Abstract
After traumatic events (accidental or surgical), the respiratory tract activates specific and prolix repairing mechanisms which tend to claw back the primitive differentiated state. The attempt of reactivation of the normal tissue functions is called ‘remodeling’ and its aim is to reinstate the modeling mechanisms that existed before the damaging event or the pathology’s establishment. Endoscopic sinus surgery represents the gold standard treatment for inflammatory, malformative, benign, and, in selected cases, malignant diseases. The surgical technique is commonly described as minimally invasive as the nostrils are used as an access route and therefore does not leave any external scars. Currently, the surgical procedures, even though minimally invasive regarding the way in, are in fact widely destructive towards the surgical target. The healing process and re-epithelialization will depend on the amount of bony tissue that has been exposed and it will be important to stratify the different surgical typologies in order to foresee the increasing difficulty of mucosal healing process. As far as upper inflammatory diseases are concerned, recent studies demonstrated how intranasal hyaluronic acid can positively regulate mucosal glands secretion and modulate inflammatory response, being a useful tool for the improvement of remodeling after endoscopic sinus surgery. Acid has shown to be able to regulate mucosal glands secretion and modulate the inflammatory response.
Keywords: embryogenesis, endoscopic sinus surgery, hyaluronan, mucociliary transport, remodeling, rhinosinusitis
Introduction
Acquisition, maintenance, and loosening of differentiated state
During embryogenesis the tissue components of the respiratory tract differentiate from endo, meso, and ectodermal origin cellular clones that, due to a projected sequence of interactions, gain a specific differentiated state, or terminal phenotype, giving birth to more than 40 different cellular populations. These cellular clones are represented by proto-differentiated cells that arise from endodermic-lined staminal cells and are identifiable in pre-respiratory epitheliums, together with basal cells who have the possibility to differentiate themselves into calciform, ciliate or, ‘brush cells’, granting the normal cellular epithelial turnover (Figure 1). Lately, over the course of the life, the differentiated tissue’s state is maintained by a prolixcontrol-system that requires the continuous production of various metabolites, growth factors, and hormones. The peripheral nervous system contributes to modulate basal cells differentiation process as well, through the colinergic nerve-endings that are conditioned by the release of P-substance (PS) and calctinonin gene-related protein (CGRP).
Figure 1.
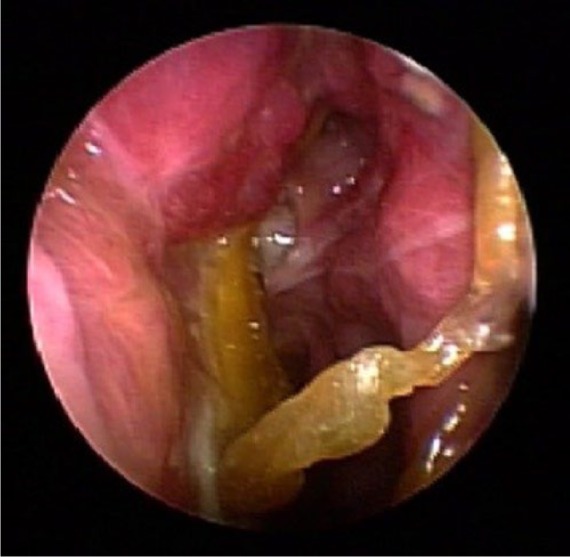
Endoscopic view without therapy.
