Abstract
Chronic inflammatory demyelinating polyneuropathy (CIDP) is a debilitating condition caused by autoimmune demyelination of peripheral nerves. CIDP is associated with increased IL-10, a cytokine with well-described anti-inflammatory effects. However, the role of IL-10 in CIDP is unclear. In this study, we demonstrate that IL-10 paradoxically exacerbates autoimmunity against peripheral nerves. In IL-10-deficient mice, protection from neuropathy was associated with an accrual of highly activated CD4+ T cells in draining lymph nodes and absence of infiltrating immune cells in peripheral nerves. Accumulated CD4+ T cells in draining lymph nodes of IL-10-deficient mice expressed lower sphingosine-1-phostphate receptor 1 (S1pr1), a protein important in lymphocyte egress. Additionally, IL-10 stimulation in vitro induced S1pr1 expression in lymph node cells in a STAT3-dependent manner. Together, these results delineate a novel mechanism in which IL-10-induced STAT3 increases S1pr1 expression and CD4+ T cell migration to accelerate T cell-mediated destruction of peripheral nerves.
Introduction
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP) is the most common acquired autoimmune peripheral neuropathy, affecting as many as 9 in 100,000 people (1). Sometimes considered the chronic form of Guillain-Barré Syndrome, CIDP is characterized by relapsing-remitting or progressive sensory dysfunction, paresthesia, and weakness due to autoimmune demyelination of nerves in the peripheral nervous system (PNS) (2). First-line treatment options include glucocorticoids, intravenous immunoglobulin (IVIg), and plasma exchange, which utilize nonspecific mechanisms of action against CIDP, do not improve disease symptoms in one-third of patients, and do not achieve remission or cure in over 70% of patients (3, 4). Thus, current treatments fail to address considerable CIDP disease burden. Better understanding of CIDP pathogenesis is an important step toward developing new mechanism-based therapies with greater efficacy.
Substantial evidence suggests T cells are critical for the development of CIDP (1). First, non-obese diabetic (NOD) mice develop spontaneous autoimmune peripheral polyneuropathy (SAPP) that resembles CIDP due to defective negative selection of T cells (Meyer zu Horste et al., 2014; Su et al., 2012). Second, T cell-deficient mice fail to develop experimental autoimmune neuritis (EAN), which is an induced model of CIDP (7). Third, CD4+ T cells are sufficient to transfer SAPP (Meyer zu Horste et al., 2014; Salomon et al., 2001; Su et al., 2012). Finally, T cells are present in sural nerve infiltrates of CIDP patients (1).
A key function of T cells is the secretion of cytokines (1, 9), and CIDP has been associated with increased interferon gamma (IFN-γ) (10, 11), and tumor necrosis factor alpha (TNF-α) (12). Blockade of IFN-γ and TNF-α in SAPP mouse models have revealed disease-promoting roles for these cytokines (Bour-Jordan et al., 2005; Zeng et al., 2013; Zhang et al., 2012). Interestingly, increased interleukin 10 (IL-10) expression has also been associated with CIDP (10) (16). However, the role of IL-10 in CIDP pathogenesis is unclear.
IL-10 has multiple anti-inflammatory effects, which include suppressing production of pro-inflammatory cytokines, chemokines, and costimulatory molecules in macrophages and dendritic cells (17, 18). IL-10 dampens autoimmunity in multiple disease models, including rheumatoid arthritis (RA) (19, 20) and multiple sclerosis (MS) (21, 22). Although IL-10 typically suppresses inflammation, it has also been reported to promote immune responses. For example, IL-10 secretion by T helper 2 (Th2) cells promotes differentiation and antibody secretion in B cells (23, 24), which is strongly associated with inflammation in systemic lupus erythematosus (SLE) (25) and allergy (26). Furthermore, IL-10 stimulates the expansion and differentiation of effector CD8+ T cells, which boosts anti-tumor immunity in mice (Zhang et al., 2016). Thus, while IL-10 is most commonly regarded as an anti-inflammatory cytokine, it is a pleiotropic cytokine that can also promote inflammation in certain immune contexts.
IL-10 binding to IL-10 receptor 1 leads to phosphorylation of signal transducer and activator of transcription 3 (STAT3) (18). Along with increased IL-10, CD4+ T cells from CIDP patients with active disease have higher pSTAT3 (10). Separately, pSTAT3 has been shown to induce S1PR1 expression (28). S1PR1 is essential for lymphocyte egress from lymph nodes (29), and has been implicated in SAPP pathogenesis (30). The concurrent roles of pSTAT3 in IL-10 signaling and S1PR1 transcriptional induction suggest that IL-10 may contribute to S1PR1 expression and S1PR1-dependent lymphocyte migration. However, multiple cytokines can activate STAT3, and it is unclear whether IL-10-induced pSTAT3 upregulates S1PR1 expression.
In this study, we found that, like CIDP patients, mice with SAPP have robust induction of IL-10. IL-10 expression was increased in inflamed peripheral nerves and T cells isolated from spleens and lymph nodes. Unexpectedly, IL-10-deficient mice were protected from neuropathy development, suggesting IL-10 is a pathogenic cytokine in SAPP. IL-10 deficiency was associated with accumulation of activated CD4+ T cells in the draining lymph nodes and decreased PNS infiltration. These findings suggested that impaired T cell egress may underlie SAPP protection in IL-10 deficiency. Indeed, IL-10-deficient CD4+ T cells lacked S1pr1 expression and displayed reduced migration to the S1PR1 ligand sphingosine-1-phosphate (S1P). Moreover, IL-10 induced S1PR1 mRNA expression, and IL-10-mediated S1pr1 expression required STAT3. Together, these findings illustrate a previously unappreciated role of IL-10 in promoting S1PR1-dependent lymphocyte migration and autoimmune destruction of peripheral nerves.
Materials and Methods
Mice
NOD.AireGW/+ mice were generated as previously described (Su et al., 2008). NOD.Scid (JAX stock #001303), NOD.μMT−/− (JAX stock #004639), and NOD.Il10−/− (JAX stock #004266) mice were purchased from The Jackson Laboratory. Mice were housed in a specific-pathogen-free (SPF) barrier facility at the University of North Carolina, Chapel Hill. Clinical neuropathy, determined by hind limb weakness, and diabetes, determined by presence of glucose in urine, were assessed at least once per week as described previously (32). Diabetic mice were treated daily with intraperitoneal insulin until used in experiments or euthanasia (due to >20% weight loss). Only female mice were utilized due to higher SAPP incidence relative to males (Su et al., 2012). Mice were utilized at 22 weeks of age unless otherwise noted. Due to the rapid progression of disease and death following initial symptoms, mice that developed SAPP prior to 22 weeks of age were harvested at SAPP onset. NOD.AireGW/+ Il10+/− or NOD.AireGW/+ Il10−/− mice were used as splenocyte donors. Experiments complied with the Animal Welfare Act and the National Institutes of Health guidelines for the ethical care and use of animals in biomedical research.
Immunohistochemistry
Mouse sciatic nerves were embedded in Optimal Cutting Temperature (OCT) compound, frozen at −20°C for 2 hours, and then stored at −80°C. Six micron-thick longitudinal sections were fixed with cold acetone, blocked with 2.5% goat serum, and then stained with anti-IL-10 antibody (Biolegend clone JES5-16E3) for 2 hours at room temperature. After washing with PBS + 0.1% Tween, sections were incubated with anti-rat-HRP antibody for 30 minutes at room temperature. DAB solution (Vector Laboratories) was applied to the sections as the chromogen. Brightness of all images was increased by 10 in Adobe photoshop.
Flow cytometry
Flow cytometry was performed as previously described (33). Briefly, single-cell suspensions were isolated from spleens or lymph nodes by crushing with forceps, or from sciatic nerves by mincing and digestion in 2 mg/ml collagenase. Cells were stained with live/dead fixable yellow dye (Life Technologies), anti-mouse CD4 (Biolegend clone GK1.5), anti-mouse CD44 (eBioscience clone IM7), anti-mouse CD62L (eBioscience clone MEL-14), anti-mouse S1PR1 (R&D clone 713412), anti-mouse IFN-γ (eBioscience clone XMG1.2), and anti-mouse IL-10 (BD clone JES5-16E3) antibodies. For intracellular cytokine staining, cells were stimulated with PMA/ionomycin for 4 hours at 37°C 5% CO2, and permeabilized using BD Cytofix/Cytoperm according to manufacturer’s instructions. Cells were analyzed on a CyAn ADP Analyzer (Beckman Coulter). Data were analyzed using FlowJo X.
Histology
Harvested organs were fixed in 10% buffered formalin for at least 96 hours, washed in 30% ethanol for 20 minutes, and then stored in 70% ethanol. Organs were embedded in paraffin, sectioned, and stained with H&E by the UNC Animal Histopathology Core. Immune infiltration was scored while blinded to genotype as previously described (Su et al., 2008). For sciatic nerves, scores of 0, 1, 2, 3, and 4 indicate 0%, 1–25%, 26–50%, 51–75%, and >75% infiltration, respectively. Colon histology was scored in a blinded fashion by a board-certified veterinary pathologist as previously described (34). Briefly, H&E-stained distal colonic sections were scored based on an additive histological injury system that included mucosal ulceration, epithelial hyperplasia, and lamina propria mononuclear and neutrophil infiltrates. Pancreatic islet infiltration was scored in a blinded fashion as previously described (35). Briefly, H&E-stained pancreatic sections were scored based on average degree of islet infiltration. Brightness of all images was increased by 50 in Adobe photoshop.
Electrophysiology
Sciatic nerve conduction studies were performed as described (36), using a Teca Synergy T2X EMG system.
Antibody treatment
Anti–B7-1 Ab (clone 16.10A1) and anti–B7-2 Ab (clone GL1) were generous gifts of Greg Szot (UCSF) or purchased from BioXCell. To induce neuropathy, 14-day-old NOD.Il10+/− or NOD.Il10−/− mice were treated with 50 μg Ab or isotype controls (2A3 and Armenian Hamster IgG) every other day for seven treatments (Salomon et al., 2001; Zeng et al., 2013).
Enzyme-linked Immunospot Assay (ELISpot)
ELISpot assays were performed according to manufacturer’s protocol (BD). One million splenocytes or 8 × 105 lymph node cells were cultured alone, with synthetic ovalbumin peptide (323–339) (Invivogen), synthetic myelin protein zero peptide (180–199) (Genemed Synthesis), or PMA/ionomycin in DMEM + 10% FBS for 17 hours at 37°C 5% CO2. Spots were enumerated using the AID iSpot Reader.
Adoptive Transfer
Adoptive transfer of CD4+ T cells from spleen was performed as previously described (Su et al., 2012). Tissue culture plates were coated with 1 μg/mL anti-CD3ε (eBioscience clone 145-2C11) and 1 μg/mL anti-CD28 (BD Pharmingen clone 37.51) in sterile PBS overnight at 4°C. Plates were washed gently with sterile PBS. Whole spleen from 22-wk-old NOD.AireGW/+ Il10+/− or NOD.AireGW/+ Il10−/− donors were cultured with plate-bound anti-mouse CD3 and anti-mouse CD28 in DMEM (Gibco) + 10% FBS (Sigma-Aldrich) for 4 days at 37°C 5% CO2. CD4+ T cells were purified from cultures using the Magnisort CD4+ T cell Enrichment Kit (Invitrogen) and 1 × 106 CD4+ T cells were transferred to NOD.Scid Il10+/− or NOD.Scid Il10−/− recipients. Diabetic mice were excluded as donors.
