Abstract
A very large number of disorders affect the orbit, and many of these occur in the setting of systemic disease. This lecture covers selected aspects of orbital diseases with systemic associations in which the author has a particular clinical or research interest. Spontaneous orbital haemorrhage often occurs in the presence of bleeding diatheses. Thrombosis of orbital veins and ischaemic necrosis of orbital and ocular adnexal tissues occur with thrombophilic disorders, vasculitis, and certain bacterial and fungal infections. Non-infectious orbital inflammation commonly occurs with specific inflammatory diseases, including Graves’ disease, IgG4-related disease, sarcoidosis, Sjögren’s syndrome and granulomatosis with polyangiitis, all of which have systemic manifestations. IgG4-related ophthalmic disease is commoner than all these except Graves’ orbitopathy. Some of these orbital inflammatory diseases are associated with an increased risk of B-cell lymphoma, usually marginal zone lymphoma of MALT type. Ocular adnexal lymphoma also has an association with infectious agents including Helicobacter pylori and Chlamydia psittaci. Orbital metastasis may be the first presentation of systemic malignancy. A number of orbital neoplasms occur in the setting of familial cancer syndromes, including Neurofibromatosis types 1 and 2. Study of the genetics and molecular biology of orbital diseases such as Graves’ orbitopathy and idiopathic orbital inflammatory disease will yield useful information on their diagnosis and management.
Introduction
The orbit is said to have a greater variety of disease than any other region in the body, and many of these diseases have systemic associations. I will not cover these in encyclopaedic detail, but will instead focus on aspects of orbital disease with systemic associations that have been of particular clinical and research interest to me.
Orbital vascular disease
Two generations ago, Duke-Elder’s multi-volume textbook was on every ophthalmologist’s bookshelf. Volume 13, part 2 of this seminal work has a section on spontaneous orbital haemorrhage.1 Under haemorrhagic diatheses, it states ‘the most important are haemophilia and scurvy’. I doubt any of you see cases of either disease now. But scurvy is an incredibly important disease, which profoundly influenced world history. More sailors died of scurvy in the age of discovery than died in direct maritime conflict.
In 1747, James Lind, father of the clinical trial, studied 12 sailors with advanced scurvy, divided into 6 groups of 2. They had the same navy diet, with various supplements. The two given oranges and lime recovered so quickly that they helped nurse the others. Translation of this research into practice sadly took several decades.
Sir Thomas Barlow, physician to 3 monarchs, characterized infantile scurvy in the 1880s, a disease that thereafter bore his name.2 In the latter part of the 19th century, scurvy had again become common in infants in developed countries like the US as parents began feeding their babies with proprietary infant foods and heated milk products, devoid of Vitamin C.3
One relatively common manifestation of infantile scurvy was acute proptosis. Some infants died, and one was reported to have an extensive superior subperiosteal haemorrhage as the cause of its extreme proptosis.4
If you think scurvy is a thing of the past then think again. A 2008 survey of low-income UK residents found 25% of men and 16% of women in the deficient range, and a further 20% in the depleted range.5 Morbidly obese patients have a similarly high rate of deficiency and depletion with implications for bleeding and wound healing.6
Frank scurvy is occasionally still encountered. Sloan et al recently reported a young child with bleeding gums and bilateral subperiosteal orbital haemorrhages, cured with Vitamin C infusion. Iron overload from multiple blood transfusions for thalassemia had interfered with Vitamin C absorption.7 Much of what we do as diagnosticians is recognize patterns of disease. In this case bleeding gums equaled scurvy.
Non-traumatic subperiosteal haemorrhage that may occur in scurvy has a readily recognizable pattern on imaging, with rounded opacities against the orbital roof, unilateral or bilateral, which on sagittal imaging have a lenticular shape, sometimes with visible fluid levels within them.
Non-traumatic subperiosteal haemorrhage (NTSOH) occurs in several settings: a sudden rise in cranial venous pressure as in vomiting or strangulation; paranasal sinus infection, and with bleeding diatheses.8 There is the sudden onset of proptosis, hypoglobus, vertical diplopia and the characteristic imaging appearance.
Of most relevance to this lecture are those patients with NTSOH occurring with bleeding diatheses, of which scurvy is one.
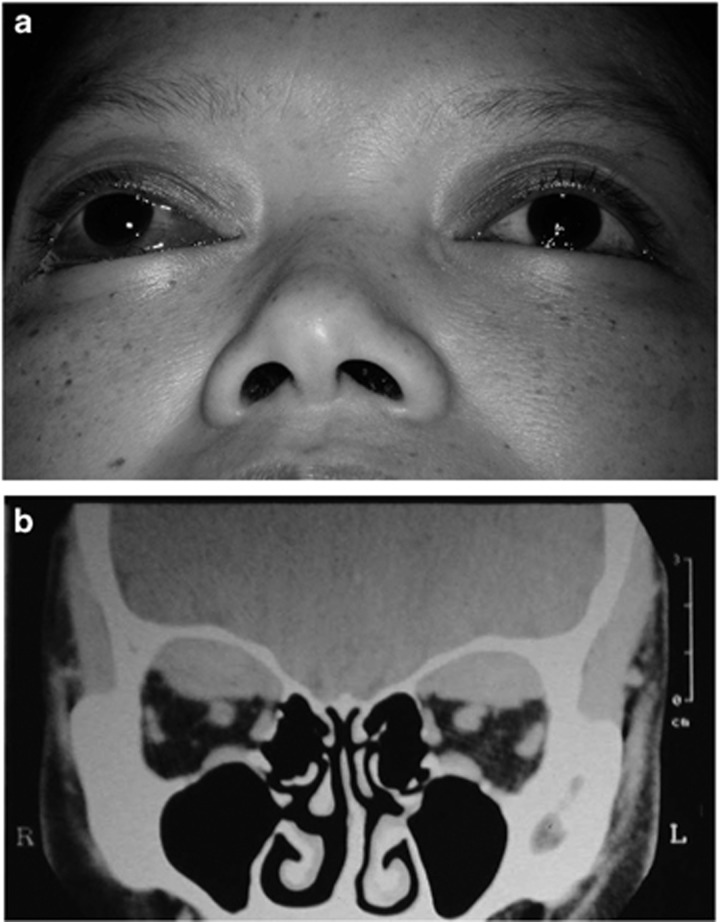
Case: Here is a tragic example. A 36-year-old mail-order bride from the Philippines, and abandoned in pregnancy by her Australian husband, suffered a massive post-partum haemorrhage, requiring a 30-unit blood transfusion and emergency hysterectomy. She developed disseminated intravascular coagulation and was intubated in ITU for 4 days. With no family or visitors, her bilateral proptosis went unnoticed and on extubation said ‘I can’t see’. She had no light perception on the right and visual acuity of 6/60 on the left with a small island of central vision and bilateral superior subperiosteal collections of blood. (Figure 1) There was minimal improvement in vision after surgical drainage.
Figure 1.
(a) A 36-year old woman with visual loss from bilateral proptosis due to bilateral superior subperiosteal haemorrhage complicating massive post-partum haemorrhage and disseminated intravascular coagulation. (b) A coronal CT scan showed bilateral superior subperiosteal haemorrhages.
A variety of bleeding disorders has predisposed to this entity, the commonest being liver disease, followed by scurvy. (Table 1) They tend to be younger adult patients, the bleed is always superior, and is often bilateral. Many have poor vision at presentation, some require surgical intervention, and a few do not recover vision, unlike all the other patients with a different cause for their haemorrhage, in whom visual recovery is the norm.9
Table 1. Aetiology of 24 published cases of non-traumatic subperiosteal orbital haemorrhage occurring with bleeding diatheses.
| Liver disease | 7 |
| Scurvy | 6 |
| Anticoagulation | 4 |
| DIC, post-partum haemorrhage | 2 |
| Thrombocytopaenia | 1 |
| Henoch-Schönlein purpura | 1 |
| Von Willebrand disease | 1 |
| Christmas disease | 1 |
| Chronic myeloid leukaemia | 1 |
Abbreviation: DIC, disseminated intravascular coagulation.
Rare cases of intracranial extradural haemorrhage have been recorded in association with this entity.10
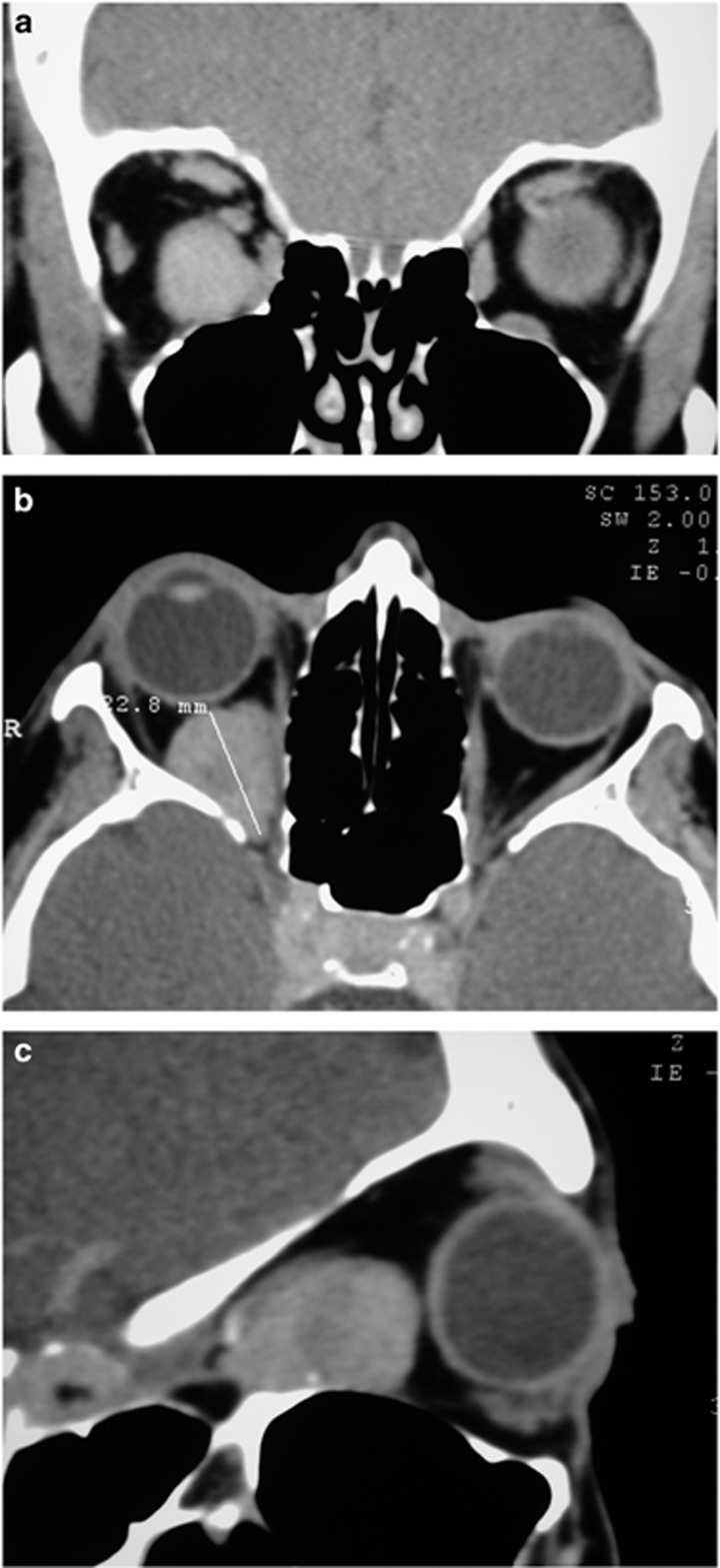
Another group of non-traumatic orbital haemorrhage is that occurring in relation to extra-ocular muscles.11, 12 Again, there is a pattern to be recognized. These haemorrhages always occur in the inferior orbit, sometimes with fluid levels within them, and the bleed is well circumscribed and rounded anteriorly, with tapering towards the orbital apex (Figure 2).
