Abstract
To examine the effects of short high-intensity interval training (HIIT) on body composition, physical performance and plasma lipids in overweight/obese compared to normal-weight young men. Nine overweight/obese and nine normal-weight men (control group) aged 17 to 20 years underwent a HIIT programme three times per week for eight weeks. Body composition, indices of aerobic [maximal aerobic velocity (MAV) and maximal oxygen uptake (VO2max)] and anaerobic [squat jump (SJ), counter-movement jump (CMJ), five-jump test (FJT), 10-m and 30-m sprint] performances, as well as fasting plasma lipids, were assessed in the two groups at PRE and POST HIIT. The HIIT programme resulted in significant reductions in body mass (-1.62%, P=0.016, ES=0.11) and fat mass (-1.59%, P=0.021, ES=0.23) in obese, but not in normal-weight subjects. MAV (+5.55%, P=0.005, ES=0.60 and +2.96%, P=0.009, ES=0.82), VO2max (+5.27%, P=0.006, ES=0.63 and +2.88%, P=0.009, ES=0.41), FJT (+3.63%, P=0.005, ES=0.28 and +2.94%, P=0.009, ES=0.52), SJ (+4.92%, P=0.009, ES=0.25 and +6.94%, P=0.009, ES=0.70) and CMJ (+6.84%, P=0.014, ES=0.30 and +6.69%, P=0.002, ES=0.64) significantly increased in overweight/obese and normal-weight groups, respectively. 30-m sprint time significantly decreased in both groups (-1.77%, P=0.038, ES=0.12 and -0.72%, P=0.030, ES=0.16). Plasma total cholesterol (-11.8%, P=0.026, ES=0.96), LDL cholesterol (-11.9%, P=0.050, ES=0.77) and triglycerides (-21.3%, P=0.023, ES=1.08) significantly decreased in the obese group, but not in the normal-weight group. The eight-week HIIT programme resulted in a slight improvement in physical fitness and a significant decrease in plasma lipids in the obese. Short duration HIIT may contribute to an improved cardiometabolic profile in the obese.
Keywords: Body composition, Interval training, Physical performance, Obesity, Lipids
INTRODUCTION
Obesity has been identified as a major health problem worldwide, especially in youth [1]. It is associated with increased risk of cardiovascular diseases, type 2 diabetes mellitus, osteoarthritis, respiratory problems and cancer [2] as well as reduced life expectancy [3]. Physical activity is an efficient method for the prevention and treatment of obesity. It promotes physical and mental health, thereby improving the quality of life [4, 5]. Continuous training is considered beneficial for maintaining cardiovascular fitness [6-8]. Endurance exercise training demonstrated significant improvement in lipid profile in normal-weight active and sedentary and in obese individuals [3, 7, 9]. High-intensity interval training (HIIT) is a mode of exercise training that consist of 30 s all-out sprints separated by recovery intervals [10]. A body of evidence has demonstrated comparable or superior improvements in cardiometabolic fitness using HIIT in comparison to moderate endurance training [10-19]. Accordingly, it was suggested that HIIT provides a useful alternative to traditional endurance-based exercise recommendations for health promotion. The use of HIIT interventions is often perceived as unpractical and intolerable by middle-aged and aged individuals. However, youth and especially those with weight excess have difficulty, and perhaps little interest, in practising long duration endurancebased activities. They more easily adhere to low-volume and more time-efficient high-intensity intermittent training. So, further studies are needed to support the role of alternative evidence-based exercise recommendations. The purpose of this study was to examine the effects of a HIIT programme on body composition, physical performance and plasma lipids in obese youth. The working hypothesis of the present study was that HIIT intervention is effective in improving body composition and lipid profile in the obese.
MATERIALS AND METHODS
Study subjects
Twenty young males aged 17 to 20 years gave their written consent to participate in the study. The study was approved by the Scientific and Ethics Committee of High Institute of Sports and Physical Education of Kef and the protocol was conducted according to the Declaration of Helsinki. Participants were students attending two secondary schools in the city of Dahmani (Kef, Tunisia). They were randomly selected and divided according to body mass index (BMI) into an overweight/obese group (OG; BMI>25 kg/m2, n=10) and a normal-weight group (NWG, BMI<25 kg/m2, n=10). All participants provided selected information on life style factors and medical history. None of the participants was a former or current smoker or was using medication and none had a history of disease or injury that would prevent physical activity. A medical examination was applied to each participant prior to inclusion, revealing no contraindication to physical exercise. Participants in both groups were undergoing two to three hours of physical education lessons. During the study, a participant had difficulty adapting to intense exercise and decided to stop participating in the programme. Another participant was absent several times and was subsequently excluded from the study. The remaining participants, nine in each group, attended all sessions of the training programme and no one was injured.
Study protocol
The protocol was conducted from February to April 2014. The temperature varied between 17°C and 23°C and the humidity ranged from 70% to 75%. Participants were familiarized with different exercises and tests during the week preceding training programme onset. They had indices of body composition and biochemical parameters recorded as well as selected physical performance measures at baseline (pre) and post-intervention.
