Abstract
Photosystem II (PSII) catalyses the photoinduced oxygen evolution and, by producing reducing equivalents drives, in concert with PSI, the conversion of carbon dioxide to sugars. Our knowledge about the architecture of the reaction centre (RC) complex and the mechanisms of charge separation and stabilisation is well advanced. However, our understanding of the processes associated with the functioning of RC is incomplete: the photochemical activity of PSII is routinely monitored by chlorophyll-a fluorescence induction but the presently available data are not free of controversy. In this work, we examined the nature of gradual fluorescence rise of PSII elicited by trains of single-turnover saturating flashes (STSFs) in the presence of a PSII inhibitor, permitting only one stable charge separation. We show that a substantial part of the fluorescence rise originates from light-induced processes that occur after the stabilisation of charge separation, induced by the first STSF; the temperature-dependent relaxation characteristics suggest the involvement of conformational changes in the additional rise. In experiments using double flashes with variable waiting times (∆τ) between them, we found that no rise could be induced with zero or short ∆τ, the value of which depended on the temperature - revealing a previously unknown rate-limiting step in PSII.
Introduction
Photosystem II (PSII), or water-plastoquinone oxidoreductase, is a large multi-subunit homodimeric protein complex embedded in the thylakoid membranes of cyanobacteria, algae and vascular plants. The structure of the reaction centre complex (RC) is known at a resolution of 1.9 Å1 and our knowledge about the primary and secondary photochemical reactions is also well advanced2–4. The electron transfer from the primary donor P680 to pheophytin (Pheo) occurs in several picoseconds; subsequent electron transfer steps on the acceptor and donor sides, respectively - from Pheo− to QA, the first quinone electron acceptor, and from a tyrosine residue (YZ) to P680+ - stabilise the charge-separated state. These reactions are followed by electron and proton transfer reactions between the primary and secondary quinone acceptors, QA and QB - on the acceptor side, and between YZ and the S-states of the Mn4CaO5 cluster, i.e. the oxygen-evolving complex (OEC) - on the donor side.
Recent time-resolved serial femtosecond crystallography experiments on PSII crystals uncovered structural changes accompanying the reactions around the QB/non-heme iron and the Mn4CaO5 cluster5. This raises the question if the variable chlorophyll fluorescence, which is proportional to the quantum efficiency of PSII6, contains any component originating from similar conformational changes, as proposed earlier by some authors7,8.
The origin of chlorophyll-a fluorescence transients has been debated in the past decades and remains controversial. According to the mainstream concept9,10, in dark-adapted leaves or thylakoid membranes the multiphasic rise from the minimum to the maximum fluorescence level, Fo-to-Fm, elicited by a long saturating flash or by a series of intense short flashes, reflects solely the reduction of QA. In contrast, some authors have suggested that additional reactions and/or conformational changes must be taken into account7,8,11–13. The modified version of the mainstream model10 offers explanation on the complexity of the fluorescence induction, the so-called OJIP curve, which contains a wealth of information on the functioning of the photosynthetic apparatus under different experimental conditions6,10,14–17. The letters J and I signify intermediate states between the minimum and maximum fluorescence levels, O and P correspond to Fo and Fm levels, respectively, with Fo ascribed to all QA oxidized and Fm to all QA reduced. According to this model “in order to reach [Fm], it is necessary, and sufficient, to have QA completely reduced in all the active PSII centers”. In sharp contrast to this concept, Vredenberg12 has suggested that “full reduction of QA is neither sufficient nor required for reaching the Fm”; instead, the fluorescence rise is proposed to be “promoted by photo-electrochemical and electrical events in the vicinity of the membrane-bound RC”. Analysis of OJIP transients in a wide temperature-range revealed (an) additional process(es) of different physical origin in the fluorescence rise once QA is reduced; Schansker and co-workers suggested the involvement of light-induced conformational changes in the variable fluorescence, Fv = Fm − Fo8.
A basic problem with the mainstream model is that in the presence of DCMU (3-(3′,4′ dichlorophenyl)-1,1′ dimethylurea), which inhibits the electron transfer between QA and QB, and thus permits only one stable charge separation, Fm is not reached upon excitation by a single-turnover saturating flash (STSF)11. The first STSF induces an intermediary level, hereafter referred to as F1, typically 75–85% of Fm and Fm can be reached only gradually by further excitations11, e.g. by several additional STSFs (Supplementary Figure S1). It was shown that, in contrast to the Fo-to-F1 rise, leading to the reduction of QA, the consecutive flash-induced rises are associated with non-electrogenic reactions - showing their different physical origin. These data have been explained18 by assuming the presence of another quencher, different from QA, called Q2. However, the identity of Q2 has remained elusive.
Another set of data in the literature, which motivated our study, was that the kinetics of the fast fluorescence rise had been shown to depend on the length rather than on the intensity of the excitation flashes19,20. With sub nanosecond flashes the rise was purely exponential while with increasingly longer flashes, in the microseconds to milliseconds range, the kinetics became more and more sigmoidal. The authors interpreted their observations within the frameworks of the theory of “interunit transfer of excitations”, i.e. the theory of connectivity between PSII units21. However, they noted that “the basic phenomena underlying [the observed] differences should be investigated further”20. These data, by taking into account that the shape variations of the rise kinetics were coupled with changes in the Fm/Fo ratio20, could be brought into harmony with the theory of connectivity22 but other explanations cannot be ruled out. In fact, it has been proposed that sigmoidicity might arise from overlapping exponential kinetic components23. Indeed, Schansker and co-workers8, via recording OJIP curves at different, physiological and cryogenic temperatures, established that the overlapping components possessed different temperature dependences and observed the loss of sigmoidicity below −10 °C. (However, a role of connectivity in the sigmoidicity at physiological temperatures could not be ruled out).
