Abstract
The goals of this study were to report the seasonal shedding patterns of strongyle and Parascaris spp. eggs in repeated fecal samples for mares (n = 38) and foals (n = 39), and to evaluate the efficacy of ivermectin treatment in mares from 2 selected horse breeding farms in central Saskatchewan. Median strongyle fecal egg counts (FEC) peaked in July and August in adult horses. The farms differed significantly (P = 0.0005) in regard to strongyle shedding categories (< 200; 200 to 500; and > 500 eggs/g) over time, but for each individual horse (both farms combined) these categories did not differ over time (P = 0.13) on samples collected in grazing season. When evaluating 3 samples collected fall, summer and fall in 2 consecutive grazing seasons, 94% of horses that shed < 200 eggs/g on 2 initial samples, remained in the same category on the third sample. Mares on each farm didn’t differ statistically in shedding categories when comparing September samples from 2 consecutive years (Farm A: P = 0.56, Farm B: P = 0.06). Peak strongyle shedding occurred late fall in the first year of life for foals on Farm A, and in July in the second year of life for foals on Farm B. Parascaris spp. FEC were greatest in foals ≤ 6 months of age, with peak observed when foals were 5 to 6 months old. Ivermectin was 100% effective at reducing strongyle FEC 2 weeks after treatment in adult horses. Horses in Saskatchewan had relatively high strongyle shedding levels, which were significantly different between the farms, and high prevalence of Oxyuris equi. Strongyle shedding consistency was observed for FECs collected from mares in grazing season (July to September).
Résumé
Caractéristiques de l’excrétion des oeufs de parasites pendant une période de 1 an chez les poulains et leurs mères dans deux fermes du centre de la Saskatchewan. Cette étude avait pour but de dresser un rapport sur les tendances d’excrétion saisonnière des oeufs des strongyles et de Parascaris spp. dans des prélèvements fécaux répétés pour les juments (n = 38) et les poulains (n = 39) et d’évaluer l’efficacité du traitement à l’ivermectine chez deux juments provenant de deux fermes d’élevage de chevaux dans le centre de la Saskatchewan. Les comptes médians d’oeufs fécaux des strongyles ont culminé en juillet et en août chez les chevaux adultes. Les fermes présentaient des différences significatives (P = 0,0005) à l’égard des catégories d’excrétion des strongyles (< 200; de 200 à 500; et > 500 oeufs/g) dans le temps, mais, pour chaque cheval individuel (les deux fermes combinées), ces catégories ne présentaient pas de différences à la longue (P = 0,13) pour les échantillons prélevés durant la saison de pâturage. Lors de l’évaluation des reois échantillons prélevés à l’automne, à l’été et à l’automne pendant deux saisons de pâturage consécutives, 94 % des chevaux qui avaient excrété < 200 oeufs/g pour deux prélèvements initiaux, sont demeurés dans la même catégorie pour le troisième échantillon. Les juments de chaque ferme ne présentaient pas de différences statistiques pour les catégories d’excrétion lorsque l’on comparait les échantillons de septembre provenant de deux années consécutives (Ferme A : P = 0,56, Ferme B : P = 0,06). L’excrétion des strongyles a culminé à la fin de l’automne pendant la première année de vie pour les poulains de la Ferme A et en juillet de la deuxième année de vie pour les poulains de la Ferme B. Les comptes d’oeufs fécaux de Parascaris spp. étaient les plus importants chez les poulains âgés de ≤ 6 mois et le point culminant était observé lorsque les poulains étaient âgés de 5 ou 6 mois. L’ivermectine était efficace à 100 % pour réduire les comptes d’oeufs fécaux 2 semaines après le traitement chez les chevaux adultes. Les chevaux de la Saskatchewan ont présenté des taux d’excrétion relativement élevés de strongyles, qui étaient significativement différents entre les fermes, et une forte prévalence d’Oxyuris equi. La constance de l’excrétion des strongyles a été observée pour les comptes d’oeufs fécaux obtenus auprès des juments pendant la saison de pâturage (de juillet à septembre).
(Traduit par Isabelle Vallières)
Introduction
Intestinal parasitism is a common health concern in horses worldwide. Intensive anthelmintic treatment schedules that have utilized frequent dosing and rotation among drug classes have accelerated the development of anthelmintic resistance (1–12). For over 20 y, some European and, more recently, North American countries, have been implementing selective treatment protocols for strongyles based on testing for fecal egg count (FEC) (1,11,13,14). The threshold for anthelmintic treatment in such protocols is usually set at 150 to 200 eggs/g, and recommended frequency of individual horse repeated FECs vary, depending on country or veterinary practice (14–17). Furthermore, prophylactic treatments are administered to low shedders, with the frequency (once to twice a year) and timing dependent on climate and management of the herd and pasture (1,13,14). It is recommended to assess treatment efficacy with an FEC reduction test (FECRT), every 3 y (adult horses) or yearly (foals and weanlings) on at least 6 horses/farm, and to avoid treatments at times when parasite burdens on pasture are low (1). FEC-based parasite control programs aim to reduce total number of treatments, preserve parasitic refugia, allow for identification and targeted treatment of horses at high risk, and to slow the development of anthelmintic resistance (1,16–20).
In order to categorize horses’ shedding level (low < 200, 200 to 500, or > 500 eggs/g), FEC should be performed during a time when previous anthelmintic treatments are no longer effective and climatic conditions are optimal for parasite transmission (1). For example, an FEC should be performed 16 wk after mox-idectin treatment (4 wk beyond egg reappearance period for this drug) during grazing season. The general recommendation is to perform more than one FEC at more than one time point (1).
