ABSTRACT
The Alere i respiratory syncytial virus (RSV) assay is an isothermal nucleic acid amplification test capable of detecting RSV directly from respiratory specimens, with results being available in ≤13 min after test initiation. The objective of this study was to evaluate the performance characteristics of the Alere i RSV assay in a point-of-care setting by using direct nasopharyngeal (NP) swab specimens (direct NP) and nasopharyngeal swab specimens eluted and transported in viral transport medium (VTM NP). The study was a prospective, multicenter, clinical trial conducted at 9 sites across the United States to evaluate the clinical performance of the Alere i RSV assay with respiratory specimens obtained from both children (age, <18 years) and older adults (age, >60 years). The performance of the Alere i RSV assay was compared with that of the reference method, the Prodesse ProFlu+ real-time reverse transcriptase PCR (RT-PCR) assay. All specimens with discrepant test results were tested further by a second FDA-cleared PCR assay (the Verigene respiratory virus plus nucleic acid test; Luminex Inc., TX). A total of 554 subjects with signs and symptoms of respiratory infections were enrolled, and respiratory samples were collected in this study. In comparison with the ProFlu+ real-time RT-PCR, the overall sensitivity and specificity of Alere i RSV assay for the detection of RSV were 98.6% (95% confidence interval [CI], 94.4 to 99.7%) and 98.0% (95% CI, 95.8 to 99.1%), respectively, for direct NP and 98.6% (95% CI, 94.4 to 99.7%) and 97.8% (95% CI, 95.5 to 98.9%), respectively, for VTM NP. The Alere i RSV is a highly sensitive and specific molecular assay ideal for rapid RSV detection in patients in the point-of-care setting due to its minimal hands-on time and rapid result availability.
KEYWORDS: respiratory syncytial virus, nucleic acid amplification, point-of-care testing, sample to answer, automation
INTRODUCTION
Respiratory syncytial virus (RSV) is one of the most frequently detected pathogens in the lower respiratory tract, especially in young children. RSV is the most common cause of bronchiolitis and pneumonia in children younger than 1 year of age (1–3). RSV infection is associated with 2.1 million medical care visits and >55,000 hospitalizations of children under 5 years of age annually in the United States alone (4). In older adults, RSV is responsible for 177,000 hospitalizations and 14,000 deaths annually in the United States (5).
As clinical signs and symptoms are often similar in many respiratory illnesses, it can be difficult to distinguish between viral and bacterial infections (6). Inaccurate and delayed diagnosis of respiratory infections may lead to unnecessary antibiotic prescriptions and improper antiviral treatment (7, 8). The rapid and accurate diagnosis of a respiratory infection can improve overall patient management, reduce the length of hospital stay, and decrease health care costs (9, 10).
Several methods are available for detection of RSV, including viral culture, rapid antigen detection tests (RADTs), direct florescent-antibody (DFA) tests, and nucleic acid-based molecular methods (11). Over the past several years, the turnaround time (TAT) for viral detection has improved significantly with shell vial culture, but it can still be 16 to 48 h (12). RADTs generally have a TAT of less than 15 min and can be performed in the point-of-care (POC) setting. However, the sensitivity of these assays is highly variable, with sensitivities showing wide variations ranging from 44.0 to 95.0% (13–15). DFA tests have reported sensitivities ranging from 80.0% to 100.0% but take considerably longer than RADTs and are technically demanding (16, 17). Recently, nucleic acid-based molecular detection methods have become the “gold standard” due to their accuracy and a relatively shorter TAT of a few hours (13, 18–20). However, molecular methods are expensive, require a more experienced laboratory technologist, and cannot be performed in POC settings. To overcome these problems, several manufacturers have developed molecular assays that are rapid and easy to perform in POC settings. The Alere i RSV assay (Alere Scarborough) is an isothermal nucleic acid-based detection method that is intended to have a higher sensitivity than traditional RADTs but comparable TATs. The aim of this study was to evaluate the performance characteristics of the Alere i RSV assay by using direct nasopharyngeal (NP) swab specimens (direct NP) and nasopharyngeal swab specimens eluted and transported in viral transport medium (VTM NP) obtained from patients with suspected respiratory infection.