Training programme
The training programme was carried out for eight consecutive weeks with three sessions per week according to Buchan et al. [10, 12, 13]. HIIT sessions were performed in Dahmani stadium in the afternoon on days without physical education classes. The HIIT protocol consisted of two series of 30-second runs at 100-110% of the maximal aerobic velocity (MAV) interspersed with 30 seconds of active recovery. Training progression was implemented by increasing the number of repetitions from the 3rd week, and increasing the intensity of the work from the 5th week (5% increase of the MAV every two weeks) (Table 1). During the programme, participants were requested to maintain their normal eating habits.
Table 1.
Eight weeks of high-intensity interval training (HIIT) programme.
| Week of training | 1-2 | 3-4 | 5-6 | 7-8 |
|---|---|---|---|---|
| Number of series | 2 | 2 | 2 | 2 |
| Number of races per series | 8 | 10 | 10 | 10 |
| Run/active recuperation time, second | 30/30 | 30/30 | 30/30 | 30/30 |
| Percent of MAV (run/active recuperation) | (100/50) | (100/50) | (105/50) | (110/50) |
| Passive recovery time, min | 5 | 5 | 5 | 5 |
| Training load, ATU | 600 | 750 | 775 | 800 |
MAV, maximal aerobic velocity; ATU, arbitrary training units
Example: [2 x (8 x 30s/30s); 100%/50% MAV; passive recovery time = 5 min]. It means that the subject had to run 2 series of 8 times 30 s: composed of 30 s running at 100% of MAV and 30 s active recovery at 50% of MAV. The subject recovers passively for 5 min between each two series. Each session is repeated 3 times a week. Example of training load calculation for training sessions during the first week: [(100 + 50)/2] x 4 x 2 = 600 ATU.
Participants were dividd into four working subgroups according to the MAV obtained through the Vameval (MAV, 10 to 12 km/h and 12 to 14 km/h for OG; and 14 to 15 km/h and 15 to 16 km/h for NWG). All sessions included three different periods; the sessions started with a standardized warm-up, which consisted of 10 min of continuous jogging at moderate intensity (50% of MAV), followed by 5 min of dynamic stretching exercises and 5 short bursts of 20-m accelerations on the track. Then, the subjects performed their training programme. They had to run for 30 seconds a given distance marked by two cones (cones 1 and 2). An acoustic signal was given at the start (cone 1) and at the end of the sprint (cone 2). The 30-second active recovery was also controlled. From the arrival cone (cone 2), the subject had to run at 50% of MAV in the opposite direction for 15 seconds and then return to the arrival cone (cone 2). In order to help the participant to manage the recovery period, a signal was given at half recovery (after 15 seconds). For the following sprint, the subject had to run in the opposite direction starting from the arrival cone, and so on. At the end of the session, the subject cooled down for about 10 min by running at low intensity and performing static stretching. Training session data, mainly timing of the sprint and the recovery period, were recorded by the same investigator (NO).
Anthropometric measurements
Anthropometric measurements were performed with the subjects bare-footed and lightly clothed. The height (m) was measured with a standing stadiometer and recorded with a precision of 0.1 cm. Subject body mass (kg) was measured with a TPRO 3100 electronic balance (Terraillon). Body mass index (BMI) was calculated: BMI (kg/m2)=body mass/height2. Skinfold thickness was determined in triplicate at four sites (biceps, triceps, subscapular and suprailiac), using a calibrated Harpenden caliper (Holtain Instruments, Pembrokeshire, UK). The mean of the three values was recorded for each site. Body density (D) was calculated according to the equations of Durnin and Wormersley [20] for men aged 17-19 years as D=1.162 – [0.063 (log Σ)] and for men aged 20-29 years as D=1.1631-[0.0632 (log Σ)], where Σ is the sum of the four skinfolds (in mm). Percentage body fat (BF) was calculated from D using Siri’s equation [21] as follows: BF=(4.95/D – 4.50) x 100.
Physical testing
All the subjects were subjected to different field and laboratory tests, during which they were strongly encouraged to attain their maximum performance. All the tests were carried out between 3 and 5 PM.
Incremental running test: The Vameval test [22] was performed on a 400-m outdoor running track. The participants were asked not to perform any physical effort during the 48 hours prior to the test. Twenty cones were placed on the track every 20 m as a reference. The test starts at a running speed of 8 km/h and increases by 0.5 km/h every minute until exhaustion. Participants adjusted their running speed to the cones placed at 20-m intervals. They were encouraged throughout the test. The test ended when the subject could no longer maintain the required running speed dictated by the audio beep for 2 consecutive occasions. Heart rate was recorded during the test, using a Polar heart rate monitor (Polar S810, Kempele, Finland), and heart rate maximum (HRmax) was recorded at the end of the final level reached.
