Significance
Plants generate organs throughout their life as a consequence of the maintenance of postembryonic stem cell niches in meristems. The molecular mechanisms controlling stem cell homeostasis and organ emergence in shoot meristems have been well described, but the manner in which environmental signals influence them to generate plasticity is largely unknown. Using the shoot apical meristem of Arabidopsis as a model system, we show that plants can adapt their organogenesis rate to changes in the availability of nitrate in the soil within a few days, thanks to long-range signaling by cytokinin hormone precursors that travel through the plant, are converted to active hormones at the shoot meristem, and modulate the expression of WUSCHEL, a key regulator of stem cell homeostasis.
Keywords: Arabidopsis, shoot apical meristem, plant nutrition, plant development, cytokinin hormones
Abstract
The shoot apical meristem (SAM) is responsible for the generation of all the aerial parts of plants. Given its critical role, dynamical changes in SAM activity should play a central role in the adaptation of plant architecture to the environment. Using quantitative microscopy, grafting experiments, and genetic perturbations, we connect the plant environment to the SAM by describing the molecular mechanism by which cytokinins signal the level of nutrient availability to the SAM. We show that a systemic signal of cytokinin precursors mediates the adaptation of SAM size and organogenesis rate to the availability of mineral nutrients by modulating the expression of WUSCHEL, a key regulator of stem cell homeostasis. In time-lapse experiments, we further show that this mechanism allows meristems to adapt to rapid changes in nitrate concentration, and thereby modulate their rate of organ production to the availability of mineral nutrients within a few days. Our work sheds light on the role of the stem cell regulatory network by showing that it not only maintains meristem homeostasis but also allows plants to adapt to rapid changes in the environment.
Plants have evolved specific mechanisms to adapt their growth and physiology to the availability of mineral nutrients in their environment (1). Various hormones such as auxin, abscisic acid, gibberellin, and cytokinin have been shown to act in this process either locally or systemically (1). Cytokinins in particular play an essential role in plant response to nitrate, where they act as second messengers (2). For example, cytokinins promote lateral root development in areas rich in NO3 if the overall NO3 availability for the plant is low (3). In the shoot, cytokinins have been shown to modulate key traits such as leaf size (4, 5) and branch number (6) in response to nitrate.
Cytokinins have also been shown to be critical for the maintenance of stem cell homeostasis in the shoot apical meristem (SAM). By modulating the expression of WUSCHEL (WUS), encoding for a homeodomain transcription factor expressed in the center of the meristem, cytokinins promote stem cell proliferation, thus controlling the size of the meristem and the rate of shoot organogenesis (7–10). Using grafting experiments, a recent study showed that a systemic signal of a cytokinin precursor [trans-zeatin riboside (tZR)], traveling from root to shoot through xylem, could influence the size of the vegetative meristem (11). However, it remains unclear whether cytokinin signaling can allow the SAM to respond to changes in nutrient levels in the environment. Here, we examined how a core stem cell regulator in the SAM dynamically responds to changes in mineral nutrient levels in the soil and whether systemic cytokinin signals can account for the dynamic adaptation of meristem function to nutrient levels. We used the inflorescence meristem of Arabidopsis as a model, as this structure produces all the flowers of the plant, and is therefore a key target for crop improvement.
Results
Plant Nutritional Status Influences Shoot Meristem Function.
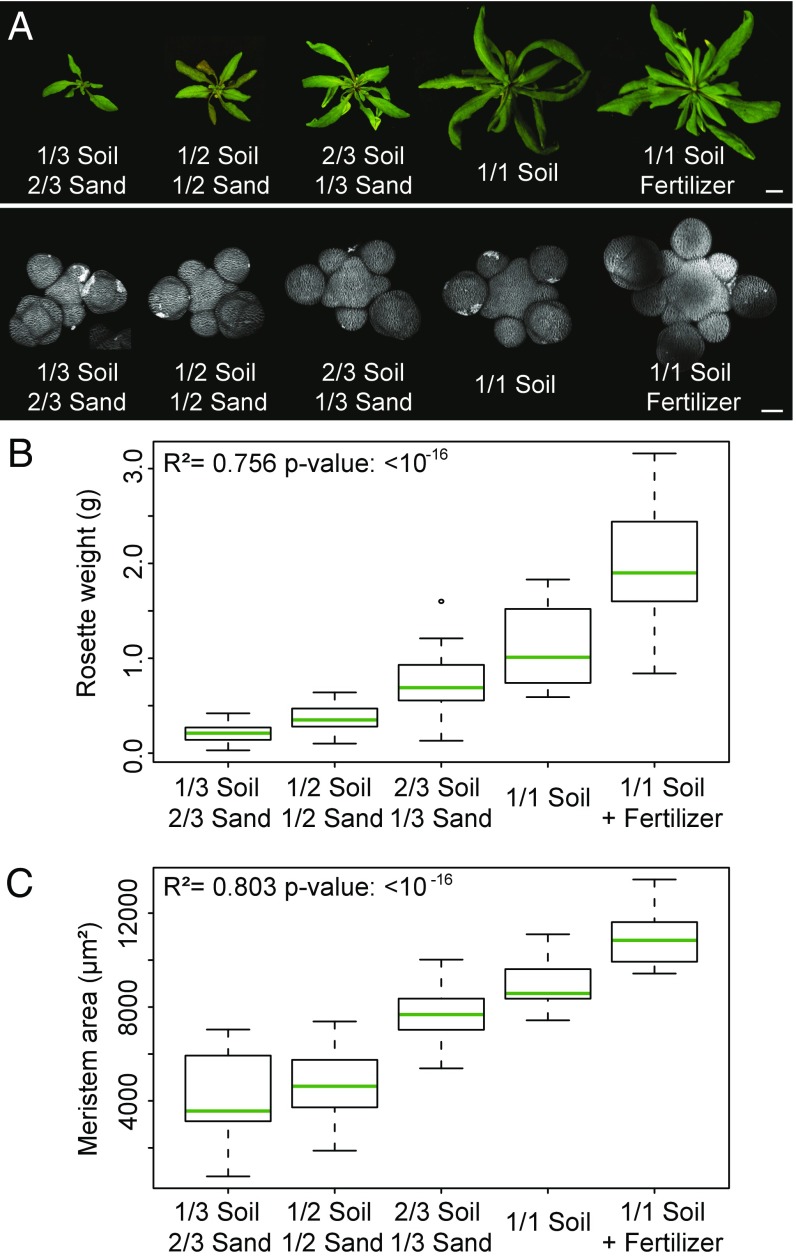
We first studied how fixed levels of nutrients affect meristem function by growing plants on soil containing diverse levels of nutrients (SI Appendix, Table S1). To assess meristem function, we measured the size of the meristem, the plastochron ratio (a feature that is inversely proportional to the organogenesis rate of the SAM) (10), and the number of flowers produced by the primary inflorescence (SI Appendix, SI Material and Methods). We observed that bigger, well-nourished plants had larger meristems and produced more flowers than smaller, more poorly nourished plants (Fig. 1 and SI Appendix, Fig. S1 A and B). We found a very close correlation between the weight of the rosette and the size of the meristem in individual plants (SI Appendix, Fig. S1C), showing that shoot development is coordinated when plants are grown under different nutritional conditions.
