Abstract
Lung ailments in rheumatic diseases present unique challenges for diagnosis and management and are a source of significant morbidity and mortality for patients. Unlike the idiopathic interstitial pneumonias, patients with rheumatic diseases experience lung disease in the context of a systemic disease that may make it more difficult to recognize and that may present greater risks with treatment. Despite recent advances in our awareness of these diseases, there is still a significant lack of understanding of natural history to elucidate which patients will have disease that is progressive and thus warrants treatment. What we do know is that a subset of patients with rheumatic disease experience parenchymal lung disease that can prognostically resemble idiopathic pulmonary fibrosis, such as in rheumatoid arthritis, and that others can have aggressive inflammatory lung disease in the context of autoimmune myositis, systemic sclerosis, or an undifferentiated autoimmune process. As we enter into a paradigm shift where we view lung health as a cornerstone of our care of patients with rheumatic diseases, we hopefully will improve our ability to identify those patients at highest risk for pulmonary disease and progression, and offer emerging treatments which will result in better outcomes and a better quality of life.
Key Words: interstitial lung disease, myositis, rheumatoid arthritis, scleroderma
Abbreviations: AIM, autoimmune myositis; AIP, acute interstitial pneumonia; ANA, antinuclear antibody; ANCA, antineutrophil cytoplasmic antibody; AZA, azathioprine; CCP, cyclic citrullinated peptides; CyC, cyclophosphamide; DAD, diffuse alveolar damage; DAH, diffuse alveolar hemorrhage; Dlco, diffusion capacity for carbon monoxide; EGPA, eosinophilic granulomatosis with polyangiitis; GPA, granulomatosis with polyangiitis; HRCT, high-resolution CT; ILD, interstitial lung disease; IPF, idiopathic pulmonary fibrosis; IVIG, IV immunoglobulin; LIP, lymphocytic interstitial pneumonia; MALT, mucosa-associated lymphoid tissue; MCP, metacarpophalangeal; MDA5, melanoma differentiation-associated gene 5; MPA, microscopic polyangiitis; MMF, mycophenolate mofetil; MTP, metatarsophalangeal; NSIP, nonspecific interstitial pneumonia; OP, organizing pneumonia; PAH, pulmonary arterial hypertension; PFT, pulmonary function test; RA, rheumatoid arthritis; RA-ILD, rheumatoid arthritis-associated interstitial lung disease; RF, rheumatoid factor; SLE, systemic lupus erythematosus; SSc, systemic sclerosis; UIP, usual interstitial pneumonia
Lung complications in patients with rheumatic disease are common and may lead to significant morbidity and mortality. In some cases, patients with a known rheumatic syndrome will experience respiratory symptoms that will require evaluation and treatment, and in other patients, lung symptoms may be the first manifestation of an underlying rheumatic disease. In either case, the ability to secure a diagnosis of either entity early in the course offers the best chance to intervene and limit morbidity and mortality. In this paper, we offer a practical approach to identifying patients with pulmonary disease who may have an underlying rheumatic disease using important phenotypic features elicited by the history, physical examination, and laboratory, radiologic, and histopathologic data. Furthermore, we suggest an approach to screening and routine surveillance through highlighting demographic, serologic, and emerging biomarker data that may help identify those patients with known rheumatic disease who are at highest risk for the development of lung complications. Finally, we discuss established and emerging treatment options for this group of patients. This discussion focuses on interstitial and airway manifestations, given the significant morbidity associated with these conditions.
Interstitial Lung Disease: Radiologic and Histopathologic Patterns Seen in Rheumatic Diseases
The most common patterns of interstitial lung disease (ILD) observed in rheumatologic diseases include nonspecific interstitial pneumonia (NSIP), usual interstitial pneumonia (UIP), organizing pneumonia (OP), lymphocytic interstitial pneumonia (LIP), acute interstitial pneumonia (AIP)/diffuse alveolar damage (DAD), and rarely desquamative interstitial pneumonia (DIP). The rheumatic diseases most commonly affected by ILD include (in order of descending frequency) systemic sclerosis (SSc)/scleroderma, autoimmune myositis (AIM), rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), Sjögren syndrome, and undifferentiated rheumatoid disorders. Distinguishing between ILD associated with an idiopathic interstitial pneumonitis and a rheumatic disease is important, given that there may be a more favorable prognosis in patients with rheumatic disease, although survival in rheumatic disease-related UIP is similar to idiopathic pulmonary fibrosis (IPF).1 In some patients, more than one histologic pattern of disease may be present, for example, NSIP and OP seen together in AIM.
Nonspecific Interstitial Pneumonia
NSIP is commonly seen in most rheumatic diseases, especially in AIM and SSc. The prognosis associated with NSIP is more favorable compared with that of UIP, although fibrotic NSIP may parallel UIP. Diagnosing NSIP by high-resolution CT (HRCT) alone can be challenging due to less defined features seen on imaging, in some cases a biopsy is necessary. However, patients with rheumatic disease and ground-glass changes without honeycombing or traction bronchiectasis and without infection most likely have a predominantly inflammatory process such as cellular NSIP, which may be amenable to empirical anti-inflammatory therapy.
Usual Interstitial Pneumonia
The UIP pattern in rheumatic disease portends the worst prognosis, with a 5-year mortality of > 50%.2 It can be seen in most rheumatic diseases but is most frequently seen in RA (up to 60% of RA-associated ILD [RA-ILD]).3 Traditionally, disease-modifying antirheumatic drugs (DMARDs) such as mycophenolate mofetil (MMF) or azathioprine (AZA) have been used in patients with UIP, but little prospective data support their use, and the preferred treatment is unknown. Data from the Prednisone, Azathioprine, and N-Acetylcysteine: A Study That Evaluates Response in Idiopathic Pulmonary Fibrosis (PANTHER-IPF) trial showing antiinflammatory therapy to be deleterious in UIP related to IPF raises concerns that a similar approach in UIP associated with rheumatic disease may be detrimental.4 In that context, prospective trials using antifibrotic agents in such patients are ongoing and anticipated. Given the poor prognosis seen in UIP in general, early referral for transplantation should be considered.
Organizing Pneumonia
OP is characterized by intraluminal fibrosis in distal airways associated with interstitial inflammation. Characteristic radiological findings include consolidation, nodules, and unique features such as the atoll sign/reversed halo sign,5 which is a central ground-glass opacity surrounded by denser airspace consolidation of a crescentic or ring shape (Figs 1A, 1B). It can be seen in most rheumatic diseases and is especially notable in AIM, and particularly the antisynthetase syndrome. There is evidence that prognosis is worse in OP related to rheumatic disease vs cryptogenic OP.6 The presence of OP in a previously well individual with or without new systemic complaints merits a detailed investigation for an underlying autoimmune disorder, especially the antisynthetase syndrome.
