Abstract
Inhalants, including toluene, target the addiction neurocircuitry and are often one of the first drugs of abuse tried by adolescents. The medial prefrontal cortex (mPFC) is involved in regulating goal-directed/reward-motivated behaviors and different mPFC sub-regions have been proposed to promote (prelimbic, PRL) or inhibit (infralimbic, IL) these behaviors. While this dichotomy has been studied in the context of other drugs of abuse, it is not known whether toluene exposure differentially affects neurons within PRL and IL regions. To address this question, we used whole-cell electrophysiology and determined the intrinsic excitability of PRL and IL pyramidal neurons in adolescent rats 24 h following a brief exposure to air or toluene vapor (10 500 p.p.m.). Prior to exposure, fluorescent retrobeads were injected into the NAc core (NAcc) or shell (NAcs) sub-regions to identify projection-specific mPFC neurons. In toluene treated adolescent rats, layer 5/6 NAcc projecting PRL (PRL5/6) neurons fired fewer action potentials and this was associated with increased rheobase, increased spike duration, and reductions in membrane resistance and amplitude of the Ih current. No changes in excitability were observed in layer 2/3 NAcc projecting PRL (PRL2/3) neurons. In contrast to PRL neurons, layer 5 IL (IL5) and layer 2/3 (IL2/3) NAcc projecting neurons showed enhanced firing in toluene-exposed animals and in IL5 neurons, this was associated with a reduction in rheobase and AHP. For NAcs projecting neurons, toluene exposure significantly decreased firing of IL5 neurons and this was accompanied by an increased rheobase, increased spike duration, and reduced Ih amplitude. The intrinsic excitability of PRL5, PRL2/3, and IL2/3 neurons projecting to the NAcs was not affected by exposure to toluene. The changes in excitability observed 24 h after toluene exposure were not observed when recordings were performed 7 days after the exposure. Finally, there were no changes in intrinsic excitability of any region in rats exposed to toluene as adults. These findings demonstrate that specific projections of the reward circuitry are uniquely susceptible to the effects of toluene during adolescence supporting the idea that adolescence is a critical period of the development that is vulnerable to drugs of abuse.
Introduction
Volatile organic solvents (VOS) have a number of important uses and are found in a large number of household and commercial products (eg, spray paints, cleaners, and adhesives). Because these agents are legal and relatively inexpensive to obtain (Lubman et al, 2008; Balster et al, 2009), VOS are voluntary inhaled by individuals seeking intoxication and euphoria (Balster et al, 2009). Although misuse of volatile organic solvents to achieve intoxication or a ‘high’ is prevalent among different socio-economic populations of all ages, inhalants are often one of the first drugs of abuse tried by adolescents and young adults (NSDUH, 2013). Use of these compounds by these individuals is of particular concern as adolescence is a critical period for brain development and refinement (Spear, 2000). Inhalant abuse during adolescence may disturb normal brain maturation and cause lasting deficits in cognition as well as impairments in the control of normal behavior (Rosenberg et al, 2002; Crews et al, 2007).
Similar to other drugs of abuse, inhalants such as toluene (methylbenzene) have high abuse potential and rats and mice exhibit conditioned place preference for toluene (Gerasimov et al, 2003; Lee et al, 2006); a behavior used to determine interactions of a drug with reward circuitry (Kobrin et al, 2015). Moreover, both mice (Blokhina et al, 2004) and nonhuman primates (Weiss et al, 1979) will self-administer toluene validating its use as a drug of abuse. The rewarding effects of toluene, like other drugs, are likely mediated by its interaction with key brain regions of the reward circuitry including the medial prefrontal cortex (mPFC), nucleus accumbens (NAc), and ventral tegmental area (VTA). In fact, toluene stimulates the firing of VTA neurons both in vivo (Riegel et al, 2007) and in ex vivo slices (Nimitvilai et al, 2016) and dopamine levels in the mPFC and NAc are elevated following toluene exposure (Gerasimov et al, 2002; Koga et al, 2007). Previous work from this laboratory also shows that a brief in vivo exposure to toluene vapor causes long-lasting (~3 weeks) and selective modifications in glutamatergic synaptic plasticity of VTA DA neurons that project to the NAc (Beckley et al, 2013). Although inputs to the NAc that arise from the mPFC are crucial for the associative learning (Block et al, 2007; Burgos-Robles et al, 2013) and recall of reward-related responses to other drugs of abuse (eg, cocaine, methamphetamine, ecstasy) (Peters et al, 2009; Rocha and Kalivas, 2010; Ball and Slane, 2012), nothing is known regarding whether these connections are also affected by toluene exposure.
The NAc is divided into the core (NAcc) and shell (NAcs) based on features including cytoarchitecture (Meredith et al, 1992; Groenewegen et al, 1999) and electrophysiological characteristics (O'Donnell and Grace, 1993). The mPFC in rodents can be sub-divided into prelimbic (PRL) and infralimbic (IL) sub-regions and mPFC inputs into the NAc occur are arranged in a topographical gradient with dorsal PRL neurons primarily projecting to the NAcc and ventral IL neurons targeting the NAcs (Ding et al, 2001). In addition, afferent projections to the NAcc arise from superficial (2/3) and deep (5/6) layers of the mPFC, while those going to the NAcs are primarily located in deep layers of the mPFC with little input from superficial layers (Ding et al, 2001). Results from recent studies demonstrate that behavioral responses to drugs with high abuse potential (eg, cocaine, methamphetamine, ecstasy) appear to be under the dichotomous control of the projection-specific inputs of the PRL/IL mPFC to the NAcc and NAcs (Peters et al, 2009; Rocha and Kalivas, 2010; Ball and Slane, 2012). To date, there is little known regarding the actions of toluene exposure on mPFC-NAc neurocircuitry and whether any potential changes show layer or age specificity.
To address this question, we conducted whole-cell electrophysiological recordings from NAc-projecting PRL/IL pyramidal neurons located in layers 2/3 and layers 5/6 of adolescent and adult rats exposed to air or toluene. To identify mPFC pyramidal neurons that projected to NAc sub-regions, fluorescent retrobeads were injected into the NAcc or NAcs prior to vapor exposure. The findings of this study show that toluene induces reversible and sub-region selective changes in mPFC excitability in adolescent, but not adult rats.
Materials and methods
Animals
Eighty male Sprague-Dawley rats were purchased at age P21 from Harlan Labs (Indianapolis, IN) and randomly assigned to air or toluene treatment groups. All rats were pair-housed in an environmentally controlled facility at the Medical University of South Carolina under a 12 h light/dark cycle. Food and water were provided ad libitum. Rats were handled in accordance with the Medical University of South Carolina Institutional Animal Care and Use Committee protocols.
Retrobead Labeling
Fluorescent retrograde tracers (Lumafluor; Naples, FL) were injected bilaterally (150 nl per side) into the NAc core (rostral to bregma; in mm +2.0, lateral ±1.4, ventral −6.0) or NAc shell (rostral to bregma; in mm +2.0, lateral ±0.8, ventral −6.2; coordinates modified for age from (Paxinos and Watson, 2005) of isoflurane anesthetized rats aged P29-P30. Rats were allowed to recover in their home cage for a minimum of 10 days prior to toluene exposure.