Vertical jump measures: Squat jump (SJ) and counter movement jump (CMJ) were carried out as described by Bosco et al. [23], using an Optojump system (Globus; Microgate Ltd., Italy). The two tests differ in the starting position, which is a standing position for CMJ and 90° of flexion of the knee joints for SJ. Participants are instructed to jump as high as possible while keeping their hands on their hips. Performance in SJ and CMJ was expressed in flight height (cm). For each test, participants performed three trials with one min of recovery in between. The best performance was retained.
Horizontal jump measures: The five-jump test (FJT) was carried out as described by Chamari et al. [24]. It consists of five successive horizontal jumps. The subject begins with feet together and ends in the same position. Starting at the right station, the subject performs five strides. He jumps on one leg (right or left), raising the knee and the arms in front. During the fifth stride the subject brings back both legs together to return to the starting position. Performance was expressed as total distance (m).
10 m and 30 m sprints: The performance was evaluated at 10 m and 30 m, during a 30-m run, after a warm-up for 10 minutes comprising runs of low intensity, followed by three accelerations and muscle stretching of the lower limbs [25]. The subjects started from a standing position and the sprint time was registered with photo-electric cells (Microgate SARL, Italy) positioned at the start line and at 10 m and 30 m. The photoelectric cells were placed at shoulder height. Each subject performed two tests; the best time was chosen.
Blood sampling and methods of analysis
Fasting blood was obtained from the antecubital vein in heparinized tubes in pre- and post-intervention. Blood samples were centrifuged at 2000 x g for 25 min and plasma was frozen at -40°C until analysis (within 3 months). Total cholesterol (TC) and HDL cholesterol (HDL-C) and triglyceride (TG) were assessed by the enzymatic method on an Architect C8000 auto analyzer (Abbott Laboratories, Abbott Park, IL) using respective reagent kits. LDL cholesterol (LDL-C) was calculated using the Friedewald formula [26].
Statistical analysis
Statistical analysis was performed using the SPSS version 16.0 software package (SPSS Inc., Chicago, IL). The assumption of normality of the data was confirmed using the Kolmogorov-Smirnov test and homogeneity of variance was verified using Levene’s test. The independent-samples t-test was used to compare basal variables between groups (OG vs. NWG). The paired-samples t-test was used to compare pre-training and post-training variables in each group (OG or NWG). Changes in dependent variables resulting from the training programme were assessed by two-way (time*group) repeated measures analysis of variance. The effect size (ES) was calculated using Cohen’s d; these calculations were based on Cohen’s classification of a small (0.2 < ES <0.5), moderate (0.5 < ES <0.8) and large (ES ≥ 0.8) effect size [27]. A p-value <0.05 based on two-sided calculation was considered significant.
RESULTS
At inclusion, age and height were similar for both two groups, but body mass, BMI and BF were significantly higher (P<0.001) in OG. The eight-week HIIT programme resulted in a significant decrease in body mass (P=0.016; ES=0.11), BMI (P=0.015; ES=0.12) and BF (P=0.021; ES=0.23) in OG, but not in NWG (Table 2). PRE training MAV and VO2max were significantly lower (P<0.001) in OG than NWG. After completing the training programme, the MAV and VO2max significantly increased in both groups (OG and NWG) [MAV (+5.55%, P=0.005, ES=0.60 and +2.96%, P=0.009, ES=0.82), VO2max (+5.27%, P=0.006, ES=0.63 and +2.88%, P=0.009, ES=0.41), respectively]. The FJT, SJ and CMJ significantly increased in OG and NWG (FJT, +3.63%, P=0.005, ES=0.28 and +2.94%, P=0.009, ES=0.52; SJ, +4.92%, P=0.009, ES=0.25 and +6.94%, P=0.009, ES=0.70; CMJ, +6.84%, ES=0.014, ES=0.30 and +6.69%, P=0.002, ES=0.64, respectively). Compared to PRE training values, 10-m sprint and 30-m sprint times significantly improved in OG [-1.14%, P=0.032, ES=0.20 and -0.77%, P=0.038, ES=0.12, respectively]. In NWG, only the 30-m sprint time significantly improved (-0.72%, P=0.030, ES=0.16) (Table 3).
TABLE 2.
Anthropometric characteristics at baseline (PRE training) and following high-intensity interval training programme (POST training) in overweight/obese and normal-weight groups.
| Normal-weight group (n=9) | Overweight/obese group (n=9) | Interaction (time * group)a | ||||
|---|---|---|---|---|---|---|
| PRE training | POST training | PRE training | POST training | F | P | |
| Age (years) | 18.1±0.93 | 18.3±1.22 | ||||
| Height (m) | 1.75±0.03 | 1.74±0.07 | ||||
| Body mass (kg) | 62.6±4.61 | 62.5±4.90 | 93.7±16.9† | 92.0±15.9* | 5.96 | 0.027 |
| Body mass index (kg/m2) | 20.5±1.51 | 20.5±1.67 | 30.8±4.56† | 30.3±4.25* | 6.24 | 0.024 |
| Percentage of body fat | 12.0±3.28 | 11.9±3.10 | 22.5±1.87† | 22.1±1.82* | 2.17 | 0.160 |
Data are expressed as mean ± SD
P< 0.001 (compared to pre-training values in normal-weight group)
P< 0.05 (compared to Pre training values in the same group)
, comparison was performed using two-way repeated measures ANOVA.
