Abstract
Vibrio cholerae, a pandemic diarrheagenic bacterium, is able to synthesize the essential vitamin riboflavin through the riboflavin biosynthetic pathway (RBP) and also to internalize it through the RibN importer. In bacteria, the way riboflavin biosynthesis and uptake functions correlate is unclear. To gain insights into the role of the riboflavin provision pathways in the physiology of V. cholerae, we analyzed the transcriptomics response to extracellular riboflavin and to deletions of ribD (RBP-deficient strain) or ribN. Many riboflavin-responsive genes were previously reported to belong to the iron regulon, including various iron uptake genes. Real time PCR analysis confirmed this effect and further documented that reciprocally, iron regulates RBP and ribN genes in a riboflavin-dependent way. A subset of genes were responding to both ribD and ribN deletions. However, in the subset of genes specifically affected in the ∆ribD strain, the functional terms protein folding and oxidation reduction process were enriched, as determined by a Gene Ontology analysis. In the gene subset specifically affected in the ∆ribN strain, the cytochrome complex assembly functional term was enriched. Results suggest that iron and riboflavin interrelate to regulate its respective provision genes and that both common and specific effects of biosynthesized and internalized riboflavin exist.
Introduction
Redox reactions, consisting of electron transfers from an oxidizing molecule to a reducing one, lie at the core of many central physiological processes. These include oxidative phosphorylation, cell signaling, photosynthesis, DNA repair, carbohydrates metabolism, oxygen storage, photosensitization and protein folding among many other1–3. In order to complete these reactions, enzymes usually require redox cofactor molecules which include nicotinamide-derived molecules, iron-sulfur clusters, thiamin, deazaflavin and transition metals like cooper, manganese, cobalt and zinc3–8. However, iron is by far the most widespread metal redox cofactor, while molecules derived from riboflavin (also named vitamin B2), such as flavin mononucletoide (FMN) and flavin adenine dinucleotide (FAD) constitute the main organic electron transfer cofactors, with an importance similar to that of iron6. Genes encoding flavoproteins may comprise up to 3.5% of the genome of a species9. Flavins are probably the most versatile cofactors, being able to catalize one- and two-electron transfers, which allows their participation in electron bifurcation reactions10. These molecules may also catalize non-redox reactions and are increasingly recognized as covalent catalysts, acting in the formation of flavin-substrate adduct intermediates9,11.
There is evidence that flavins may act as signaling molecules in bacteria. Riboflavin and its breakage derivative lumichrome are able to mimic N-acyl homoserine lactone for activation of quorum sensing pathways in Pseudomonas aeruoginosa and riboflavin is a chemoattractant to S. oneidensis12,13. Riboflavin may as well be secreted by some bacteria to be used as electron shuttle to reduce Fe+3 into its more soluble Fe+2 form and to complete the extracellular respiratory chain14–17. In addition, this vitamin frequently represents a metabolic currency during bacteria-host or intermicrobial trade interactions18,19.
Most bacteria are able to biosynthesize riboflavin through the riboflavin biosynthetic pathway (RBP). This pathway starts with guanosine triphosphate (GTP) and ribulose-5-phosphate to synthesize riboflavin using the RibA (GTP cyclohydrolase II), RibD (pyrimidine deaminase/reductase), RibH (lumazine synthase), RibB (3,4-dihydroxybutanone phosphate synthase) and RibE (riboflavin synthase) enzymes20,21. The nomenclature of RBP enzymes varies among bacterial species and Escherichia coli names22 are thoroughly used here. In bacterial genomes, RBP genes could form an operon or be positioned in different loci. In various species, some RBP genes are duplicated or multiplicated23. In some cases, duplicated RBP gene orthologs appear to implement modularity to riboflavin production, where the RBP uses subsets of genes to provide riboflavin for specific purposes, such as secretion or interactions with the host24,25. Bacteria may also use importer proteins to internalize riboflavin from the surroundings. Although many bacterial species rely exclusively on riboflavin uptake, many others possess both riboflavin biosynthesis and uptake. It is hypothesized that this overlay allows bacteria to take advantage of changing environments, turning on riboflavin uptake and stopping biosynthesis in nutrient rich niches, while granting autonomy when facing stringent conditions. It is also posible that riboflavin importers procure flavins for specific functions in riboflavin-prototrophic species23,26–28.
Vibrio cholerae are Gram negative proteobacteria responsible for cholera, a pandemic disease affecting mainly developing countries, characterized by acute, life-threatening diarrhea29. Global cholera burden has recently been estimated in around 2.8 million cases with 95,000 deaths per year30. Most V. cholerae strains are innocuous indigenous members of estuarine and seawater microbiota, with a few strains from serotypes O1 and O139 causing almost all of cholera cases29–32. In these bacteria, development of virulence is not only associated with the acquisition of virulence factors but also of specific alleles of virulence adaptive polymorphisms rotating in environmental species, which confer selective advantages like host colonization properties32. Importantly, environmental water conditions such as temperature, salinity, pH and sunlight exposure have a major impact in the development of cholera epidemics and thus outbreaks are expected to increase due to global warming31,33.
Cholera is mostly a waterborne disease, and after human consumption, V. cholerae expresses several virulence factors. Cholera toxin is the main inducer of diarrhea. This toxin translocates into host epitelial cells to promote constitutive activation of the adenylate cyclase, causing an increase in Cl− and water efflux. Initial adhesion to host intestine is promoted by the toxin coregulated pilus. In addition, other V. cholerae virulence factors such as the flagellum, the HapA metalloprotease, Zot and RTX toxins and different iron acquisition systems are also expressed in order to favor host colonization33,34. In the environment, Vibrio cholerae is primarily found associated to abiotic surfaces and to chitin carpaces of acquatic organisms as microcolonies or biofilms, but also as planktonic cells. Biofilm formation is required during the host pathogenic phase and biofilm structures are detected in faeces from infected humans34. In addition, these bacteria are able to enter metabolically dormant viable but non culturable and persister states in response to harsh environmental conditions, which may allow bacteria to face physical and nutritional changes in niches or to survive in atypical environments such as fomites35,36. Thus, this bacterium has a complex life cycle and likely, both V. cholerae riboflavin requirements and availability are highly variable among the different environments and physiological states in which it may be found. Although there is no estimation of the number of flavin-requiring proteins in V. cholerae, a structural genomics approach calculated the proportion of genes coding for flavoenzymes in more than 1% in the related species Vibrio fischeri9. V. cholerae encodes a full RBP organized into a large operon and two monocistronic units. Together with genes not directly involved in riboflavin biosynthesis, the RBP operon contains ribD, ribE, ribH and a gene belonging to a family of hybrid ribBA genes common in proteobacteria. In addition, RibA and RibB monocystronic homologs are encoded in the genome of V. cholerae37,38. The ribB gene conserves a putative FMN riboswitch, which is a regulatory element forming alternative structures in the 5´ untranslated region of the messenger RNA to control expression depending on FMN binding status. The RBP is dispensable when V. cholerae grows in rich medium39, as this species also has a RibN riboflavin importer40. Unlike some orthologs in other proteobacteria, ribN in V. cholerae lacks a FMN riboswitch. We recently reported that when growing in the presence of extracellular riboflavin in standard minimal media, the expression of the monocistronic ribB gene is diminished while expression of the rest of the RBP genes and of ribN is not affected38.
In spite of the ubiquitous importance of riboflavin in bacterial physiology, no high throughput approach has been applied to study the response elicited by any bacterial species to this metabolite. Given the complex ecophysiological features of V. cholerae, this organism may comprise a suited model to study the way riboflavin biosynthesis and transport interplay to accomplish bacterial riboflavin needs. This study analized the transcriptomics response to extracellular riboflavin and compared the effects of the elimination of endogenous biosynthesis or uptake through the RibN importer. This allowed the identification of a set of genes responding to exogenous riboflavin, as well as to outline specific effects of synthesized or internalized riboflavin.
Materials and Methods
Strains and growth conditions
V. cholerae N16961 strain and its ∆ribD and ∆ribN derivatives were grown overnight in LB plates at 37 °C. 5 ml of LB broth were inoculated with a colony of the plate cultures and incubated at 37 °C in an orbital shaker at 150 rpm until they reached an OD600nm of 1.0. Next, cultures were centrifuged and pellet washed twice with T minimal medium41 and resuspended in 1 ml of fresh T. 10 ml of plain T medium or T + 2 µM riboflavin were inoculated with 10 µl of the resuspensions and incubated at 37 °C and 180 rpm until an OD600nm of 0.8. 1 ml of each culture was centrifuged and subjected to RNA extraction. When indicated, iron was omitted in T media and 3 ml of cultures at OD600nm = 0.3 were harvested for RNA extraction. This growth protocol was performed three times independently for each condition and was similar for RNA subjected to transcriptomics and Real Time PCR (RT-PCR).
RNA extractions, retrotranscription, RNAseq and RT-PCR
RNA extraction was performed with the Thermo Scientific Genejet RNA purification kit according to manufacturer’s instructions. RNA extracts were digested with Turbo DNA-free DNAase at 37 °C for 1 hour. For RNAseq, rRNA was removed using the Ribo-Zero removal kit and cDNA libraries were constructed using the TruSeq mRNA stranded kit, according to manufacturer’s instructions. Next, RNA was sequenced using the Illumina HySeq platform to produce 100 bp paired-end reads, with ~40 million reads per sample. Sequencing raw data files, processed sequence data files and metadata information was deposited at the Gene Expression Omnibus database from NCBI (GSE107538). rRNA removal, cDNA libraries generation and RNAseq were performed at Genoma Mayor (Santiago, Chile).