With regard to the kinetics of the fast rise, the question can be raised why and how the length of the exciting flash determines both the magnitude (the Fm/Fo ratio) and the shape of the fluorescence rise (exponential with short flashes and sigmoidal with long ones)19,20. A reasonable explanation might be that the long flashes trigger a second process, which then raises the fluorescence yield (increases the Fm/Fo ratio) and, because of the kinetic overlap with the first process (the reduction of QA), the rise becomes sigmoidal. It is important to see, which also follows from the experiments of Joliot and Joliot11, that the second reaction occurs after the first one is completed (“the reduction of C550 attains its maximum level after the first saturating actinic flash…, unlike the fluorescence yield, [for which, i.e. for Fm] several [additional] flashes are required”). Based on these data11,19,20, it can be argued that there must be a waiting time between the two types of reactions; in other terms, a rate limitation in PSII – at least as reflected by the reaction(s) leading to the fluorescence rise. Heuristically, (a) rate-limiting step(s) must be involved in the fluorescence rise: we know that the fluorescence yield after the first STSF excitation does not depend on its intensity19,20 – the saturation of flashes is routinely checked, e.g. by firing two STSFs simultaneously, which should not affect the fluorescence levels. Nevertheless, to the best of our knowledge, such rate-limiting steps have not been identified and characterized.
The major aim of this work was to address the above two questions, i.e. (i) on the origin of the F1-to-Fm fluorescence increment in DCMU-inhibited PSII RC, and (ii) on the basic features of the (previously unidentified) rate-limiting step(s), which is now suggested to determine to a significant extent the kinetics of fluorescence rise. To this end, we investigated the gradual increase of fluorescence yield using STSF excitations on DCMU-treated whole cyanobacterial cells, plant thylakoid membrane preparations and cyanobacterial PSII core particles in a broad temperature range, between about 170 and 300 K. Our data show that in order to reach Fm, it is necessary to have QA reduced but additional processes succeeding this step, most probably conformational changes, must be involved. Further, by using double-STSF excitations, we show that the fluorescence rise kinetics is largely determined by temperature-dependent rate-limiting reactions in PSII. We propose the involvement of dielectric relaxation processes due to the presence of large static and transient electric fields, generated by the STSF-induced stable and transient charge separations with open and closed reaction centres, respectively.
Results
Characterization of the STSF-induced fluorescence rises
In order to discriminate between the Fo-to-F1 and the F1-to-Fm rises and to elucidate the origin of the F1-to-Fm increment, we recorded the STSF-induced variations of the fluorescence yield on dark-adapted PSII core particles, isolated from Thermosynechococcus vulcanus cells, and isolated spinach thylakoid membranes in the presence of DCMU.
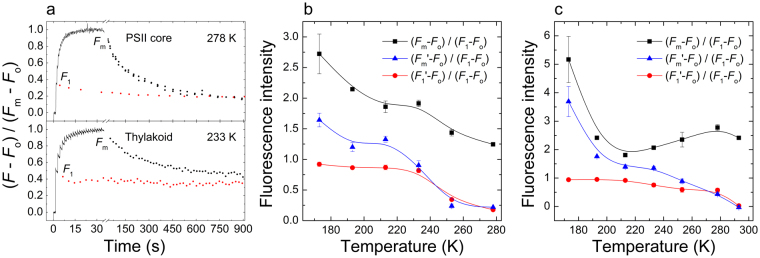
As shown in Fig. 1a (upper traces), in PSII core at 5 °C (278 K), the first STSF induced a fluorescence increase from Fo to F1, to a level which was only about 35% of Fm. An additional train of flashes consisting of 10–12 STSFs was required to reach Fm. At this temperature, which is relatively low compared to the 55 °C growth temperature of cells, charge recombination is limited. Accordingly, during 15 min in darkness F1 decreased only by about 20%. (Indeed, in thermophilic cyanobacteria, the thermoluminescence band arising from S2QA− recombination has been found at 35 °C24, indicating a reasonably high stability of the charge separated state at 5 °C). Similar features – large F1-to-Fm increment and an almost stable F1 level – can be observed in spinach thylakoid membranes at cryogenic temperatures (Fig. 1a lower trace). In contrast to the stability of F1, the Fm levels decayed considerably more rapidly, confirming that the physical processes determining the two levels differ from each other (see Introduction). Whereas the Fo-to-F1 transient can be unequivocally assigned to the reduction of QA, the F1-to-Fm rise must have a different origin, which remains to be identified. At higher temperatures, 20 °C (293 K) for PSII core and 5 °C (278 K) for thylakoid membranes, the build-up characteristics of the fluorescence yield upon the excitation of the samples with a train of STSFs (Supplementary Figure S2) were qualitatively very similar to those at lower temperatures (Fig. 1a). The main differences were in the decays; in particular, in the relaxation of the F1 levels, which accelerated with the increase of temperature, evidently due to recombination processes leading to the reoxidation of QA−.
Figure 1.
Temperature dependence of chlorophyll-a fluorescence induction. Kinetic traces at different temperatures (a) and parameters of STSF-induced fluorescence transients as a function of temperature in isolated spinach thylakoid membranes (b) and in isolated PSII core complexes of T. vulcanus (c). In panel a, red and black data points belong to transients induced by a single STSF and a train of STSFs, respectively. Fm′ and F1′ denote the corresponding fluorescence levels measured after 15 min in the dark following the excitation(s), and thus the black, blue and red curves represent the temperature dependences of Fm, Fm′ and F1′, respectively, normalized to F1. In panel a, the decay kinetics of F1 and Fm levels were measured on separate samples. In all cases when measuring the decay kinetics, in order to avoid its actinic effect, the measuring beam was turned on and off intermittently.
Activation energy associated with the relaxation of the fluorescence increment
In order to separate more clearly the two types of components of the fluorescence rise induced by a train of SFSFs, i.e. the Fo-to-F1 rise and the F1-to-Fm increment, we carried out experiments in a broad temperature range. In particular, we wanted to study the nature of the F1-to-Fm increment without the influence of F1 relaxation. This could be performed in the temperature ranges where F1 levels exhibited high stability on the timescale of minutes. As shown in Fig. 1b and c, for DCMU-treated thylakoid membranes and PSII core, respectively, in broad, low temperature ranges, Fm levels decayed at all temperatures (compare black squares with blue triangles), while the F1 levels remained nearly unchanged (red circles). Decay of Fm was observed even at 77 K, as checked on a leaf immersed in liquid nitrogen (not shown). The increments, in general, were larger at low temperatures, albeit the temperature dependence did not follow a simple rule in either sample – a phenomenon worth of further investigations. In PSII core, contributions to the increment from PSI are evidently ruled out. For thylakoid membranes, this is excluded by the data obtained with different optical filters, with one of them preferentially allowing the detection of PSII emission, while the other suited both for PSII and PSI emission (Supplementary Figure S3). Hence, we confirmed that F1-to-Fm originated solely from PSII.