Figure 1.
High-resolution CT images. A, Note the rind of ground-glass opacity surrounded by a denser consolidation, known as the atoll sign, in a patient with dermatomyositis and antisynthetae syndrome (Jo-1 antibody positive) and biopsy proven organizing pneumonia. B, Reverse halo sign, which is characterized by ground-glass attenuation surrounded by a ring of denser consolidation. C, Ruptured lung nodule with pneumothorax in a patient with rheumatoid arthritis (RA) and additional rheumatoid nodule in the left lung. D, Mucosa-associated lymphoid tissue (MALT) lymphoma in a 50-year-old woman with Sjögren syndrome and multiple lung nodules. E, Ruptured nodule on right with pneumothorax in a patient with RA and multiple RA nodules.
Lymphocytic Interstitial Pneumonia
LIP is an interstitial process characterized by a lymphoplasmacytic infiltrate, nodules, and lymphoid follicles and is typically seen in Sjögren syndrome but can also be seen in RA, SSc, and other rheumatic syndromes. In some cases, nodular lesions may appear to be inflammatory and associated with known LIP but in fact may represent lymphoma, especially the mucosa-associated lymphoid tissue (MALT) type (Figs 1C, 1D). Given that patients with both Sjögren syndrome and RA are at higher risk for lymphoma, close surveillance and consideration for biopsy are warranted.7
Other Important Lung Manifestations in Rheumatic Diseases
Airway disease, including follicular bronchiolitis, constrictive or obstructive bronchiolitis, bronchiectasis, and emphysema, may occur and may coexist with underlying ILD, especially in RA.8, 9, 10 Airway changes may be noted early in RA, and emerging evidence suggests that upper respiratory exposure to antigens, such as citrulline, and resultant autoimmunity may initiate or propagate RA.11, 12 In particular, bronchiectasis is common in RA and may increase the risk of infection with RA treatment especially with biological agents. The presence of obstruction on pulmonary function tests (PFTs) in the face of declining respiratory status with air trapping/mosaicism on CT scans raises the issue of reversible and treatable airway disease (follicular bronchiolitis) vs obliterative bronchiolitis, which is not responsive to medical therapy unless an offending drug is identified and removed. Of note, many patients may also have concomitant emphysema in the context of a previous smoking history.
Rheumatoid nodulosis in the lung is frequently seen in association with RA and although often asymptomatic, nodules have been noted to rupture, with resulting pneumothorax and infection (Fig 1E). In addition, inflammatory nodules in RA and Sjögren syndrome may present a diagnostic quandary, as they can mimic malignancy.
Pleural inflammation and effusions are common in many rheumatic syndromes, including RA, SLE, and occasionally in SSc and Sjögren syndrome. Exudative effusions are most typically seen in RA with characteristic low glucose and pH values.13 Persistent pleural inflammation has been postulated as a cause of shrinking lung syndrome seen in SLE.14
Pulmonary Disease and Suspected Underlying Rheumatic Syndrome: Clues to a Diagnosis
Key historical and examination findings may reveal a pattern of disease consistent with a rheumatic syndrome that can aid in formulating a diagnosis (Table 1). Some individuals with ILD have minimal autoimmune phenotypic or serologic features that are suggestive of, but not definitive for, a rheumatic syndrome, earning a designation of interstitial pneumonia with autoimmune features (IPAF).15 If a specific rheumatologic diagnosis is considered likely by phenotype, a reasonable and potentially more cost-effective approach to serologic testing can be used. If a specific histologic or radiological pattern is recognized in the absence of a clear phenotype, a defined set of serologic tests may be reasonable as well (Table 2). When the suspicion of a rheumatic syndrome is present but no clear pattern emerges from the history or physical examination, a more broad approach to laboratory testing should be considered.
Table 1.
Rheumatic Diseases and Their Associated Historical and Physical Findings, Imaging Features, and Serologic Characteristics
| Rheumatic Disease | Historical and Physical Findings | Common Imaging Features | Typical Serologic Characteristics |
|
|---|---|---|---|---|
| Autoantibody | Special Consideration | |||
| SSc/scleroderma | Raynaud syndrome, gastroesophageal reflux disease, calcinosis cutis, skin thickening, ulcers on the fingertips, limited oral aperture, nail fold capillary microscopic abnormalities, pericarditis | NSIP UIP OP AIP/DAD DAH |
ANA | Nucleolar pattern, anticentromere pattern |
| SCL-70, TH/TO, RNA polymerase III, U3 RNP | … | |||
| AIM | Dysphagia, proximal muscle weakness, photosensitive rash on hands/chest/face/back, mechanics hands, antisynthetase syndrome (Raynaud syndrome, mechanics hands, fever, arthritis, ILD) | NSIP OP UIP AIP/DAD Pneumomediastinum |
Creatinine kinase, aldolase | … |
| MDA5 | Skin nodules, dermatomyositis, and ILD | |||
| Jo-1, PL-12, PL-7 OJ, EJ, ZO, KS | Antisynthetase syndrome | |||
| Anti-PM/SCL, Ku | Seen with overlap of myositis and SSc | |||
| Anti-Ro/SSA | Ro 52-kD associated with AIM and SSc and ILD | |||
| Rheumatoid arthritis | Swollen joints in the hands (especially wrists, MCP joints), knees, and feet (including the MTP joints); rheumatoid nodules; inflammatory eye disease | UIP NSIP OP Bronchiolitis Lung nodules |
RF | Higher rate of false positive results with RF alone |
| Anticyclic citrullinated peptides | … | |||
| SLE | Joint pain, alopecia, oral ulcers, pleurisy, rash, photosensitivity, pericarditis | Acute pneumonitis NSIP/UIP DAH OP |
Anti-Smith | … |
| Anti-Ro/SSA Anti-La/SSB |
Ro 52-kD associated with SSc and AIM and ILD | |||
| Sjögren syndrome | Dry eye and mouth, parotid swelling (Fig 4C), neuropathy | LIP Follicular bronchiolitis Cystic lung disease NSIP UIP OP |
Anti-Ro/SSA Anti-La/SSB |
… |
| RF | … | |||
| Mixed connective tissue disease | Variable signs and symptoms primarily associated with SLE, SSc, and AIM | NSIP LIP |
ANA | Nuclear speckled pattern |
| Anti-RNP Ribonucleoprotein | Features of different rheumatic diseases/mixed connective tissue disease including SSc, AIM, SLE, with higher risk of ILD and PAH | |||
| Vasculitis | Palpable purpura, necrotic skin lesions, otitis media, epistaxis, nasal septal perforation, mononeuritis multiplex, hemoptysis, oral ulcers | DAH UIP |
ANCA | GPA, MPA, and EGPA |
| Antiglomerular basement membrane | ||||
| RF | GPA, cryoglobulinemia | |||
AIM = autoimmune myositis; AIP/DAD = acute interstitial pneumonia/diffuse alveolar damage; ANA = antinuclear antibodies; ANCA = antineutrophil cytoplasmic antibody; DAH = diffuse alveolar hemorrhage; EGPA = eosinophilic granulomatosis with polyangiitis; GPA = granulomatosis with polyangiitis; ILD = interstitial lung disease; LIP = lymphocytic interstitial pneumonia; MCP = metacarpophalangeal; MPA = microscopic polyangiitis; MTP = metatarsophalangeal; NSIP = nonspecific interstitial pneumonia; OP = organizing pneumonia; PAH = pulmonary arterial hypertension; RF = rheumatoid factor; SLE = systemic lupus erythematosus; SSc = systemic sclerosis; UIP = usual interstitial pneumonia.