Modeling and remodeling
The continuous structural remodeling is nourished by a molecular coincidental turnover that takes place together with general omeostasis and that is called ‘post-differentiation tissue modeling’ or simply ‘modeling’. Respiratory function itself, telomeres ageing and specific pathologies, lead the respiratory tract to a progressive loosening of the differentiated state assessed through phenotypes specific markers expression. This may happen during pathological processes in which their ‘natural history’ can express different tissue states, not compatible with the exploitation of a normal physiological function. Similarly, after traumatic events (accidental or surgical), the respiratory tract activates specific and prolix repairing mechanisms which tend to claw back the primitive differentiated state. The attempt of reactivation of the normal tissue functions is called ‘remodeling’ and its aim is to reinstate the modeling mechanisms that existed before the damaging event or the pathology’s establishment. As a rule, a para-physiological condition that grants a partial and acceptable reactivation of the tissue’s functions is reached through the remodeling process. However, there are pathological situations in which the characteristic symptoms create various degrees of dysfunction (Figure 2). That’s why remodeling is a crucial phenomenon, and its abnormal process may lead to either the lost or to the missed reacquisition of the ancient differentiated state. To date there are no therapeutic approaches oriented on the regulation of the remodeling process. However, on the basis of many experimental results and observations, also hyaluronan seems to play a central role on pre-respiratory tissue physiology by helping to define some fundamental mechanisms.1
Figure 2.
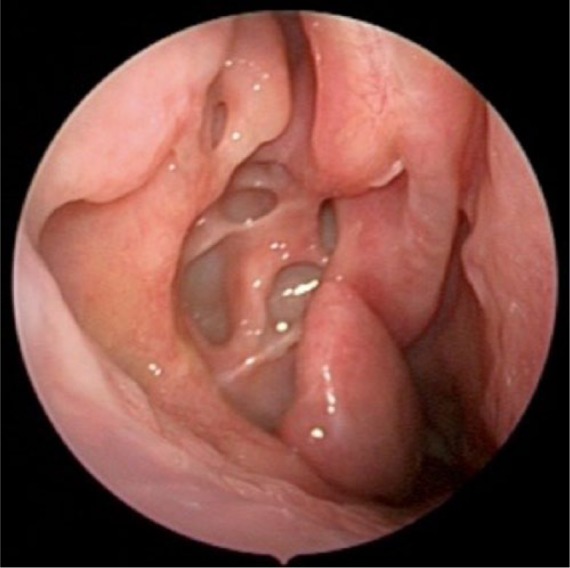
Anatomo-pathological characteristics of remodeling.
Anatomo-pathological characteristics of remodeling
Remodeling is an elaborate dynamic process which depends on many variables and develops at various intervals and in different ways, according to the originating causes. Remodeling, which may may be influenced by previous events either in perinatal/postnatal period or occurring in the first years of life, may also depend on duration and intensity of the related flogosis and on the complexity of the produced immunitary response. Although remodeling is an individual and genetically defined tissue response, into the respiratory tract (in particular the upper airways tract) it shows some particular recurring characteristics such as:
- Epithelial hyperplasia that may be associated to significant structural changes. These may determine a decreased upper respiratory tract air flow.
- Loosening of epithelial differentiation with impairment of specific functions, as the decreased production of tensioactive and defense factors, partial deactivation of mucociliary clearance, thickening of extracellular matrix of the submucosal space, attributable to an imbalance between synthesis and demolition of its macromolecular components; all the above mentioned factors cause unavoidable consequences on microvascular and neurosensorial functions.
- Heap of plasmatic proteins, like potentially flogistic catabolites and edemigenic proteins that are able to interfere with normal metabolism.1 It is important to remember that hyaluronan is produced from serous glands of the submucosal layer and once expelled amplifies the mucociliary clearance and concentrates inside of the vascular adventitia and basal membrane, where it assimilates with extracellular matrix carrying out an important anti-edemigenic action.2–4
Pre-respiratory mucosal mechanisms for differentiation loosening
The mucosal differentiated state maintenance is based on the continuous cellular interaction established between the epithelial component and the fibroblasts of the submucosal layer. This interaction is based on bidirectional exchanges between signal-molecules like TGF-β and E-prostaglandine. The adulterated epithelial production of suppressive or proliferative factors and/or the fibroblasts’ inability in answering in an adequate way may change the normal molecular cross-talk and determine an abnormal remodeling. Moreover the unavoidable oxidative stress associated with the remodeling process promotes a fibrotic healing either inducing the loss of the epithelial cellular differentiation into mesenchymal-type cells or changing the differentiation of the basal cells. In particular, respiratory tract epithelial cells in the presence of Hydrogen Peroxide modify their morphological aspect and reduce the epithelial phenotype specific markers’ production E-cadherin and ZO-1, increasing both the mesenchymal markers expression (vimentin and α-actin) and matrix extracellular components secretion. The oxidative stress itself promotes TGF-β1 expression and this can be avoided, together with the related epithelial differentiation loosening, by means of antioxidant treatments (feniltert-butylnitrone [PBN]) and the use of dismutase3 superoxid (SOD3).