The cost associated with repeated fecal sampling could be reduced by reducing the frequency of testing. From a limited number of studies (21–24) it is believed that adult horses (> 3 y old) remain consistent in their shedding levels unless their immune system is compromised, or they are moved to a new environment (with different environmental parasitic contamination). In a study analyzing sequential FECs as a part of a selective anthelmintic program in Danish horses, Nielsen et al (14) evaluated FECs collected twice yearly (spring and fall) over a 3-year period, and estimated an 84% probability of a FEC result < 200 eggs/g for horses with the 2 previous FEC results < 200 eggs/g. When FEC exceeded 200 eggs/g on the previous 2 samples, the probability for a horse exceeding 200 eggs/g on the third sample was 59% (22). In another study from the Netherlands, 55.2% of horses had consistently low FECs (< 100 eggs/g) in 2 consecutive samples taken 6 wk apart (21), and for 303 horses in Germany the egg shedding remained fairly consisted on 9 samples collected over 1 y (24).
Environmental conditions such as temperature and humidity play an important role in egg and larval survival, as well as larval development time on a pasture. Extremely cold winters in western Canada greatly prolong or completely halt the development of strongyle eggs to infective L3 larval stage and decrease viability of eggs (25,26). A study by Polley (25) on the ecology of free-living stages of strongyles on the Canadian prairie showed a total inhibition of strongyle free living larval development for October, December, and January (25). Taken together, the climatic characteristics of different regions should be considered when optimizing a targeted parasite control program.
To our knowledge, strongyle and Parascaris spp. infections have not been investigated in seasonally pastured horses in Saskatchewan. Therefore, the goal of our study was to investigate parasite egg shedding over a 12-month period in 2 mare and foal herds under different management conditions, and estimate the strongyle egg shedding consistency in adult horses. Additionally, we report the efficacy of ivermectin on strongyle egg reduction for adult horses.
Materials and methods
Animals and farm management
All procedures and experimental protocols using animals were approved by the University of Saskatchewan’s Committee on Animal Care and Supply, and the University of Saskatchewan Animal Research Ethics Board. Horse owners consented to samples being obtained from their animals and to abide by the requirements of the study, including ceasing anthelmintic treatment of all horses during the study except for 1 treatment administered by investigators.
Fecal samples were collected from all mares and their foals on 2 equine breeding farms (Farms A and B) located within a 120 km radius from Saskatoon, Saskatchewan. Twenty-three mare-foal pairs were included from Farm A, in which mares represented numerous breeds (Standardbreds, Thoroughbreds, and Arabians) and foals were sired by a Friesian stallion. On Farm B, there were 15 Quarter Horse (QH) mares with 16 QH foals, as 1 mare had an additional foster foal. All foals included were born between May and July 2010. Horses on Farm A were housed in a 30 m × 60 m corral pen and fed round bale hay ad libitum throughout fall to summer and pastured on 48 hectares during summer months. The history of parasite control on this farm included no anthelmintic treatments for 5 y before starting the study, with the exception of male weanlings, which had been treated with ivermectin paste at the time of castration every year by a local veterinarian. Horses on Farm B were housed in a 4-hectare pen and fed round bale hay ad libitum between November and late July, then pastured on 13 hectares in August through October. The history of parasite control on this farm included twice yearly anthelmintic treatments: mares and foals were administered ivermectin (injectable bovine formulation administered orally) and oral pyrantel pamoate, respectively, before pasture turnout (usually in July), followed by administration of ivermectin (same formulation as described) to all horses upon moving to winter pens in November. On both farms, pastures and pens had not been maintained in the past 5 y in terms of manure removal or rotation of pastures.
Sample collection and analysis
The study was performed between July 2010 and September 2011. Fecal samples were collected in July, August, September, November, and December of 2010, and March, May, July, and September of 2011 (mares only). Foals born in 2010 were sampled in July, August, September, November, and December 2010, and in March, June, and July 2011.
At each sampling, approximately 10 g of feces were obtained directly from the rectum of mares and foals. Samples were transported to the laboratory in a cooler with an ice pack. Samples were stored at 4°C and examined within 48 h of collection. A modified Wisconsin double centrifugal sugar flotation technique was used for all fecal egg counts (FEC) as previously described (27), with a minimum detection limit of 1 egg/g. Briefly, the sample was diluted with water, filtered through cheese cloth, and centrifuged at 300 × g for 10 min. Supernatant was then discarded and the sample was mixed with Sheather’s solution (Jorgensen Laboratories, Loveland, Colorado, USA; specific gravity 1.27) using a vortex mixer. The sample was then again centrifuged at 300 × g for 10 min. Slides were examined at 10× magnification under a standard light microscope. Eggs were counted and classified according to their morphology. All samples were processed and interpreted by the first author.
Determination of strongyle susceptibility to ivermectin in mares
In November 2010, following a collection of fecal samples for FEC, 0.2 μg/kg of ivermectin (Eqvalan; Mérial Limited, Baie d’Urfé, Quebec) was administered orally by the first author to all horses. The weight of the horses was estimated by the first and second authors and the breeder, and mean weight was used to calculate dose of ivermectin per horse. Ten to 14 d after anthelmintic treatment, the FEC was repeated and a fecal egg count reduction test (FECRT) was calculated as 100× (1-egg count after treatment/egg count pre-treatment) (1).
Statistical analysis
All data, including horse number, date, FEC, and FECRT were entered into an Excel spreadsheet (Microsoft; Redmond, Washington, USA). Analyses were performed using R version 3.3.2, “nlme” package Version 3.1-129 for linear mixed effects model analysis, “ordinal” package Version 2015.6-28 was used for ordinal mixed effects model analysis, and “lsmeans” package Version 2.25-5 was used for pairwise comparison analysis. Statistical significance was set at P < 0.05 for all tests.
Fecal egg count data are highly skewed (visual justification was made through histograms), therefore median and range (minimum, maximum) are reported in summary statistics. Log-transformation on the FEC egg count was used for further data analysis. Specifically, 1 egg is added to all egg counts before log-transformation to avoid undefined values from logarithmic transformation. Additionally, FECs collected 10 to 14 d after deworming (December 2010) were removed from further analysis. Three FEC categories (< 200, 200 to 500, > 500 eggs/g) were used to categorize the egg shedding level.
Linear mixed effects models were used to examine the effects of farm and time (as fixed effects) on the log-transformed egg count data (as outcomes), after accounting for the dependence structure due to repeated measures in horses (as random effect). The assumptions of normality and equal variance were assessed using the specific models.
