ABSTRACT
OXA-427 is a new class D carbapenemase encountered in different species of Enterobacteriaceae in a Belgian hospital. To study the dispersal of this gene, we performed a comparative analysis of two plasmids containing the blaOXA-427 gene, isolated from a Klebsiella pneumoniae strain and an Enterobacter cloacae complex strain. The two IncA/C2 plasmids containing blaOXA-427 share the same backbone; in the K. pneumoniae strain, however, this plasmid is cointegrated into an IncFIb plasmid, forming a 321-kb megaplasmid with multiple multiresistance regions.
KEYWORDS: carbapenemase, megaplasmids, OXA-427
TEXT
The dissemination of carbapenem resistance in Enterobacteriaceae is a major public health concern, as carbapenem antibiotics are a last-resort therapy for these common pathogens. The new OXA-427 carbapenemase presents a profile of wide resistance to β-lactams, including broad-spectrum penicillins, extended-spectrum cephalosporins, and carbapenems (1, 2). The blaOXA-427 gene was detected in different Enterobacteriaceae species (n = 6) and in different patients (n = 9) hospitalized at a university hospital in Belgium, in different hospital wards, between March 2012 and June 2014 (1); this suggests a high level of interspecies transmission of this resistance gene, probably associated with horizontal gene transfer of a specific plasmid. Recent studies, based on long-read sequencing data, suggest that mobility of resistance genes occurs at multiple nested genetic levels, with transmission of strains between individuals, transfer of plasmids between strains, and transposition of resistance genes between plasmids (3, 4). We report the comparative analysis of two plasmids containing the blaOXA-427 gene, for future study of the potential mechanism of interspecies transmission of the blaOXA-427 gene.
Two OXA-427-producing isolates, one of Klebsiella pneumoniae (KLPN57) and the other of the Enterobacter cloacae complex (ENCL58), were cultured from clinical samples from two different patients, in January 2013 and October 2013, respectively. Genomic DNA was extracted from these isolates and PacBio sequencing was performed at the Genomics Core laboratory (Leuven, Belgium), according to the protocol used by Conlan et al. (5). Genomic DNA was sheared into 10- to 15-kb fragments and converted into SMRTbell template libraries. Each strain was sequenced on a single SMRT cell with the PacBio RSII system, using P6 polymerase binding and C4 sequencing kits. The sequencing coverage for both plasmids ranged from 160-fold to 400-fold coverage. Genome assemblies were performed using HGAP 3 as part of SMRTAnalysis 2.3.0, annotation of the genes was performed by using RAST (http://rast.nmpdr.org), and the functional assignments for the predicted open reading frames (ORFs) were confirmed by using BLASTP with the Protein Data Bank (PDB) from NCBI (http://www.ncbi.nlm.nih.gov). IS finder and ResFinder were used for identification of mobile elements and antibiotic resistance genes, respectively (6, 7). PubMLST was used for multilocus sequence typing (MLST) (8). The plasmid maps were drawn by using SnapGene software.
