Abstract
Integration of multiple cellular signals provides new opportunities in understanding oxygen sensing and response mechanisms in plants.
Plants grow in a dynamic environment and continuously face numerous stress conditions in parallel. This fluctuating environment pushed the evolution of extensive metabolic flexibility (Sweetlove et al., 2010; van Dongen et al., 2011). Flooding is an example of an environmental condition that combines many different stress factors simultaneously, including limited gas diffusion (e.g. of oxygen, carbon dioxide, and ethylene), temperature stress, light quality and quantity, and osmotic and oxidative stress. Changes in the internal concentration of oxygen, but also e.g. ethylene, are crucial early signals for the initiation of adaptation strategies to flooding (Sasidharan and Voesenek, 2015; Sasidharan et al., 2017). Initially, a plant cell responds to nearly all these different types of stresses by using the same set of second messengers (such as reactive oxygen species [ROS], calcium, nitric oxide [NO], and lipid molecules) and by activating signaling cascades through kinases (Bjornson et al., 2016; Zhu, 2016). This raises the question of how cells detect and differentiate between stresses.
One fundamental aim in low-oxygen research is to unravel how organisms detect and react to the lack of oxygen and respond to an impairment of their energy metabolism (Ratcliffe, 2013). In both plants (Licausi et al., 2011a; Gibbs et al., 2011, 2014; Weits et al., 2014; White et al., 2017) and animals (Loenarz and Schofield, 2011; Ratcliffe, 2013; Bishop and Ratcliffe 2014; Hamanaka et al., 2016), key transcriptional regulators have been identified that regulate the hypoxic response via the integration of cellular signals that are required for their activation (Box 1). Here, we discuss the nature and origin of these various signals under hypoxia that initiate adaptation responses at the transcriptional and (post-) translational level and their potential integration points. In addition, we postulate that it is the integration of different signal inputs that defines a response specific to oxygen limitation. Further, we will discuss the state of current methods and technologies to measure plant internal oxygen concentrations.
THE CONCEPT OF AN INTEGRATIVE OXYGEN SENSING MECHANISM
Abiotic stress perception appears not to rely on the detection of only one specific effector or ligand. Despite all efforts to reveal the primary sensors for diverse abiotic stresses, unequivocal identification remains arduous (Zhu, 2016). In Arabidopsis (Arabidopsis thaliana), the N-end rule pathway of targeted proteolysis was shown to act as a mechanism that controls protein levels of subgroup VII ETHYLENE-RESPONSE FACTOR (ERFVII) transcription factors by regulating their protein turnover in an oxygen-concentration-dependent manner (Gibbs et al., 2011; Licausi et al., 2011a; Box 1). Remarkably, protein abundance of RELATED TO APETALA2.12 (RAP2.12), an ERFVII member in Arabidopsis, is also positively regulated by HYDRAULIC CONDUCTIVITY OF ROOT1 (HCR1), a Raf-like MAPKKK, which could phosphorylate RAP2.12 in vitro. This N-end rule-independent stabilization mechanism is proposed to allow the integration of cation (specifically potassium) availability with the oxygen concentration in the soil (Shahzad et al., 2016).
Low-oxygen stress impairs diverse plant cellular functions, including those in the cytosol, nucleus, mitochondria, and endoplasmic reticulum (ER). Actually, molecular oxygen is among the most-utilized molecules in eukaryotic cellular metabolism (Raymond and Segrè, 2006) with more than 350 oxygen-dependent reactions. Thus, decreasing oxygen levels might be indirectly sensed due to an impairment of various metabolic pathways when the cellular oxygen concentration changes around or below the Michaelis-Menten constant KM for oxygen of the enzymes involved. As such, any oxygen-dependent enzyme has the potential to act as a kind of sensor, suggesting simultaneous oxygen sensing at multiple sites of the cell. Then again, it might well be that one or a few oxygen-concentration-dependent enzymes evolved to primarily monitor aerobic metabolism.
Taken together, three scenarios of cellular oxygen sensing can be discussed: (1) decentralized sensing at many sites of the cell by e.g. oxygen-consuming enzymes, (2) a primary “oxygen sensor” detects a drop in oxygen to initiate all signaling cascades, and (3) a combination of scenario 1 and 2. A primary oxygen sensor (scenario 2) would detect the cellular oxygen concentration but not the oxygen availability. However, differentiation between “concentration” and “availability” is important, since a low oxygen concentration can result in a high flux of oxygen into the tissue and does not necessarily lead to low-oxygen stress. The advantage of a decentralized oxygen-sensing mechanism (scenario 1) would be that it allows for fine-tuning the response to low-oxygen conditions to a particular situation. By adding up various oxygen-concentration-related signals, such integrative signaling would trigger hypoxic responses only when the actual oxygen concentration does indeed disturb cellular homeostasis. The decentralized oxygen-sensing model further suggests that perturbations of processes in various organelles, at the plasma membrane and in the cytosol can initiate low-oxygen-stress signaling and that these various triggers must be integrated to activate appropriate transcriptional reprogramming and cellular adaptation responses. A specific balance between multiple stress-induced signals could provide a mechanism—like a “cellular fingerprint”—to discriminate between different stress types.
In this review, we summarize signaling pathways from different cellular compartments that are initiated by low-oxygen stress and discuss potential integration points for the multiple signal inputs resulting in hypoxia-specific adaptive responses.
N-END RULE PATHWAY
In Arabidopsis, members of the ERFVII transcription factor family (Box 1) contain a conserved N-terminal domain that makes them oxygen- and NO-dependent substrates of the N-end rule pathway of targeted proteolysis (Gibbs et al., 2015).
While the N-end rule is acting as a safeguard mechanism to limit ERFVII abundance under aerobic conditions, several observations suggest that its control is limited to certain Met-Cys proteins, environmental conditions, and developmental phases of the plant. Arabidopsis N-end rule mutant seedlings show constitutive expression of about half of the core hypoxia-responsive genes (Gibbs et al., 2011), indicating that the other hypoxia genes are not controlled by this pathway. In addition, introduction of a GUS reporter driven by the ALCOHOL DEHYDROGENASE1 (ADH1) promoter in Arabidopsis wild-type seedlings or a proteolysis6 (prt6) mutant defective in ERFVII turnover revealed constitutive activation of the promoter in certain tissues of the prt6 mutant, but not in all as compared to the wild-type. Interestingly, in both the wild-type and the prt6 mutant an increased GUS signal was observed after 6 h of hypoxia (Gibbs et al., 2011). Thus, in N-end rule mutants, the hypoxic response is not entirely saturated, nor do mutations in this pathway affect all plant tissues in a similar manner. While constitutive expression of a subset of hypoxic genes is observed in N-end rule mutant seedlings, a recent report revealed that this constitutive expression is less pronounced in adult mutant plants (Giuntoli et al., 2017). As submergence of rosette-stage plants just prior to bolting still results in the activation of hypoxic genes, it strongly argues for additional, N-end rule-independent mechanisms that control either ERFVII activity or alternative transcriptional cascades. In addition, at least one ERFVII, SUBMERGENCE1 A (SUB1A) from rice (Oryza sativa), is not a target of the N-end rule pathway, although it possesses the N-terminal consensus sequence as found in Arabidopsis ERFVIIs (Gibbs et al., 2011). This further suggests alternative regulatory pathways.
MITOCHONDRIAL DYSFUNCTION AS TRIGGER FOR HYPOXIA SIGNALING
Limited oxygen availability affects respiration, which impacts on a whole palette of mitochondrial parameters and processes (see Box 2), including reactive oxygen and nitrogen species (ROS/RNS) homeostasis, redox status of NAD(P)H and antioxidant pools, ATP/ADP ratio, the proton-motive force, and calcium and metabolite levels, which in principle could all trigger mitochondrial signaling (Schwarzländer and Finkemeier, 2013). Indeed, roles for these diverse signals in mitochondrial retrograde responses have been established and are discussed by Wagner et al. (2018) in this Focus Issue. The role of mitochondrial retrograde signals in hypoxia has been partly investigated; for example, mitochondrial ROS signals were shown to impact on anoxia tolerance (Chang et al., 2012). Likewise, in animals, mitochondrial electron transport chain (mETC)-derived ROS signals appear to provoke adaptation responses upon hypoxia (Hamanaka et al., 2016). Interestingly, complex III inhibition under aerobic conditions by antimycin-A treatment in part causes induction of hypoxia-responsive genes in Arabidopsis (Ng et al., 2013a), which could be due to disruption of the ATP/ADP ratio and/or increased mitochondrial ROS production. Furthermore, metabolic signals derived from mitochondria might be integrated into cellular reprogramming during hypoxia. This is supported by the notion that overexpression of the mitochondrial UNCOUPLING PROTEIN1 (UCP1), which decreases stress-induced ROS production, results in a hypoxic metabolic pattern and transcriptomic signature (Barreto et al., 2016). UCP proteins in animals have been linked to the induction of hypoxic responses (Dromparis et al., 2013), suggesting a universal role of this protein family in controlling low-oxygen responses.