We analysed the temperature dependence of Fm decay of thylakoid membranes and PSII core. The analysis was performed on the decay kinetics between about 170 and 210 K, for thylakoids, and about 170 and 250 K, for PSII core. In these ranges, the corresponding F1 values remained constant and charge recombination involving QA− could be neglected. The calculated activation energies, 13.8 ± 0.8 kJ/mol and 11.3 ± 3.6 kJ/mol, for thylakoid membranes and PSII core, respectively (Fig. 2), agree reasonably well with each other and the values derived from the temperature dependences of the OJIP fluorescence rise components in thylakoid membranes8.
Figure 2.
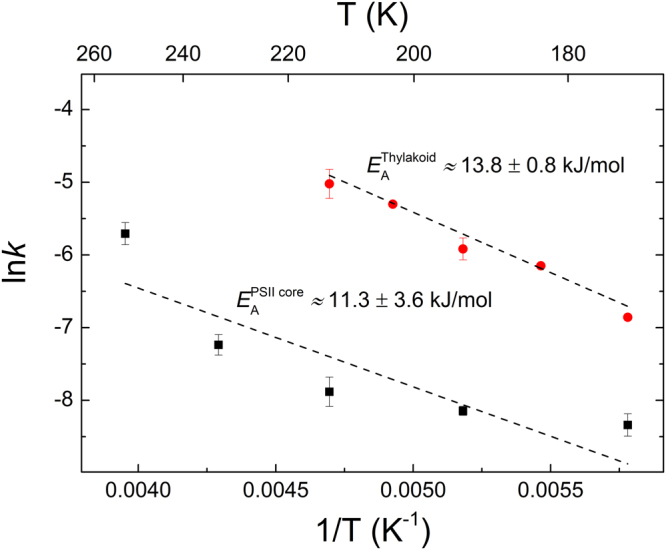
Activation energies associated with the fluorescence relaxation in thylakoid membranes and PSII core particles. The activation energies (EA) were calculated from the decay of Fm in a temperature range where the decay of F1 was negligible.
Rate-limiting steps in the reactions associated with the fluorescence increments
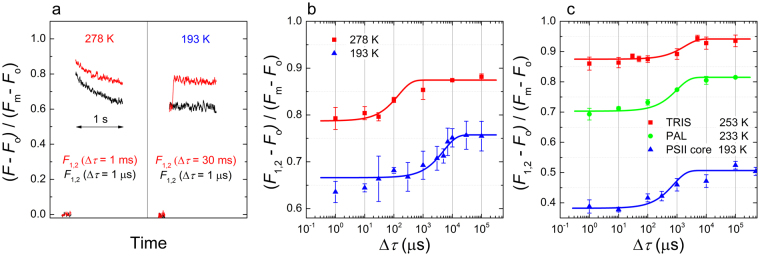
In order to understand the mechanism underlying the variations in the fast fluorescence rise kinetics20 (exponential with short pulses and sigmoidal with longer flashes), we performed double-STSF induced fluorescence kinetic measurements. We observed that when the two flashes were fired simultaneously or with a short dark interval between them, no fluorescence increment could be observed (Fig. 3). There was a minimum waiting time (∆τ), which was required after the first STSF, for the second STSF to induce any observable F1-to-F2 increment. This suggests that a rate-limiting step is associated with the fluorescence increment. The waiting half-time of the increment was temperature-dependent: in thylakoids it varied between several hundred microseconds at 5 °C (278 K), about 1.3 ms at −10 °C (283 K) (not shown) and almost 10 ms at −80 °C (193 K) (Fig. 3a,b). The existence of these rate limitations could be clearly discerned in all other samples tested: TRIS-washed thylakoid membranes, PSII core particles, intact cyanobacterial cells (the PAL mutant of Synechocystis PCC 6803, lacking the phycobilisome antenna25) (Fig. 2c). TRIS washing was used to remove the OEC from the PSII supercomplex. Hence, we can rule out the involvement of the Mn4CaO5 cluster. (This was unlikely, also because in the presence of DCMU the second and consecutive flashes can not induce turnover in the S-states of OEC). Isolation artefacts were ruled out by using intact cyanobacterial cells (the PAL mutant Synechocystis sp. PCC6803 – this mutant was used to avoid possible interference and high background emission from this antenna that is anchored to the thylakoid membrane).
Figure 3.
Chlorophyll-a fluorescence induced by double flashes. Kinetic traces of the F1-to-F2 increment with two different dark times (Δτ) between the first and second flashes, at two different temperatures in isolated spinach thylakoid membranes (a) and dependences of the F1-to-F2 increments on the waiting time (Δτ) between the flashes in thylakoid membranes (b), and in TRIS-washed thylakoid membranes, intact cells of the PAL mutant of Synechocystis PCC6803 and in the PSII core particles (c) at different temperatures as indicated. F1,2 denotes the fluorescence intensity level following the first double STSF.
The existence of rate-limiting step shows again that the processes induced by the first and the consecutive flashes are of different physical origins. In turn, as pointed out in the Introduction, two independent processes may, at least in part, be responsible for the sigmoidal rise of the fluorescence. In good agreement with this notion, the fluorescence induction kinetics of solubilised PSII core exhibited a sigmoidal rise (Supplementary Figure S4). Evidently, in this sample connectivity between the dimeric PSII supercomplexes can be ruled out (but still allowing intradimer energy transfer between the monomers26). Nevertheless, the rise, which also depended on the temperature, could be fitted reasonably well with two exponentials. It is to be noted, however, that for a more rigorous mathematical analysis of this fast fluorescence rise kinetics, and the OJIP curves in general, the rate-limiting step(s) should be taken into account. When fitting with two exponentials the waiting time is ignored, or considered to be negligible relative to the rise times. Also, it implicitly assumes that Fm can be reached in one additional step, which is not the case. Even with the largest ∆τ, Fm could not be reached by the second STSF, and the number of STSFs inducing Fm remained invariant on the mode of excitation – as long as two simultaneously fired STSFs were counted as one and ∆τ was sufficiently long. Further, the rate-limiting step was present not only in F1-to-F2 but also between later steps (Supplementary Figure S5) – suggesting similar processes during the train of additional STSFs.