TABLE 3.
Indices of aerobic and anaerobic performance and plasma lipids at inclusion (PRE training) and after 8 weeks of a high-intensity interval training (POST training) programme in overweight/obese and normal-weight groups.
| Normal-weight group (n=9) | Overweight/obese group (n=9) | Interaction (time * group)a | ||||
|---|---|---|---|---|---|---|
| PRE training | POST training | PRE training | POST training | F | P | |
| MAV (km/h) | 14.9±0.53 | 15.4±0.74** | 11.5±1.15††† | 12.1±0.96** | 0.64 | 0.434 |
| VO2max (ml/kg/min) | 54.1±1.84 | 55.6±2.58** | 42.0±4.03††† | 44.2±3.37** | 0.58 | 0.459 |
| HR max (beat/min) | 190±10.01 | 189±10.20 | 193±8.63 | 192±7.58 | 0.03 | 0.859 |
| 10-m sprint time (s) | 2.01±0.11 | 2.01±0.14 | 2.40±0.17††† | 2.37±0.15* | 1.13 | 0.304 |
| 30-m sprint time (s) | 4.62±0.19 | 4.59±0.20* | 5.63±0.47††† | 5.58±0.43* | 0.30 | 0.591 |
| Squat jump (cm) | 28.2±2.77 | 30.0±2.71** | 19.8±4.18††† | 20.8±4.21** | 2.31 | 0.148 |
| CMJ (cm) | 30.0±3.24 | 32.0±3.43** | 21.6±4.92††† | 23.0±5.11** | 0.74 | 0.404 |
| Five-jump test (m) | 11.1±0.53 | 11.4±0.69** | 8.99±1.18††† | 9.30±1.15** | 0.01 | 0.910 |
| Cholesterol (mg/dL) | 136±20.3 | 127±20.2 | 175±36.0†† | 150±15.8* | 2.39 | 0.142 |
| Triglycerides (mg/dL) | 82.5±31.1 | 68.4±16.5 | 122±39.0† | 90.0±21.2* | 1.25 | 0.280 |
| LDL cholesterol (mg/dL) | 85.2±19.2 | 79.2±15.4 | 113±30.0† | 96.2±13.1* | 1.62 | 0.221 |
| HDL cholesterol (mg/dL) | 36.3±6.15 | 36.4±7.23 | 37.0±2.12 | 37.2±3.21 | 0.09 | 0.764 |
Note: Data are expressed as mean ± SD; MAV, maximal aerobic velocity; VO2max, maximal oxygen uptake; HR max, maximum heart rate; CMJ, counter-movement jump
, P < 0.05
, P < 0.01 (compared to pre-training values in the same group)
, P< 0.05
, P < 0.01
, P < 0.001 (compared to pre-training values in normal-weight group).
, comparison was performed using two-way repeated measures ANOVA.
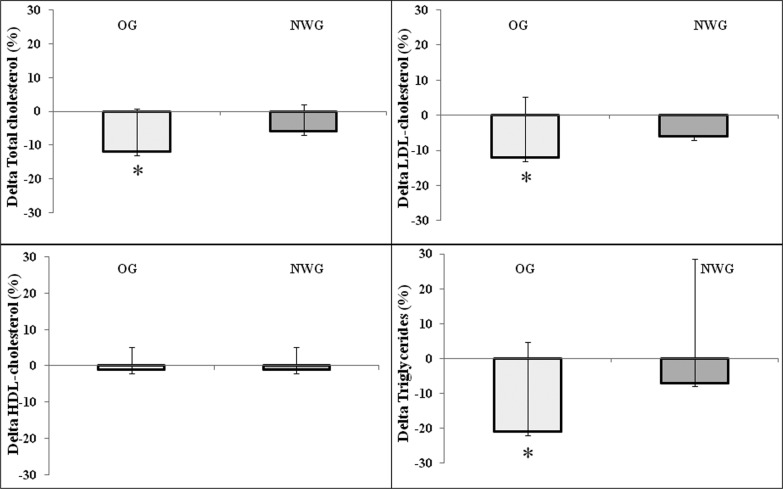
Baseline plasma TC, LDL-C and TG concentrations were significantly higher in OG compared to NWG (Table 3). Compared to PRE training values, plasma TC, TG and LDL-C significantly decreased in OG (-11.8%, P=0.026, ES=0.96 and -21.3%, P=0.023, ES=1.08 and -11.9%, P=0.050, ES=0.77, respectively) after 8-week HIIT. In NWG, the decrease was of low magnitude and non-significant (-6.40%, P=0.062, ES=0.48 and -6.56%, P=0.332, ES=0.60; and -5.40%, P=0.171, ES=0.37, respectively). HDL-C concentrations remained unchanged in both groups POST training (Figure 1). Repeated measures detected a significant difference between OG and NWG for body mass and BMI, only (Table 2). No significant differences were detected for any other variables (Table 3).