An important feature of plant mitochondria is that the proton-translocating complexes I, III, and IV (see Box 2) are capable of forming so-called supercomplexes (Dudkina et al., 2005). Remarkably, mitochondrial supercomplex assembly is oxygen dependent, as in potato tubers, low oxygen promotes dissociation of complex I from the supercomplex, while reassembly is observed under reoxygenation (Ramírez-Aguilar et al., 2011). Supplying isolated mitochondria with succinate under a low pH also caused dissociation of complex I as observed for low-oxygen conditions. Substrate-dependent supercomplex formation was also found in mouse cells, in which the plasticity of the mETC complexes appears to be used to fine-tune respiration (Lapuente-Brun et al., 2013). That said, it still needs to be demonstrated that supercomplex formation also affects respiration rates in plant mitochondria. Taken together, the release of complex I from the supercomplex does not appear to be the initial trigger for hypoxic responses in plants but is already part of the adaptation response that is initiated by a drop in pH and a change in substrate availability.
When the oxygen concentration is reduced below a certain threshold, respiration is impaired (Nikoloski and van Dongen 2011). However, it remains to be determined at what subcellular oxygen concentration mitochondrial dysfunction is initiated. Precise determination of the mitochondrial oxygen concentration is especially important, as the affinity for oxygen of Cytochrome c Oxidase likely depends on cellular conditions such as pH, redox status, and oxygen concentration (Krab et al., 2011). Also, it is to be determined whether this respiratory impairment is specific to hypoxia or if other abiotic stresses cause similar mitochondrial constraints, too. UCPs, TYPE II NAD(P)H DEHYDROGENASEs, and ALTERNATIVE OXIDASE (AOX) as part of the mitochondrial energy dissipation pathways (see Box 2; Maxwell et al., 1999; Geisler et al., 2007; Millar et al., 2011) have roles in the response to diverse abiotic and biotic stresses, which in general cause an energy crisis (Pu et al., 2015; Zhu, 2016). Beyond that, mitochondrial ROS production with signaling function can be observed under salt or drought stress (Hernandez et al., 1993; Pastore et al., 2007), indicating that stress signals released upon mitochondrial dysfunction are not hypoxia specific per se. A fundamental biological question that needs further attention is how cells distinguish between hypoxia-induced mitochondrial signals and those induced by other stresses.
THE ER AS SIGNALING HUB
Protein folding in the ER is essential for the stability and functionality of many secreted and membrane-bound proteins. Although the ER employs different proteins and pathways for this task, it is inevitable that some proteins get misfolded especially during stress (Hegde and Ploegh, 2010). ER stress results in the activation of the unfolded protein response, which enhances the function of the protein folding machinery and the protein quality control system, see Liu and Howell (2016) for a detailed overview. The major pathway for oxidative protein folding is present in the ER and relies on protein disulfide isomerase (PDI) and ER thiol oxidase (ERO) proteins (Aller and Meyer; 2013). PDI oxidizes proteins and is regenerated by ERO through the transfer of electrons from PDI to molecular oxygen. The KM of the ERO protein from yeast was found to be 4–10 µm for oxygen, which is similar to that of AOX in plants (Gross et al., 2006). If plant ERO1 proteins display a similar KM as their yeast counterpart for oxygen, then they would be functionally impaired under hypoxia and might act as sensors for low-oxygen conditions. Interestingly, only the disulfide bond formation during the folding phase in the ER involving EROs was shown to be oxygen dependent in mammalian cells, while subsequent glycosylation, ER export, Golgi processing, and transit to the extracellular space were not (Koritzinsky et al., 2013). As oxygen-dependent folding is directly impaired upon low-oxygen stress, it has been suggested that the ER constitutes a cellular oxygen sensor in animals that contributes to signaling and survival during hypoxic conditions (Wouters and Koritzinsky, 2008). Future work is required to understand if this putative ER pathway also plays a role in plants. Of note, in mitochondria, oxidative protein folding relies on a flavin adenine dinucleotide (FAD)-dependent thiol oxidase that acts in an oxygen-dependent manner (Ang et al., 2014), suggesting that other protein folding pathways might have a role in low-oxygen stress.
Interestingly, the ER also represents an important crossroad for mitochondrial stress signals. Disturbance of mitochondrial function results in a signal to the nucleus, the so-called mitochondrial retrograde regulation (MRR; Rhoads and Subbaiah 2007). MRR relies on both ROS and calcium signals, and recently, several of the protein components of the pathway were uncovered. Transmembrane-domain-containing NO APICAL MERISTEM/ARABIDOPSIS TRANSCRIPTION ACTIVATION FACTOR/CUP-SHAPED COTYLEDON (NAC) transcription factors were found to act as important modulators of the transcriptional response during MRR (De Clercq et al., 2013; Ng et al., 2013b). Both ANAC013 and ANAC017 mediate MRR responses but are located at the ER, indicating that mitochondrial signals are received at the ER. In this case, the mitochondrial signal is proposed to activate a yet-unknown protease that releases the NAC transcription factors from the ER membrane, which subsequently travel to the nucleus to modulate expression. Many of the MRR-induced genes are also hypoxia responsive, including the ERFVII transcription factor gene HYPOXIA RESPONSIVE ERF2, (HRE2), encoding a regulator of anaerobic responses in Arabidopsis (Licausi et al., 2010). Therefore, it would be worthwhile to analyze the contribution of ER-mediated MRR to low-oxygen tolerance in plants.
Energy Signaling
The harmful character of low-oxygen stress lies in its impact on oxidative phosphorylation in the mitochondrion, which delivers energy equivalents in the form of ATP (Millar et al., 2011). However, a low local oxygen concentration does not necessarily lead to an energy crisis when the flux of oxygen toward the mitochondria can keep pace with respiratory oxygen consumption. Therefore, it is of importance that any changes in the ATP-producing processes within mitochondria are communicated to other compartments to induce appropriate adaptive responses. By doing so, differentiation is required between (1) an energy crisis that is induced by mitochondrial dysfunction due to hypoxia, and (2) a low-energy status that is caused by high ATP consumption due to metabolic activity. Only sensing cellular oxygen levels without taking into account the actual energy charge of the cell could provoke cost-intensive adaptation responses in tissues with high respiratory activity. For detailed discussion of mitochondrial energy signaling, we would like to refer to the Update of Wagner et al. (2018) in this Focus Issue.
Eukaryotic energy sensing is largely conserved and involves the two kinases Suc nonfermenting1-related kinase1 (SnRK1) and target of rapamycin (TOR) in both plants and animals (Broeckx et al., 2016). Plant SnRK1 is activated in response to energy-depleting stress conditions to promote plant survival. Loss of SnRK1 function impairs submergence tolerance in plants (Cho et al., 2012). It is important to note that SnRK1 appears to sense the AMP/ATP ratio and ADP/ATP ratio in cells, linking its activation directly to an energy crisis (Hardie, 2011). Many different stresses can cause an energy crisis; not surprisingly, most of the core anaerobic genes are not hypoxia specific (van Dongen and Licausi, 2015).