For RT-PCR analysis, the AffinityScript QPCR cDNA Synthesis kit (Agilent Technologies) was used for cDNA synthesis according to manufacturer’s instructions. As a negative control, a reaction with no reverse transcriptase was included for each sample in each run. RT-PCR was performed using the Brilliant II SYBR Green QPCR Master Mix kit in a One-Step Applen Biosystems (Life Technologies) thermocycler. Relative expression in the indicated conditions was determined through the ∆∆Ct method as developed before42. The 16 s ribosomal RNA gene was used for normalization. For the assessment of the relative expression by RT-PCR of ribB, ribN, ribD and gyrB, the sets of primers used were ribB Fw/ribB Rv, ribN Fw/ribN Rv, ribD Fw/ribD Rv and gyrB Fw/gyrB Rv38, respectively. Other RT-PCR primers are as follows: for tonB1, tonB1 Fw (5′- GGTGTTTGCCATGCCTGCTGG-3′)/tonB1 Rv (5′-GCGGCTTCACCTTCGGCTTAG-3′); for sodA, sodA Fw (5′-GCCAAGCGATATTCATCCAAGG-3′)/sodA Rv (5′-GCTCAGTGGCCTATCTTCATGC-3′).
RNAseq data analysis
Quality control visualization and analysis (adapter and quality trimming) was performed using FastQC version 0.11.2 (http://www.bioinformatics.bbsrc.ac.uk/projects/fastqc/) and Trim_galore version 0.4.1 (http://www.bioinformatics.babraham.ac.uk/projects/trim_galore/), respectively. Reads were mapped to the genome of Vibrio cholerae 01 biovar El Tor str. N16961 (RefSeq, NCBI) using Bowtie2 version 2.1.043. In all of the samples the alignment percentage of reads was above 98%. Differential expression analysis between samples was performed with the Bioconductor package edgeR version 3.18.144 using negative binomial model and exact test based on quantile-adjusted conditional maximum likelihood method (qCML). Genes with a statistically significant change in expression (P < 0.05) were selected for further analysis. Analyses of enrichment of Gene Ontology (GO) terms of biological processes in the indicated subsets of genes were performed on the online platform of the Gene Ontology Consortium (www.geneontology.org), and statistically significant (P < 0.05) functional terms were retrieved.
Results
Overview of the experiment
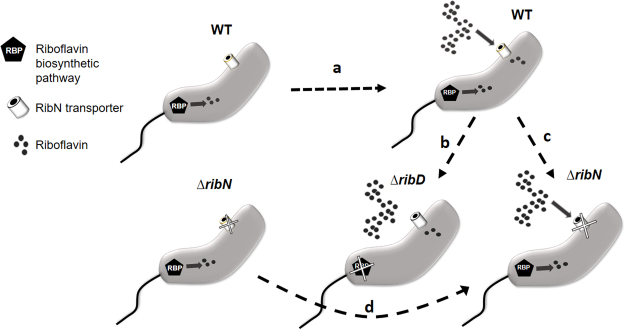
In V. cholerae, exogenous riboflavin downregulates the expression of the FMN riboswitch-containing gene ribB38. In order to identify other genes whose expression is affected in response to riboflavin, we performed RNAseq in V. cholerae N16961 cultures growing in T minimal medium with or without riboflavin. Also, to start elucidating putative differential roles of the riboflavin provision pathways, we included in this analysis the ∆ribD and ∆ribN derivative strains. The V. cholerae ∆ribD is a riboflavin auxotroph unable to grow in T media without riboflavin, while the ∆ribN does not has an impairment to grow without riboflavin compared to the WT45. A general overview of strains, growth conditions and transcriptomics comparisons is presented in Fig. 1. Four transcriptomics comparisons were performed as follows: WT growing without riboflavin versus WT with riboflavin (Comparison a in Fig. 1), WT versus ∆ribD both with riboflavin (b), WT versus ∆ribN both with riboflavin (c). The genes showing a difference of at least one fold in expression in any of these comparisons were selected and are shown in Table 1. Additionally, a comparison of ∆ribN without riboflavin versus ∆ribN with riboflavin (d) was performed. Genes showing more than one fold change in this comparison and also found in any of the three previous comparisons are indicated in Table 1. In all cases, the genes selected presented a statistically significant change in expression (P < 0.05).
Figure 1.
Schematic representation of the V. cholerae strains and conditions compared in the transcriptomics analysis. V. cholerae WT and its derivative mutant strains were grown in minimal T medium with or without riboflavin as indicated. Four transcriptomics comparisons were performed. In order to identify genes whose expression is regulated by riboflavin, transcriptomes of WT in T versus WT in T plus riboflavin were compared (comparison (a)). Comparison of WT versus ΔribD (b) allowed to identify genes affected by the lack of riboflavin biosynthesis. Comparison (c) identified genes affected by the lack of riboflavin transport through RibN. Finally, comparison of the ΔribN strain with and without riboflavin pinpointed genes affected by riboflavin independently of its uptake through RibN.
Table 1.
List of genes whose expression is affected in response to exogenous riboflavin or deletions of ribD or ribN.
| Gene ID | Gene Name | Gene Description | Fold Change (Log2) | ||
|---|---|---|---|---|---|
| WT → WT RF+ | WT RF + → ΔribD RF+ | WT RF + → ΔribN RF+ | |||
| VC0010 | amino acid ABC transporter periplasmic amino acid-binding portion | 1.809 | |||
| VC0018 | ibpA | 16 kDa heat shock protein A | −1.740 | ||
| VC0027 | threonine dehydratase | −1.226 | −1.191 | ||
| VC0028 | dihydroxy-acid dehydratase | −1.087 | |||
| VC0030 | ilvM | acetolactate synthase II small subunit | 1.116 | ||
| VC0053 | hypothetical protein | 1.074 | |||
| VC0089 | cytochrome c551 peroxidase | 1.076 | |||
| VC0102 | hypothetical protein | −1.160 | |||
| VC0138 | hypothetical protein | −1.564 | |||
| VC0139 | DPS family protein | −1.561 | −1.012 | ||
| VC0143 | hypothetical protein | −1.028 | |||
| VC0162 | ketol-acid reductoisomerase | 1.881 | 1.161 | ||
| VC0199 | hemolysin secretion ATP-binding protein%2 C putative | −1.428 | |||
| VC0200 | fhuA | OMT ferrichrome | −2.453◊ | ||
| VC0201 | fhuC | IMT ferrichrome | −1.308◊ | ||
| VC0202 | iron(III) ABC transporter%2 C periplasmic iron-compound-binding protein | −1.349◊ | |||
| VC0211 | pyrE | orotate phosphoribosyltransferase | 1.218 | ||
| VC0216 | methyl-accepting chemotaxis protein | 1.316 | |||
| VC0301 | hypothetical protein | −1.087 | −1.191 | ||
| VC0364 | bfd | bacterioferritin-associated ferredoxin | −1.459◊ | ||
| VC0365 | bfr | bacterioferritin | −1.124 | ||
| VC0366 | rpsF | ribosomal protein S6 | 1.152 | ||
| VC0367 | primosomal replication protein N | 1.136 | |||
| VC0368 | rpsR | ribosomal protein S18 | 1.099 | ||
| VC0382 | hypothetical protein | 1.060 | |||
| VC0383 | hypothetical protein | 1.198 | |||
| VC0384 | sulfite reductase (NADPH) flavoprotein alpha-component | 1.208 | |||
| VC0420 | conserved hypothetical protein | −1.050 | |||
| VC0426 | hypothetical protein | −1.708 | |||
| VC0430 | immunogenic protein | 1.017 | |||
| VC0438 | conserved hypothetical protein | −1.114 | |||
| VC0488 | extracellular solute-binding protein putative | 1.067 | |||
| VC0491 | hypothetical protein | 1.026 | |||
| VC0492 | hypothetical protein | 1.273 | |||
| VC0503 | conserved hypothetical protein | −1.667 | |||
| VC0515 | conserved hypothetical protein | 1.158 | |||
| VC0546 | hypothetical protein | −1.215 | |||
| VC0548 | csrA | carbon storage regulator | −1.264 | −1.109 | |
| VC0549 | hypothetical protein | 1.054 | |||
| VC0550 | oxaloacetate decarboxylase alpha subunit | 1.010 | |||
| VC0589 | ABC transporter ATP-binding protein | −1.010 | |||
| VC0607 | pseudogene | 1.102 | |||
| VC0608 | fbpA | Iron(III) ABC transporter | −1.439◊ | ||
| VC0625 | hypothetical protein | −1.140 | |||
| VC0633 | ompU | outer membrane protein OmpU | 1.362 | −1.261 | |
| VC0651 | conserved hypothetical protein | −1.750 | |||
| VC0652 | protease putative | −2.054 | |||