FTY720 Treatment
AireGW/+ mice were treated with DMSO or 1 mg/kg FTY720 (Cayman Chemical). FTY720 was first dissolved in DMSO (25g in 1 mL DMSO), aliquoted, and frozen at −20°C. Mice were given intraperitoneal injections of DMSO or FTY720 dissolved 1:100 in 100 μL sterile water on every week day (5 days per week) from 14 weeks of age (before SAPP development) until utilization. Mice were harvested when they developed SAPP, or when all DMSO-treated controls developed SAPP.
Chemotaxis Assay
Chemotaxis was measured using transwell assays as described by (37). Migration of lymph node cells was measured using a 24-well transwell plate (Corning Life Sciences) with 6.5 mm polycarbonate filters and five μm pores. The lower chamber was coated overnight at 4°C with 600 μl of 100 μg/ml human collagen type IV (Sigma-Aldrich) in 0.5 M acetic acid, washed with PBS, and air-dried. Two million cells were resuspended in 100 μl RPMI 1640 medium with 0.1% fatty acid-free BSA (Sigma-Aldrich), 100 U/ml penicillin G, 2mM L-glutamine, and 25 mM HEPES buffer. Cells were placed on the Transwell inserts. S1P (20 nM in 600 μL) was added in the same medium to the lower chamber. Migration was performed for four hours at 37°C 5% CO2. Migrated cells were counted with a hemocytometer. Migration assays without S1P were performed in parallel to assess baseline migration. Net migration to S1P was calculated by subtracting the number of cells that migrated nonspecifically from the number of cells that migrated to S1P.
IL-10 Stimulation and Real Time RT-PCR
Five hundred thousand cells from fresh sciatic and lumbar lymph nodes of NOD.Il10−/− mice were stimulated with recombinant human IL-10 (100 ng/mL from Peprotech), stimulated with IL-10 and STA-21 (10 μM from Santa Cruz Biotech), or incubated without stimulation in XVIVO 15 + Transferrin serum-free media (Lonza) for 30 minutes at 37°C 5% CO2. RNA was isolated from cells using the Zymo RNA MicroPrep kit. Superscript II (Invitrogen) reverse transcriptase was used to create cDNA. TaqMan universal PCR Master Mix (Applied Biosystems) was used for qPCR. Commercially available TaqMan primer-probe sets for IL-10 and S1Pr1 were used (Applied Biosystems). Cyclophilin A was used as an internal control and detected with the primer-probe set reported by (Su et al., 2008). Reactions were run on a Quantstudio 6 Flex system (Life Technologies) and analyzed as described (Su et al., 2008).
Statistics
Data were analyzed with Graphpad Prism 6 using one-sample two-tailed Student’s t tests, unpaired two-tailed Student’s t tests or one-way ANOVA. The Bonferroni or Tukey corrections for multiple comparisons were used when appropriate. Mantel-Cox log-rank tests were used to compare survival curves. R (v3.3.1) was used to perform Fisher’s Exact test and false discovery rate (FDR) adjustments. p<0.05 was considered significant, unless the threshold was reduced for the Bonferroni correction.
Results
IL-10 promotes SAPP in two mouse models
IL-10 is increased in CIDP patients (10), but the functional significance of IL-10 in CIDP is unknown. To study this, we used a strain of NOD mice that develops SAPP due to a dominant-negative G228W mutation in the Autoimmune Regulator gene (NOD.AireGW/+; henceforth referred to as AireGW/+) (Su et al., 2012). SAPP in AireGW/+ mice shares key features with CIDP, including infiltration of peripheral nerves by CD4+ T cells and F4/80+ macrophages (38–40), interferon gamma (IFN-γ) production by CD4+ T cells (10, 11), and demyelination of peripheral nerves (Dalakas, 2015; Su et al., 2012). Whether increased IL-10 expression is associated with SAPP in this model, however, is not known. IL-10 mRNA expression was compared by qPCR in whole sciatic nerve from NOD wild-type (WT) vs. AireGW/+ mice with SAPP (Fig. 1A). IL-10 mRNA was not detected in WT nerves or young (< 10 wks old) AireGW/+ nerves, whereas neuropathic (> 15 wks old) AireGW/+ nerves expressed abundant IL-10. Additionally, immunohistochemical staining for IL-10 protein revealed increased protein expression in neuropathic AireGW/+ nerves compared to WT (Fig. 1B). Thus, similar to CIDP, SAPP in AireGW/+ mice is associated with increased IL-10 expression.
Figure 1. IL-10 deficiency is protective in SAPP.
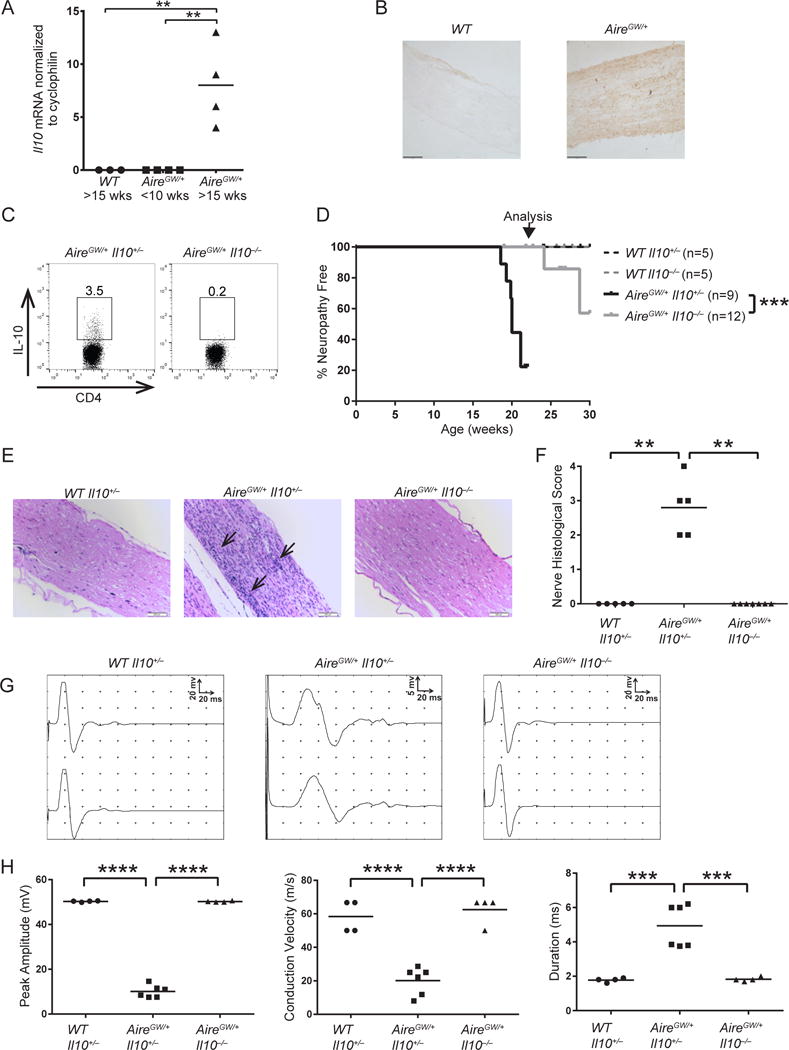
Relative IL-10 mRNA expression (A) and immunohistochemical stains of IL-10 (B) (magnification 200×) in sciatic nerves of NOD.WT (WT) mice >15 weeks old and NOD.AireGW/+ (AireGW/+) mice <10 weeks old or > 15 weeks old. Scale bar = 100 μm. p-value was calculated using two-tailed unpaired Student’s t test with Welch’s correction; *, p < 0.05. (C) Representative flow cytometry plots of splenic CD4+ T cells from IL-10-sufficient (IL-10+/−) and IL-10-deficient (IL-10−/−) AireGW/+ mice stained for intracellular IL-10. Gated on lymphocytes, singlets, and live cells. Numbers on plots represent frequencies of IL-10-expressing cells. WT Il10+/−, WT Il10−/−, AireGW/+ Il10+/−, and AireGW/+ Il10−/− mice were bred and monitored for SAPP. (D) SAPP incidence curves. Arrow points to age at which nerves were harvested for histology and EMG. p-value was calculated using Mantel-Cox log-rank test of AireGW/+ Il10+/− versus AireGW/+ Il10−/− mice; ***, p < 0.001. (E) H&E-stained sciatic nerves from 22-wk-old mice (magnification 200×). Arrow points to areas of dense immune cell infiltration. Scale bar = 200μm. (F) Cumulative sciatic nerve infiltration scores. p-values were calculated using Fisher’s Exact test with Bonferroni’s correction; **, p < 0.01. (G) Representative proximal and distal compound muscle action potentials from sciatic nerves of 22-wk-old mice. Note that the y-axis (amplitude) scale is 20 mV in WT Il10+/− and AireGW/+ Il10−/− mice, but is reduced to 5 mV in AireGW/+ Il10+/− mice. (H) Cumulative peak amplitude, conduction velocity, and duration of compound muscle action potentials from 22-wk-old mice. p-values were calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; ***, p < 0.001, ****p < 0.0001. Each symbol represents an individual mouse.
To determine IL-10’s role in SAPP pathogenesis, we crossed NOD.Il10−/− mice (Serreze et al., 2001) with NOD.AireGW/+ mice to generate NOD.AireGW/+ Il10+/− and NOD.AireGW/+ Il10−/− mice. Lack of IL-10 protein expression in NOD.AireGW/+ Il10−/− mice was confirmed by intracellular IL-10 staining and flow cytometry of CD4+ T cells (Fig. 1C). On the C3H/HeJBir and 129/SvEv backgrounds, IL-10 deficient mice are susceptible to colitis, growth retardation, anemia, and early death (34, 42, 43). Such findings could potentially prevent our ability to observe SAPP in NOD.AireGW/+ mice. However, we did not observe colitis or its associated symptoms in NOD.WT Il10−/− or NOD.AireGW/+ Il10−/− mice (Supp. Fig. 1 and data not shown). These results are consistent with prior reports that colitis is mild and transient on the NOD background (44), and suggest that NOD.AireGW/+ Il10−/− mice can be used to understand the impact of IL-10 on SAPP development.