Toluene Vapor Exposure
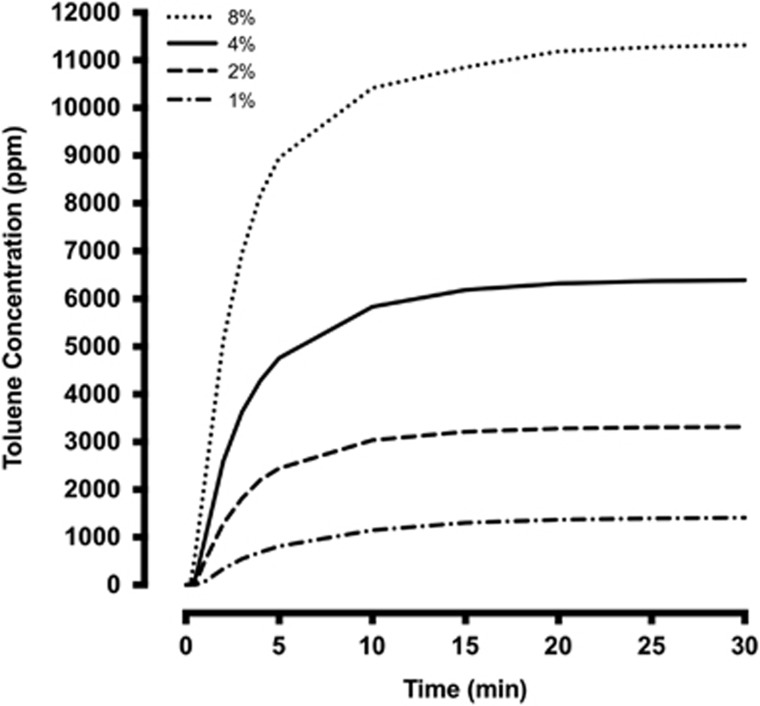
Adolescent (P40–43) and adult (P96–99) rats were habituated to a 30 × 30 × 30 cm anesthesia chamber (Plas Labs, Lansing, MI) fitted with an inlet and an outlet for 60 min. The next day, rats were placed in the chamber and allowed to habituate in air for 10 min followed by administration of either air or toluene vapor (Sigma-Aldrich, St Louis, MO) using a sevoflurane vaporizer (a concentration within the range used by human solvent abusers). Toluene-exposed rats received two 10-minute exposures to toluene (ramped from 0 to 10 500 p.p.m.) separated by a 10-minute air purge and a final 20-minute air washout. Airflow was maintained at 4 l/min at all times. Air-treated controls were placed in the chamber for the same total duration as the toluene treated rats (60 min). A portable gas detector (DOD Technologies, Cary, IL) was used to monitor chamber toluene concentrations during the exposure periods (Figure 1).
Figure 1.
Vaporizer output curves at 1, 2, 4, and 8% settings. Air flow was set at 4 l/min for all settings. Measurements were taken at 15 s, 30 s, 45 s, 1 min, 2 min, 3 min, 4 min, 5 min, 10 min, 15 min, 20 min, 25 min, and 30 min. Maximum output is reached at ~10 min.
Preparation of Brain Slices
One or seven days after exposure to air or toluene, rats were anesthetized with urethane (3.0 mg/kg, i.p.) and perfused with an ice-cold sectioning solution consisting of (in mM): 200 sucrose, 1.9 KCl, 6 MgSO4, 1.4 NaH2PO4, 25 NaHCO3, 0.5 CaCl2, 10 glucose, and 0.4 ascorbic acid; pH 7.35–7.45 with 310–320 mOsm. The brains were then immediately harvested and coronal brain sections (300 μm) containing the mPFC were sliced on a Leica VT1000S vibratome (Leica Biosystems, Buffalo Grove, IL) in oxygenated (95% O2; 5% CO2) sectioning solution and then transferred to a holding chamber containing normal artificial cerebrospinal fluid (aCSF; in mM): 125 NaCl, 2.5 KCl, 25 NaHCO3, 1.4 NaH2PO4, 1.3 MgCl2, 2 CaCl2, and 10 glucose; pH 7.35–7.45, with 310–320 mOsm (Beckley et al, 2013). Brain slices were incubated at 34 °C for 30 min and allowed to recover at room temperature for an additional 45 min.
Ex Vivo Electrophysiology
Brain slices were transferred to the recording chamber and perfused with oxygenated and heated (~34 °C) aCSF at a flow rate of 2 ml/min. The temperature was maintained during the course of the recordings with in-line and bath heaters (Warner Instruments, Hamden, CT). Pyramidal neurons within layers 2/3 or 5/6 of the PRL and IL sub-regions were visually identified using a Zeiss FS2 microscope (Zeiss, Thorndale, NY) according to landmarks illustrated in a rat brain atlas (Paxinos and Watson, 2005). Recording pipettes were constructed from thin-walled borosilicate capillary glass tubing (I.D.=1.17 mm, O.D. 1.50 mm; Warner Instruments, Hamden, CT), pulled with a horizontal pipette puller (P-97 Sutter Instrument Co., Novata, CA). Pipettes were filled with an internal solution containing (in mM): 120 K-gluconate, 10 HEPES, 10 KCl, 2 MgCl2, 2 Na2ATP, 0.3 NaGTP, 1 EGTA and 0.2% biocytin; pH 7.35–7.45, and 285–295 mOsm and had resistances ranging from 3–5 MΩ. After a stable gigaohm seal was formed, light suction was applied to break through the cell membrane and achieve whole-cell access. Neurons with an access resistance of neurons greater than 20 mOhm were not used for analysis. Recorded events were acquired with an Axon MultiClamp 700A (Molecular Devices, Union City, CA), digitized at a sampling rate of 10 kHz (filtered at 4 kHz) with an Instrutech ITC-18 analog-digital converter (HEKA Instruments, Bellmore, NY) controlled by AxographX software (Axograph Scientific, Sydney, Australia) running on a Macintosh G4 computer (Apple, Cupertino, CA). The resting membrane potential (RMP) of all neurons was first recorded and then adjusted to −70 mV for electrophysiological assessments of excitability. Action potential firing was induced by a series of 500 ms current steps (−400 pA to +500 pA) incremented in +25 pA steps. Recordings were analyzed offline for the number of spikes in response to each current step, threshold (mV), rheobase (pA), action potential peak amplitude (mV), action potential half-width (ms), after-hyperpolarization (AHP; mV) and Ih -current (mV) using AxographX software. In a subset of experiments, ZD7288 (20 μM; Tocris Bioscience, Bristol, UK) was added to the aCSF for 10 min to block hyperpolarization-activated cyclic nucleotide-gated (HCN) channels. In another subset of experiments, the recording aCSF was supplemented with kynurenic acid (1 mM; Tocris Bioscience, Minneapolis, MN), strychnine (1 μM Tocris Bioscience, Minneapolis, MN), picrotoxin (100 μM Tocris Bioscience, Minneapolis, MN), and CGP55845 (1 μM Tocris Bioscience, Minneapolis, MN) to block N-methyl-D-aspartate (NMDA), glycine, GABAA, and GABAB receptors respectively.
Statistical Analysis
Data were analyzed using Graphpad Prism Software v7 (GraphPad Software Inc., La Jolla, CA) and SPSS (v24; IBM, Armonk, NY). Membrane properties of neurons from air and toluene exposed rats were compared with unpaired t-tests. Membrane properties of neurons from air and toluene exposed rats were compared before and after application of ZD7288, picrotoxin, CGP, strychnine, and kyneurinic acid with paired t-tests. Current-voltage relationships were analyzed with mixed analysis of variance (Anova) and Sidaks post hoc contrasts. Differences were considered significant with α<0.05. Data are presented as mean and SEM. The number of neurons tested under each condition ranged from 6–12.