| VC0654 | conserved hypothetical protein | −1.471 | |||
| VC0655 | acetyltransferase putative | −1.182 | |||
| VC0706 | sigma-54 modulation protein putative | −1.297 | |||
| VC0707 | hypothetical protein | −1.043 | |||
| VC0708 | bamD | conserved hypothetical protein | −1.124 | ||
| VC0711 | clpB | clpB protein | −2.092 | −1.022 | |
| VC0734 | malate synthase A | 3.115 | |||
| VC0735 | hypothetical protein | 3.069 | |||
| VC0736 | isocitrate lyase | 1.788 | |||
| VC0748 | aminotransferase NifS class V | −1.034 | |||
| VC0749 | NifU-related protein | −1.254 | |||
| VC0750 | hesB | hesB family protein | −1.166 | ||
| VC0753 | ferredoxin | −1.008 | −1.100 | ||
| VC0754 | conserved hypothetical protein | −1.079 | |||
| VC0765 | conserved hypothetical protein | −1.475 | |||
| VC0771 | vibB | vibriobactin-specific isochorismatase | −1.315 | ||
| VC0824 | tpx | tagD protein | 1.636 | ||
| VC0855 | dnaK | dnaK protein | −1.560 | ||
| VC0856 | dnaJ | dnaJ protein | −1.504 | ||
| VC0863 | conserved hypothetical protein | 1.115 | |||
| VC0878 | rpmE2 | ribosomal protein L31P family | −1.276 | ||
| VC0879 | rpmJ | ribosomal protein L36 putative | −1.121 | ||
| VC0895 | hypothetical protein | −1.190 | |||
| VC0905 | metQ | D-methionine transport system substrate-binding protein | 1.230 | ||
| VC1049 | aphB | transcriptional regulator LysR family | −1.111 | ||
| VC1075 | conserved hypothetical protein | −1.086 | |||
| VC1077 | hypothetical protein | −1.136 | |||
| VC1091 | oligopeptide ABC transporter periplasmic oligopeptide-binding protein | 2.133 | |||
| VC1114 | bioC | biotin synthesis protein BioC | −1.556 | ||
| VC1115 | bioD | dethiobiotin synthetase | −1.750 | ||
| VC1117 | htpX | heat shock protein HtpX | −1.069 | ||
| VC1139 | phosphoribosyl-AMP cyclohydrolase/phosphoribosyl-ATP pyrophosphohydrolase | 1.072 | |||
| VC1147 | iron-containing alcohol dehydrogenase | 1.203 | |||
| VC1157 | response regulator | 1.183 | |||
| VC1169 | trpA | tryptophan synthase alpha subunit | 1.028 | ||
| VC1175 | hypothetical protein | 1.153 | |||
| VC1206 | gntR | histidine utilization repressor | 1.631◊ | ||
| VC1217 | conserved hypothetical protein | −1.070 | |||
| VC1224 | hypothetical protein | −1.101 | |||
| VC1226 | thiopurine methyltransferase | −1.344 | |||
| VC1227 | hypothetical protein | −1.250 | |||
| VC1235 | sodium/dicarboxylate symporter | 1.325 | |||
| VC1248 | methyl-accepting chemotaxis protein | 1.355 | |||
| VC1264 | irpA | fuction unknown, COG3487 | −1.406◊ | ||
| VC1266 | hypothetical periplasmic lipoprotein, like to irpA, COG3488 | −1.086 | |||
| VC1278 | transcriptional regulator MarR family | 2.100 | |||
| VC1279 | transporter BCCT family | 4.896 | |||
| VC1280 | hypothetical protein | 1.144 | |||
| VC1314 | transporter putative | 1.487 | |||
| VC1315 | sensor histidine kinase | 1.179 | |||
| VC1324 | hypothetical protein | 1.104 | |||
| VC1343 | peptidase M20A family | −1.335 | |||
| VC1373 | DnaK-related protein | −1.039 | |||
| VC1386 | chaperone | −1.079 | |||
| VC1414 | taq | thermostable carboxypeptidase 1 | 1.145 | ||
| VC1489 | hypothetical protein | −1.609 | −1.454 | ||
| VC1510 | hypothetical protein | 1.168 | 1.016 | ||
| VC1511 | formate dehydrogenase cytochrome B556 subunit | 1.521 | 1.102 | ||
| VC1512 | formate dehydrogenase iron-sulfur subunit | 1.604 | 1.100 | ||
| VC1513 | pseudogene | 2.147 | 1.251 | ||
| VC1514 | hypothetical protein | 2.306 | 1.395 | ||
| VC1515 | chaperone formate dehydrogenase-specific putative | 2.761 | 1.920 | ||
| VC1516 | iron-sulfur cluster-binding protein | 2.750 | 2.064 | ||
| VC1517 | hypothetical protein | 1.484 | 1.143 | ||
| VC1518 | hypothetical protein | 1.735 | 1.252 | ||
| VC1523 | conserved hypothetical protein | 1.852 | 1.043 | ||
| VC1524 | ABC transporter permease protein | 1.617 | |||
| VC1547 | exbB | exbB related linked to tonB2 | −1.006 | ||
| VC1548 | hypothetical, linked to tonB2 | −1.083 | |||
| VC1551 | glycerol-3-phosphate ABC transporter permease protein | −1.055 | |||
| VC1559 | hypothetical protein | −1.371 | |||
| VC1560 | catalase/peroxidase | −1.450 | |||
| VC1563 | conserved hypothetical protein | 1.068 | |||
| VC1564 | hypothetical protein | 1.155 | |||
| VC1565 | tolC | outer membrane protein TolC putative | 1.202 | ||
| VC1581 | nuoL | NADH dehydrogenase putative | 2.736 | ||
| VC1582 | conserved hypothetical protein | 1.969 | |||
| VC1688 | hypothetical protein | −1.127 | |||
| VC1704 | metE | 5-methyltetrahydropteroyltriglutamate–homocysteine methyltransferase | 3.435 | ||
| VC1719 | torR | DNA-binding response regulator TorR | −1.718 | ||
| VC1731 | conserved hypothetical protein | −1.084 | |||
| VC1808 | hypothetical protein | 1.396 | |||
| VC1823 | fruA | PTS system fructose-specific IIB component | 1.385 | ||
| VC1865 | hypothetical protein | −1.376 | |||
| VC1871 | conserved hypothetical protein | −1.034 | |||
| VC1949 | pvcA | pvcA protein | 1.021 | ||
| VC1950 | biotin sulfoxide reductase | −1.785 | |||
| VC1951 | yecK | cytochrome c-type protein YecK | −1.854 | ||
| VC1956 | mltB | lytic murein transglycosylase putative | −1.242 | ||
| VC1957 | conserved hypothetical protein | −1.314 | |||
| VC1958 | hypothetical protein | −1.144 | |||
| VC1962 | lipoprotein | −1.070 | −1.215 | ||
| VC1971 | menE | o-succinylbenzoic acid–CoA ligase | 1.181 | ||
| VC1972 | menA | o-succinylbenzoate-CoA synthase | −1.587 | ||
| VC1973 | menB | naphthoate synthase | −2.445 | ||
| VC1974 | menH | conserved hypothetical protein | −2.129 | ||
| VC2001 | yeaD | conserved hypothetical protein | 1.019 | ||
| VC2007 | transcriptional regulator ROK family | 1.118 | |||
| VC2013 | ptsG | PTS system glucose-specific IIBC component | 1.038 | ||
| VC2036 | asd | aspartate-semialdehyde dehydrogenase | 1.069 | ||
| VC2045 | sodA | superoxide dismutase Fe | −1.249 | −1.328 | |
| VC2051 | ccmG | cytochrome c biogenesis protein | −1.131 | ||
| VC2052 | ccmF | cytochrome c-type biogenesis protein CcmF | −1.306 | ||
| VC2053 | ccmE | cytochrome c-type biogenesis protein CcmE | −1.828 | ||
| VC2054 | ccmD | heme exporter protein D | −1.708 | ||
| VC2055 | ccmC | heme exporter protein C | −1.490 | ||
| VC2076 | feoC | putative ferrous iron transport protein C | −1.421◊ | ||
| VC2077 | feoB | ferrous iron transport protein B | −1.489◊ | ||
| VC2078 | feoA | ferrous iron transport protein A | −1.172 | ||
| VC2149 | hypothetical protein | −1.007 | |||
| VC2174 | ushA | UDP-sugar hydrolase | 1.318 | ||
| VC2221 | hypothetical protein | 1.443 | |||
| VC2271 | ribD | riboflavin-specific deaminase | −1.385 | ||
| VC2272 | nrdR | conserved hypothetical protein | 1.858 | ||
| VC2323 | conserved hypothetical protein | −1.227 | |||
| VC2352 | NupC family protein | 1.381 | 1.164 | ||
| VC2357 | hypothetical protein | 1.362 | |||
| VC2361 | grcA | formate acetyl transferase-related protein | 1.163◊ | 1.092 | |
| VC2363 | thrB | homoserine kinase | 1.009 | ||
| VC2364 | thrA | aspartokinase I/homoserine dehydrogenase threonine-sensitive | 1.391 | ||
| VC2367 | hypothetical protein | −1.123 | |||
| VC2368 | arcA | aerobic respiration control protein FexA | −1.409 | ||
| VC2371 | conserved hypothetical protein | −1.303 | |||
| VC2372 | hypothetical protein | −1.395 | |||
| VC2373 | gltD | glutamate synthase large subunit | 1.126 | ||
| VC2417 | recJ | single-stranded-DNA-specific exonuclease RecJ | −1.098 | ||