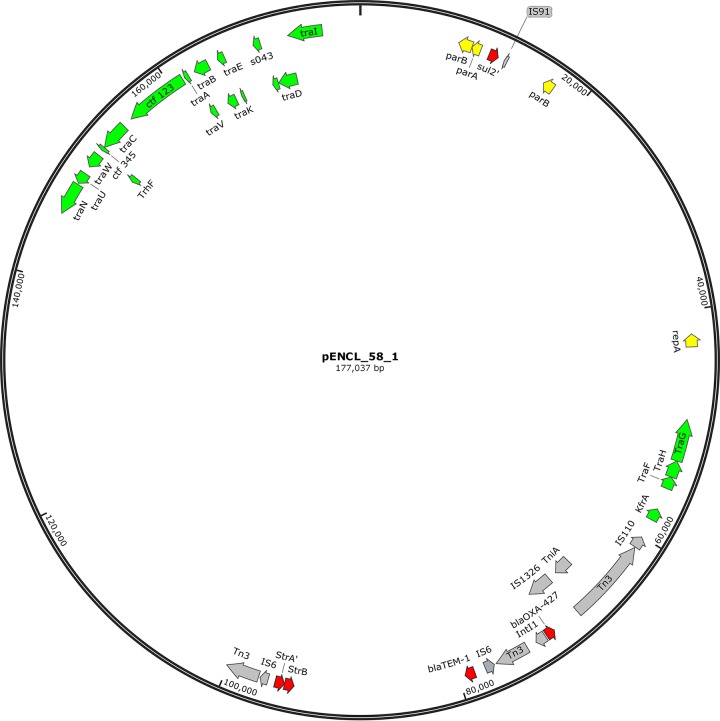
The genomic content of Enterobacter cloacae strain ENCL58 consists of a 5.0-Mb genome with sequence type 171 (ST-171) and three plasmids (177 kb, 121 kb, and 84 kb). The 177,037-bp plasmid pENCL58_01, carrying the blaOXA-427 gene, is an IncA/C2 plasmid of plasmid multilocus sequence type 3, 100% identical to KX869741.1 (1) (Fig. 1). The other two plasmids (IncFIb and IncFII types) carry no known resistance genes. The IncA/C2 plasmid shows overall high levels of homology with other IncA/C2 plasmids found in Enterobacter aerogenes (GenBank accession number FO203354.1; 99% sequence identity and 87% coverage), Klebsiella pneumoniae (GenBank accession number JQ010984.1; 99% sequence identity and 86% coverage), and Salmonella enterica (GenBank accession number KM670336.1; 99% sequence identity and 75% coverage). The plasmid pENCL58_01 contains 223 coding sequences, including three functional antibiotic resistance genes (blaOXA-427, blaTEM-1B, and strB) and two truncated resistance genes (strA and sul2). The blaTEM-1 gene and the strB-strA gene combination are located adjacent to the blaOXA-427 resistance transposon, in the direction away from repA, and both are associated with the presence of IS15 elements, making this a resistance island (RI). In this plasmid, two transfer regions were identified, one located from position 49313 to position 57324 and including 3 tra genes and kfrA (for plasmid maintenance) and one located from position 145665 to position 173831 and including 12 tra genes and trhF.
FIG 1.
Circular representation of pENCL58_01, harboring bla0XA-427. Only genes for partitioning/replication (yellow), conjugative transfer (green), antibiotic resistance (red), and mobility (gray) are depicted. Prime symbols indicate truncated genes.
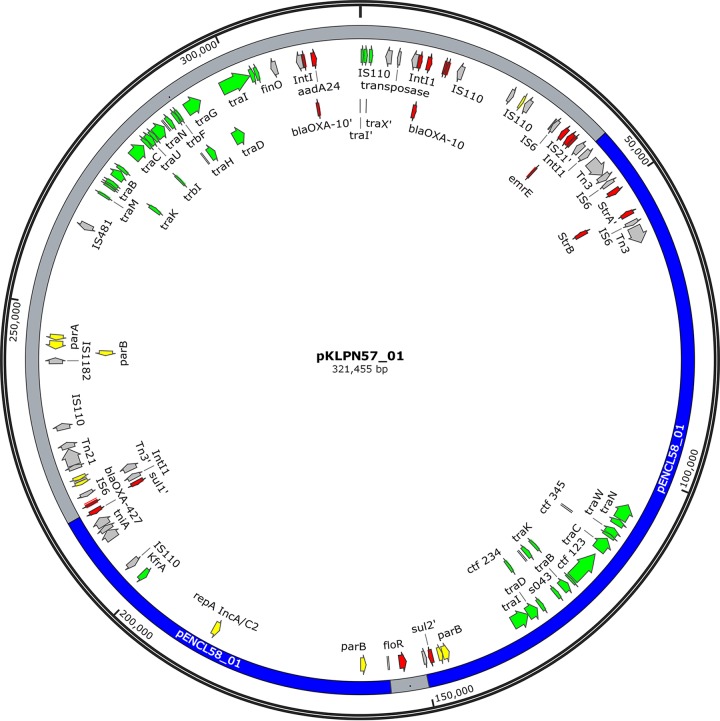
The assembly of the Klebsiella pneumoniae strain KLPN57 reads resulted in a genome of 5.23 Mb with ST-1 and three plasmids (321 kb, 175 kb, and 77 kb). The blaOXA-427 gene was identified on the 321,455-bp plasmid pKLPN57_01, which carries 417 predicted coding sequences (Fig. 2). The other plasmids are of type IncFIb, with resistance genes blaCTX-M-15, blaTEM-1B, aph(3′)-Ia, aadA2, and sul1, and type IncFII, with qnrS1.