Discussion
The major aim of this work was to resolve some of the controversies over the origin and kinetics of the chlorophyll-a fluorescence induction of PSII, perhaps the most widely used probe of PSII activity. In particular, as argued in the Introduction, in contrast to the expectations based on the ‘mainstream’ model: (i) the fluorescence maximum in DCMU-treated samples cannot be reached by one STSF excitation, and (ii) the fast fluorescence kinetics depends on the length, rather than on the intensity of the flash excitation of PSII. In order to understand the underlying physical mechanisms responsible for these phenomena, which are in conflict with the most widely accepted model, we performed experiments on a variety of different samples and at different, physiological and cryogenic temperatures, in the presence of DCMU, which inhibits the electron transfer between the primary and secondary quinone acceptors, QA and QB. As will be discussed in more detail below: (i) by using trains of STSFs, we provide irrevocable evidence that the first STSF and the consecutive flashes induce different reactions; (ii) our data, derived from experiments using double-STSF excitations, reveal that the stepwise fluorescence rises after the first STSF do not occur in the absence of sufficiently long waiting times (∆τ) between STSFs – showing the existence of rate-limiting steps, with strong temperature dependence but of presently unidentified nature. As a corollary, we show, also experimentally, that the sigmoidal rise does not require the connectivity between PSII supercomplexes.
The origin of F1-to-Fm fluorescence increment differs from that of the Fo-to-F1 rise
Our experiments have shown, in agreement with some earlier data (see Introduction) that the underlying physical mechanisms induced by the first STSF and the consecutive flashes, responsible for the Fo-to-F1 rise and the F1-to-Fm increment, respectively, are of different nature. This is most clearly seen at cryogenic temperatures where the charge-separated state is stabilised (Fig. 1). In this region, both in isolated plant thylakoid membranes and cyanobacterial PSII core particles, the F1 levels remain constant but the F1-to-Fm increments display strong, temperature-dependent decay characteristics. Hence, we can conclude that, in accordance with the mainstream model10, the reduction of QA is necessary to account for the fluorescence rise from its minimum to maximum value (Fo-to-Fm). However, in contrast to this model, our data show that it is not a sufficient condition. This conclusion is in perfect agreement with that drawn by Joliot and Joliot11, who have shown that, in isolated spinach thylakoid membranes at room temperature, the first STSF induces an intense electric field but the consecutive steps are not electrogenic. Hence, the experiments of Joliot and Joliot, at room temperature, and our data, also at cryogenic temperatures and on PSII core particles, show that the first flash, reducing QA, must be followed by several additional excitations to reach Fm. The length of this process (i.e., the number of excitations involved) depends strongly on the temperature, so does the magnitude of the F1-to-Fm increment relative to the F1 level – which can be quite high (~500% for PSII core at 170 K).
As concerns the origin of F1-to-Fm increment, there are essentially two types of possibilities: (i) the additional rise originates from the activity (release) of a quencher different from QA – called Q218 or (ii) the fluorescence yield is modulated via additional light-induced reactions.
Considerations on the involvement of the hypothetical quencher Q2
When taking into account the molecular architecture of PSII RC1, we can find candidates for the additional quencher (Q2). The non-heme iron, which is situated between QA and QB and plays a key role in the protonation of the reduced QB27, has been proposed28 to be identical with Q2. However, this is unlikely to be the case, since the oxidized form of this non-heme iron can easily be reduced by a STSF29, and in the presence of DCMU it remains stably reduced30 - facts which would be difficult to reconcile with the decay of the fluorescence increment (Fig. 1). The reduction of QC, the third (plasto)quinone found in the crystal structure of PSII from Thermosynechococcus elongatus31, might be involved in light-induced redox reactions32, and thus may act as a quencher. However, QC is unlikely to be present in our PSII core particles – given the fact that it is not found in the crystal structure of the same preparation1. Also, given its position in the quinone exchange cavity31, it is unclear how QC would be reduced in the presence of DCMU and reoxidized under the conditions when QA remains stably reduced at low temperature (cf. Fig. 1). In a theoretical model, in which “cyt b559 [cytochrome b559] accepts electrons from the reduced primary electron acceptor in PSII, pheophytin, and donates electrons to the oxidized primary electron donor in PSII (P680+)”, it has been proposed that “Fm increases with an increasing amount of initially reduced cyt b559”33. However, direct reduction of the cyt b559 by reduced pheophytin seems highly unlikely because of the relatively large distance between the two molecules31. Indeed, in the presence of protonophoric compounds, such as CCCP (carbonylcyanide-m-chlorophenylhydrazon), Fv/Fm has been shown to increase34, and fast photoreduction of the autooxidized cyt b559 has also been observed; however, the STSF-induced photoreduction has been shown to proceed via QB−, the semireduced secondary quinone acceptor, a reaction inhibited by DCMU35. Taken together, these putative quenchers do not explain our observations. The same holds true for the double-hit trapping model, which hypothesizes that the [PheoQA] acceptor pair acts as a two electron trap36. With this pair of acceptor molecules it would be difficult to explain the multi-step F1-to-Fm increments observed in our experiments. Also, the slowly-turning over, non-QB RCs, a fraction of the heterogeneous population of PSII in the thylakoid membranes, generate a stable charge separation upon the first STSF and remain silent for seconds afterwards37. The involvement of a presently unidentified, low-quantum-yield photochemical reaction, such as between the tyrosine residues and P680+ 38, the quenching by P680+ 39 or the participation of components of inactive branch40 cannot be ruled out. However, the product(s) (the quencher) must persist for long time periods (to maintain F1 constant or the higher levels steps only slowly decaying), minutes at low temperatures; also, its concentration should be largely independent of the intensity of the STSF (a single or double STSF should produce the same amount of quencher); and the release of this putative quencher should be possible to induce by light (to produce the increment) but only after a suitable waiting time. These conditions do not seem to apply to these potential quenchers, components participating in the ultrafast photochemical reactions of the RC.
