotid arteries (CCA) were assessed by B-mode ultrasound according to a standardized protocol.22 Analysis of the images was performed at the University of Pennsylvania Vascular Research Centers by an experienced technician, blinded to participant characteristics. Images of distal CCA were obtained at far walls ≈1 cm proximal to bifurcation on both sides. Distal CCA has been associated with the least variability in measurement.22 The minimum, mean, and maximum values of IMT were calculated on both right and left CCA, separately. IMT was measured at the baseline assessment in participants without OSA and at baseline and following 4 months of PAP treatment in participants with OSA who had adequate PAP adherence.
As supplemental measurements of carotid artery wall characteristics, we computed right and left CCA wall mass using the following formula: ρL(πRe2 −πRi2), where ρ is the arterial wall density (ρ = 1.06), L is the length of the arterial segment (L = 1 cm), and Re and Ri are the mean external and internal radius, respectively.23 Bilateral circumferential wall stress was calculated as (mean blood pressure × internal diameter)/2 × wall thickness according to Lamé’s equation.24,25
Obesity Measurements
Participants were classified as obese or nonobese based on their waist circumference at baseline. While participants were standing with feet together, the waist circumference was taken horizontally around the abdomen at the midpoint between the highest point of the iliac crest and lowest part of the costal margin in the mid-axillary line with the Gulick II Tape Measure (model 67020). Measurements were taken at the end of a normal exhalation and recorded to the nearest 0.1 cm. Two measures that were within 0.5 cm were averaged. Waist circumference was chosen as an a priori definition of obesity in this study in order to better capture visceral obesity level, which is often used as a clinical determinant of OSA and associated with increased risk for cardiometabolic diseases.26,27 Specifically, men and women were defined as obese if they had a waist circumference >107 cm and >96 cm, respectively, and nonobese otherwise.
Statistical Analysis
Continuous variables are summarized using means and standard deviations and categorical variables with frequencies and percentages. Demographic characteristics are compared between groups (eg, controls vs. OSA) using t tests (continuous) and chi-squared tests (categorical).
For assessing an overall effect of OSA, combining left- and right-sided measures, we utilized linear mixed models controlling for multiple measures per individual. To assess the effect of OSA on baseline carotid artery measures on the left and right side, separately, we utilized linear regression models. Both models examined whether there were differences in IMT measures between OSA patients and controls, controlling for relevant covariates. Covariates of interest, specified a priori, included: study site (Iceland/Penn), age, BMI, gender, race, smoking status, alcohol use, HDL, fasting glucose, lipid-lowering medication use, and the number of antihypertensive drugs (0, 1, or ≥2). In addition to the effects in the overall population, we also examined the effect of OSA within obese and nonobese patients separately.
To examine the effect of PAP treatment on changes in carotid atherosclerosis, we first calculated participant-specific change scores as follow-up minus baseline values. Then, among OSA participants adherent to PAP, we used linear (left, right) and mixed (combined) regression models with change score as the outcome. To determine whether there was a significant change from baseline, we tested the significance of the model intercept term; in an unadjusted linear regression model, this is equivalent to a paired t test. To assure a meaningful interpretation of the intercept term and provide a valid assessment of the within group PAP effect, covariates were mean centered at zero. Models in all participants were adjusted for the covariates listed above, in addition to baseline AHI. To test whether obesity had a significant impact on carotid artery measure changes on PAP, we tested whether change scores were different in obese and nonobese participants, using similar methods to those described for examination of the OSA effect on baseline measures.
Analyses were performed using SAS software, Version 9.4 (SAS institute, Cary, NC). Given six a priori measures of carotid artery atherosclerosis examined (ie, average IMT, maximum IMT, minimum IMT, diameter, arterial wall mass, circumferential stress), statistical significance was based on a Bonferroni corrected p-value of .0083; a p < .05 was considered suggestive evidence for an association.
RESULTS
Participant Characteristics
Table 1 compares baseline characteristics of participants with OSA and control participants. A total of 206 OSA cases and 53 controls had available baseline carotid IMT measurements. While the two groups were similar in age and race, the OSA group had more men than the control group (85.4% vs. 67.9%; p = .003). The OSA group showed significantly higher BMI and waist circumference compared to the control group. The OSA group also had a higher prevalence of hypertension and use of antihypertensive medications. No differences between groups were present in the prevalence of other diseases, including coronary artery disease, heart failure, and diabetes, although OSA participants had higher fasting blood glucose levels compared to controls. Use of lipid-lowering medications was also similar between the two groups, but the OSA group showed significantly lower HDL and higher triglyceride values than the control group. Using the criteria of waist circumference, a total of 110 participants (73 participants with OSA and 37 controls) were classified as nonobese and 147 participants (131 OSA and 16 controls) as obese. OSA patients generally had severe disease, with a mean AHI of 35.3 ± 15.3 events/hour, an ODI of 23.2 ± 14.3 events/hour, and percentage time with SpO2 < 90% of 6.6 ± 10.2%, and a minimum SpO2 of 79.1 ± 8.0%. Controls showed a mean AHI of 4.1 ± 2.7. In those who completed the 16-week treatment, residual AHI was 5.5 ± 5.2 events/hour and ODI was 2.3 ± 2.4 events/hour. A total of 118 OSA participants adherent to treatment had available carotid IMT data at baseline and follow-up.