Figure 2.
(a) A coronal CT scan of the orbits with a well-circumscribed spontaneous orbital haemorrhage related to the inferior rectus muscle sheath. (b) An axial image shows the lesion with a rounded anterior surface. (c) On a sagittal image the lesion tapers towards the orbital apex.
Patients present with painful proptosis, diplopia, sometimes nausea and vomiting, usually on waking. Most are elderly, and importantly all resolve spontaneously. There is no underlying lesion responsible. The median age is the late 60 s, and about a quarter have decreased vision at presentation. Unlike superior subperiosteal bleeding, a minority of these patients have identifiable risk factors which include antiplatelet agents. Only a small number require surgical drainage and all recovered fully.9
One other rare entity associated with spontaneous bleeding is amyloidosis. The amyloid protein infiltrates blood vessels and orbital and adnexal amyloidosis may present with bleeding.13 Rare patients may present with spontaneous eyelid haemorrhages as well as ophthalmoplegia,14 and the systemic amyloidosis may be associated with multiple myeloma.15
The opposite of bleeding is thrombosis and ischaemic necrosis. Thrombosis of the superior ophthalmic vein usually occurs in association with cavernous sinus thrombosis and other dural venous sinus thromboses.
Case: A 44-year-old man presented with 3 weeks of worsening left headache, and a day of left eye swelling, reduced vision, and painful eye movements. There was some neck pain and photophobia, fever and weight loss. There was no facial rash or other skin lesions. He was a heavy smoker and drinker.
He was afebrile, alert with slightly reduced left vision, swollen left eyelids and reduced left eye movements. He had negative blood cultures, a normal lumbar puncture but a CRP of 279.
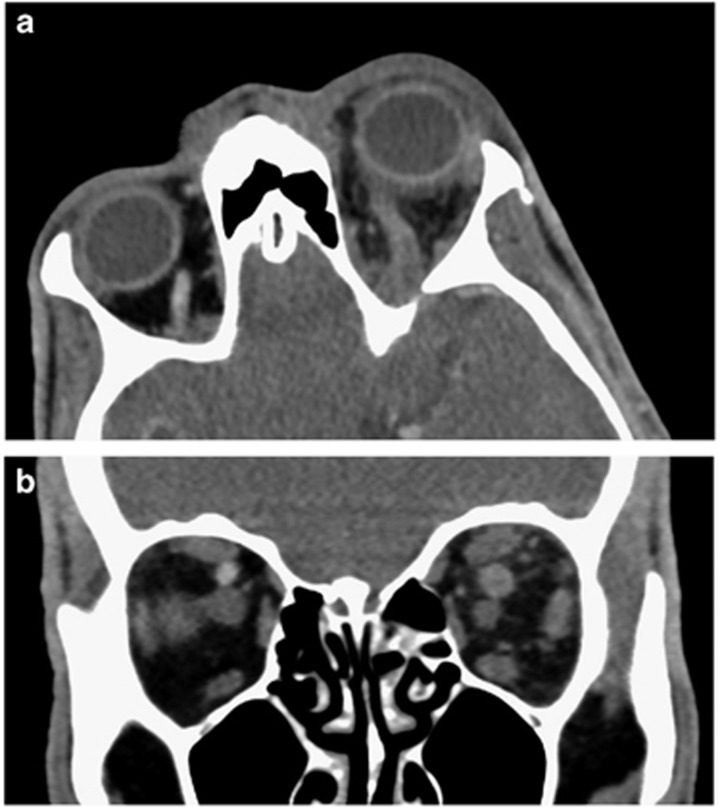
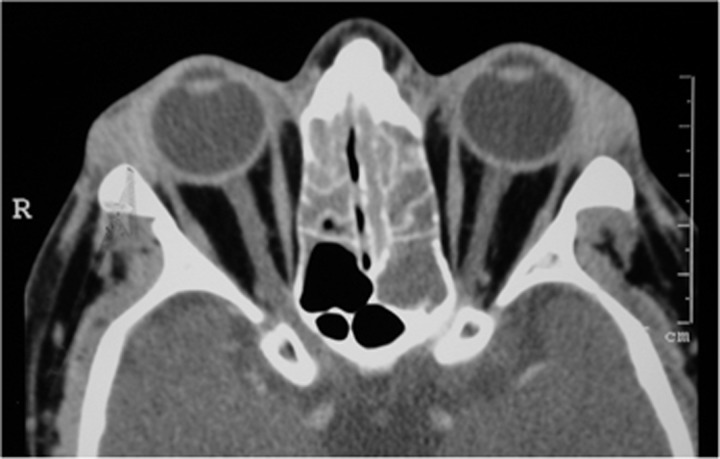
A CT scan with contrast showed a dilated left superior ophthalmic vein, which did not enhance as much as the normal contralateral vein (Figure 3). We thought he had a thrombosed superior ophthalmic vein and arranged an urgent MRI. This revealed thrombosis not only in the superior ophthalmic vein but also in both sides of the cavernous sinus (Figure 4) extending as far as the jugular bulb on the right and involving the inferior petrosal sinus.
Figure 3.
(a) An axial CT scan with contrast of a patient with thrombosis of the left superior ophthalmic vein (SOV), which is enlarged and shows less contrast enhancement than the normal contralateral SOV. (b) A contrast-enhanced coronal CT scan of the orbits demonstrates a larger left SOV with less contrast enhancement.
Figure 4.
(a) An axial T1-weighted orbital MRI with contrast shows a filling defect in the left superior ophthalmic vein due to thrombosis. (b) The thrombosis involves both sides of the cavernous sinus (arrows).
A thrombophilia screen was negative, and he was anticoagulated. Further investigation led to the discovery of a lung lesion, which was an early carcinoma, and the presumed cause of his thrombophilia.
Superior ophthalmic vein thrombosis usually occurs in association with cavernous sinus thrombosis and beyond as in this man. Risk factors include prothrombotic disorders, infection, and malignancy, but may also occur as part of the natural history of carotid cavernous sinus fistulae.
Thrombosis of major venous tributaries does not lead to tissue necrosis but other conditions can. These include various forms of vasculitis, and infective agents which are angiotropic or whose exotoxins lead to small vessel closure (Table 2).
Table 2. Causes of orbital and ocular adnexal tissue necrosis.
| Vasculitis |
| Granulomatosis with polyangiitis |
| Giant cell arteritis |
| Herpes zoster ophthalmicus |
| Infection |
| Mucormycosis |
| Group A beta-haemolytic Streptococcus |
| Pseudomonus aeruginosa |
| Pyoderma gangrenosum |
Ischaemic necrosis in granulomatosis with polyangiitis (GPA or Wegener’s granulomatosis) occurs over a long time frame (Figure 5). Rarely giant cell arteritis can lead to visible tissue necrosis, as may herpes zoster.
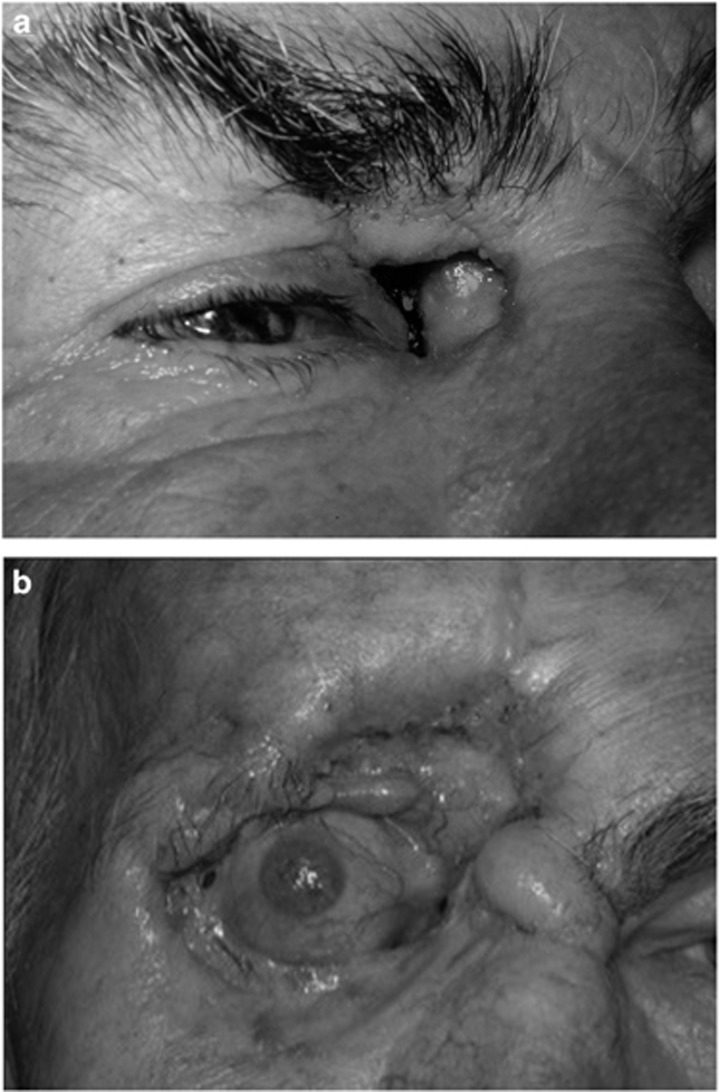
Figure 5.