Results
Prefrontal Cortex Projections to the NAcc and NAcs
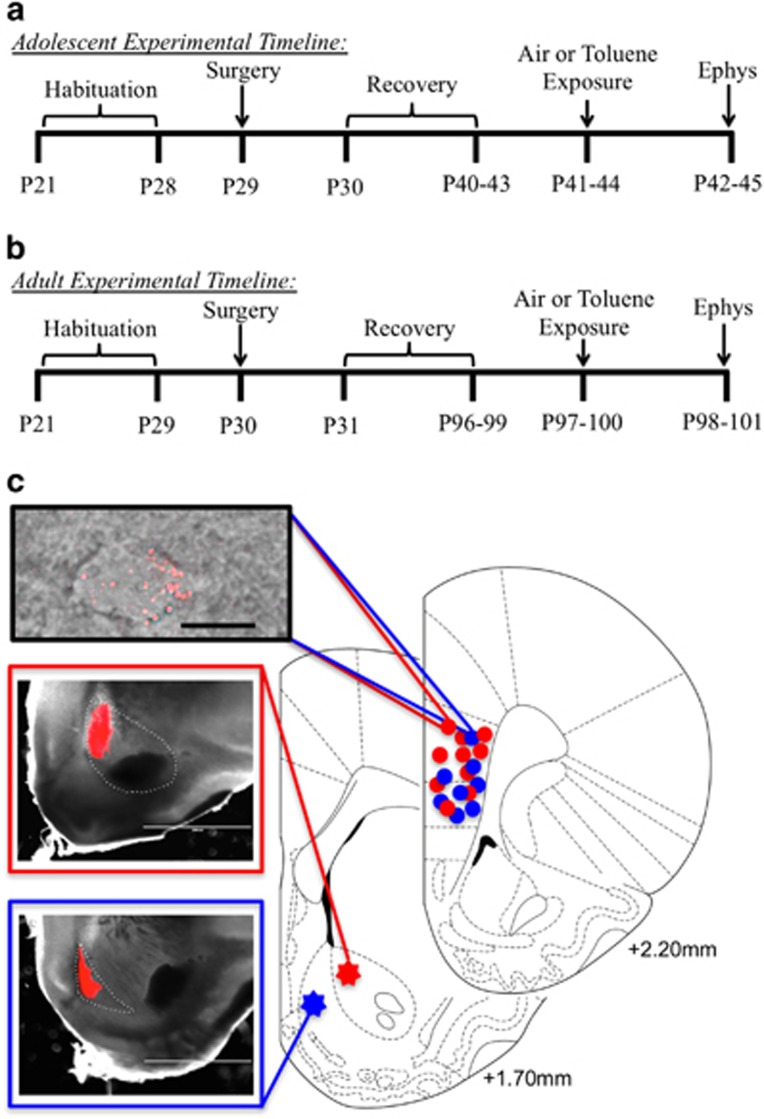
Rats were injected with fluorescent retrobeads into either the NAcc (n=44) or NAcs (n=36) on postnatal day 29–30 (Figure 2a and b). Figure 2c shows representative examples of injections demonstrating that retrobeads were confined to either the NAcc or NAcs. A recovery period of 10–14 days for adolescents and 65–69 days for adults yielded reliable labeling of core- or shell-projecting pyramidal neurons in the prefrontal cortex (Figure 2c). Pyramidal neurons projecting to the NAcc were located throughout the PRL and IL of the mPFC while NAcs-projecting pyramidal neurons were primarily located in the IL of the mPFC. Labeling of NAcs-projecting neurons in the ventral portions of the PRL was also observed particularly in PRL layers 5/6.
Figure 2.
Experimental timeline and localization of retrograde tracer in NAc and mPFC. Timeline shows procedures and ages of rats for experiments involving adolescent (a) and adult (b) animals. (c) Representative images and atlas drawings showing location of retrobead injections and expression. Atlas diagrams of cortical rat brain slices (redrawn from Paxinos and Watson, 2005) show representative locations of retrobead injections in the NAc and bead labelled mPFC neurons used for recording. Images show a PRL L5 pyramidal neuron expressing fluorescent retrobeads (top; scale bar 25 μm) and examples of retrobead injections in core (middle) and shell (bottom) of the NAc.
Toluene Exposure Induces Projection- and Sub-Region Specific Alterations in the Intrinsic Excitability of mPFC Pyramidal Neurons from Adolescent Rats
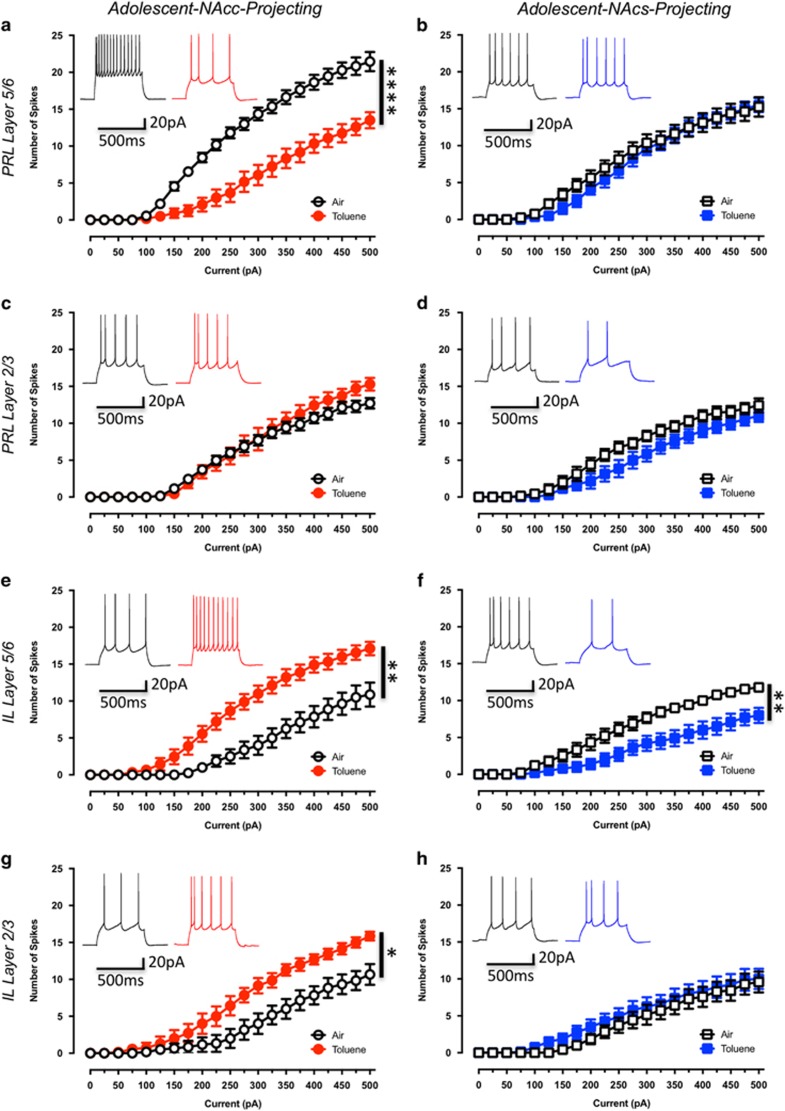
Approximately 2 weeks after injecting the retrograde tracers, adolescent rats (P41–44) were habituated to the vapor chamber and on the next day exposed to either toluene or air. Following exposure, rats were returned to their home cage and slices were prepared for recordings the following day. Evoked firing was significantly reduced in PRL5/6-NAcc neurons from toluene-exposed rats as compared to those from air-exposed control rats (drug effect: F(1,21)=27.94, p<0.0001 Figure 3a). Firing of PRL2/3-NAcc neurons was not affected by toluene exposure (drug effect: F(1,12)=0.36, p=0.56; Figure 3c). In contrast to PRL5/6-NAcc neurons, evoked firing of IL5/6-NAcc neurons was significantly enhanced in toluene exposed rats (drug effect: F(1,15)=16.46, p<0.001; Figure 3e). A similar increase in firing was also observed in IL2/3-NAcc neurons (drug effect: F(1,11)=7.62, p<0.05; Figure 3g). To determine whether the differential effects of toluene on mPFC firing were dependent on the projection target, the intrinsic excitability of neurons projecting to the NAcs was determined. Toluene treatment significantly reduced evoked firing of IL5/6-NAcs projecting neurons (drug effect: F(1,17)=9.70, p<0.01; Figure 3f) but did not affect intrinsic excitability of PRL5/6-NAcs (drug effect: F(1,14)=0.34, p=0.57; Figure 3b), PRL2/3-NAcs (drug effect: F(1,12)=3.03, p=0.11; Figure 3d), or IL2/3-NAcs (drug effect: F(1,13)=0.57, p=0.46; Figure 3h) neurons. These findings suggest that specific connections within mPFC-NAc reward circuitry are uniquely disrupted following a brief exposure to toluene.
Figure 3.
Toluene exposure produces sub-region and projection-target selective alterations in current-evoked firing of adolescent mPFC neurons. Figures show the effect of toluene exposure on firing of prelimbic and infralimbic mPFC neurons projecting to the core (a—Air: n=11, Toluene: n=12; c—Air: n=7, Toluene: n=7; e—Air: n=8, Toluene: n=9; g—Air: n=6, Toluene: n=7) or shell (b—Air: n=8, Toluene: n=8, d—Air: n=7, Toluene: n=7; f—Air: n=9, Toluene: n=10; h—Air: n=7, Toluene: n=8) of the NAc. Traces in each figure show examples of firing (evoked with a 250 pA current step) from air (left trace) or toluene (right trace) exposed rats measured 24 h after exposure to 10 500 p.p.m. toluene vapor. Data are presented as mean and SEM. Symbol (*) indicates main effect of toluene treatment on firing (two-way repeated measures ANOVA; p<0.05).