Given their utility, NOD.AireGW/+ Il10+/− mice and NOD.AireGW/+ Il10−/− littermates were monitored for SAPP for 30 weeks. Consistent with our previous data, 80% of the IL-10-sufficient AireGW/+ mice developed SAPP by 22 weeks of age (AireGW/+ Il10+/−, Fig. 1D) (Su et al., 2012). Strikingly, compared to IL-10-sufficient AireGW/+ mice, IL-10-deficient AireGW/+ mice had a significant delay in SAPP (AireGW/+ Il10−/−, Fig. 1D), which suggests that IL-10 accelerates SAPP development. None of the IL-10-sufficient or deficient Aire WT mice developed SAPP during the study (WT Il10+/− or Il10−/−, Fig. 1D), indicating that IL-10 deficiency does not impair nerve function. Additionally, extensive cellular immune infiltrate was seen in H&E-stained sciatic nerves of IL-10-sufficient AireGW/+ mice, but was absent in IL-10-deficient AireGW/+ mice (Fig. 1E, 1F). Finally, SAPP in IL-10-sufficient AireGW/+ sciatic nerves was associated with electrophysiological changes consistent with demyelination, including prolonged distal motor latencies, slowed conduction velocity, and increased compound muscle action potential (CMAP) duration due to temporal dispersion relative to WT nerves (Fig. 1G, 1H). Conversely, abnormalities were not detected in sciatic nerves of IL-10-deficient AireGW/+ mice (Fig. 1G, 1H). IL-10-deficiency therefore protects against SAPP development, nerve infiltration, and electrophysiological changes in AireGW/+ mice. These findings that suggest that IL-10 promotes SAPP were surprising, given IL-10’s well-described functions as an immunoregulatory cytokine in a number of autoimmune diseases (17).
SAPP has been described in multiple mouse models, including NOD mice deficient in costimulatory molecules B7-1/2 (32). To determine whether the effect of IL-10 deficiency on SAPP was specific only to Aire-deficient mice or generalizable to other SAPP models, we treated IL-10-sufficient and IL-10-deficient WT mice with anti-mouse B7-1 and anti-mouse B7-2 to induce autoimmune peripheral neuropathy (Salomon et al., 2001; Zeng et al., 2013). By approximately 12 weeks of age, 100% of the anti-B7-1/2 treated, IL-10-sufficient WT mice had SAPP compared to only 22% of IL-10-deficient WT mice (Fig. 2A). At 12 weeks of age, sciatic nerves of IL-10-sufficient WT mice were heavily infiltrated, whereas sciatic nerves of IL-10-deficient WT mice had significantly less infiltration (Fig. 2B, 2C). These data suggest IL-10’s promotion of autoimmunity against peripheral nerves is not specific to the Aire-deficient model, but is a more generalizable feature of SAPP pathogenesis.
Figure 2. IL-10 deficiency is protective in anti-B71/anti-B7-2-induced autoimmune peripheral polyneuropathy.
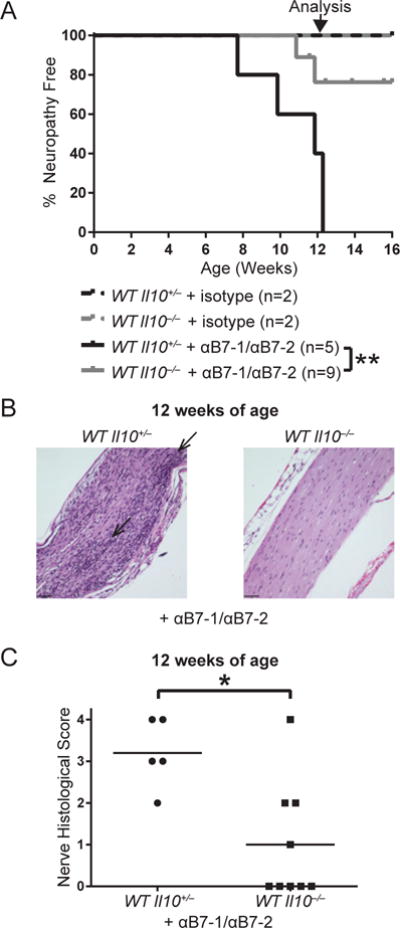
Anti-B7-1/anti-B7-2 or isotype treated WT or WT Il10−/− mice were monitored for SAPP. (A) SAPP incidence curves. p-value was calculated using Mantel-Cox log-rank test of anti-B7-1/anti-B7-2-treated WT Il10+/− versus WT Il10−/− mice; **, p < 0.01. Arrow points to age at which nerves were harvested for histology in B and C. (B) H&E-stained sciatic nerves from 12-week-old mice (magnification 200×). Arrows point to areas of dense immune cell infiltration. Scale bar = 60 μm. (C) Cumulative sciatic nerve infiltration scores from 12-week-old mice. p-value was calculated using Fisher’s Exact test; *, p < 0.05. Each symbol represents an individual mouse.
It is unclear whether IL-10’s effect in promoting SAPP reflects a more global, generalized role in promoting autoimmunity against all target tissues. In addition to SAPP, AireGW/+ mice also develop autoimmune diabetes (Su et al., 2012), allowing us to concurrently assess the role of IL-10 in SAPP and diabetes in these mice. Although IL-10 deficiency delayed SAPP development, lack of IL-10 did not affect autoimmune diabetes development in AireGW/+ mice (Supp. Fig. 2). This finding is consistent with previous reports that IL-10 does not alter diabetes incidence in NOD.WT mice (Serreze et al., 2001). Thus, IL-10 appears to promote autoimmunity in a tissue-specific manner.
IL-10 produced by CD4+ T cells promotes SAPP development
Many cell types in the innate and adaptive immune systems produce IL-10 (18). Therefore, it is not clear which IL-10-producing cell type is important in promoting SAPP. A key source of IL-10 is CD4+ T cells, and CD4+ T cells are sufficient to transfer SAPP in multiple models (Bour-Jordan et al., 2005; Meyer zu Horste et al., 2014; Su et al., 2012). Given the critical role for CD4+ T cells in SAPP, we examined IL-10 expression in CD4+ T cells from WT and neuropathic AireGW/+ mice. IL-10 expression in CD4+ T cells from the spleen and nerve-draining lymph node of neuropathic AireGW/+ mice was significantly increased compared to WT mice (Fig. 3A, 3B). Furthermore, while CD4+ T cells were absent in WT sciatic nerve, >10% of infiltrating CD4+ T cells expressed IL-10 in AireGW/+ sciatic nerves. Thus, increased IL-10 expression within CD4+ T cells is associated with SAPP development.
Figure 3. IL-10 produced by CD4+ T cells promotes SAPP.
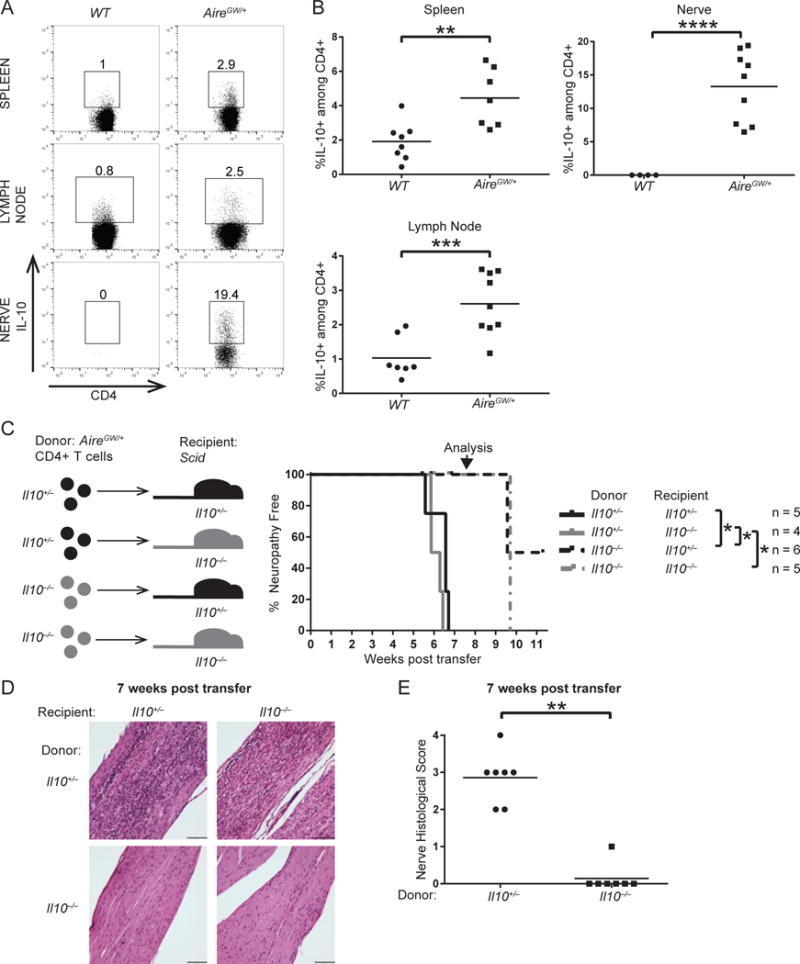
Spleens, lumbar lymph nodes, and sciatic nerves of 22-wk-old WT mice and AireGW/+ mice stained for intracellular IL-10. (A) Flow cytometry plots of CD4+ T cells. Gated on lymphocytes, singlets, and live cells. Numbers on plots represent frequencies of IL-10-expressing cells. (B) Cumulative frequencies of CD4+IL-10+ T cells. p-values were calculated using two-tailed unpaired Student’s t test with Welch’s correction; **, p < 0.01, ***, p < 0.001, ****, p < 0.0001. (C) Schematic (left) and SAPP incidence curve (right) of NOD.Scid Il10+/− or NOD.Scid Il10−/− recipient mice receiving purified CD4+ T cells adoptively transferred from 22-wk-old AireGW/+ Il10+/− or AireGW/+ Il10−/− donor mice. p-values were calculated using Mantel-Cox log-rank test with false discovery rate adjustment for multiple comparisons; *, p < 0.05. Arrow points to time when nerves were harvested for histology. (D) H&E-stained sciatic nerves harvested 7 weeks post CD4+ T cell adoptive transfer from the groups described in C. (magnification 200×). Scale bar = 98 μm. (E) Cumulative infiltration scores of sciatic nerves from recipient mice 7 weeks post transfer. Grouped by donor genotype. p-value was calculated using Fisher’s Exact test; **, p < 0.01.
Given increased CD4+ T cell IL-10 expression with SAPP, we next sought to determine whether IL-10 production by CD4+ T cells is sufficient to promote SAPP. Purified AireGW/+ CD4+ T cells that were IL-10 sufficient or deficient were transferred to immunodeficient NOD.PrkdcScid/Scid (Scid) (45) recipients that were either IL-10 sufficient or deficient (outlined in Fig. 3C, left). IL-10 sufficient donor CD4+ T cells induced SAPP in IL-10-sufficient and deficient recipients 6–7 weeks post-transfer (Fig. 3C, right). Conversely, IL-10-deficient donor CD4+ T cells induced SAPP in IL-10-sufficient and deficient recipients 10 weeks post transfer, which was a significant delay relative to IL-10-sufficient donors (Fig. 3C, right). At 7 weeks post-transfer, recipients of IL-10-deficient donor CD4+ T cells displayed significantly less sciatic nerve infiltration than recipients of IL-10-sufficient donor CD4+ T cells (Fig. 3D, 3E). These data demonstrate that IL-10 production by CD4+ T cells promotes SAPP development.