| VC2418 | dsbC | thiol:disulfide interchange protein DsbC | −1.200 | ||
| VC2419 | xerD | integrase/recombinase XerD | −1.173 | ||
| VC2466 | rseA | sigma-E factor negative regulatory protein RseA | −1.130 | ||
| VC2486 | hypothetical protein | −1.035 | |||
| VC2490 | leuA | 2-isopropylmalate synthase | 1.135 | ||
| VC2508 | argF | ornithine carbamoyltransferase | −1.487 | ||
| VC2509 | hypothetical protein | −1.032 | |||
| VC2510 | pyrB1 | aspartate carbamoyltransferase catalytic subunit | 1.319 | ||
| VC2511 | pyrB2 | aspartate carbamoyltransferase regulatory subunit | 1.394 | ||
| VC2524 | ksdC | conserved hypothetical protein | −1.199 | ||
| VC2543 | hypothetical protein | 1.076 | |||
| VC2544 | fbp | fructose-16-bisphosphatase | 1.614 | ||
| VC2560 | cysN | sulfate adenylate transferase subunit 2 | 1.463 | ||
| VC2562 | cpdB | 2′3′-cyclic-nucleotide 2′-phosphodiesterase | 1.206 | ||
| VC2565 | elaA | elaA protein | −1.108 | ||
| VC2568 | fklB | peptidyl-prolyl cis-trans isomerase FKBP-type | 1.042 | ||
| VC2637 | peroxiredoxin family protein/glutaredoxin | −1.378 | |||
| VC2644 | argC | N-acetyl-gamma-glutamyl-phosphate reductase | −1.289 | ||
| VC2645 | argE | acetylornithine deacetylase | −1.080 | ||
| VC2656 | frdA | fumarate reductase flavoprotein subunit | 1.103 | ||
| VC2657 | frdB | fumarate reductase iron-sulfur protein | 1.360 | ||
| VC2658 | frdC | fumarate reductase 15 kDa hydrophobic protein | 1.708 | ||
| VC2659 | frdD | fumarate reductase 13 kDa hydrophobic protein | 1.699 | ||
| VC2674 | hslU | protease HslVU ATPase subunit HslU | −1.330 | ||
| VC2675 | hslV | protease HslVU subunit HslV | −1.258 | ||
| VC2689 | pfkA | 6-phosphofructokinase isozyme I | −1.076 | ||
| VC2699 | dcuA | C4-dicarboxylate transporter anaerobic | 1.040 | ||
| VC2706 | conserved hypothetical protein | 1.577 | 1.529 | ||
| VC2720 | nfuA | conserved hypothetical protein | −1.197 | −1.084 | |
| VC2738 | pckA | phosphoenolpyruvate carboxykinase | 1.086 | ||
| VCA0011 | malT | malT regulatory protein | 1.882 | ||
| VCA0013 | malP | maltodextrin phosphorylase | 1.713 | ||
| VCA0014 | malQ | 4-alpha-glucanotransferase | 1.698 | ||
| VCA0015 | hypothetical protein | 1.630 | |||
| VCA0016 | 14-alpha-glucan branching enzyme | 1.642 | |||
| VCA0025 | transporter NadC family | 1.244 | |||
| VCA0053 | ppnP | purine nucleoside phosphorylase | 1.062 | ||
| VCA0087 | hypothetical protein | −1.004 | |||
| VCA0139 | hypothetical protein | −1.146 | −1.236 | ||
| VCA0180 | pepT | peptidase T | −1.364 | ||
| VCA0205 | C4-dicarboxylate transporter anaerobic | 1.170 | 1.136 | ||
| VCA0216 | hypothetical membrane, linked to VCA0215 and VCA0217 | −1.395 | |||
| VCA0231 | vctR | linked to vctA, function unknown | −1.327 | ||
| VCA0245 | cmtB | PTS system IIA component | 1.105 | ||
| VCA0246 | sgaT | SgaT protein | 1.073 | ||
| VCA0268 | methyl-accepting chemotaxis protein | −1.056 | 1.152 | ||
| VCA0269 | decarboxylase group II | 1.218 | |||
| VCA0344 | hypothetical protein | 1.012 | |||
| VCA0511 | nrdG | anaerobic ribonucleoside-triphosphate reductase | 1.175 | ||
| VCA0516 | ptsIIB | PTS system fructose-specific IIBC component | 2.838 | ||
| VCA0517 | fruK | 1-phosphofructokinase | 1.948 | −1.919 | |
| VCA0518 | ptsIIA | PTS system fructose-specific IIA/FPR component | 1.778 | −1.113 | |
| VCA0523 | cqsA | aminotransferase class II | 2.585 | ||
| VCA0540 | formate transporter 1 putative | −2.633 | −4.612 | ||
| VCA0550 | hypothetical protein | −1.096 | |||
| VCA0551 | hypothetical protein | −1.394 | |||
| VCA0592 | nudG | MutT/nudix family protein | 1.661 | ||
| VCA0621 | transcriptional regulator SorC family | −1.283 | |||
| VCA0628 | SecA-related protein | 1.536 | |||
| VCA0665 | dcuC | C4-dicarboxylate transporter anaerobic | −1.512 | ||
| VCA0721 | hypothetical protein | −1.014 | |||
| VCA0752 | trx2 | thioredoxin 2 | −1.252 | ||
| VCA0773 | methyl-accepting chemotaxis protein | 1.209 | |||
| VCA0784 | hypothetical protein | −1.566 | |||
| VCA0819 | groES | chaperonin 10 Kd subunit | −1.227 | ||
| VCA0820 | groEL | chaperonin 60 Kd subunit | −1.119 | ||
| VCA0821 | hypothetical protein | −1.116 | |||
| VCA0823 | ectC | ectoine synthase | 1.304 | ||
| VCA0824 | ectB | diaminobutyrate–pyruvate aminotransferase | 1.820 | ||
| VCA0825 | ectA | L-24-diaminobutyric acid acetyltransferase | 1.691 | ||
| VCA0867 | ompW | outer membrane protein OmpW | 1.639 | ||
| VCA0897 | devB | devB protein | −1.127 | ||
| VCA0898 | gnd | 6-phosphogluconate dehydrogenase decarboxylating | −1.401 | −1.262 | |
| VCA0907 | hutZ | heme binding | −1.430 | −1.047 | |
| VCA0908 | hutX | Unknown, linked to hutZ | −1.626 | −1.091 | |
| VCA0909 | hutW | unknown, linked to hutZ | −3.049◊ | ||
| VCA0910 | tonB1 | tonB1 protein | −3.208◊ | ||
| VCA0911 | exbB1 | TonB system transport protein ExbB1 | −3.328◊ | ||
| VCA0912 | exbD1 | TonB system transport protein ExbD1 | −2.996◊ | 2.023 | |
| VCA0913 | hutB1 | hemin ABC transporter%2 C periplasmic hemin-binding protein HutB | −2.383◊ | ||
| VCA0914 | hutB2 | hemin ABC transporter%2 C permease protein%2 C putative | −1.808◊ | ||
| VCA0944 | malF | maltose ABC transporter permease protein | 1.853 | ||
| VCA0945 | malE | maltose ABC transporter periplasmic maltose-binding protein | 1.986 | ||
| VCA0954 | cheV | chemotaxis protein CheV putative | −1.029 | ||
| VCA0965 | GGDEF family protein | −1.396 | |||
| VCA0966 | hypothetical protein | −1.335 | |||
| VCA0967 | hypothetical protein | −1.507 | −1.135 | ||
| VCA0968 | hypothetical protein | −1.527 | −1.190 | ||
| VCA0979 | methyl-accepting chemotaxis protein | 1.006 | |||
| VCA0981 | hypothetical protein | 1.008 | |||
| VCA0985 | oxidoreductase/iron-sulfur cluster-binding protein | −1.381 | |||
| VCA0988 | methyl-accepting chemotaxis protein | −1.119 | |||
| VCA1006 | organic hydroperoxide resistance protein putative | −1.130 | |||
| VCA1007 | hypothetical protein | −1.064 | |||
| VCA1009 | hypothetical protein | −1.260 | |||
| VCA1010 | conserved hypothetical protein | −3.403 | |||
| VCA1014 | hypothetical protein | 1.080 | |||
| VCA1027 | malM | maltose operon periplasmic protein putative | 1.060 | ||
| VCA1028 | lamB | maltoporin | 2.485 | ||
| VCA1060 | ribB | 34-dihydroxy-2-butanone 4-phosphate synthase | −3.58 | 1.476 | 2.938 |
| VCA1063 | speC | ornithine decarboxylase inducible | 1.067 | ||
| VCA1064 | hypothetical protein | 1.366 | |||
| VCA1069 | methyl-accepting chemotaxis protein | 1.383 | |||
| VCA1099 | ABC transporter permease protein | 1.081 | |||
A total of 277 genes are differentially expressed in response to at least one of the three first conditions compared (Table 1). The results of the indicated comparisons is summarized as a Venn diagram in Fig. 2. 31 regulated genes were differentially expressed in the WT strain in response to extracellular riboflavin (Table 1). 177 genes were significantly affected by the mutation in ribD, of which 34 were also affected in the ribN mutant. A total of 108 genes were affected by the elimination of ribN growing in riboflavin, 74 of which were not affected by the ribD elimination. One gene was affected in the three comparisons, which corresponded to the FMN riboswitch-regulated ribB (VCA1060). These data are consistent with the notion that although the functions of riboflavin biosynthesis and transport through RibN overlap, there may also exist specific functions for each riboflavin provision pathway.