Hyaluronan owns a remarkable antioxidant activity. It is released together with a small scavenger molecular pool (10,000 dalton) and with lactoferrin which is provided with an important anti-bacterial activity.
Nasal mucosa
Nasal mucosa
Nasal mucosa, defined as lung’s ‘guard dog’,5 regulates the pretreatment of inhaled air, controlling its temperature, humidity, and sterility. These functions are the result of prolix interactions between the covering epithelium and the submucosal components. The epithelial element is made of calciform cells that produce and release a tensioactive mixture that is stratified together with mucus components framing a film with pseudoplastic not Newtonian characteristics which is able to switch between a gel-shape into a solution-shape due to the metacronal beat of the nearby ciliate cells. Ciliate cells (200 lashes/cell) through 16/s coordinated swingings determine a flow that in little more than 10 min (3–25 mm/min) moves a guzzled particle from the choanal space towards the orofarynx taking advantage of the gel/solution changes of the film. Hyaluronan is the main glycosimoglycan (80%) produced by the pre-respiratory mucosa and represents the main component of the film covering the upper airways. Together with condroitin sulfate and heparan sulfate it stratifies on the mucosal epithelial cells organizing three-dimensional nets (called coating) in which mucus molecules are anchored, allowing their function without their removal by mucociliary transport.6,7
Remodeling and the nasal cycle
The main aim of remodeling is functional reinstatement. This may be evaluated through the mucosal main functions recovery as it is observed during the nasal cycle. The cycle is generated from hydrostatic pressure variations inside the sinusoidal microcirculation that leads to the opening of microchannels located in the latero-basal portions of the epithelial cells of the mucosa; this action does not compromise the barrier effect but it activates the ejection of a plasmatic flow stratifying into the nasal cavities with the function of diluting and removing unwanted material from the intercellular spaces, like sweat proteins, immunoglobulins, and allergens. As a consequence, an exuded microfilm is formed, that is considered as a real defensive line.8 This important defense mechanism may change into a pro-flogistic mechanism when it is transformed into a plasmatic extravasation that expands from the post-capillary veins to the sinusoids, producing an exude rich in proflogogen molecules. The existence of an alternate congestion-decongestion cycle is well known from over a century, even if its precise function is still unknown. Besides, its validation is not attended in all individuals9–21 and it does not seem to follow a circadian rhythm with significant differences between morning and evening.
The basement membrane
The basement membrane’s remodeling
The basement membrane is a connective, deformable and flexible matrix that establishes a microenviroment that hosts a neurovascular net and the seromucosal gland’s adenomerous. From a structural point of view it is made by proteoglycans (high and low molecular weight) able to resist against compressive forces, and three types of collagen (I, III, and IV) that unload traction forces. Its molecular constituents’ production and turnover is directly controlled by fibroblasts. In order to maintain the structure it is mandatory to have a control system that grants the balanced mix of matrix components and their programmed turnover that is not going to modify the composition. Molecular components turnover is regulated by metalloprotease (MMP). Hyaluronan is directly involved both into MMP synthesis control and their inhibition (Isnard N2001). The regulation is managed by small fragments that generate from its controlled depolymerization.15
Tissue repairing
Irrespective of the surgical trauma, a wound always determines the tissue interruption. In the injured area there are deep microscopic alterations that may lead to formation of real craters. All upper airway tissue activates the repair mechanisms finalized to restrain the lesion and restore the damaged area; all essential procedures needed for a new tissue synthesis oriented towards healing.