IL-10 does not promote SAPP through its effects on B cells
IL-10 production by CD4+ T helper 2 (Th2) cells can promote inflammation by enhancing B cell proliferation, differentiation, and antibody production (Go, 1990; Moore et al., 2001; Rousset et al., 1992). We therefore sought to test whether IL-10 promotion of SAPP development requires B cells. IL-10 sufficient AireGW/+ mice were crossed with NOD.μMT−/− mice that lack mature B cells (Serreze et al., 1996), to generate AireGW/+ μMT+/− (B cell-sufficient) and AireGW/+ μMT−/− (B cell-deficient) progeny. Interestingly, SAPP occurred with the same incidence in B cell-sufficient and deficient AireGW/+ mice (Fig. 4A), suggesting that IL-10 does not promote SAPP through its effects on B cells. Furthermore, sciatic nerves from B cell-sufficient and deficient mice exhibited similar nerve conduction changes (Fig. 4B, 4C). Since B cells do not accelerate SAPP development, it is unlikely that B cell enhancement is the mechanism by which IL-10 promotes SAPP.
Figure 4. B cell deficiency does not protect from SAPP.
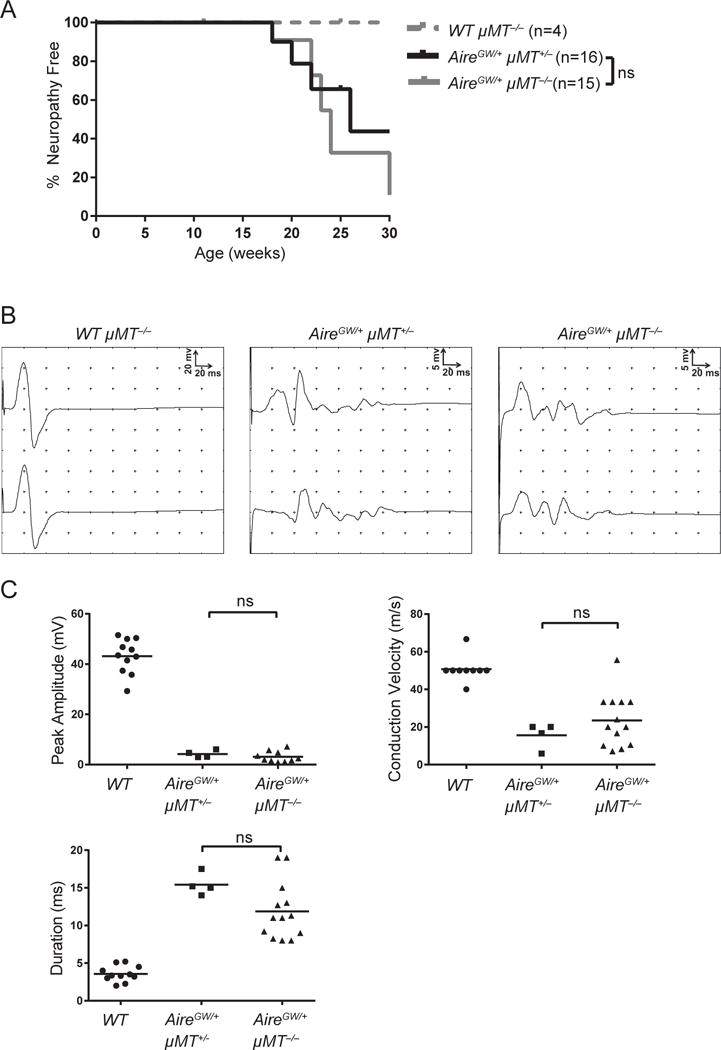
WT μMT−/−, AireGW/+ μMT+/−, and AireGW/+ μMT−/− mice were monitored for SAPP. (A) SAPP incidence curve. p-value was calculated using Mantel-Cox log-rank test of AireGW/+ μMT+/− versus AireGW/+ μMT−/− mice; ns indicates no significant difference observed. (B) Representative proximal and distal compound muscle action potentials from sciatic nerves of 22-wk-old mice. (C) Cumulative peak amplitude, conduction velocity, and duration of compound muscle action potentials of 22-wk-old mice. p-value was calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; ns indicates no significant difference observed. Each symbol represents an individual mouse.
IL-10-deficient AireGW/+ mice harbor highly activated CD4+ T cells
IL-10 has a well-described role in suppressing T cell activation (Moore et al., 2001). Since IL-10 unexpectedly promoted SAPP (Fig. 1 and 2), whether IL-10 would suppress or enhance T cell activation was not clear. Thus, we characterized CD4+ T cell activation in IL-10-deficient AireGW/+ mice that were protected from SAPP. Frequencies of CD4+CD44hiCD62Llo activated T cells were compared in the spleens of WT, IL-10-sufficient AireGW/+, and IL-10-deficient AireGW/+ mice. IL-10-deficient AireGW/+ mice had significantly higher frequencies of activated CD4+ T cells relative to IL-10-sufficient WT mice and AireGW/+ mice (Fig. 5A, 5B). We next tested whether IL-10 would suppress or enhance CD4+ T cell expression of IFN-γ, a cytokine critical for SAPP development (Bour-Jordan et al., 2005; Zeng et al., 2013). While IL-10-deficient AireGW/+ mice had significantly greater frequencies and absolute numbers of IFN-γ-producing CD4+ T cells in the spleen compared to WT mice, numbers of IFN-γ-producing CD4+ T cells were not different compared to IL-10-sufficient AireGW/+ mice (Fig. 5C, D). In the draining lymph node, IL-10-deficient AireGW/+ mice had greater absolute numbers of IFN-γ-producing CD4+ T cells compared to both WT and IL-10-sufficient AireGW/+ mice (Fig. 5E, F). Thus, despite protecting from SAPP, IL-10 deficiency was associated with equivalent or increased CD4+ T cell activation and IFN-γ production.
Figure 5. CD4+ T cells in IL-10-deficient AireGW/+ mice are highly activated.
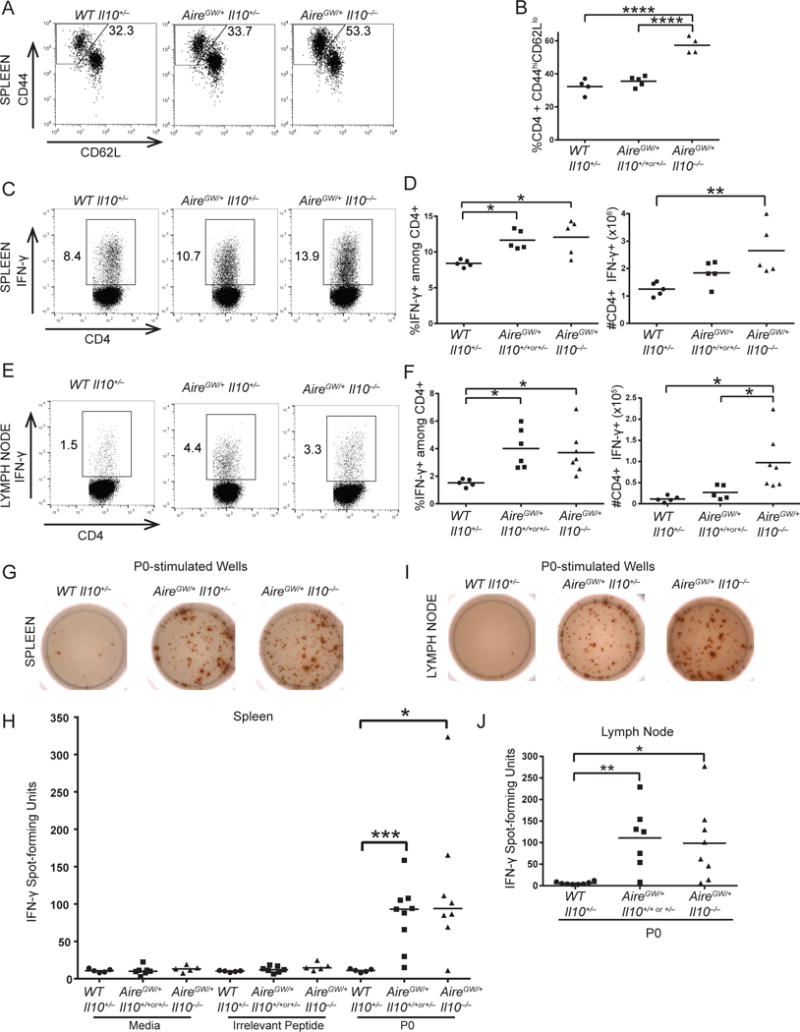
CD4+ T cells from spleens and nerve-draining lymph nodes of 22-wk-old WT, AireGW/+ Il10+/+ or +/−, and AireGW/+ Il10−/− mice were analyzed. (A) Flow cytometry plots of splenic CD4+ T cells stained for CD44 and CD62L. Gated on lymphocytes, singlets, live, CD4+ cells. Numbers on plots represent frequencies of CD44hiCD62Llo cells. (B) Cumulative frequencies of CD4+CD44hiCD62Llo activated T cells from spleens. p-values were calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; ****, p < 0.0001. (C) Flow cytometry plots of splenic CD4+ T cells stained for intracellular IFN-γ. Gated on lymphocytes, singlets, and live cells. Numbers on plots represent frequencies of IFN-γ-expressing cells. (D) Cumulative frequencies and numbers of CD4+IFN-γ+ T cells from spleens. p-values were calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; *, p < 0.05, **, p < 0.01. (E) Flow cytometry plots of lymph node CD4+ T cells stained for intracellular IFN-γ. Gated on lymphocytes, singlets, and live cells. Numbers on plots represent frequencies of IFN-γ-expressing cells. (F) Cumulative frequencies and numbers of CD4+IFN-γ+ T cells from lymph nodes. p-values were calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; *, p < 0.05. (G) ELISpot wells detecting IFN-γ from P0-stimulated splenocytes. (H) Cumulative numbers of IFN-γ spots from media only, ovalbumin-stimulated, or P0-stimulated splenocytes. Horizontal bars represent medians. p-values were calculated using two-tailed, unpaired Student’s t test with Welch’s correction and Bonferroni’s correction; *, p < 0.025, *** p < 0.001. (I) ELISpot wells detecting IFN-γ from P0-stimulated lymph node cells. (J) Cumulative numbers of IFN-γ spots from P0-stimulated lymph node cells. p-values were calculated using two-tailed, unpaired Student’s t test with Welch’s correction and Bonferroni’s correction; *, p < 0.025, ** p < 0.01. Each symbol represents an individual mouse.