MATERIALS AND METHODS
Clinical specimens.
This prospective, multicenter clinical trial was conducted at 9 different sites across the United States during the 2015-2016 respiratory season. Pediatric patients (age, <18 years) and older adults (age, >60 years) with a suspected respiratory infection that met the inclusion criteria (at least two of the following symptoms: congested or runny nose, dry cough, irritability, decreased activity, breathing difficulties, myalgia, temperature of >100°F) were enrolled in this study. Any subject receiving palivizumab (Synagis), an FDA-approved antibody against RSV infection, was excluded. After informed consent to participate in this study was obtained, two specimens were collected from the nostril with the most visible drainage or congestion by the use of foam-tipped NP swabs (included in the Alere i RSV assay kit). Then, each swab was transported as a direct NP and VTM NP eluted in 3 ml transport medium. The study was approved by each site's institutional review board.
Alere i RSV assay nucleic acid amplification testing.
All samples collected were tested either within 2 h (when stored at room temperature) or within 24 h (when stored at 4°C) in the POC settings by nonlaboratory personnel by following the quick reference guide. All specimens were tested by either directly placing the swab in the buffer (direct NP) or adding 200 μl of universal transport medium (VTM NP) to the buffer in the sample receiver. Briefly, the test base and sample receiver are placed into Alere instrument and heated for 3 min (warm-up stage). Following the heating step, the specimen is added directly to the sample receiver. The transfer cartridge is then used to transfer the sample to the test base. Once this step has been confirmed by the user, the test proceeds and results are available within 10 min. This assay identifies positive test results as soon as an amplification threshold is reached (usually within 8 to 10 min after initiating the test). For the study, positive and negative quality control swabs were processed every day before testing of study specimens. VTM NP and direct NP with invalid results were repeated once.
Nucleic acid extraction for reference method.
Total nucleic acid was extracted from 200 μl of the viral transport medium (VTM) containing 20 μl of a universal internal control according to the manufacturer's instructions (Alere Scarborough, Inc.). In addition, a positive control and a negative control were added to monitor for PCR amplification and contamination, respectively. In brief, total RNA was extracted with a NucliSENS easyMAG automated extraction system (bioMérieux Inc., Durham, NC) according to the manufacturer's instructions, and the nucleic acid was eluted in 55 μl of elution buffer. The sample eluate was aliquoted into two separate Eppendorf tubes and stored at −80°C until further testing. All nucleic acid was extracted within 72 h of sample collection.
Real-time RT-PCR.
Reverse transcriptase PCR (RT-PCR) was performed with the Prodesse ProFlu+ assay for detection of influenza A virus, influenza B virus, and RSV (Hologic Inc., San Diego, CA). The assay was performed on all tested specimens, along with the recommended controls, according to the manufacturer's instructions on a Cepheid SmartCycler II real-time instrument with Dx software (version 1.7b). For discrepant analysis, a second FDA-cleared assay was used (Verigene respiratory virus plus nucleic acid test; Luminex Inc., TX).
Statistical analysis.
Statistical analysis of the difference in the diagnostic yields among the assays was performed using Fisher's exact test. Reportable data were summarized in two-by-two data tables according to an established method.