Baseline Carotid Artery Measures Between Participants With OSA and Controls
Covariate-adjusted estimates of carotid artery variables at baseline in the OSA and control groups are shown in Table 2. As discussed in the methods, we compared the variables on the left and right sides together through mixed-model analyses, as well as in left and right CCA separately using linear regression. There were no significant differences in carotid IMT, diameter, or arterial wall mass between the two groups. OSA participants had a higher level of circumferential stress on the left side compared to controls that was of borderline significance (p = .026), but the difference was not significant on the right side (p = .291) or in the combined analysis (p = .066). Further, we examined the differences in carotid artery variables separately within nonobese (Table 3) and obese (Table 4) participants. Using the criteria of waist circumference, a total of 110 participants (73 participants with OSA and 37 controls) were classified as nonobese and 147 participants (131 OSA and 16 controls) as obese. The difference in circumferential stress between OSA participants and controls was seen primarily in obese participants, with nominally higher circumferential stress levels in obese OSA participants versus obese controls in combined analyses (63.6 vs. 55.7; p = .044), although not meeting the Bonferroni corrected p value for significance and trends toward higher stress on both the right (p = .083) and left (p = .060) sides. As in the overall sample, there were no significant differences between those with and without OSA for other measures (IMT, diameter, arterial wall mass) when stratified by obesity.
Table 2.
Adjusted Comparisons of Carotid Artery Measurements at Baseline.
| Measure | Controls (AHI < 10) | OSA (15 ≤ AHI ≤ 75) | p ‡ | ||
|---|---|---|---|---|---|
| N* | Estimate (95% CI)† | N* | Estimate (95% CI)† | ||
| Combined | |||||
| Average IMT (mm) | 47 | 0.70 (0.66, 0.74) | 191 | 0.69 (0.67, 0.71) | .716 |
| Maximum IMT (mm) | 47 | 0.74 (0.70, 0.78) | 191 | 0.73 (0.71, 0.75) | .671 |
| Minimum IMT (mm) | 47 | 0.65 (0.62, 0.69) | 191 | 0.65 (0.63, 0.67) | .904 |
| Diameter (mm) | 47 | 7.22 (7.04, 7.41) | 191 | 7.30 (7.22, 7.39) | .461 |
| Arterial wall mass | 47 | 0.86 (0.80, 0.93) | 191 | 0.87 (0.84, 0.89) | .984 |
| Circumferential stress | 47 | 58.97 (54.95, 63.00) | 178 | 63.33 (61.37, 65.30) | .066 |
| Right side | |||||
| Average IMT (mm) | 47 | 0.68 (0.64, 0.72) | 187 | 0.68 (0.66, 0.70) | .928 |
| Maximum IMT (mm) | 47 | 0.72 (0.67, 0.76) | 187 | 0.72 (0.70, 0.74) | .892 |
| Minimum IMT (mm) | 47 | 0.63 (0.59, 0.68) | 187 | 0.64 (0.62, 0.66) | .828 |
| Diameter | 47 | 7.29 (7.08, 7.50) | 187 | 7.30 (7.20, 7.40) | .902 |
| Arterial wall mass | 47 | 0.85 (0.78, 0.91) | 187 | 0.85 (0.82, 0.88) | .924 |
| Circumferential stress | 47 | 61.23 (56.65, 65.80) | 174 | 64.06 (61.83, 66.29) | .291 |
| Left side | |||||
| Average IMT (mm) | 46 | 0.72 (0.67, 0.77) | 187 | 0.71 (0.68, 0.73) | .561 |
| Maximum IMT (mm) | 46 | 0.77 (0.71, 0.82) | 187 | 0.74 (0.72, 0.77) | .475 |
| Minimum IMT (mm) | 46 | 0.68 (0.63, 0.73) | 187 | 0.67 (0.64, 0.69) | .747 |
| Diameter (mm) | 46 | 7.20 (6.98, 7.41) | 187 | 7.32 (7.22, 7.42) | .299 |
| Arterial wall mass | 46 | 0.89 (0.81, 0.96) | 187 | 0.89 (0.85, 0.92) | .974 |
| Circumferential stress | 46 | 56.54 (52.05, 61.03) | 174 | 62.43 (60.27, 64.59) | .026 |
AHI = apnea-hypopnea index; CI = confidence interval; IMT = intima-media thickness; OSA = obstructive sleep apnea.
*Sample size with available data;
†Linear (right, left) or mixed (combined) model derived estimated value of measure at baseline, adjusted for site, BMI, age, gender, race, current smoking status, current alcohol use, HDL, lipid lowering medication use, number of antihypertensive medications (0, 1, or ≥2), and glucose;
‡ p-value comparing controls and OSA at baseline.