Toluene-Induced Dysfunction of mPFC Pyramidal Neuron Intrinsic Excitability is Transient
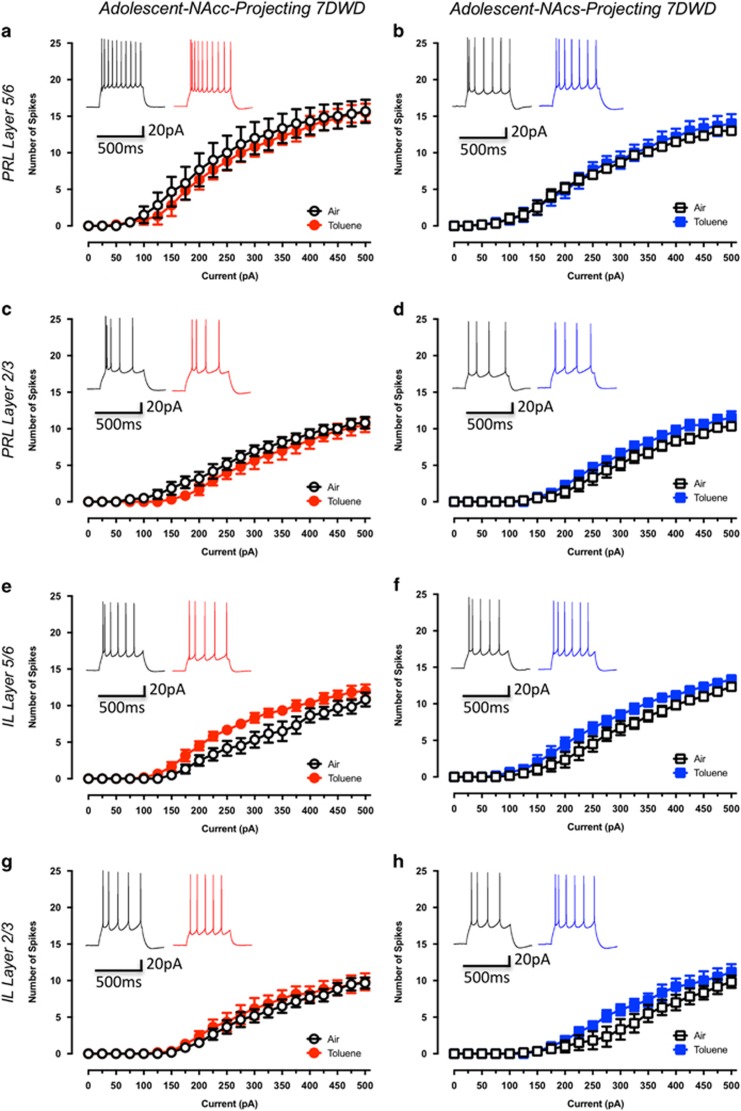
To determine whether toluene-induced changes in firing persist beyond 24 h, current-evoked firing was assessed in adolescent rats 7 days following air or toluene exposure. In contrast to the changes observed 1 day after toluene exposure, NAcc-projecting pyramidal neurons showed no change in firing at 7 days post-exposure (PRL5/6-NAcc: drug effect: F(1,10)=0.16, p=0.70; Figure 4a, PRL2/3-NAcc: drug effect: F(1,10)=1.11, p=0.32; Figure 4c, IL5/6-NAcc: drug effect: F(1,10)=4.58, p=0.06; Figure 4e, IL2/3-NAcc: drug effect: F(1,10)=0.29, p=0.60; Figure 4g). Similarly, there were no differences in firing of NAcs-projecting pyramidal neurons following a 7-day recovery from toluene exposure (PRL5/6-NAcs: drug effect: F(1,10)=0.14, p=0.71; Figure 4b, PRL2/3-NAcs: drug effect: F(1,10)=1.43, p=0.26; Figure 4d, IL5/6-NAcs: drug effect: F(1,10)=2.70, p=0.13; Figure 4f, IL2/3-NAcs: drug effect: F(1,10)=1.55, p=0.24; Figure 4h). These findings indicate that a single binge exposure to toluene produces transient changes in excitability of mPFC neurons that varies based on both the layer and the NAc projection target.
Figure 4.
Toluene-induced alterations to the intrinsic excitability of mPFC-NAc neurons of adolescent rats is transient. Figures show the effect of toluene exposure on firing of prelimbic and infralimbic mPFC neurons projecting to the core (a—Air: n=6, Toluene: n=6; c—Air: n=6, Toluene: n=6; e—Air: n=6, Toluene: n=6; g—Air: n=6, Toluene: n=6) or shell (b—Air: n=6, Toluene: n=6; d—Air: n=6, Toluene: n=6; f—Air: n=6, Toluene: n=6; h—Air: n=6, Toluene: n=6) of the NAc. Traces in each figure show examples of firing (evoked with a 250 pA current step) from air (left trace) or toluene (right trace) exposed rats 7 day following exposure to 10 500 p.p.m. toluene vapor. Data are presented as mean and SEM.
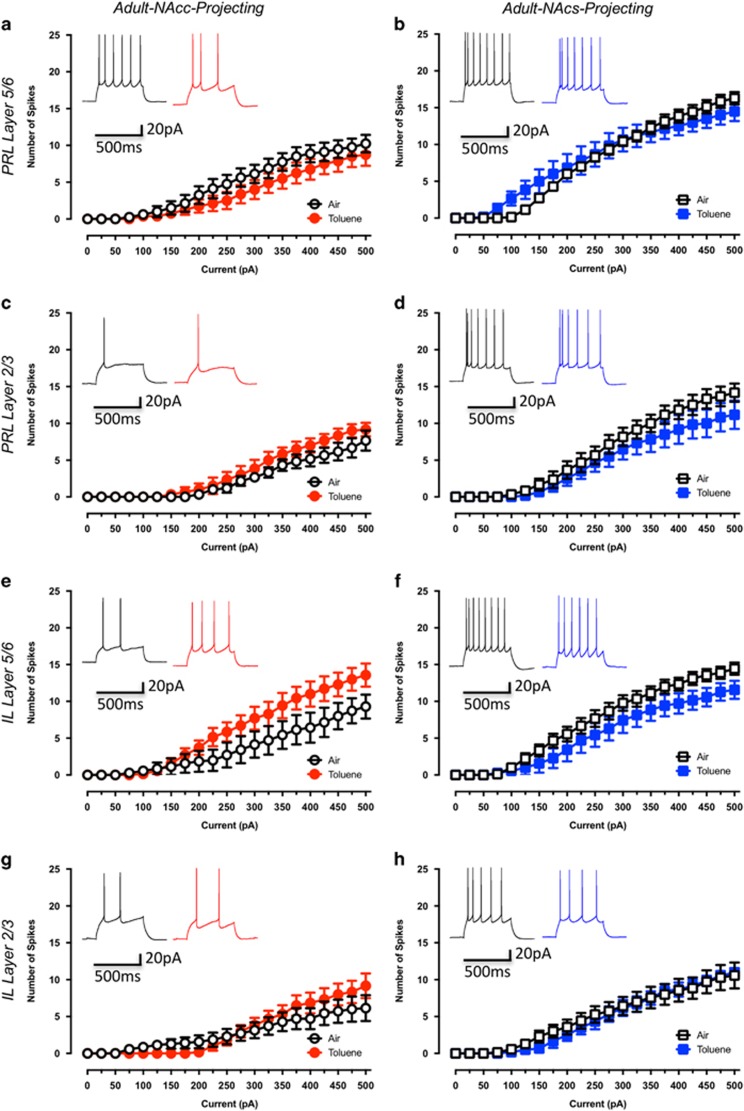
Adult Rats Exhibit Reduced Sensitivity to Toluene-induced Alterations in the Intrinsic Excitability of mPFC Pyramidal Neurons
To extend the findings obtained in adolescent rats, we assessed the consequences of a brief exposure to toluene on core- or shell-projecting mPFC pyramidal neurons from adult rats. These animals were 98–101 days old at the time of exposure and recording. Unlike adolescent animals, toluene exposure during adulthood did not alter the intrinsic excitability of NAcc-projecting pyramidal neurons in the different mPFC sub-regions tested (PRL5/6-NAcc: drug effect: F(1,14)=1.14, p=0.30; Figure 5a, PRL2/3-NAcc: drug effect: F(1,10)=1.47, p=0.25; Figure 5c, IL5/6-NAcc: drug effect: F(1,12)=2.24, p=0.16; Figure 5e, IL2/3-NAcc: drug effect: F(1,11)=0.14, p=0.72; Figure 5g). Similarly, evoked firing of NAcs-projecting pyramidal neurons from toluene-treated adult rats was similar to air controls in all sub-regions (PRL5/6-NAcs: drug effect: F(1,11)=0.02, p=0.87; Figure 5b, PRL2/3-NAcs: drug effect: F(1,10)=1.23, p=0.29; Figure 5d, IL5/6-NAcs: drug effect: F(1,14)=2.07, p=0.17; Figure 5f, IL2/3-NAcs: drug effect: F(1,101=0.07, p=0.79; Figure 5h).