The possible role of dielectric relaxation following the charge separations
The fluorescence yield of chlorophyll-a can also be modulated by minor variations in the physicochemical environment of the molecules and protein complexes and/or subtle conformational changes of the complexes. Spectral properties and fluorescence lifetimes of different fluorophores can be sensitive to variations in the dielectric (micro)environment. The peak position of the absorption spectra of the lipophylic dye merocyanine 540 has been shown to depend strongly on the dielectric constant of the medium41. Similar, albeit less marked dependence has been reported for chlorophyll-a and bacteriochlorophyll-a; the lifetime (fluorescence yield) of these molecules also depend on the refractive index / dielectric constant of the solvent42. Substantial variations in the fluorescence lifetime of chlorophyll-a have also been observed in plant light-harvesting protein complex II upon the addition and removal of detergents or varying the lipidic environment of the complexes43,44. It is thus proposed that variations in the dielectric microenvironment of RC chlorophyll-a can be responsible for the F1-to-Fm increment.
The OJIP rise in thylakoid membranes has been shown to be sensitive to the transmembrane electrochemical potential gradient12,45, generated by charge separation and consecutive electron and proton transfer steps. While we can not rule out the influence of this factor on the F1-to-Fm rise, it is clear that in solubilised PSII core the increments occur in the absence of membrane potential. At the same time, however, the local electric field, due e.g. to (S2/YZ)+QA−, the strength of which can be orders of magnitude higher than that of the uniform transmembrane field, induces a redistribution of ions in the electrolyte surrounding the reaction centre46. It is thus reasonable to assume that these events affect the fluorescence yield of chlorophyll-a. Its possible interplay with the variations in the dielectric microenvironment - via dielectric relaxation – is discussed below.
The exact physical mechanism of the processes underlying the F1-to- Fm increment remains to be elucidated. The increments can most easily be interpreted in terms of light-induced conformational changes8 – possessing activation energy values (Fig. 2) similar to those determined earlier8 under substantially different experimental conditions. With regard to the nature of conformational changes, we hypothesize that they are coupled to the dielectric relaxation in the RC matrix, which is exposed to the strong local electric field of (S2/YZ)+QA−. Similar mechanism has been proposed to be responsible for the light-induced structural changes in the purple-bacterial RC47. We would like to stress that this mechanism does not assume additional quencher molecule(s) but rather relies on modulation of the fluorescence quantum yield via the physicochemical environment of chromophores in the RC complex. The dielectric relaxation is a complex phenomenon even for simple proteins; it may contain different components at different temperatures and hydration conditions48. In intrinsic membrane proteins or lipoproteins, inherent heterogeneity of the dielectric matrix may further complicate the situation49. Nevertheless, a justifiable assumption, the fine adjustments (components of the dielectric relaxation) in the dielectric matrix are hindered more and more at lower temperatures, explaining the increase of the F1-to-Fm increment and the number of excitations required at low temperatures (Fig. 1). Fluorescence and absorbance transient measurements with submicrosecond time resolution should clarify if at ambient temperatures the Fo-to-F1 fluorescence rise is related solely to the reduction of QA or also contains an additional component. Excitations in the presence of reduced QA evidently induce transient charge separations, P+Pheo− or YZ+Pheo− in the ns-µs time domain. The large transient local electric fields46 might induce conformational changes in the proteins and/or rearrangements in the dielectric matrix. As an alternative explanation, the involvement of dissipation-induced heat packages50 cannot be ruled out; they might transiently’melt’ the RC matrix and facilitate its dielectric adjustments. In either case, the F1-to-Fm increment might be part of the dark-to-light transition of the PSII RC following the reduction of QA. Dark- and light-adapted conformational states have also been proposed for purple bacterial RC51.
Origin of the rate-limiting steps
The physical mechanism of the waiting-time related transient inhibition of the flash-induced reaction responsible for the fluorescence increment (Fig. 3) also remains to be identified but a similar, dielectric-relaxation / conformational-change based mechanism could be at play. It is also to be clarified – by ultrafast absorbance and fluorescence transient measurements - if the rate limitation is at the level of primary photochemistry (e.g. the turnover of P+Pheo−) or at the level of the state of PSII, a state determining its fluorescence yield. In this latter case, the rate limitation is confined to the propagation of the effect of STSF-induced photoreaction(s) to the site of the fluorophore. It is interesting to note that the waiting times determined in our samples are commensurate with the half-times of the electron transfer between QA and QB, which also displays strong temperature dependence and is related to protein dynamics52–54. It is tempting to speculate that the two mechanisms are harmonized. This may constitute a hitherto unknown photoprotection mechanism, which might be capable of down-regulating the turnover of RC upon excess excitation of PSII.
Materials and Methods
Thylakoid membranes were isolated from fresh market spinach leaves essentially as described earlier37, with minor modifications. Briefly, deveined leaves were homogenized in 20 mM tricine (pH 8.0), 400 mM NaCl, 2 mM MgCl2, filtered through a nylon mesh and centrifuged for 7 min at 6000 g. The pellet was resuspended in 20 mM tricine (pH 8.0), 150 mM NaCl and 5 mM MgCl2, homogenized and centrifuged for 7 min at 6,000 g. The final pellet was resuspended in 20 mM MES (pH 6.5), 400 mM sucrose, 15 mM NaCl, 5 mM MgCl2 and stored, until use, in liquid nitrogen at a concentration of 2–3 mg/ml Chl content. Identical results were obtained with fresh preparations. PSII core complex of T. vulcanus was isolated as described earlier55,56. PAL mutant Synechocystis sp. PCC6803 cells25 were grown photoautotrophically in BG11 medium supplemented with 5 mM HEPES–NaOH (pH 7.5), and 8 μg/ml chloramphenicol. Cells were grown at 30 °C, under continuous illumination at the intensity of 30 μmol photons m−2 s−1. Cultures were aerated on a gyratory shaker operating at 100 rpm. TRIS washed thylakoid membranes were prepared by the following protocol. The thylakoid membranes were incubated in 1 M TRIS buffer (pH 8.0) for 30 min in complete darkness and shaken from time to time. The suspension was then centrifuged for 7 min at 6,000 g. The pellet was resuspended in 20 mM MES (pH 6.5), 400 mM sucrose, 15 mM NaCl, 5 mM MgCl2 and the sample was used freshly.