Table 2.
Approach to Ordering Specific Serologic Tests Based on HRCT or Histologic Pattern on Biopsy, or Both, in the Context of Absence of a Clear Phenotype
| Pattern | Serologic Tests to Order |
|---|---|
| Definite UIP | RF, CCP, ANA, ANCA |
| NSIP, OP, undefined pattern, or AIP | ANA, Ro, La, RNP, Scl-70, RF, CCP, myositis antibody panel, creatinine kinase, aldolase, MDA5 |
| Bronchiolitis (including obliterative bronchiolitis) | RF, anti-CCP |
| DAH | ANCA, ANA, antiphospholipid antibody, cryoglobulins, antiglomerular basement membrane antibody |
| LIP | ANA, Ro, La, RF, anti-CCP |
CCP = anticyclic citrullinated peptides. See Table 1 legend for expansion of other abbreviations.
Assessment of Hospitalized Patients With Rapidly Progressive ILD
The presence of an underlying rheumatic disease always needs to be considered in a patient with unexplained progressive or rapidly progressive respiratory disease. Clues to suggest a rheumatic disease include sclerodactyly, limited oral aperture, nail fold capillary changes, telangiectasia, and adult-onset Raynaud syndrome suggestive of the SSc spectrum of diseases (Fig 2); skin rashes on the extremities or face, or both, nodules, diffuse erythema, plaques on the extensor surfaces, periungual erythema, nail fold capillary microscopic changes, and “mechanics hands,” which are fissures and cracking seen on the tips and radial aspect of the fingers, suggestive of dermatomyositis or antisynthetase syndrome, or both (Fig 3); symmetrical inflammatory arthritis in the hands and feet suggestive of RA; and skin rash, livedo reticularis, and joint pain, especially in young female patients, suggestive of SLE (although similar acute presentations have been noted in male patients). In the context of diffuse alveolar hemorrhage (DAH), the presence of sinusitis with or without epistaxis, otitis, nasal septal perforation (which requires otoscopic evaluation), palpable purpura, and mononeuritis multiplex all suggest the possibility of vasculitis, including antineutrophil cytoplasmic antibody (ANCA)-associated disease. Rarely DAH may be a presenting feature in SLE and in the antiphospholipid antibody syndrome. In such patients with a rapidly progressive course, early evaluation by a rheumatologist or dermatologist, or both, is reasonable, as opposed to waiting for serologic test results that frequently take days or longer.
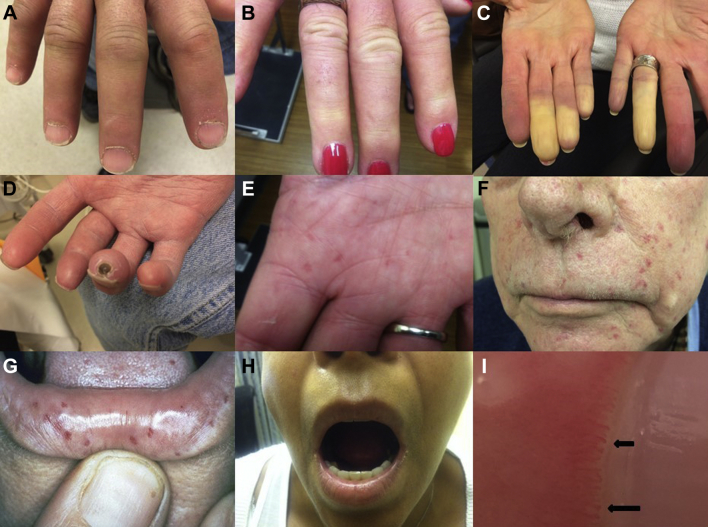
Figure 2.
Clinical characteristics seen in systemic sclerosis/scleroderma. A, Sclerodactyly; note the lack of vertical skin creases on the extensor surfaces of the fingers from the proximal interphalangeal joint to the distal interphalangeal joint. B, Active Raynaud syndrome with sclerodactyly. C, Sharp demarcation in perfusion of the distal digits in a patient with active Raynaud syndrome. D, Ischemic ulceration in a patient with severe Raynaud syndrome. E, Palmar telangiectasia. F, Facial telangiectasia. G, Telangiectasia on the lip and tongue. H, Limited oral aperture. I, Dilated nail fold capillaries, which indicate a vasculopathy seen in scleroderma and other rheumatic syndromes.
Figure 3.
Clinical characteristics seen in the autoimmune myositis. A, Diffuse erythema of the face in a patient with dermatomyositis and interstitial lung disease (ILD). B, Periungual erythema (down arrows) of fingers with ischemic vascular changes (up arrow) in the periungual area in a patient with dermatomyositis. C, Eyelid erythema and scaling seen in dermatomyositis. D, Gottren papules over the extensor surfaces of the fingers with periungual erythema in dermatomyositis. E, Cracking in the distal tips of the fingers of a patient with antisynthetase syndrome: “mechanics hands.” F, Mechanics hands in the antisynthetase syndrome. G, Nodular erythematous lesions on the palmar surface of the hand seen in melanoma differentiation-associated gene 5 (MDA5) antibody-related ILD. H, Healing ulcerating plaques on the dorsal surface of the hand in a patient with MDA-5 antibody-related ILD. I, Pneumomediastinum in dermatomyositis.
Clinical Considerations in Select Rheumatic Diseases
Systemic Sclerosis/Scleroderma
SSc is a systemic disorder characterized by excessive fibroblast proliferation, autoimmunity, and endothelial dysfunction leading to organ dysfunction involving the skin, lung, heart, and kidneys. Characteristic early features in SSc include sclerodactyly (characterized by thickening of the skin on the dorsal surfaces of the hands and fingers, which can over time extend to the extremities, trunk, and face), Raynaud syndrome with cutaneous ulcers, telangiectasia, a limited oral aperture, and nail fold capillary changes (Fig 2). SSc may be diffuse or limited in nature based on the extent of skin disease. The development of new-onset Raynaud syndrome in an adult should raise the suspicion of an emerging rheumatic disease.