Figure 2.
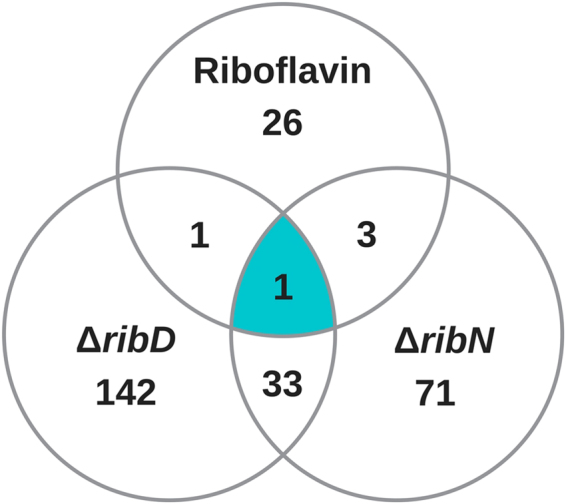
Summary of the results of transcriptomics comparisons. Venn diagram showing the distribution of the genes affected by exogenous riboflavin, the deletion of the riboflavin biosynthetic gene ribD and the deletion of the riboflavin transporter gene ribN, as determined by transcriptomics.
The riboflavin regulon of V. cholerae includes many iron regulated genes
The first transcriptomic comparison assessed the effect of riboflavin in the WT strain. The gene ribB was found at the top of the list of genes regulated by riboflavin, being highly repressed. This pattern is consistent with our previous report, although the degree of repression (roughly 12-fold) was higher than in our earlier determination (2-fold decrease)38. A previous RNA microarray study identified 84 genes regulated by iron in V. cholerae46. Most of the genes identified here as responding to riboflavin, are also members of such iron regulon (21 out of 31). V. cholerae possesses several transport systems dedicated to the uptake of various iron forms. These include the genes for synthesis and utilization of the vibriobactin siderophore, the ferrous iron transport system FeoABC, the ferric iron acquisition system FbpABC, the Hut heme transport and the VctPDGC system47–49. Likely, these systems are differentially required depending on the iron source available on each stage of the V. cholerae life cycle49. Genes related to most of these iron acquisition systems, except for the VctPDGC system, were found to be moderately repressed by riboflavin (Table 1), while all of such systems are known to be repressed by iron46,48. Other genes belonging to the iron regulon that were also detected responding to extracellular riboflavin included the bacterioferritin operon bfd-bfr and a few proteins with unknown function like the encoded by the VC1264, VC1266 and VC0143 open reading frames (ORFs). In addition, ybtA, coding for a member of the AraC family of transcriptional regulators involved in regulation of siderophore production in Yersinia50, was also repressed by riboflavin. Genes identified here which have not previously reported to be regulated by iron include hutC, coding for a transcriptional regulator of the histine utilization operon, glcA, coding for the autonomous glycyl radical cofactor protein and two methyl-accepting chemotaxis protein genes. While most of the genes identified in this comparison were repressed by riboflavin, hutC and glcA were activated. In our previous study, contrary to its effect on the WT strain, riboflavin induced the expression of ribB in a ∆ribN strain45. This suggested that riboflavin may induce changes in transcription in a manner independent of its internalization through RibN. For this reason, in order to globally identify effects of riboflavin independent of its uptake through RibN, we included a comparison of the transcriptomes in response to riboflavin in the ∆ribN strain. This analysis revealed that 16 of the genes affected by riboflavin in the WT are also affected in this strain (indicated by asterisks in Table 1 and full list in Table S1). This suggests that at least for these cases, the regulatory effect of riboflavin is independent of its internalization through RibN. In order to identify general functional relationships among the genes responsive to riboflavin, we performed analysis of enrichment of Gene Ontology (GO) terms of biological processes associated to this set. Such analysis seek to identify functional terms, as defined by the PANTHER classification system, overrepresented in a given group of genes51,52. Three GO biological processes were found statistically overrepresented (P < 0.05). These corresponded to iron ion transmembrane transport, cellular responses to iron ion and iron ion homeostasis.
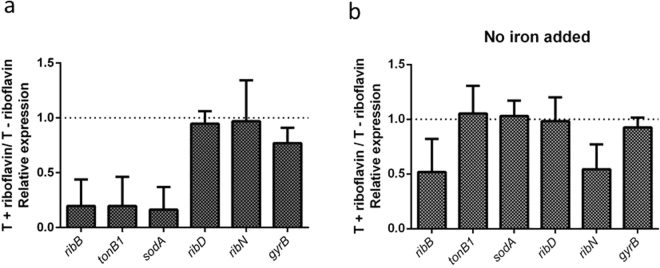
To validate the transcriptomic comparison, we determined the relative expression of ribB and tonB1 in T medium and T plus riboflavin by RT-PCR. The tonB1 gene encodes a component of one of the two TonB-ExbB-ExbD complexes that harness membrane proton motive force for its heterologous use in various iron transport systems in V. cholerae47. Thus, it seems to be an adequate gene to monitor the expression of iron acquisition systems. The expression of ribB and tonB1 was reduced 4-fold in response to added riboflavin (Fig. 3a). This is in agreement with the transcriptomics results although a higher effect of riboflavin was detected by RT-PCR. To assess if there may be additional genes known to be regulated by iron that are also regulated by riboflavin but missed in our transcriptomics analysis, we determined the expression of sodA. This gene is known to be repressed by iron46. Notably, the expression of sodA was reduced 4.13-fold by riboflavin. As controls, we determined the expression of the riboflavin biosynthetic gene ribD and of the ribN gene. We have previously demonstrated that the expression of these genes is not affected by riboflavin in standard T media38 and their expression did not change in response to riboflavin in our transcriptomics results. Accordingly, the expression of these two genes was not affected by riboflavin as determined by RT-PCR. One additional control was used, gyrB, which was not affected by the presence of exogenous riboflavin according to transcriptomics. The RNA of this gene was only slighty reduced by riboflavin (0.29-fold) as determined by RT-PCR.
Figure 3.
Effect of riboflavin on the expression of genes under different iron conditions. Relative expression of the indicated genes with and without riboflavin in T media (a) or T without added iron (b), as determined by RT-PCR. WT V. cholerae was grown until medium exponential phase at 37 °C, RNA extracted and RT-PCR assayed as described in Materials and Methods. Results shown are the average and standard deviation of three independent experiments.
Riboflavin and iron reciprocally regulate their provision genes
Thus far, results indicate that riboflavin regulates many genes that are also regulated by iron. The experiments were performed in standard T media. The recipe for this medium includes 20 µM FeCl and may be considered an iron-replete condition when compared to minimal media without added iron46,53. It is reported that in such conditions, the iron aquisiton systems of V. cholerae are mainly repressed47. Thus, in the case of iron uptake genes, riboflavin seems to enhance the repression produced in high iron conditions. Along these lines, we aimed to determine the effect of riboflavin on the expression of iron regulated genes under iron-restrictive conditions. These conditions are known to induce the expression of iron uptake systems. For this, we grew V. cholerae in T media without any added iron and determined the effect of riboflavin by RT-PCR. Notably, the expression of tonB1 and sodA, as well as that of the controls ribD and gyrB, remained around the same with or without riboflavin in such iron-restrictive conditions (Fig. 3b). This may indicate that the negative modulatory effect of riboflavin is surpassed under the highly inducing conditions triggered by iron restriction. Strikingly, the expression of ribN was diminished by half by riboflavin in this condition, in contrast with the results obtained in iron repleted media, were riboflavin has no effect. This suggests that riboflavin modulates the expression of ribN but only during iron restriction. To corroborate that tonB1 is being induced in response to iron restriction in our experiments, we compared the expression of this gene when growing without iron versus standard T media. We assessed this in media with and without riboflavin. Irrespective of the presence of riboflavin, the expression of tonB1 is highly increased (more than 10-fold) in low iron media, although the induction effect was higher without riboflavin (Fig. 4). Remarkably, although iron has no effect in the expression of ribD when riboflavin is present, in riboflavin-free media this gene was highly repressed in iron-restrictive conditions. The same occurred for the ribN gene. Nonetheless, a different effect applied for the ribB gene. In the absence of riboflavin, iron had no effect, while iron restriction increased the expression of this gene 3-fold in the presence of exogenous riboflavin. Collectively, results indicate that riboflavin and iron interplay affects the expression of iron and riboflavin provision genes in a gene-specific manner.
Figure 4.
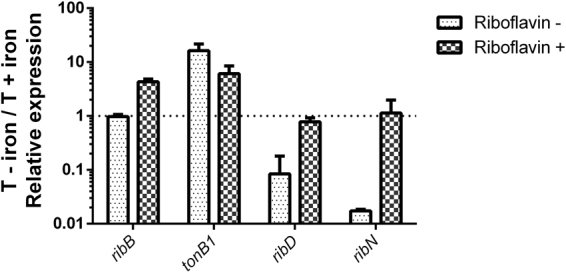
Effect of iron in the expression of genes under different riboflavin conditions. The relative expression of the genes in T without iron versus complete T, with and without riboflavin as indicated. Growth conditions were similar as those described in Fig. 3. Results shown are the average and standard deviation of three independent experiments.