RESULTS
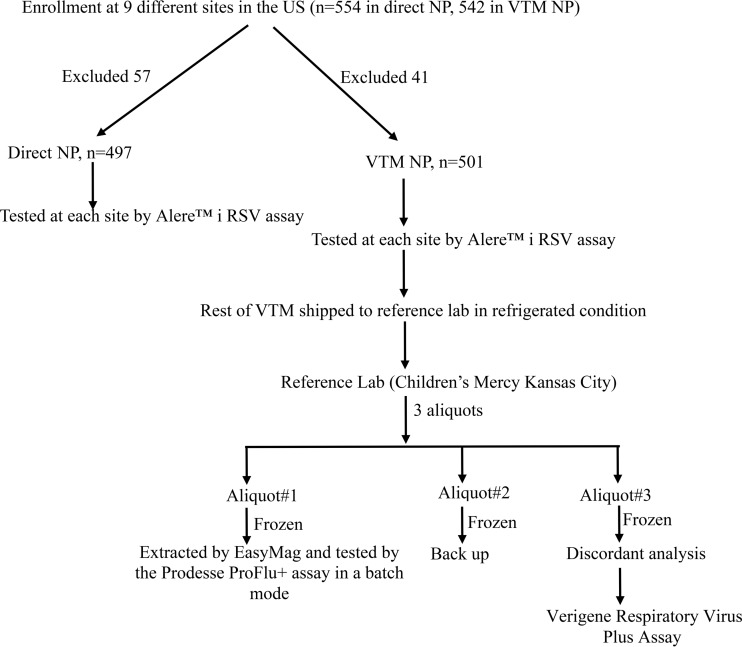
A flowchart of subject enrollment, sample collection, and testing is given in Fig. 1. More than 90% of the subject population in both the direct NP and VTM NP groups were children <18 years of age. Among the 554 subjects enrolled (children, n = 501; adults, n = 53) in the direct NP group, 57 samples (10.0%) were excluded due to various reasons, such as (i) invalid Alere i RSV assay results (4.5%, 25/554), (ii) invalid Prodesse ProFlu+ assay results (1.0%, 6/554), (iii) failure of the subject to meet the inclusion criteria (2.0%, 12/554), (iv) the use of inappropriate storage conditions or testing outside a 24-h window (2.0%, 13/554), and (v) failure to perform Alere i RSV control testing on the day of sample testing (0.2%, 1/554). Of 542 subjects enrolled (children, n = 488; adults, n = 54) in the VTM NP groups, 41 samples (7.5%) were excluded due to (i) invalid Alere i RSV results (2.0%, 11/542), (ii) invalid Prodesse ProFlu+ results (1.0%, 5/542), (iii) failure of the subject to meet the inclusion criteria (2.0%, 12/542), (iv) the use of inappropriate storage conditions or testing outside a 24-h window (2.0%, 12/542), and (v) failure to perform Alere i RSV control testing on the day of sample testing (0.2%, 1/542). After exclusion, the total sample size was 497 (children, n = 455; adults, n = 42) in the direct NP group and 501 (children, n = 459; adults, n = 42) in the VTM NP group. The rates of inhibition for the Alere i RSV assay were 4% and 2% in the direct NP group and the VTM NP group, respectively. Overall, the sensitivity and specificity of the Alere i RSV assay for both groups were ≥98.0% compared to the results of the Prodesse ProFlu+ assay (Table 1). A total of 19 samples were found to be discrepant, of which 15 samples were false positive (FP) by the Alere i RSV assay. Following discrepant analysis by a second FDA-cleared assay, 12 of the 15 FP samples (direct NP, n = 6; VTM NP, n = 6) were confirmed to be true positive (TP). RSV was detected by a second reference method in the remaining 4 samples false negative (FN) by the Alere i RSV assay (2 in each group). All samples were collected and tested at 9 different sites by nonlaboratory personnel. The sensitivity and specificity values of the Alere i RSV assay at each site are given in Table 2. These results indicate that regardless of the sample type and operator, the Alere i RSV assay demonstrated a high sensitivity and a high specificity in detecting RSV in respiratory samples obtained from patients with suspected respiratory infection.
FIG 1.
Flowchart of number of subjects enrolled and tested. Schematic diagram of symptomatic subjects enrolled in both the direct NP and VTM NP groups. Respiratory samples were collected and tested on-site by the Alere i RSV assay, and the rest of the samples were shipped to the reference lab for testing by the Prodesse ProFlu+ assay (the reference method).
TABLE 1.
Performance of the Alere i RSV assay versus real-time RT-PCRd
| Specimen type | No. of samples with the following result: |
% sensitivity (95% CI) | % specificity (95% CI) | % PPV (95% CI) | % NPV (95% CI) | |||
|---|---|---|---|---|---|---|---|---|
| TP | TN | FP | FN | |||||
| Direct NP | 137 | 351 | 7a | 2c | 98.6 (94.4–99.7) | 98.0 (95.8–99.1) | 95.1 (89.8–97.8) | 99.4 (97.7–99.9) |
| VTM NP | 138 | 353 | 8b | 2c | 98.6 (94.4–99.7) | 97.8 (95.5–98.9) | 94.5 (89.1–97.4) | 99.4 (97.7–99.9) |
RSV was detected in 6/7 false-positive samples by the Verigene respiratory virus plus assay.