In addition to suppressing T cell activation, IL-10 also downregulates expansion of self-antigen specific T cells (47). Since IL-10 unexpectedly promoted SAPP, however, we sought to determine how IL-10 deficiency would alter the precursor frequency of PNS-specific T cells. Pathogenic CD4+ T cells target the self-antigen myelin protein zero (P0) (Su et al., 2012). For this reason, we assessed the expansion of P0-specific CD4+ T cells using an ELISpot detecting IFN-γ. One million splenocytes from WT, IL-10-sufficient AireGW/+, and IL-10-deficient AireGW/+ mice were left unstimulated (Media), stimulated with MHC II-restricted ovalbumin irrelevant peptide, stimulated with MHC II-restricted P0 peptide, or stimulated with PMA/ionomycin for a positive control (data not shown). IL-10-deficient AireGW/+ splenocytes stimulated with P0 elicited a median of 94 IFN-γ spots, which was significantly higher than WT (11 spots) and similar to IL-10-sufficient AireGW/+ mice (93 spots) (Fig. 5G, 5H). We also tested cells from the nerve-draining lymph nodes for response to P0. Similar to spleen, the number of P0-specific T cells was significantly higher in IL-10-deficient AireGW/+ lymph nodes (99 spots) relative to WT (6 spots) and similar to IL-10-sufficient AireGW/+ mice (111 spots) (Fig. 5I, 5J). Thus, IL-10-deficiency protected from the development of SAPP, but does not decrease the frequency of nerve-specific CD4+ T cells in spleen or draining lymph nodes.
IL-10 deficiency is associated with enlarged lumbar lymph nodes, impaired lymphocyte migration, and reduced S1pr1 expression in CD4+ T cells
IL-10 deficiency was associated with minimal immune infiltrate in the sciatic nerves (Fig. 1 and 2) and, concurrently, activated T cells in the spleen and lymph nodes (Fig. 5). One potential explanation for these disparate findings is that IL-10 is required for T cell egress from secondary lymphoid structures. Without IL-10, then, activated T cells may accumulate in the lymph nodes rather than migrating into peripheral nerves. Since reduced lymphocyte egress is associated with increased lymph node cellularity (48), we first tested whether IL-10 deficiency may affect migration by investigating lymph node size in IL-10-deficient mice. Indeed, the nerve-draining lumbar lymph nodes were enlarged in IL-10-deficient AireGW/+ mice relative to WT and IL-10-sufficient AireGW/+ mice (Fig. 6A). On average, IL-10-deficient AireGW/+ lumbar lymph nodes had >3-fold more cells than WT lymph nodes and ~3-fold more cells than IL-10-sufficient AireGW/+ lymph nodes (Fig. 6B). These findings lend support to the hypothesis that IL-10 may accelerate SAPP by promoting migration of T cells out of lymph nodes and into nerves.
Figure 6. IL-10 promotes lymphocyte migration through STAT3 dependent S1pr1 induction.
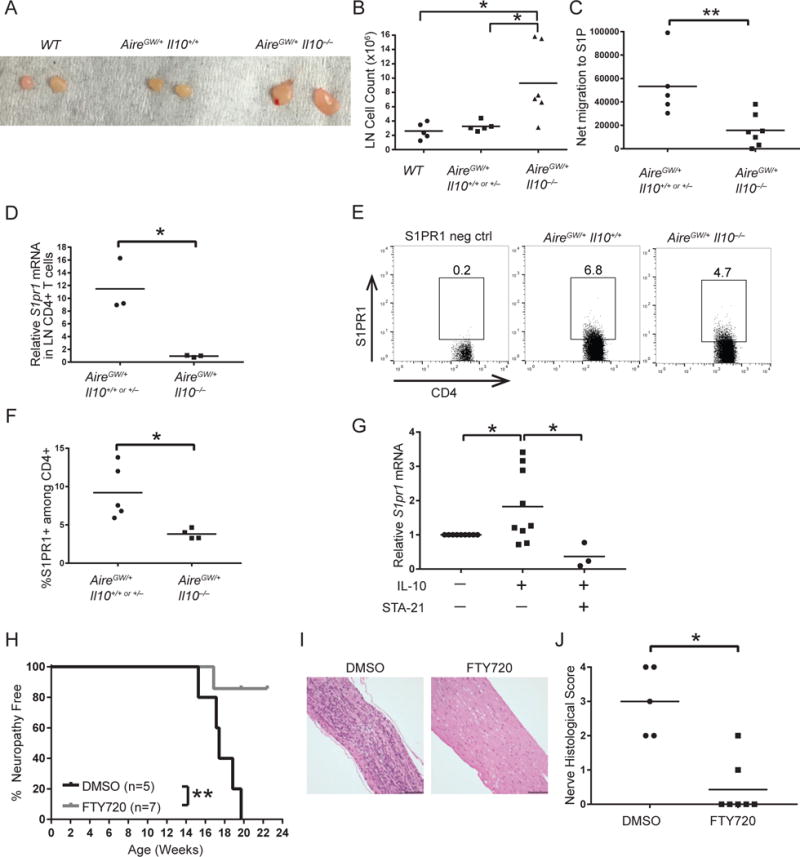
Image (A) and cumulative cell counts (B) of lumbar lymph nodes from 22-wk-old WT, AireGW/+ Il10+/+ or +/−, and AireGW/+ Il10−/− mice. p-values were calculated using one-way ANOVA with Tukey’s correction for multiple comparisons; *, p < 0.05. (C) Net migration in vitro to sphingosine-1-phosphate (S1P) of lymph node cells from 22-wk-old AireGW/+ Il10+/+ or +/− and AireGW/+ Il10−/− mice. p-value was calculated using two-tailed unpaired Student’s t test with Welch’s correction; **, p < 0.01. CD4+ T cells from lumbar lymph nodes of 22-wk-old AireGW/+ Il10+/+ or +/− and AireGW/+ Il10−/− mice were analyzed for S1PR1 expression. (D) Relative S1pr1 mRNA expression. p-value was calculated using two-tailed unpaired Student’s t test with Welch’s correction; *, p < 0.05. (E) Flow cytometry plots of lumbar lymph node CD4+ T cells stained for extracellular S1PR1. Gated on lymphocytes, singlets, and live cells. Numbers on plots represent frequencies of S1PR1-expressing cells. “S1PR1 neg ctrl” indicates negative control for S1PR1 staining. (F) Cumulative frequencies of CD4+S1PR1+ T cells. p-value was calculated using two-tailed unpaired Student’s t test with Welch’s correction; *, p < 0.05. (G) Relative S1pr1 mRNA expression of lumbar lymph node cells from WT Il10−/− mice stimulated with recombinant IL-10 or IL-10 plus STAT3 inhibitor (STA-21). p-values were calculated using one-sample t test (Unstimulated versus IL-10) and two-tailed unpaired Student’s t test (IL-10 versus IL-10 + STA-21) with false discovery rate adjustment for multiple comparisons; * indicates p < 0.05. DMSO or FTY720-treated AireGW/+ mice were monitored for SAPP. (H) SAPP incidence curve. p-value was calculated using Mantel-Cox log-rank test; **, p < 0.01. (I) H&E-stained sciatic nerves (magnification 200×). Scale bar = 98 μm. (J) Cumulative sciatic nerve infiltration scores. p-value was calculated using Fisher’s Exact test; *, p < 0.05.
T cells exit lymph nodes by expressing S1PR1 on the cell surface and following gradients of S1P (29). To test IL-10’s effects on lymphocyte migration, we measured chemotaxis to S1P of lymph node cells from IL-10-sufficient and IL-10-deficient AireGW/+ mice in a transwell assay. IL-10-deficient AireGW/+ lymph node cells had significantly reduced net migration to S1P, with a >3-fold reduction in migrating cells relative to IL-10-sufficient AireGW/+ mice (Fig. 6C). These findings suggest that IL-10 increases T cell migration to S1P.
We next sought to determine whether reduced chemotaxis to S1P by IL-10-deficient AireGW/+ lymphocytes may be due to reduced S1PR1 expression. To test this, we measured S1pr1 mRNA in CD4+ T cells purified from lumbar lymph nodes of IL-10-sufficient and deficient AireGW/+ mice. IL-10-deficient AireGW/+ CD4+ T cells expressed 12-fold lower levels of S1pr1 mRNA relative to IL-10-sufficient AireGW/+ controls (Fig. 6D). Conversely, IL-10 deficiency did not affect S1pr1 mRNA in lymph node CD4+ T cells of WT mice (Supp. Fig. 3). To determine whether reduced S1pr1 mRNA correlated with reduced surface protein expression, we measured S1PR1 surface protein on CD4+ T cells from lumbar lymph nodes of IL-10-sufficient and deficient AireGW/+ mice. A significantly lower frequency of CD4+ T cells from lymph nodes of IL-10-deficient AireGW/+ mice expressed S1PR1 surface protein relative to IL-10-sufficient AireGW/+ mice (Fig. 6E, 6F). Thus, S1pr1 mRNA and protein were reduced in IL-10-deficient AireGW/+ CD4+ T cells. Together, these findings support a model in which IL-10 upregulates S1PR1 on CD4+ T cells to enhance their migration into the PNS and promote SAPP development.
IL-10 induces S1pr1 mRNA in a STAT3-dependent manner
How IL-10 upregulates S1pr1 expression is unclear. IL-10 signaling has been shown to activate STAT3, and, separately, STAT3 has been shown to upregulate S1PR1 transcription (17, 28). Based on these findings, we reasoned that IL-10 may induce S1pr1 in T cells via STAT3 activation. To test this, we stimulated lymph node cells with recombinant IL-10 in vitro and measured relative expression of S1pr1 by qPCR. Lymph node cells from IL-10 deficient (WT Il10−/−) mice were used to avoid preexisting induction of S1pr1 by IL-10. We stimulated whole lumbar lymph node instead of sorted CD4+ T cells because: 1. the lymph node is predominantly composed of T cells, and 2. the additional time required for sorting exposes the cells to an S1P-low environment, which may lead to S1pr1 resensitization (37, 49). IL-10 induced S1pr1 mRNA by ~ 1.8-fold compared to unstimulated cells (Fig. 6G). Thus, addition of IL-10 is sufficient to upregulate S1pr1 in lymph node cells. Notably, STA-21, a STAT3 inhibitor (50), abrogated induction of S1pr1 by IL-10 (Fig. 6G). Together these data suggest that IL-10 activates STAT3 to induce S1pr1 transcription.
S1pr functional antagonist FTY720 suppresses SAPP development
S1PR1 expression was reduced in IL-10-deficient AireGW/+ mice (Fig. 6D–6F), suggesting that decreased S1PR1 may underlie SAPP protection in IL-10-deficient AireGW/+ mice. S1PR1 blockade is being investigated for therapeutic potential in multiple autoimmune diseases (51), and is currently in clinical use for the treatment of MS (52), an autoimmune disease of the central nervous system (CNS). However, it is unclear whether loss of S1PR1 prevents SAPP development in AireGW/+ mice. To test this, we blocked S1PR1 activity in AireGW/+ mice using the S1PR functional antagonist FTY720 (53). FTY720-treated AireGW/+ mice were protected from SAPP development relative to DMSO-treated controls (Fig. 6H). Additionally, on H&E stained sections of sciatic nerves, FTY720 treatment was associated with reduced infiltration (Fig. 6I, 6J). Thus, S1PR1 plays a key role in promoting SAPP and supports a model in which IL-10-mediated upregulation of S1PR1 promotes SAPP development.