Stresses that provoke a cellular “energy crisis” evoke translational repression, which has also been observed during hypoxia (Branco-Price et al., 2008; Mustroph et al., 2009). In eukaryotic cells, growth in response to energy availability is in part controlled by the TOR protein kinase (Dobrenel et al., 2016). TOR controls translation initiation and has been shown to be inactivated in mammalian cells upon hypoxia (Wouters and Koritzinsky, 2008). In plants, a picture has emerged in which TOR kinase is active under favorable energy conditions, while SnRK1 is stimulated upon energy limitation (Tomé et al., 2014). Moreover, TOR and SnRK1 appear to act antagonistically. So far, the function of SnRK1 under low-oxygen conditions in plants is well established; however, a role for TOR is still unexplored.
ROS Signaling
Cellular imbalances as a result of abiotic stress cause a change in ROS homeostasis. In addition, during initial stress exposure, a so-called oxidative burst is observed, which acts as a signal to initiate adaptive responses (Schippers and Schmidt, 2016). Although counterintuitive, mitochondrial ROS function in retrograde signaling during oxygen deprivation to adjust energy management in yeast and animal cells (Guzy et al., 2007; Hamanaka et al., 2016). Also in plants, a mitochondrial ROS burst is observed upon a drop in oxygen levels (Chang et al., 2012). As application of antimycin-A mimics this burst, it is suggested that superoxide (O2−) derived from complex III might act as a signaling molecule during mitochondrial dysfunction upon oxygen deprivation. In order to prevent the build-up of high levels of superoxide, mitochondria actively scavenge it by employing MANGENESE SUPEROXIDE DISMUTASE (MnSOD). The hydrogen peroxide (H2O2) produced during this reaction either oxidizes mitochondrial proteins or exits the organelle to induce downstream signaling responses. To exit mitochondria, H2O2 would rely on aquaporins (Bienert et al., 2007).
Within the cytosol, ROS as signaling molecules are rather short lived due to the high cytosolic antioxidant capacity. Moreover, to provide mitochondrial specificity to the ROS signal, sensing and translation of this signal is expected to occur near the mitochondrial surface. So far, release of H2O2 by mitochondria during oxygen deprivation into the cytosol has been linked to the activation of MITOGEN-ACTIVATED PROTEIN KINASE3 (MPK3) and MPK6 in Arabidopsis (Chang et al., 2012). The fact that these MPKs are involved in a broad range of biotic and abiotic stress pathways (Rodriguez et al., 2010) implies that they are integrated into local sensory complexes to maintain ROS-source information to activate stress-specific responses. As loss-of-function mutants of either MPK3 or MPK6 do not largely affect the activation of anaerobic genes (Chang et al., 2012), additional components in the mitochondrial ROS-signaling pathway related to low-oxygen stress await to be uncovered. Here, it is of interest to note that mitochondria are often in direct contact with the ER (Mueller and Reski, 2015), which therefore might be involved in relaying or translating the mitochondrial ROS signal.
Next to the transient mitochondrial oxidative burst upon oxygen deprivation, a plasma membrane-localized burst occurs through activation of calcium-dependent NADPH oxidases (Blokhina et al., 2001; Baxter-Burrell, 2002). As RESPIRATORY BURST OXIDASE HOMOLOG (RBOH)-dependent ROS production occurs during nearly every stress, it remains unclear how such signals can be rendered specific for low-oxygen stress.
Arabidopsis contains 10 NADPH oxidases, called RBOHs, of which three—RBOHD, RBOHF, and RBOHI—have been implicated in low-oxygen responses (Pucciariello et al., 2012; Liu et al., 2017; Lin et al., 2017). Although fermentative genes including ADH1 have been shown to depend on single RBOH activity, the exact mechanism through which each RBOH contributes to low-oxygen signaling needs further investigation. Recently, RBOHI activity providing submergence tolerance could be linked to auxin signaling (Lin et al., 2017). NADPH oxidases are activated by RHO-LIKE SMALL G-PROTEIN OF PLANTS (ROP) and calcium-dependent kinases (Baxter-Burrell et al., 2002; Suzuki et al., 2011). ROP promotes the oxidative burst during oxygen deprivation by activating NADPH oxidases. The burst is transient, as H2O2 activates ROP GTPASE ACTIVATING PROTEIN4 (RopGAP4), which deactivates ROP signaling (Baxter-Burrell et al., 2002). Plants lacking RopGAP4 show impaired low-oxygen tolerance. Recently, it was found that the ERFVII transcription factor RAP2.12 activates the expression of HYPOXIA RESPONSIVE UNIVERSAL STRESS PROTEIN1 (HRU1), which, like RopGAP4, acts to dampen NADPH oxidase activity (Gonzali et al., 2015). Potentially, RopGAP4 and HRU1 not only limit the length of the initial apoplastic ROS burst but might be involved in protecting the plants from an oxidative burst upon reoxygenation.
Studies on mammalian cells suggest a feed-forward loop between the mitochondrial ROS burst and the activation of NADPH oxidases (Kröller-Schön et al., 2014). So far, this concept has not been studied in plant cells, but it would be intriguing to be investigated further.
Calcium Signaling
NADPH oxidase-driven ROS production depends on calcium, while ROS in turn can modulate calcium uptake into the cytosol (Mazars et al., 2010; Suzuki et al., 2011), which lets both second messengers be part of a tightly linked signaling cross-talk. Stress induces an increase in cytoplasmic calcium levels, which activates diverse calcium-binding proteins that decode the calcium signal into a cellular and transcriptional response (Dodd et al., 2010). Among these calcium-activated proteins are kinases, enzymes, and transcription factors. The increase in cytosolic calcium levels occurs through the release of calcium from the apoplast and/or cellular organelles (Edel et al., 2017).
Early anoxia studies revealed a rise in cytosolic calcium levels by using an AEQUORIN-based reporter in Arabidopsis (Sedbrook et al., 1996). Upon oxygen deprivation, a biphasic calcium response was observed, with a first initial short-lived calcium spike occurring within minutes of anoxia, followed by a second, prolonged calcium response that lasted several hours. In addition, the authors showed that inhibition of calcium fluxes by calcium channel blockers resulted in a partial inhibition of ADH1 expression under anoxia. Also in maize, the use of such blockers impairs the expression of ADH1 under low-oxygen stress (Subbaiah et al., 1994). Still, the extent to which calcium fluxes and signaling events influence anaerobic gene expression is largely unexplored in plants.
Cytosolic calcium homeostasis is rapidly restored after a calcium spike by active transport mechanisms (Sanders et al., 2002). In plants, energization relies either on calcium pumps powered by ATP hydrolysis or by Ca2+/H+ antiporters powered by a proton-motive force (Dodd et al., 2010). The energy-consuming transport of calcium is essential to obtain the basal levels, against which calcium release channels operate transiently to elevate free cytosolic calcium levels upon a stress stimulus. Thus, after the cytosolic calcium spike, prestress levels need to be restored to keep the system sensitive toward a follow-up transient increase in calcium (Bose et al., 2011). These properties suggest that calcium signaling might be restricted to the initial phase of low-oxygen stress, as increasing ATP constrains and the pH change under prolonged stress conditions might impair calcium homeostasis mechanisms.
The origin of calcium released into the cytosol under low-oxygen stress in plants is partly explored. Both plasma membrane calcium channel blockers as well as intracellular calcium channel blockers in part block calcium-dependent hypoxic responses (Sedbrook et al., 1996; Yemelyanov et al., 2011). Potential intracellular calcium sources for the cytosolic spike during oxygen deprivation might be the vacuole, the ER, and mitochondria. Although roles for ER and mitochondria in cytosolic calcium release have been described in mammalian cells (Rizzuto et al., 2012; Wiel et al., 2014), it is still unclear to which extent these organelles contribute to calcium signaling under abiotic stresses in plants.
In rice, CALCINEURIN B-LIKE INTERACTING PROTEIN KINASE15 (OsCIPK15) has been shown to be required for the expression of genes encoding glycolytic enzymes, including ADH (Lee et al., 2009; Meyer et al., 2010). Excitingly, OsCIPK15 is activated by SnRK1, linking it to the energy-sensing network. Energy saving as an adaptation response to low-oxygen stress can be achieved by controlled selective translation of mRNA (Sorenson and Bailey-Serres, 2014), a process that is regulated in a calcium-dependent manner. CALMODULIN38 (CML38) from Arabidopsis acts as a potential calcium sensor that colocalizes under low-oxygen stress to mRNA ribonucleoprotein particles,. including stress granules. The latter are formed under hypoxia to serve as storage of translationally inhibited mRNA (Lokdarshi et al., 2016).