Figure 1.
Variation (in percentage) of plasma total cholesterol, LDL and HDL cholesterol, and triglyceride after 8 weeks of high-intensity interval training programme in overweight/obese (OG) and normal-weight (NWG) groups *, P<0.05 (significant difference PRE/POST training in the same group).
DISCUSSION
The HIIT programme resulted in a slight body mass loss and significant improvement of lipid profile in OG, as well as an improvement of indices of aerobic and anaerobic performance in both groups. These findings agree with previous HIIT studies showing beneficial effects of intermittent exercises on body mass in overweight and obese subjects [17, 18, 28, 29]. However, no significant changes in body composition were observed in overweight/obese males following two or four weeks of intermittent training [30, 31]. In active or sedentary normal-weight young men, interval training has contradictory results, with either improvement [15, 32] or no effect on body composition [14, 33]. Thus, the impact of intermittent training on body composition may depend on factors such as the training duration and intensity, the method of body composition measurement and dietary intervention.
Following HIIT, indices of aerobic capacity (MAV and VO2max) significantly increased in both overweight/obese and normal-weight subjects, which agrees with the available literature [17, 28, 29, 32, 33]. In the same context, a recent study of Sawyer et al. [34] demonstrated a positive effect of 8-week HIIT on VO2max in obese adults. Tjønna et al. [35] concluded that interval training is more effective than a multidisciplinary approach (exercise, diet and psychological advice) in improving VO2max in overweight and obese adolescents. Additionally, studies of Buchan et al. [10, 12] confirmed the impact of a 7-week HIIT programme on aerobic performance in normal weight subjects. It was suggested that interval training allows one to decrease the production of lactic acid and to improve the use of phosphocreatine during exercise [36]. Changes in aerobic capacity indices might also be related to changes in body composition, especially in obese subjects [28]. However, weight loss is not mandatory for exercise-induced effects on improving aerobic and anaerobic capacity [37].
Intermittent training has been proven to improve anaerobic performance in trained and active populations [11, 38]. However, studies assessing the effects of HIIT on anaerobic parameters in the obese are rare. Our study showed that an eight-week HIIT programme is effective to improve anaerobic performance in both normal-weight and overweight/obese subjects. These findings agrees with the studies of Buchan et al. [10, 12, 13, 39] that demonstrated significant enhancements in anaerobic markers (vertical jump performance, 10 m sprint speed) and cardiorespiratory fitness after HIIT in normal-weight adolescents. The improvement may be related to the increase of activities of key enzymes involved in glycogenolysis and anaerobic glycolysis in skeletal muscle after interval training [40]. The HIIT was more effective in enhancing anaerobic indices in obese than non-obese subjects. These differences may be due to changes in body composition in the obese, as well as psychological factors. The training programme may favour the development of positive self-esteem and increase perceived physical value and feelings of competence (motivation, management of stress, confidence, etc.), which would be more evident in the obese [41].
The HIIT programme resulted in a significant improvement in plasma lipid profile in sedentary overweight/obese subjects, but not in active normal-weight subjects. While abundant data suggest a beneficial effect of continuous training on plasma lipids in overweight and obese subjects [8,9], data on the effect of HIIT on lipid profile in the obese are sparse and inconsistent. Koubaa et al. [8] observed an improvement in plasma lipids after 12-week interval training in obese children. Also, Racil et al. [18, 42] observed an improvement in plasma lipids, cardiometabolic variables, blood leptin concentration and ratings of perceived exertion after a 12-week HIIT programme in obese adolescent females. However, no significant changes in blood lipids were observed in young overweight/obese males after either two-week [43] or 12-week HIIT programmes [17, 19]. In normal-weight subjects, the effect of training on plasma lipids is also inconsistent. Some studies reported significant changes [14, 44] while others showed no significant effect [16, 32]. The discrepancies could be related to differences in subject characteristics (ethnicity, lipid-lowering drug use, smoking, alcohol consumption, diet, and previous physical activity), training intensity and duration, and the atmosphere characteristics (e.g. geography, climate, and season). In the present study, while the variations of TC, TG and LDL-C were only significant in the obese, their health benefit would be important even in non-obese subjects [32, 45].
The present study has some limitations. The relatively small number of participants in each group may have underpowered the study. Some external factors such as dietary intake and energy expenditure that may have affected the body composition and lipid profile were not controlled. Also, the study includes young male subjects, which makes the findings unsuitable for all individuals. Future investigations should focus on the effect of HIIT in other categories of individuals with different gender, age classes and phenotypes, and provide a particular awareness of the psychological impact of the training programme in the obese.