Discussion
Similar to other autoimmune conditions, such as RA, Sjogren’s syndrome, and Grave’s disease (17, 54–56), IL-10 expression is upregulated in PBMCs of patients with autoimmune demyelinating polyneuropathy. Unlike most other autoimmune conditions, however, we report here that IL-10 promotes autoimmune peripheral neuropathy rather than suppressing the autoimmune response. Moreover, IL-10 deficiency was associated with an accumulation of activated CD4+ T cells in the draining lymph nodes and a lack of immune cell infiltration in nerves, which suggests a defect in lymphocyte egress from lymph nodes. Indeed, IL-10 deficiency was associated with reduced S1PR1-mediated lymphocyte migration, and IL-10 was sufficient to induce S1pr1 expression. These findings delineate a previously unappreciated mechanism by which IL-10 may function to promote autoimmune disease.
IL-10 has been reported to promote immune responses in other inflammatory contexts. For example, IL-10 enhances B cell-mediated immunity (23, 24). However, our data indicate that SAPP in Aire-deficient mice can occur independently of B cells. Thus, it is unlikely that IL-10 is functioning through B cell activation to promote SAPP. IL-10 also promotes CD8+ T cell-mediated immunity (Zhang et al., 2016). Although it is possible that this IL-10-dependent mechanism may be contributing to SAPP development, our adoptive transfer experiments show that CD8+ T cells are dispensable for SAPP transfer since NOD.Scid mice lack CD8+ T cells (45). Instead, our data suggest IL-10 induces S1pr1 expression, which promotes CD4+ T cell-mediated migration from lymph nodes and autoimmunity against the PNS.
On the surface, our findings may seem to contradict a recent report that IL-10 protected NOD.B7-2−/− mice from SAPP (57). Quan et al., 2015 show that transfer of dendritic cells (DCs) pretreated with IL-10 protects from neuropathy. These seemingly contradictory results, however, may not be mutually exclusive. It is possible that IL-10 has distinct effects on different immune cell types in SAPP pathogenesis. While IL-10 may have anti-inflammatory effects on DC’s, it may also promote CD4+ T cell egress from lymph nodes in SAPP. Overall, however, IL-10 appears to have a SAPP-accelerating effect, since mice with global IL-10 deficiency are protected from SAPP.
IL-10 protects against inflammation in multiple tissues, such as the colon, joints, and the CNS (17). What leads IL-10 to promote autoimmunity against the PNS, and not against other organs, is not clear. Peripheral nerves fundamentally differ from other tissues in several ways. For instance, a blood-nerve barrier surrounds the PNS, making it an immunologically privileged site (58). Furthermore, since nerve tissue and nerve antigens are widely dispersed throughout the body, the mechanisms behind nerve inflammation and tolerance may have unique characteristics. Also, tertiary lymphoid structures within target organs have been reported to be an important source of autoimmune T cells in autoimmune diabetes, MS, SLE, and RA (59), but are not seen in the PNS. Thus, differences in location (e.g., tertiary vs. secondary) of critical lymphoid structures may be key in accounting for differences between organs.
CIDP has been observed in several patients with mutations in AIRE (60); however, the majority of CIDP patients have no known AIRE deficiency. Thus, an important question is whether results obtained in the Aire deficiency model of SAPP can be generalized. We observed that IL-10 deficiency protected mice from SAPP not only in the Aire deficiency (AireGW/+) model, but also in the Aire-independent, anti-B7-1/2 antibody-induced model. Thus, our findings suggest that IL-10’s promotion of SAPP pathogenesis is a common feature of autoimmune peripheral neuropathies. Since the Aire-deficient and anti-B7-1/2 antibody models are both on the NOD mouse background, it remains possible that IL-10’s promotion of PNS autoimmunity is specific to the NOD background. SAPP models reported to date are all on the NOD background (5, 6, 8, 61, 62), so it is not currently possible to test IL-10’s effects on SAPP in other mouse strains. Given the efficacy of S1PR1 as a therapeutic target (51), there has been much effort in delineating factors controlling its expression. Although post-transcriptional and post-translational regulation of S1PR1 have been well-studied (49, 63–67), not as many transcriptional regulators have been found. Three known transcriptional regulators include vascular endothelial growth factor (VEGF) (68), Krüppel-like factor 2 (KLF2) (49, 69), and STAT3 (28). Lee et al., 2010 found that STAT3-dependent induction of S1pr1 is partially driven by IL-6, since IL-6 signals through STAT3. Here, we show a previously unappreciated function of IL-10 in inducing S1pr1 transcription through STAT3.
Our results have multiple clinical implications. We show that IL-10 paradoxically promotes SAPP, which suggests increased IL-10 observed in CIDP patients may be a marker of disease progression rather than resolution. Furthermore, IL-10 is currently under investigation to treat autoimmune and inflammatory diseases, including RA (19, 20), MS (21, 22), and Crohn’s disease (70). Our data suggest that IL-10 treatment may have previously unanticipated effects: IL-10 therapy may exacerbate disease in CIDP patients and/or unmask PNS autoimmunity in susceptible individuals. FTY720 is currently under investigation for clinical use to treat CIDP, and our data provide support for its use in treating autoimmune peripheral neuropathy (71). Separately, autoimmune peripheral neuropathies are an increasingly common side-effect of current treatments and infections. For example, immune checkpoint inhibitors are being extensively tested in skin, lung, and kidney cancers, but may cause PNS autoimmunity (72, 73). Furthermore, recent Zika virus outbreaks have been strongly linked to an acute autoimmune peripheral neuritis that resembles Guillain-Barré syndrome (74, 75). Thus, there is mounting need to understand the pathogenesis of autoimmune peripheral neuropathies. This study demonstrates a critical role for IL-10 in promoting lymphocyte migration and accelerating PNS autoimmunity, and has far-reaching implications for current treatment strategies in cancer and autoimmunity.
Supplementary Material
Acknowledgments
We thank Edward Miao, Yisong Wan, Roland Tisch, Justin Wilson, Laurel Kartchner, and Hsing-Hui Wang for feedback on this manuscript. We thank Joshua Starmer and Dominic Moore for assistance with statistics.
Animal histopathology was performed in the LCCC Animal Histopathology Core Facility at the University of North Carolina at Chapel Hill. We thank the UNC Flow Cytometry core for assistance with flow cytometry experiments.
Abbreviations
- Aire
autoimmune regulator
- CIDP
chronic inflammatory demyelination polyneuropathy
- CNS
central nervous system
- EAE
experimental autoimmune encephalitis
- GW
G228W mutation in Aire
- LN
lymph node
- MS
multiple sclerosis
- NOD
non-obese diabetic
- P0
myelin protein zero
- PNS
peripheral nervous system
- RA
rheumatoid arthritis
- S1P
sphingosine-1-phosphate
- S1PR1
sphingosine-1-phosphate receptor 1
- SAPP
spontaneous autoimmune peripheral polyneuropathy
- SLE
systemic lupus erythematosus
- WT
wild-type
Footnotes
This work is supported by the National Institutes of Health (NIH)/National Institute for Neurological Disorders and Stroke (NINDS) R01 grant (to M.A.S) and F30 grant (to C-J.S.).
References
- 1.Dalakas MC. Pathogenesis of immune-mediated neuropathies. Biochim Biophys Acta-Mol Basis Dis. 2015;1852:658–666. doi: 10.1016/j.bbadis.2014.06.013. [DOI] [PubMed] [Google Scholar]
- 2.Dalakas MC. Advances in the diagnosis, pathogenesis and treatment of CIDP. Nat Rev Neurol. 2011;7:507–517. doi: 10.1038/nrneurol.2011.121. [DOI] [PubMed] [Google Scholar]
- 3.Ropper AH. Current treatments for CIDP. Neurology. 2003;60:S16–S22. doi: 10.1212/wnl.60.8_suppl_3.s16. [DOI] [PubMed] [Google Scholar]
- 4.Kuwabara S. Long term prognosis of chronic inflammatory demyelinating polyneuropathy: a five year follow up of 38 cases. J Neurol Neurosurg Psychiatry. 2006;77:66–70. doi: 10.1136/jnnp.2005.065441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Su MA, Davini D, Cheng P, Giang K, Fan U, DeVoss JJ, Johannes KPA, Taylor L, Shum AK, Valenzise M, Meloni A, Bour-Jordan H, Anderson MS. Defective Autoimmune Regulator-Dependent Central Tolerance to Myelin Protein Zero Is Linked to Autoimmune Peripheral Neuropathy. J Immunol. 2012;188:4906–4912. doi: 10.4049/jimmunol.1200493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Meyer zu Horste G, Mausberg AK, Cordes S, El-Haddad H, Partke H-J, Leussink VI, Roden M, Martin S, Steinman L, Hartung H-P, Kieseier BC. Thymic Epithelium Determines a Spontaneous Chronic Neuritis in Icam1 tm1Jcgr NOD Mice. J Immunol. 2014;193:2678–2690. doi: 10.4049/jimmunol.1400367. [DOI] [PubMed] [Google Scholar]
- 7.Brosnan JV, Craggs RI, King RHM, Thomas PK. Reduced susceptibility of T cell-deficient rats to induction of experimental allergic neuritis. J Neuroimmunol. 1987;14:267–282. doi: 10.1016/0165-5728(87)90014-2. [DOI] [PubMed] [Google Scholar]
- 8.Salomon B, Rhee L, Bour-Jordan H, Hsin H, Montag A, Soliven B, Arcella J, Girvin AM, Padilla J, Miller SD, Bluestone JA. Development of spontaneous autoimmune peripheral polyneuropathy in B7-2-deficient NOD mice. J Exp Med. 2001;194:677–84. doi: 10.1084/jem.194.5.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Abraham PM, Quan SH, Dukala D, Soliven B. CD19 as a therapeutic target in a spontaneous autoimmune polyneuropathy. Clin Exp Immunol. 2014;175:181–191. doi: 10.1111/cei.12215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Madia F, Frisullo G, Nociti V, Conte A, Luigetti M, Grande AD, Patanella AK, Iorio R, Tonali PA, Batocchi AP, Sabatelli M. pSTAT1, pSTAT3, and T-bet as markers of disease activity in chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst. 2009;14:107–117. doi: 10.1111/j.1529-8027.2009.00220.x. [DOI] [PubMed] [Google Scholar]