Table 3.
Adjusted Comparisons of Carotid Artery Measurements at Baseline Among Nonobese* Participants.
| Measure | Controls (AHI < 10) | OSA (15 ≤ AHI ≤ 75) | p § | ||
|---|---|---|---|---|---|
| N † | Estimate (95% CI)‡ | N † | Estimate (95% CI)‡ | ||
| Combined | |||||
| Average IMT (mm) | 32 | 0.66 (0.61, 0.71) | 68 | 0.69 (0.66, 0.71) | .386 |
| Maximum IMT (mm) | 32 | 0.70 (0.65, 0.75) | 68 | 0.72 (0.69, 0.75) | .513 |
| Minimum IMT (mm) | 32 | 0.61 (0.57, 0.66) | 68 | 0.65 (0.62, 0.68) | .199 |
| Diameter (mm) | 32 | 7.06 (6.85, 7.27) | 68 | 7.20 (7.06, 7.33) | .317 |
| Arterial wall mass | 32 | 0.79 (0.73, 0.86) | 68 | 0.85 (0.80, 0.89) | .221 |
| Circumferential stress | 32 | 61.48 (56.82, 66.13) | 65 | 62.18 (59.13, 65.22) | .818 |
| Right side | |||||
| Average IMT (mm) | 32 | 0.65 (0.60, 0.70) | 68 | 0.69 (0.66, 0.72) | .202 |
| Maximum IMT (mm) | 32 | 0.69 (0.64, 0.74) | 68 | 0.73 (0.69, 0.76) | .263 |
| Minimum IMT (mm) | 32 | 0.6 (0.55, 0.65) | 68 | 0.66 (0.62, 0.69) | .107 |
| Diameter (mm) | 32 | 7.15 (6.90, 7.39) | 68 | 7.24 (7.08, 7.39) | .569 |
| Arterial wall mass | 32 | 0.79 (0.72, 0.87) | 68 | 0.86 (0.81, 0.91) | .171 |
| Circumferential stress | 32 | 63.59 (58.79, 68.40) | 65 | 61.52 (58.36, 64.68) | .509 |
| Left side | |||||
| Average IMT (mm) | 31 | 0.67 (0.62, 0.73) | 67 | 0.68 (0.65, 0.72) | .817 |
| Maximum IMT (mm) | 31 | 0.72 (0.66, 0.77) | 67 | 0.72 (0.68, 0.75) | .987 |
| Minimum IMT (mm) | 31 | 0.63 (0.57, 0.68) | 67 | 0.65 (0.61, 0.68) | .558 |
| Diameter (mm) | 31 | 6.95 (6.73, 7.17) | 67 | 7.15 (7.01, 7.29) | .156 |
| Arterial wall mass | 31 | 0.79 (0.72, 0.87) | 67 | 0.84 (0.79, 0.89) | .391 |
| Circumferential stress | 31 | 59.26 (53.33, 65.19) | 64 | 62.79 (58.94, 66.63) | .362 |
AHI = apnea-hypopnea index; CI = confidence interval; IMT = intima-media thickness; OSA = obstructive sleep apnea.
*Nonobese defined as a waist circumference ≤ 107 cm in men or ≤ 96 cm in women;
†Sample size with available data;
‡Linear (right, left) or mixed (combined) model derived estimated value of measure at baseline, adjusted for site, BMI, age, gender, race, current smoking status, current alcohol use, HDL, lipid lowering medication use, number of antihypertensive medications (0, 1 or ≥2), and glucose;
‡ p-value comparing controls and OSA at baseline.
Table 4.
Adjusted Comparisons of Carotid Artery Measurements at Baseline Among Obese* Participants.