Among all rheumatic diseases, ILD is most common in SSc, occurring in > 50% of patients, and most frequently involves NSIP.16 Clinically significant ILD tends to develop earlier. It is seen more frequently in diffuse SSc (in which skin involvement is above the elbows or knees) but is well characterized in those with limited SSc and even in patients with SSc without skin involvement (sine scleroderma).17 Pulmonary arterial hypertension (PAH) occurs in 8% to 12% of SSc and is the leading cause of death.18 Concomitant ILD and PAH portends a worse prognosis and warrants an aggressive treatment approach, with early consideration for transplantation.19 The patients at greatest risk for the development of progressive ILD in SSc include those who are SCL-70-antibody positive, African Americans, older patients, and male patients, with an early decline in FVC and diffusion capacity for carbon monoxide (Dlco) and a greater extent of disease seen on HRCT.20, 21, 22, 23 Emerging biomarkers, cytokines, and chemokines may help focus efforts for screening those patients at highest risk for the development of disease and progression with a favorable response to treatment.24, 25
Autoimmune Myositis, Previously Known as Idiopathic Inflammatory Myopathies
AIM encompass patients with muscle inflammation/weakness and malar/eyelid/periungual erythema, scaly erythematous plaques over the extensor surface of the hands (“Grotten papules”), and erythema over the back of the arms and chest (Figs 3A-D). Much of the morbidity and mortality relates to associated lung disease, including respiratory muscle dysfunction, aspiration, and ILD, most notably NSIP and OP followed by UIP and AIP.26 A worse prognosis is noted in older age groups, acute and subacute presentations with low initial FVC, amyopathic cases, and when concomitant PAH is present.27, 28
The antisynthetase syndrome deserves special consideration given the very high rates of ILD, which can be rapidly progressive and potentially fatal.29, 30 Although this disorder is classically characterized by mechanics hands (Figs 3E, 3F), inflammatory arthritis, myositis, fever, Raynaud syndrome, and ILD, in many cases patients will have only some of these features. ILD may present with consolidation or diffuse ground-glass and reticular opacities consistent with OP or NSIP and in some cases may progress to AIP. Although this syndrome is most often associated with the Jo-1 antibody, rarer antibodies such as PL-12 and PL 7 are closely linked to ILD as the primary manifestation, and other features such as myositis may be absent. Although Jo-1 testing is often available for rapid turnaround, the rarer antibodies are often part of a broader panel of myositis-specific and myositis-related antibodies, which are of variable reliability depending on the laboratory and assay used, and it may take significant time to obtain the results. However, if there is a high index of suspicion and testing is pending or initially negative, therapy with aggressive immunosuppression should be considered.
Recently, a novel antibody known as antimelanoma differentiation-associated gene 5 (MDA5) has been associated with myositis or amyopathic myositis and active and progressive ILD in a significant number of patients, leading to severe lung dysfunction and death.31, 32 Patients may have cutaneous lesions that are nodular or with a characteristic induration and ulceration (Figs 3G, 3H) or other cutaneous features seen in dermatomyositis.33 Pneumomediastinum has been noted in this population, as well as in patients with dermatomyositis without MDA534 (Fig 3I). Testing for this antibody is limited to select laboratories, so if there is a high clinical suspicion for this entity, aggressive therapy should be initiated while testing is pending, including early consideration for lung transplantation.
Rheumatoid Arthritis
RA is characterized by swollen joints in the hands, especially wrists and metacarpophalangeal (MCP) joints, knees, and feet, including the metatarsophalangeal (MTP) joints, rheumatoid nodules (Fig 4A), and inflammatory eye disease. All compartments of the respiratory system can be involved, but the most important manifestations include ILD and airway disease. ILD occurs clinically in 10% of patients and subclinically in another 30% of patients.35, 36 In some cases, ILD may precede the development of articular disease. The UIP pattern occurs in 60% of patients with RA-ILD, with a survival that may be similar to that of IPF.2, 37 Demographic data suggest that older age, male sex, smoking, and seropositivity are risk factors for a higher risk of disease. The role of anticitrullinated protein antibodies (ACPA) and citrullination in the development of lung disease is an area of active research.38, 39 A combination of demographic risk factors with emerging serologic and genetic biomarkers may help identify appropriate patients for screening with physiological and radiological testing.40, 41, 42
Figure 4.
Additional clinical findings suggestive of a rheumatic syndrome. A, Nodulosis of the extensor elbow region in rheumatoid arthritis. B, Dilated esophagus with air-fluid level in a 68-year-old man with systemic sclerosis. C, Right parotid swelling in a 58-year-old woman with Sjögren syndrome.
Drug-induced lung disease has been described in most DMARD and biological agents frequently used in RA.43 Methotrexate rarely causes an acute pneumonitis (likely < 1%), and sulfasalazine and gold can rarely lead to obliterative bronchiolitis. Tumor necrosis factor inhibitors have been implicated in case reports as worsening underlying ILD, although larger national registries offer no clear correlation between the use of these agents and the development or progression of ILD.44, 45 In general, if drug-induced lung toxicity is suspected, the suspected agent should be removed, the patient assessed for infection, and a course of glucocorticoids considered if symptoms persist or worsen.
ANCA-Associated Vasculitis
ANCA-associated vasculitis includes microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA), and eosinophilic granulomatosis with polyangiitis (EGPA). The respiratory system is often involved in all three types, along with palpable purpura, necrotic skin lesions, otitis media, epistaxis, nasal septal perforation, mononeuritis multiplex, and oral ulcers. Although DAH and hemoptysis are the most frequent and potentially life-threatening lung complications of vasculitis, ILD in the UIP pattern has also been described, particularly in those with MPA.46, 47 It is unknown if the development of fibrosis is related to the underlying inflammatory process associated with vasculitis in the lung, so the presence of UIP in this group is treated separately from the underlying lung vasculitis.
Approaches to Screening for ILD in Rheumatic Diseases
Delineation of screening strategies for lung disease in patients with rheumatic diseases are consensus based. Early referral by the rheumatologist or primary care provider to pulmonary medicine practitioners is important in the face of symptoms (dyspnea, cough), physical examination findings (ambulatory hypoxemia, clubbing, bibasilar crackles), decrements on baseline PFTs (restrictive lung disease or diminished 6-min walk distance), and ILD seen on HRCT. Conversely, given that many patients with ILD may have an underlying rheumatic disease, a detailed assessment by a rheumatologist is an important component of the evaluation.