Relative fluorescence yields were measured using a PAM 101 fluorometer (Walz, Effeltrich, Germany). The frequency of the modulated measuring light (low intensity, non-actinic) was 1.6 kHz. Variable fluorescence was induced by 0.5 J single-turnover saturating Xe flashes (General Radio 1539-A, USA) of 3 µs duration at half-peak intensity. The sample was placed at the sample holder of a thermoluminescence apparatus in order to control the temperature. The timing of the flashes was controlled by using a home-designed programmable digital pulse generator. The decays of each measurements were recorded by using NI DAQ 6001 via custom-designed LabVIEW software. Least-squares optimization was used to estimate the decay parameters. The optimization algorithms were implemented in Matlab (The MathWorks, Natick, MA). For chlorophyll-a fluorescence transient measurements the chlorophyll concentration of the thylakoid membranes were diluted to ~100 μg/ml in 20 mM MES (pH 6.5), 400 mM sucrose, 15 mM NaCl, 5 mM MgCl2; the PSII core complexes to ~40 μg/ml in 30 mM MES (pH 6.0), 20 mM NaCl, 3 mM CaCl2 and 5% glycerol; the PAL mutants to ~50 μg/ml in BG11 medium. DCMU was dissolved in dimethyl sulfoxide (DMSO) and added to all samples immediately before the fluorescence measurements at a final concentration of 40 μM (the final concentration of DMSO did not exceed 1%). Before the measurements, the samples were dark adapted for 5 min at room temperature.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Electronic supplementary material
Acknowledgements
This work was supported by a Strategic Priority Research Program (XDB17030100), a Key Research Project for Frontier Science (QYZDY-SSW-SMC003) from CAS, China, and a National Natural Science Foundation of China grant 31470339 (to JRS), grants from the National Research Development and Innovation Office of Hungary (OTKA K 112688, KH 124985 and TÉT_15-1-2016-0144 to GG, GINOP-2.3.2-15-2016-00058 to UB and OTKA PD 121225 to MM) and from the Moravian-Silesian Region (Project title: Strengthening international cooperation in science, research and education; ID: 01211/2016/RRC to VŠ). We are indebted to Prof. Govindjee for critical reading of the manuscript and helpful comments, Prof. William A. Cramer and Dr. László Nagy for stimulating discussions and Dr. Ghada Ajlani for the PAL mutant.
Author Contributions
G.G. conceived the study and, together with G.S. and M.M., designed the experiments, which were performed by M.M., G.S. and L.K. The PSII core and the PAL mutant cells were provided by J.R.S., Q.Z. and G.H. and U.B., respectively. The paper was written by G.G., M.M., G.S., PH.L., V.Š. and J.R.S.
Competing Interests
The authors declare no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-21195-2.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Umena Y, Kawakami K, Shen JR, Kamiya N. Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 angstrom. Nature. 2011;473:55–U65. doi: 10.1038/nature09913. [DOI] [PubMed] [Google Scholar]
- 2.Cardona T, Sedoud A, Cox N, Rutherford AW. Charge separation in photosystem II: A comparative and evolutionary overview. Biochim Biophys Acta-Bioenergetics. 2012;1817:26–43. doi: 10.1016/j.bbabio.2011.07.012. [DOI] [PubMed] [Google Scholar]
- 3.Vinyard DJ, Ananyev GM, Dismukes GC. Photosystem II: The reaction center of oxygenic photosynthesis. Annu Rev Biochem. 2013;82:577–606. doi: 10.1146/annurev-biochem-070511-100425. [DOI] [PubMed] [Google Scholar]
- 4.Romero E, Novoderezhkin VI, van Grondelle R. Quantum design of photosynthesis for bio-inspired solar-energy conversion. Nature. 2017;543:355–365. doi: 10.1038/nature22012. [DOI] [PubMed] [Google Scholar]
- 5.Suga M, et al. Light-induced structural changes and the site of O = O bond formation in PSII caught by XFEL. Nature. 2017;543:131–135. doi: 10.1038/nature21400. [DOI] [PubMed] [Google Scholar]
- 6.Genty B, Briantais JM, Baker NR. The relationship between the quantum yield of photosynthetic electron-transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta. 1989;990:87–92. doi: 10.1016/S0304-4165(89)80016-9. [DOI] [Google Scholar]
- 7.Moise N, Moya I. Correlation between lifetime heterogeneity and kinetics heterogeneity during chlorophyll fluorescence induction in leaves: 1. Mono-frequency phase and modulation analysis reveals a conformational change of a PSII pigment complex during the IP thermal phase. Biochim Biophys Acta-Bioenergetics. 2004;1657:33–46. doi: 10.1016/j.bbabio.2004.04.001. [DOI] [PubMed] [Google Scholar]
- 8.Schansker G, Toth SZ, Kovacs L, Holzwarth AR, Garab G. Evidence for a fluorescence yield change driven by a light-induced conformational change within photosystem II during the fast chlorophyll a fluorescence rise. Biochim Biophys Acta-Bioenergetics. 2011;1807:1032–1043. doi: 10.1016/j.bbabio.2011.05.022. [DOI] [PubMed] [Google Scholar]
- 9.Duysens, L. M. N. & Sweers, H. E. In Studies on Microalgae and Photosynthetic Bacteria 353–372 (Japanese Society of Plant Physiologists, University of Tokyo Press, 1963).
- 10.Stirbet AG. Chlorophyll a fluorescence induction: a personal perspective of the thermal phase, the J-I-P rise. Photosynth Res. 2012;113:15–61. doi: 10.1007/s11120-012-9754-5. [DOI] [PubMed] [Google Scholar]
- 11.Joliot P, Joliot A. Comparative-study of the fluorescence yield and of the C550 absorption change at room-temperature. Biochim Biophys Acta. 1979;546:93–105. doi: 10.1016/0005-2728(79)90173-7. [DOI] [PubMed] [Google Scholar]
- 12.Vredenberg W. A simple routine for quantitative analysis of light and dark kinetics of photochemical and non-photochemical quenching of chlorophyll fluorescence in intact leaves. Photosynth Res. 2015;124:87–106. doi: 10.1007/s11120-015-0097-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Delosme R. Study of the induction of fluorescence in green algae and chloroplasts at the onset of an intense illumination. Biochim Biophys Acta. 1967;143:108–128. doi: 10.1016/0005-2728(67)90115-6. [DOI] [PubMed] [Google Scholar]
- 14.Papageorgiou, G. C. & Govindjee. Chlorophyll a Fluorescence: A Signature of Photosynthesis. (Springer, 2004).