| Measure | Controls (AHI < 10) | OSA (15 ≤ AHI ≤ 75) | p § | ||
|---|---|---|---|---|---|
| N † | Estimate (95% CI)‡ | N † | Estimate (95% CI)‡ | ||
| Combined | |||||
| Average IMT (mm) | 15 | 0.73 (0.66, 0.80) | 123 | 0.7 (0.68, 0.73) | .409 |
| Maximum IMT (mm) | 15 | 0.77 (0.70, 0.85) | 123 | 0.75 (0.72, 0.77) | .504 |
| Minimum IMT (mm) | 15 | 0.7 (0.63, 0.77) | 123 | 0.66 (0.64, 0.68) | .272 |
| Diameter (mm) | 15 | 7.37 (7.00, 7.73) | 123 | 7.42 (7.30, 7.54) | .790 |
| Arterial wall mass | 15 | 0.92 (0.81, 1.04) | 123 | 0.9 (0.86, 0.94) | .655 |
| Circumferential stress | 15 | 55.71 (48.59, 62.83) | 113 | 63.56 (61.05, 66.07) | .044 |
| Right side | |||||
| Average IMT (mm) | 15 | 0.72 (0.65, 0.78) | 119 | 0.68 (0.65, 0.70) | .271 |
| Maximum IMT (mm) | 15 | 0.76 (0.68, 0.83) | 119 | 0.72 (0.69, 0.75) | .394 |
| Minimum IMT (mm) | 15 | 0.69 (0.62, 0.75) | 119 | 0.63 (0.61, 0.66) | .130 |
| Diameter (mm) | 15 | 7.4 (7.02, 7.77) | 119 | 7.36 (7.24, 7.49) | .881 |
| Arterial wall mass | 15 | 0.91 (0.79, 1.03) | 119 | 0.86 (0.82, 0.90) | .395 |
| Circumferential stress | 15 | 57.38 (48.92, 65.84) | 109 | 65.41 (62.40, 68.41) | .083 |
| Left side | |||||
| Average IMT (mm) | 15 | 0.75 (0.66, 0.85) | 120 | 0.73 (0.70, 0.76) | .645 |
| Maximum IMT (mm) | 15 | 0.79 (0.69, 0.89) | 120 | 0.77 (0.73, 0.80) | .682 |
| Minimum IMT (mm) | 15 | 0.72 (0.62, 0.81) | 120 | 0.69 (0.66, 0.72) | .610 |
| Diameter (mm) | 15 | 7.34 (6.93, 7.75) | 120 | 7.46 (7.32, 7.60) | .588 |
| Arterial wall mass | 15 | 0.94 (0.79, 1.08) | 120 | 0.93 (0.88, 0.98) | .916 |
| Circumferential stress | 15 | 54.16 (46.77, 61.55) | 110 | 61.78 (59.16, 64.39) | .060 |
AHI = apnea-hypopnea index; CI = confidence interval; IMT = intima-media thickness; OSA = obstructive sleep apnea.
*Obese defined as a waist circumference >107 cm in men or >96 cm in women;
†Sample size with available data;
‡Linear (right, left) or mixed (combined) model derived estimated value of measure at baseline, adjusted for site, BMI, age, gender, race, current smoking status, current alcohol use, HDL, lipid lowering medication use, number of anti-hypertensive medications (0, 1 or ≥2), and glucose;
§ p-value comparing controls and OSA at baseline.
Changes in Carotid Artery Measurements Following PAP Treatment
Table 5 shows changes in carotid artery measures following PAP treatment among participants with OSA who were adherent to treatment, both overall and stratified by obesity status. When considering all adherent OSA participants, no significant changes in carotid IMT, arterial-wall mass, and circumferential stress were observed following PAP treatment, but there was a statistically significant increase in carotid artery diameter in the combined model (0.13 [0.06, 0.20]; p = .0004]. When assessing the left and right sides separately, carotid artery diameter significantly increased following PAP treatment on the left (mean change [95% confidence interval {CI}] = 0.18 [0.10, 0.26] mm; p < .0001) and the right side (0.08 [0.01, 0.17]; p = .046). While the increases in diameter were not significantly different between nonobese and obese OSA patients, average increases were generally larger in the nonobese (Table 5). Specifically, in nonobese OSA participants, average diameter following PAP treatment increased by 0.17 mm (95% CI: 0.05, 0.29; p = .007) when combining the left and right and 0.24 mm (95% CI: 0.10, 0.37; p = .001) on the left side.
Table 5.
Adjusted Mean Absolute Changes in Carotid Artery Measures in PAP-dherent OSA Patients Following 4 Months of Treatment, Overall and Stratified by Obesity.
| Measure | Overall | Nonobese* | Obese* | p § | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | β (95% CI)† | p ‡ | N | β (95% CI)† | p ‡ | N | β (95% CI)† | p ‡ | ||
| Combined | ||||||||||
| Average IMT (mm) | 115 | −0.003 (−0.02, 0.01) | .724 | 41 | −0.01 (−0.03, 0.02) | .550 | 74 | 0.000 (−0.02, 0.02) | .995 | .644 |
| Maximum IMT (mm) | 115 | 0.003 (−0.01, 0.02) | .711 | 41 | −0.004 (−0.03, 0.03) | .798 | 74 | 0.01 (−0.01, 0.03) | .535 | .583 |
| Minimum IMT (mm) | 115 | −0.01 (−0.02, 0.01) | .429 | 41 | −0.01 (−0.03, 0.02) | .622 | 74 | −0.01 (−0.03, 0.01) | .543 | .968 |
| Diameter (mm) | 115 | 0.13 (0.06, 0.20) | .0004 | 41 | 0.17 (0.05, 0.29) | .007 | 74 | 0.10 (0.01, 0.18) | .037 | .335 |