Taken together, despite the unquestionably eminent role of calcium in the initial response to low-oxygen stress, major gaps exist regarding the regulation of calcium fluxes, homeostasis, and the role of specific calcium sensor and signaling proteins in adaptation to hypoxia.
MEASURING OXYGEN CONCENTRATIONS IN PLANTS
A major bottleneck for the research on low-oxygen stress is that accurate measurements of in situ oxygen concentrations in plants are difficult. Tools and methods to resolve the dynamic variation in oxygen levels with spatial and temporal resolution are needed for a better understanding of low-oxygen signaling at the cellular level.
Over the years, numerous methods for accurate measurement of oxygen have been developed (Box 3). Invasive oxygen measurements using Clark-type oxygen electrodes and optical oxygen sensors are commonly used and led to our current understanding on the dynamics and distribution of oxygen in plants. Still, the development of noninvasive oxygen measurement methods in plants is largely lacking behind as compared to the mammalian field, were cancer research has driven the development of a broad range of methods for the detection of hypoxia within animal tissue, such as soluble oxygen sensitive fluorescent dyes, genetically encoded sensors, positron emission tomography (PET), and 18F NMR (NMR) (Zhang et al., 2010; Dmitriev and Papkovsky, 2015; Fleming et al., 2015).
Clark-type electrodes are based on the diffusion of oxygen through a membrane to the oxygen-reducing cathode, which generates a current that is proportional to the oxygen concentration (Clark et al., 1953). A disadvantage is that oxygen is consumed at the cathode. For the measurements of oxygen in tissue, this drawback has been partially overcome by the development of miniaturized microsensors, which consume significantly less oxygen. These sensors, for which commercially available models are available with a tip diameter of 5 to 10 µm, are also significantly less invasive compared to larger models. Despite the oxygen consumption and destructiveness of the Clark-type electrodes, they have been widely used and have been employed to measure the oxygen concentration in numerous plants such as rice, seagrass, green alga cultures, and in the roots of several wetland plant species (Melis et al., 2000; Visser et al., 2000; Binzer et al., 2005; Colmer and Pedersen, 2008).
Optical oxygen sensors are popular due to their wide possible applications and their lack of oxygen consumption during the measurements. The majority of oxygen optodes are based on the quenching of a luminescent probe by oxygen, called phosphorescence quenching, resulting in increased phosphorescence when the oxygen concentration decreases. Oxygen-sensitive phosphorescent dyes can be coated on the inside of culture flasks or microplates to measure the oxygen concentration in a sample over time. Another application includes the creation of thin films coated with an oxygen-sensitive fluorescent dye, which was successfully used to map the oxygen concentration in plant roots (Rudolph-Moor et al., 2012). Micro-optodes, which consist of an optical fiber with a tip coated by an oxygen-sensitive probe can be used to measure the oxygen concentration within plant tissue (Licausi et al., 2011b). However, insertion of micro-optodes into the tissue is destructive and facilitates oxygen diffusion into the sample, which can lead to faulty measurements.
Genetically encoded oxygen reporters provide a potential solution to overcome the drawback associated with the invasiveness of Clark-type electrodes and micro-optode oxygen sensors (Ast et al., 2012). Several genes have been identified that display specific and strong up-regulation upon low oxygen (Mustroph et al., 2010). The expression of these genes may therefore be used as an indication of underlying hypoxic conditions. Activation of promoters of hypoxia-inducible genes, including HYPOXIA RESPONSE ATTENUATOR1, PLANT CYS OXIDASE1 (PCO1), and PCO2 has been clearly demonstrated (Chung and Ferl, 1999; Giuntoli et al., 2014; Weits et al., 2014; Gasch et al., 2016).
OXYGEN MEASUREMENTS IN NONPLANT ORGANISMS
Oxygen-sensitive phosphorescence probes are used to measure the intracellular oxygen concentration in different animal tissues (Vikram et al., 2007). The probes are either directly injected into the desired tissue or taken up by the tissue through passive self-loading (Dmitriev and Papkovsky, 2015). Afterward, the phosphorescence signal is imaged using a fluorescent microscope to map the oxygen concentration.
Several parameters need to be examined before phosphorescent probes can be applied in plants, e.g. sensitivity to pH, stability, toxicity, and probe distribution. Systemic distribution is another roadblock, as several dyes are injected intravenously in animals and rely on transport via the cardiovascular system to the tissue. A red-light-emitting iridium-based probe Ir(btp)2(acac) (BTP) was developed and used to image tumor hypoxia in vivo (Zhang et al., 2010). The phosphorescence of the probe is quenched by oxygen, but when the concentration drops below 5% v/v O2 the signal increases. However, cellular uptake of BTP appears to be albumin-mediated, and its uptake into plant cells may therefore not occur efficiently. Modification of these oxygen probes, such as the addition of hydrophilic groups or domains that interact with transport proteins, may facilitate uptake by the cell. Importantly, as chlorophyll displays red autofluorescence, phosphorescent oxygen probes for plants should be chosen with distinct emission spectra.
For studying the oxygen concentration in bacteria, a genetically encoded fluorescent protein-based biosensor for oxygen was established (Potzkei et al., 2012). This FRET-based probe contains an oxygen-sensitive yellow fluorescent protein (YFP) coupled with a hypoxia-insensitive flavin-binding fluorescent protein (FbFP). Maturation of the YFP chromophore requires oxygen and therefore FRET efficiency from the FbFP donor to the YFP acceptor depends on the oxygen availability. Remarkably, the YFP/FbFP emission ratio could be calibrated to the dissolved oxygen concentration in a range from 0% to 33%, allowing a precise quantification of the intracellular oxygen concentration.
PET has been widely applied to image hypoxia within tumors in mammals. PET-based detection of oxygen is based on the imaging of 18F-containing imidazoles, which passively diffuse in to cells and bind strongly to macromolecules of hypoxic cells (Fleming et al., 2015). This can be combined with x-ray computed tomography to provide additional anatomical information of the tissue, allowing precise mapping of the oxygen concentration within the tissue (Krause et al., 2006). Another minimally invasive method to determine oxygen content in mammalian tissues is 19F (NMR). This spectroscopic technique is based on the measurement of the spin lattice relaxation rate of perfluorocarbons, which has a linear relation to the tissue oxygen concentration. Another minimally invasive method to determine oxygen content in mammalian tissues is Fluorine-19 nuclear magnetic resonance (19F NMR). 19F NMR allows imaging of the absolute oxygen concentration in tissue (Yu et al., 2005). Imaging of the oxygen concentration using PET or NMR is not limited by the thickness of the issue, and it therefore represents an effective technique to map the oxygen concentration in a noninvasive manner of thick or difficult-to-access tissue. PET and NMR-based oxygen measurements may also prove useful for plants; however, their use in mammals relies on the injection of probes in to the blood stream, and their distribution in plant tissue must therefore be carefully tested.
CONCLUSION
Oxygen deprivation affects diverse organelles, which give rise to unique stress signals that must be processed and integrated to initiate an appropriate adaptive response of the plant cell. The diverse and widespread consequences of a drop in oxygen for cellular homeostasis strongly support a view in which oxygen sensing does not occur through one primary sensor but happens decentralized and simultaneously at multiple sites in the cell (Figure 1).
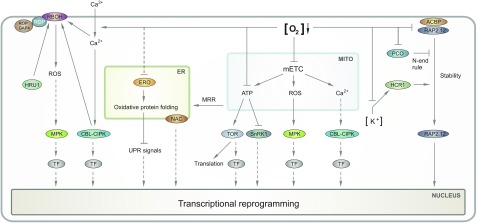
Figure 1.