Fig. 1.
Plant nutritional status influences meristem function. (A) Morphology of the rosette (Top) and of the SAM (Bottom) of representative WT plants grown on soils of different nutritive quality. (Scale bar, Top, 1 cm; Bottom, 50 µm.) (B and C) Rosette weight (B) and meristem size (C) of WT plants grown on soils of different nutritive quality (1/3 soil 2/3 sand: n = 17; 1/2 soil 1/2 sand: n = 22; 2/3 soil 1/3 sand: n = 23; 1/1 soil: n = 21; 1/1 soil + fertilizer: n = 26, pool of two independent experiments). Error bars correspond to the mean ± SD. Data were compared using linear models.
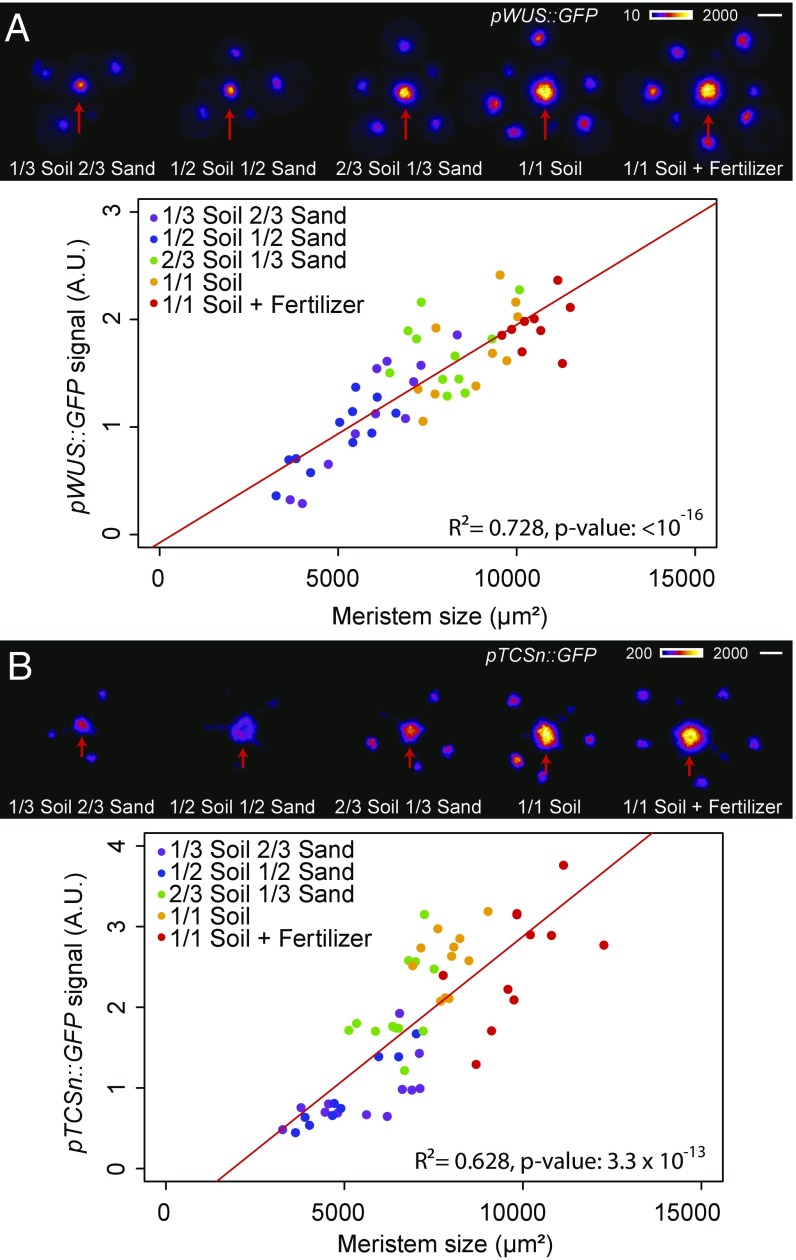
We next examined whether the observed changes in meristem size were linked to changes in stem cell homeostasis by examining fluorescent reporters for the key meristem regulatory genes, WUS and CLAVATA3 (CLV3). We developed a pipeline based on projections of 3D confocal stacks to automatically extract and analyze gene expression domains using a generalized exponential fit function (SI Appendix, Fig. S2 and SI Material and Methods). The intensity of the signal and the size of the domain of expression of pWUS::GFP were strongly correlated with the size of the SAM in the different growth conditions (Fig. 2A and SI Appendix, Fig. S3 A and B), which was also confirmed using a translational fusion (SI Appendix, Fig. S3C). CLV3 did not show such behavior in all experimental repeats, and only the size of its expression domain consistently correlated weakly with the size of the SAM (SI Appendix, Fig. S4). Such an uncoupling between WUS and CLV3 expression has been described in vegetative meristems of plants grown under different light levels (12) and suggests that these two genes could be differentially regulated by both nutrients and light.