Restoration is a process based on many mechanisms’ sequential integration and involves tens of different cellular populations activating more than 500 genes that prime process able to reinstate the damaged tissue integrity and restore a reactivating remodeling mechanism.
This is made through a process in which progressive and partially crossed phases are recognizable; hemostasis, inflammation (through migration and cellular colonization), and tissue remodeling. The first mechanism is hemostasis.
Its efficacy is linked to the synchronous interaction of arterial and venous vessels, platelet cells, and coagulation factors. In order to activate the different phases an inertial state needs to be established inside the injured area, like a sort of isolation from chemical-mechanical stress proceeding from adjacent tissue. The final aim is to seal the area with a ‘biological cement’ that grants the essential static tissue conditions needed for a correct developing of the hemostatic phase. Platelet cells supervise hemostasis, which represents the first healing step, by taking part into the flogosis activation and the related angiogenesis. Platelet cells constitute main factors of the new healing tissue. Circulating platelet cells are in a hibernation state but when they reach the damaged tissue area they activate themselves. They radically change their morphology (transition from a ‘smooth’to ‘thorny’ form) and express on their membrane different adhesion molecules that promote a selective link with damaged areas by allowing adhesion and interaction with leukocytes and neutrophils, and by favoring their enrolment from circulating blood as well. Therefore platelet cells become leading actors in tissue repairing process. In addition to ‘neutrophil-platelet’ interaction, mostly mediated by P-selectin towards β-2 e β-3 integrins, platelet cells interact with endothelial cells and monocytes, activating pro-inflammatory cytokines production. It is necessary that all damaged tissues activate their mechanisms to prevent the potential bacterial growth from speeding into the injured area, through the production of molecule acting as ‘danger signals’ to be quickly sorted out from immunological system in order to start a controlled flogosis without interfering with healing mechanisms. This is possibly due to the development of a net made of signals responsible for automatization and fastening of tissue response produced by the coordinated action of platelet cells, neutrophils, and monocytes on tissue Hyaluronic Acid; this action, which can supported by hyaluronidase system, produces plenty of different molecular weight fragments that make up the net of needed signals.22
Role of hyaluronan in tissue repair
Tissue hyaluronan fragmentation is managed by a hyaluronidase system displaced in many cellular compartments.
Hyal 2, chained to the cellular membrane through glycosylphosfatidylinositol (GPI), acts on extracellular matrix hyaluronic; Hyal 1 and Hyal 3, present at endoplasmatic network and lysosomial level, further depolymerize hyaluronan once endocytosed.Hyaluronan is diffusely represented along all airways and mainly at nasal mucosa level, being a potential reservoir from where signal molecules for reparation can be acquired. All hyaluronidase system components can be activated by oxygen free radicals (ROS) that are profusely generated during the first phases of tissue repair process. Thus several fragments of smaller weight are formed. Low weight fragments act as ‘danger signals’ while tetrasaccharides have a great anti-apoptotic activity. While other different weight fragments, which may act as intracellular protein synthesis inductors, are able to increase the response to shock (Heat shock protein), other fragments contribute into signal molecule transduction. Pre-respiratory mucosal 3 hyaluronidases may act coordinatively: thanks to their activity on hyaluronan an ‘upregulation’ of TNF-αand IL-1 β is established, similar to what happens in asthmatic patients when stimulated by allergens.19 Hyaluronan fragments that interact with neutrophil and monocyte surface receptors (CD44, RHAMM, TLR4) can stimulate cytokines production and many flogosis mediators activation (like activation of the nuclear factor inhibitor: factor-κBα/nuclear-kB pathway) which are able to upregulate the exp-ression of interleukin-1β and TNF-α17 and the expression of many other potential flogosis mediators. From a general point of view, low-weight fragments induce activation while high-weight fragments determine suppression, being able to prevent extreme expression of flogosis as well. Hyaluronan fragments can interact with CD44 and TLR-4 and therefore regulate monocytic transformation into macrophage. Furthermore, monocytes own three ialuronidases which can generate a pool of HA-active fragments inside healing tissues.23 In particular, from the interaction with TLR4 signals are generated, which are able to start neutrophils degranulation and induce autolysis; the latter produces debris and DNA creating traps (NET) which concentrate, recall, and block other neutrophils enhancing the action.