Figure 5.
Toluene exposure during adulthood has no effect on the intrinsic excitability of NAc projecting mPFC neurons. Figures show the effect of toluene exposure on firing of prelimbic and infralimbic mPFC neurons projecting to the core (a—Air: n=8, Toluene: n=8; c—Air: n=6, Toluene: n=6; e—Air: n=7, Toluene: n=7; g—Air: n=7, Toluene: n=6) or shell (b—Air: n=7, Toluene: n=6; d—Air: n=6, Toluene: n=6; f—Air: n=9, Toluene: n=7; h—Air: n=7, Toluene: n=6) of the NAc. Traces in each figure show examples of current-evoked firing (evoked with a 250 pA current step) from air (left trace) or toluene (right trace) exposed adult rats 24 h following exposure to 10 500 p.p.m. toluene vapor. Data are presented as mean and SEM.
During the course of these studies, we noted that adolescent rats emerged from the chamber mostly sedated and exhibited a near complete loss of motor function that persisted for several minutes. In contrast, while adult rats showed clear signs of intoxication (motor incoordination, ataxia) following toluene exposure, they were not fully sedated. To determine whether increasing sedation of adult rats would reveal changes in firing, a group of adult rats received a single 20-min exposure to toluene that produced levels of sedation and loss of motor function comparable to that in adolescent rats. However, despite this enhanced exposure protocol, no changes were observed in the intrinsic excitability of NAcc-projecting mPFC pyramidal neurons in these animals (PRL5/6-NAcc: drug effect: F(1,12)=0.04, p=0.83; Supplementary Figure 1a, IL5/6-NAcc: drug effect: F(1,12)=1.42, p=0.26; Supplementary Figure 1b). These findings suggest that the adolescent brain is particularly susceptible to toluene.
Effects of Toluene on the Electrophysiological Properties of Neurons in the mPFC
To identify potential mechanisms that underlie toluene’s effect on mPFC neuron excitability, we assessed the electrophysiological characteristics of pyramidal neurons from adolescent and adult rats exposed to air or toluene (Supplementary Information, Supplementary Table 1). As compared to neurons from air controls, PRL5/6-NAcc neurons from toluene-exposed rats had increases in rheobase (t(21)=4.63, p<0.05) and spike duration (t(21)=2.20, p<0.05) with reductions in membrane resistance (t(21)=2.33, p<0.05), and Ih amplitude (t(21)=4.05, p<0.05). IL5/6-NAcc neurons of toluene-exposed rats had a reduced rheobase (t(15)=2.88, p<0.05) and reduced AHP (t(15)=2.35, p<0.05) as compared to air-exposed controls. Although no significant change in evoked firing was observed in PRL2/3-NAcc neurons from toluene treated animals, these neurons had a reduction in AHP amplitude (t(12)=2.88, p<0.05) as compared to neurons from air-exposed rats. There were no significant changes in the membrane properties of IL2/3-NAcc neurons between air- and toluene-exposed rats. IL5/6-NAcs neurons from toluene-exposed rats had an increased rheobase (t(17)=2.64, p<0.05), increased spike duration (t(17)=2.17, p<0.05), and reduced Ih amplitude (t(17)=3.25, p<0.05) as compared to air-exposed controls. There were no significant changes between air- and toluene-exposed rats in the membrane properties of PRL5/6-NAcs, PRL2/3-NAcs or IL2/3-NAcs neurons. In addition, there were no significant differences in the membrane properties of NAcc- or NAcs-projecting neurons of any prefrontal sub-region from adolescent rats 7 days after toluene exposure (Supplementary Information,Supplementary Table 2). However, membrane resistance was increased in PRL2/3-NAcs (t(10)=3.47, p<0.05; Supplementary Information, Supplementary Table 2), IL5/6-NAcs (t(10)=4.43, p<0.05; Supplementary Information,Supplementary Table 2), and IL2/3-NAcs (t(10)=2.79, p<0.05; Supplementary Table 2) neurons. The membrane properties of NAcc- or NAcs-projecting neurons in toluene-treated adult rats were not different from controls (Supplementary Information, Supplementary Table 1).
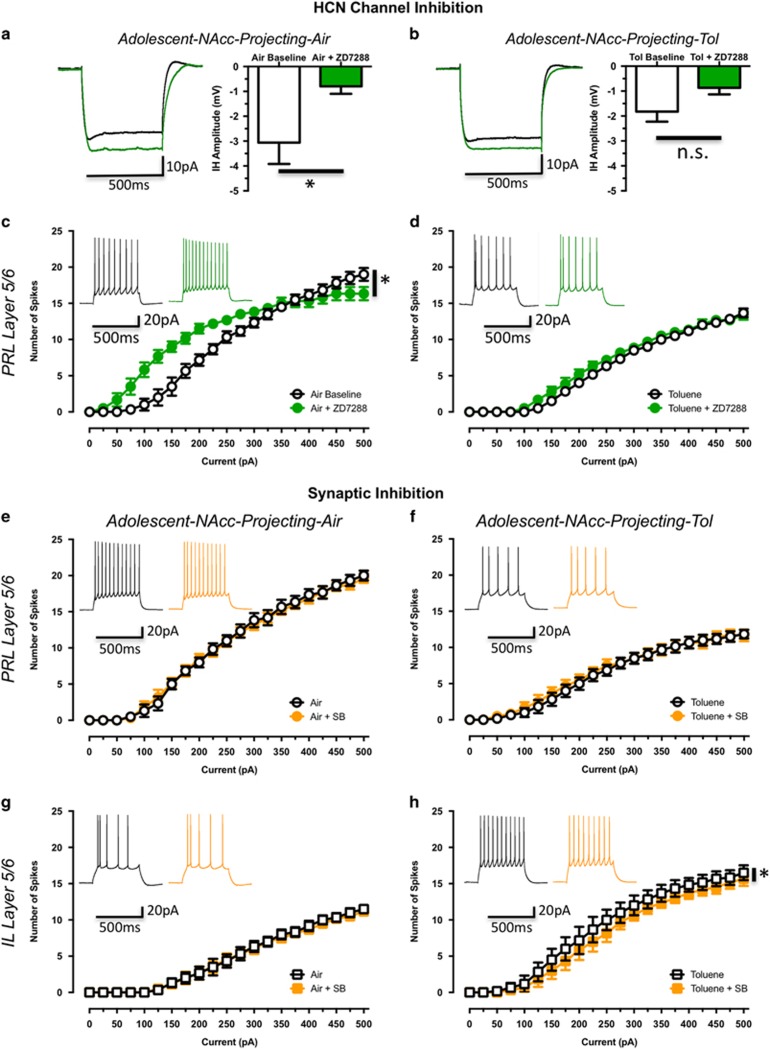
A partial least squares (PLS) analysis was then performed to determine which membrane properties data were most related to the changes in spiking (Beckley et al, 2015). The three variables with the highest variable in projection (VIP) scores were rheobase, action potential duration and Ih analysis. Of these Ih can be directly manipulated and thus current-evoked firing was measured in the absence and presence of the Ih channel blocker ZD7288. These studies were done with PRL5/6-NAcc neurons as they showed significant reductions in Ih current amplitude following toluene exposure (Supplementary Information, Supplementary Table 1). Spiking was first measured under basal conditions and then ZD7288 (20 μM) was applied for 10 min. As blockade of Ih normally hyperpolarizes the RMP (Maccaferri and McBain, 1996; Albertson et al, 2011) that in itself affects firing, all neurons were re-clamped to −70 mV following exposure to ZD7288. In PRL5/6-NAcc neurons from air-exposed animals, ZD7288 significantly reduced Ih amplitude (t(5)=3.37, p<0.05; Supplementary Information, Supplementary Table 3; Figure 6a) and left-shifted the current-spiking relationship (ZD effect: F(1,10)=5.31, p<0.05; Figure 6c). In the presence of ZD7288, firing was enhanced at modest depolarizing currents (+25 to +250 pA) but decreased at higher current steps. In PRL5/6-NAcc neurons from toluene-exposed animals, firing in the absence of ZD7288 was reduced as compared to air controls (Figure 6b) and spike duration was increased (t(10)=2.45, p<0.05; Supplementary Information,Supplementary Table 3), consistent with previous findings. Ih amplitude was also reduced in these neurons as compared to air controls (t(10)=2.30, p<0.05; Supplementary Information,Supplementary Table 3). Following exposure to ZD7288, there was a modest, but statistically insignificant reduction in Ih (t(5)=2.82, p=0.17; Supplementary Information, Supplementary Table 3; Figure 6b) or firing (ZD effect: F(1,10)=1.49, p=0.25; Figure 6d) in PRL5/6 neurons from toluene-treated animals. Together, these findings suggest that changes in HCN channel function are not the major factor underlying alterations in firing of mPFC neurons following exposure to toluene.