Integration of multiple low-oxygen stress-signaling pathways from diverse cellular origins. According to this model, signaling to the nucleus occurs through various pathways. From left to right, low-oxygen stress results in an NADPH-oxidase-derived oxidative burst, which eventually leads to posttranslational activation of diverse transcription factors (TFs). The influx of extracellular calcium stimulates a signaling cascade involving CALCINEURIN-B-LIKE PROTEINs (CBLs) and CBL-INTERACTING PROTEIN KINASEs (CIPKs) and the activation of NADPH oxidases, called RESPIRATORY BURST OXIDASE HOMOLOGs (RBOHs). RBOHs are in addition stimulated to produce ROS by interacting with RHO-LIKE SMALL G-PROTEINS OF PLANTS (ROP). The resulting ROS burst is subsequently dampened through the action of ROP GTPase ACTIVATING PROTEIN4 (RopGAP4), which targets ROP, as well as by HYPOXIA-RESPONSIVE UNIVERSAL STRESS PROTEIN1 (HRU1), which forms a complex with ROP and RBOH. Furthermore, oxygen limitation might hamper oxidative protein folding in the endoplasmic reticulum (ER) through impairing the activity of ER THIOL OXIDASE (ERO), which subsequently results in an unfolded protein response (UPR). In addition, low-oxygen stress causes mitochondrial dysfunction, which is communicated to the ER via mitochondrial retrograde regulation (MRR). Dormant NAC TFs at the ER are proteolytically released to transduce the MRR signal to the nucleus. Further, impairment of the mitochondrial electron transport chain (mETC) is followed by a mitochondrial oxidative burst and potentially an efflux of stored calcium into the cytosol. The oxidative burst results in the activation of MITOGEN-ACTIVATED PROTEIN KINASE3 (MPK3) and MPK6, which may regulate hypoxic gene expression by phosphorylation of transcriptional regulators. Disturbance of the mETC further leads to a drop in ATP levels, which is sensed by the kinases TARGET OF RAPAMYCIN (TOR) and SUC NONFERMENTING1-RELATED KINASE1 (SnRK1), whereby TOR is inactivated, while Snrk1 is induced under ATP limitation. Stabilization of RELATED TO APETALA2.12 (RAP2.12) and other subgroup VII ERF TFs occurs through inactivation of the N-end rule pathway under hypoxic conditions. Under aerobic conditions, this active degradation pathway involves oxygen-dependent oxidation of the TF proteins that is catalyzed by PLANT CYS OXIDASE (PCO). Additionally, RAP2.12 protein abundance is increased by HYDRAULIC CONDUCTIVITY ROOT1 (HCR1), a Raf-like MAPKKK which targets the TF under oxygen-limiting and K+-sufficient conditions. Finally, integration of the diverse subcellular signal inputs most likely occurs at the level of TFs, which are potentially regulated by posttranslational modification and induce transcriptional reprogramming resulting in hypoxia adaptation.
At the transcriptional level, at least 120 transcription factors are differentially expressed upon low-oxygen stress in Arabidopsis (Licausi et al., 2011c). Still, the roles for most of these transcription factors or the kind of signals they perceive upon low-oxygen stress are unknown. So far, it is unclear which signals activate ERFVIIs upon low-oxygen stress in Arabidopsis. The non-N-end rule ERFVII target SUB1A of rice was shown to be a MPK phosphorylation target, (Singh and Sinha, 2016), suggesting posttranslational signal integration. Interestingly, MPK3/6 activity in Arabidopsis is induced by a mitochondrial signal upon oxygen deprivation (Chang et al., 2012). Also, transcription factor targets of the SnRK1 kinase might play a crucial role in oxygen signaling (Baena-González et al., 2007). Furthermore, mitochondrial- or ER-to-nucleus signaling was shown to rely on transcription factors that need to be proteolytically activated (Iwata et al., 2008; De Clercq et al., 2013). One of the main challenges in the future is to identify all transcription factors that act as signal integration points during low-oxygen stress, to decipher the signals they perceive and the mechanisms through which they act in a spatial and temporal manner.
To date, a number of retrograde signaling molecules have been described, including ROS, calcium, and diverse metabolites (Kmiecik et al., 2016). Still, it is hardly known how these signals act during low-oxygen stress. That said, the oxidative burst observed in mitochondria during oxygen deprivation was shown to activate MPKs (Chang et al., 2012). Potentially, calcium and ROS might act in synergy to form an integrative signal, as observed during salt stress (Evans et al., 2016). Organellar signaling does not only involve signaling to the nucleus but also between organelles (Kleine and Leister, 2016). Here, the question raises whether low oxygen-induced signaling of one organelle can be prioritized over another.
Abiotic stresses in general impair energy metabolism and provoke mitochondrial dysfunctioning, which cause the activation of SnRK and AOX. In addition, signaling molecules like calcium and ROS are vital to sense all different kinds of stresses. Despite this remarkable overlap in stress responses at the cellular and molecular level, cells can differentiate between stresses and respond specifically. To be able to do so, plants might be capable of reading stress signal signatures (i.e. ROS waves or calcium spikes) and sense the temporal and spatial context. In yeast and human cell systems, it was shown that cellular stress sensors act in scaffolds (Kang et al., 2004). In plants, the idea of signaling scaffolds has only recently started to emerge (Demir et al., 2013,). Future research (see “Outstanding Questions”) should place emphasis on the multiple signaling pathways that are activated by oxygen limitation, their downstream integration, and cross-talk to reveal how the adaptation to low-oxygen stress is fine-tuned and adjusted to a particular situation encountered by plants. This will also require the development of innovative methods to measure intracellular molecular oxygen levels with a high spatial and temporal resolution.
Footnotes
The research on abiotic stress responses in the van Dongen laboratory is financed by RWTH Aachen University and the German Science Foundation.
Articles can be viewed without a subscription.
References
- Aller I, Meyer AJ (2013) The oxidative protein folding machinery in plant cells. Protoplasma 250: 799–816 [DOI] [PubMed] [Google Scholar]
- Ang SK, Zhang M, Lodi T, Lu H (2014) Mitochondrial thiol oxidase Erv1: Both shuttle cysteine residues are required for its function with distinct roles. Biochem J 460: 199–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ast C, Schmälzlin E, Löhmannsröben H-G, van Dongen JT (2012) Optical oxygen micro- and nanosensors for plant applications. Sensors (Basel) 12: 7015–7032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baena-González E, Rolland F, Thevelein JM, Sheen J (2007) A central integrator of transcription networks in plant stress and energy signalling. Nature 448: 938–942 [DOI] [PubMed] [Google Scholar]
- Barreto P, Okura V, Pena IA, Maia R, Maia IG, Arruda P (2016) Overexpression of mitochondrial uncoupling protein 1 (UCP1) induces a hypoxic response in Nicotiana tabacum leaves. J Exp Bot 67: 301–313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baxter-Burrell A, Yang Z, Springer PS, Bailey-Serres J (2002) RopGAP4-dependent Rop GTPase rheostat control of Arabidopsis oxygen deprivation tolerance. Science 296: 2026–2028 [DOI] [PubMed] [Google Scholar]
- Bienert GP, Møller AL, Kristiansen KA, Schulz A, Møller IM, Schjoerring JK, Jahn TP (2007) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282: 1183–1192 [DOI] [PubMed] [Google Scholar]
- Binzer T, Borum J, Pedersen O (2005) Flow velocity affects internal oxygen conditions in the seagrass Cymodocea nodosa. Aquat Bot 83: 239–247 [Google Scholar]