| Arterial wall mass | 115 | 0.01 (−0.01, 0.03) | .203 | 41 | 0.01 (−0.03, 0.05) | .535 | 74 | 0.01 (−0.01, 0.04) | .339 | .954 |
| Circumferential stress | 109 | 0.91 (−1.08, 2.90) | .367 | 38 | 2.76 (−0.73, 6.25) | .119 | 71 | −0.09 (−2.59, 2.40) | .941 | .204 |
| Right side | ||||||||||
| Average IMT (mm) | 115 | −0.01 (−0.02, 0.01) | .560 | 41 | −0.01 (−0.04, 0.02) | .366 | 74 | 0.000 (−0.02, 0.02) | .991 | .477 |
| Maximum IMT (mm) | 115 | −0.002 (−0.02, 0.02) | .811 | 41 | −0.01 (−0.05, 0.02) | .389 | 74 | 0.01 (−0.02, 0.03) | .690 | .370 |
| Minimum IMT (mm) | 115 | −0.01 (−0.03, 0.01) | .348 | 41 | −0.01 (−0.05, 0.02) | .389 | 74 | −0.01 (−0.03, 0.02) | .650 | .678 |
| Diameter (mm) | 115 | 0.08 (0.001, 0.17) | .046 | 41 | 0.12 (−0.03, 0.26) | .117 | 74 | 0.06 (−0.05, 0.17) | .272 | .545 |
| Arterial wall mass | 115 | 0.004 (−0.02, 0.03) | .728 | 41 | −0.002 (−0.05, 0.04) | .942 | 74 | 0.01 (−0.02, 0.04) | .644 | .750 |
| Circumferential stress | 109 | 1.23 (−1.10, 3.56) | .297 | 38 | 3.46 (−0.62, 7.54) | .096 | 71 | −0.09 (−3.01, 2.83) | .951 | .177 |
| Left side | ||||||||||
| Average IMT (mm) | 113 | 0.00 (−0.02, 0.02) | .996 | 40 | −0.001 (−0.04, 0.03) | .959 | 73 | 0.000 (−0.03, 0.02) | .977 | .981 |
| Maximum IMT (mm) | 113 | 0.01 (−0.01, 0.03) | .431 | 40 | 0.01 (−0.03, 0.05) | .698 | 73 | 0.01 (−0.02, 0.04) | .550 | .973 |
| Minimum IMT (mm) | 113 | −0.004 (−0.02, 0.02) | .708 | 40 | 0.001 (−0.03, 0.03) | .949 | 73 | −0.01 (−0.03, 0.02) | .582 | .719 |
| Diameter (mm) | 113 | 0.18 (0.10, 0.26) | <.0001 | 40 | 0.24 (0.10, 0.37) | .001 | 73 | 0.13 (0.04, 0.23) | .008 | .234 |
| Arterial wall mass | 113 | 0.02 (−0.003, 0.05) | .077 | 40 | 0.03 (−0.02, 0.07) | .253 | 73 | 0.02 (−0.01, 0.05) | .269 | .786 |
| Circumferential stress | 107 | 0.59 (−1.57, 2.75) | .587 | 37 | 2.09 (−1.74, 5.91) | .281 | 70 | −0.09 (−2.81, 2.62) | .945 | .374 |
CI = confidence interval; IMT = intima-media thickness; OSA = obstructive sleep apnea; PAP = positive airway pressure.
*Nonobese or obese defined using a waist circumference cut-point of >107 cm in men or >96 cm in women;
†Linear (right, left) or mixed (combined) model derived estimate of within group change from baseline on PAP (calculated as follow-up minus baseline values), adjusted for site, AHI, age, gender, race, current smoking status, current alcohol use, HDL, lipid lowering medication use, number of antihypertensive medications (0, 1 or ≥2), glucose, and BMI (overall model only);
‡ p value testing whether within group change on PAP is significantly different from zero;
§ p value comparing changes in nonobese versus obese OSA.
DISCUSSION
To the best of our knowledge, this is the largest study examining the effect of OSA and PAP on carotid atherosclerosis. We found no significant difference, after adjusting for known cardiovascular risk factors, in carotid IMT, diameter, or arterial-wall mass between participants with untreated moderate-to-severe OSA and controls (AHI < 10 events/hour), overall or within obese and nonobese participants separately. There was suggestive evidence of an association between AHI and circumferential stress, particularly in the more obese participants. In those individuals in the OSA group who were adherent to PAP treatment, no significant changes were observed in carotid IMT, arterial-wall mass, or circumferential stress. Only the diameter of carotid arteries significantly increased over the 4-month treatment.
Whether OSA itself is responsible for an increased risk for carotid atherosclerosis remains controversial, although several studies show a tendency for increased carotid IMT in OSA compared to non-OSA participants.11–13 Potential mechanisms for the OSA effect on increased IMT is that oxidative stress, due to the intermittent hypoxia reoxygenation in OSA, can produce oxygen-free radicals that result in increased proinflammatory cytokines and coagulating factors4,5 that are known risk factors for inflammation and atherosclerosis. However, OSA patients often have comorbid conditions including hypertension, obesity, and hyperlipidemia, that are also critical factors accelerating the progress of atherosclerosis. Hence, to test the independent effects of OSA, it is important to minimize the confounding effects of concomitant cardiovascular risk factors.