An ordinal logistic mixed effects model was used to examine the effects of farm and time (as fixed effects) on the 3 FEC shedding categories (as outcomes), after accounting for the repeated measures in horses (as random effect). In addition, post-hoc analysis using Tukey method for P-value adjustment was done for pairwise comparisons between groups.
Shedding consistency was approached with the following 3 methods. Friedman test was used to test for consistency in shedding categories in mares over time, and Wilcox signed rank test with continuity correction was used when only 2 time points were compared (September 2010 and September 2011). Lastly, we looked for consistency on 3 samples taken in consecutive grazing seasons (September 2010, July 2011, and September 2011), as described by Nielsen et al (22). Namely, the cutoff of 200 eggs/g was used, and results were presented as percentage of horses remaining in the same category (< 200 eggs/g or ≥ 200 eggs/g) on third sample, as on 2 previous ones.
Results
Mares
Ninety-seven percent (201/207) of fecal samples were recovered from mares on Farm A and 96% (130/135) from Farm B. On occasion, rectal feces were unavailable at the time of collection.
Strongyle eggs were recovered at least once from 89.5% of all mares’ (Farm A and Farm B) fecal samples. Parascaris spp. and Oxyuris equi eggs were found in 3% and 2% of mares’ fecal samples, respectively. No cestode eggs were detected. Median strongyle FEC was 212, range 0 to 2490 eggs/g. Peak median strongyle FEC in mares was recorded in August 2010 for Farm A and July 2011 for Farm B.
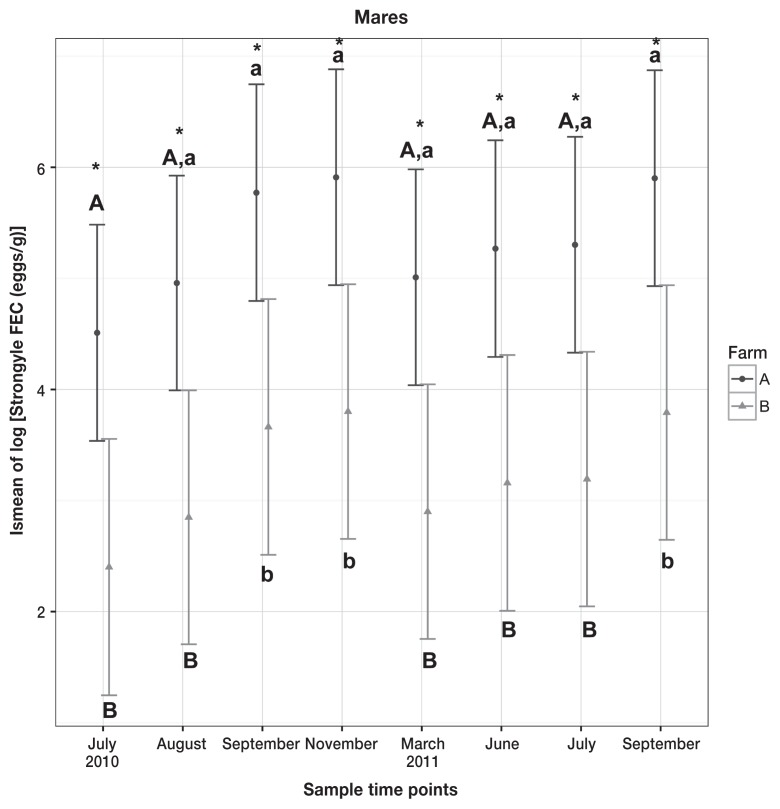
Results from linear mixed effects models showed that both farm (P = 0.0027) and time (P = 0.0009) had statistically significant effects on the log of strongyle FECs in mares, and the farms were significantly different at every collection time. The results from the post-hoc pairwise comparison are shown in Figure 1.
Figure 1.
Means and 95% confidence intervals of lsmeans from linear mixed effects model on log of strongyle FEC in mares. Differences between the farms are marked with asterisks and differences within the farm are marked with letters, with significance set as P < 0.05. Upper- and lowercase letter A represents Farm A, and upper- and lowercase letter B represents Farm B.
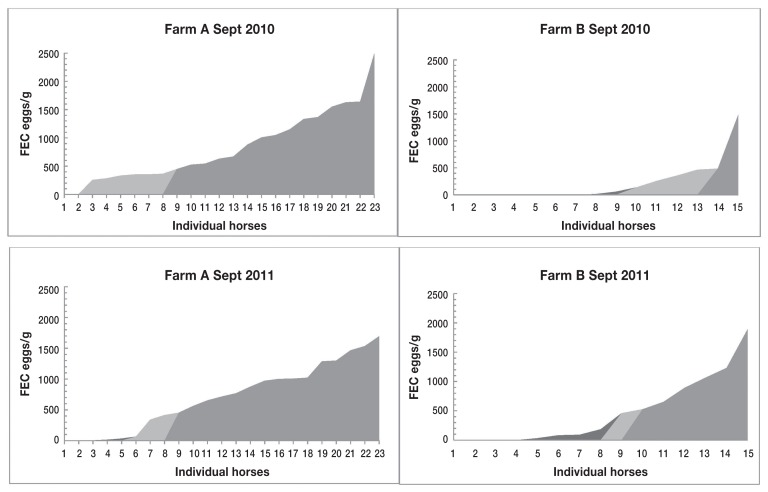
The months of September 2010 and 2011 were used to compare egg shedding distribution and magnitude for Farm A and Farm B (Figure 2). Most horses on Farm A (60% in both years) had high FEC, and most on Farm B (66% and 53%, respectively) had low FEC (Table 1).
Figure 2.
Shedding distribution of strongyle eggs in adult mares from each farm in September 2010 and September 2011. Black area represents low shedders (< 200 eggs/g); light gray area represents moderate shedders (200 to 500 eggs/g), and dark gray area represents high shedders (> 500 eggs/g).
Table 1.