RSV was detected in 6/8 false-positive samples by the Verigene respiratory virus plus assay.
RSV was detected in all 4 false-negative samples by the Verigene respiratory virus plus assay.
Abbreviations: TP, true positive; TN, true negative; FP, false positive; FN, false negative; PPV, positive predictive value; NPV, negative predictive value; CI, confidence interval.
TABLE 2.
Detection of RSV by the Alere i RSV assay at individual sitesa
| Sample type and site | No. of samples with the following result: |
% sensitivity (95% CI) | % specificity (95% CI) | Prevalence (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Total | TP | TN | FP | FN | ||||
| Direct NP | ||||||||
| Overall | 497 | 137 | 351 | 7b | 2c | 98.6 (94.4–99.7) | 98.0 (95.8–99.1) | 28.0 |
| Site 001A | 139 | 74 | 63 | 2d | 0 | 100.0 (93.80–100.0) | 96.9 (88.3–99.5) | 53.0 |
| Site 002A | 29 | 11 | 15 | 2d | 1c | 91.6 (59.7–99.5) | 88.2 (62.2–97.9) | 38.0 |
| Site 009A | 82 | 30 | 51 | 1d | 0 | 100.0 (85.8–100.0) | 98.0.0 (88.4–99.9) | 37.0 |
| Site 045A | 1 | 0 | 1 | 0 | 0 | NA | NA | 0 |
| Site 059A | 17 | 2 | 15 | 0 | 0 | 100.0 (19.8–100.0) | 100.0 (74.6–100.0) | 12 |
| Site 061A | 106 | 8 | 97 | 1 | 0 | 100.0 (59.8–100.0) | 91.5 (93.6–99.9) | 7.0 |
| Site 067A | 13 | 0 | 13 | 0 | 0 | N/A | 100.0 (71.6–100.0) | 0 |
| Site 071A | 82 | 8 | 73 | 0 | 1c | 100.0 (59.8–100.0) | 98.6 (91.7–99.9) | 10.0 |
| Site 074A | 28 | 4 | 23 | 1d | 0 | 100.0 (39.6–100.0) | 95.8 (76.8–99.7) | 18.0 |
| VTM NP | ||||||||
| Overall | 501 | 138 | 353 | 8e | 2c | 98.6 (94.4–99.7) | 97.8 (95.5–98.9) | 27.0 |
| Site 001A | 140 | 74 | 64 | 1 | 1c | 98.6 (91.8–99.9) | 98.5 (90.6–99.9) | 53.0 |
| Site 002A | 30 | 12 | 16 | 2d | 0 | 100.0 (69.8–100.0) | 88.8 (63.9–98.0) | 40.0 |
| Site 009A | 82 | 29 | 50 | 2d | 1c | 96.6 (80.1–99.8) | 96.1 (85.6–99.3) | 35.0 |
| Site 045A | 1 | 0 | 1 | 0 | 0 | NA | NA | 0 |
| Site 059A | 17 | 2 | 15 | 0 | 0 | 100.0 (19.8–100.0) | 100.0 (74.6–100.0) | 12.0 |
| Site 061A | 107 | 8 | 96 | 3f | 0 | 100.0 (59.7–100.0) | 97.0 (90.7–99.2) | 7.0 |
| Site 067A | 13 | 0 | 13 | 0 | 0 | NA | 100.0 (71.6–100.0) | 0 |
| Site 071A | 82 | 9 | 73 | 0 | 0 | 100.0 (62.9–100.0) | 100.0 (93.8–100.0) | 11.0 |
| Site 074A | 29 | 4 | 25 | 0 | 0 | 100.0 (39.6–100.0) | 100.0 (83.4–100.0) | 14.0 |
Abbreviations: TP, true positive; TN, true negative; FP, false positive; FN, false negative; CI, confidence interval; NA, not applicable.
RSV was detected in 6/7 false-positive samples by the Verigene respiratory virus plus assay.