Fig. 2.
Plant nutritional status influences stem cell homeostasis in the SAM. Expression of pWUS::GFP (A) and pTCSn::GFP (B) in WT plants grown on soils of different nutritive quality [n = 55 (A) and n = 57 (B)]. (Top) Representative plants. (Scale bars: 50 µm.) Red arrows point to the center of the inflorescence meristem. (Bottom) Total fluorescence signal in the inflorescence meristem vs. meristem size (SI Appendix, SI Material and Methods). Data were fitted using linear models.
As cytokinins modulate WUS and CLV3 expression (7–9), we examined the expression of pTCSn::GFP, a reporter of cytokinin response, when plants were grown with different levels of nutrients. Similar to what was observed with WUS, the intensity of the signal and the size of the domain of expression of the pTCSn::GFP reporter correlated with the size of the meristem in the different growth conditions (Fig. 2B and SI Appendix, Fig. S5), thus showing that plants growing with higher levels of nutrients exhibit higher levels of cytokinin signaling in the SAM. As altering cytokinin signaling is known to affect meristem homeostasis (13–18), we further studied the phenotype in the meristem of various mutants of cytokinin metabolism and their response to mineral nutrition.
Cytokinins Are Involved in the Response of the Meristem to Mineral Nutrients.
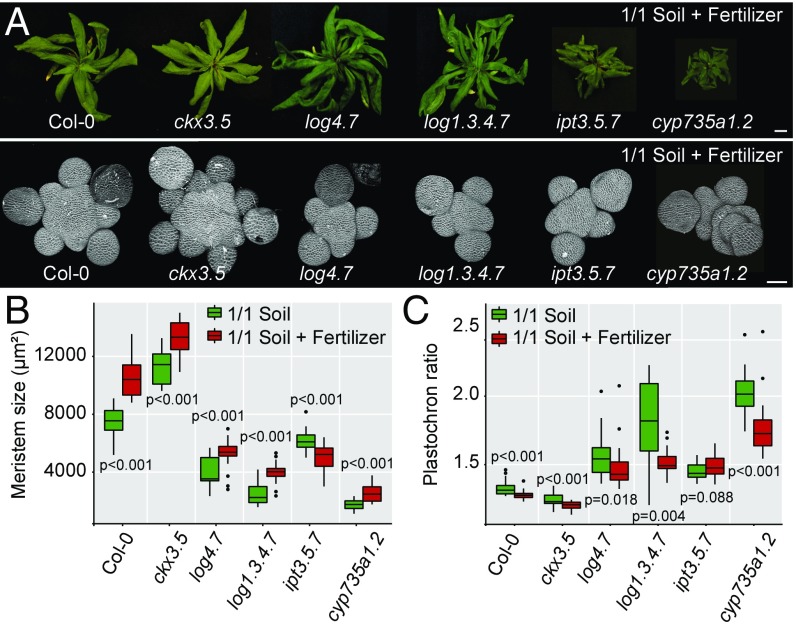
First, we looked at the phenotype of mutants altered in the successive steps of production of cytokinins via ISOPENTENYL TRANSFERASE (IPT) and LONELYGUY (LOG) enzymes (19): ipt3-1 ipt5-2 ipt7-1 (referred to here as ipt3.5.7) for the first step of biosynthesis and log4-3 log7-1 (log4.7) and log1-2 log3-2 log4-3 log7-1 (log1.3.4.7) for the second step of biosynthesis; for mutants for the conversion of cytokinins via CYTOCHROME P450 MONOOXYGENASE 735A (CYP735A) enzymes (20): cyp735a1-1 cyp735a2-1 (cyp735a1.2); and for the degradation of cytokinins via CYTOKININ OXIDASE (CKX) enzymes (13): ckx3-1 ckx5-2 (ckx3.5) in plants grown on soil supplied with fertilizer. Mutant shoots grew almost normally in this condition, and only the ipt3.5.7 and cyp735a1.2 mutants showed a consistent decrease in rosette weight (Fig. 3A and SI Appendix, Fig. S6A). However, meristem size and organogenesis rate were strongly affected by the mutations (Fig. 3 and SI Appendix, Fig. S6 B–D). The ipt3.5.7, log4.7, log1.3.4.7, and cyp735a1.2 mutants, altered in various steps of trans-zeatin (tZ)production, showed smaller inflorescence meristems that produced fewer organs. In contrast, the ckx3.5 mutant, which displays higher levels of cytokinins, showed larger inflorescence meristems that produced more organs, as previously described (13). We also quantified the size of the domain of expression of WUS by in situ hybridization (SI Appendix, Fig. S7). We observed changes in the size of the WUS expression domain in all genotypes that correlated with the changes in meristem size we quantified, thus supporting the idea that cytokinin metabolism controls meristem function through the stem cell regulatory network.
Fig. 3.