Categorization of strongyle egg shedding in adult mares’ fecal samples collected in September 2010 (Farm A, n = 22; Farm B, n = 14) and September 2011 (Farm A, n = 22; Farm B, n = 15), as percentage of horses in each category and their percentage contribution to overall egg shedding at that time.
| September 2010 | September 2011 | |||||||
|---|---|---|---|---|---|---|---|---|
|
|
|
|||||||
| Farm A | Farm B | Farm A | Farm B | |||||
|
|
|
|
|
|||||
| Fecal egg count (eggs/g) | Horses (%) | Eggs (%) | Horses (%) | Eggs (%) | Horses (%) | Eggs (%) | Horses (%) | Eggs (%) |
| < 200 | 8.7 | 0.01 | 66.66 | 6.62 | 26.09 | 0.74 | 53.33 | 5.53 |
| 200 to 500 | 30.43 | 12.71 | 26.67 | 48.04 | 13.04 | 7.48 | 6.67 | 6.46 |
| > 500 | 60.87 | 87.28 | 6.67 | 45.34 | 60.87 | 91.78 | 40.00 | 88.01 |
Consistency of shedding of strongyle eggs in mares
Results from the ordinal mixed effects model on shedding categories (low, moderate, and high) showed that both farm (P = 0.0006) and time (P = 0.0001) had statistically significant effects on the strongyle FEC egg shedding categories. For an animal in Farm A, the estimated odds of being in higher shedding category (> 500 eggs/g) were 16.7 [95% confidence interval (CI): 3.33 to 100, P = 0.0006] times higher, compared to an animal in Farm B.
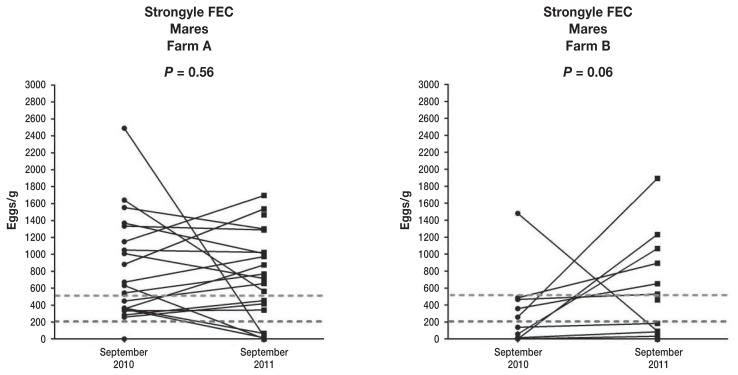
When comparing the actual strongyle FEC egg counts in the September months between 2010 and 2011 using Wilcox signed rank test with continuity correction, no statistical significant difference was found in Farm A or Farm B (P = 0.56 and P = 0.06, respectively) (Figure 3).
Figure 3.
Comparison of September 2010 and September 2011 strongyle FECs for individual mares on Farms A and B.
Shedding categories for each of the mares did not statistically differ over time for samples collected in grazing season (all samples, excluding December 2010, March and June 2011) in both farms; P-values from the Friedman test were 0.13, 0.22, and 0.09 for both farms, Farm A and Farm B, respectively. When only post deworming (December 2010) sample was excluded, we found no statistical difference (P = 0.12) in shedding categories over time in mares in Farm B, but there was a significant difference (P = 0.0036) in mares in Farm A.
Lastly, consistency of strongyle FECs was assessed for 3 samples collected in consecutive grazing seasons in September 2010, July 2011, and September 2011. Of horses excreting low FEC (< 200 eggs/g) on 2 former samples (17/38), 94% (16/17) remained in the same category for the third fecal examination. Among horses with FEC results ≥ 200 eggs/g on the first 2 samples (10/38), 8/10 remained in the same category on the third fecal examination. Altogether 27/38 (71%) horses were consistent on all 3 samples either under low or moderate/high shedding categories.
Foals
Ninety-four percent (173/183) of fecal samples were recovered from foals on Farm A and 96% (124/128) from Farm B. On occasion, rectal feces were unavailable at the time of collection. Strongyle eggs were recovered from 71.9%, Parascaris spp. eggs from 48.7%, and Oxyuris equi eggs from 7.8% of all foals’ samples.
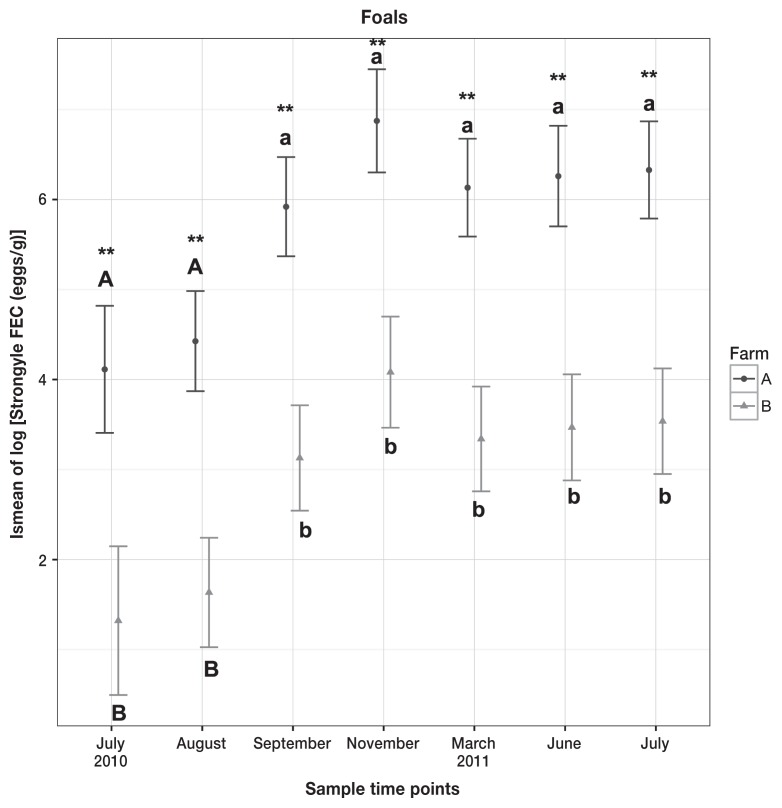
Median strongyle FEC for foals was 108 eggs/g, range 0 to 1987 eggs/g. Peak median strongyle FEC occurred in July 2011 on Farm A, and November 2010 on Farm B. Results from linear mixed effects models showed that both farm (P < 0.0001) and time (P < 0.001) had statistically significant effects on the log of strongyle FEC egg counts in foals, and the results from the post-hoc pairwise comparison are shown in Figure 4.