(a) A man with the ‘limited’ form of granulomatosis with polyangiitis (GPA) presented with sinus and orbital disease and a chronic fistula from the skin to the frontal sinus. (b) Nine years later, the area of tissue necrosis had enlarged despite immunosuppression.
The fungal infection, mucormycosis may lead to eschar formation (tissue necrosis), usually as a late clinical sign. Infection with group A beta-hemolytic streptococci can lead to necrotizing fasciitis which can sometimes involve retroseptal structures fulminantly, and some patients with pseudomonas infection can develop tissue necrosis. Pyoderma gangrenosum, a neutrophilic dermatosis, can lead to tissue loss, and may be associated with diabetes, rheumatoid arthritis and inflammatory bowel disease.
Mucormycosis
Case: An 82-year-old man with known myelodysplasia and anaemia, but normal white cells, had required regular blood transfusions. He was on 25 mg prednisolone, and had type 2 diabetes, well-controlled with oral hypoglycaemics. He presented with 3 days of malaise, complete right ptosis, lid swelling, and complete loss of right vision with a normal fundus. There was complete internal and external ophthalmoplegia, and right proptosis and chemosis (Figure 6). His CT scan showed proptosis and patchy orbital fat changes, and some minor changes in the sphenoid and maxillary sinuses (Figure 7).
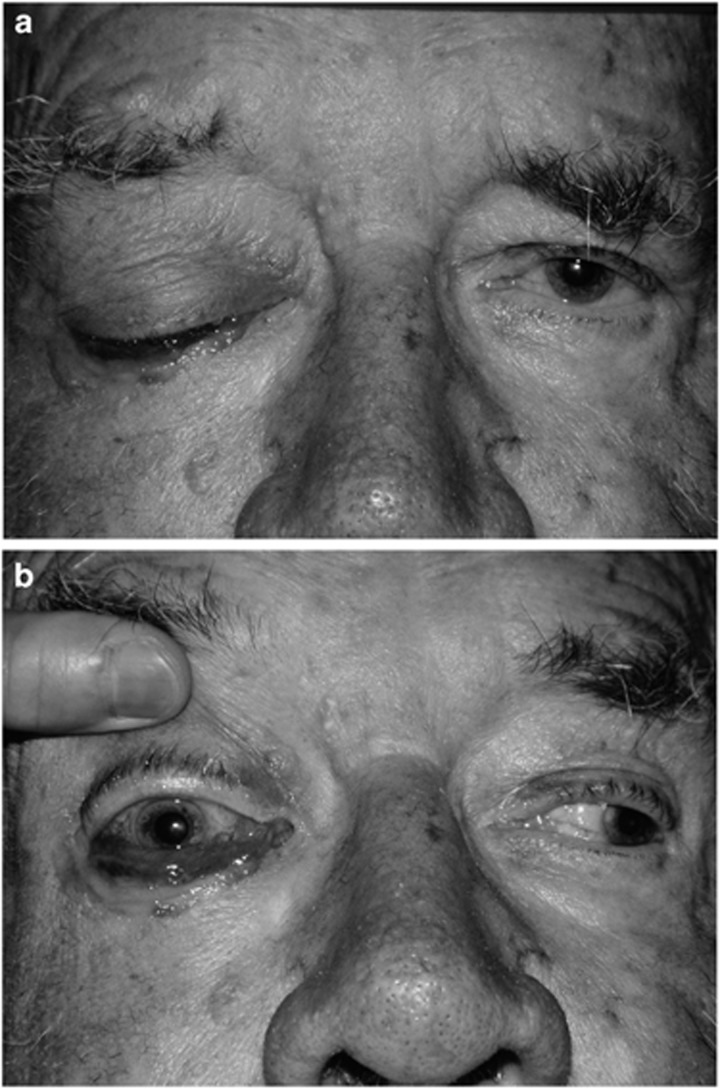
Figure 6.
(a) An 82-year-old man with myelodysplasia, on 25 mg prednisolone, presented with 3 days of malaise, a blind right eye, and (b) complete external and internal ophthalmoplegia.
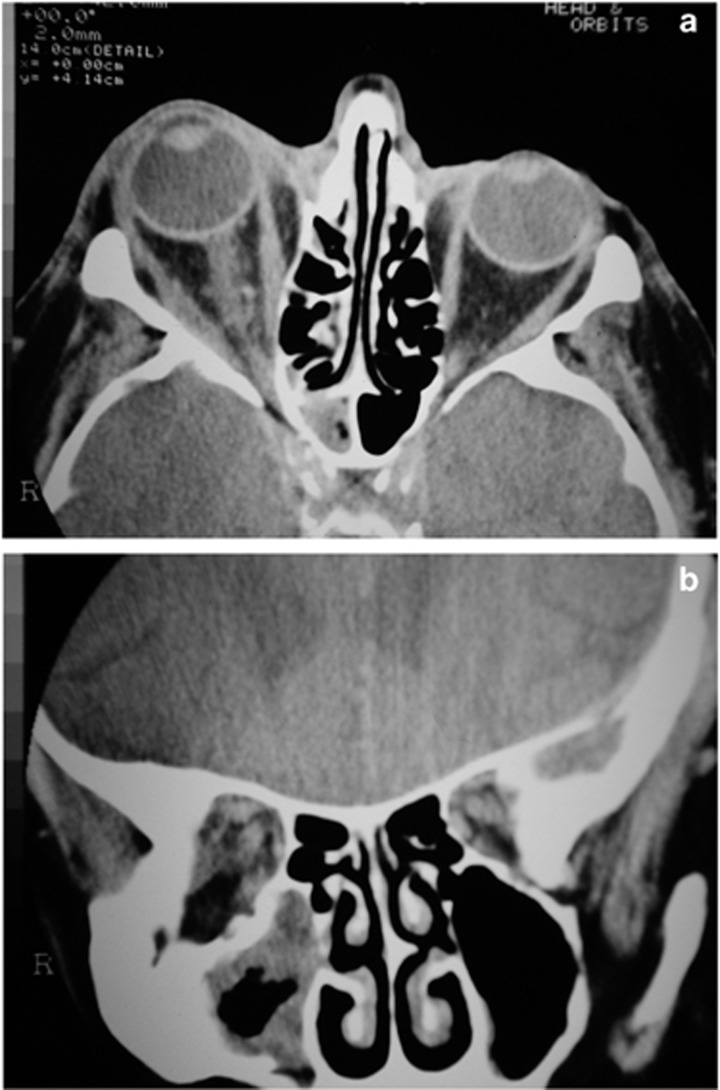
Figure 7.
(a) An axial CT scan (of the patient in Figure 6) showed proptosis, patchy orbital tissue change and (b) mild adjacent sinus disease. A biopsy of the sinuses confirmed mucormycosis.
We were concerned about possible mucormycosis and arranged urgent sinus biopsies which showed non-septate hyphae. He had immediate orbital exenteration and fronto-ethmoidectomy and mucormycosis was found involving the orbital apex tissues. IV amphotericin was started.
On day 5, he suffered a right middle cerebral artery stroke, and the next day suddenly collapsed and died. At post mortem he had ruptured a mycotic internal carotid aneurysm, but to our surprise was found to have evidence of haemochromatosis affecting liver and pancreas. Abnormal iron storage had preceded his blood transfusions.
Mucormycosis has well known risk factors: diabetes with or without ketoacidosis, renal disease or transplantation with immunosuppression, or immunosuppression for haematological malignancies, and iron chelating agents for iron overload in dialysis patients.16
Interestingly, both ketoacidosis and iron chelating agents probably alter iron availability for this organism, and our patient’s haemochromatosis was probably a factor in the development of his mucormycosis.17
Non-infective orbital inflammation
Non-infective orbital inflammation is traditionally divided into specific causes and idiopathic orbital inflammation, once known as ‘pseudotumour’. By far the commonest type is thyroid eye disease or Graves’ orbitopathy (GO).
GO of course is very strongly associated with thyroid gland dysfunction, usually Graves’ thyrotoxicosis. Many orbitopathy patients present before or after the onset of hyperthyroidism. Five to 10% are euthyroid, but half of these become hyperthyroid within 18 months. A small proportion of orbitopathy patients will have primary hypothyroidism. Looked at the other way, 30–50% of Graves’ disease patients develop clinical signs of orbitopathy, and as many as 60–90% when assessed by CT.
There are other extra-thyroid manifestations of Graves’ disease apart from the orbitopathy. Many patients complain about the swelling in the periorbital region above and below their orbital rims. This has been well demonstrated to be due to tissue changes in the retro- and sub-orbicularis oculi fat, so it is not just the orbital contents affected by this disease.18 Soft tissue changes in the glabellar region with deepened glabellar folds are also common. Some patients feel that their whole face becomes swollen by the disease.
Pre-tibial myxedema can be a particularly troublesome and difficult to treat manifestation. It occurs in only 4% of patients with Graves’ orbitopathy, and more commonly when the orbitopathy is severe. A fifth of patients with pre-tibial changes will have acropachy as well. Thyroid acropachy, in which finger clubbing is the commonest change, is rarer still.19
Those of us who treat Graves’ orbitopathy patients know how common mental health can be affected. This is more than just the difficulty of adjusting to profound changes in one’s facial appearance. There is a higher incidence of anxiety and major depression in orbitopathy patients,20 and interestingly, a link between levels of TSH receptor antibody and depression scores has been shown.21
Other auto-immune disorders such as vitiligo and alopecia may occur in association with Graves’ disease, yet another burden for these patients to bear.