Systemic Sclerosis
Given the frequency of lung disease in SSc and the increased morbidity and mortality associated with concurrent PAH, assessment for early evidence of ILD and PAH in this population is considered standard of care. All patients should be screened with baseline PFTs, including spirometry, total lung capacity and diffusion capacity, as well as echocardiography and HRCT imaging.48 Although a normal HRCT scan at baseline is encouraging for a low rate of progression, an abnormal baseline scan implies a variable risk of progression.49 If all baseline screening is normal, follow-up annual PFTs should be performed. If there is a decline of > 10% FVC or > 15% Dlco, repeated HRCT imaging and echocardiography should be considered. If at any time the HRCT scan demonstrates > 20% fibrotic lung disease (extensive), treatment should be considered; < 20% fibrotic ILD can be followed with PFTs every 3 to 6 months.23 The 6-min walk test, although not validated in SSc, can be a useful test during initial and follow-up evaluations to suggest underlying ILD or emerging PAH. Technical limits related to Raynaud syndrome may be overcome with forehead or ear probes.50 Assessment for an esophageal motility abnormality, a common problem in SSc and other rheumatic diseases, is warranted to assess for aspiration risk given the potential for exacerbation of lung injury51 (Fig 4B).
Autoimmune Myositis
Patients with AIM, especially those with the antisynthetase syndrome, should be screened with baseline PFTs and HRCT scans given the significant risk for ILD. If the HRCT scan is abnormal and the patient is symptomatic with an FVC < 70%, treatment should be considered. If the patient is not symptomatic and the FVC is > 70%, PFTs should be performed every 3-6 months. Patients should also undergo an assessment for esophageal dysmotility. In addition, the variable risk of PAH developing in AIM requires vigilance and appropriate surveillance.28
Rheumatoid Arthritis
In RA, demographic predictors exist that point toward a screening algorithm using PFT results and HRCT scans to identify ILD in high-risk individuals such as older male smokers. Emerging biomarker data may offer further evidence as to which groups are at highest risk for ILD.42 If the screening HRCT scan is abnormal, serial PFT results should be assessed every 3-6 months. A decision to treat should be based on the ILD pattern, the presence of symptoms, and the degree of restriction on PFTs. A decline in FVC of 10% in RA-ILD suggests a higher risk of mortality and may help guide decisions to treat.37 Of note, since many patients with RA are ever smokers, CT scanning for cancer surveillance may be indicated, offering an opportunity for assessment of subclinical ILD.
Treatment Strategies: Present and Future
Historically, those with rheumatic disease and ILD were treated with a wide array of predominantly anti-inflammatory agents, but approaches to treatment are evolving with emerging therapies targeting fibrosis.52, 53 Table 3 provides our approach regarding screening and treatment options. In inflammatory diseases in which cellular NSIP or OP is prominent, we frequently use MMF, although other agents such as cyclophosphamide (CyC), AZA, or calcineurin inhibitors may be efficacious. In patients with predominant fibrotic disease (fibrosing NSIP or UIP), these agents appear to be only marginally efficacious, and emerging antifibrotic therapies such as pirfenidone and nintedanib may prove to be useful, with prospective trials highly anticipated.54 In ILD, in which the predominant lung lesion is dominated by lymphoid follicles or an LIP pattern (such as in airway disease in RA or Sjögren syndrome), B-cell deletion, such as with rituximab, may be a sensible choice of therapy.55 Recent efforts have focused on the development of outcome measures specific to the rheumatic diseases that could be applied to prospective trials to enrich appropriate inclusion.56, 57
Table 3.
Options for Screening and Treatment of ILD for Specific Rheumatic Diseases
| Rheumatic Disease | Screening | Treatment |
|---|---|---|
| SSc | Baseline PFTs, HRCT, and echocardiography for all patients; if normal, yearly PFTs. If there is a decline in FVC > 10% or Dlco > 15% consider repeated CT imaging and echocardiography. Consider treatment in any patient with fibrotic lung disease > 20% on HRCT (extensive); if < 20%, PFTs every 3-6 mo. | MMF: Use up to 3 g/d CyC: An option for 6-9 mo with conversion to MMF or AZA In early rapidly progressive disease, consider suitability for stem cell transplantation at selected centers |
| AIM | Baseline PFTs and HRCT in all patients. If CT results are abnormal and patient is symptomatic with FVC < 70%, consider treatment. If patient is not symptomatic with FVC > 70%, PFTs every 3-6 mo. | For moderate disease: MMF, AZA, or tacrolimus with steroids Rituximab should be considered in antisynthetase syndrome In MDA5-ILD, aggressive therapy including high-dose glucocorticoids and at least 1 other steroid-sparing agents such as CyC, Rituxan or MMF should be used |
| Rheumatoid arthritis | Baseline PFTs in high-risk individuals (smokers, male sex, older age). If abnormal results, consider HRCT | If predominantly inflammatory disease, MMF or rituximab If predominantly fibrotic disease, consider antifibrotic agents pending results from ongoing trials |
AZA = azathioprine; CyC = cyclophosphamide; Dlco = diffusion capacity for carbon monoxide; HRCT = high-resolution CT; MDA5 = melanoma differentiation-associated gene 5; MMF = mycophenolate mofetil; PFTs = pulmonary function tests. See Table 1 legend for expansion of other abbreviations.
Systemic Sclerosis
For patients with progressive or extensive SSc-ILD (> 10% decline in FVC or >15% decline in Dlco), retrospective data and two prospective treatment trials showed a modest benefit using oral CyC or MMF.58, 59, 60, 61 Based on better tolerability and less toxicity in the Scleroderma Lung Study II,60 MMF has become our first-line therapy (up to 3 g/d), although CyC for 6 to 9 months with conversion to MMF or AZA is a second option. Recent experience with autologous bone marrow transplantation in SSc suggests improved survival in selected patients with significant or rapidly progressive lung disease, and ongoing trials are in progress.62, 63
Autoimmune Myositis
In AIM with moderate ILD, MMF, AZA, or tacrolimus with steroids are all options. Rituximab should be particularly considered in the antisynthetase syndrome. In MDA5-ILD, aggressive therapy including high-dose glucocorticoids and at least one other steroid-sparing agent such as CyC or MMF should be used. Some centers suggest additional therapy with both B-cell-deleting therapy and IV immunoglobulin (IVIG) until clinical stability is achieved, although no prospective data support this approach. Given the poor prognosis that may be associated with this syndrome, early consideration for lung transplantation is imperative.
Rheumatoid Arthritis
In RA-ILD, disease that is predominantly inflammatory is often treated with MMF or rituximab. It is postulated that fibrotic disease would probably respond better to antifibrotic therapy given the overlap between the RA-UIP phenotype and IPF, and studies are currently ongoing and highly anticipated.