FIG 2.
Circular representation of pKLPN57_01, harboring bla0XA-427. The plasmid parts identical to those in pENCL58_01 (>99% similarity) are indicated in blue. Only genes for partitioning/replication (yellow), conjugative transfer (green), antibiotic resistance (red), and mobility (gray) are depicted. Prime symbols indicate truncated genes.
The plasmid pKLPN57_01 contains resistance genes for aminoglycoside resistance (aadA24, aacA4, and strB), β-lactam resistance (blaOXA-10, blaTEM-1B, and blaOXA-427), chloramphenicol resistance (floR), and sulfonamide resistance (sul1). Five additional resistance genes (strA, blaOXA-2, sul2, blaOXA-10, and aacA4) are truncated and potentially dysfunctional. Interestingly, two types of repA genes, coding for plasmid replication proteins, were detected on that plasmid. These repA genes belong to two distinct plasmid incompatibility groups, namely, IncA/C2 and IncFIb. Further analysis confirmed that pKLPN57_01 is composed of a novel plasmid belonging to the IncFIb group and an IncA/C2 plasmid highly similar to pENCL58_01, which was identified in Enterobacter cloacae ENCL58 (Fig. 3). Additionally, the plasmid carries two parts of a repA gene related to colW separated by the IncA/C2 region, together constituting the complete replication gene. Based on the presence of different repA genes and the large number of mobile genetic elements, the pKLPN57_01 plasmid is in fact a megaplasmid of 321 kb and could be a result of multiple fusions of different plasmids and/or mobile elements. The IncFIb region showed the highest level of homology with a K. pneumoniae plasmid (GenBank accession number CP021541.1; 99% identity and 68% coverage) and the colW region with a Achromobacter xylosoxidans subsp. denitrificans plasmid (GenBank accession number HF679279.1; 100% identity and 37% coverage). Unfortunately, we were not able to identify the sources of these plasmid elements.
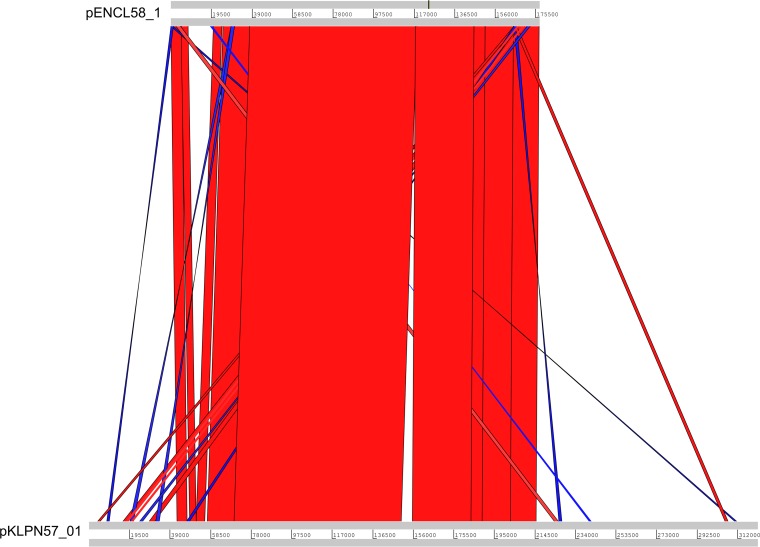
FIG 3.