Remodeling and endoscopic surgery
Endoscopic sinus surgery represents the gold standard treatment for many diseases: flogistic, malformative, benign, and, in selected cases, malignant neoplasms. The instruments are cutting and grasping pliers and motorized instruments, like intranasal drills and ultrasonic/mechanical aspirators. The surgical technique is commonly described as minimally invasive as it employs the nostrils as the access route and therefore does not leave external scars. Currently, the endosopic surgical procedures are carried out depending on the pathology to be treated, and even if they remain minimally invasive as far as the way in is concerned, they are widely destructive upon the surgical target. This is clearly seen during removal of malign neoformations, according to the oncological safety plane that requires the removal of the lesion together with healthy surrounding tissue. In case of inflammatory diseases the surgical procedure requires the sinusal ostium opening (frontal, sphenoidal, and maxillary) to be enlarged, if required. This grants the restoration of the two main functions: sinus ventilation and mucociliary transport, which are fundamental for the recovery of the sinus wellness.13 In order to rehabilitate the sinus ostiums it is frequently necessary to remove anatomical structures that establish the ostium prechambers, like in the case of frontal sinus. Thus, in order to open the sinus in correspondence of its natural ostium it is required to decompose its recess, by dissecting the bony structures surrounding it, like the uncinate process, the ethmoidal bulla, and the agger nasi cell. This procedure is carried out respecting the sinonasal mucosa that covers these structures so at the end of the intervention the mucosal trauma is very limited. In case of pathological pansinusal involvement the surgical procedure includes the complete dissection of all anterior and posterior ethmoidal cells, with the exposition of the ethmoidal roof. In this case the extended removal of bony structures necessarily entails a wide nasal mucosal trauma that, even if limited by using cutting and not stripping instruments, occurs in a significant way for all endoscopic procedures and even more widely if the pathology to be treated requires it. In case of benign or malign sinonasal lesions removal, the aggressiveness of the procedure progressively increases according to the sinonasal structures to be removed. The healing process and re-epithelialization will depend on the amount of bony tissue that has been exposed and it will be important to stratify the different surgical typologies in order to foresee the increasing difficulty of mucosal healing process.
Endoscopic endonasal surgery tries to be as less traumatic as possible for the nasal mucosa that gets hurt anyway. The postoperative and healing period is generally divided into an early period (first weeks) and late period (up to 6 months).11 The first one is mainly influenced by the surgical act, by the first postoperative treatments and by the pathology that was treated; the latter being relevant for the healing during the late healing period. Hoseman et al. thought that the healing process takes place in four phases: hematic crust formation (up to 10 days), lymphedema (up to 30 days), mesenchymal regeneration (up to 3 months), and finally crust formation (after 3 months). Infective process are able to inhibit the healing process and lead to a mucosal regeneration decreas.24 A wound healing is a very prolix, coordinated, and multistep process that involves the clot formation, an inflammatory reaction and at the end tissues maturation and remodeling. This process is influenced by a lot of endogenic and esogenic factors as radiations, nutrition, systemic factors, and the surgical technique that was employed. In 2004 a study has investigated how the techniques which are more commonly used in the postoperative period may alter and influence nasal healing. This study has evidenced how nasal packings positioned at the end of the surgical procedure, if left many hours inside the nostrils, can provoke an infection and promote fibrin formation, which will later become a scar. The authors therefore recommend nasal packings be removed within 24 h after surgery and suggest avoiding their indiscriminate use if intraoperative bleeding was not massive. Other devices that are used to maintain the separating space between different structures in order to avoid adherences are more frequently used when the space is very narrow and the mucosa has been seriously damaged. Furthermore it is suggested to clean the nostrils with saline solution in the immediate postoperative period (preferably using big volumes at low pressures, in order to reach the space inside paranasal sinuses without damaging the mucosa).