Cytokinins allow the adaptation of meristem function to plant nutritional status. (A) Morphology of the rosette (Top) and of the SAM (Bottom) of representative WT and CK-associated mutant plants. (Scale bar, Top, 1 cm; Bottom, 50 µm.) (B and C) Meristem size (B) and plastochron ratio (C) of WT and CK-associated mutant plants grown on soil either without fertilizer (green, g) or with fertilizer (red, r); [Col-0: n = 22 (g) and 25 (r); ckx3.5: n = 23 (g) and 27 (r); log4.7: n = 19 (g) and 24 (r); log1.3.4.7: n = 14 (g) and 25 (r); ipt3.5.7: n = 36 (g) and 20 (r); cyp735a1.2: n = 26 (g) and 20 (r), pool of two independent experiments]. Data were compared using Student’s t test. Error bars correspond to the mean ± SD.
We next looked at the effect of mineral nutrition on the phenotype of the mutants by comparing plants grown on soil with or without fertilizer. Of all mutants, only ipt3.5.7 mutants did not respond to fertilizer with an increase in meristem size and organogenesis rate, as observed in WT. Instead, it displayed a surprising statistically significant decrease in meristem size (Fig. 3). Although all the enzymes involved in cytokinin metabolism we studied can modulate meristem homeostasis, IPT enzymes, which catalyze the first step of cytokinin production, appear to be the only ones necessary for the response of the meristem to changes in nutrient availability in the soil. This result is supported by the fact that nitrate, the main mineral nutrient, can directly modulate the expression of IPT3 and, to a lesser extent, of IPT5 in seedlings (21). To further support that the response of meristems to mineral nutrients relies on cytokinin precursors produced by IPT enzymes, we used mass spectrometry to compare CK levels in inflorescences of plants grown without or with fertilizer. Despite technical variability in the mass spectrometry measurements between replicates (SI Appendix, SI Material and Methods), we observed a statistically significant increase in the levels of tZR precursors, the dephosphorylated products of IPT enzymes, in the two experimental replicates, further supporting the importance of these enzymes in the response of the meristem to nutrients (SI Appendix, Fig. S8 and Tables S2 and S3). We did not find significant changes in the levels of the active cytokinin tZ in the whole inflorescence, but found a significant increase in the levels of the degradation products of tZ: tZROG (trans-zeatin-O-glucoside riboside) and tZ7G (trans-zeatin-7-glucoside) (SI Appendix, Tables S2 and S3). This lack of changes in tZ levels could be a result of tZ being a transient molecule mainly synthesized locally in the meristem, notably through the action of LOG4 and LOG7 enzymes (7–9).
Cytokinin Precursors Act as Long-Range Signals in the Control of Meristem Function.
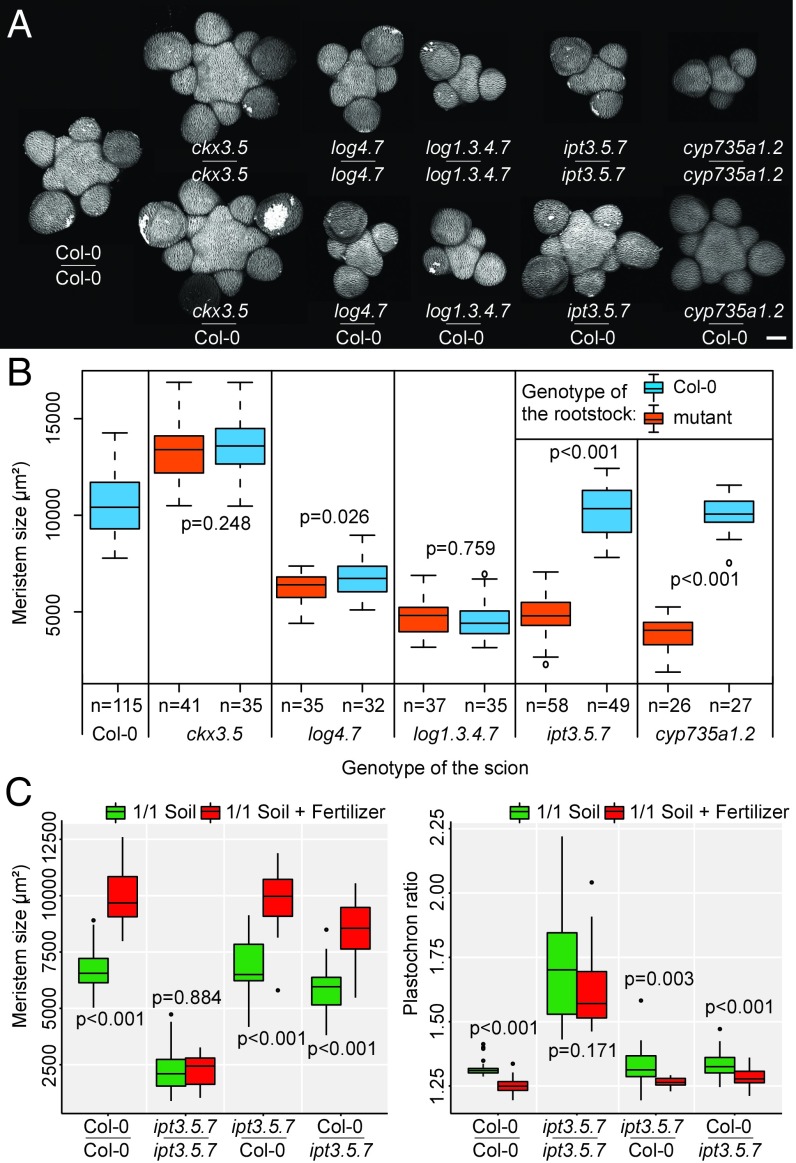
We further studied the spatial origin of the signal triggering the response of the meristem to mineral nutrition. Grafting experiments have previously shown that cytokinins can act either as local or as systemic signals in the control of root and shoot development (11, 20, 22). We performed grafting experiments on our set of mutants to study whether a systemic signal is involved in the control of meristem function. Adding a WT root was able to rescue the phenotype in the meristem of ipt3.5.7 and cyp735a1.2 mutants, but not of the ckx3.5 and the two log mutants (Fig. 4 A and B and SI Appendix, Fig. S9). Grafting a mutant root onto a WT scion led to a minor but statistically significant decrease in meristem size for log1.3.4.7, ipt3.5.7, and cyp735a1.2 mutant roots (SI Appendix, Fig. S10). These experiments show that expressing IPT and CYP735A in either part of the plant is sufficient but not completely necessary for proper meristem function, and that LOG and CKX must act largely in the shoot to regulate meristem function. Our results are in agreement with the recent work by Osugi et al. (11), who showed that the phenotype in the vegetative meristem of the ipt triple