Pairwise comparison of pENCL58_01 (upper sequence) and pKLPN57_01 (lower sequence). The intI1 gene of pENCL58_01 is used as a starting point. At the starting position, the homologous plasmid is disrupted and integrated into the pKLPN57_01 megaplasmid. Connecting bars represent regions with BLASTN matches of >99.9% in the same (red) and inverted (blue) orientations, visualized with the Artemis Comparison Tool (13).
The genetic environment downstream of the blaOXA-427 gene, characterized by IS1326 and a Tn3 family transposon, is identical in the two plasmids. Upstream of the blaOXA-427 gene, however, additional resistance genes are present in pKLPN57_01, compared to pENCL58_01 (Fig. 1 and 2).
In Fig. 3, the alignment of pENCL58 and pKLPN57 is shown. In the pKLPN57_01 megaplasmid, the IncA/C2 region is flanked by identical class I integron integrase genes, intI1, at both sites. At the 5′ site, the intI1 gene is fused with a resistance integron. At the 3′ site, the intI1 gene of IncA/C2 is fused with a partial colW-like repA gene by a transposase gene of the Tn3 family and an IS6 insertion sequence. In both regions of fusion, there is an abundance of Tn3 family transposases and IS6 and IS110 insertion sequences. The region homologous to pENCL58 is divided into two parts by a transposon flanked on both sides by IS91, carrying the resistance gene floR.
We conclude that the megaplasmid in the K. pneumoniae isolate is a result of cointegration of an IncA/C2-type plasmid harboring blaOXA-427 with an IncF1b-type plasmid. The ability of Tn3 and insertion sequences to mediate cointegration of plasmids is well known (9). Plasmid cointegrates have been reported previously for carbapenemase-producing Enterobacteriaceae strains (5, 10); they showed IncFIa-type plasmid backgrounds and IS26 insertion sequences flanking the regions of fusion. To our knowledge, however, cointegrates formed from large resistance plasmids with sizes of >300 kb have not been reported previously. The plasmid cointegration seems to represent an important molecular pathway for interspecies transfer of plasmids and their resistance genes. Cointegration with disruption of replicons, as observed in pKLPN57_01, may allow plasmid incompatibility to be overcome (11). Next, cointegration with a plasmid that has coevolved in K. pneumoniae may provide the transferred plasmid with specific conjugation and restriction modification machinery (12). The cointegration of plasmids into pKLPN57_01 has probably taken place in recent years. Future studies will need to clarify whether megaplasmids of the size presented in our study are persistent or represent a temporary initial stage in the evolution of antibiotic resistance plasmids.
Accession number(s).
The annotated plasmid sequences of pKLPN57_01 and pENCL58_01 were deposited in DDBJ/EMBL/GenBank under accession numbers LT882698 and LT882699, respectively.
ACKNOWLEDGMENTS
We thank Artur Sabat (University of Groningen, University Medical Center Groningen, Groningen, the Netherlands) for reviewing the sequence analysis and Y. Pfeifer (Robert Koch Institute) for kindly providing the Escherichia coli j53 azide-resistant strain.