Genes affected both in the ribD and ribN mutants
We have recently hypothesized that riboflavin transport, instead of merely replacing for the RBP, may afford riboflavin for specific physiological functions23. The results of the transcriptomics comparisons performed here show that 34 of the genes whose expression is affected by the elimination of riboflavin biosynthesis are also affected by the elimination of the RibN importer (Table S2). This clearly suggests that functional overlap between riboflavin biosynthesis and internalization occurs. Five of these genes belong to the iron regulon. These are the VC1515, VCA1516, VC1514, VCA0908 and VCA0907 ORFs. VC1514 encodes a protein of unknown function putatively encoded in the same operon as VC1515 and VC1516. The latter genes code for a putative chaperone of a formate dehydrogenase and an iron-sulfur cluster binding protein, respectively. Thus, this system seems to be involved in redox reactions although its exact function is unknown. The transcription of these genes is increased in both ribD and ribN mutants. VCA908 and VCA907 are encoded within a putative operon that codes for proteins involved in heme utilization. These ORFs were found to be downregulated in response to ribD and ribN eliminations. The set of genes regulated in the two conditions also included ribB, which was upregulated as a result of both mutations. As this gene conserves an FMN riboswitch that represses expression in response to FMN, its induction probably reflects a reduction in intracellular riboflavin levels produced in both mutants. Genes encoding proteins involved in amino acids metabolism, such as VC0162 coding for a ketol-acid reductoisomerase and VC0027, coding for threonine dehydratase, as well as enzymes involved in redox metabolism such as VC2045, coding for a superoxide dismutase and VC0753, encoding a ferredoxin, were also included in this subgroup. An analysis of enrichment of GO terms of biological processes associated to this set rendered no significant overrepresentation.
Most of the genes within this group followed the same pattern of regulation irrespective of whether the elimination was on ribD or ribN. However, three genes were differentially affected by the mutations. VCA0517 and VCA0518, encoded in an operon of a phosphotransferase system for fructose transport, were upregulated roughly 3.5 times in the ∆ribD strain but downregulated 3.7 and 2.1 times respectively in the ∆ribN strain. Likewise, the gene for OmpU, one of the major porins in this species, was upregulated 2.5 times in the ∆ribD strain but downregulated 2.3 times in the ∆ribN strain. These represent an intriguing group, as the transcription of these genes seems to be reciprocally regulated by riboflavin biosynthesis and riboflavin uptake through RibN.
Genes specifically affected in response to ribD elimination
The transcription of 139 genes was significantly affected by the elimination of ribD but not by the elimination of ribN. This comprised the largest set of genes defined by any of our comparisons (Table S3). The VC1279 ORF, encoding a putative member of the Betaine/Carnitine/Choline Transporters (BCCT) family, was the highest regulated gene, being induced 29.6 times in response to ribD elimination. Also atop of the list were the genes VC1704, encoding a 5-methyltetrahydropteroyltriglutamate-homocysteine methyltransferase required for cystein and methionin biosynthesis and VC0734, coding for a malate synthase. The list included some iron regulated genes, such as exbD1, related to the TonB1 system and many genes related to the PTS system for fructose and glucose uptake. In the GO terms enrichment analysis for this subset two terms were overrepresented: protein folding and oxidation-reductions process.
Genes specifically affected in response to ribN mutation
We identified genes whose expression changed in the ribN mutant but not in the ribD mutant. 73 genes corresponded to this pattern of regulation (Table S4). In this list two genes involved in heme export, VC2054 and VC2055 were downregulated, which could also be related to the riboflavin-iron metabolic interplay highlighted across the transcriptomics results. Notably, many ribosome assembly genes also appeared in this set of genes. Among the most regulated genes are menB and menH, both involved in menaquinone (vitamin K) biosynthesis, bioD, required for biotin biosynthesis, VC1950, which encodes a biotin sulfoxide reductase that allows biotin salvatage and yecK and ccmE, two genes involved in cytochrome c-type biogenesis. The list included other genes also involved in biotin biosynthesis and cytochrome c-type biogenesis such as bioC and ccmF, respectively. All of these proteins were downregulated in the ∆ribN strain. Thus it seems that the lack of transport of riboflavin through RibN downregulates menaquinone, biotin and cytochrome c biosynthesis. Accordingly, the GO term cytochrome complex assembly was significantly enriched in this subset of genes. Menaquinone and cytochromes participate in electron transfer chains. Notably, the ArcA response regulator that control aerobic respiration was also downregulated. Thus, these results suggest that internalized riboflavin may be involved in respiratory chain processes in V. cholerae.
Discussion
This study assessed the effect of riboflavin on gene expression in V. cholerae. Many of the genes affected by riboflavin are known to be regulated by the iron levels in the media. The determination of the expression of genes by RT-PCR added sodA to the list of genes downregulated by riboflavin. Thus, our transcriptomics analysis may be underestimating the number of genes regulated by riboflavin and the overlapping of iron and riboflavin regulons could be more extensive. Genes belonging to five out of six iron acquisition systems present in this species were negatively modulated by the presence of riboflavin in T media. These systems are known to be repressed in iron-rich environments and induced under iron deprivement. When assessed its effect in low iron, riboflavin no longer repressed iron regulated genes. Thus, riboflavin seems to accent a high iron condition in the expression of iron uptake systems and possibly other iron regulated genes, while having no repressive effect during iron starvation. Contrarywise, the riboflavin transport gene ribN, which is expressed independently of riboflavin in T media with iron, was negatively modulated by exogenous riboflavin during iron starvation. Reciprocally, iron repressed the expression of ribD and ribN but only in the absence of exogenous riboflavin, while inducing the expression of ribB in the presence of riboflavin. These three genes are encoded in separated transcriptional elements. Noteworthy, ribB is the only one conserving a FMN riboswitch38, likely rendering the expression of this gene coupled to the levels of intracellular riboflavin. This may be responsible for its differential regulation. The increase in expression of ribB in low iron may reflex a decrease in intracellular riboflavin levels. This may seem paradoxical given that this effect only occurs in the presence of extracellular riboflavin. However, we have previously observed an increase in ribB transcription in the presence of riboflavin in a ribN mutant38, and such result replicated in this transcriptomics analysis. This suggested that the presence of extracellular riboflavin increases intracellular riboflavin requirements. Thus, this increase may be exacerbated in low iron conditions, which may explain this result. In general, the expression of iron and riboflavin provision genes was found to be modulated by the presence of both iron and riboflavin in the media in a coordinated fashion. At least in the case of riboflavin provision genes, this regulation is gene-specific. Altogether, this may reflex a paramount regulatory crosstalk between the two most important redox cofactors in nature. The iron-riboflavin interregulatory effect may be common also in other bacteria. RBP genes have been found upregulated under iron-deficiency conditions by high throughput approaches in different bacteria such as Caulobacter crescentus54, Methylocystis55 and Clostridium acetobutylicum56. The physiological relevance of this is unknown. One probable explanation is that in these species the lack of iron could be compensated by enhancing the biosynthesis of riboflavin, another important redox cofactor. Indeed, flavodoxins seem to substitute for ferredoxins in electron transfer reactions under iron starving conditions in different organisms across kingdoms57–60. Nonetheless in V. vulnificus, a bacterial species philogenetically related to V. cholerae, the RBP genes are downregulated under iron restriction61, which is a similar effect to what we observed in this study for ribD and ribN. Our work provided evidence of the reciprocal phenomenon for the first time, in which the availability of riboflavin alters iron metabolism in bacteria. Altogether, the overlay between riboflavin and iron regulons suggests the existence of a network interconnecting riboflavin and iron homeostasis and probably a common regulatory mechanism. This seems an important feature that grants further study.
The way riboflavin biosynthesis and uptake correlate to fulfill the flavin needs in riboflavin opportunistic species is still unclear. Nonetheless, some studies shed light into the role of riboflavin transporters in bacterial physiology. The RibM riboflavin importer is able to provide flavins to a RBP-deficient mutant of Corynebacterium glutamicum when growing with extracellular riboflavin, albeit the levels of the intracellular riboflavin pools are lower than those in the WT strain62. In Staphylococcus aureus, the Energy coupling factor (ECF)-RibU riboflavin uptake system is able to fully substitute for the RBP during in vitro growth with riboflavin traces and also during mouse infection63. Overexpression of RibU, the substrate-binding component of this system, helps overcome heat stress in Lactococcus lactis64,65. The RfuABCD riboflavin uptake system in Borrelia burgdorferi is required to set an efficient oxidative stress response and for colonization in the murine model66. In the case of RibN, it is required for full colonization of pea plant roots at early stages by the riboflavin prototroph Rhizobium leguminosarum40. In V. cholerae, riboflavin biosynthesis is sufficient to grow in river water but RibN provides a competitive advantage45. Here, transcriptome comparisons suggest that riboflavin biosynthesis and uptake have common and specific effects in gene transcription, which may be related to functions performed by these two riboflavin provision pathways. Remarkably, GO functional terms were distinctively defined in the subsets affected by each deletion. While protein folding and oxidation-reductions process were enriched in the genes specifically affected by the lack of riboflavin biosynthesis, cytochrome complex assembly was enriched in the set of genes pointedly affected by the ribN mutation. Other genes involved in electron transport chain were also affected in the ∆ribN specific set. Hence, this study may serve as a start point to characterize cellular functions requiring exogenous riboflavin in this species. Notably, the number of genes affected by the elimination of riboflavin biosynthesis was significantly higher than those affected by the presence of external riboflavin or abrogation of RibN. This may pose that biosynthesized riboflavin is engaged in more physiological functions than exogenous riboflavin. The fact that extracellular riboflavin downregulates the monicistronically encoded ribB but does not affects the expression of the main RBP operon on which other ribB homolog may be encoded also supports this view38. This effect could allow to retain the capacity to fully biosynthesize riboflavin in the presence of exogenous riboflavin. Importantly, the elimination of RibN does not necessarily abolish riboflavin uptake, as the presence of additional riboflavin transport systems has not been experimentally determined in this strain. This could be accomplished by the determination of the levels of riboflavin needed to support growth in a double ∆ribD/∆ribN strain. However, our attempts to obtain such strain have failed even in the presence of high riboflavin concentrations in the media. Nonetheless, the increase in expression of ribB induced by exogenous riboflavin in the ribN mutant may suggest that riboflavin is not entering the cell by a different transporter.