mutant, but not the log sextuple mutant, could be complemented by grafting a WT root. These results also make sense in light of the expression patterns of the different genes, as LOG and CKX are expressed in meristems (7, 9, 13), whereas IPTs and CYP735a1-2 are expressed predominantly in the vascular tissues of the root and, to a lesser extent, of the shoot (16, 23); only IPT7 expression has been shown to be induced by SHOOT MERISTEMLESS in the vegetative meristem (24). We next looked at the response to mineral nutrition of grafted combinations of WT and ipt3.5.7 mutant plants. Contrary to self-grafted mutants, ipt3.5.7 mutant plants grafted with WT root or WT shoot and growing with fertilizer showed larger meristems and reduced plastochron ratios than the same plants growing without fertilizer (Fig. 4C). IPT activity exclusively in the root or solely in the shoot is thus sufficient, but not necessary, for the meristem to respond to changes in the nutritional environment.
Fig. 4.
Cytokinin precursors act as long-range signals in the control of SAM homeostasis. (A) Representative meristems of WT and cytokinin-associated mutant plants self-grafted or grafted with a WT rootstock and grown on soil supplied with fertilizer. (Scale bar, 50 µm.) (B) Meristem size of WT and cytokinin-associated mutant plants self-grafted or grafted with a WT rootstock and grown on soil supplied with fertilizer (pools of two independent experiments; the numbers of biological replicates are displayed in the figure). Data were compared using Student’s t tests. Error bars correspond to the mean ± SD. (C) Meristem size (Left) and plastochron ratio (Right) of self-grafted or reciprocally grafted WT (Col-0) and ipt3.5.7 mutant plants grown on soil (green, g) or on soil supplied with fertilizer (red, r); [pools of two independent experiments; rootstock: Col-0 and scion: Col-0: n = 26 (g) and n = 22 (r); rootstock: ipt3.5.7 and scion: ipt3.5.7: n = 22 (g) and n = 15 (r); rootstock: Col-0 and scion: ipt3.5.7: n = 18 (g) and n = 21 (r); rootstock: ipt3.5.7 and scion: Col-0: n = 26 (g) and n = 28 (r)]. Data were compared using Student’s t tests. Error bars correspond to the mean ± SD.
To analyze the dynamics between production of cytokinin precursors and modulation of meristem function, we applied tZR and tZ to meristems expressing pTCSn::GFP and pWUS::GFP cut from the plant and grown in vitro. In the absence of added cytokinin, both pTCSn::GFP and pWUS::GFP signal strongly decreased over a 48-h time course, suggesting that extrinsic cytokinin is needed to sustain meristem homeostasis (Fig. 5). In contrast, adding 50 µM either of tZR or tZ to the medium up-regulated pTCSn::GFP expression ectopically and allowed the maintenance of the pWUS::GFP signal in the center of the meristem. Inducing IPT3 in cut meristems in the absence of extrinsic cytokinin also maintained, at least partially, WUS expression (SI Appendix, Fig. S11). Together with the results from the grafting experiments, these data support a model in which stem cell homeostasis is controlled by a systemic signal of cytokinin precursors (tZR) produced by IPT enzymes outside the meristem. As tZR alone cannot trigger cytokinin response (11), this systemic signal must be locally converted to active cytokinins by LOG enzymes in the shoot meristem.
Fig. 5.
Cytokinin precursors are sufficient to maintain meristem homeostasis in vitro. Effect of tZR and tZ application on the expression of pTCSn::GFP (A) and pWUS::GFP (B) in cut meristems grown in vitro (pools of two independent experiments, pTCSn::GFP: n = 11 for each condition, pWUS::GFP: n = 12 for each condition). (Top) Representative plants. (Scale bars, 50 µm.) Red arrows point to the center of the inflorescence meristem. (Bottom) Total fluorescence signal in the inflorescence meristem at 24h and 48h divided by the signal at 0h. Data were compared using Student’s t test. Error bars correspond to the mean ± SD.
Cytokinins Allow a Rapid Response of Meristems to Changes in Nitrate Availability.