Rapidly Progressive Disease
In patients with rapidly progressive disease, in the face of ambiguous physical findings but when a rheumatic disease is suspected, aggressive management with high-dose glucocorticoids and IV cyclophosphamide may be warranted. In some cases, rituximab may be added, particularly in AIM with the antisynthetase syndrome or suspected MDA5 antibody-associated ILD. If myositis and severe weakness is evident on examination or by elevated muscle enzymes, IVIG may be added temporarily to augment muscle function and offer other potential benefits with limited adverse events. Plasmapheresis may be offered in the context of severe immune-mediated lung disease such as DAH, with the hope of clearing immune complexes while awaiting the onset of action of other immunomodulating agents. Finally, patients with disease that is extensive or progressive or those who have a response to therapy that is suboptimal or ineffective need consideration for lung transplantation.64, 65
In summary, the intersection between pulmonary and rheumatologic disease represents a unique and often ambiguous paradigm of medicine that requires vigilance and meticulous assessment but most importantly a multidisciplinary approach involving pulmonary medicine, rheumatology, radiology, pathology, thoracic surgery and other disciplines. We hope that with greater recognition and understanding of these disorders, improved diagnostic and screening strategies, and more targeted therapies, both outcomes and quality of life for this group of challenging patients will improve.
Acknowledgments
Financial/nonfinancial disclosures: The authors have reported to CHEST the following: T. J. D. and P. F. D. are involved in a clinical trial with Genentech.
Other contributions: We thank Lindsey MacFarlane, MD, for her review of this manuscript. CHEST worked with the authors to ensure that the Journal policies on patient consent to report information were met.
Role of sponsors: The sponsor had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript.
Footnotes
FUNDING/SUPPORT: This work was supported by the National Institutes of Health [Grant No. 1 K23 HL119558 to T. J. D.].
References
- 1.Park J.H., Kim D.S., Park I.N. Prognosis of fibrotic interstitial pneumonia: idiopathic versus collagen vascular disease-related subtypes. Am J Respir Crit Care Med. 2007;175(7):705–711. doi: 10.1164/rccm.200607-912OC. [DOI] [PubMed] [Google Scholar]
- 2.Strand M.J., Sprunger D., Cosgrove G.P. Pulmonary function and survival in idiopathic vs secondary usual interstitial pneumonia. Chest. 2014;146(3):775–785. doi: 10.1378/chest.13-2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bongartz T., Nannini C., Medina-Velasquez Y. Incidence and mortality of interstitial lung disease in rheumatoid arthritis: a population-based study. Arthritis Rheum. 2010;62:1583–1591. doi: 10.1002/art.27405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Idiopathic Pulmonary Fibrosis Clinical Research Network. Raghu G., Anstrom K.J., King T.E., Jr. Prednisone, azathioprine, and N-acetylcysteine for pulmonary fibrosis. N Engl J Med. 2012;366(21):1968–1977. doi: 10.1056/NEJMoa1113354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Godoy M.C., Viswanathan C., Marchiori E. The reversed halo sign: update and differential diagnosis. Br J Radiol. 2012;85(1017):1226–1235. doi: 10.1259/bjr/54532316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yoo J.W., Song J.W., Jang S.J. Comparison between cryptogenic organizing pneumonia and connective tissue disease-related organizing pneumonia. Rheumatology (Oxford) 2011;50(5):932–938. doi: 10.1093/rheumatology/keq410. [DOI] [PubMed] [Google Scholar]
- 7.Tian X., Yi E.S., Ryu J.H. Lymphocytic interstitial pneumonia and other benign lymphoid disorders. Semin Respir Crit Care Med. 2012;33(5):450–461. doi: 10.1055/s-0032-1325156. [DOI] [PubMed] [Google Scholar]
- 8.Perez T., Remy-Jardin M., Cortet B. Airways involvement in rheumatoid arthritis: clinical, functional, and HRCT findings. Am J Respir Crit Care Med. 1998;157(5 Pt 1):1658–1665. doi: 10.1164/ajrccm.157.5.9710018. [DOI] [PubMed] [Google Scholar]
- 9.Devouassoux G., Cottin V., Liote H. Characterisation of severe obliterative bronchiolitis in rheumatoid arthritis. Eur Respir J. 2009;33(5):1053–1061. doi: 10.1183/09031936.00091608. [DOI] [PubMed] [Google Scholar]
- 10.Wilczynska M.M., Condliffe A.M., McKeon D.J. Coexistence of bronchiectasis and rheumatoid arthritis: revisited. Respir Care. 2013;58(4):694–701. doi: 10.4187/respcare.01857. [DOI] [PubMed] [Google Scholar]
- 11.Wilsher M., Voight L., Milne D. Prevalence of airway and parenchymal abnormalities in newly diagnosed rheumatoid arthritis. Respir Med. 2012;106(10):1441–1446. doi: 10.1016/j.rmed.2012.06.020. [DOI] [PubMed] [Google Scholar]
- 12.Reynisdottir G., Karimi R., Joshua V. Structural changes and antibody enrichment in the lungs are early features of anti-citrullinated protein antibody-positive rheumatoid arthritis. Arthritis Rheumatol. 2014;66(1):31–39. doi: 10.1002/art.38201. [DOI] [PubMed] [Google Scholar]
- 13.Balbir-Gurman A., Yigla M., Nahir A.M., Braun-Moscovici Y. Rheumatoid pleural effusion. Semin Arthritis Rheum. 2006;35(6):368–378. doi: 10.1016/j.semarthrit.2006.03.002. [DOI] [PubMed] [Google Scholar]
- 14.Henderson L.A., Loring S.H., Gill R.R. Shrinking lung syndrome as a manifestation of pleuritis: a new model based on pulmonary physiological studies. J Rheumatol. 2013;40(3):273–281. doi: 10.3899/jrheum.121048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fischer A., Antoniou K.M., Brown K.K. An official European Respiratory Society/American Thoracic Society research statement: interstitial pneumonia with autoimmune features. Eur Respir J. 2015;46(4):976–987. doi: 10.1183/13993003.00150-2015. [DOI] [PubMed] [Google Scholar]
- 16.Bouros D., Wells A.U., Nicholson A.G. Histopathologic subsets of fibrosing alveolitis in patients with systemic sclerosis and their relationship to outcome. Am J Respir Crit Care Med. 2002;165(12):1581–1586. doi: 10.1164/rccm.2106012. [DOI] [PubMed] [Google Scholar]
- 17.Fischer A., Swigris J.J., Groshong S.D. Clinically significant interstitial lung disease in limited scleroderma: histopathology, clinical features, and survival. Chest. 2008;134(3):601–605. doi: 10.1378/chest.08-0053. [DOI] [PubMed] [Google Scholar]