In a recent review, Nadeem et al.28 analyzed 16 studies on the association between OSA and carotid IMT and concluded that OSA patients had significantly greater IMT than controls. However, the independent effect of AHI on IMT was modest, and only a few of the 16 studies investigated the possible confounding effect of other cardiovascular risk factors by including controls with hypertension or metabolic syndrome. In addition, most studies in the analysis examined cross-sectional associations with relatively small sample sizes—less than 40 in each group. Therefore, controversy persists on the independent effect of OSA per se on carotid atherosclerosis.
The present study compared carotid IMT between 206 participants with moderate-to-severe OSA and 53 controls. After adjusting for relevant confounders, we found no significant difference in carotid IMT or diameter between participants with OSA and controls in either obese or nonobese participants. These results are consistent with some previous studies. Gorzewska et al.29 studied 28 nonobese OSA patients with no history of cardiovascular disease or significant abnormalities in glucose, hemoglobin A1c, and lipids. No significant differences were reported in carotid IMT between OSA cases and controls, and there were no associations between IMT versus AHI and the level of oxygen desaturation. In addition, a 13-year observational study recently reported that baseline AHI was not a significant predictor of future increased IMT but did find a relationship with future carotid plaque.30 While we found no difference in carotid IMT between participants with moderate-to-severe OSA and controls, there was a suggestive (p < .05) association between OSA and circumferential stress, primarily among obese individuals. Enlargement of luminal diameter and blood-flow change is associated with higher tensile stress in the artery walls.17,31 In our study, however, CCA diameter was not significantly different between OSA participants and controls. One can speculate that the increased levels of circumferential wall stress may be a result of a combination of geometric remodeling of the wall and blood pressure effects among participants with OSA. Future studies should attempt to replicate the suggestive association between circumferential stress and OSA, as well as explore these possible mechanisms.
Previous studies on the effect of PAP treatment on carotid IMT in participants with OSA also report conflicting results. Drager et al.6 randomly assigned 24 patients with severe OSA who were free of comorbidities to receive no treatment or CPAP and found a significant reduction in carotid IMT after 4 months of treatment. Hui et al.16 showed a significant improvement of carotid IMT following 6 and 12 months of CPAP treatment. In a recent study, however, reduction in carotid IMT following 3-month CPAP treatment was not significant.17 It is unclear whether the results in the latter study may have been due to the shorter treatment period. In the current study, there was no significant decrease in carotid IMT and arterial-wall mass over the 4-month treatment period among adherent patients. Furthermore, we did not find any difference in PAP effect on wall thickness between obese and nonobese participants. An unexpected finding of the present study was a significant increase in CCA diameter after PAP treatment. When we separated data into the left and right artery and by obesity level, the increase in diameter was significant in the left carotid artery and the direction of effect was similar on the right side. Although the change was not significantly different between obese and nonobese OSA patients, the diameter increase was larger and only statistically significant within the nonobese. One can speculate that the negative effect of obesity limits the impact of PAP on improving CCA diameter; however, results should be replicated given the relatively small sample of nonobese OSA participants. Despite the increase of diameter, there was no statistically significant change in circumferential stress after treatment. The mechanisms behind the increase in diameter are unknown. Intermittent hypoxia in OSA induces endothelial dysfunction, mediated by damage of endothelial cells by increase of reactive oxygen species and superoxide production, thus suppressing the endothelium-dependent vasodilation due to the suppressing of the endothelial nitric oxide.32 In addition, fragmentation of sleep by end-apneic arousals induces prolonged sympathetic nerve activation which is associated with alterations in nitric oxide synthase and vasodilation.33,34 Therefore, it is speculated that PAP treatment may improve endothelial repair capacity, thus, increasing internal elastic lamina in the present study. However, few studies make similar comparisons of diameter change in carotid arteries after PAP treatment in OSA patients, and a small change in diameter (0.12~0.30 mm) in the present study makes it difficult to assert clinical significance. Drager et al.6 showed no change in carotid artery diameter following PAP treatment for 4 months, inconsistent with our findings. Further studies are needed to confirm the effect of PAP on vasodilation of carotid arteries.
Our study has some limitations. First, nonapneic controls and non-PAP-adherent OSA patients in the present study did not undergo follow-up examination of carotid arteries. We only performed follow-up ultrasound testing in PAP-adherent OSA participants. Thus, it is difficult to rule out alternative explanations on the change in carotid artery diameter over time or to show possible effects of PAP treatment on impeding progression of atherosclerotic changes. Second, the sample size of controls, especially obese controls, was relatively small, which may limit low statistical power to demonstrate the differences between obese and nonobese participants. Although differences between the groups in measured cardiovascular risk factors were adequately taken into account in multivariate models in the present study, residual confounding cannot be ruled out. Third, as controls in this study were also selected from the patients referred to the sleep centers for evaluation of suspected OSA, it is possible that some of the controls have heavy snoring that may increase carotid IMT by transmission of the vibration energy to the vessel walls and mitigate the difference in IMT between OSA and controls. Last, the follow-up period might have been too short to evaluate changes in carotid atherosclerosis. Elevated IMT in the vessels of carotid arteries is an early sign of carotid atherosclerosis and progresses slowly over time. Therefore, 4 months of PAP treatment may not be sufficient to observe the effect of PAP on improvement of atherosclerotic changes in the carotid arteries, and longer follow-up studies are needed.