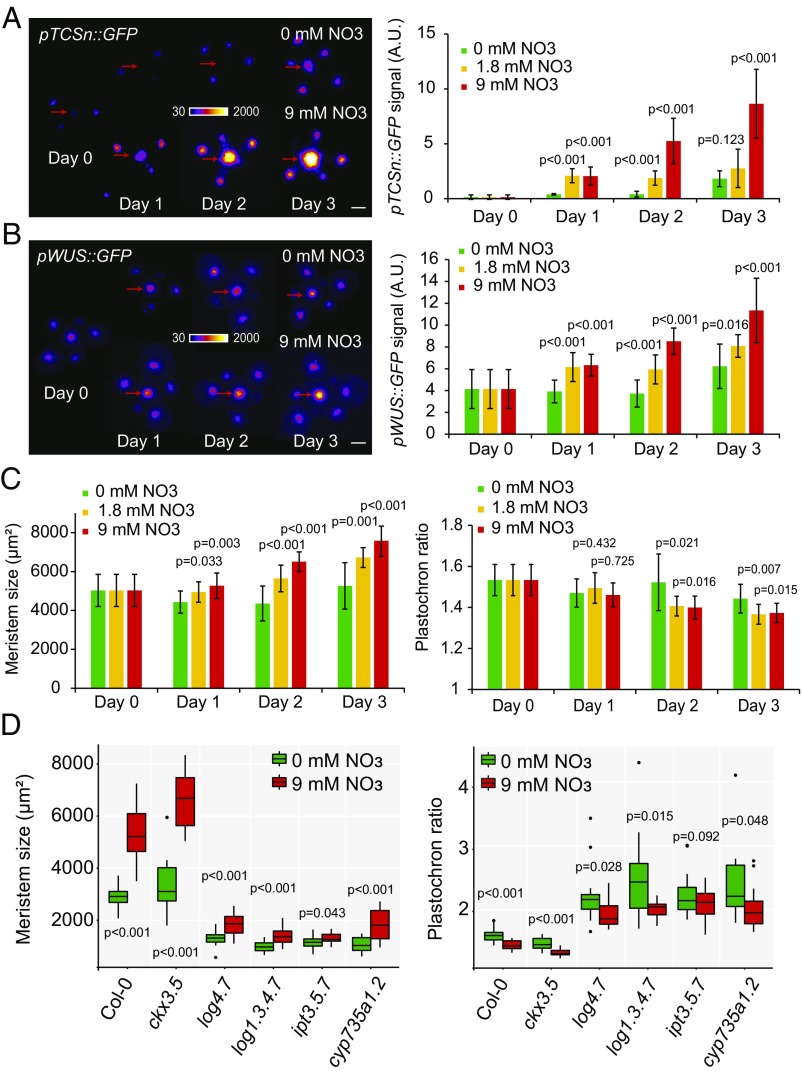
Nitrogen is a major nutrient that can modulate various aspects of plant development (25), and the availability of which in the soil can vary rapidly during the lifecycle of a plant (26). Adding nitrate (NO3) to nitrogen-deficient plants leads to a rapid and transient increase in cytokinin levels (11, 21, 27, 28). Given the rapid response of meristems to changes in cytokinin levels we observed in vitro, we looked to see whether the meristem could dynamically respond to changes in nitrate concentration in vivo. We grew plants on sand, watering them with a nutritive solution containing a low concentration of nitrate (1.8 mM NO3 as the only source of nitrogen) to induce a deficiency (6). Once the plants bolted, we watered them with a nutritive solution containing different concentrations of NO3 (0, 1.8, or 9 mM) and quantitatively analyzed the dynamics of response to this treatment. In the SAM, the treatment led to the dose-dependent induction of pTCSn::GFP and of pWUS::GFP expression within only 1 d, and to an increase of meristem size and of the organogenesis rate within 2–3 d (Fig. 6 A–C and SI Appendix, Fig. S12). Similar to what was observed when plants were grown on soils of different nutritive qualities, the nitrate treatment affected the size of CLV3 expression domain more than its levels (SI Appendix, Fig. S13). We then checked whether this response relied on changes in cytokinin levels in the plant. In the root, we confirmed that the addition of nitrate to deficient plants led to a rapid, dose-dependent, and transient induction of nitrate signaling (inferred from the level of expression of the nitrate responsive gene NITRATE REDUCTASE 1) and of IPT3 and IPT5 (SI Appendix, Fig. S14A). By measuring the levels of cytokinin species 2 d after the treatment by mass spectrometry, we also confirmed that the treatment led to a significant increase in the concentration of cytokinin precursors (tZR and/or trans-zeatin ribose phosphate, depending on the tissues) and products of degradation (tZ7G, tZ9G, and/or tZROG, depending on the tissues; SI Appendix, Fig. S14B and Table S4). Finally, we analyzed the meristem response in our set of mutants 3 d after a nitrate treatment (9 mM NO3). Similar to what we observed on plants grown in soil, the response of ipt3.5.7 mutant plants to nitrate in the meristem was strongly reduced, although statistically significant in one of the two experimental repeats, whereas response in the other mutant backgrounds was as in WT (Fig. 6D and SI Appendix, Fig. S15). In summary, our data show that the addition of nutrients leads to a response of the stem cell regulatory network within a day, and that the rapid changes in meristem properties can be explained by the same cytokinin-based mechanism of signal propagation we found for different fixed nutrient conditions (Fig. 3).
Fig. 6.
Nitrate modulates meristem homeostasis through IPTs. (A–C) Effect of nitrate resupply on pTCSn::GFP expression (A), pWUS::GFP expression (B), and meristem size and plastochron ratio (C). The treatment was performed at day 0, and different meristems were dissected and imaged each day (n = 8–12). (A and B, Left) Representative plants and (Right) quantification of total florescent signal. (Scale bars, 50 µm.) Red arrows point to the center of the inflorescence meristem. Data were compared using Student’s t tests. Error bars correspond to the mean ± SD. (D) Meristem size and plastochron ratio of WT and cytokinin-associated mutants 3 d after treatment with a nutritive solution containing either 0 mM (green, g) or 9 mM NO3 (red, r); [Col-0: n = 21 (g) and 24 (r), ckx3.5: n = 13 (g) and 12 (r); log4.7: n = 17 (g) and 18 (r); log1.3.4.7: n = 13 (g) and 15 (r); ipt3.5.7: n = 18 (g) and 19 (r); cyp735a1.2: n = 14 (g) and 14 (r)]. Data were compared using Student’s t test. Error bars correspond to the mean ± SD.