- 11.Csurhes PA, Sullivan AA, Green K, Pender MP, McCombe PA. T cell reactivity to P0, P2, PMP-22, and myelin basic protein in patients with Guillain-Barre syndrome and chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Neurosurg Psychiatry. 2005;76:1431–1439. doi: 10.1136/jnnp.2004.052282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Beppu M, Sawai S, Misawa S, Sogawa K, Mori M, Ishige T, Satoh M, Nomura F, Kuwabara S. Serum cytokine and chemokine profiles in patients with chronic inflammatory demyelinating polyneuropathy. J Neuroimmunol. 2015;279:7–10. doi: 10.1016/j.jneuroim.2014.12.017. [DOI] [PubMed] [Google Scholar]
- 13.Zhang HL, Hassan MY, Zheng XY, Azimullah S, Quezada HC, Amir N, Elwasila M, Mix E, Adem A, Zhu J. Attenuated EAN in TNF-α deficient mice is associated with an altered balance of M1/M2 macrophages. PLoS One. 2012;7 doi: 10.1371/journal.pone.0038157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bour-Jordan H, Thompson HL, Bluestone J. Distinct Effector Mechanisms in the Development of Autoimmune Neuropathy versus Diabetes in Nonobese Diabetic Mice. J Immunol. 2005;175:5649–5655. doi: 10.4049/jimmunol.175.9.5649. [DOI] [PubMed] [Google Scholar]
- 15.Zeng XL, Nagavalli A, Smith CJ, Howard JF, Su MA. Divergent Effects of T Cell Costimulation and Inflammatory Cytokine Production on Autoimmune Peripheral Neuropathy Provoked by Aire Deficiency. J Immunol. 2013;190:3895–3904. doi: 10.4049/jimmunol.1203001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sanvito L, Makowska A, Mahdi-Rogers M, Hadden RDM, Peakman M, Gregson N, Nemni R, Hughes RAC. Humoral and cellular immune responses to myelin protein peptides in chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Neurosurg Psychiatry. 2009;80:333–338. doi: 10.1136/jnnp.2008.159798. [DOI] [PubMed] [Google Scholar]
- 17.Moore KW, de Waal Malefyt R, Coffman RL, O’Garra A. Interleukin-10 and the Interleukin-10 Receptor. Annu Rev Immunol. 2001;19:683–765. doi: 10.1146/annurev.immunol.19.1.683. [DOI] [PubMed] [Google Scholar]
- 18.Kwilasz AJ, Grace PM, Serbedzija P, Maier SF, Watkins LR. The therapeutic potential of interleukin-10 in neuroimmune diseases. Neuropharmacology. 2015;2:55–69. doi: 10.1016/j.neuropharm.2014.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Keravala A, Lechman ER, Nash J, Mi Z, Robbins PD. Human, viral or mutant human IL-10 expressed after local adenovirus-mediated gene transfer are equally effective in ameliorating disease pathology in a rabbit knee model of antigen-induced arthritis. Arthritis Res Ther. 2006;8:R91. doi: 10.1186/ar1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lubberts E, Joosten LAB, Van Den Bersselaar L, Helsen MMA, Bakker AC, Xing Z, Richards CD, Van Den Berg WB. Intra-articular IL-10 gene transfer regulates the expression of collagen-induced arthritis (CIA) in the knee and ipsilateral paw. Clin Exp Immunol. 2000;120:375–383. doi: 10.1046/j.1365-2249.2000.01217.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sloane E, Ledeboer A, Seibert W, Coats B, van Strien M, Maier SF, Johnson KW, Chavez R, Watkins LR, Leinwand L, Milligan ED, Van Dam AM. Anti-inflammatory cytokine gene therapy decreases sensory and motor dysfunction in experimental Multiple Sclerosis: MOG-EAE behavioral and anatomical symptom treatment with cytokine gene therapy. Brain Behav Immun. 2009;23:92–100. doi: 10.1016/j.bbi.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cua DJ, Hutchins B, LaFace DM, Stohlman SA, Coffman RL. Central Nervous System Expression of IL-10 Inhibits Autoimmune Encephalomyelitis. J Immunol. 2001;166:602–608. doi: 10.4049/jimmunol.166.1.602. [DOI] [PubMed] [Google Scholar]
- 23.Go NF. Interleukin 10, a novel B cell stimulatory factor: unresponsiveness of X chromosome-linked immunodeficiency B cells. J Exp Med. 1990;172:1625–1631. doi: 10.1084/jem.172.6.1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rousset F, Garcia E, Defrance T, Peronne C, Vezzio N, Hsu D, Kasteleini ROB, Moore KW, Banchereau J. Interleukin 10 is a potent growth and differentiation factor for activated human B lymphocytes. Proc Natl Acad Sci. 1992;89:1890–1893. doi: 10.1073/pnas.89.5.1890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Joo H, Coquery C, Xue Y, Gayet I, Dillon SR, Punaro M, Zurawski G, Banchereau J, Pascual V, Oh S. Serum from patients with SLE instructs monocytes to promote IgG and IgA plasmablast differentiation. J Exp Med. 2012;209:1335–1348. doi: 10.1084/jem.20111644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Akdis CA, Akdis M. Mechanisms of immune tolerance to allergens: role of IL-10 and Tregs. J Clin Invest. 2014;124:30–38. doi: 10.1172/JCI78891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang H, Wang Y, Hwang ES, He YW. Interleukin-10 : An Immune-Activating Cytokine in Cancer Immunotherapy. J Clin Oncol. 2016;34:3576–3578. doi: 10.1200/JCO.2016.69.6435. [DOI] [PubMed] [Google Scholar]
- 28.Lee H, Deng J, Kujawski M, Yang C, Liu Y, Herrmann A, Kortylewski M, Horne D, Somlo G, Forman S, Jove R, Yu H. STAT3-induced S1PR1 expression is crucial for persistent STAT3 activation in tumors. Nat Med. 2010;16:1421–1428. doi: 10.1038/nm.2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Matloubian M, Lo CG, Cinamon G, Lesneski MJ, Xu Y, Brinkmann V, Allende ML, Proia RL, Cyster JG. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature. 2004;427:355–360. doi: 10.1038/nature02284. [DOI] [PubMed] [Google Scholar]
- 30.Kim HJ, Jung CG, Dukala D, Bae H, Kakazu R, Wollmann R, Soliven B. Fingolimod and related compounds in a spontaneous autoimmune polyneuropathy. J Neuroimmunol. 2009;214:93–100. doi: 10.1016/j.jneuroim.2009.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Su MA, Giang K, Žumer K, Jiang H, Oven I, Rinn JL, DeVoss JJ, Johannes KPA, Lu W, Gardner J, Chang A, Bubulya P, Chang HY, Peterlin BM, Anderson MS. Mechanisms of an autoimmunity syndrome in mice caused by a dominant mutation in Aire. J Clin Invest. 2008;118:1712–1726. doi: 10.1172/JCI34523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Salomon B, Rhee L, Bour-Jordan H, Hsin H, Montag a, Soliven B, Arcella J, Girvin aM, Padilla J, Miller SD, Bluestone Ja. Development of spontaneous autoimmune peripheral polyneuropathy in B7-2-deficient NOD mice. J Exp Med. 2001;194:677–84. doi: 10.1084/jem.194.5.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shum AK, DeVoss J, Tan CL, Hou Y, Johannes K, O’Gorman CS, Jones KD, Sochett EB, Fong L, Anderson MS. Identification of an Autoantigen Demonstrates a Link Between Interstitial Lung Disease and a Defect in Central Tolerance. Sci Transl Med. 2009;1:9ra20–9ra20. doi: 10.1126/scitranslmed.3000284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Madsen K, Doyle J, Jewell L, Tavernini M, Fedorak R. IL10 colitis scoring Gastroenterology 1999.pdf. 1999:1107–1114. doi: 10.1016/s0016-5085(99)70013-2. [DOI] [PubMed] [Google Scholar]
- 35.Leiter EH. The NOD Mouse: A Model for Insulin-Dependent Diabetes Mellitus. Curr Protoc Immunol. 2001;1–23 doi: 10.1002/0471142735.im1509s24. [DOI] [PubMed] [Google Scholar]
- 36.Xia RH, Yosef N, Ubogu EE. Dorsal caudal tail and sciatic motor nerve conduction studies in adult mice: Technical aspects and normative data. Muscle and Nerve. 2010;41:850–856. doi: 10.1002/mus.21588. [DOI] [PubMed] [Google Scholar]
- 37.Sensken SC, Nagarajan M, Bode C, Graler MH. Local Inactivation of Sphingosine 1-Phosphate in Lymph Nodes Induces Lymphopenia. J Immunol. 2011;186:3432–3440. doi: 10.4049/jimmunol.1002169. [DOI] [PubMed] [Google Scholar]
- 38.Bouchard C, Lacroix C, Plante V, Adams D, Chedru F, Guglielmi JM, Said G. Clinicopathologic findings and prognosis of chronic inflammatory demyelinating polyneuropathy. Neurology. 1999;52:498–503. doi: 10.1212/wnl.52.3.498. [DOI] [PubMed] [Google Scholar]
- 39.Cornblath DR, Griffin DE, Welch D, Griffin JW, McArthur JC. Quantitative analysis of endoneurial T-cells in human sural nerve biopsies. J Neuroimmunol. 1990;26:113–118. doi: 10.1016/0165-5728(90)90082-x. [DOI] [PubMed] [Google Scholar]
- 40.Schmidt B, Toyka KV, Kiefer R, Full J, Hartung HP, Pollard J. Inflammatory infiltrates in sural nerve biopsies in Guillain-Barre syndrome and chronic inflammatory demyelinating neuropathy. Muscle Nerve. 1996;19:474–87. doi: 10.1002/(SICI)1097-4598(199604)19:4<474::AID-MUS8>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 41.Serreze DV, Chapman HD, Post CM, Johnson EA, Suarez-Pinzon WL, Rabinovitch A. Th1 to Th2 Cytokine Shifts in Nonobese Diabetic Mice: Sometimes an Outcome, Rather Than the Cause, of Diabetes Resistance Elicited by Immunostimulation. J Immunol. 2001;166:1352–1359. doi: 10.4049/jimmunol.166.2.1352. [DOI] [PubMed] [Google Scholar]
- 42.Kühn R, Löhler J, Rennick D, Rajewsky K, Müller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 43.Sellon RK, Tonkonogy S, Schultz M, Dieleman LA, Grenther W, Balish E, Rennick DM, Sartor RB. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect Immun. 1998;66:5224–5231. doi: 10.1128/iai.66.11.5224-5231.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Asghar MN, Emani R, Alam C, Helenius TO, Grönroos TJ, Sareila O, Din MU, Holmdahl R, Hänninen A, Toivola DM. In Vivo Imaging of Reactive Oxygen and Nitrogen Species in Murine Colitis. Inflamm Bowel Dis. 2014;20:1435–1447. doi: 10.1097/MIB.0000000000000118. [DOI] [PubMed] [Google Scholar]