In conclusion, the present study showed that OSA may not be an independent risk factor for carotid atherosclerosis. Comparing moderate-to-severe OSA patients with nonapneic controls, after adjustment for potential confounding factors, we observed only a borderline association with circumferential stress, primarily among the obese individuals. There were no associations with other measures of carotid atherosclerosis. Among those participants with OSA who were adherent to PAP treatment, we did not find any changes in carotid IMT, regardless of obesity level, but the luminal diameter significantly increased, particularly in the nonobese participants. Further long-term studies are needed to explore the effect of PAP on carotid remodeling, potential left-to-right side differences in this effect, and potential differences between obese and nonobese participants.
FUNDING
This study was supported by National Institute of Health (HL094307).
DISCLOSURE STATEMENT
AIP is The John L. Miclot Professor of Medicine at the University of Pennsylvania. Funds for this endowment were provided by the Philips Respironics Foundation. ESA is a consultant for Nox Medical, Reykjavik, Iceland (unrelated to manuscript). All the other authors have no conflict of interest to declare.
REFERENCES
- 1. Chami HA, Resnick HE, Quan SF, Gottlieb DJ. Association of incident cardiovascular disease with progression of sleep-disordered breathing. Circulation. 2011; 123(12): 1280–1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Hla KM, Young T, Hagen EW et al. . Coronary heart disease incidence in sleep disordered breathing: the Wisconsin Sleep Cohort Study. Sleep. 2015; 38(5): 677–684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lorenz MW, von Kegler S, Steinmetz H, Markus HS, Sitzer M. Carotid intima-media thickening indicates a higher vascular risk across a wide age range: prospective data from the Carotid Atherosclerosis Progression Study (CAPS). Stroke. 2006; 37(1): 87–92. [DOI] [PubMed] [Google Scholar]
- 4. Damiani MF, Zito A, Carratù P et al. . Obstructive sleep apnea, hypertension, and their additive effects on atherosclerosis. Biochem Res Int. 2015; 2015: 984193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Ciccone MM, Scicchitano P, Zito A et al. . Correlation between inflammatory markers of atherosclerosis and carotid intima-media thickness in obstructive sleep apnea. Molecules. 2014; 19(2): 1651–1662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi GF. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med. 2007; 176(7): 706–712. [DOI] [PubMed] [Google Scholar]
- 7. Cho JG, Witting PK, Verma M et al. . Tissue vibration induces carotid artery endothelial dysfunction: a mechanism linking snoring and carotid atherosclerosis? Sleep. 2011; 34(6): 751–757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Almendros I, Acerbi I, Puig F, Montserrat JM, Navajas D, Farré R. Upper-airway inflammation triggered by vibration in a rat model of snoring. Sleep. 2007; 30(2): 225–227. [DOI] [PubMed] [Google Scholar]
- 9. Ciccone MM, Scicchitano P, Mitacchione G et al. . Is there a correlation between OSAS duration/severity and carotid intima-media thickness? Respir Med. 2012; 106(5): 740–746. [DOI] [PubMed] [Google Scholar]
- 10. Fox N, Ayas N, Park JE et al. . Carotid intima media thickness in patients with obstructive sleep apnea: comparison with a community-based cohort. Lung. 2014; 192(2): 297–303. [DOI] [PubMed] [Google Scholar]
- 11. Altin R, Ozdemir H, Mahmutyazicioğlu K et al. . Evaluation of carotid artery wall thickness with high-resolution sonography in obstructive sleep apnea syndrome. J Clin Ultrasound. 2005; 33(2): 80–86. [DOI] [PubMed] [Google Scholar]
- 12. Yun CH, Jung KH, Chu K et al. . Increased circulating endothelial microparticles and carotid atherosclerosis in obstructive sleep apnea. J Clin Neurol. 2010; 6(2): 89–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Meng S, Fang L, Wang CQ, Wang LS, Chen MT, Huang XH. Impact of obstructive sleep apnoea on clinical characteristics and outcomes in patients with acute coronary syndrome following percutaneous coronary intervention. J Int Med Res. 2009; 37(5): 1343–1353. [DOI] [PubMed] [Google Scholar]
- 14. Salepci B, Fidan A, Ketenci SC et al. . The effect of obstructive sleep apnea syndrome and snoring severity to intima-media thickening of carotid artery. Sleep Breath. 2015; 19(1): 239–246. [DOI] [PubMed] [Google Scholar]