- Bishop T, Ratcliffe PJ (2014) Signaling hypoxia by hypoxia-inducible factor protein hydroxylases: A historical overview and future perspectives. Hypoxia (Auckl) 2: 197–213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjornson M, Dandekar A, Dehesh K (2016) Determinants of timing and amplitude in the plant general stress response. J Integr Plant Biol 58: 119–126 [DOI] [PubMed] [Google Scholar]
- Blokhina OB, Chirkova TV, Fagerstedt KV (2001) Anoxic stress leads to hydrogen peroxide formation in plant cells. J Exp Bot 52: 1179–1190 [PubMed] [Google Scholar]
- Bose J, Pottosin II, Shabala SS, Palmgren MG, Shabala S (2011) Calcium efflux systems in stress signaling and adaptation in plants. Front Plant Sci 2: 85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Branco-Price C, Kaiser KA, Jang CJ, Larive CK, Bailey-Serres J (2008) Selective mRNA translation coordinates energetic and metabolic adjustments to cellular oxygen deprivation and reoxygenation in Arabidopsis thaliana. Plant J 56: 743–755 [DOI] [PubMed] [Google Scholar]
- Broeckx T, Hulsmans S, Rolland F (2016) The plant energy sensor: Evolutionary conservation and divergence of SnRK1 structure, regulation, and function. J Exp Bot 67: 6215–6252 [DOI] [PubMed] [Google Scholar]
- Chang R, Jang CJ, Branco-Price C, Nghiem P, Bailey-Serres J (2012) Transient MPK6 activation in response to oxygen deprivation and reoxygenation is mediated by mitochondria and aids seedling survival in Arabidopsis. Plant Mol Biol 78: 109–122 [DOI] [PubMed] [Google Scholar]
- Cho YH, Hong JW, Kim EC, Yoo SD (2012) Regulatory functions of SnRK1 in stress-responsive gene expression and in plant growth and development. Plant Physiol 158: 1955–1964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung H-J, Ferl RJ (1999) Arabidopsis alcohol dehydrogenase expression in both shoots and roots is conditioned by root growth environment. Plant Physiol 121: 429–436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark LC Jr., Wolf R, Granger D, Taylor Z (1953) Continuous recording of blood oxygen tensions by polarography. J Appl Physiol 6: 189–193 [DOI] [PubMed] [Google Scholar]
- Colmer TD, Pedersen O (2008) Oxygen dynamics in submerged rice (Oryza sativa). New Phytol 178: 326–334 [DOI] [PubMed] [Google Scholar]
- De Clercq I, Vermeirssen V, Van Aken O, Vandepoele K, Murcha MW, Law SR, Inzé A, Ng S, Ivanova A, Rombaut D, et al. (2013) The membrane-bound NAC transcription factor ANAC013 functions in mitochondrial retrograde regulation of the oxidative stress response in Arabidopsis. Plant Cell 25: 3472–3490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demir F, Horntrich C, Blachutzik JO, Scherzer S, Reinders Y, Kierszniowska S, Schulze WX, Harms GS, Hedrich R, Geiger D, et al. (2013) Arabidopsis nanodomain-delimited ABA signaling pathway regulates the anion channel SLAH3. Proc Natl Acad Sci USA 110: 8296–8301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dmitriev RI, Papkovsky DB (2015) Intracellular probes for imaging oxygen concentration: How good are they? Methods Appl Fluoresc 3: 034001. [DOI] [PubMed] [Google Scholar]
- Dobrenel T, Caldana C, Hanson J, Robaglia C, Vincentz M, Veit B, Meyer C (2016) TOR signaling and nutrient sensing. Annu Rev Plant Biol 67: 261–285 [DOI] [PubMed] [Google Scholar]
- Dodd AN, Kudla J, Sanders D (2010) The language of calcium signaling. Annu Rev Plant Biol 61: 593–620 [DOI] [PubMed] [Google Scholar]
- Dromparis P, Paulin R, Sutendra G, Qi AC, Bonnet S, Michelakis ED (2013) Uncoupling protein 2 deficiency mimics the effects of hypoxia and endoplasmic reticulum stress on mitochondria and triggers pseudohypoxic pulmonary vascular remodeling and pulmonary hypertension. Circ Res 113: 126–136 [DOI] [PubMed] [Google Scholar]
- Dudkina NV, Eubel H, Keegstra W, Boekema EJ, Braun HP (2005) Structure of a mitochondrial supercomplex formed by respiratory-chain complexes I and III. Proc Natl Acad Sci USA 102: 3225–3229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edel KH, Marchadier E, Brownlee C, Kudla J, Hetherington AM (2017) The evolution of calcium-based signalling in plants. Curr Biol 27: R667–R679 [DOI] [PubMed] [Google Scholar]
- Evans MJ, Choi WG, Gilroy S, Morris RJ (2016) A ROS-assisted calcium wave dependent on the AtRBOHD NADPH oxidase and TPC1 cation channel propagates the systemic response to salt stress. Plant Physiol 171: 1771–1784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming IN, Manavaki R, Blower PJ, West C, Williams KJ, Harris AL, Domarkas J, Lord S, Baldry C, Gilbert FJ (2015) Imaging tumour hypoxia with positron emission tomography. Br J Cancer 112: 238–250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasch P, Fundinger M, Müller JT, Lee T, Bailey-Serres J, Mustroph A (2016) Redundant ERF-VII transcription factors bind to an evolutionarily conserved cis-motif to regulate hypoxia-responsive gene expression in Arabidopsis. Plant Cell 28: 160–180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler DA, Broselid C, Hederstedt L, Rasmusson AG (2007) Ca2+-binding and Ca2+-independent respiratory NADH and NADPH dehydrogenases of Arabidopsis thaliana. J Biol Chem 282: 28455–28464 [DOI] [PubMed] [Google Scholar]
- Gibbs DJ, Conde JV, Berckhan S, Prasad G, Mendiondo GM, Holdsworth MJ (2015) Group VII ethylene response factors coordinate oxygen and nitric oxide signal transduction and stress responses in plants. Plant Physiol 169: 23–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbs DJ, Lee SC, Isa NM, Gramuglia S, Fukao T, Bassel GW, Correia CS, Corbineau F, Theodoulou FL, Bailey-Serres J, et al. (2011) Homeostatic response to hypoxia is regulated by the N-end rule pathway in plants. Nature 479: 415–418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbs DJ, Md Isa N, Movahedi M, Lozano-Juste J, Mendiondo GM, Berckhan S, Marín-de la Rosa N, Vicente Conde J, Sousa Correia C, Pearce SP, et al. (2014) Nitric oxide sensing in plants is mediated by proteolytic control of group VII ERF transcription factors. Mol Cell 53: 369–379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giuntoli B, Lee SC, Licausi F, Kosmacz M, Oosumi T, van Dongen JT, Bailey-Serres J, Perata P (2014) A trihelix DNA binding protein counterbalances hypoxia-responsive transcriptional activation in Arabidopsis. PLoS Biol 12: e1001950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giuntoli B, Shukla V, Maggiorelli F, Giorgi FM, Lombardi L, Perata P, Licausi F (2017) Age-dependent regulation of ERF-VII transcription factor activity in Arabidopsis thaliana. Plant Cell Environ 40: 2333–2346 [DOI] [PubMed] [Google Scholar]
- Gonzali S, Loreti E, Cardarelli F, Novi G, Parlanti S, Pucciariello C, Bassolino L, Banti V, Licausi F, Perata P (2015) Universal stress protein HRU1 mediates ROS homeostasis under anoxia. Nat Plants 1: 15151. [DOI] [PubMed] [Google Scholar]
- Gross E, Sevier CS, Heldman N, Vitu E, Bentzur M, Kaiser CA, Thorpe C, Fass D (2006) Generating disulfides enzymatically: Reaction products and electron acceptors of the endoplasmic reticulum thiol oxidase Ero1p. Proc Natl Acad Sci USA 103: 299–304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzy RD, Mack MM, Schumacker PT (2007) Mitochondrial complex III is required for hypoxia-induced ROS production and gene transcription in yeast. Antioxid Redox Signal 9: 1317–1328 [DOI] [PubMed] [Google Scholar]
- Hamanaka RB, Weinberg SE, Reczek CR, Chandel NS (2016) The mitochondrial respiratory chain is required for organismal adaptation to hypoxia. Cell Reports 15: 451–459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardie DG. (2011) AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes Dev 25: 1895–1908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hegde RS, Ploegh HL (2010) Quality and quantity control at the endoplasmic reticulum. Curr Opin Cell Biol 22: 437–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez JA, Corpas FJ, Gomez M, Del Río LA, Sevilla F (1993) Salt‐induced oxidative stress mediated by activated oxygen species in pea leaf mitochondria. Physiol Plant 89: 103–110 [Google Scholar]