Figure 6.
Effects of blockers of Ih channels and synaptic transmission on firing of PRL5/6-NAcc mPFC neurons from adolescent rats exposed to air or toluene vapor. (a and b) Traces show representative responses of neurons from air (a; n=6) or toluene-exposed (b; n=6) rats to hyperpolarizing current steps in the absence (black) and presence (green) of 20 μM ZD7288. Summary plots show amplitude of Ih in the absence and presence of ZD7288. (c and d) Effect of ZD7288 on current-evoked firing of neurons from air (c) or toluene-exposed (d) rats in the absence (black) and presence (green) of ZD7288. (e–h) Effect of blockers of synaptic transmission on current evoked firing of PRL5/6- and IL5/6-NAcc mPFC neurons from adolescent rats 24 h following exposure to air (e; n=6, g; n=6) or 10 500 p.p.m. toluene vapor (f; n=6, h; n=6). Traces in (e–h) show examples of current-evoked firing (evoked with a 250 pA current step) in neurons from air (left) or toluene (right) treated animals. Data are presented as mean and SEM. Symbol (*) value significantly (p<0.05) different from control (a and b; unpaired t-test; c–h; two-way repeated measures ANOVA).
In a final set of studies, recordings were carried out in the presence of synaptic blockers to determine whether alterations in synaptic activity may have contributed to changes in intrinsic excitability observed in toluene-treated animals. Following baseline measurements of firing, aCSF containing a cocktail of blockers (picrotoxin, CGP55845, strychnine, and kyneurinic acid) was perfused and current-evoked firing was again measured. There was no significant effect of blockers on the intrinsic excitability of PRL5/6-NAcc (SB effect: F(1,5)=0.08, p=0.79; Figure 6e) or IL5/6-NAcc (SB effect: F(1,5)=0.32, p=0.59; Figure 6g) neurons from air-treated rats. As previously observed (Figures 3a, e and 6c, d), binge exposure to toluene reduced firing of PRL5/6-NAcc neurons and enhanced firing of IL5/6-NAcc neurons as compared to respective air controls. Although synaptic blockers had no effect on firing of PRL5/6 neurons from toluene-treated animals (SB effect: F(1,5)=0.64, p=0.46; Figure 6f), there was a very small but statistically significant reduction in the firing of IL5/6-NAcc neurons from toluene-treated rats (SB effect: F(1,5)=7.28, p<0.05; Figure 6h). Analysis of membrane properties revealed a minor but significant increase in the AHP of IL5/6-NAcc neurons following perfusion with the synaptic blocker cocktail (Supplementary Table 3). These findings suggest that changes in evoked firing following toluene exposure are not due to altered synaptic signaling.
Discussion
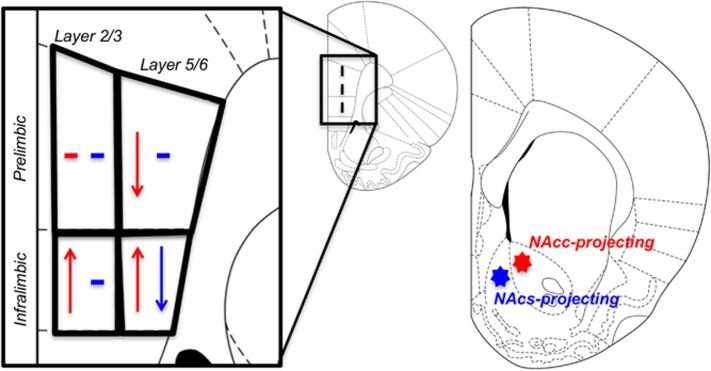
The major finding of the current study shows that a single binge exposure to toluene induces profound changes in mPFC excitability that are manifested in a projection/sub-region-specific and age-dependent manner (summarized in Figure 7). In adolescent rats, pyramidal neurons projecting to the NAcc showed changes in evoked firing that varied by mPFC sub-region with PRL neurons showing reduced firing and IL neurons enhanced firing after toluene exposure. The decrease in PRL firing was also layer specific with only deep layer neurons exhibiting changes in firing. Moreover, pyramidal neurons projecting to the NAcs also exhibited reduced firing, but only those from the deep layer IL sub-region. The toluene-induced changes in firing were apparent within 24 h of toluene vapor exposure but fully recovered after 7 days. The changes in mPFC excitability were only observed in adolescent rats, as toluene exposure in adulthood had no effect on evoked firing or measures of intrinsic excitability. These findings suggest that the vulnerability of adolescent mPFC neurons to abused inhalants such as toluene may be one important factor that contributes to the higher reported use of these agents by children and adolescents (NSDUH, 2013).
Figure 7.
Summary of mPFC intrinsic excitability changes in adolescent rats after 1 day withdrawal. Effects on mPFC-NAcc projecting neurons are shown in red. Changes in mPFC-NAcs projecting neurons are shown in blue. ↑ indicates an increase in intrinsic excitability. ↓ indicates a decrease in intrinsic excitability.—indicates no changes to intrinsic excitability.
The development of inhalant abuse likely involves drug-induced modifications to the physiology of the mesocorticolimbic reward circuitry. While the concentrations of toluene capable of producing alterations in neural circuitry vary somewhat based on the brain region examined, exposure to several thousand p.p.m. of toluene appears to be required. This is consistent with the use of volatile solvents for intoxication by humans that involves brief exposures to relatively high concentrations (5000–15 000 p.p.m.) of vapor (reviewed by Bowen et al, 2006). In a previous study from this laboratory, exposure to 5700 p.p.m. significantly enhanced the AMPA/NMDA ratio of NAcc and NAcs projecting VTA DA neurons but not those that project to the mPFC (Beckley et al, 2013). Exposures to similar concentrations (3000–7000 p.p.m.) of toluene have been shown to produce significant increases in extracellular dopamine in the mPFC and accumbens (Gerasimov et al, 2002; Koga et al, 2007) a consequence similar to that of other drugs of abuse, such as cocaine, that also modify reward circuitry (Gerasimov et al, 2002). Moreover, toluene concentrations greater than 800 p.p.m. are necessary for inducing conditioned place preference (Gerasimov et al, 2003; Lee et al, 2006). In concert with findings from Beckley et al (2013), the results of the present study report add to the growing appreciation of the selectivity of action of abused inhalants such as toluene and emphasize the need to identify projection-specific neurons when assessing drug action.
Behavioral responses (ie, extinction and reinstatement of drug-seeking) to drugs with high abuse potential (ie, cocaine, methamphetamine) have been suggested to be under the dichotomous control of projection-specific mPFC inputs with PRL-NAcc neurons promoting drug seeking and IL-NAcs neurons blocking this behavior (Peters et al, 2009; Rocha and Kalivas, 2010; Ball and Slane, 2012). While a compelling hypothesis, recent evidence suggests that the PRL/IL model of drug seeking may be too simplistic (Moorman et al, 2015). For example, context-induced reinstatement of heroin seeking was enhanced rather than inhibited by activation of IL-NAcs neurons (Bossert et al, 2011; Bossert et al, 2012) and reinstatement of alcohol self-administration was facilitated by inactivation not activation of PRL-NAcc (Willcocks and McNally, 2013). In the present study, the intrinsic excitability of both PRL-NAcc and IL-NAcs projections was significantly suppressed by a binge exposure to toluene while activity was enhanced in IL-NAcc neurons. How these changes relate to the voluntary use of abused inhalants is not yet known due to the lack of reliable models of toluene self-administration and reinstatement. Nonetheless, taken together with findings in the literature, these results suggest that different drugs of abuse may target unique ensembles of neurons within discrete mPFC subregions that contributes to differences in drug seeking and relapse behavior observed between different classes of abused substances.