- 18.Mathai S.C., Hassoun P.M. Pulmonary arterial hypertension associated with systemic sclerosis. Expert Rev Respir Med. 2011;5(2):267–279. doi: 10.1586/ers.11.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mathai S.C., Hummers L.K., Champion H.C. Survival in pulmonary hypertension associated with the scleroderma spectrum of diseases: impact of interstitial lung disease. Arthritis Rheum. 2009;60(2):569–577. doi: 10.1002/art.24267. [DOI] [PubMed] [Google Scholar]
- 20.Winstone T.A., Assayag D., Wilcox P.G. Predictors of mortality and progression in scleroderma-associated interstitial lung disease: a systematic review. Chest. 2014;146(2):422–436. doi: 10.1378/chest.13-2626. [DOI] [PubMed] [Google Scholar]
- 21.Ryerson C.J., O'Connor D., Dunne J.V. Predicting mortality in systemic sclerosis-associated interstitial lung disease using risk prediction models derived from idiopathic pulmonary fibrosis. Chest. 2015;148(5):1268–1275. doi: 10.1378/chest.15-0003. [DOI] [PubMed] [Google Scholar]
- 22.Khanna D., Nagaraja V., Tseng C.H. Predictors of lung function decline in scleroderma-related interstitial lung disease based on high-resolution computed tomography: implications for cohort enrichment in systemic sclerosis-associated interstitial lung disease trials. Arthritis Res Ther. 2015;17:372. doi: 10.1186/s13075-015-0872-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Goh N.S., Desai S.R., Veeraraghavan S. Interstitial lung disease in systemic sclerosis: a simple staging system. Am J Respir Crit Care Med. 2008;177(11):1248–1254. doi: 10.1164/rccm.200706-877OC. [DOI] [PubMed] [Google Scholar]
- 24.De Lauretis A., Sestini P., Pantelidis P. Serum interleukin 6 is predictive of early functional decline and mortality in interstitial lung disease associated with systemic sclerosis. J Rheumatol. 2013;40(4):435–446. doi: 10.3899/jrheum.120725. [DOI] [PubMed] [Google Scholar]
- 25.Volkmann E.R., Tashkin D.P., Roth M.D. Changes in plasma CXCL4 levels are associated with improvements in lung function in patients receiving immunosuppressive therapy for systemic sclerosis-related interstitial lung disease. Arthritis Res Ther. 2016;18(1):305. doi: 10.1186/s13075-016-1203-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Marie I., Hachulla E., Cherin P. Interstitial lung disease in polymyositis and dermatomyositis. Arthritis Rheum. 2002;47(6):614–622. doi: 10.1002/art.10794. [DOI] [PubMed] [Google Scholar]
- 27.Fujisawa T., Hozumi H., Kono M. Prognostic factors for myositis-associated interstitial lung disease. PloS One. 2014;9(6):e98824. doi: 10.1371/journal.pone.0098824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hervier B., Meyer A., Dieval C. Pulmonary hypertension in antisynthetase syndrome: prevalence, aetiology and survival. Eur Respir J. 2013;42(5):1271–1282. doi: 10.1183/09031936.00156312. [DOI] [PubMed] [Google Scholar]
- 29.Solomon J., Swigris J.J., Brown K.K. Myositis-related interstitial lung disease and antisynthetase syndrome. J Bras Pneumol. 2011;37(1):100–109. doi: 10.1590/s1806-37132011000100015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lega J.C., Fabien N., Reynaud Q. The clinical phenotype associated with myositis-specific and associated autoantibodies: a meta-analysis revisiting the so-called antisynthetase syndrome. Autoimmun Rev. 2014;13(9):883–891. doi: 10.1016/j.autrev.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 31.Chen Z., Cao M., Plana M.N. Utility of anti-melanoma differentiation-associated gene 5 antibody measurement in identifying patients with dermatomyositis and a high risk for developing rapidly progressive interstitial lung disease: a review of the literature and a meta-analysis. Arthritis Care Res (Hoboken) 2013;65(8):1316–1324. doi: 10.1002/acr.21985. [DOI] [PubMed] [Google Scholar]
- 32.Hall J.C., Casciola-Rosen L., Samedy L.A. Anti-melanoma differentiation-associated protein 5-associated dermatomyositis: expanding the clinical spectrum. Arthritis Care Res (Hoboken) 2013;65(8):1307–1315. doi: 10.1002/acr.21992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Narang N.S., Casciola-Rosen L., Li S., Chung L., Fiorentino D.F. Cutaneous ulceration in dermatomyositis: association with anti-melanoma differentiation-associated gene 5 antibodies and interstitial lung disease. Arthritis Care Res (Hoboken) 2015;67(5):667–672. doi: 10.1002/acr.22498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Le Goff B., Cherin P., Cantagrel A. Pneumomediastinum in interstitial lung disease associated with dermatomyositis and polymyositis. Arthritis Rheum. 2009;61(1):108–118. doi: 10.1002/art.24372. [DOI] [PubMed] [Google Scholar]
- 35.Olson A.L., Swigris J.J., Sprunger D.B. Rheumatoid arthritis–interstitial lung disease–associated mortality. Am J Resp Crit Care Med. 2011;183:372–378. doi: 10.1164/rccm.201004-0622OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kelly C.A., Saravanan V., Nisar M. Rheumatoid arthritis-related interstitial lung disease: associations, prognostic factors and physiological and radiological characteristics—a large multicentre UK study. Rheumatology (Oxford) 2014;53(9):1676–1682. doi: 10.1093/rheumatology/keu165. [DOI] [PubMed] [Google Scholar]
- 37.Solomon J.J., Chung J.H., Cosgrove G.P. Predictors of mortality in rheumatoid arthritis-associated interstitial lung disease. Eur Respir J. 2016;47(2):588–596. doi: 10.1183/13993003.00357-2015. [DOI] [PubMed] [Google Scholar]
- 38.Giles J.T., Danoff S.K., Sokolove J. Association of fine specificity and repertoire expansion of anticitrullinated peptide antibodies with rheumatoid arthritis associated interstitial lung disease. Ann Rheum Dis. 2014;73(8):1487–1494. doi: 10.1136/annrheumdis-2012-203160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harlow L., Rosas I.O., Gochuico B.R. Identification of citrullinated hsp90 isoforms as novel autoantigens in rheumatoid arthritis-associated interstitial lung disease. Arthritis Rheum. 2013;65(4):869–879. doi: 10.1002/art.37881. [DOI] [PubMed] [Google Scholar]
- 40.Fingerlin T.E., Murphy E., Zhang W. Genome-wide association study identifies multiple susceptibility loci for pulmonary fibrosis. Nat Genet. 2013;45(6):613–620. doi: 10.1038/ng.2609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen J., Doyle T.J., Liu Y. Biomarkers of rheumatoid arthritis-associated interstitial lung disease. Arthritis Rheumatol. 2015;67(1):28–38. doi: 10.1002/art.38904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Doyle T.J., Patel A.S., Hatabu H. Detection of rheumatoid arthritis-interstitial lung disease is enhanced by serum biomarkers. Am J Respir Crit Care Med. 2015;191(12):1403–1412. doi: 10.1164/rccm.201411-1950OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Roubille C., Haraoui B. Interstitial lung diseases induced or exacerbated by DMARDS and biologic agents in rheumatoid arthritis: a systematic literature review. Semin Arthritis Rheum. 2014;43(5):613–626. doi: 10.1016/j.semarthrit.2013.09.005. [DOI] [PubMed] [Google Scholar]