- Iwata Y, Fedoroff NV, Koizumi N (2008) Arabidopsis bZIP60 is a proteolysis-activated transcription factor involved in the endoplasmic reticulum stress response. Plant Cell 20: 3107–3121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang MI, Kobayashi A, Wakabayashi N, Kim SG, Yamamoto M (2004) Scaffolding of Keap1 to the actin cytoskeleton controls the function of Nrf2 as key regulator of cytoprotective phase 2 genes. Proc Natl Acad Sci USA 101: 2046–2051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleine T, Leister D (2016) Retrograde signaling: Organelles go networking. Biochim Biophys Acta 1857: 1313–1325 [DOI] [PubMed] [Google Scholar]
- Kmiecik P, Leonardelli M, Teige M (2016) Novel connections in plant organellar signalling link different stress responses and signalling pathways. J Exp Bot 67: 3793–3807 [DOI] [PubMed] [Google Scholar]
- Koritzinsky M, Levitin F, van den Beucken T, Rumantir RA, Harding NJ, Chu KC, Boutros PC, Braakman I, Wouters BG (2013) Two phases of disulfide bond formation have differing requirements for oxygen. J Cell Biol 203: 615–627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krab K, Kempe H, Wikström M (2011) Explaining the enigmatic K(M) for oxygen in cytochrome c oxidase: A kinetic model. Biochim Biophys Acta 1807: 348–358 [DOI] [PubMed] [Google Scholar]
- Krause BJ, Beck R, Souvatzoglou M, Piert M (2006) PET and PET/CT studies of tumor tissue oxygenation. Q J Nucl Med Mol Imaging 50: 28–43 [PubMed] [Google Scholar]
- Kröller-Schön S, Steven S, Kossmann S, Scholz A, Daub S, Oelze M, Xia N, Hausding M, Mikhed Y, Zinssius E, et al. (2014) Molecular mechanisms of the crosstalk between mitochondria and NADPH oxidase through reactive oxygen species-studies in white blood cells and in animal models. Antioxid Redox Signal 20: 247–266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapuente-Brun E, Moreno-Loshuertos R, Acín-Pérez R, Latorre-Pellicer A, Colás C, Balsa E, Perales-Clemente E, Quirós PM, Calvo E, Rodríguez-Hernández MA, et al. (2013) Supercomplex assembly determines electron flux in the mitochondrial electron transport chain. Science 340: 1567–1570 [DOI] [PubMed] [Google Scholar]
- Lee KW, Chen PW, Lu CA, Chen S, Ho TH, Yu SM (2009) Coordinated responses to oxygen and sugar deficiency allow rice seedlings to tolerate flooding. Sci Signal 2: ra61. [DOI] [PubMed] [Google Scholar]
- Licausi F, Giorgi FM, Schmälzlin E, Usadel B, Perata P, van Dongen JT, Geigenberger P (2011b) HRE-type genes are regulated by growth-related changes in internal oxygen concentrations during the normal development of potato (Solanum tuberosum) tubers. Plant Cell Physiol 52: 1957–1972 [DOI] [PubMed] [Google Scholar]
- Licausi F, Kosmacz M, Weits DA, Giuntoli B, Giorgi FM, Voesenek LA, Perata P, van Dongen JT (2011a) Oxygen sensing in plants is mediated by an N-end rule pathway for protein destabilization. Nature 479: 419–422 [DOI] [PubMed] [Google Scholar]
- Licausi F, van Dongen JT, Giuntoli B, Novi G, Santaniello A, Geigenberger P, Perata P (2010) HRE1 and HRE2, two hypoxia-inducible ethylene response factors, affect anaerobic responses in Arabidopsis thaliana. Plant J 62: 302–315 [DOI] [PubMed] [Google Scholar]
- Licausi F, Weits DA, Pant BD, Scheible WR, Geigenberger P, van Dongen JT (2011c) Hypoxia responsive gene expression is mediated by various subsets of transcription factors and miRNAs that are determined by the actual oxygen availability. New Phytol 190: 442–456 [DOI] [PubMed] [Google Scholar]
- Lin IS, Wu YS, Chen CT, Chen GH, Hwang SG, Jauh GY, Tzen JT, Yang CY (2017) AtRBOH I confers submergence tolerance and is involved in auxin-mediated signaling pathways under hypoxic stress. Plant Growth Regul 83: 277–285 [Google Scholar]
- Liu JX, Howell SH (2016) Managing the protein folding demands in the endoplasmic reticulum of plants. New Phytol 211: 418–428 [DOI] [PubMed] [Google Scholar]
- Liu B, Sun L, Ma L, Hao FS (2017) Both AtrbohD and AtrbohF are essential for mediating responses to oxygen deficiency in Arabidopsis. Plant Cell Rep 36: 947–957 [DOI] [PubMed] [Google Scholar]
- Loenarz C, Schofield CJ (2011) Physiological and biochemical aspects of hydroxylations and demethylations catalyzed by human 2-oxoglutarate oxygenases. Trends Biochem Sci 36: 7–18 [DOI] [PubMed] [Google Scholar]
- Lokdarshi A, Conner WC, McClintock C, Li T, and Roberts DM (2016) Arabidopsis CML38, a calcium sensor that localizes to ribonucleoprotein complexes under hypoxia stress. Plant Phys 170: 1046–1059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxwell DP, Wang Y, McIntosh L (1999) The alternative oxidase lowers mitochondrial reactive oxygen production in plant cells. Proc Natl Acad Sci USA 96: 8271–8276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazars C, Thuleau P, Lamotte O, Bourque S (2010) Cross-talk between ROS and calcium in regulation of nuclear activities. Mol Plant 3: 706–718 [DOI] [PubMed] [Google Scholar]
- Melis A, Zhang L, Forestier M, Ghirardi ML, Seibert M (2000) Sustained photobiological hydrogen gas production upon reversible inactivation of oxygen evolution in the green alga Chlamydomonas reinhardtii. Plant Physiol 122: 127–136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer AK, Longin CF, Klose C, Hermann A (2010) New regulator for energy signaling pathway in plants highlights conservation among species. Sci Signal 3: jc5. [DOI] [PubMed] [Google Scholar]
- Millar AH, Whelan J, Soole KL, Day DA (2011) Organization and regulation of mitochondrial respiration in plants. Annu Rev Plant Biol 62: 79–104 [DOI] [PubMed] [Google Scholar]
- Mueller SJ, Reski R (2015) Mitochondrial dynamics and the ER: The plant perspective. Front Cell Dev Biol 3: 78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mustroph A, Lee SC, Oosumi T, Zanetti ME, Yang H, Ma K, Yaghoubi-Masihi A, Fukao T, Bailey-Serres J (2010) Cross-kingdom comparison of transcriptomic adjustments to low-oxygen stress highlights conserved and plant-specific responses. Plant Physiol 152: 1484–1500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mustroph A, Zanetti ME, Jang CJ, Holtan HE, Repetti PP, Galbraith DW, Girke T, Bailey-Serres J (2009) Profiling translatomes of discrete cell populations resolves altered cellular priorities during hypoxia in Arabidopsis. Proc Natl Acad Sci USA 106: 18843–18848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S, Giraud E, Duncan O, Law SR, Wang Y, Xu L, Narsai R, Carrie C, Walker H, Day DA, et al. (2013a) Cyclin-dependent kinase E1 (CDKE1) provides a cellular switch in plants between growth and stress responses. J Biol Chem 288: 3449–3459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S, Ivanova A, Duncan O, Law SR, Van Aken O, De Clercq I, Wang Y, Carrie C, Xu L, Kmiec B, et al. (2013b) A membrane-bound NAC transcription factor, ANAC017, mediates mitochondrial retrograde signaling in Arabidopsis. Plant Cell 25: 3450–3471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikoloski Z, van Dongen JT (2011) Modeling alternatives for interpreting the change in oxygen-consumption rates during hypoxic conditions. New Phytol 190: 273–276, author reply 276–278 [DOI] [PubMed] [Google Scholar]
- Pastore D, Trono D, Laus MN, Di Fonzo N, Flagella Z (2007) Possible plant mitochondria involvement in cell adaptation to drought stress. A case study: Durum wheat mitochondria. J Exp Bot 58: 195–210 [DOI] [PubMed] [Google Scholar]