RSV was not detected by the Verigene respiratory virus plus assay.
RSV was detected by the Verigene respiratory virus plus assay.
RSV was detected in 6/8 false-positive samples by the Verigene respiratory virus plus assay.
RSV was detected in 2/3 false-positive samples by the Verigene respiratory virus plus assay.
Because both children (age, <18 years) and older adults (age, >60 years) were enrolled in this study, we analyzed the sensitivity and specificity of the Alere i RSV assay separately in these age groups. In children, the sensitivity and specificity of the Alere i RSV assay were ≥97.5% for both sample types (Table 3). In the older-adult group, only 5 samples were TP for RSV, and all of them were detected by the Alere i RSV assay (Table 3), making the assay 100% sensitive and specific.
TABLE 3.
Detection of RSV in children and older adults by the Alere i RSV assaya
| Sample type and population | No. of samples with the following result: |
% sensitivity (95% CI) | % specificity (95% CI) | Prevalence (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Overall | TP | TN | FP | FN | ||||
| Direct NP | ||||||||
| Children | 455 | 132 | 314 | 7b | 2c | 98.5 (94.1–99.7) | 97.8 (95.3–99.0) | 29.0 |
| Older adults | 42 | 5 | 37 | 0 | 0 | 100 (46.2–100) | 100 (88.3–100.0) | 12.0 |
| VTM NP | ||||||||
| Children | 459 | 133 | 316 | 8d | 2c | 98.5 (94.2–99.7) | 97.5 (95.0–98.8) | 29.0 |
| Older adults | 42 | 5 | 37 | 0 | 0 | 100 (46.2–100) | 100 (88.3–100.0) | 12.0 |
Abbreviations: TP, true positive; TN, true negative; FP, false positive; FN, false negative; CI, confidence interval.
RSV was detected in 6/7 false-positive samples by the Verigene respiratory virus plus assay.
RSV was not detected by the Verigene respiratory virus plus assay.
RSV was detected in 6/8 false-positive samples by the Verigene respiratory virus plus assay.
The median threshold cycle (CT) value for samples that were positive by both the Alere i RSV assay and the Prodesse ProFlu+ assay was 25.0 (range, 18.9 to 36.6). Since the Alere i RSV assay does not generate a CT value, we could not determine the CT value for samples that were Alere i RSV positive but Prodesse ProFlu+ negative (n = 8). Two samples from the direct NP group that were missed by the Alere i RSV assay but detected by the Prodesse ProFlu+ assay had midrange CT values (27.4 and 33.7). In contrast, 2 samples from the VTM NP group missed by the Alere i RSV assay had CT values of 36.9 and 36.5.
DISCUSSION
Ideally, a diagnostic assay in the POC setting should be fast, accurate, and easy to perform by any nonlaboratory personnel. The most common diagnostic assays for RSV detection performed in the POC setting are RADTs. The majority of the RADTs have good specificities; however, the sensitivities of these assays vary widely in published reports, partly due to the assay used and the population tested. Although second-generation RADTs that employ an objective digital reader, such as the BD Veritor and Quidel Sofia assays, have been reported to have better sensitivity (range, 71.0% to 90.0%) (15, 21) than first-generation subjective-read RADT assays, such as the BinaxNOW and BD Directigen EZ assays (sensitivity range, 59.0 to 86.0%) (13, 14), they still fall short of optimal clinical performance. More sensitive (>95.0%) nucleic acid-based molecular assays are usually performed at moderate- to high-complexity laboratories and require technical expertise and specialized instrumentation. A simple nucleic acid-based diagnostic assay with a TAT similar to that of RADTs but with a higher sensitivity than RADTs is an ideal assay in the POC setting. In this prospective multicenter clinical trial, we evaluated the clinical performance of the Alere i RSV assay in the POC setting using two different respiratory sample types from patients with suspected respiratory infection. We found that the Alere i RSV assay was highly sensitive (98.6%) and specific (≥97.5%) in detecting RSV in both the direct NP and VTM NP groups, with results being available in 13 min or less.