Crusts play an important role within the healing process and nasal and paranasal mucosa re-epithelialization.
Their presence is a natural step towards the definitive mucosal healing, but they inevitably determine a decreasing airflow inside the nostrils, and consequently the increasing probability to develop infections as well.24 Crusting defaces the normal airflow and mucociliary transport until its inhibition. This may determine a vicious cycle that causes nostrils’ dirtiness producing more crust formation and so on. Since the 1980s, several authors have stressed the concept of endoscopic postoperative medication to be performed up to 2 months after the surgical procedure. With regard to this matter it is necessary to underline the importance of the crust. The main problem occurs when the crust closes the sinus natural ostium, being a relevant cause for mucociliary transport interruption, mucosal secretion stasis, and increase in infections of the sinus. Also, the prolonged closure of the ostium facilitates the scar surplus and the consequent stenosis of the ostium itself.
Despite a general agreement on the need of postoperative nasal washings there are no guidelines yet on how these should be done. Nasal washing can stop the vicious cycle started because of the crusts’ presence, through their non-traumatic dissolution together with sticky mucus removal.24 During decades many different drugs were tested intranasally in the postoperative period in order to reduce fibrin, crusts, and consequently scars. In 1999, a study demonstrated that 0.2% Hyaluronic Acid topical administration (as nasal ointment) reduced crust formation and increased nasal airflow. During the postoperative period it is fundamental to keep the sinus ostia opened in order to support mucociliary drainage and sinus ventilation process, which are able to lead towards a considerable re-epithelialization. This result is obtained by a combination of intranasal endoscopic medications with removal of crusts obstructing sinus ostia and nasal washing using drugs able to support the cleaning from crusts and stickiness, blocking fibrin the nasal mucosa cellularity and the inflammatory status. The study carried formation and promoted re-epithelialization. Recent studies demonstrated how low-molecular-weight Hyaluronic Acid acts like a mucosal lubricant and how, in high concentrations, is able to positively influence bio-mechanical forces, hydric balance and cellular functions, growth factor activity, and cytokine behavior. Hyaluronic Acid has shown that it can regulate mucosal gland secretion and modulate the inflammatory response.
Based on these concepts we have recently performed a study in order to demonstrate that the topical administration (through the use of a nebulizer) of Hyaluronic Acid versus saline solution improves outcome on 75 pediatric patients affected by inflammatory diseases of the upper airways (rhinitis, rhinosinusitis, adenoiditis, pharyngitis, otitis media, tubotympanitis, and tonsillitis) after 3 months of treatment (divided into 15-day cycles and 15-day suspensions), and demonstrated a significant and objective improvement of mucociliary transport and of microbiological, cytological, endoscopic, and clinical status.25
Remodeling
During rhinitis and allergic rhinitis, the remodeling process shows some abnormalities on some kind of proteoglycans (lumican and biglycan).17,18 Collagens are those usually produced (I, III, and IV) but those who are at the level of the basal membrane tend to increase in quantity.26 There is also an increased expression of a metalloprotease type MMP-9 directly involved into the matrix turnover and associated to a substantial increase ofTIMP-1 andTIMP-2 mRNA. Changes in microcirculation are detected, with an increase in capacity and resistance after stimulation tests.27 The number of calciform cells inside the inferior tubinates mucosa is not influenced by either the presence of rhinitis or allergic rhinitis.28 During allergic rhinitis the natural exposition to the antigen is related to an increased nasal mucosa infiltration of T-helper and CD25+.29
Conclusion
Sinonasal wellbeing is fundamental for the quality of life of our patients and for the condition of the whole body, as the various nasal functions are necessary to allow all the related respiratory activities. Hyaluronan allows to maintain the correct trophism of the nasal mucosa, damage of the nasal mucosa and the cellular barrier alternates the nasal function, being the damage related not only to specific nasal pathologies but also to the surgical procedures used for their treatment and for the treatment of sinonasal anatomical alterations.