Collectively, this study comprises an integral analysis of the response induced by availability of riboflavin in V. cholerae on what constitutes, to the best of our knowledge, the first approach to a riboflavin regulon in bacteria.
Electronic supplementary material
Acknowledgements
We thank Daniela Gutiérrez for logistic support for transcriptomics analysis. This work was funded by CONICYT-FONDECYT (Chile) Grant Number 1150818.
Author Contributions
I.S.C. performed cultures, RNA extractions, RT-PCR, conceived experiments and analized results. L.L.A. performed transcriptomics analysis, analized results and contributed to paper writing. A.F.F. discussed results and provided technical support in experiments. I.V.S.D.O. analyzed results and prepared tables. V.A.G.A. conceived the study, analized results and wrote the paper.
Competing Interests
The authors declare no competing interests.
Footnotes
Ignacio Sepúlveda-Cisternas and Luis Lozano Aguirre contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-21302-3.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bilan DS, Belousov VV. Genetically encoded probes for NAD +/NADH monitoring. Free Radic. Biol. Med. 2016;100:32–42. doi: 10.1016/j.freeradbiomed.2016.06.018. [DOI] [PubMed] [Google Scholar]
- 2.Mellor SB, Vavitsas K, Nielsen AZ, Jensen PE. Photosynthetic fuel for heterologous enzymes: the role of electron carrier proteins. Photosynth. Res. 2017;134:329–342. doi: 10.1007/s11120-017-0364-0. [DOI] [PubMed] [Google Scholar]
- 3.Sánchez M, Sabio L, Gálvez N, Capdevila M, Dominguez-Vera JM. Iron chemistry at the service of life. IUBMB Life. 2017;69:382–388. doi: 10.1002/iub.1602. [DOI] [PubMed] [Google Scholar]
- 4.Gnandt E, Dörner K, Strampraad MFJ, de Vries S, Friedrich T. The multitude of iron–sulfur clusters in respiratory complex I. EBEC 2016 19th Eur. Bioenerg. Conf. 2016;1857:1068–1072. doi: 10.1016/j.bbabio.2016.02.018. [DOI] [PubMed] [Google Scholar]
- 5.Greening C, et al. Physiology, Biochemistry, and Applications of F420- and Fo-Dependent Redox Reactions. Microbiol. Mol. Biol. Rev. MMBR. 2016;80:451–493. doi: 10.1128/MMBR.00070-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Monteverde DR, Gómez-Consarnau L, Suffridge C, Sañudo-Wilhelmy SA. Life’s utilization of B vitamins on early Earth. Geobiology. 2017;15:3–18. doi: 10.1111/gbi.12202. [DOI] [PubMed] [Google Scholar]
- 7.Palmer LD, Skaar EP. Transition Metals and Virulence in Bacteria. Annu. Rev. Genet. 2016;50:67–91. doi: 10.1146/annurev-genet-120215-035146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Paul CE, Hollmann F. A survey of synthetic nicotinamide cofactors in enzymatic processes. Appl. Microbiol. Biotechnol. 2016;100:4773–4778. doi: 10.1007/s00253-016-7500-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Macheroux P, Kappes B, Ealick SE. Flavogenomics–a genomic and structural view of flavin-dependent proteins. FEBS J. 2011;278:2625–2634. doi: 10.1111/j.1742-4658.2011.08202.x. [DOI] [PubMed] [Google Scholar]
- 10.Peters JW, Miller A-F, Jones AK, King PW, Adams MW. Electron bifurcation. Curr. Opin. Chem. Biol. 2016;31:146–152. doi: 10.1016/j.cbpa.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 11.Piano V, Palfey BA, Mattevi A. Flavins as Covalent Catalysts: New Mechanisms Emerge. Trends Biochem. Sci. 2017;42:457–469. doi: 10.1016/j.tibs.2017.02.005. [DOI] [PubMed] [Google Scholar]
- 12.Rajamani S, et al. The vitamin riboflavin and its derivative lumichrome activate the LasR bacterial quorum-sensing receptor. Mol. Plant-Microbe Interact. MPMI. 2008;21:1184–1192. doi: 10.1094/MPMI-21-9-1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim BJ, et al. Oxygen Tension and Riboflavin Gradients Cooperatively Regulate the Migration of Shewanella oneidensis MR-1 Revealed by a Hydrogel-Based Microfluidic Device. Front. Microbiol. 2016;7:1438. doi: 10.3389/fmicb.2016.01438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Crossley RA, et al. Riboflavin biosynthesis is associated with assimilatory ferric reduction and iron acquisition by Campylobacter jejuni. Appl. Environ. Microbiol. 2007;73:7819–7825. doi: 10.1128/AEM.01919-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.von Canstein H, Ogawa J, Shimizu S, Lloyd JR. Secretion of flavins by Shewanella species and their role in extracellular electron transfer. Appl. Environ. Microbiol. 2008;74:615–623. doi: 10.1128/AEM.01387-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Okamoto A, Hashimoto K, Nealson KH, Nakamura R. Rate enhancement of bacterial extracellular electron transport involves bound flavin semiquinones. Proc. Natl. Acad. Sci. USA. 2013;110:7856–7861. doi: 10.1073/pnas.1220823110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wu C, et al. Electron acceptor dependence of electron shuttle secretion and extracellular electron transfer by Shewanella oneidensis MR-1. Bioresour. Technol. 2013;136:711–714. doi: 10.1016/j.biortech.2013.02.072. [DOI] [PubMed] [Google Scholar]
- 18.Magnúsdóttir S, Ravcheev D, de Crécy-Lagard V, Thiele I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 2015;6:148. doi: 10.3389/fgene.2015.00148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Meseguer AS, et al. Buchnera has changed flatmate but the repeated replacement of co-obligate symbionts is not associated with the ecological expansions of their aphid hosts. Mol. Ecol. 2017;26:2363–2378. doi: 10.1111/mec.13910. [DOI] [PubMed] [Google Scholar]
- 20.Abbas CA, Sibirny AA. Genetic control of biosynthesis and transport of riboflavin and flavin nucleotides and construction of robust biotechnological producers. Microbiol. Mol. Biol. Rev. MMBR. 2011;75:321–360. doi: 10.1128/MMBR.00030-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Haase I, Gräwert T, Illarionov B, Bacher A, Fischer M. Recent advances in riboflavin biosynthesis. Methods Mol. Biol. Clifton NJ. 2014;1146:15–40. doi: 10.1007/978-1-4939-0452-5_2. [DOI] [PubMed] [Google Scholar]
- 22.Vitreschak AG, Rodionov DA, Mironov AA, Gelfand MS. Regulation of riboflavin biosynthesis and transport genes in bacteria by transcriptional and translational attenuation. Nucleic Acids Res. 2002;30:3141–3151. doi: 10.1093/nar/gkf433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.García-Angulo VA. Overlapping riboflavin supply pathways in bacteria. Crit. Rev. Microbiol. 2017;43:196–209. doi: 10.1080/1040841X.2016.1192578. [DOI] [PubMed] [Google Scholar]
- 24.Bonomi HR, et al. An atypical riboflavin pathway is essential for Brucella abortus virulence. PloS One. 2010;5:e9435. doi: 10.1371/journal.pone.0009435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Frelin O, et al. A directed-overflow and damage-control N-glycosidase in riboflavin biosynthesis. Biochem. J. 2015;466:137–145. doi: 10.1042/BJ20141237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hemberger S, et al. RibM from Streptomyces davawensis is a riboflavin/roseoflavin transporter and may be useful for the optimization of riboflavin production strains. BMC Biotechnol. 2011;11:119. doi: 10.1186/1472-6750-11-119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gutiérrez-Preciado A, et al. Extensive Identification of Bacterial Riboflavin Transporters and Their Distribution across Bacterial Species. PloS One. 2015;10:e0126124. doi: 10.1371/journal.pone.0126124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jaehme M, Slotboom DJ. Diversity of membrane transport proteins for vitamins in bacteria and archaea. Biochim. Biophys. Acta. 2015;1850:565–576. doi: 10.1016/j.bbagen.2014.05.006. [DOI] [PubMed] [Google Scholar]
- 29.Clemens, J. D., Nair, G. B., Ahmed, T., Qadri, F. & Holmgren, J. Cholera. Lancet Lond. Engl. 10.1016/S0140-6736(17)30559-7 (2017). [DOI] [PubMed]
- 30.Ali M, Nelson AR, Lopez AL, Sack DA. Updated global burden of cholera in endemic countries. PLoS Negl. Trop. Dis. 2015;9:e0003832. doi: 10.1371/journal.pntd.0003832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chowdhury FR, Nur Z, Hassan N, von Seidlein L, Dunachie S. Pandemics, pathogenicity and changing molecular epidemiology of cholera in the era of global warming. Ann. Clin. Microbiol. Antimicrob. 2017;16:10. doi: 10.1186/s12941-017-0185-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shapiro BJ, Levade I, Kovacikova G, Taylor RK, Almagro-Moreno S. Origins of pandemic Vibrio cholerae from environmental gene pools. Nat. Microbiol. 2016;2:16240. doi: 10.1038/nmicrobiol.2016.240. [DOI] [PubMed] [Google Scholar]