In the past few years, nucleic acid-based detection tests have been developed for use in the POC setting. These assays are based on either the real-time PCR technology or isothermal amplification. The Alere i Influenza A&B assay (Alere Scarborough, ME) is an isothermal amplification assay which utilizes the nicking enzyme amplification reaction (NEAR) and was the first Clinical Laboratory Improvement Amendments (CLIA)-waived molecular assay cleared by the FDA in 2015, followed by the Alere i Strep A assay later in 2015 and the Alere i RSV assay in 2016. Both the Liat Influenza A/B and RSV assay (Roche Diagnostics, CA) and the Xpert Xpress Flu/RSV test (Cepheid Inc., CA) are CLIA-waived real-time PCR-based assays with TATs of between 20 and 30 min.
In this prospective study, we found that the Alere i RSV assay has a high sensitivity (98.5%) and a high specificity (≥97.5%) in detecting RSV in respiratory specimens of both the direct NP and VTM NP types from children. Similar results were obtained from hospitalized children in Germany (22). Using previously frozen, retrospective NP swab specimens eluted in universal transport medium, Peters et al. found that the sensitivity and specificity of the assay were 100.0% and 97.0%, respectively, compared to the results of real-time RT-PCR (22). However, the performance characteristics of the assay for older adult patients are not yet known. In our study, 42 adult patients were enrolled, of whom 5 were TP and none were FN, making the assay 100% sensitive and specific. As this is a relatively small sample size, additional studies are warranted to fully understand the assay performance in this patient population.
Rapid and accurate diagnostic assays for infectious disease have tremendous benefits for patient management. They have been shown to (i) reduce unnecessary antibiotic prescription, (ii) avoid chest X-ray use, (iii) reduce the length of hospitalization, and (iv) reduce overall patient charges (7, 9, 10, 23). Molecular-based diagnostic assays for infectious diseases in the POC setting are relatively new, and outcome studies are limited. In a recent study, the authors found that introduction of the Liat Influenza A/B and RSV assay in the emergency department (ED) had a significant impact on clinical management, including antimicrobial stewardship (24, 25). Another study reported that implementation of the Xpert Flu A/B assay reduced the mean wait time for patients in the ED by 9.1 h, decreased the mean isolation time of hospitalized patients by 23.7 h, and reduced the overall cost of the health care visit (26). Although no outcome study on rapid, nucleic acid-based detection of RSV alone in the POC setting is available thus far, on the basis of the results of the studies mentioned above, it is possible that this assay may have a similar impact on patient management.
One study published recently reported that the Alere i RSV assay is able to detect RSV within 5 to 7 min, with the total assay duration for negative samples being 13 min from the time of test initiation (22). In this study, we had a similar experience. The hands-on time for the assay was less than 2 min, with the total TAT being about 8 to 10 min (including a 3-min warm-up stage) for RSV-positive samples and 13 min for negative samples, making it an ideal assay in the POC setting.
In this study, all the testing by the Alere i RSV assay was conducted by nonlaboratory personnel, such as nurses, research enrollment staffs, and physician assistants, without any training on laboratory testing. These personnel were provided with the instruction booklet and quick reference guide by Alere Scarborough, which describes the step-by-step procedures necessary to perform the test. No major errors were observed due to operator handling of the assay across all 9 sites, suggesting that the test is easy to use. In addition, the Alere i instrument has a small footprint and so can be placed in an area with limited bench space. Since the instrument displays the result as either positive or negative (with occasional invalid results being reported), it eliminates the need for operator interpretation that is associated with traditional RADTs.
In summary, the Alere i RSV assay demonstrated high sensitivity (>98.0%) and high specificity (≥97.5%) in both the direct NP and VTM NP groups with respiratory samples obtained from children (age, <18 years) and older adults (age, >60 years) with suspected respiratory infection. The performance of the assay was very high in children (sensitivity, >98.0%; specificity, ≥97.5%). Although the sensitivity and specificity of the assay were both 100.0% in older adults, due to the small number of positive samples, it was difficult to adequately determine the performance of the assay in this age group. More studies in elderly patients are needed to determine the clinical performance of the assay in this population.
ACKNOWLEDGMENT
This clinical trial was supported by Alere Scarborough, ME.