Figure 4.
Means and 95% confidence intervals of lsmeans from linear mixed effects model on log of strongyle FEC in foals. Differences between the farms are marked with asterisks, differences within the farm are marked with letters, with significance set as P < 0.05. Upper-and lowercase letter A represents Farm A, and upper- and lowercase letter B represents Farm B.
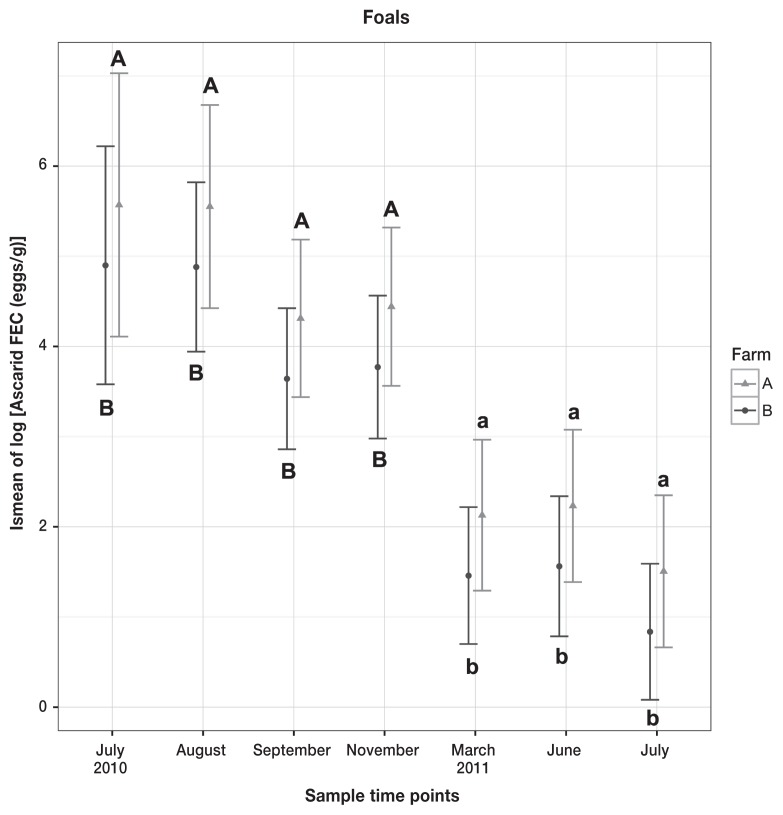
Median Parascaris spp. FEC for foals was 0 eggs/g, range 0 to 1554. Peak shedding occurred in 2010, in the months of August on Farm A, and November on Farm B. Only time had a statistically significant effect (P < 0.001) on the log of ascarid FEC egg counts in foals (Figure 5).
Figure 5.
Means and 95% confidence intervals of lsmeans from linear mixed effects model on log of ascarid FEC in foals. Differences within the farm (P < 0.05) are marked with letters, with significance set as P < 0.05. Upper- and lowercase letter A represents Farm A, and upper- and lowercase letter B represents Farm B. No significant difference was observed between the farms at any time point.
Fecal egg count reduction test
Ivermectin treatment was 100% effective in reducing strongyle shedding in all adult horses.
Discussion
The present study provides information on repeated FECs for adult horses and foals in 2 farms in Saskatchewan with the focus on magnitude and consistency of strongyle egg shedding over time.
The management practices of both breeding farms in our study included a period of time from fall to early summer when horses were crowded in smaller pens, from which manure and organic matter had not been removed for years. Additionally, lack of FEC-based parasite control programs and specific treatments against encysted cyathostomes could have contributed to our herds having both high percentage of strongyle positive samples in adult horses (89.5%) and relatively high median strongyle FECs (212, range: 0 to 2490 eggs/g), in particular in Farm A (416, range: 0 to 2490 eggs/g). The percentage of positive strongyle FECs was higher than reported for some European countries (48.1% in Germany, 61.1% Italy and 60.9% in the UK), but similar to that previously reported in Kentucky (99%) and Nicaragua (94%) (15,16,28).
Hesitance of some horse owners to perform fecal egg examinations likely originates from perceived cost. The frequency of FECs could be diminished based on assumption of shedding consistency among adult horses (> 3 y old) (1). Growing, yet limited knowledge is available on repeatability of FEC results (1,21–24). We looked into patterns of shedding among adult horses, including peak times and consistency, accounting for climate characteristics of our region. The Canadian prairies are unique in regards to their long-lasting, cold winters. With winter air temperatures often below –20°C for extended periods, such an environment is seemingly less favorable for parasite survival. However, snow cover can keep the temperature to which the parasites are actually exposed more constant and closer to the freezing point, which can greatly prolong egg and larva survival. In a study performed 25 y previously, Polley (25) observed complete inhibition of development of strongyle larvae during the months of October, December, and January in the Saskatoon area. However, a small proportion of eggs placed in experimental plots throughout winter were viable the following spring and these contributed to the peak in number of infective larvae recorded in August and September. Peak strongyle FEC in our study occurred just before (July) and overlapped (August) with the times when environmental larval loads are expected to be high.