While evidence is accumulating that abused inhalants share many characteristics with other more well-known drugs of abuse, most solvents including toluene possess a strong odor that could induce changes on its own. Thus, it is possible that the toluene-induced changes in the intrinsic excitability of mPFC observed in the present study may have arisen from odor-based contextual learning. However, we think this is unlikely for several reasons. Results from behavioral studies have consistently demonstrated that odor-based contextual learning is mediated by the orbital and insular cortices rather than those in more medial regions (Otto and Eichenbaum, 1992; Schoenbaum and Eichenbaum, 1995; Di Pietro et al, 2004). In addition, findings from a related study demonstrated that the dorsal agranular insular cortex, not the mPFC, regulates drug-seeking guided by olfactory cues (Di Pietro et al, 2006). Finally, in the present study, toluene had no effect on mPFC excitability in adult animals despite similar and in some cases more prolonged exposures. While we can not completely rule out the possibility that odor plays a role in the effects of toluene vapor on mPFC excitability, we think it more likely that these changes are mediated by direct actions of toluene on targets that regulate neuronal excitability.
As mentioned above, even a prolonged exposure to toluene vapor had little effect on the intrinsic excitability of mPFC neurons from adult animals suggesting that the adolescent mPFC-NAc circuitry is uniquely susceptible to toluene-induced modification. This could reflect age-dependent changes in either the rewarding or aversive effects of toluene. This idea is consistent with results from a number of studies involving other drugs of abuse including cocaine, alcohol, and nicotine (reviewed by Schramm-Sapyta et al, 2009). Alternatively, the blunted action of toluene in adult animals may reflect developmental differences in the biodistribution or metabolism of toluene. We noted that adolescent rats exposed to two 10-minute exposures to 10 500 p.p.m. toluene emerge from the chamber mostly sedated and exhibit a near complete loss of motor function that persists for several minutes. Adult rats subjected to the same exposure paradigm also showed clear signs of intoxication (motor incoordination, ataxia) but did not appear to fully sedated unless exposed for a longer period of time. The former observation is consistent with others (Samuel-Herter et al, 2014) wherein motor activity following toluene exposure recovers more quickly in adult rats than adolescents and young adults and in this study, the level of sedation did not contribute to the mPFC pathophysiology observed in adolescent rats. Interestingly, repeated exposures to 4000 p.p.m. toluene stimulates locomotor activity in both adolescents and adult rats, this effect is more robust in adults (Bowen et al, 2007). While differences in toluene metabolism may underlie the reported age-related differences in locomotor activity, recovery from intoxication, and mPFC physiology, the available literature on toluene biodistribution and metabolism is limited and often contradictory. For example, 1–2-year-old rats have been reported to experience greater toluene loads in the brain than 4-month-old animals following orally administered toluene (Gordon et al, 2010). However, the rate of formation of benzyl alcohol (the primary metabolite of toluene via hydroxylation) was shown to be four-times higher in mature male rats as a result of increased cytochrome p450 content (Nakajima et al, 1992). Future studies of toluene-induced reward (eg, toluene self-administration, toluene conditioned place preference) in adolescent and adult animals are needed to examine the relationship between brain concentrations of toluene and alterations in brain reward circuitry.
In the current study, binge exposure to toluene-induced opposite effects on current-evoked firing that were region, layer and projection target specific. Although these changes were accompanied by alterations in several electrophysiological parameters including spike duration, AHP and Ih amplitude; none of these parameters was sufficient to predict changes to intrinsic excitability. Among the changes observed, a reduction in Ih -current amplitude was associated with reduced firing in IL5/6-NAcs and PRL5/6-NAcc neurons. Hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are the primary mediators of Ih -currents and blockade of these channels with ZD7288 would be expected to enhance firing near the rheobase when depolarized from the same membrane potential as untreated neurons (Maccaferri and McBain, 1996; Albertson et al, 2011). In the current study, ZD7288 reduced firing of PRL5/6-NAcc neurons from control animals at maximal current step while firing around the rheobase was increased. Ih -currents and spiking was reduced in neurons from toluene-treated animals and ZD7288 had no further effect on firing in these neurons. In contrast, no changes were observed in the Ih amplitude of IL5/6-NAcc neurons from toluene-exposed rats but firing was enhanced and this was accompanied with a reduced AHP. The AHP is a major component of inter-spike interval timing and is regulated by calcium-activated K+ channels, including SK and BK channels (Sah and Faber, 2002). We have previously reported that BK channels can be inhibited by toluene (Del Re et al, 2006) suggesting that a persistent reduction in the AHP may have contributed to the increase in firing of these neurons. However, like changes in Ih, PRL2/3-NAcc neurons also exhibited a reduction in the AHP, but showed no significant increase in firing suggesting that alone, toluene-induced changes in AHP are not solely responsible for alterations in intrinsic excitability. Rather, the findings of the study suggest that following toluene exposure, there are multiple and neuronal-selective changes in yet to be identified factors that regulate the intrinsic excitability of mPFC neurons.
In summary, the present study revealed that toluene exposure induces projection- and sub-region specific alterations in the intrinsic excitability of mPFC pyramidal neurons during the critical period of adolescence. These findings add to a growing awareness that drug-induced dysfunction of NAc projecting mPFC neurons is a critical process that contributes to maladaptive behaviors related to drug-seeking and the transition from recreational drug use to addiction. They also suggest that the immature state of prefrontal neuronal circuitry during adolescence may generate a more vulnerable brain state to drug exposure during this time.
Funding and disclosure
The authors declare no conflict of interest.