- 15.Strasser, R. J., Tsimilli-Michael, M. & Srivastava, A. Analysis of the chlorophyll a fluorescence transient in Chlorophyll a Fluorescence: A Signature of Photosynthesis (eds G.C. Papageorgiou & Govindjee) 463–495 (Springer, 2004).
- 16.Kalaji HM, et al. Frequently asked questions about in vivo chlorophyll fluorescence: practical issues. Photosynth Res. 2014;122:121–158. doi: 10.1007/s11120-014-0024-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stirbet A, Riznichenko GY, Rubin AB. & Govindjee. Modeling chlorophyll a fluorescence transient: relation to photosynthesis. Biochemistry. Biokhimiia. 2014;79:291–323. doi: 10.1134/S0006297914040014. [DOI] [PubMed] [Google Scholar]
- 18.Joliot P, Joliot A. A photosystem II electron-acceptor which is not a plastoquinone. Febs Lett. 1981;134:155–158. doi: 10.1016/0014-5793(81)80590-X. [DOI] [Google Scholar]
- 19.Valkunas L, Geacintov NE, France L, Breton J. The dependence of the shapes of fluorescence induction curves in chloroplasts on the duration of illumination pulses. Biophys J. 1991;59:397–408. doi: 10.1016/S0006-3495(91)82233-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.France LL, Geacintov NE, Breton J, Valkunas L. The dependence of the degrees of sigmoidicities of fluorescence induction curves in spinach-chloroplasts on the duration of actinic pulses in pump-probe experiments. Biochim Biophys Acta. 1992;1101:105–119. doi: 10.1016/0167-4838(92)90474-R. [DOI] [Google Scholar]
- 21.Joliot, A. & Joliot, P. Étude cinétique de la réaction photochimique libérant l’oxygéne au cours de la photosynthése. CR Acad Sci Paris258, 4622–4625 (in French) (1964). [PubMed]
- 22.Stirbet A. Excitonic connectivity between photosystem II units: what is it, and how to measure it? Photosynth Res. 2013;116:189–214. doi: 10.1007/s11120-013-9863-9. [DOI] [PubMed] [Google Scholar]
- 23.Vredenberg WJ. Analysis of initial chlorophyll fluorescence induction kinetics in chloroplasts in terms of rate constants of donor side quenching release and electron trapping in photosystem II. Photosynth Res. 2008;96:83–97. doi: 10.1007/s11120-007-9287-5. [DOI] [PubMed] [Google Scholar]
- 24.Govindjee KH, Inoue Y. Thermo-luminescence and oxygen evolution from a thermophilic blue-green-alga obtained after single-turnover light-flashes. Photochem Photobiol. 1985;42:579–585. doi: 10.1111/j.1751-1097.1985.tb01613.x. [DOI] [Google Scholar]
- 25.Ajlani G, Vernotte C. Construction and characterization of a phycobiliprotein-less mutant of Synechocystis sp. PCC 6803. Plant Mol Biol. 1998;37:577–580. doi: 10.1023/A:1005924730298. [DOI] [PubMed] [Google Scholar]
- 26.Caffarri S, Tibiletti T, Jennings RC, Santabarbara S. A comparison between plant photosystem I and photosystem II architecture and functioning. Curr Protein Pept Sc. 2014;15:296–331. doi: 10.2174/1389203715666140327102218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shevela D, Eaton-Rye JJ, Shen JR. & Govindjee. Photosystem II and the unique role of bicarbonate: a historical perspective. Biochim Biophys Acta. 2012;1817:1134–1151. doi: 10.1016/j.bbabio.2012.04.003. [DOI] [PubMed] [Google Scholar]
- 28.Muh F, Zouni A. The nonheme iron in photosystem II. Photosynth Res. 2013;116:295–314. doi: 10.1007/s11120-013-9926-y. [DOI] [PubMed] [Google Scholar]
- 29.Brettel K, Setif P, Mathis P. Flash-induced absorption changes in photosystem-I at low-temperature - evidence that the electron acceptor-A1 is vitamin-K1. Febs Lett. 1986;203:220–224. doi: 10.1016/0014-5793(86)80746-3. [DOI] [Google Scholar]
- 30.Ikegami I, Katoh S. Studies on chlorophyll fluorescence in chloroplasts II. Effect of ferricyanide on the induction of fluorescence in the presence of 3-(3,4-dichlorophenyl)-1,1-dimethylure. Plant Cell Physiol. 1973;14:829–836. [Google Scholar]
- 31.Guskov A, et al. Cyanobacterial photosystem II at 2.9-angstrom resolution and the role of quinones, lipids, channels and chloride. Nat Struct Mol Biol. 2009;16:334–342. doi: 10.1038/nsmb.1559. [DOI] [PubMed] [Google Scholar]
- 32.Muh F, Glockner C, Hellmich J, Zouni A. Light-induced quinone reduction in photosystem II. Biochim Biophys Acta-Bioenergetics. 2012;1817:44–65. doi: 10.1016/j.bbabio.2011.05.021. [DOI] [PubMed] [Google Scholar]
- 33.Lazar D, Ilik P, Kruk J, Strzalka K, Naus J. A theoretical study on effect of the initial redox state of cytochrome b(559) on maximal chlorophyll fluorescence level (F-M) implications for photoinhibition of photosystem II. J Theor Biol. 2005;233:287–300. doi: 10.1016/j.jtbi.2004.10.015. [DOI] [PubMed] [Google Scholar]
- 34.Samson G, Bruce D. Origins of the low yield of chlorophyll a fluorescence induced by single turnover flash in spinach thylakoids. Biochim Biophys Acta-Bioenergetics. 1996;1276:147–153. doi: 10.1016/0005-2728(96)00072-2. [DOI] [Google Scholar]
- 35.Barabas K, Garab G. 2 Populations of the high-potential form of cytochrome-B-559 in chloroplasts treated with 2-(3-Chloro-4-Trifluoromethyl)Anilino-3,5-Dinitrothiophene (Ant 2p) Febs Lett. 1989;248:62–66. doi: 10.1016/0014-5793(89)80432-6. [DOI] [Google Scholar]
- 36.Vredenberg W. Kinetic analyses and mathematical modeling of primary photochemical and photoelectrochemical processes in plant photosystems. Biosystems. 2011;103:138–151. doi: 10.1016/j.biosystems.2010.10.016. [DOI] [PubMed] [Google Scholar]
- 37.Chylla RA, Garab G, Whitmarsh J. Evidence for slow turnover in a fraction of photosystem II complexes in thylakoid membranes. Biochim Biophys Acta. 1987;894:562–571. doi: 10.1016/0005-2728(87)90136-8. [DOI] [Google Scholar]
- 38.Schlodder E, Cetin M, Lendzian F. Temperature dependence of the oxidation kinetics of Tyr(Z) and Tyr(D) in oxygen-evolving photosystem II complexes throughout the range from 320 K to 5 K. Biochim Biophys Acta-Bioenergetics. 2015;1847:1283–1296. doi: 10.1016/j.bbabio.2015.07.005. [DOI] [PubMed] [Google Scholar]
- 39.Shinkarev VPG. Insight into the relationship of chlorophyll a fluorescence yield to the concentration of its natural quenchers in oxygenic photosynthesis. Proceedings of the National Academy of Sciences of the United States of America. 1993;90:7466–7469. doi: 10.1073/pnas.90.16.7466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schreiber, U. Assessment of maximal fluorescence yield: donor-side dependent quenching and QB quenching. In Plant spectrophotometry: applications and basic research (eds O van Kooten & JFH Snel) 23–47 (Rozenberg Publishers, 2002).