Discussion
Taken together, our results show that the meristem can adapt the rate of shoot organogenesis to the availability of nitrate in the soil. Mechanistically, this phenomenon correlates with the ability of WUS and cytokinin signaling output to quantitatively respond to variations in nitrate levels. As the main inflorescence of WT plants growing on soil only produced an average of 3.1 ± 0.4 flowers per day (n = 16), the response of the SAM to changes in nitrate levels, which leads to significant changes in the rate of organ production in only 2 d, should be seen as very rapid in comparison with the pace of morphogenesis in this tissue. The timing of the response to nutrients is very similar to the response to induced perturbations of the core network (29), in both cases causing expression domain changes in a day followed by changes in growth and size. Our findings thus expand the understanding of the function of the stem cell regulatory network in the SAM. They show that this network not only acts to maintain the integrity of the SAM during organogenesis but also allows a very rapid adaptation of SAM function to a nutritional cue.
A recent study showed that vegetative meristems of seedlings germinating in the dark displayed reduced WUS expression, which was controlled by the activity of CKX enzymes (12). Given the pleiotropic effects of cytokinins (2), we can hypothesize that different environmental signals could be integrated through cytokinin signaling and lead to different developmental responses in the meristem depending on whether they modulate, locally or globally, different aspects of cytokinin signaling.
Although it was developed in Arabidopsis, this model can in the future be applied to crops, where cytokinin metabolism and action on stem cell regulation should be conserved. In rice, where LOG mutants were first characterized (15), quantitative trait loci for increased grain productivity have been mapped to genes involved in the regulation of cytokinins, including notably a CKX enzyme-encoding gene (30, 31). In maize, weak mutants of FASCIATED-EAR3, a receptor able to bind a CLV3 homolog and involved in the control of WUS expression, also shows increased seed yield (32). Our work, which provides an integrative model based on nutrient availability, cytokinin metabolism, and its effect on stem cell regulation and meristem function (SI Appendix, Fig. S16), allows a better characterization of the influence of mineral nutrients on plant architecture and could be used to better understand plant response to environmental inputs and to develop cultivars with increased yield.
Methods
Detailed information on plant material, growth conditions, construction of inducible lines, image acquisition and analysis, grafting, mass spectrometry, qPCR, and in situ hybridization is provided in SI Appendix, SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Tanya Waldie and Maaike de Jong for sharing their knowledge on developmental plasticity and for help with the transient nitrate treatments, Jeremy Gruel for fruitful discussions on stem cell regulation, Paul Tarr for providing material and for comments on the manuscript, Jason Daff for his horticulture expertise, Raymond Wightman for help with imaging, and Hugo Tavares for help with statistical analysis. We also thank Chillie Zeng, Mark Bennett, and Rosa Lopez-Cobollo from Imperial College London for their help during the preparation and the analysis of cytokinin species by mass spectrometry. This work is supported by the Gatsby Charitable Foundation [fellowship GAT3395/DAA (to E.M.M.), GAT3272/GLC (to J.C.W.L.), and GAT3395-PR4 (to H.J.)]. E.M.M. also acknowledges support from the Howard Hughes Medical Institute and the Gordon and Betty Moore Foundation (through Grant GBMF3406). Research in the laboratory of J.C.W.L. was made possible by the award of a European Research Council under the European Union’s Seventh Framework Programme (FP/2007-2013)/ERC Grant Agreement 338060. P.F.-J. acknowledges a postdoctoral fellowship provided by the Herchel Smith Foundation.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1718670115/-/DCSupplemental.
References
- 1.Krouk G, et al. A framework integrating plant growth with hormones and nutrients. Trends Plant Sci. 2011;16:178–182. doi: 10.1016/j.tplants.2011.02.004. [DOI] [PubMed] [Google Scholar]
- 2.Werner T, Schmülling T. Cytokinin action in plant development. Curr Opin Plant Biol. 2009;12:527–538. doi: 10.1016/j.pbi.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 3.Ruffel S, et al. Nitrogen economics of root foraging: Transitive closure of the nitrate-cytokinin relay and distinct systemic signaling for N supply vs. demand. Proc Natl Acad Sci USA. 2011;108:18524–18529. doi: 10.1073/pnas.1108684108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rahayu YS, et al. Root-derived cytokinins as long-distance signals for NO3–Induced stimulation of leaf growth. J Exp Bot. 2005;56:1143–1152. doi: 10.1093/jxb/eri107. [DOI] [PubMed] [Google Scholar]
- 5.Walch-Liu P, Neumann G, Bangerth F, Engels C. Rapid effects of nitrogen form on leaf morphogenesis in tobacco. J Exp Bot. 2000;51:227–237. doi: 10.1093/jexbot/51.343.227. [DOI] [PubMed] [Google Scholar]