- 45.Blunt T, Finnie NJ, Taccioli GE, Smith GCM, Demengeot J, Gottlieb TM, Mizuta R, Varghese AJ, Alt FW, Jeggo PA, Jackson SP. Defective DNA-dependent protein kinase activity is linked to V(D)J recombination and DNA repair defects associated with the murine scid mutation. Cell. 1995;80:813–823. doi: 10.1016/0092-8674(95)90360-7. [DOI] [PubMed] [Google Scholar]
- 46.Serreze DV, Chapman HD, Varnum DS, Hanson MS, Reifsnyder PC, Richard SD, Fleming SA, Leiter EH, Shultz LD. B lymphocytes are essential for the initiation of T cell-mediated autoimmune diabetes: analysis of a new “speed congenic” stock of NOD.Ig mu null mice. J Exp Med. 1996;184:2049. doi: 10.1084/jem.184.5.2049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Groux H, Garra A, Bigler M, Rouleau M, Antonenko S, Vries JE, Roncarolo M-GG, O’Garra a, Bigler M, Rouleau M, Antonenko S, deVries JE, Roncarolo M-GG. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature. 1997;389:737–742. doi: 10.1038/39614. [DOI] [PubMed] [Google Scholar]
- 48.Green DS, Center DM, Cruikshank WW. Human immunodeficiency virus type 1 gp120 reprogramming of CD4+ T-cell migration provides a mechanism for lymphadenopathy. J Virol. 2009;83:5765–5772. doi: 10.1128/JVI.00130-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cyster JG, Schwab SR. Sphingosine-1-Phosphate and Lymphocyte Egress from Lymphoid Organs. Annu Rev Immunol. 2012;30:69–94. doi: 10.1146/annurev-immunol-020711-075011. [DOI] [PubMed] [Google Scholar]
- 50.Jerez A, Clemente MJ, Makishima H, Koskela H, LeBlanc F, Ng KP, Olson T, Przychodzen B, Afable M, Gomez-Segui I, Guinta K, Durkin L, Hsi ED, McGraw K, Zhang D, Wlodarski MW, Porkka K, Sekeres MA, List A, Mustjoki S, Loughran TP, Maciejewski JP. STAT3 mutations unify the pathogenesis of chronic lymphoproliferative disorders of NK cells and T-cell large granular lymphocyte leukemia. Blood. 2012;120:3048–3057. doi: 10.1182/blood-2012-06-435297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mao-Draayer Y, Sarazin J, Fox D, Schiopu E. The sphingosine-1-phosphate receptor: A novel therapeutic target for multiple sclerosis and other autoimmune diseases. Clin Immunol. 2017;175:10–15. doi: 10.1016/j.clim.2016.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Calabresi PA, Radue EW, Goodin D, Jeffery D, Rammohan KW, Reder AT, Vollmer T, Agius MA, Kappos L, Stites T, Li B, Cappiello L, von Rosenstiel P, Lublin FD. Safety and efficacy of fingolimod in patients with relapsing-remitting multiple sclerosis (FREEDOMS II): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Neurol. 2014;13:545–556. doi: 10.1016/S1474-4422(14)70049-3. [DOI] [PubMed] [Google Scholar]
- 53.Mandala S, Hajdu R, Bergstrom J, Quackenbush E, Xie J, Milligan J, Thornton R, Shei G, Card D, Keohane C, Rosenbach M, Hale J, Lynch CL, Rupprecht K, Parsons W, Rosen H. Alteration of Lymphocyte Trafficking by Sphingosine-1-Phosphate Receptor Agonists. Science. 2002;296:346–350. doi: 10.1126/science.1070238. [DOI] [PubMed] [Google Scholar]
- 54.Mirakian R, Hammond LJ, Bottazzo GF. TH1 and TH2 cytokine control of thyrocyte survival in thyroid autoimmunity. Nat Immunol. 2001;2:371. doi: 10.1038/87661. [DOI] [PubMed] [Google Scholar]
- 55.Lopatin U, Yao X, Williams RK, Bleesing JJ, Dale JK, Wong D, Teruya-Feldstein J, Fritz S, Morrow MR, Fuss I, Sneller MC, Raffeld M, Fleisher TA, Puck JM, Strober W, Jaffe ES, Straus SE. Increases in circulating and lymphoid tissue interleukin-10 in autoimmune lymphoproliferative syndrome are associated with disease expression. Blood. 2001;97:3161–3170. doi: 10.1182/blood.v97.10.3161. [DOI] [PubMed] [Google Scholar]
- 56.Lalani I, Bhol K, Ahmed AR. Interleukin-10: biology, role in inflammation and autoimmunity. Ann Allergy Asthma Immunol. 1997;79:469–483. doi: 10.1016/S1081-1206(10)63052-9. [DOI] [PubMed] [Google Scholar]
- 57.Quan S, Kim HJ, Dukala D, Sheng JR, Soliven B. Impaired Dendritic Cell Function in a Spontaneous Autoimmune Polyneuropathy. J Immunol. 2015;194:4175–4184. doi: 10.4049/jimmunol.1401766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kanda T, Numata Y, Mizusawa H. Chronic inflammatory demyelinating polyneuropathy: decreased claudin-5 and relocated ZO-1. J Neurol Neurosurg Psychiatry. 2004;75:765–769. doi: 10.1136/jnnp.2003.025692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Neyt K, Perros F, GeurtsvanKessel CH, Hammad H, Lambrecht BN. Tertiary lymphoid organs in infection and autoimmunity. Trends Immunol. 2012;33:297–305. doi: 10.1016/j.it.2012.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Valenzise M, Aversa T, Salzano G, Zirilli G, De Luca F, Su M. Novel insight into Chronic Inflammatory Demyelinating Polineuropathy in APECED syndrome: molecular mechanisms and clinical implications in children. Ital J Pediatr. 2017;43:11. doi: 10.1186/s13052-017-0331-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Louvet C, Kabre BG, Davini DW, Martinier N, Su Ma, DeVoss JJ, Rosenthal WL, Anderson MS, Bour-Jordan H, Bluestone Ja. A novel myelin P0-specific T cell receptor transgenic mouse develops a fulminant autoimmune peripheral neuropathy. J Exp Med. 2009;206:507–514. doi: 10.1084/jem.20082113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Briet C, Bourdenet G, Rogner UC, Becourt C, Tardivel I, Drouot L, Arnoult C, do Rego JC, Prevot N, Massaad C, Boyer O, Boitard C. The Spontaneous Autoimmune Neuromyopathy in ICOSL−/− NOD Mice is CD4+ T-cell and Interferon-γ Dependent. Front Immunol. 2017;8:1–14. doi: 10.3389/fimmu.2017.00287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Arnon TI, Xu Y, Pham T, An J, Coughlin S, Dorn GW, Cyster JG. GRK2-Dependent S1PR1 Desensitization Is Required for Lymphocytes to Overcome Their Attraction to Blood. Science. 2011;333 doi: 10.1126/science.1208248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bankovich AJ, Shiow LR, Cyster JG. CD69 Suppresses Sphingosine 1-Phosophate Receptor-1 (S1P1) Function through Interaction with Membrane Helix 4. J Biol Chem. 2010;285:22328–22337. doi: 10.1074/jbc.M110.123299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shiow LR, Rosen DB, Brdickova N, Xu Y, An J, Lanier LL, Cyster JG, Matloubian M. CD69 acts downstream of interferon-a/b to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature. 2006;440 doi: 10.1038/nature04606. [DOI] [PubMed] [Google Scholar]
- 66.Willinger T, Ferguson SM, Pereira JP, De Camilli P, Flavell RA. Dynamin 2 – dependent endocytosis is required for sustained S1PR1 signaling. J Exp Med. 2014;211:685–700. doi: 10.1084/jem.20131343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Xin Q, Li J, Dang J, Bian X, Shan S, Yuan J, Qian Y, Liu Z, Liu G, Yuan Q, Liu N, Gao F, Liu Q. miR-155 Deficiency Ameliorates Autoimmune Inflammation of Systemic Lupus Erythematosus by Targeting S1pr1 in Fas lpr/lpr Mice. J Immunol. 2015;194:5437–5445. doi: 10.4049/jimmunol.1403028. [DOI] [PubMed] [Google Scholar]
- 68.Igarashi J, Erwin PA, Dantas AP, Chen H, Michel T. VEGF induces S1P1 receptors in endothelial cells: Implications for cross-talk between sphingolipid and growth factor receptors. Proc Natl Acad Sci. 2003;100:10664–10669. doi: 10.1073/pnas.1934494100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Carlson CM, Endrizzi BT, Wu J, Ding X, Weinreich MA, Walsh ER, Wani MA, Lingrel JB, Hogquist KA, Jameson SC. Kruppel-like factor 2 regulates thymocyte and T-cell migration. Nature. 2006;442:299–302. doi: 10.1038/nature04882. [DOI] [PubMed] [Google Scholar]
- 70.Asadullah K, Sterry W, Volk HD. Interleukin-10 Therapy — Review of a New Approach. Med Immunol. 2003;55:241–269. doi: 10.1124/pr.55.2.4. [DOI] [PubMed] [Google Scholar]
- 71.Hughes RAC, Dalakas M, Latov N, Léger JM, Merkies ISJ, Nobile-Orazio E, Agoropoulou C, Häring DA, Zhang-Auberson L, Von Rosenstiel P, Hartung HP. Oral fingolimod (FTY720) for the treatment of chronic inflammatory demyelinating polyradiculoneuropathy (CIDP): Study design of the phase 3 forcidp trial. J Peripher Nerv Syst. 2013;18:S48–S49. [Google Scholar]
- 72.Zimmer L, Goldinger SM, Hofmann L, Loquai C, Ugurel S, Thomas I, Schmidgen MI, Gutzmer R, Utikal JS, Göppner D, Hassel JC, Meier F, Tietze JK, Forschner A, Weishaupt C, Leverkus M, Wahl R, Dietrich U, Garbe C, Kirchberger MC, Eigentler T, Berking C, Gesierich A, Krackhardt AM, Schadendorf D, Schuler G, Dummer R, Heinzerling LM. Neurological, respiratory, musculoskeletal, cardiac and ocular side-effects of anti-PD-1 therapy. Eur J Cancer. 2016;60:210–225. doi: 10.1016/j.ejca.2016.02.024. [DOI] [PubMed] [Google Scholar]
- 73.Kourie HR, Klastersky JA. Side-effects of checkpoint inhibitor-based combination therapy. Curr Opin Oncol. 2016;28:306–313. doi: 10.1097/CCO.0000000000000295. [DOI] [PubMed] [Google Scholar]
- 74.Parra B, Lizarazo J, Jiménez-Arango JA, Zea-Vera AF, González-Manrique G, Vargas J, Angarita JA, Zuñiga G, Lopez-Gonzalez R, Beltran CL, Rizcala KH, Morales MT, Pacheco O, Ospina ML, Kumar A, Cornblath DR, Muñoz LS, Osorio L, Barreras P, Pardo CA. Guillain–Barré Syndrome Associated with Zika Virus Infection in Colombia. N Engl J Med. 2016;375:1513–1523. doi: 10.1056/NEJMoa1605564. [DOI] [PubMed] [Google Scholar]
- 75.Cao-lormeau VM, Blake A, Mons S, Lastere S, Roche C, Vanhomwegen J, Dub T, Baudouin L, Teissier A, Larre P, Vial A, Decam C, Choumet V, Halstead S, Willison H, Musset L, Manuguerra J, Despres P, Fournier E, Mallet H, Musso D, Fontanet A, Neil J, Ghawche F. Guillain-Barré Syndrome outbreak caused by ZIKA virus infection in French Polynesia. Lancet. 2016;387:1531–1539. doi: 10.1016/S0140-6736(16)00562-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