Currently, endoscopic endonasal surgery is considered the gold standard treatment for a variety of pathologies. However, even if a minimally invasive approach is carried out, it does not mean that treatment is minimally invasive as well. Depending on the pathology being treated, the surgical procedure employed can be really aggressive and destructive. In any case (even for less invasive procedures) the nasal mucosa is going to be damaged in various degrees and the outcome is granted also by the healing process after the surgery.
The remodeling potential of the Hyaluronan represents a fundamental element in the treatment of such conditions.
Its topical use increases and improves the healing process supporting the nasal functions restore and the mucosal trophism. Our experience confirm that the administration of Hyaluronan can protect the sinonasal epithelium from inflammatory and surgical damage although further studies are needed to strengthen the results obtained by now.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- 1. Ahmed SK, Williams JL, Drake-Lee A, et al. (2009) Staining of nasal mucosa to examine remodelling. Journal of Laryngology & Otology 123: 234–237. [DOI] [PubMed] [Google Scholar]
- 2. Banerji S, Wright AJ, Noble M, et al. (2007) Structures of the Cd44-hyaluronan complex provide insight into a fundamental carbohydrate-protein interaction. Nature Structural & Molecular Biology 14: 234–239. [DOI] [PubMed] [Google Scholar]
- 3. Baraniuk JN, Shizari T, Sabol M, et al. (1996) Hyaluronan is exocytosed from serous, but not mucous cells, of human nasal and tracheobronchial submucosal glands. Journal of Investigative Medicine 44: 47–52. [PubMed] [Google Scholar]
- 4. Bard JB, McBride WH, Ross AR. (1983) Morphology of hyaluronidase-sensitive cell coats as seen in the SEM after freeze-drying. Journal of Cell Science 62: 371–383. [DOI] [PubMed] [Google Scholar]
- 5. Geurkink N. (1983) Nasal anatomy, physiology, and function. Journal of Allergy and Clinical Immunology 72: 123–128. [DOI] [PubMed] [Google Scholar]
- 6. Kultti A, Rilla K, Tiihonen R, et al. (2006) Hyalu-ronan synthesis induces microvillus-like cell surface protrusions. Journal of Biological Chemistry 281: 15821–15828. [DOI] [PubMed] [Google Scholar]
- 7. Ganbo T, Hisamatsu K, Inoue H, et al. (1995) The effects of leukotrienes C4 and D4 on ciliary activity of human paranasal sinus mucosa in vitro. Rhinology 33: 199–202. [PubMed] [Google Scholar]
- 8. Eccles RB. (2000) The nasal cycle in respiratory defence. Acta Otorhinolaryngologica Belgica 54: 281–286. [PubMed] [Google Scholar]
- 9. Hallén H, Geisler C, Haeggström A, et al. (1996) Variations in congestion of the nasal mucosa in man. Clinical Otolaryngology & Allied Sciences 21: 396–399. [DOI] [PubMed] [Google Scholar]
- 10. Hayen W, Goebeler M, Kumar S, et al. (1999) Hyaluronan stimulates tumor cell migration by modulating the fibrin fiber architecture. Journal of Cell Science 112: 2241–2251. [DOI] [PubMed] [Google Scholar]
- 11. Hosemann W, Wigand ME, Gode U, et al. (1991) Normal wound healing of the paranasal sinuses: Clinical and experimental investigations. European Archives of Oto-Rhino-Laryngology 248: 390–394. [DOI] [PubMed] [Google Scholar]
- 12. Hostettler KE, Roth M, Burgess JK, et al. (2008) Airway epithelium-derived transforming growth factor-beta is a regulator Offibroblast proliferation in both fibrotic and normal subjects. Clinical & Experimental Allergy 38: 1309–1317. [DOI] [PubMed] [Google Scholar]
- 13. Huang PM, Syrkina O, Yu L, et al. (2010) High MW hyaluronan inhibits smoke improves inhalation-induced lung injury and survival. Respirology 15: 1131–1139. [DOI] [PubMed] [Google Scholar]