CONCLUSIONS
The eight-week HIIT programme improved aerobic and anaerobic performances, body composition and the plasma lipid profile in sedentary overweight/obese young men. These findings suggest that intermittent training may improve the cardio-metabolic profile in the obese. Further research is needed to confirm this assumption and to clarify the underlying mechanisms.
Acknowledgments
The authors are grateful to the participants for their valuable contribution to achieve the training program and for their kindness and courage
Conflict of interest
The authors declare that they have no conflict of interest.
REFERENCES
- 1.Speiser PW, Rudolf MC, Anhalt H, Camacho-Hubner C, Chiarelli F, Eliakim A, Freemark M, Gruters A, Hershkovitz E, Iughetti L, Krude H, Latzer Y, Lustig RH, Pescovitz OH, Pinhas-Hamiel O, Rogol AD, Shalitin S, Sultan C, Stein D, Vardi P, Werther GA, Zadik Z, Zuckerman-Levin N, Hochberg Z. Obesity Consensus Working Group Childhood obesity. J Clin Endocrinol Metab. 2005;90(3):1871–8187. doi: 10.1210/jc.2004-1389. [DOI] [PubMed] [Google Scholar]
- 2.Calle EE, Thun MJ, Petrelli JM, Rodriguez C, Heath CW., Jr Body-mass index and mortality in a prospective cohort of U.S. adults. N Engl J Med. 1999;341(15):1097–1105. doi: 10.1056/NEJM199910073411501. [DOI] [PubMed] [Google Scholar]
- 3.Poirier P, Després JP. Exercise in weight management of obesity. Cardiol Clin. 2001;19(3):459–470. doi: 10.1016/s0733-8651(05)70229-0. [DOI] [PubMed] [Google Scholar]
- 4.Gueugnon C, Mougin F, Simon-Rigaud ML, Nègre V, Touati S, Regnard J. Interest of individualized interval training in obese teenagers. Science & Sports. 2011;26(4):229–232. [Google Scholar]
- 5.Buchan DS, Ollis S, Thomas NE, Baker JS. Physical activity behaviour: an overview of current and emergent theoretical practices. J Obes. 2012;2012:546459. doi: 10.1155/2012/546459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Haskell WL, Lee IM, Pate RR, Powell KE, Blair SN, Franklin BA, Macera CA, Heath GW, Thompson PD, Bauman A. Physical activity and public health: updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Med Sci Sports Exerc. 2007;39(8):1423–1434. doi: 10.1249/mss.0b013e3180616b27. [DOI] [PubMed] [Google Scholar]
- 7.Ben Ounis O, Elloumi M, Ben Chiekh I, Zbidi A, Amri M, Lac G, Tabka Z. Effects of two-month physical-endurance and diet-restriction programs on lipid profiles and insulin resistance in obese adolescent boys. Diabetes Metab. 2008;34(6 Pt 1):595–600. doi: 10.1016/j.diabet.2008.05.011. [DOI] [PubMed] [Google Scholar]
- 8.Koubaa A, Trabelsi H, Masmoudi L, Elloumi M, Sahnoun Z, Zeghal KM, Hakim A. The effects of intermittent and continuous training on body composition, cardiorespiratory fitness and lipid profile in obese adolescents. Iosr Journal of Pharmacy. 2013;3(2):31–37. [Google Scholar]
- 9.Antunes Bde M, Monteiro PA, Silveira LS, Cayres SU, Silva CB, F IF Jr. Effect of concurrent training on risk factors and hepatic steatosis in obese adolescents. Rev Paul Pediatr. 2013;31(3):371–376. doi: 10.1590/S0103-05822013000300015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Buchan DS, Ollis S, Young JD, Cooper SM, Shield JP, Baker JS. High intensity interval running enhances measures of physical fitness but not metabolic measures of cardiovascular disease risk in healthy adolescents. BMC Public Health. 2013;13:498. doi: 10.1186/1471-2458-13-498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wong PL, Chaouachi A, Chamari K, Dellal A, Wisloff U. Effect of preseason concurrent muscular strength and high-intensity interval training in professional soccer players. J Strength Cond Res. 2010;24(3):653–660. doi: 10.1519/JSC.0b013e3181aa36a2. [DOI] [PubMed] [Google Scholar]
- 12.Buchan DS, Ollis S, Thomas NE, Buchanan N, Cooper SM, Malina RM, Baker JS. Physical activity interventions: effects of duration and intensity. Scand J Med Sci Sports. 2011;21(6):e341–e350. doi: 10.1111/j.1600-0838.2011.01303.x. [DOI] [PubMed] [Google Scholar]
- 13.Buchan DS, Ollis S, Thomas NE, Malina RM, Baker JS. School-based physical activity interventions: challenges and pitfalls. Child Care Health Dev. 2012;38(1):1–2. doi: 10.1111/j.1365-2214.2011.01258.x. [DOI] [PubMed] [Google Scholar]