Studies evaluating repeated strongyle FEC determined that adult horses (> 3 y of age) tend to fall under the “70/30” rule, whereby the majority of the population sheds a low to moderate amounts of eggs, and a minority of the population (15% to 30%) sheds as much as 83% of all eggs in the grazing season (1,20). Kaplan and Nielsen (20) estimated that “treating all adult horses exceeding a strongyle FEC of 200 EPG, only leads to treating about 50% of the horse population, but still provides about 95% reduction of the overall egg shedding.” Based on our data, Farm B seemed to fit these criteria in the month of September 2010, and to a lesser extent in 2011, in which 93.37% and 60% of horses shed < 500 eggs/g, respectively (Table 1 and Figure 2). In contrast, as many as 60% of horses on Farm A were in the high shedding category for both years. Both farms were also significantly different in regard to shedding categories of individual horses over all FEC collection times. Animals from Farm A had 16.7 times higher odds to be in a shedding category over 500 eggs/g than did animals from Farm B. This particular difference could potentially originate from the historical differences in anthelmintic use in these farms. Other risk factors for gastrointestinal parasites such as use of pasture, high population density, failure to remove manure, housing younger and aged horses together, and lack of advice on parasite control (21–23,29–32) were similar for both farms. It is also possible that there were higher levels of contamination of pens and pastures on Farm A, and/or that environmental parasite stages had higher survival rates due to variability in microhabitat that was beyond the scope of our study.
At the level of the individual horse, we examined the consistency of shedding over time on repeated samples using 3 different approaches. Analysis of shedding categories (< 200, 200 to 500, and > 500 eggs/g) over time for both farms combined showed no statistical difference for all samples excluding the post deworming one (December 10), March and June 2011. The exclusion was based on the assumption that adult horses FECs are most reliable in the grazing season (1) when exposure is high enough to distinguish the natural ability of horses’ immune systems to decrease the shedding level versus low shedding due to low environmental exposure (cold months in the Northern hemisphere). Interestingly, Farm B was no different when March and June samples were included in analysis, and this is likely due to fact that animals in Farm B had overall higher odds to be in lower shedding categories; early spring and summer samples would be expected to be overall lower than in the grazing season. We also compared 2 years’ September samples for individual horses separately from each farm. Although statistically there was no difference in shedding levels from one season to another, Farm B was borderline significant at P < 0.06. This means that some animals switched categories from 1 year to another based on FECs performed in the peak of environmental exposure to parasites, and months after ERP of anthelmintic treatment. Although studies including larger number of animals are warranted, repeating FECs from one grazing season to another should be considered.
Nielsen et al (22) examined strongyle shedding consistency in 3 consecutive samples from Danish horses that were under strategic parasite control. Fecal exams were performed twice yearly (spring and fall) and anthelmintic treatments administered to all horses with FEC > 200 eggs/g. These authors found that horses with FEC < 200 eggs/g on 2 consecutive samples had 84% probability for the same result on the third sampling. Moderate and high shedders (> 200 eggs/g) had a 59% chance to be within the same category on a third sample. To perform a similar analysis, we used fall, summer and fall samples available, and the likelihood of samples remaining in the same category was even more pronounced, with 94% of low shedders and 80% of moderate and high shedders combined remaining in their respective categories on the third FEC. Repeated fecal samples have inherently high variability for reasons such as uneven egg distribution in feces, seasonal changes in egg production by internal parasites, use of different flotation techniques, changes in environmental contamination (density of horses or other management interventions), or a horse’s health, immune status, and age (13,16,31). Our data set was also limited by small number of animals (39 adult horses on 2 farms) while Nielsen (22) used 424 horses on 10 farms, so it is difficult to extrapolate the results to the general horse population of Saskatchewan, and more studies are warranted.
Ivermectin dosed at 0.2 mg/kg body weight reduced strongyle shedding by 100% in adult horses in our study. Considering the historical sparse use of dewormers on Farm A for the past 5 y, this is an expected outcome. Farm B had a history of using the injectable bovine formulation of ivermectin orally. The bioavailability of this product is unknown. Potential under-dosing related to such practice could have selected resistant parasites over time. This does not seem to be the case based on FECRT, yet we did not examine the egg recurrence times, which would provide a more accurate picture of early signs of anthelmintic resistance (1). We did not measure but estimated the weight of the horses due to safety concerns such as working with previously unhandled horses in squeeze chutes. The weights were estimated by 2 veterinarians working with actual horses’ weights on a daily basis in an equine hospital, and by an experienced breeder on each farm. Although not ideal, the alternative method of using weight tape (most commonly used in the field) could have resulted in a similar margin of error as a visual estimate. Reavell (33) looked into visual versus weight tape weight estimates in adult horses, and found similar 8% to 12% error for both methods. Another study found that weight tape estimate was significantly different from horses’ actual weight, with a mean error of 65.81 kg (34).
Strongyle shedding levels in foals differed significantly between the farms. For all collection times that difference was pronounced at P < 0.001, with foals from Farm A shedding significantly more eggs than foals from Farm B. This correlates well with the high shedding levels observed in adult horses on Farm A, consistent with a potential high degree of pasture contamination. One cannot exclude other factors, such as differences in immune status or genetics of horses on the 2 farms, but this was beyond the scope of the present study.
Parascaris spp. is a ubiquitous ascarid parasite of foals and weanlings. Occasionally, Parascaris spp. eggs are recovered from adult horses’ feces, generally in immune-compromised animals kept in close association with foals (1,35). First appearance of Parascaris spp. eggs in foal manure begins at approximately 3 mo of age (the prepatent period following ingestion of larvated eggs is approximately 10 wk) and subsides at about 18 mo of age (1). In our study, the peak of Parascaris spp. shedding occurred in November for both farms, when foals were approximately 5 (Farm B) to 6 (Farm A) mo of age. Opposite to strongyle shedding in both mares and foals, the farm had no effect on ascarid shedding levels, and the only significant difference was time. This finding is striking considering the historical differences in parasite control in foals between the 2 farms — none on Farm A, and 2 treatments in the first year of life for Farm B. However, as there is no direct correlation between FECs for Parascaris spp. and real worm counts in equids (15); FECs may not represent real ascarid infection levels for these foals. Therefore the FECs reported here provide qualitative (decrease in shedding in second year of life) rather than quantitative information.
Oxyuris equi eggs were found more often than expected in 7.8% of samples from foals and 2% of samples from adult horses. Historically, Oxyuris equi was more commonly found in weanlings and yearlings; however, in recent years it seems to be increasing in adult horse populations (1,36). All samples in this study were collected directly from the rectum and rectal sleeves had contact with the perianal area. This likely contributed to a number of positive results, together with poor parasite control practices especially on Farm A, and macrocyclic lactone use in adults on Farm B, which may not be as effective in clearing the parasite from the host (36).