REFERENCES
- 1.Bogaerts P, Naas T, Saegeman V, Bonnin RA, Schuermans A, Evrard S, Bouchahrouf W, Jove T, Tande D, de Bolle X, Huang TD, Dortet L, Glupczynski Y. 2017. OXA-427, a new plasmid-borne carbapenem-hydrolysing class D β-lactamase in Enterobacteriaceae. J Antimicrob Chemother 72:2469–2477. doi: 10.1093/jac/dkx184. [DOI] [PubMed] [Google Scholar]
- 2.Bogaerts P, Thierry N, Evrard S, Bouchahrouf W, Saegeman V, Lasserre C, Tande D, De Bolle X, Huang TD, Glupczynski Y. 2015. OXA-427, a new plasmidic ESBL class D OXA-carbapenemase recovered from Enterobacteriaceae clinical isolates. Abstr ECCMID 2015, abstr P1307. [Google Scholar]
- 3.Sheppard AE, Stoesser N, Wilson DJ, Sebra R, Kasarskis A, Anson LW, Giess A, Pankhurst LJ, Vaughan A, Grim CJ, Cox HL, Yeh AJ, Sifri CD, Walker AS, Peto TE, Crook DW, Mathers AJ. 2016. Nested Russian doll-like genetic mobility drives rapid dissemination of the carbapenem resistance gene blaKPC. Antimicrob Agents Chemother 60:3767–3778. doi: 10.1128/AAC.00464-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mathers AJ, Stoesser N, Sheppard AE, Pankhurst L, Giess A, Yeh AJ, Didelot X, Turner SD, Sebra R, Kasarskis A, Peto T, Crook D, Sifri CD. 2015. Klebsiella pneumoniae carbapenemase (KPC)-producing K. pneumoniae at a single institution: insights into endemicity from whole-genome sequencing. Antimicrob Agents Chemother 59:1656–1663. doi: 10.1128/AAC.04292-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Conlan S, Thomas PJ, Deming C, Park M, Lau AF, Dekker JP, Snitkin ES, Clark TA, Luong K, Song Y, Tsai YC, Boitano M, Dayal J, Brooks SY, Schmidt B, Young AC, Thomas JW, Bouffard GG, Blakesley RW, Mullikin JC, Korlach J, Henderson DK, Frank KM, Palmore TN, Segre JA. 2014. Single-molecule sequencing to track plasmid diversity of hospital-associated carbapenemase-producing Enterobacteriaceae. Sci Transl Med 6:254ra126. doi: 10.1126/scitranslmed.3009845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Siguier P, Perochon J, Lestrade L, Mahillon J, Chandler M. 2006. ISfinder: the reference centre for bacterial insertion sequences. Nucleic Acids Res 34:D32–D36. doi: 10.1093/nar/gkj014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carattoli A, Zankari E, Garcia-Fernandez A, Voldby Larsen M, Lund O, Villa L, Moller Aarestrup F, Hasman H. 2014. In silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob Agents Chemother 58:3895–3903. doi: 10.1128/AAC.02412-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Larsen MV, Cosentino S, Rasmussen S, Friis C, Hasman H, Marvig RL, Jelsbak L, Sicheritz-Ponten T, Ussery DW, Aarestrup FM, Lund O. 2012. Multilocus sequence typing of total-genome-sequenced bacteria. J Clin Microbiol 50:1355–1361. doi: 10.1128/JCM.06094-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shapiro JA. 1979. Molecular model for the transposition and replication of bacteriophage Mu and other transposable elements. Proc Natl Acad Sci U S A 76:1933–1937. doi: 10.1073/pnas.76.4.1933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chavda KD, Chen L, Jacobs MR, Rojtman AD, Bonomo RA, Kreiswirth BN. 2015. Complete sequence of a blaKPC-harboring cointegrate plasmid isolated from Escherichia coli. Antimicrob Agents Chemother 59:2956–2959. doi: 10.1128/AAC.00041-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lin DL, Ramirez MS, Tran T, Tolmasky ME. 2013. A cointegrate-like plasmid that facilitates dissemination by conjugation of the extended-spectrum β-lactamase CTX-M-17. Antimicrob Agents Chemother 57:5191–5192. doi: 10.1128/AAC.01365-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ramirez MS, Traglia GM, Lin DL, Tran T, Tolmasky ME. 2014. Plasmid-mediated antibiotic resistance and virulence in Gram-negatives: the Klebsiella pneumoniae paradigm. Microbiol Spectr 2:1–15. doi: 10.1128/microbiolspec.PLAS-0016-2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carver TJ, Rutherford KM, Berriman M, Rajandream MA, Barrell BG, Parkhill J. 2005. ACT: the Artemis Comparison Tool. Bioinformatics 21:3422–3423. doi: 10.1093/bioinformatics/bti553. [DOI] [PubMed] [Google Scholar]