Orbital myositis
The non-specific or idiopathic orbital inflammations can be classified by rate of onset, type of inflammation or anatomical location. Orbital myositis is a well-recognized sub-group. Orbital myositis affects more women, usually as young adults, with typically an acute onset of unilateral painful eye movement, a single muscle affected and a rapid response to oral steroids. The medial, superior and lateral recti are most commonly affected, and the affected muscle usually shows fusiform enlargement with enhancement, often but not always with tendon involvement. Recurrence is relatively common.22, 23
Orbital myositis may be associated with systemic disease, typically autoimmune processes, and other inflammatory and infective disease. It is rarely seen as a paraneoplastic process, may be drug-related or related to giant cell myocarditis.
There is a long list of specific inflammatory processes that have been associated with orbital myositis (Table 3). After Graves’ orbitopathy, inflammatory bowel disease is one of the commoner ones.
Table 3. Autoimmune and inflammatory diseases associated with orbital myositis.
| Graves’ orbitopathy |
| IgG4-related disease |
| Crohn’s disease/ulcerative colitis |
| Rheumatoid arthritis |
| Psoriatic arthritis |
| Systemic lupus erythematosis |
| Undifferentiated CT disease |
| NOD-2 autoimmune disease |
| Celiac disease |
| Churg Strauss syndrome |
| Behçet’s disease |
| Lyme disease |
| Whipple’s disease |
| Herpes zoster |
Abbreviation: NOD-2, nucleotide-binding oligomerization domain containing 2.
Extraocular muscle enlargement and dysfunction has also been described as a paraneoplastic phenomenon in a number of malignancies, including breast cancer, lung cancer and seminoma, with multiple muscles usually involved.
A number of drugs have been reported to cause myositis. Statins are well-known to cause a widespread myopathy, but have also been reported to cause orbital myositis.24
In patients with inflammatory bowel disease and orbital myositis, most have multiple muscles involved bilaterally. In some, the myositis precedes the bowel symptoms by many months, is often recurrent and resistant to steroids. However, in other aspects the myositis associated with inflammatory bowel disease mirrors the typical idiopathic single muscle form, with very similar demographics.
One particularly important form of orbital myositis is that associated with giant cell myocarditis, which of itself is rare, with heart failure and arrhythmias common, and an 89% rate of death or cardiac transplantation.25 There is a strikingly similar list of autoimmune diseases associated with giant-cell myocarditis, again the commonest being inflammatory bowel disease.
Of the patients with orbital myositis and giant cell myocarditis, most again have bilateral disease with multiple affected muscles. Cardiac symptoms usually occur within 1-2 months of the myositis. Half died and another 2 needed heart transplants.
Case: With all this in mind here is an interesting recent case of a 33-year-old woman with a week of painful bilateral eye swelling. She had a known axillary angiosarcoma, now beyond resection, and was on no medications. She had marked chemosis and lid swelling, but normal vision, with near complete ophthalmoplegia on the right and less severe reduced ductions on the left. Imaging showed multiple affected muscles with tendon involvement (Figure 8).
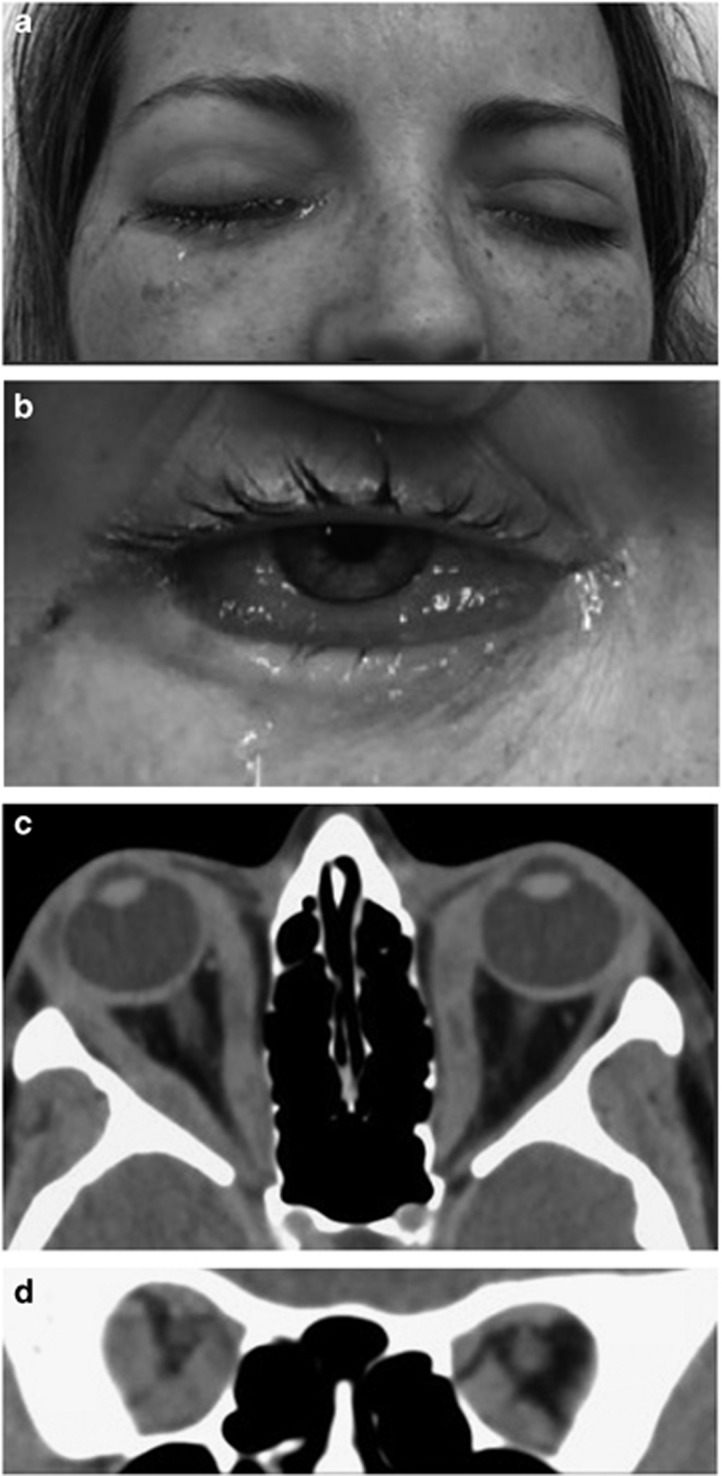
Figure 8.
(a) A 33-year-old woman with a known axillary angiosarcoma presented with 1 week of bilateral painful eye swelling with ophthalmoplegia and (b) conjunctival chemosis. (c) An axial and (d) coronal CT scan showed multiple extraocular muscles enlarged, including their tendinous insertions. A muscle biopsy found granulomatous orbital myositis.
Blood tests were normal apart from an elevated CRP. A PET scan showed her axillary sarcoma and there was intense uptake in the extraocular muscles. A lateral rectus muscle biopsy showed granulomatous inflammation. She responded well to intravenous corticosteroids. As yet we do not know whether this is a paraneoplastic phenomenon or whether she is at risk of developing myocarditis. An important message is to beware in patients with atypical myositis, and have a low threshold for biopsy.
Specific forms of orbital inflammation
Sarcoidosis.
Ocular adnexal sarcoidosis often presents with bilateral lacrimal gland inflammation and enlargement, often with concurrent lung and mediastinal disease. However, the presentation may be more complex.
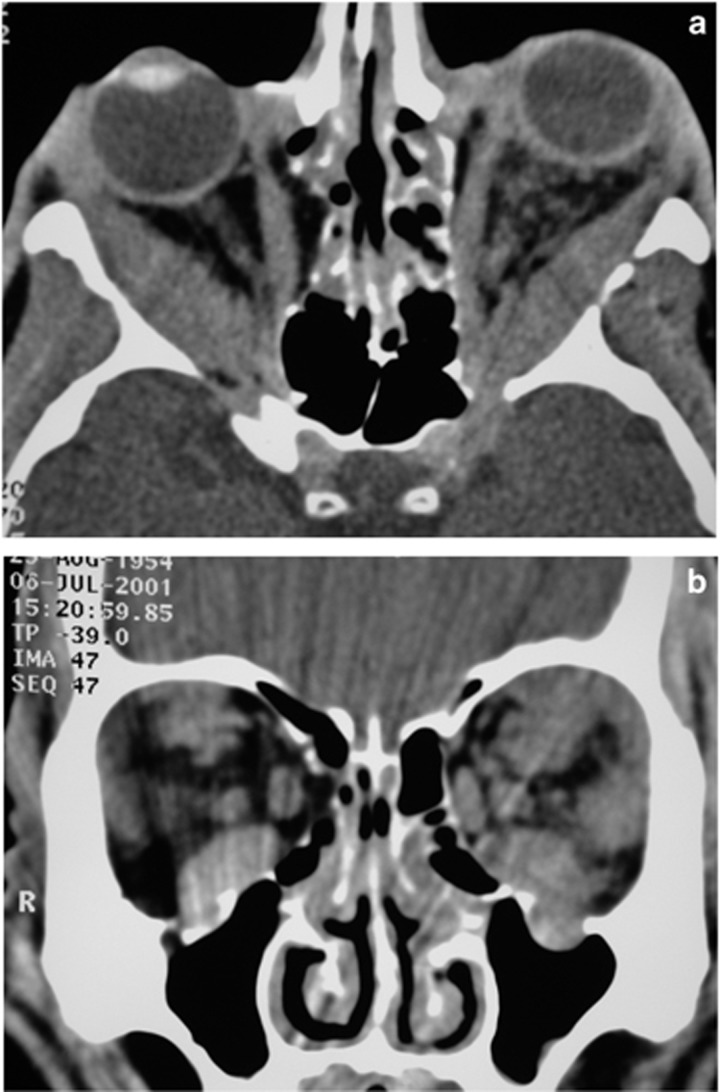
Case: A 40-year-old man from India, had a year of bilateral progressive lacrimal gland enlargement and marked nasal obstruction (Figure 9). A year earlier, in India, he had epididymitis biopsied showing granulomatous inflammation associated with hilar lymphadenopathy on chest X-ray. In India that was assumed to be tuberculosis and he was put on quadruple therapy without improvement.