We did not find cestode eggs in any of the fecal samples. Cestode eggs pose a diagnostic challenge as they are contained within proglottids and are shed intermittently upon disintegration of the proglottid. The recommended diagnostic protocol is to examine FECs 24 h after cestocidal treatment (37), or potential inclusion of a salivary enzyme-linked immunosorbent assay (ELISA) test, which was not done in this study.
In conclusion, the parasite egg shedding in this study was generally high in prevalence and magnitude, and there were significant differences between the 2 farms with different management practices. There was a consistency of strongyle egg shedding in adult horses over time for samples collected in the grazing season. Further studies incorporating larger groups of horses and facilities are warranted to offer more guidance for parasite control strategies under Saskatchewan-specific environmental conditions and equine management practices.
Acknowledgments
Funding was provided by Equine Health Research Fund from the Western College of Veterinary Medicine’s Equine. We acknowledge Drs. Katharina Lohman and Steve Manning, Western College of Veterinary Medicine, University of Saskatchewan for their contribution to the idea for this study, and recruitment of animals. We also thank the owners of Farms A and B for their cooperation in this study. Ivermectin paste was supplied at no charge by Mérial. CVJ
Footnotes
Use of this article is limited to a single copy for personal study. Anyone interested in obtaining reprints should contact the CVMA office (hbroughton@cvma-acmv.org) for additional copies or permission to use this material elsewhere.
References
- 1.American Association of Equine Practitioners [ www.aaep.org] c2016. Available from: http://www.aaep.org/custdocs/AAEPParasiteControlGuidelines.pdf.
- 2.Kaplan RM. Anthelmintic resistance in nematodes of horses. Vet Res. 2002;33:491–507. doi: 10.1051/vetres:2002035. [DOI] [PubMed] [Google Scholar]
- 3.Molento MB, Nielsen MK, Kaplan RM. Resistance to avermectin/milbemycin anthelmintics in equine cyathostomins — Current situation. Vet Parasitol. 2012;185:16–24. doi: 10.1016/j.vetpar.2011.10.013. [DOI] [PubMed] [Google Scholar]
- 4.Lyons ET, Tolliver S, Ionita M, Lewellen A, Collins S. Field studies indicating reduced activity of ivermectin on small strongyles in horses on a farm in Central Kentucky. Parasitol Res. 2008;103:209–215. doi: 10.1007/s00436-008-0959-7. [DOI] [PubMed] [Google Scholar]
- 5.Kaplan RM, Klei TR, Lyons ET, et al. Prevalence of anthelmintic resistant cyathostomes on horse farms. J Am Vet Med Assoc. 2004;225:903–910. doi: 10.2460/javma.2004.225.903. [DOI] [PubMed] [Google Scholar]
- 6.Slocombe JO, de Gannes RV. Cyathostomes in horses in Canada resistant to pyrantel salts and effectively removed by moxidectin. Vet Parasitol. 2006;140:181–184. doi: 10.1016/j.vetpar.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 7.Slocombe JO, Coté JF, de Gannes RV. The persistence of benzimidazole-resistant cyathostomes on horse farms in Ontario over 10 years and the effectiveness of ivermectin and moxidectin against these resistant strains. Can Vet J. 2008;49:56–60. [PMC free article] [PubMed] [Google Scholar]
- 8.Molento MB, Antunes J, Bentes RN, Coles GC. Anthelmintic resistant nematodes in Brazilian horses. Vet Rec. 2008;162:384–385. doi: 10.1136/vr.162.12.384. [DOI] [PubMed] [Google Scholar]
- 9.Lyons ET, Tolliver S, Ionita M, Collins S. Evaluation of parasiticidal activity of fenbendazole, ivermectin, oxibendazole, and pyrantel pamoate in horse foals with emphasis on Ascarids (Parascaris equorum) in field studies on five farms in Central Kentucky in 2007. Parasitol Res. 2008;103:287–291. doi: 10.1007/s00436-008-0966-8. [DOI] [PubMed] [Google Scholar]
- 10.Larsen ML, Ritz C, Petersen SL, Nielsen MK. Determination of ivermectin efficacy against cyathostomins and Parascaris equorum on horse farms using selective therapy. Vet J. 2011;188:44–47. doi: 10.1016/j.tvjl.2010.03.009. [DOI] [PubMed] [Google Scholar]
- 11.von Samson-Himmelstjerna G, Fritzen B, Demeler J, et al. Cases of reduced cyathostomin egg-reappearance period and failure of Parascaris equorum count reduction following ivermectin treatment as well as survey on pyrantel efficacy on German horse farms. Vet Parasitol. 2007;144:74–80. doi: 10.1016/j.vetpar.2006.09.036. [DOI] [PubMed] [Google Scholar]
- 12.Reinemeyer CR. Diagnosis and control of anthelmintic-resistant Parascaris equorum. Parasit Vectors. 2009;2( Suppl 2):S8. doi: 10.1186/1756-3305-2-S2-S8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nielsen MK, Fritzen B, Duncan JL, et al. Practical aspects of equine parasite control: A review based upon a workshop discussion consensus. Equine Vet J. 2010;42:460–468. doi: 10.1111/j.2042-3306.2010.00065.x. [DOI] [PubMed] [Google Scholar]
- 14.Nielsen MK, Reist B, Kaplan RM, Pfister K, van Doorn DCK, Becher A. Equine parasite control under prescription-only conditions in Denmark — Awareness, knowledge, perception, and strategies applied. Vet Parasitol. 2014;204:64–72. doi: 10.1016/j.vetpar.2013.10.016. [DOI] [PubMed] [Google Scholar]