- 6.Müller D, et al. Cytokinin is required for escape but not release from auxin mediated apical dominance. Plant J. 2015;82:874–886. doi: 10.1111/tpj.12862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chickarmane VS, Gordon SP, Tarr PT, Heisler MG, Meyerowitz EM. Cytokinin signaling as a positional cue for patterning the apical-basal axis of the growing Arabidopsis shoot meristem. Proc Natl Acad Sci USA. 2012;109:4002–4007. doi: 10.1073/pnas.1200636109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gordon SP, Chickarmane VS, Ohno C, Meyerowitz EM. Multiple feedback loops through cytokinin signaling control stem cell number within the Arabidopsis shoot meristem. Proc Natl Acad Sci USA. 2009;106:16529–16534. doi: 10.1073/pnas.0908122106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gruel J, et al. An epidermis-driven mechanism positions and scales stem cell niches in plants. Sci Adv. 2016;2:e1500989. doi: 10.1126/sciadv.1500989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Landrein B, et al. Meristem size contributes to the robustness of phyllotaxis in Arabidopsis. J Exp Bot. 2015;66:1317–1324. doi: 10.1093/jxb/eru482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Osugi A, et al. Systemic transport of trans-zeatin and its precursor have differing roles in Arabidopsis shoots. Nat Plants. 2017;3:17112. doi: 10.1038/nplants.2017.112. [DOI] [PubMed] [Google Scholar]
- 12.Pfeiffer A, et al. Integration of light and metabolic signals for stem cell activation at the shoot apical meristem. Elife. 2016;5:e17023. doi: 10.7554/eLife.17023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bartrina I, Otto E, Strnad M, Werner T, Schmülling T. Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell. 2011;23:69–80. doi: 10.1105/tpc.110.079079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Higuchi M, et al. In planta functions of the Arabidopsis cytokinin receptor family. Proc Natl Acad Sci USA. 2004;101:8821–8826. doi: 10.1073/pnas.0402887101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kurakawa T, et al. Direct control of shoot meristem activity by a cytokinin-activating enzyme. Nature. 2007;445:652–655. doi: 10.1038/nature05504. [DOI] [PubMed] [Google Scholar]
- 16.Miyawaki K, et al. Roles of Arabidopsis ATP/ADP isopentenyltransferases and tRNA isopentenyltransferases in cytokinin biosynthesis. Proc Natl Acad Sci USA. 2006;103:16598–16603. doi: 10.1073/pnas.0603522103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nishiyama R, et al. Analysis of cytokinin mutants and regulation of cytokinin metabolic genes reveals important regulatory roles of cytokinins in drought, salt and abscisic acid responses, and abscisic acid biosynthesis. Plant Cell. 2011;23:2169–2183. doi: 10.1105/tpc.111.087395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Werner T, et al. Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell. 2003;15:2532–2550. doi: 10.1105/tpc.014928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kieber JJ, Schaller GE. Cytokinins. Arabidopsis Book. 2014;12:e0168. doi: 10.1199/tab.0168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kiba T, Takei K, Kojima M, Sakakibara H. Side-chain modification of cytokinins controls shoot growth in Arabidopsis. Dev Cell. 2013;27:452–461. doi: 10.1016/j.devcel.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 21.Takei K, et al. AtIPT3 is a key determinant of nitrate-dependent cytokinin biosynthesis in Arabidopsis. Plant Cell Physiol. 2004;45:1053–1062. doi: 10.1093/pcp/pch119. [DOI] [PubMed] [Google Scholar]
- 22.Matsumoto-Kitano M, et al. Cytokinins are central regulators of cambial activity. Proc Natl Acad Sci USA. 2008;105:20027–20031. doi: 10.1073/pnas.0805619105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Miyawaki K, Matsumoto-Kitano M, Kakimoto T. Expression of cytokinin biosynthetic isopentenyltransferase genes in Arabidopsis: Tissue specificity and regulation by auxin, cytokinin, and nitrate. Plant J. 2004;37:128–138. doi: 10.1046/j.1365-313x.2003.01945.x. [DOI] [PubMed] [Google Scholar]
- 24.Yanai O, et al. Arabidopsis KNOXI proteins activate cytokinin biosynthesis. Curr Biol. 2005;15:1566–1571. doi: 10.1016/j.cub.2005.07.060. [DOI] [PubMed] [Google Scholar]
- 25.Kiba T, Kudo T, Kojima M, Sakakibara H. Hormonal control of nitrogen acquisition: Roles of auxin, abscisic acid, and cytokinin. J Exp Bot. 2011;62:1399–1409. doi: 10.1093/jxb/erq410. [DOI] [PubMed] [Google Scholar]
- 26.Giehl RF, von Wirén N. Root nutrient foraging. Plant Physiol. 2014;166:509–517. doi: 10.1104/pp.114.245225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Takei K, Sakakibara H, Taniguchi M, Sugiyama T. Nitrogen-dependent accumulation of cytokinins in root and the translocation to leaf: Implication of cytokinin species that induces gene expression of maize response regulator. Plant Cell Physiol. 2001;42:85–93. doi: 10.1093/pcp/pce009. [DOI] [PubMed] [Google Scholar]
- 28.Peterson JB, Miller CO. Cytokinins in Vinca rosea L. Crown gall tumor tissue as influenced by compounds containing reduced nitrogen. Plant Physiol. 1976;57:393–399. doi: 10.1104/pp.57.3.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reddy GV, Meyerowitz EM. Stem-cell homeostasis and growth dynamics can be uncoupled in the Arabidopsis shoot apex. Science. 2005;310:663–667. doi: 10.1126/science.1116261. [DOI] [PubMed] [Google Scholar]
- 30.Ashikari M, et al. Cytokinin oxidase regulates rice grain production. Science. 2005;309:741–745. doi: 10.1126/science.1113373. [DOI] [PubMed] [Google Scholar]
- 31.Wu Y, et al. The QTL GNP1 encodes GA20ox1, which increases grain number and yield by increasing cytokinin activity in rice panicle meristems. PLoS Genet. 2016;12:e1006386. doi: 10.1371/journal.pgen.1006386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Je BI, et al. Signaling from maize organ primordia via FASCIATED EAR3 regulates stem cell proliferation and yield traits. Nat Genet. 2016;48:785–791. doi: 10.1038/ng.3567. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.