Figure 9.
An axial CT scan of a man with bilateral lacrimal gland enlargement and rhinosinusitis due to sarcoidosis.
Biopsy of his lacrimal gland showed non-caseating granulomatous inflammation, and tissue was negative for acid-fast bacilli and grew nothing on culture. His anti-TB therapy was ceased and he was successfully treated with steroids and methotrexate.
Rhinosinusitis may occur with sarcoidosis and many of these patients also develop nasolacrimal duct obstruction, which may require surgery.26
Another aspect of sarcoidosis encountered in orbital practice is neuro-sarcoid with optic nerve involvement.
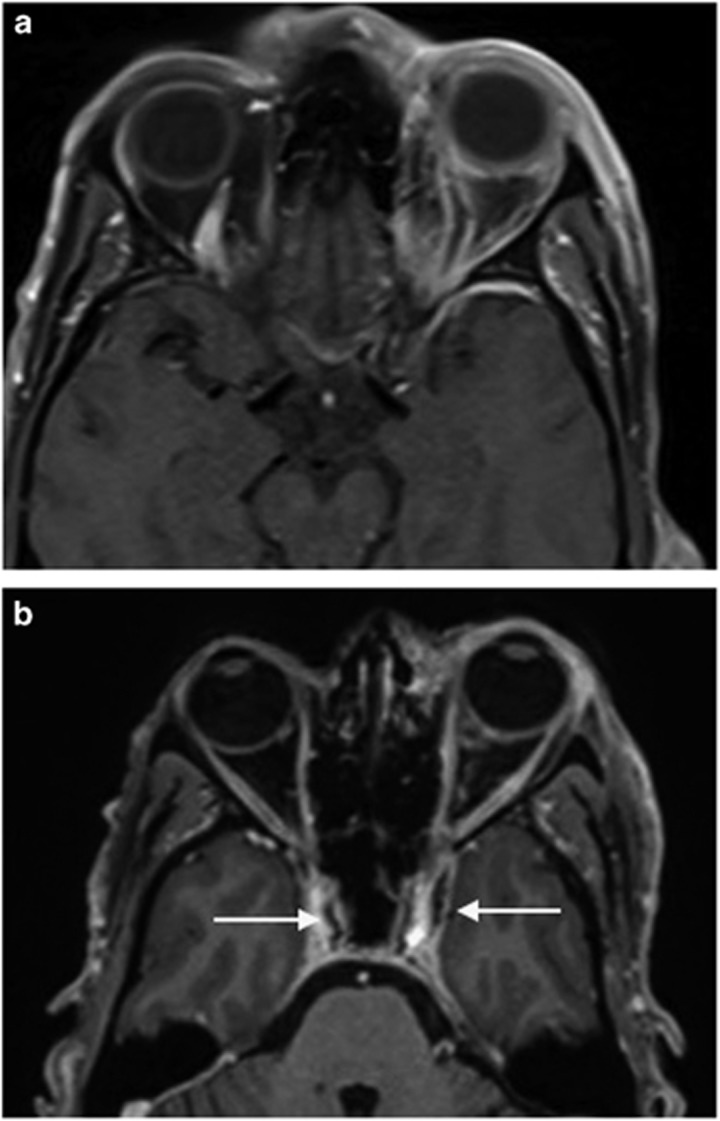
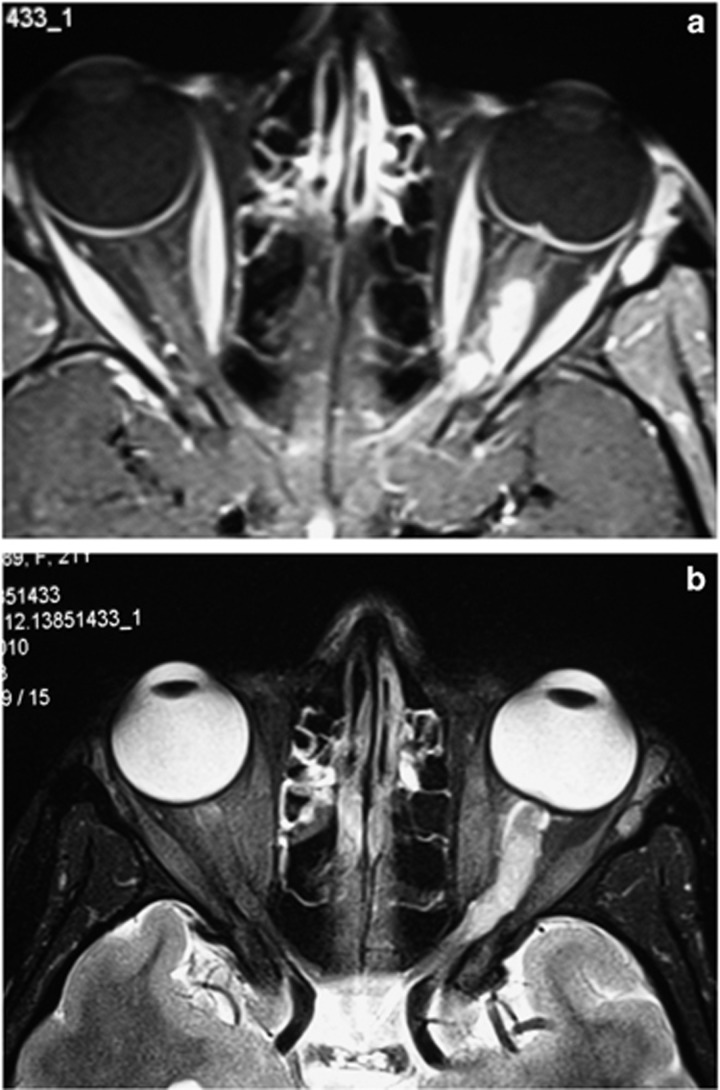
Case: A 21-year-old female presented with some months of failing left vision, to hand motions with a swollen disc, an afferent defect and mild proptosis. Changes on her scans were odd, with the suggestion of both nerve sheath and intrinsic optic nerve involvement (Figure 10).
Figure 10.
(a) A T1-weighted orbital MRI with contrast shows optic nerve disease and optic disc swelling. (b) A T2-weighted orbital MRI shows optic nerve disease. There appeared to be both optic nerve sheath and intrinsic optic nerve disease which on biopsy was found to be due to neuro-sarcoid.
We performed an optic nerve sheath and small optic nerve intrinsic biopsy and identified non-caseating granulomatous inflammation consistent with neuro-sarcoid.
In cases where an optic neuropathy is otherwise difficult to diagnose, consideration can be given to direct optic nerve biopsy.27
Sarcoidosis much more commonly causes uveitis than orbital disease, and oddly the two rarely occur together. Most have lacrimal gland disease, or anterior inferior orbital disease. Half will have known systemic disease at presentation, and systemic disease may develop some years later.28, 29, 30
IgG4-related disease.
Another inflammatory cause of lacrimal gland enlargement is IgG4-related disease.
Case: A 48-year-old woman had 6 months of painless puffy upper eyelids and reduced exercise tolerance and nasal stuffiness. There was oedema of the upper lids and lateral conjunctiva with palpably enlarged lacrimal and submandibular glands. Scans showed enlarged lacrimal glands with slightly hazy borders and significant rhinosinusitis.
Blood tests showed an elevated CRP and marked elevation of serum IgG4. Lacrimal gland biopsy showed an inflammatory process with both B and T lymphocytes, but also plasma cells and eosinophils, and some storiform fibrosis. Stains for IgG and IgG4 showed large numbers of IgG4+ plasma cells in a ratio well over 40% compared to the IgG+ plasma cells.
A PET CT scan showed uptake in the lacrimal glands but also strangely in the extraocular muscles, which had been normal on MRI, as well as the submandibular glands, and within the tracheo-bronchial tree. A diagnosis of IgG4-related disease was made, with involvement of the lacrimal and submandibular glands, and the upper and lower airways.
Until recently, a list of the commoner forms of specific orbital inflammation would have included Graves’ orbitopathy, sarcoidosis, Sjögren’s syndrome, granulomatosis with polyangiitis (GPA, or Wegener’s granulomatosis), and xanthogranulomatous disease amongst the more common types. We now have to add IgG4-related disease, which has reduced the proportion of patients with idiopathic orbital inflammation. In terms of relative incidence, IgG4-related disease should be second only to Graves’ orbitopathy.
IgG4-related disease (IgG4-RD) is a disease first hinted at in 2001 when elevated levels of serum IgG4 were noted in patients with autoimmune pancreatitis.31 By 2003 it had expanded to a systemic autoimmune disease,32 and by 2007 sclerosing dacryoadenitis was recognized as a manifestation.33, 34
Commonly involved organs are pancreas, hepatobiliary tract, salivary glands, lymph nodes and retroperitoneum, with the orbit high up this list, but almost any organ can be involved.
The diagnosis partly depends on finding significant numbers of IgG4+ plasma cells within a dense lymphoplasmacytic infliltrate with fibrosis, and in larger organs obliterative phlebitis, and often, large numbers of eosinophils. The numbers of IgG4+ plasma cells is defined, and more importantly their ratio to IgG+ plasma cells should be over 40%.
A consensus statement on the pathological features has been published,35 as well as combined diagnostic criteria that includes clinical features and serum IgG4 levels, but uses a cut-off of 10 IgG4+ cells per high power field.36 Confusingly, diagnostic criteria for IgG4-related ophthalmic disease have set the level of IgG4+ cells at over 50 per high power field.37
But it is important to recognize that high numbers of IgG4+ plasma cells do not on their own make a diagnosis of IgG4-RD. They can occur in many other diseases including GPA or Wegener’s granulomatosis, and xanthogranulomatous disease.38 And the histology should be appropriate with no necrosis, and no granulomas, giant cells or neutrophils. And the cutoffs for numbers of IgG4+ cells are not absolute. Lower numbers may be accepted in the presence of extensive fibrosis or the appropriate clinical features.