REFERENCES
- 1.Tregoning JS, Schwarze J. 2010. Respiratory viral infections in infants: causes, clinical symptoms, virology, and immunology. Clin Microbiol Rev 23:74–98. doi: 10.1128/CMR.00032-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mansbach JM, Piedra PA, Teach SJ, Sullivan AF, Forgey T, Clark S, Espinola JA, Camargo CA Jr, MARC-30 Investigators. 2012. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med 166:700–706. doi: 10.1001/archpediatrics.2011.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Korppi M. 2014. Rhinovirus bronchiolitis: to be or not to be? Acta Paediatr 103:997–999. doi: 10.1111/apa.12737. [DOI] [PubMed] [Google Scholar]
- 4.Hall CB, Weinberg GA, Iwane MK, Blumkin AK, Edwards KM, Staat MA, Auinger P, Griffin MR, Poehling KA, Erdman D, Grijalva CG, Zhu Y, Szilagyi P. 2009. The burden of respiratory syncytial virus infection in young children. N Engl J Med 360:588–598. doi: 10.1056/NEJMoa0804877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.National Foundation for Infectious Diseases. 2016. Respiratory syncytial virus in older adults: a hidden annual epidemic: a report by the National Foundation for Infectious Diseases. National Foundation for Infectious Diseases, Bethesda, MD: http://www.nfid.org/publications/reports/rsv-report.pdf. [Google Scholar]
- 6.Jennings LC, Anderson TP, Werno AM, Beynon KA, Murdoch DR. 2004. Viral etiology of acute respiratory tract infections in children presenting to hospital: role of polymerase chain reaction and demonstration of multiple infections. Pediatr Infect Dis J 23:1003–1007. doi: 10.1097/01.inf.0000143648.04673.6c. [DOI] [PubMed] [Google Scholar]
- 7.Byington CL, Castillo H, Gerber K, Daly JA, Brimley LA, Adams S, Christenson JC, Pavia AT. 2002. The effect of rapid respiratory viral diagnostic testing on antibiotic use in a children's hospital. Arch Pediatr Adolesc Med 156:1230–1234. doi: 10.1001/archpedi.156.12.1230. [DOI] [PubMed] [Google Scholar]
- 8.Adcock PM, Stout GG, Hauck MA, Marshall G. 1997. Effect of rapid viral diagnosis on the management of children hospitalized with lower respiratory tract infection. Pediatr Infect Dis J 16:842–846. doi: 10.1097/00006454-199709000-00005. [DOI] [PubMed] [Google Scholar]
- 9.Rogers BB, Shankar P, Jerris RC, Kotzbauer D, Anderson EJ, Watson JR, O'Brien LA, Uwindatwa F, McNamara K, Bost JE. 2015. Impact of a rapid respiratory panel test on patient outcomes. Arch Pathol Lab Med 139:636–641. doi: 10.5858/arpa.2014-0257-OA. [DOI] [PubMed] [Google Scholar]
- 10.Barenfanger J, Drake C, Leon N, Mueller T, Troutt T. 2000. Clinical and financial benefits of rapid detection of respiratory viruses: an outcomes study. J Clin Microbiol 38:2824–2828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bawage SS, Tiwari PM, Pillai S, Dennis V, Singh SR. 2013. Recent advances in diagnosis, prevention, and treatment of human respiratory syncytial virus. Adv Virol 2013:595768. doi: 10.1155/2013/595768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Babady NE, Tang YW. 2015. Respiratory syncytial virus and human metapneumovirus, p 1498–1518. In Jorgensen JH, Pfaller MA, Carroll KC, Funke G, Landry ML, Richter SS, Warnock DW (ed), Manual of clinical microbiology, 11th ed, vol 2 ASM Press, Washington, DC. [Google Scholar]
- 13.Goodrich JS, Miller MB. 2007. Comparison of Cepheid's analyte-specific reagents with BD Directigen for detection of respiratory syncytial virus. J Clin Microbiol 45:604–606. doi: 10.1128/JCM.01325-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ohm-Smith MJ, Nassos PS, Haller BL. 2004. Evaluation of the Binax NOW, BD Directigen, and BD Directigen EZ assays for detection of respiratory syncytial virus. J Clin Microbiol 42:2996–2999. doi: 10.1128/JCM.42.7.2996-2999.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kanwar N, Hassan F, Nguyen A, Selvarangan R. 2015. Head-to-head comparison of the diagnostic accuracies of BD Veritor™ system RSV and Quidel® Sofia® RSV FIA systems for respiratory syncytial virus (RSV) diagnosis. J Clin Virol 65:83–86. doi: 10.1016/j.jcv.2015.02.008. [DOI] [PubMed] [Google Scholar]