- 14. Ionov R, El-Abed A, Goldmann M, et al. (2004) Interactions of lipid monolayers with the natural biopolymer hyaluronic acid. Biochimica et Biophysica Acta 1667: 200–207. [DOI] [PubMed] [Google Scholar]
- 15. Joris L, Dab I, Quinton PM. (1993) Elemental composition of human airway surface fluid in healthy and diseased airways. American Review of Respiratory Diseases 148: 1633–1637. [DOI] [PubMed] [Google Scholar]
- 16. Isnard N, Legeais JM, Renard G, et al. (2001) Effect of hyaluronan on MMP expression and activation. Cell Biology International 25: 735–739. [DOI] [PubMed] [Google Scholar]
- 17. Kim TH, Lee JY, Lee HM, et al. (2010) Remodelling of nasal mucosa in mild and severe persistent allergic rhinitis with special reference to the distribution of collagen, proteoglycans, and lymphatic vessels. Clinical & Experimental Allergy 40: 1742–1754. [DOI] [PubMed] [Google Scholar]
- 18. Jordan S, Trigg CJ, Davies RJ. (1994) Lymphocyte infiltration and thickness of the nasal mucous membrane in perennial and seasonal allergic rhinitis. Journal of Allergy and Clinical Immunology 93: 635–643. [DOI] [PubMed] [Google Scholar]
- 19. Krasiński R, Tchórzewski H. (2007) Hyaluronan-mediated regulation of inflammation. Postepy Higieny i Medycyny Doswiadczalnej (Online) 61: 683–689. [PubMed] [Google Scholar]
- 20. Krasteva G, Kummer W. (2012) “Tasting” the airway lining fluid. Histochemistry and Cell Biology 138: 365–383. [DOI] [PubMed] [Google Scholar]
- 21. Krejcova D, Pekarova M, Safrankova B, et al. (2009) The effect of different molecular weight hyaluronan on macrophage physiology. Neuroendocrinology Letters 30 (Suppl. 1): 106–111. [PubMed] [Google Scholar]
- 22. De la, Motte C, Nigro J, Vasanji A, et al. (2009) Platelet-derived hyaluronidase 2 cleaves hyaluronan into fragments that triggermonocyte-mediated production of proinflammatory cytokines. American Journal of Pathology 174: 2254–2264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Girard N, Maingonnat C, Bertrand P, et al. (2002) Human monocytes synthesize hyaluronidase. British Journal of Haematology 119: 199–203. [DOI] [PubMed] [Google Scholar]
- 24. Manue V, Johannssen H, Salhy J, et al. (1999) Prospective randomized investigation for evaluation of postoperative changes in the microbial climate of paranasal mucosa by the use of different dissoluting techniques during postoperative. Rhinology 37: 113–116. [PubMed] [Google Scholar]
- 25. Macchi A, Castelnuovo P, Terranova P, et al. (2013) Effects of sodium hyaluronate 9 mg in children with recurrent upper respiratory tract infections: Results from a randomized controlled study. International Journal of Immunology and Pharmacology 26: 127–135. [DOI] [PubMed] [Google Scholar]
- 26. Montero Mora P, Blanco E, Matta Campos JJ, et al. (2003) Nasal remodeling in patient with perennia allergic rhinitis. Revista Alergia Mexico 50: 79–82. [PubMed] [Google Scholar]
- 27. Chen S. (2010) Persistent allergic rhinitis with special reference to the distribution of collagen, proteoglycans, and lymphaticvessels. Clinical and Experimental Allergy 40: 1742–1754. [DOI] [PubMed] [Google Scholar]
- 28. Berger G, Marom Z, Ophir D. (1997) Goblet cell density of the inferior turbinates in patients with perennial allergic and nonallergic rhinitis. American Journal of Rhinology 11: 233–236. [DOI] [PubMed] [Google Scholar]
- 29. Calderón MA, Lozewicz S, Prior A. (1992) Changes in the nasal mucosal microvascular with allergic rhinitis. Zhonghua Er Bi Yan Hou Ke Za Zhi 27: 39–40. [PubMed] [Google Scholar]