- 44.Dixon W.G., Hyrich K.L., Watson K.D. Influence of anti-TNF therapy on mortality in patients with rheumatoid arthritis-associated interstitial lung disease: results from the British Society for Rheumatology Biologics Register. Ann Rheum Dis. 2010;69(6):1086–1091. doi: 10.1136/ard.2009.120626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Curtis J.R., Sarsour K., Napalkov P., Costa L.A., Schulman K.L. Incidence and complications of interstitial lung disease in users of tocilizumab, rituximab, abatacept and anti-tumor necrosis factor alpha agents, a retrospective cohort study. Arthritis Res Ther. 2015;17:319. doi: 10.1186/s13075-015-0835-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Arulkumaran N., Periselneris N., Gaskin G. Interstitial lung disease and ANCA-associated vasculitis: a retrospective observational cohort study. Rheumatology (Oxford) 2011;50(11):2035–2043. doi: 10.1093/rheumatology/ker236. [DOI] [PubMed] [Google Scholar]
- 47.Schirmer J.H., Wright M.N., Vonthein R. Clinical presentation and long-term outcome of 144 patients with microscopic polyangiitis in a monocentric German cohort. Rheumatology (Oxford) 2016;55(1):71–79. doi: 10.1093/rheumatology/kev286. [DOI] [PubMed] [Google Scholar]
- 48.Denton C.P., Hughes M., Gak N. BSR and BHPR guideline for the treatment of systemic sclerosis. Rheumatology (Oxford) 2016;55(10):1906–1910. doi: 10.1093/rheumatology/kew224. [DOI] [PubMed] [Google Scholar]
- 49.Hoffmann-Vold A.M., Aalokken T.M., Lund M.B. Predictive value of serial high-resolution computed tomography analyses and concurrent lung function tests in systemic sclerosis. Arthritis Rheumatol. 2015;67(8):2205–2212. doi: 10.1002/art.39166. [DOI] [PubMed] [Google Scholar]
- 50.Garin M.C., Highland K.B., Silver R.M., Strange C. Limitations to the 6-minute walk test in interstitial lung disease and pulmonary hypertension in scleroderma. J Rheumatol. 2009;36(2):330–336. doi: 10.3899/jrheum.080447. [DOI] [PubMed] [Google Scholar]
- 51.Richardson C., Agrawal R., Lee J. Esophageal dilatation and interstitial lung disease in systemic sclerosis: a cross-sectional study. Semin Arthritis Rheum. 2016;46(1):109–114. doi: 10.1016/j.semarthrit.2016.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Richeldi L., du Bois R.M., Raghu G. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071–2082. doi: 10.1056/NEJMoa1402584. [DOI] [PubMed] [Google Scholar]
- 53.King T.E., Jr., Bradford W.Z., Castro-Bernardini S. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2083–2092. doi: 10.1056/NEJMoa1402582. [DOI] [PubMed] [Google Scholar]
- 54.Khanna D., Albera C., Fischer A. An open-label, phase ii study of the safety and tolerability of pirfenidone in patients with scleroderma-associated interstitial lung disease: the LOTUSS Trial. J Rheumatol. 2016;43(9):1672–1679. doi: 10.3899/jrheum.151322. [DOI] [PubMed] [Google Scholar]
- 55.Andersson H., Sem M., Lund M.B. Long-term experience with rituximab in anti-synthetase syndrome-related interstitial lung disease. Rheumatology (Oxford) 2015;54(8):1420–1428. doi: 10.1093/rheumatology/kev004. [DOI] [PubMed] [Google Scholar]
- 56.Khanna D., Mittoo S., Aggarwal R. Connective tissue disease-associated interstitial lung diseases (CTD-ILD)—report from OMERACT CTD-ILD Working Group. J Rheumatol. 2015;42(11):2168–2171. doi: 10.3899/jrheum.141182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Saketkoo L.A., Mittoo S., Huscher D. Connective tissue disease related interstitial lung diseases and idiopathic pulmonary fibrosis: provisional core sets of domains and instruments for use in clinical trials. Thorax. 2014;69(5):428–436. doi: 10.1136/thoraxjnl-2013-204202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fischer A., Brown K.K., Du Bois R.M. Mycophenolate mofetil improves lung function in connective tissue disease-associated interstitial lung disease. J Rheumatol. 2013;40(5):640–646. doi: 10.3899/jrheum.121043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tashkin D.P., Elashoff R., Clements P.J. Cyclophosphamide versus placebo in scleroderma lung disease. N Engl J Med. 2006;354(25):2655–2666. doi: 10.1056/NEJMoa055120. [DOI] [PubMed] [Google Scholar]
- 60.Tashkin D.P., Roth M.D., Clements P.J. Mycophenolate mofetil versus oral cyclophosphamide in scleroderma-related interstitial lung disease (SLS II): a randomised controlled, double-blind, parallel group trial. Lancet Respir Med. 2016;4(9):708–719. doi: 10.1016/S2213-2600(16)30152-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Oldham J.M., Lee C., Valenzi E. Azathioprine response in patients with fibrotic connective tissue disease-associated interstitial lung disease. Respir Med. 2016;121:117–122. doi: 10.1016/j.rmed.2016.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Del Papa N., Onida F., Zaccara E. Autologous hematopoietic stem cell transplantation has better outcomes than conventional therapies in patients with rapidly progressive systemic sclerosis. Bone Marrow Transplant. 2017;52(1):53–58. doi: 10.1038/bmt.2016.211. [DOI] [PubMed] [Google Scholar]
- 63.Kloth C., Maximilian Thaiss W., Preibsch H. Quantitative chest CT analysis in patients with systemic sclerosis before and after autologous stem cell transplantation: comparison of results with those of pulmonary function tests and clinical tests. Rheumatology (Oxford) 2016;55(10):1763–1770. doi: 10.1093/rheumatology/kew259. [DOI] [PubMed] [Google Scholar]
- 64.Courtwright AM, El-Chemaly S, Dellaripa PF, Goldberg HJ. Survival and outcomes after lung transplantation for non-scleroderma connective tissue-related interstitial lung disease [published online ahead of print December 31, 2016. J Heart Lung Transplant. http://dx.doi.org/10.1016/j.healun.2016.12.013. [DOI] [PubMed]
- 65.Eberlein M., Mathai S.C. Lung transplantation in scleroderma. Time for the pendulum to swing? Ann Am Thorac Soc. 2016;13(6):767–769. doi: 10.1513/AnnalsATS.201512-801ED. [DOI] [PubMed] [Google Scholar]