So the diagnosis depends on integrating histopathology with the clinical and serological features.
Just how common is this disease? In Japan, it represents a fifth of all orbital lymphoproliferative disease, and is commoner than other non-thyroid forms of inflammation.39 Retrospective studies of orbital biopsies from the USA40 and France41 diagnosed as idiopathic inflammation or reactive lymphoid hyperplasia showed 40–50% may have had IgG4-related disease.
Orbital IgG4-RD can follow several patterns. Lacrimal gland disease is commonest. Extraocular muscle disease with trigeminal nerve enlargement is another pattern, as is fibrosing inflammation.42
IgG4-related dacryoadenitis is typically a chronic sclerosing type, with bilateral disease being commoner.
Case: Here is a typical case with lacrimal gland disease. A 58-year-old man had presented in 2011 with a big left submandibular gland, removed and labeled as Mickulicz disease. The same occurred in the other submandibular gland. He had a history of sinus disease needing surgery, mild asthma, and more recent swelling of his lacrimal glands and parotids. In days gone by this would have been called Mickulicz disease.
His serum IgG4 was markedly elevated and a biopsy of his lacrimal gland showed features typical of IgG4-RD, confirmed on review of the pathology of his submandibular glands.
IgG4-RD has a very distinctive tendency to involve the infra-orbital nerve, and sometimes other trigeminal nerve branches. If present, it is usually seen with enlarged extraocular muscles, lacrimal glands and paranasal sinus disease and eosinophilia.43
Curiously even in the absence of clinical orbital disease, patients with autoimmune pancreatitis have larger than normal infra-orbital nerves,44 and large nerves are statistically associated with IgG4-related ophthalmic disease.45
Case: A 51-year-old man serves as an illustrative case. Way back in 1991 he presented with an enlarged left lacrimal gland, lymphadenopathy, asthma, and chronic sinusitis, diagnosed on lacrimal gland biopsy as reactive lymphoid hyperplasia, responding to steroids.
Much later in 2008 he returned with bilateral proptosis and lesions centred on the lacrimal glands. Repeat biopsy found features of IgG4-related disease. His scans showed marked enlargement of the infra-orbital nerves and sinus disease.
As just described, IgG4-RD may evolve from one pattern to another over time.
Case: A 37-year-old man, again first seen in 1991 presented with bilateral lacrimal and submandibular gland disease diagnosed as reactive lymphoid hyperplasia on biopsy, treated with steroids and lost to follow-up. This is the previously called Mickulicz disease pattern.
He came back in 2001, now with several years of increasing proptosis, and now had enlarged extra-ocular muscles and infra-orbital nerves. Biopsies again showed reactive lymphoid hyperplasia, which on review in 2007 showed features of IgG4-related disease. His scans in 2001 showed enlarged extraocular muscles and infraorbital nerves, as well as patchy infiltrative disease in the superior orbits (Figure 11).
Figure 11.
A middle-aged man had presented with bilateral lacrimal and submandibular gland enlargement diagnosed as reactive lymphoid hyperplasia. Ten years later he developed (a) bilateral proptosis with enlargement of extraocular muscles on an axial CT scan, and (b) enlargement of the infra-orbital nerves, and infiltrative disease of the superior orbits. Review of his biopsies confirmed IgG4-related disease.
How common is systemic disease in the presence of IgG4-related ophthalmic disease? Putting four east-Asian series together gives a rate of 40%,46 and we have shown that bilateral disease is statistically associated with systemic disease.47 Disease elsewhere may precede or follow the orbital disease, and systemic disease is probably best detected with PET scanning. PET scans can also show response to treatment.
To screen for systemic disease, a careful history and clinical examination is critical, and renal and liver function should be checked. Imaging modalities other than PET scan include CT and Gallium scanning.
The next case highlights the utility of PET scans in this disease.
Case: A 72-year-old woman seen first in 2004 had several years of left eye swelling, and chronic sinusitis requiring multiple operations, and on CT, had enlarged muscles, patchy infiltrate, and a big left infraorbital nerve.
Biopsy from the left orbit in 2004 diagnosed reactive lymphoid hyperplasia. Repeat biopsy in 2007 diagnosed IgG4-RD and she was started on steroids and methotrexate, with a good response to treatment.
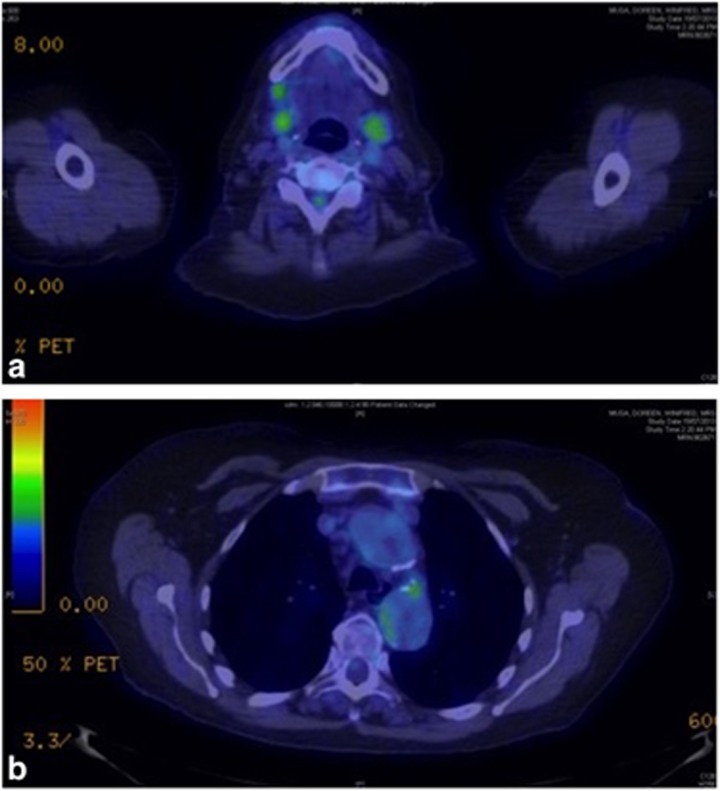
However, in 2012 she had to cease the methotrexate because of repeated infections. She then developed left ureteric obstruction, and MRI and PET scans showed retroperitoneal fibrosis with obstructive nephropathy, and also aortitis of the thoracic aorta. Her PET CT showed cervical nodal disease as well as thoracic aortic disease (Figure 12).
Figure 12.
A 72-year-old woman with IgG4-related disease developed widespread disease including (a) cervical lymphadenopathy and (b) thoracic aorta disease due to IgG4-related disease, shown on PET scanning.
A recent paper on IgG4-related large vessel disease, found a fifth of 160 patients with IgG4-related disease had large vessel disease, and as many as a third of these had aortitis with aneurysm formation.48 Thoracic disease is commoner than abdominal and in this series, 3 aneurysms dissected or had a contained rupture.
IgG4-related disease is also associated with lymphoma.
Case: A case of ours, a 52-year-old woman, presented in 2008 with bilateral lacrimal gland and cervical nodal disease with a background of biliary cirrhosis. Scans showed enlarged infraorbital nerves and a large left frontal nerve. Her serum IgG4 was markedly elevated and biopsies confirmed IgG4-related disease, which was treated with steroids and hydroxychloroquine.
Four years later she noted worsening left vision and MRI showed now a perio-optic nerve lesion on that side. Repeat biopsy now showed marginal zone lymphoma of MALT type.
In Japanese series of IgG4-related disease, about 10% develop lymphoma and in our own series, 5 of 30 cases have done so, most with MALT lymphoma.42
Chronic orbital inflammation, infection, and lymphoma
It is well recognized that other chronic inflammatory diseases increase the risk of lymphoma.
Case: A 41-year-old woman was known for 14 years to have primary Sjögren’s syndrome. 6 years after that diagnosis, her lacrimal glands enlarged and a biopsy was consistent with Sjögren’s syndrome. Five years later she developed marked increase in the right lacrimal gland swelling.
Scans curiously showed a large gland with a big cyst within the orbital lobe. An excisional biopsy showed a large cyst lined by ductal epithelium with MALT lymphoma within the wall.49
Another patient with a different chronic and relapsing inflammatory disease, relapsing polychondritis, had seen me over 10 years with episodes of orbital inflammation, enlarged muscles coming and going, but no other signs of thyroid orbitopathy. She recently presented with 3 months of marked bilateral proptosis. Bilateral infiltrative masses revealed MALT lymphoma on biopsy, presumably related to her orbital inflammation secondary to relapsing polychondritis.
Sjögren’s syndrome is the best-known example of chronic inflammation leading to lymphoma with a near 14-fold increase in the incidence of non-Hodgkin’s lymphoma.50 Even thyroid orbitopathy may predispose to lymphoma51 and as we have seen IgG4-related ophthalmic disease also can lead to lymphoma.
Of course many other inflammatory diseases are associated with cancer, and another association is gastritis due to infection with Helicobacter pylori and gastric MALT lymphoma and also gastric adenocarcinoma.
The link between infection with Helicobacter pylori and gastritis and stomach and duodenal ulceration was first made by two Australians, Barry Marshal and Robin Warren from Perth. For many years they were ridiculed for their suggestions, but were finally vindicated, winning the 2005 Nobel prize for medicine. Marshall went so far to fulfill Koch’s postulates as to himself swallow a beaker of broth full of what was then called Campylobacter pyloridis, inducing acute gastritis confirmed on biopsy.
Of ocular adnexal lymphomas, a proportion have been found to be associated with infectious agents also, including Helicobacter pylori, Chlamydia psittaci, and hepatitis C virus.
A large multicenter French study looked at the link between gastric H pylori infection and ocular adnexal lymphoma (OAL).52 Three groups—83 with OAL, 101 with extragastric and extraorbital lymphoma, and 156 controls without lymphoma, had gastric biopsies. 45% of the OAL group had H pylori infection, 25% of the other lymphoma patients had it, and only 12 of the controls. This gave a P value <.001 for OAL vs controls, and <.01 for OAL vs other lymphomas.