- 15. Gunbatar H, Sertogullarindan B, Ekin S, Akdag S, Arisoy A, Sayhan H. The correlation between red blood cell distribution width levels with the severity of obstructive sleep apnea and carotid intima media thickness. Med Sci Monit. 2014; 20: 2199–2204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hui DS, Shang Q, Ko FW et al. . A prospective cohort study of the long-term effects of CPAP on carotid artery intima-media thickness in obstructive sleep apnea syndrome. Respir Res. 2012; 13: 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kostopoulos K, Alhanatis E, Pampoukas K et al. . CPAP therapy induces favorable short-term changes in epicardial fat thickness and vascular and metabolic markers in apparently healthy subjects with obstructive sleep apnea-hypopnea syndrome (OSAHS). Sleep Breath. 2016; 20(2): 483–493. [DOI] [PubMed] [Google Scholar]
- 18. Schwartz AR, Patil SP, Laffan AM, Polotsky V, Schneider H, Smith PL. Obesity and obstructive sleep apnea: pathogenic mechanisms and therapeutic approaches. Proc Am Thorac Soc. 2008; 5(2): 185–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rocha VZ, Libby P. Obesity, inflammation, and atherosclerosis. Nat Rev Cardiol. 2009; 6(6): 399–409. [DOI] [PubMed] [Google Scholar]
- 20. Minoguchi K, Yokoe T, Tazaki T et al. . Increased carotid intima-media thickness and serum inflammatory markers in obstructive sleep apnea. Am J Respir Crit Care Med. 2005; 172(5): 625–630. [DOI] [PubMed] [Google Scholar]
- 21. Iber C, Ancoli-Israel S, Chesson AL, Quan SF. for the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, 1st ed. Westchester, IL: American Academy of Sleep Medicine, 2007. [Google Scholar]
- 22. Roman MJ, Naqvi TZ, Gardin JM, Gerhard-Herman M, Jaff M, Mohler E. Clinical application of noninvasive vascular ultrasound in cardiovascular risk stratification: a report from the American Society of Echocardiography and the Society of Vascular Medicine and Biology. J Am Soc Echocardiogr. 2006; 19(8): 943–954. [DOI] [PubMed] [Google Scholar]
- 23. Girerd X, Mourad JJ, Copie X et al. . Noninvasive detection of an increased vascular mass in untreated hypertensive patients. Am J Hypertens. 1994; 7(12): 1076–1084. [DOI] [PubMed] [Google Scholar]
- 24. Nichols W, O’Rourke MF. Aging, high blood pressure and disease in humans. In: Arnold E, ed. McDonald’s Blood Flow in Arteries: Theoretical, Experimental and Clinical Principles. 3rd ed. London, United Kingdom: Oxford University Press; 1990: 398–420. [Google Scholar]
- 25. Fung Y. Bioviscoelastic solids. Mechanical Properties of Living Tissues. 2nd ed New York, NY: Springer Verlag, 1993:242–320. [Google Scholar]
- 26. Bouchi R, Takeuchi T, Akihisa M et al. . High visceral fat with low subcutaneous fat accumulation as a determinant of atherosclerosis in patients with type 2 diabetes. Cardiovasc Diabetol. 2015; 14: 136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Luchnikova E, Shchekotov V. 7A.05: Visceral fat level determined using the bioelectrical impedance as a method to assess obstructive sleep apnea risk. J Hypertens. 2015; 33 Suppl 1: e90. [Google Scholar]
- 28. Nadeem R, Harvey M, Singh M et al. . Patients with obstructive sleep apnea display increased carotid intima media: a meta-analysis. Int J Vasc Med. 2013; 2013: 839582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Gorzewska A, Specjalski K, Drozdowski J et al. . Intima-media thickness in patients with obstructive sleep apnea without comorbidities. Lung. 2013; 191(4): 397–404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gunnarsson SI, Peppard PE, Korcarz CE et al. . Obstructive sleep apnea is associated with future subclinical carotid artery disease: thirteen-year follow-up from the Wisconsin sleep cohort. Arterioscler Thromb Vasc Biol. 2014; 34(10): 2338–2342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kappus RM, Fahs CA, Smith D et al. . Obesity and overweight associated with increased carotid diameter and decreased arterial function in young otherwise healthy men. Am J Hypertens. 2014; 27(4): 628–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Lavie L. Oxidative stress inflammation and endothelial dysfunction in obstructive sleep apnea. Front Biosci. 2012; 4: 1391–1403. [DOI] [PubMed] [Google Scholar]
- 33. Ip MS, Lam B, Chan LY et al. . Circulating nitric oxide is suppressed in obstructive sleep apnea and is reversed by nasal continuous positive airway pressure. Am J Respir Crit Care Med. 2000; 162(6): 2166–2171. [DOI] [PubMed] [Google Scholar]
- 34. Teramoto S, Kume H, Matsuse T et al. . Oxygen administration improves the serum level of nitric oxide metabolites in patients with obstructive sleep apnea syndrome. Sleep Med. 2003; 4(5): 403–407. [DOI] [PubMed] [Google Scholar]