- Potzkei J, Kunze M, Drepper T, Gensch T, Jaeger K-E, Büchs J (2012) Real-time determination of intracellular oxygen in bacteria using a genetically encoded FRET-based biosensor. BMC Biol 10: 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pu X, Lv X, Tan T, Fu F, Qin G, Lin H (2015) Roles of mitochondrial energy dissipation systems in plant development and acclimation to stress. Ann Bot 116: 583–600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pucciariello C, Parlanti S, Banti V, Novi G, Perata P (2012) Reactive oxygen species-driven transcription in Arabidopsis under oxygen deprivation. Plant Physiol 159: 184–196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramírez-Aguilar SJ, Keuthe M, Rocha M, Fedyaev VV, Kramp K, Gupta KJ, Rasmusson AG, Schulze WX, van Dongen JT (2011) The composition of plant mitochondrial supercomplexes changes with oxygen availability. J Biol Chem 286: 43045–43053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratcliffe PJ. (2013) Oxygen sensing and hypoxia signalling pathways in animals: The implications of physiology for cancer. J Physiol 591: 2027–2042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raymond J, Segrè D (2006) The effect of oxygen on biochemical networks and the evolution of complex life. Science 311: 1764–1767 [DOI] [PubMed] [Google Scholar]
- Rhoads DM, Subbaiah CC (2007) Mitochondrial retrograde regulation in plants. Mitochondrion 7: 177–194 [DOI] [PubMed] [Google Scholar]
- Rizzuto R, De Stefani D, Raffaello A, Mammucari C (2012) Mitochondria as sensors and regulators of calcium signalling. Nat Rev Mol Cell Biol 13: 566–578 [DOI] [PubMed] [Google Scholar]
- Rudolph-Moor N, Esser HG, Carminati A, Moradi BA, Hilger A, Kardjilov N, Nagl S, Oswald SE (2012) Dynamic oxygen mapping in the root zone by fluorescence dye imaging combined with neutron radiography. J Soils Sediments 12: 63–74 [Google Scholar]
- Sanders D, Pelloux J, Brownlee C, Harper JF (2002) Calcium at the crossroads of signaling. Plant Cell 14(Suppl): S401–S417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasidharan R, Hartman S, Liu Z, Martopawiro S, Sajeev N, van Veen H, Yeung E, Voesenek LACJ (2017) Signal dynamics and interactions during flooding stress. Plant Physiol, (in press). Published online November 2, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasidharan R, Voesenek LA (2015) Ethylene-mediated acclimations to flooding stress. Plant Physiol 169: 3–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schippers JHM, Schmidt R (2016) The role of ROS and redox signaling during the initial cellular response to abiotic stress. In Gupta DK, Palma JM, Corpas FJ, eds, Redox State as a Central Regulator of Plant-Cell Stress Responses. Springer International Publishing, Cham, Switzerland, pp 253–273 [Google Scholar]
- Schwarzländer M, Finkemeier I (2013) Mitochondrial energy and redox signaling in plants. Antioxid Redox Signal 18: 2122–2144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sedbrook JC, Kronebusch PJ, Borisy GG, Trewavas AJ, Masson PH (1996) Transgenic AEQUORIN reveals organ-specific cytosolic Ca2+ responses to anoxia and Arabidopsis thaliana seedlings. Plant Physiol 111: 243–257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahzad Z, Canut M, Tournaire-Roux C, Martinière A, Boursiac Y, Loudet O, Maurel C (2016) A potassium-dependent oxygen sensing pathway regulates plant root hydraulics. Cell 167: 87–98.e14 [DOI] [PubMed] [Google Scholar]
- Singh P, Sinha AK (2016) A positive feedback loop governed by SUB1A1 interaction with MITOGEN-ACTIVATED PROTEIN KINASE3 imparts submergence tolerance in rice. Plant Cell 28: 1127–1143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorenson R, Bailey-Serres J (2014) Selective mRNA sequestration by OLIGOURIDYLATE-BINDING PROTEIN 1 contributes to translational control during hypoxia in Arabidopsis. Proc Natl Acad Sci USA 111: 2373–2378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez MC, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61: 621–649 [DOI] [PubMed] [Google Scholar]
- Subbaiah CC, Zhang J, Sachs MM (1994) Involvement of intracellular calcium in anaerobic gene expression and survival of maize seedlings. Plant Physiol 105: 369–376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki N, Miller G, Morales J, Shulaev V, Torres MA, Mittler R (2011) Respiratory burst oxidases: The engines of ROS signaling. Curr Opin Plant Biol 14: 691–699 [DOI] [PubMed] [Google Scholar]
- Sweetlove LJ, Beard KF, Nunes-Nesi A, Fernie AR, Ratcliffe RG (2010) Not just a circle: Flux modes in the plant TCA cycle. Trends Plant Sci 15: 462–470 [DOI] [PubMed] [Google Scholar]
- Tomé F, Nägele T, Adamo M, Garg A, Marco-Llorca C, Nukarinen E, Pedrotti L, Peviani A, Simeunovic A, Tatkiewicz A, et al. (2014) The low energy signaling network. Front Plant Sci 5: 353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Dongen JT, Gupta KJ, Ramírez-Aguilar SJ, Araújo WL, Nunes-Nesi A, Fernie AR (2011) Regulation of respiration in plants: A role for alternative metabolic pathways. J Plant Physiol 168: 1434–1443 [DOI] [PubMed] [Google Scholar]
- van Dongen JT, Licausi F (2015) Oxygen sensing and signaling. Annu Rev Plant Biol 66: 345–367 [DOI] [PubMed] [Google Scholar]
- Vikram DS, Zweier JL, Kuppusamy P (2007) Methods for noninvasive imaging of tissue hypoxia. Antioxid Redox Signal 9: 1745–1756 [DOI] [PubMed] [Google Scholar]
- Visser EJW, Colmer TD, Blom CWPM, Voesenek LACJ (2000) Changes in growth, porosity, and radial oxygen loss from adventitious roots of selected mono- and dicotyledonous wetland species with contrasting types of aerenchyma. Plant Cell Environ 23: 1237–1245 [Google Scholar]
- Wagner S, Van Aken O, Elsässer M, Schwarzländer M (2018) Mitochondrial signaling and oxygen stress response of plants. Plant Phys 176: 1156–1170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weits DA, Giuntoli B, Kosmacz M, Parlanti S, Hubberten H-M, Riegler H, Hoefgen R, Perata P, van Dongen JT, Licausi F (2014) Plant cysteine oxidases control the oxygen-dependent branch of the N-end-rule pathway. Nat Commun 5: 3425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White MD, Klecker M, Hopkinson RJ, Weits DA, Mueller C, Naumann C, O’Neill R, Wickens J, Yang J, Brooks-Bartlett JC, et al. (2017) Plant cysteine oxidases are dioxygenases that directly enable arginyl transferase-catalysed arginylation of N-end rule targets. Nat Commun 8: 14690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiel C, Lallet-Daher H, Gitenay D, Gras B, Le Calvé B, Augert A, Ferrand M, Prevarskaya N, Simonnet H, Vindrieux D, et al. (2014) Endoplasmic reticulum calcium release through ITPR2 channels leads to mitochondrial calcium accumulation and senescence. Nat Commun 5: 3792. [DOI] [PubMed] [Google Scholar]
- Wouters BG, Koritzinsky M (2008) Hypoxia signalling through mTOR and the unfolded protein response in cancer. Nat Rev Cancer 8: 851–864 [DOI] [PubMed] [Google Scholar]
- Yemelyanov VV, Shishova MF, Chirkova TV, Lindberg SM (2011) Anoxia-induced elevation of cytosolic Ca2+ concentration depends on different Ca2+ sources in rice and wheat protoplasts. Planta 234: 271–280 [DOI] [PubMed] [Google Scholar]
- Yu J, Kodibagkar V, Cui W, Mason R (2005) 19F: a versatile reporter for non-invasive physiology and pharmacology using magnetic resonance. Curr Med Chem 12: 819–848 [DOI] [PubMed] [Google Scholar]
- Zhang S, Hosaka M, Yoshihara T, Negishi K, Iida Y, Tobita S, Takeuchi T (2010) Phosphorescent light-emitting iridium complexes serve as a hypoxia-sensing probe for tumor imaging in living animals. Cancer Res 70: 4490–4498 [DOI] [PubMed] [Google Scholar]
- Zhu JK. (2016) Abiotic stress signaling and responses in plants. Cell 167: 313–324 [DOI] [PMC free article] [PubMed] [Google Scholar]