Acknowledgments
We thank Jared Beneroff for excellent technical assistance during the experiments carried out for this study.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Albertson AJ, Yang J, Hablitz JJ (2011). Decreased hyperpolarization-activated currents in layer 5 pyramidal neurons enhances excitability in focal cortical dysplasia. J Neurophysiol 106: 2189–2200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ball KT, Slane M (2012). Differential involvement of prelimbic and infralimbic medial prefrontal cortex in discrete cue-induced reinstatement of 3,4-methylenedioxymethamphetamine (MDMA; ecstasy) seeking in rats. Psychopharmacology (Berl) 224: 377–385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balster RL, Cruz SL, Howard MO, Dell CA, Cottler LB (2009). Classification of abused inhalants. Addiction 104: 878–882. [DOI] [PubMed] [Google Scholar]
- Beckley JT, Evins CE, Fedarovich H, Gilstrap MJ, Woodward JJ (2013). Medial prefrontal cortex inversely regulates toluene-induced changes in markers of synaptic plasticity of mesolimbic dopamine neurons. J Neurosci 33: 804–813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beckley JT, Randall PK, Smith RJ, Hughes BA, Kalivas PW, Woodward JJ (2015). Phenotype-dependent inhibition of glutamatergic transmission on nucleus accumbens medium spiny neurons by the abused inhalant toluene. Addict Biol 21: 530–546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Block AE, Dhanji H, Thompson-Tardif SF, Floresco SB (2007). Thalamic-prefrontal cortical-ventral striatal circuitry mediates dissociable components of strategy set shifting. Cereb Cortex 17: 1625–1636. [DOI] [PubMed] [Google Scholar]
- Blokhina EA, Dravolina OA, Bespalov AY, Balster RL, Zvartau EE (2004). Intravenous self-administration of abused solvents and anesthetics in mice. Eur J Pharmacol 485: 211–218. [DOI] [PubMed] [Google Scholar]
- Bossert JM, Stern AL, Theberge FR, Cifani C, Koya E, Hope BT et al (2011). Ventral medial prefrontal cortex neuronal ensembles mediate context-induced relapse to heroin. Nat Neurosci 14: 420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossert JM, Stern AL, Theberge FR, Marchant NJ, Wang HL, Morales M et al (2012). Role of projections from ventral medial prefrontal cortex to nucleus accumbens shell in context-induced reinstatement of heroin seeking. J Neurosci 32: 4982–4991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowen SE, Batis JC, Paez-Martinez N, Cruz SL (2006). The last decade of solvent research in animal models of abuse: mechanistic and behavioral studies. Neurotoxicol Teratol 28: 636–647. [DOI] [PubMed] [Google Scholar]
- Bowen SE, Charlesworth JD, Tokarz ME, Wright MJ Jr., Wiley JL (2007). Decreased sensitivity in adolescent vs. adult rats to the locomotor activating effects of toluene. Neurotoxicol Teratol 29: 599–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgos-Robles A, Bravo-Rivera H, Quirk GJ (2013). Prelimbic and infralimbic neurons signal distinct aspects of appetitive instrumental behavior. PLoS ONE 8: e57575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crews F, He J, Hodge C (2007). Adolescent cortical development: a critical period of vulnerability for addiction. Pharmacol Biochem Behav 86: 189–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Re AM, Dopico AM, Woodward JJ (2006). Effects of the abused inhalant toluene on ethanol-sensitive potassium channels expressed in oocytes. Brain Res 1087: 75–82. [DOI] [PubMed] [Google Scholar]
- Di Pietro NC, Black YD, Green-Jordan K, Eichenbaum HB, Kantak KM (2004). Complementary tasks to measure working memory in distinct prefrontal cortex subregions in rats. Behav Neurosci 118: 1042–1051. [DOI] [PubMed] [Google Scholar]
- Di Pietro NC, Black YD, Kantak KM (2006). Context-dependent prefrontal cortex regulation of cocaine self-administration and reinstatement behaviors in rats. Eur J Neurosci 24: 3285–3298. [DOI] [PubMed] [Google Scholar]
- Ding DC, Gabbott PL, Totterdell S (2001). Differences in the laminar origin of projections from the medial prefrontal cortex to the nucleus accumbens shell and core regions in the rat. Brain Res 917: 81–89. [DOI] [PubMed] [Google Scholar]
- Gerasimov MR, Collier L, Ferrieri A, Alexoff D, Lee D, Gifford AN et al (2003). Toluene inhalation produces a conditioned place preference in rats. Eur J Pharmacol 477: 45–52. [DOI] [PubMed] [Google Scholar]
- Gerasimov MR, Schiffer WK, Marstellar D, Ferrieri R, Alexoff D, Dewey SL (2002). Toluene inhalation produces regionally specific changes in extracellular dopamine. Drug Alcohol Depend 65: 243–251. [DOI] [PubMed] [Google Scholar]
- Gordon CJ, Gottipolu RR, Kenyon EM, Thomas R, Schladweiler MC, Mack CM et al (2010). Aging and susceptibility to toluene in rats: a pharmacokinetic, biomarker, and physiological approach. J Toxicol Environ Health A 73: 301–318. [DOI] [PubMed] [Google Scholar]
- Groenewegen HJ, Wright CI, Beijer AV, Voorn P (1999). Convergence and segregation of ventral striatal inputs and outputs. Ann N Y Acad Sci 877: 49–63. [DOI] [PubMed] [Google Scholar]
- Kobrin KL, Moody O, Arena DT, Moore CF, Heinrichs SC, Kaplan GB (2015). Acquisition of morphine conditioned place preference increases the dendritic complexity of nucleus accumbens core neurons. Addict Biol 21: 1086–1096. [DOI] [PubMed] [Google Scholar]
- Koga Y, Higashi S, Kawahara H, Ohsumi T (2007). Toluene inhalation increases extracellular noradrenaline and dopamine in the medial prefrontal cortex and nucleus accumbens in freely-moving rats. J Kyushu Dent Soc 61: 39–54. [Google Scholar]
- Lee DE, Gerasimov MR, Schiffer WK, Gifford AN (2006). Concentration-dependent conditioned place preference to inhaled toluene vapors in rats. Drug Alcohol Depend 85: 87–90. [DOI] [PubMed] [Google Scholar]
- Lubman DI, Yucel M, Lawrence AJ (2008). Inhalant abuse among adolescents: neurobiological considerations. Br J Pharmacol 154: 316–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maccaferri G, McBain CJ (1996). The hyperpolarization-activated current (Ih) and its contribution to pacemaker activity in rat CA1 hippocampal stratum oriens-alveus interneurones. J Physiol 497(Pt 1): 119–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meredith GE, Agolia R, Arts MP, Groenewegen HJ, Zahm DS (1992). Morphological differences between projection neurons of the core and shell in the nucleus accumbens of the rat. Neuroscience 50: 149–162. [DOI] [PubMed] [Google Scholar]
- Moorman DE, James MH, McGlinchey EM, Aston-Jones G (2015). Differential roles of medial prefrontal subregions in the regulation of drug seeking. Brain Res 1628: 130–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima T, Wang RS, Katakura Y, Kishi R, Elovaara E, Park SS et al (1992). Sex-, age- and pregnancy-induced changes in the metabolism of toluene and trichloroethylene in rat liver in relation to the regulation of cytochrome P450IIE1 and P450IIC11 content. J Pharmacol Exp Ther 261: 869–874. [PubMed] [Google Scholar]
- Nimitvilai S, You C, Arora DS, McElvain MA, Vandegrift BJ, Brodie MS et al (2016). Differential Effects of Toluene and Ethanol on Dopaminergic Neurons of the Ventral Tegmental Area. Front Neurosci 10: 434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NSDUH (2013) Substance Abuse and Mental Health Services Administration (SAMHSA). 2013 National Survey on Drug Use and Health (NSDUH). [Google Scholar]
- O'Donnell P, Grace AA (1993). Physiological and morphological properties of accumbens core and shell neurons recorded in vitro. Synapse 13: 135–160. [DOI] [PubMed] [Google Scholar]
- Otto T, Eichenbaum H (1992). Complementary roles of the orbital prefrontal cortex and the perirhinal-entorhinal cortices in an odor-guided delayed-nonmatching-to-sample task. Behav Neurosci 106: 762–775. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson CThe Rat Brain In Stereotaxic Coordinates, The New Coronal Set, Fifth Edition. Academic Press: New York, 2005. [Google Scholar]
- Peters J, Kalivas PW, Quirk GJ (2009). Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem 16: 279–288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riegel AC, Zapata A, Shippenberg TS, French ED (2007). The abused inhalant toluene increases dopamine release in the nucleus accumbens by directly stimulating ventral tegmental area neurons. Neuropsychopharmacology 32: 1558–1569. [DOI] [PubMed] [Google Scholar]
- Rocha A, Kalivas PW (2010). Role of the prefrontal cortex and nucleus accumbens in reinstating methamphetamine seeking. Eur J Neurosci 31: 903–909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg NL, Grigsby J, Dreisbach J, Busenbark D, Grigsby P (2002). Neuropsychologic impairment and MRI abnormalities associated with chronic solvent abuse. J Toxicol Clin Toxicol 40: 21–34. [DOI] [PubMed] [Google Scholar]
- Sah P, Faber ES (2002). Channels underlying neuronal calcium-activated potassium currents. Prog Neurobiol 66: 345–353. [DOI] [PubMed] [Google Scholar]
- Samuel-Herter SR, Slaght SL, McKay BE (2014). Age-dependent time courses of recovery for motor functions following acute toluene intoxication in rats. Dev Psychobiol 56: 657–673. [DOI] [PubMed] [Google Scholar]
- Schoenbaum G, Eichenbaum H (1995). Information coding in the rodent prefrontal cortex. I. Single-neuron activity in orbitofrontal cortex compared with that in pyriform cortex. J Neurophysiol 74: 733–750. [DOI] [PubMed] [Google Scholar]
- Schramm-Sapyta NL, Walker QD, Caster JM, Levin ED, Kuhn CM (2009). Are adolescents more vulnerable to drug addiction than adults? Evidence from animal models. Psychopharmacology (Berl) 206: 1–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spear LP (2000). The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24: 417–463. [DOI] [PubMed] [Google Scholar]
- Weiss B, Wood RW, Macys DA (1979). Behavioral toxicology of carbon disulfide and toluene. Environ Health Perspect 30: 39–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willcocks AL, McNally GP (2013). The role of medial prefrontal cortex in extinction and reinstatement of alcohol-seeking in rats. Eur J Neurosci 37: 259–268. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.