- 14.Musa DI, Adeniran SA, Dikko AU, Sayers SP. The effect of a high-intensity interval training program on high-density lipoprotein cholesterol in young men. J Strength Cond Res. 2009;23(2):587–592. doi: 10.1519/JSC.0b013e318198fd28. [DOI] [PubMed] [Google Scholar]
- 15.Thomas TR, Adeniran SB, Iltis PW, Aquiar CA, Albers JJ. Effects of interval and continuous running on HDL-cholesterol, apoproteins A-1 and B, and LCAT. Can J Appl Sport Sci. 1985;10(1):52–59. [PubMed] [Google Scholar]
- 16.Nybo L, Sundstrup E, Jakobsen MD, Mohr M, Hornstrup T, Simonsen L, Bülow J, Randers MB, Nielsen JJ, Aagaard P, Krustrup P. High-intensity training versus traditional exercise interventions for promoting health. Med Sci Sports Exerc. 2010;42(10):1951–1958. doi: 10.1249/SS.0b013e3181d99203. [DOI] [PubMed] [Google Scholar]
- 17.Heydari M, Freund J, Boutcher SH. The effect of high-intensity intermittent exercise on body composition of overweight young males. J Obes. 2012;2012:480467. doi: 10.1155/2012/480467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Racil G, Ben Ounis O, Hammouda O, Kallel A, Zouhal H, Chamari K, Amri M. Effects of high vs. moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur J Appl Physiol. 2013;113(10):2531–2540. doi: 10.1007/s00421-013-2689-5. [DOI] [PubMed] [Google Scholar]
- 19.Corte de Araujo AC, Roschel H, Picanço AR, do Prado DM, Villares SM, de Sá Pinto AL, Gualano B. Similar health benefits of endurance and high-intensity interval training in obese children. PLoS One. 2012;7(8):e42747. doi: 10.1371/journal.pone.0042747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Durnin JV, Womersley J. Body fat assessed from total body density and its estimation from skinfold thickness: measurements on 481 men and women aged from 16 to 72 years. Br J Nutr. 1974;32(1):77–97. doi: 10.1079/bjn19740060. [DOI] [PubMed] [Google Scholar]
- 21.Siri WE. Body composition from fluid space and density. In: Brozek J, Hanschel A, editors. Techniques for measuring body composition. Washington, DC: National Academy of Science; 1961. pp. 223–244. [Google Scholar]
- 22.Cazorla G. Proceedings of the International Symposium of Guadeloupe. Edts: Actshng and Areaps. 1990. Field tests to evaluate aerobic capacity and maximal aerobic speed; pp. 151–173. (In French) [Google Scholar]
- 23.Bosco C, Luhtanen P, Komi PV. A simple method for measurement of mechanical power in jumping. Eur J Appl Physiol Occup Physiol. 1983;50(2):273–282. doi: 10.1007/BF00422166. [DOI] [PubMed] [Google Scholar]
- 24.Chamari K, Chaouachi A, Hambli M, Kaouech F, Wisløff U, Castagna C. The five-jump test for distance as a field test to assess lower limb explosive power in soccer players. J Strength Cond Res. 2008;22(3):944–950. doi: 10.1519/JSC.0b013e31816a57c6. [DOI] [PubMed] [Google Scholar]
- 25.Impellizzeri FM, Rampinini E, Castagna C, Martino F, Fiorini S, Wisloff U. Effect of plyometric training on sand versus grass on muscle soreness and jumping and sprinting ability in soccer players. Br J Sports Med. 2008;42(1):42–46. doi: 10.1136/bjsm.2007.038497. [DOI] [PubMed] [Google Scholar]
- 26.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502. [PubMed] [Google Scholar]
- 27.Cohen J. Statistical power analysis for the behavioural sciences. 2nd edn. Hillsdale, NJ: Erbaulm; 1988. [Google Scholar]
- 28.Drigny J, Gremeaux V, Dupuy O, Gayda M, Bherer L, Juneau M, Nigam A. Effect of interval training on cognitive functioning and cerebral oxygenation in obese patients: a pilot study. J Rehabil Med. 2014;46(10):1050–1054. doi: 10.2340/16501977-1905. [DOI] [PubMed] [Google Scholar]
- 29.Lau PW, Wong del P, Ngo JK, Liang Y, Kim CG, Kim HS. Effects of high-intensity intermittent running exercise in overweight children. Eur J Sport Sci. 2015;15(2):182–190. doi: 10.1080/17461391.2014.933880. [DOI] [PubMed] [Google Scholar]