Chlamydia psittaci has been found in association with ocular adnexal MALT lymphoma, but there has been a huge geographic variation, from 87% in Italy to zero in other parts of Europe, North America and Japan.
In Italy, a prospective multi-centre trial showed a complete or partial response to oral doxycycline for 3 weeks in nearly a half of 27 cases of OAL of MALT type, with response occurring interestingly in both chlamydia positive and negative cases.53
A Korean study looked at a larger number treated with Doxycycline and found nearly two-thirds of 90 patients with OAL of MALT type had a 5-year progression-free survival.54
Orbital metastasis
Lets finally turn to some other orbital malignancies with systemic associations. Orbital metastasis is the obvious one. From published series55, 56 including our own,57 we know that breast, prostate and lung are commonest, but in our country, cutaneous melanoma was second only to breast, a reflection of our high melanoma incidence. Carcinoid tumour, a relatively rare lesion elsewhere, features prominently also.
Seventy to 85% of patients with orbital metastasis have a known primary tumour, often with other known metastases, but between 15 and 30% have their first presentation with orbital metastasis.55, 56, 57
The commonest patterns for orbital metastasis are diffuse anterior fat, extraocular muscle and bone involvement.55 Prostate cancer goes to orbital bone most frequently, breast more commonly to orbital fat, and melanoma and carcinoid tumour go preferentially to extraocular muscle.
Metastatic orbital breast cancer may have the classical presentation of enophthalmos, with restricted movements due to the infiltrating and sclerosing nature of some breast cancer disease. Breast cancer metastasis to a single extraocular muscle will often have both a paralytic and restrictive component to the motility disorder. Breast cancer less frequently metastasizes to bone, with a lesion centred on the lateral wing of the sphenoid sometimes mimicking a meningioma.
Prostate cancer preferentially affects bone, often with hyperostosis, again mimicking meningioma. But bone destruction from metastatic prostate cancer may also be seen. The degree of hyperostosis may be extreme and widespread with prostate cancer.
By contrast, melanoma has a predilection for extraocular muscle. In the past, survival for these patients was measured in months, but with newer monoclonal antibodies and immunotherapy, survival for years is now often seen.
Case: here is an example of an orbital metastasis being the first manifestation of disease. A 67-year-old woman came with 6 months of vertical diplopia and no clues to its cause. She had limited up and downgaze and a well circumscribed lesion in the levator superior rectus muscle complex.
A biopsy revealed a small cell neuroendocrine tumour or carcinoid tumour. An octreotide scan, which binds to somatostatin receptors on carcinoid tumours, located a lesion in the right iliac fossa, which was the primary lesion in the terminal ileum. The primary lesion was resected, the solitary orbital metastasis treated with radiotherapy and she maintains normal vision, with reduced motility over a 15-year follow-up.
Carcinoid metastasis to the orbit has an attraction for extraocular muscle, which may on occasions be multiple and bilateral.
Genetics, molecular biology and orbital disease
In this century, we will see major advances in our understanding of the genetics of orbital disease and its molecular biology.
There are some well-known and less well-known familial eyelid cancer syndromes, as well as some well-known and lesser known familial cancer syndromes that may impact on the orbit.
You’ll be aware of the orbital features of Neurofibromatosis type 1, with sphenoid bone dysplasia, plexiform neuromas and optic pathway gliomas being common. NF1 is due to a mutation on the long arm of chromosome 17, which codes for a tumour suppressor protein called Neurofibromin.
The less common NF2 phenotype includes bilateral vestibular schwannomas, and multiple meningiomas, including occasionally primary orbital meningiomas, as well as cataract. The NF2 gene is also a tumour suppressor, common in Schwann cells, and codes for a protein called Merlin.
Multiple endocrine neoplasia type 1 patients often develop parathyroid, pancreatic, carcinoid and pituitary tumours. The MEN1 gene is also a tumour suppressor, coding for the protein Menin.
Gardner’s syndrome, a subset of familial adenomatous polyposis (FAP), has multiple osteomas as part of its phenotype and a young man who had already undergone prophylactic colectomy for FAP, presented to us with an orbital osteoma.58 FAP is due to a mutation in the APC gene, and Gardner’s syndrome is polyposis coli plus tumours elsewhere, including skin and bone.
We recently saw a young boy with both a rhabdomyosarcoma in the left orbit and a choroid plexus carcinoma in the ventricles who sadly died from his tumours. His sister had also had an osteosarcoma. Genetic studies identified the Li-Fraumeni familial cancer syndrome. This syndrome leads to the development of many different tumours in children and young adults, including osteosarcomas and soft tissue sarcomas. Most cases are due to mutations in TP53, which codes for the well-known protein p53, sometimes referred to as the guardian of the genome for its role in DNA repair.
The study of the genetics of Graves’ orbitopathy offers an opportunity to identify patients at risk59 and to better understand the pathophysiology of the disease.60 This is but one example of the future direction of research in orbital disease.
Examining the gene expression profile of banked orbital biopsies has identified a proportion of patients with idiopathic orbital inflammation with similar gene expression to patients with GPA or Wegener’s granulomatosis.61 Such profiling may improve diagnostic rates in patients with the so-called limited form of the disease where biopsies may lack classical features and ANCA is often negative.
Conclusion
Robert Doyne was renowned as a meticulous clinical observer. I believe there will always be a place for the careful clinical observer with a broad knowledge, who can identify gaps in clinical knowledge, fill some of those gaps and make the care of our patients better now and into the future. I hope I have been able to do that in a small way.
Footnotes
The author declare no conflict of interest.
References
- Duke-Elder S. System of Ophthalmology. Henry Kimpton: London, 1974; Vol 13(part 2): chapter 12, p 819. [Google Scholar]
- Barlow T. On cases described as ‘acute rickets’ which are possibly a combination of rickets and scurvy, the scurvy being essential and the rickets variable. Medicochirurg Trans 1883; 66: 159–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The American Pediatric Society’s collective investigation on infantile scurvy in North America. Arch Pediatr 1898; 15: 481–508. [Google Scholar]
- Snow I. Eye symptoms of infantile scurvy. A case of infantile scurvy with extreme protrusion of the eyeball, shown by autopsy to be due to a large retrobulbar hematoma. Arch Pediatr 1905; 22: 576–580. [PMC free article] [PubMed] [Google Scholar]
- Mosdøl A, Erens B, Brunner EJ. Estimated prevalence and predictors of vitamin C deficiency within UK’s low-income population. J Pub Health 2008; 30: 456–460. [DOI] [PubMed] [Google Scholar]
- Riess KP, Farnen JP, Lambert PJ, Mathiason MA, Kothari SN. Ascorbic acid deficiency in bariatric surgical population. Surg Obese Relat Dis 2009; 5: 81–86. [DOI] [PubMed] [Google Scholar]
- Sloan BH, Kulwin DR, Kersten RC. Scurvy causing bilateral orbital hemorrhages. Arch Ophthalmol 1999; 117: 842–843. [DOI] [PubMed] [Google Scholar]
- Atalla M, McNab AA, Sullivan TJ, Sloan B. Non-traumatic subperiosteal orbital hemorrhage. Ophthalmology 2001; 108: 183–189. [DOI] [PubMed] [Google Scholar]
- McNab AA. Non-traumatic orbital haemorrhage. Surv Ophthalmol 2014; 59: 166–184. [DOI] [PubMed] [Google Scholar]
- Verma S, Sivanandan S, Aneesh MK, Gupta V, Seth R, Kabra S. Unilateral proptosis and extradural hematoma in a child with scurvy. Pediatr Radiol 2007; 37: 937–939. [DOI] [PubMed] [Google Scholar]
- Simon GJ, McNab AA. Idiopathic orbital hemorrhage related to the inferior rectus muscle: a rare cause for acute-onset diplopia and unilateral proptosis. Ophthalmology 2005; 112: 1838–1843. [DOI] [PubMed] [Google Scholar]
- Hakin KN, McNab AA, Sullivan TJ. Spontaneous hemorrhage within the rectus muscle. Ophthalmology 1994; 101: 1631–1634. [DOI] [PubMed] [Google Scholar]
- Yood RA, Skinner M, Rubinow A, Talarico L, Cohen AS. Bleeding manifestations in 100 patients with amyloidosis. JAMA 1983; 249: 1322–1324. [PubMed] [Google Scholar]
- Lett KS, Shah A, Gottlob I. External ophthalmoplegia as the presenting feature of systemic amyloidosis. Strabismus 2007; 15: 153–156. [DOI] [PubMed] [Google Scholar]
- Goshe JM, Schoenfield L, Emch T, Singh AD. Myeloma-associated orbital amyloidosis. Orbit 2010; 29: 274–277. [DOI] [PubMed] [Google Scholar]