- 15.von Samson-Himmelstjerna G, Traversa D, Demeler J, et al. Effects of worm control practices examined by a combined faecal egg count and questionnaire survey on horse farms in Germany, Italy and the UK. Parasit Vectors. 2009;2:S3. doi: 10.1186/1756-3305-2-S2-S3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nielsen MK, Baptiste KE, Tolliver SC, Collins SS, Lyons ET. Analysis of multiyear studies in horses in Kentucky to ascertain whether counts of eggs and larvae per gram of feces are reliable indicators of numbers of strongyles and ascarids present. Vet Parasitol. 2010;174:77–84. doi: 10.1016/j.vetpar.2010.08.007. [DOI] [PubMed] [Google Scholar]
- 17.Swiderski C, French DD. Paradigms for parasite control in adult horse populations: A review. Proc American Association of Equine Practitioners; San Diego, California. December 6–10, 2008; pp. 316–321. [Google Scholar]
- 18.Dobson RJ, Hosking BC, Jacobson CL, et al. Preserving new anthelmintics: A simple method for estimating faecal egg count reduction test (FECRT) confidence limits when efficacy and/or nematode aggregation is high. Vet Parasitol. 2012;186:79–92. doi: 10.1016/j.vetpar.2011.11.049. [DOI] [PubMed] [Google Scholar]
- 19.Waghorn TS, Leathwick DM, Miller CM, Atkinson DS. Brave or gullible: Testing the concept that leaving susceptible parasites in refugia will slow the development of anthelmintic resistance. N Z Vet J. 2008;56:158–163. doi: 10.1080/00480169.2008.36828. [DOI] [PubMed] [Google Scholar]
- 20.Kaplan RM, Nielsen MK. An evidence-based approach to equine parasite control: It ain’t the 60s anymore. Equine Vet Educ. 2010;22:306–316. [Google Scholar]
- 21.Döpfer D, Kerssens CM, Meijer YG, Boersema JH, Eysker M. Shedding consistency of strongyle-type eggs in Dutch boarding horses. Vet Parasitol. 2004;124:249–258. doi: 10.1016/j.vetpar.2004.06.028. [DOI] [PubMed] [Google Scholar]
- 22.Nielsen MK, Haaning N, Olsen SN. Strongyle egg shedding consistency in horses on farms using selective therapy in Denmark. Vet Parasitol. 2006;135:333–335. doi: 10.1016/j.vetpar.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 23.Becher AM, Mahling M, Nielsen MK, Pfister K. Selective anthelmintic therapy of horses in the Federal states of Bavaria (Germany) and Salzburg (Austria): An investigation into strongyle egg shedding consistency. Vet Parasitol. 2010;171:116–122. doi: 10.1016/j.vetpar.2010.03.001. [DOI] [PubMed] [Google Scholar]
- 24.Scheuerle MC, Stear MJ, Honeder A, Becher AM, Pfister K. Repeatability of strongyle egg counts in naturally infected horses. Vet Parasitol. 2016;228:103–107. doi: 10.1016/j.vetpar.2016.08.021. [DOI] [PubMed] [Google Scholar]
- 25.Polley L. Strongylid parasites of horses: Experimental ecology of the free-living stages on the Canadian prairie. Am J Vet Res. 1986;47:1686–1693. [PubMed] [Google Scholar]
- 26.Nielsen MK, Kaplan RM, Thamsborg SM, Monrad J, Olsen SN. Climatic influences on development and survival of free-living stages of equine strongyles: Implications for worm control strategies and managing anthelmintic resistance. Vet J. 2007;174:23–32. doi: 10.1016/j.tvjl.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 27.Cox DD, Todd AC. Survey of gastrointestinal parasitism in Wisconsin dairy cattle. J Am Vet Med Assoc. 1962;141:706–709. [PubMed] [Google Scholar]
- 28.Kyvsgaard NC, Lindbom J, Andreasen LL, Luna-Olivares LA, Nielsen MK, Monrad J. Prevalence of strongyles and efficacy of fenbendazole and ivermectin in working horses in El Sauce, Nicaragua. Vet Parasitol. 2011;181:248–254. doi: 10.1016/j.vetpar.2011.04.002. [DOI] [PubMed] [Google Scholar]
- 29.Larsen MM, Lendal S, Chriél M, Olsen SN, Bjørn H. Risk factors for high endoparasitic burden and the efficiency of a single anthelmintic treatment of Danish horses. Acta Vet Scand. 2002;43:99–106. doi: 10.1186/1751-0147-43-99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Klei TR, Chapman MR. Immunity in equine cyathostome infections. Vet Parasitol. 1999;85:123–136. doi: 10.1016/s0304-4017(99)00093-x. [DOI] [PubMed] [Google Scholar]
- 31.Vidyashankar AN, Hanlon BM, Kaplan RM. Statistical and biological considerations in evaluating drug efficacy in equine strongyle parasites using fecal egg count data. Vet Parasitol. 2012;185:45–46. doi: 10.1016/j.vetpar.2011.10.011. [DOI] [PubMed] [Google Scholar]
- 32.Corbett CJ, Love S, Moore A, Burden FA, Matthews JB, Denwood MJ. The effectiveness of faecal removal methods of pasture management to control the cyathostomin burden of donkeys. Parasit Vectors. 2014;7:48. doi: 10.1186/1756-3305-7-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reavell DG. Measuring and estimating the weight of horses with tapes, formulae and by visual assessment. Equine Vet Educ. 1999;11:314–317. [Google Scholar]
- 34.Wagner EL, Tyler PJ. A comparison of weight estimation methods in adult horses. J Equine Vet Sc. 2011;31:706–710. [Google Scholar]
- 35.Nielsen MK, Wang J, Davis R, et al. Parascaris univalens — A victim of large-scale misidentification? Parasitol Res. 2014;113:4485–4490. doi: 10.1007/s00436-014-4135-y. [DOI] [PubMed] [Google Scholar]
- 36.Reinemeyer CR, Nielsen MK. Review of the biology and control of Oxyuris equi. Equine Vet Educ. 2014;26:584–591. [Google Scholar]
- 37.Elsener J, Villeneuve A. Does examination of fecal samples 24 hours after cestocide treatment increase the sensitivity of Anoplocephala spp. detection in naturally infected horses? Can Vet J. 2011;52:158–161. [PMC free article] [PubMed] [Google Scholar]