- 16.Aslanzadeh J, Zheng X, Li H, Tetreault J, Ratkiewicz I, Meng S, Hamilton P, Tang YW. 2008. Prospective evaluation of rapid antigen tests for diagnosis of respiratory syncytial virus and human metapneumovirus infections. J Clin Microbiol 46:1682–1685. doi: 10.1128/JCM.00008-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fong CK, Lee MK, Griffith BP. 2000. Evaluation of R-Mix FreshCells in shell vials for detection of respiratory viruses. J Clin Microbiol 38:4660–4662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Borg I, Rohde G, Löseke S, Bittscheidt J, Schultze-Werninghaus G, Stephan V, Bufe A. 2003. Evaluation of a quantitative real-time PCR for the detection of respiratory syncytial virus in pulmonary diseases. Eur Respir J 21:944–951. doi: 10.1183/09031936.03.00088102. [DOI] [PubMed] [Google Scholar]
- 19.Hymas WC, Hillyard DR. 2009. Evaluation of Nanogen MGB Alert detection reagents in a multiplex real-time PCR for influenza virus types A and B and respiratory syncytial virus. J Virol Methods 156:124–128. doi: 10.1016/j.jviromet.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 20.Li H, McCormac MA, Estes RW, Sefers SE, Dare RK, Chappell JD, Erdman DD, Wright PF, Tang YW. 2007. Simultaneous detection and high-throughput identification of a panel of RNA viruses causing respiratory tract infections. J Clin Microbiol 45:2105–2109. doi: 10.1128/JCM.00210-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bell JJ, Anderson EJ, Greene WH, Romero JR, Merchant M, Selvarangan R. 2014. Multicenter clinical performance evaluation of BD Veritor™ system for rapid detection of respiratory syncytial virus. J Clin Virol 61:113–117. doi: 10.1016/j.jcv.2014.05.020. [DOI] [PubMed] [Google Scholar]
- 22.Peters RM, Schnee SV, Tabatabai J, Schnitzler P, Pfeil J. 2017. Evaluation of Alere i RSV for rapid detection of respiratory syncytial virus in children hospitalized with acute respiratory tract infection. J Clin Microbiol 55:1032–1036. doi: 10.1128/JCM.02433-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Woo PC, Chiu SS, Seto WH, Peiris M. 1997. Cost-effectiveness of rapid diagnosis of viral respiratory tract infections in pediatric patients. J Clin Microbiol 35:1579–1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen L, Tian Y, Chen S, Liesenfeld O. 2015. Performance of the Cobas(®) influenza A/B assay for rapid PCR-based detection of influenza compared to Prodesse ProFlu+ and viral culture. Eur J Microbiol Immunol 5:236–245. doi: 10.1556/1886.2015.00046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hansen G, Moore J, Hanson K, Hirigoyen D, Dahl A, Zadroga R. 2015. Experience with the Roche Cobas© Liat rapid influenza A/B assay during influenza season: analysis of test performance and qualification on the impact of patient management in the emergency department setting, poster S20. Abstr 31st Clin Virol Symp, Daytona Beach, FL. [Google Scholar]
- 26.Soto M, Sampietro-Colom L, Vilella A, Pantoja E, Asenjo M, Arjona R, Hurtado JC, Trilla A, Alvarez-Martínez MJ, Mira A, Vila J, Marcos MA. 2016. Economic impact of a new rapid PCR assay for detecting influenza virus in an emergency department and hospitalized patients. PLoS One 11:e0146620. doi: 10.1371/journal.pone.0146620. [DOI] [PMC free article] [PubMed] [Google Scholar]