- 33.Conner, J. G., Teschler, J. K., Jones, C. J. & Yildiz, F. H. Staying Alive: Vibrio cholerae’s Cycle of Environmental Survival, Transmission, and Dissemination. Microbiol. Spectr. 4 (2016). [DOI] [PMC free article] [PubMed]
- 34.Silva AJ, Benitez JA. Vibrio cholerae Biofilms and Cholera Pathogenesis. PLoS Negl. Trop. Dis. 2016;10:e0004330. doi: 10.1371/journal.pntd.0004330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jubair M, Morris JG, Ali A. Survival of Vibrio cholerae in nutrient-poor environments is associated with a novel ‘persister’ phenotype. PloS One. 2012;7:e45187. doi: 10.1371/journal.pone.0045187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Farhana I, Hossain ZZ, Tulsiani SM, Jensen PKM, Begum A. Survival of Vibrio cholerae O1 on fomites. World J. Microbiol. Biotechnol. 2016;32:146. doi: 10.1007/s11274-016-2100-x. [DOI] [PubMed] [Google Scholar]
- 37.Islam Z, Kumar A, Singh S, Salmon L, Karthikeyan S. Structural basis for competitive inhibition of 3,4-dihydroxy-2-butanone-4-phosphate synthase from Vibrio cholerae. J. Biol. Chem. 2015;290:11293–11308. doi: 10.1074/jbc.M114.611830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cisternas IS, Torres A, Flores AF, Angulo VAG. Differential regulation of riboflavin supply genes in Vibrio cholerae. Gut Pathog. 2017;9:10. doi: 10.1186/s13099-017-0159-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chao MC, et al. High-resolution definition of the Vibrio cholerae essential gene set with hidden Markov model-based analyses of transposon-insertion sequencing data. Nucleic Acids Res. 2013;41:9033–9048. doi: 10.1093/nar/gkt654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.García Angulo VA, et al. Identification and characterization of RibN, a novel family of riboflavin transporters from Rhizobium leguminosarum and other proteobacteria. J. Bacteriol. 2013;195:4611–4619. doi: 10.1128/JB.00644-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wyckoff EE, Payne SM. The Vibrio cholerae VctPDGC system transports catechol siderophores and a siderophore-free iron ligand. Mol. Microbiol. 2011;81:1446–1458. doi: 10.1111/j.1365-2958.2011.07775.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods San Diego Calif. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 43.Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat. Methods. 2012;9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Robinson MD, McCarthy DJ, Smyth GK. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinforma. Oxf. Engl. 2010;26:139–140. doi: 10.1093/bioinformatics/btp616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fuentes Flores A, Sepúlveda Cisternas I, Vásquez Solis de Ovando JI, Torres A, García-Angulo VA. Contribution of riboflavin supply pathways to Vibrio cholerae in different environments. Gut Pathog. 2017;9:64. doi: 10.1186/s13099-017-0214-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mey AR, Wyckoff EE, Kanukurthy V, Fisher CR, Payne SM. Iron and fur regulation in Vibrio cholerae and the role of fur in virulence. Infect. Immun. 2005;73:8167–8178. doi: 10.1128/IAI.73.12.8167-8178.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wyckoff EE, Mey AR, Payne SM. Iron acquisition in Vibrio cholerae. Biometals Int. J. Role Met. Ions Biol. Biochem. Med. 2007;20:405–416. doi: 10.1007/s10534-006-9073-4. [DOI] [PubMed] [Google Scholar]
- 48.Payne SM, Mey AR, Wyckoff EE. Vibrio Iron Transport: Evolutionary Adaptation to Life in MultipleEnvironments. Microbiol. Mol. Biol. Rev. MMBR. 2016;80:69–90. doi: 10.1128/MMBR.00046-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Peng ED, Wyckoff EE, Mey AR, Fisher CR, Payne SM. Nonredundant Roles of Iron Acquisition Systems in Vibrio cholerae. Infect. Immun. 2016;84:511–523. doi: 10.1128/IAI.01301-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Anisimov R, Brem D, Heesemann J, Rakin A. Transcriptional regulation of high pathogenicity island iron uptake genes by YbtA. Int. J. Med. Microbiol. IJMM. 2005;295:19–28. doi: 10.1016/j.ijmm.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 51.Ashburner M, et al. Gene Ontology: tool for the unification of biology. Nat. Genet. 2000;25:25–29. doi: 10.1038/75556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.The gene ontology consortium Gene Ontology Consortium: going forward. Nucleic Acids Res. 43, D1049–D1056 (2015). [DOI] [PMC free article] [PubMed]
- 53.Craig SA, Carpenter CD, Mey AR, Wyckoff EE, Payne SM. Positive regulation of the Vibrio cholerae porin OmpT by iron and fur. J. Bacteriol. 2011;193:6505–6511. doi: 10.1128/JB.05681-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.da Silva Neto JF, Lourenço RF, Marques MV. Global transcriptional response of Caulobacter crescentus to iron availability. BMC Genomics. 2013;14:549. doi: 10.1186/1471-2164-14-549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Balasubramanian R, Levinson BT, Rosenzweig AC. Secretion of flavins by three species of methanotrophic bacteria. Appl. Environ. Microbiol. 2010;76:7356–7358. doi: 10.1128/AEM.00935-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vasileva D, Janssen H, Hönicke D, Ehrenreich A, Bahl H. Effect of iron limitation and fur gene inactivation on the transcriptional profile of the strict anaerobe Clostridium acetobutylicum. Microbiol. Read. Engl. 2012;158:1918–1929. doi: 10.1099/mic.0.056978-0. [DOI] [PubMed] [Google Scholar]
- 57.Thamer W, et al. A two [4Fe-4S]-cluster-containing ferredoxin as an alternative electron donor for 2-hydroxyglutaryl-CoA dehydratase from Acidaminococcus fermentans. Arch. Microbiol. 2003;179:197–204. doi: 10.1007/s00203-003-0517-8. [DOI] [PubMed] [Google Scholar]
- 58.Mackey KRM, et al. Divergent responses of Atlantic coastal and oceanic Synechococcus to iron limitation. Proc. Natl. Acad. Sci. USA. 2015;112:9944–9949. doi: 10.1073/pnas.1509448112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang Y, et al. Functional pyruvate formate lyase pathway expressed with two different electron donors in Saccharomyces cerevisiae at aerobic growth. FEMS Yeast Res. 2015;15:fov024. doi: 10.1093/femsyr/fov024. [DOI] [PubMed] [Google Scholar]
- 60.Chowdhury NP, Klomann K, Seubert A, Buckel W. Reduction of Flavodoxin by Electron Bifurcation and Sodium Ion-dependent Reoxidation by NAD + Catalyzed by Ferredoxin-NAD + Reductase (Rnf) J. Biol. Chem. 2016;291:11993–12002. doi: 10.1074/jbc.M116.726299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pajuelo D, et al. Iron and Fur in the life cycle of the zoonotic pathogen Vibrio vulnificus. Environ. Microbiol. 2016;18:4005–4022. doi: 10.1111/1462-2920.13424. [DOI] [PubMed] [Google Scholar]
- 62.Takemoto N, Tanaka Y, Inui M, Yukawa H. The physiological role of riboflavin transporter and involvement of FMN-riboswitch in its gene expression in Corynebacterium glutamicum. Appl. Microbiol. Biotechnol. 2014;98:4159–4168. doi: 10.1007/s00253-014-5570-5. [DOI] [PubMed] [Google Scholar]
- 63.Wang H, et al. Dual-Targeting Small-Molecule Inhibitors of the Staphylococcus aureus FMN Riboswitch Disrupt Riboflavin Homeostasis in an Infectious Setting. Cell Chem. Biol. 2017;24:576–588.e6. doi: 10.1016/j.chembiol.2017.03.014. [DOI] [PubMed] [Google Scholar]
- 64.Chen J, Shen J, Solem C, Jensen PR. Oxidative stress at high temperatures in Lactococcus lactis due to an insufficient supply of Riboflavin. Appl. Environ. Microbiol. 2013;79:6140–6147. doi: 10.1128/AEM.01953-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen J, Shen J, Ingvar Hellgren L, Ruhdal Jensen P, Solem C. Adaptation of Lactococcus lactis to high growth temperature leads to a dramatic increase in acidification rate. Sci. Rep. 2015;5:14199. doi: 10.1038/srep14199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Showman AC, Aranjuez G, Adams PP, Jewett MW. Gene bb0318 Is Critical for the Oxidative Stress Response and Infectivity of Borrelia burgdorferi. Infect. Immun. 2016;84:3141–3151. doi: 10.1128/IAI.00430-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.