- 30.Leggate M, Carter WG, Evans MJ, Vennard RA, Sribala-Sundaram S, Nimmo MA. Determination of inflammatory and prominent proteomic changes in plasma and adipose tissue after high-intensity intermittent training in overweight and obese males. J Appl Physiol. (1985). 2012;112(8):1353–1360. doi: 10.1152/japplphysiol.01080.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alkahtani SA, King NA, Hills AP, Byrne NM. Effect of interval training intensity on fat oxidation, blood lactate and the rate of perceived exertion in obese men. Springer plus. 2013;2:532. doi: 10.1186/2193-1801-2-532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ouerghi N, Khammassi M, Boukorraa S, Feki M, Kaabachi N, Bouassida A. Effects of a high-intensity intermittent training program on aerobic capacity and lipid profile in trained subjects. Open Access J Sports Med. 2014;5:243–248. doi: 10.2147/OAJSM.S68701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ben Abderrahman A, Zouhal H, Chamari K, Thevenet D, de Mullenheim PY, Gastinger S, Tabka Z, Prioux J. Effects of recovery mode (active vs. passive) on performance during a short high-intensity interval training program: a longitudinal study. Eur J Appl Physiol. 2013;113(6):1373–1383. doi: 10.1007/s00421-012-2556-9. [DOI] [PubMed] [Google Scholar]
- 34.Sawyer BJ, Tucker WJ, Bhammar DM, Ryder JR, Sweazea KL, Gaesser GA. Effects of high-intensity interval training and moderate-intensity continuous training on endothelial function and cardiometabolic risk markers in obese adults. J Appl Physiol. (1985). 2016;121(1):279–288. doi: 10.1152/japplphysiol.00024.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tjønna AE, Stølen TO, Bye A, Volden M, Slørdahl SA, Odegård R, Skogvoll E, Wisløff U. Aerobic interval training reduces cardiovascular risk factors more than a multitreatment approach in overweight adolescents. Clin Sci. (Lond). 2009;116(4):317–326. doi: 10.1042/CS20080249. [DOI] [PubMed] [Google Scholar]
- 36.Balsom PD, Ekblom B, Söderlund K, Sjödin B, Hultman E. Creatine supplementation and dynamic high-intensity intermittent exercise. Scand J Med Sci Sports. 1993;3:143–149. [Google Scholar]
- 37.Mazurek K, Zmijewski P, Krawczyk K, Czajkowska A, Kęska A, Kapuściński P, Mazurek T. High intensity interval exercise and moderate continuous cycle training in a physical education program improves health-related fitness in young females. Biol Sport. 2016;33(2):139–144. doi: 10.5604/20831862.1198626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ziemann E, Grzywacz T, Łuszczyk M, Laskowski R, Olek RA, Gibson AL. Aerobic and anaerobic changes with high-intensity interval training in active college-aged men. J Strength Cond Res. 2011;25(4):1104–1112. doi: 10.1519/JSC.0b013e3181d09ec9. [DOI] [PubMed] [Google Scholar]
- 39.Buchan DS, Ollis S, Thomas NE, Baker JS. The influence of a high intensity physical activity intervention on a selection of health related outcomes: an ecological approach. BMC Public Health. 2010;10:8. doi: 10.1186/1471-2458-10-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Glaister M. Multiple sprint work: physiological responses, mechanisms of fatigue and the influence of aerobic fitness. Sports Med. 2005;35(9):757–777. doi: 10.2165/00007256-200535090-00003. [DOI] [PubMed] [Google Scholar]
- 41.Famose JP, Guerin F, Sarrazin P. Les croyances sur soi: clarification conceptuelle, formation, et relations à la performance sportive. Paris: Editions Revue EPS; 2005. [Google Scholar]
- 42.Racil G, Coquart JB, Elmontassar W, Haddad M, Goebel R, Chaouachi A, Amri M, Chamari K. Greater effects of high- compared with moderate-intensity interval training on cardio-metabolic variables, blood leptin concentration and ratings of perceived exertion in obese adolescent females. Biol Sport. 2016;33(2):145–152. doi: 10.5604/20831862.1198633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Whyte LJ, Gill JM, Cathcart AJ. Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metabolism. 2010;59(10):1421–1428. doi: 10.1016/j.metabol.2010.01.002. [DOI] [PubMed] [Google Scholar]
- 44.Altena TS, Michaelson JL, Ball SD, Guilford BL, Thomas TR. Lipoprotein subfraction changes after continuous or intermittent exercise training. Med Sci Sports Exerc. 2006;38(2):367–372. doi: 10.1249/01.mss.0000185088.33669.fd. [DOI] [PubMed] [Google Scholar]
- 45.Pedersen TR, Olsson AG, Faergeman O, Kjekshus J, Wedel H, Berg K, Wilhelmsen L, Haghfelt T, Thorgeirsson G, Pyörälä K, Miettinen T, Christophersen B, Tobert JA, Musliner TA, Cook TJ. Lipoprotein changes and reduction in the incidence of major coronary heart disease events in the Scandinavian Simvastatin Survival Study (4S) Circulation. 1998;97(15):1453–1460. doi: 10.1161/01.cir.97.15.1453. [DOI] [PubMed] [Google Scholar]