- Slade MP, McNab AA. Fatal mucormycosis associated with deferoxamine therapy. Am J Ophthalmol 1991; 112: 594–595. [DOI] [PubMed] [Google Scholar]
- McNab AA, McKelvie P. Iron overload is a risk factor for zygomycosis. Arch Ophthalmol 1997; 115: 919–921. [DOI] [PubMed] [Google Scholar]
- Thornton IL, Clark J, Sokol JA, Hite M, Nunery WR. Radiographic evidence of prominent retro and suboribicularis oculi fat in thyroid-associated orbitopathy. Orbit 2016; 35: 35–38. [DOI] [PubMed] [Google Scholar]
- Bartalena L, Fatourechi V. Extrathyroidal manifestations of Graves’ disease: a 2014 update. J Endocrinol Invest 2014 37: 691–700. [DOI] [PubMed] [Google Scholar]
- Bunevicius R, Velickiene D, Prange AJ. Mood and anxiety disorders in women with treated hyperthyroidism and ophthalmopathy caused by Graves’ disease. Gen Hosp Psych 2005; 27: 133–139. [DOI] [PubMed] [Google Scholar]
- Fam J, Rush AJ, Burt T, Chan ES, Siddiqui PN, Assam PN et al. Thyroid autoimmune antibodies and depressive disorder in women. Ann Acad Med Singapore 2015; 44: 284–289. [PubMed] [Google Scholar]
- Scott IU, Siatkowski RM. Idiopathic orbital myositis. Curr Opinion Rheumatol 1997; 9: 504–512. [DOI] [PubMed] [Google Scholar]
- Mombaerts I, Koornneef L. Current status in the treatment of orbital myositis. Ophthalmology 1997; 104: 402–408. [DOI] [PubMed] [Google Scholar]
- Bahrami B, Rajak S, Selva D. Orbital myositis secondary to statin therapy. Med J Aust 2015; 203: 365–366. [DOI] [PubMed] [Google Scholar]
- Cooper LT, Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis – natural history and treatment. New Eng J Med 1997; 336: 1860–1866. [DOI] [PubMed] [Google Scholar]
- Avisar I, McNab AA, Dolman PJ, Patel B, Desousa JL, Sevla D et al. Endonasal dacrycystorhinostomy for nasolacrimal duct obstruction in patients with sarcoidosis. Orbit 2013; 32: 225–230. [DOI] [PubMed] [Google Scholar]
- Khong JJ, McNab AA. Medial transconjunctival intrinsic optic nerve biopsy: surgical technique and indications. Orbit 2012; 31: 227–232. [DOI] [PubMed] [Google Scholar]
- Prabhakaran VC, Saeed P, Esmaeli B, Sullivan TJ, McNab A et al. Orbital and adnexal sarcoidosis. Arch Ophthalmol 2007; 125: 1657–1662. [DOI] [PubMed] [Google Scholar]
- Mavrikakis I, Rootman J. Diverse clinical presentations of orbital sarcoid. Am J Ophthalmol 2007; 144: 760–775. [DOI] [PubMed] [Google Scholar]
- Demirci H, Christianson MD. Orbital and adnexal involvement in sarcoidosis: analysis of clinical features and systemic disease in 30 cases. Am J Ophthalmol 2011; 151: 1074–1080. [DOI] [PubMed] [Google Scholar]
- Hamano H, Kawa S, Horiuchi A, Unno H, Furuya N, Akamatsu T et al. High serum IgG4 concentrations in patients with sclerosing pancreatitis. N Engl J Med 2001; 344: 732–738. [DOI] [PubMed] [Google Scholar]
- Kamisawa T, Egawa N, Nakajima H. Autoimmune pancreatitis is a systemic autoimmune disease. Am J Gastroenterol 2003; 98: 2811–2812. [DOI] [PubMed] [Google Scholar]
- Cheuk Y, Yuen HKL, Chan LKC. Chronic sclerosing dacryoadenitis: part of the spectrum of IgG4-related sclerosing disease? Am J Surg Pathol 2007; 31: 643–645. [DOI] [PubMed] [Google Scholar]
- Takahira M, Kawano M, Zen Y, Minato H, Yamada K, Sugiyama K. IgG4-related chronic sclerosing dacryodenitis. Arch Ophthalmol 2007; 125: 1575–1578. [DOI] [PubMed] [Google Scholar]
- Deshpande V, Zen Y, Chan JK, Yi EE, Sato Y, Yoshino T et al. Consensus statement on the pathology of IgG4-related disease. Mod Pathol 2012; 25: 1181–1192. [DOI] [PubMed] [Google Scholar]
- Umehara H, Okazaki K, Masaki Y et al. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol 2012; 22: 21–30. [DOI] [PubMed] [Google Scholar]
- Goto H, Takahira M, Azumi A. Japanese Study Group for IgG4-related ophthalmic disease. Diagnostic criteria for IgG4-related ophthalmic disease. Jpn J Ophthalmol 2015; 59: 1–7. [DOI] [PubMed] [Google Scholar]
- McKelvie P, McNab AA, Hardy T, Rathi V. Comparative study of clinical, pathological, radiological and genetic features of patients with adult ocular adnexal xanthogranulomatous disease, Erdheim-Chester disease, and IgG4-related disease of the orbit/ocular adnexa. Ophthal Plast Reconstr Surg 2017; 33: 112–119. [DOI] [PubMed] [Google Scholar]
- Japanese study group of IgG4-related ophthalmic disease. A prevalence study of IgG4-related ophthalmic disease in Japan. Jpn J Ophthalmol 2013; 57: 573–579. [DOI] [PubMed] [Google Scholar]
- Plaza JA, Garrity JA, Dogan A, Ananthamurthy A, Witzig TE, Salamäo DR. Orbital inflammation with IgG4-positive plasma cells. Arch Ophthalmol 2011; 129: 421–428. [DOI] [PubMed] [Google Scholar]
- Deschamps R, Deschamps L, Depaz R, Coffin-Pichonnet S, Belange G, Jacomet PV et al. High prevalence of IgG4-related lymphoplasmacyctic infiltrative disorder in 25 patients with orbital inflammation: a retrospective case series. Br J Ophthalmol 2013; 97: 999–1004. [DOI] [PubMed] [Google Scholar]
- McNab AA, McKelvie P. IgG4-related ophthalmic disease. Part II: clinical aspects. Ophthal Plast Reconstr Surg 2015; 31: 167–178. [DOI] [PubMed] [Google Scholar]
- Hardy TG, McNab AA, Rose GE. Enlargement of the infraorbital nerve: an important sign associated with orbital reactive lymphoid hyperplasia or IgG4-related inflammation. Ophthalmology 2014; 121: 1297–1303. [DOI] [PubMed] [Google Scholar]
- Watanabe T, Fujinaga Y, Kawakami S, Hatta T, Hamano H, Kawa S et al. Infraorbital nerve swelling associated with autoimmune pancreatitis. Jpn J Radiol 2011; 29: 194–201. [DOI] [PubMed] [Google Scholar]
- Ohshima K, Sogabe Y, Sato Y. The usefulness of infraorbital nerve enlargement on MRI imaging in clinical diagnosis of IgG4-related orbital disease. Jpn J Ophthalmol 2012; 56: 380–382. [DOI] [PubMed] [Google Scholar]
- Wu A, Andrew NH, McNab AA, Selva D. IgG4-related ophthalmic disease: pooling of published cases and literature review. Curr Allergy Asthma Rep 2015; 15: 27. [DOI] [PubMed] [Google Scholar]
- Wu A, Andrew NH, McNab AA, Selva D. Bilateral IgG4-related ophthalmic disease: an indication for systemic imaging. Br J Ophthalmol 2016; 100: 1409–1411. [DOI] [PubMed] [Google Scholar]
- Perugino CA, Wallace ZS, Meyersohn N, Oliveira G, Stone JR, Stone JH. Large vessel involvement by IgG4-related disease. Medicine (Baltimore) 2016; 95: e3344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassidy DT, McKelvie P, Harris GJ, Rose GE, McNab AA. Lacrimal gland orbital lobe cysts associated with MALT lymphoma and primary Sjögren’s syndrome. Orbit 2005; 24: 257–263. [DOI] [PubMed] [Google Scholar]
- Liang Y, Yang Z, Qin B, Zhong R. Primary Sjögren’s syndrome and malignancy risk: a systematic review and meta-analysis. Ann Rheum Dis 2014; 73: 1151–1156. [DOI] [PubMed] [Google Scholar]
- Nutting CM, Shah-Desai S, Rose GE, Norton AP, Plowman PN. Thyroid orbitopathy possibly predisposes to late-onset of periocular lymphoma. Eye 2006; 20: 645–648. [DOI] [PubMed] [Google Scholar]
- Decaudin D, Ferroni A, Vincent-Salomon A, Beldjord K, Validire P, de Cremoux P et al. Ocular adnexal lymphoma and Helicobacter pylori gastric infection. Am J Hematol 2010; 85: 645–649. [DOI] [PubMed] [Google Scholar]
- Ferreri AJ, Ponzoni M, Guidoboni M, Resti AG, Politi LS, Cortelazzo S et al. Bacteria-eradicating therapy with doxycycline in ocular adnexal MALT lymphoma: a multicenter prospective trial. J Nat Cancer Inst 2006; 98: 1375–1382. [DOI] [PubMed] [Google Scholar]
- Han JJ, Kim TM, Jeon YK, Kim MK, Khwarg SI, Kim CW et al. Long-term outcomes of first-line treatment with doxycycline in patients with previously untreated ocular adnexal marginal zone B cell lymphoma. Ann Hematol 2015; 94: 575–581. [DOI] [PubMed] [Google Scholar]
- Goldberg RA, Rootman J. Tumors metastatic to the orbit. Surv Ophthalmol 1990; 35: 1–24. [DOI] [PubMed] [Google Scholar]
- Shields JA, Shields CL, Brotman HK, Carvalho C, Perez N, Eagle RC Jr. Cancer metastatic to the orbit: the 2000 Robert M Curts Lecture. Ophthal Plast Reconstr Surg 2001; 17: 346–354. [DOI] [PubMed] [Google Scholar]
- Valenzuela A, Archibald CW, Fleming B, Ong L, O’Donnell B, Crompton JJ et al. Orbital metastasis: clinical features, management and outcome. Orbit 2009; 28: 153–159. [DOI] [PubMed] [Google Scholar]
- McNab AA. Orbital osteoma in Gardner’s syndrome. Aust N Z J Ophthalmol 1998; 26: 169–171. [DOI] [PubMed] [Google Scholar]
- Khong JJ, Wang LY, Smyth GK, McNab AA, Hardy TG, Selva D et al. Differential gene expression profiling of orbital adipose tissue in thyroid orbitopathy. Invest Ophthalmol Vis Sci. 2015; 56: 6438–6447. [DOI] [PubMed] [Google Scholar]
- Khong JJ, Burdon KP, Lu Y, Leonardos L, Laurie KJ, Walsh JP et al. Association of polymorphisms in MACRO domain containing 2 with thyroid-associated orbitopathy. Invest Ophthalmol Vis Sci 2016; 57: 3129–3137. [DOI] [PubMed] [Google Scholar]
- Rosenbaum JT, Choi D, Wilson DJ, Grossniklaus HE, Harrington CA, Sibley CH et al. Orbital pseudotumor can be a localized from of granulomatosis with polyangiitis as revealed by gene expression profiling. Exp Molec Pathol 2015; 99: 271–278. [DOI] [PMC free article] [PubMed] [Google Scholar]