- 41.Lelkes PI, Miller IR. Perturbations of membrane structure by optical probes: I. Location and structural sensitivity of merocyanine 540 bound to phospholipid membranes. The Journal of membrane biology. 1980;52:1–15. doi: 10.1007/BF01869001. [DOI] [PubMed] [Google Scholar]
- 42.Connolly JS, Samuel EB, Janzen AF. Effects of solvent on the fluorescence properties of bacteriochlorophyll a. Photochem Photobiol. 1982;36:565–574. doi: 10.1111/j.1751-1097.1982.tb04417.x. [DOI] [Google Scholar]
- 43.Ilioaia C, Johnson MP, Horton P, Ruban AV. Induction of efficient energy dissipation in the isolated light-harvesting complex of photosystem II in the absence of protein aggregation. J Biol Chem. 2008;283:29505–29512. doi: 10.1074/jbc.M802438200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Akhtar P, et al. Pigment Interactions in light-harvesting complex II in different molecular environments. J Biol Chem. 2015;290:4877–4886. doi: 10.1074/jbc.M114.607770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Antal TK, Osipov V, Matorin DN, Rubin AB. Membrane potential is involved in regulation of photosynthetic reactions in the marine diatom Thalassiosira weissflogii. J Photochem Photobiol B: Biology. 2011;102:169–173. doi: 10.1016/j.jphotobiol.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 46.Zimanyi L, Garab G. Configuration of the electric-field and distribution of ions in energy transducing biological-membranes - model-calculations in a vesicle containing discrete charges. J Theor Biol. 1989;138:59–76. doi: 10.1016/S0022-5193(89)80178-X. [DOI] [Google Scholar]
- 47.Malferrari M, Mezzetti A, Francia F, Venturoli G. Effects of dehydration on light-induced conformational changes in bacterial photosynthetic reaction centers probed by optical and differential FTIR spectroscopy. Biochim Biophys Acta-Bioenergetics. 2013;1827:328–339. doi: 10.1016/j.bbabio.2012.10.009. [DOI] [PubMed] [Google Scholar]
- 48.Panagopoulou A, Kyritsis A, Shinyashiki N, Pissis P. Protein and water dynamics in bovine serum albumin-water mixtures over wide ranges of composition. J Phys Chem B. 2012;116:4593–4602. doi: 10.1021/jp2105727. [DOI] [PubMed] [Google Scholar]
- 49.Bhaduri S, et al. Pathways of Transmembrane Electron Transfer in Cytochrome bc Complexes: Dielectric Heterogeneity and Interheme Coulombic Interactions. J Phys Chem B. 2017;121:975–983. doi: 10.1021/acs.jpcb.6b11709. [DOI] [PubMed] [Google Scholar]
- 50.Garab G. Hierarchical organization and structural flexibility of thylakoid membranes. Biochim Biophys Acta-Bioenergetics. 2014;1837:481–494. doi: 10.1016/j.bbabio.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 51.Goushcha AO, Kharkyanen VN, Scott GW, Holzwarth AR. Self-regulation phenomena in bacterial reaction centers. I. General theory. Biophys J. 2000;79:1237–1252. doi: 10.1016/S0006-3495(00)76378-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Garbers A, Reifarth F, Kurreck J, Renger G, Parak F. Correlation between protein flexibility and electron transfer from QA−* to QB in PSII membrane fragments from spinach. Biochemistry. 1998;37:11399–11404. doi: 10.1021/bi980296+. [DOI] [PubMed] [Google Scholar]
- 53.Shlyk-Kerner O, et al. Protein flexibility acclimatizes photosynthetic energy conversion to the ambient temperature. Nature. 2006;442:827–830. doi: 10.1038/nature04947. [DOI] [PubMed] [Google Scholar]
- 54.Pieper J, Renger G. Protein dynamics investigated by neutron scattering. Photosynth Res. 2009;102:281–293. doi: 10.1007/s11120-009-9480-9. [DOI] [PubMed] [Google Scholar]
- 55.Shen JR, Inoue Y. Binding and functional-properties of two new extrinsic components, cytochrome c-550 and a 12-kDa Protein, in cyanobacterial photosystem II. Biochemistry. 1993;32:1825–1832. doi: 10.1021/bi00058a017. [DOI] [PubMed] [Google Scholar]
- 56.Shen JR, Kamiya N. Crystallization and the crystal properties of the oxygen-evolving photosystem II from Synechococcus vulcanus. Biochemistry. 2000;39:14739–14744. doi: 10.1021/bi001402m. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.