SUMMARY
Helminth infection is known for generating large amounts of poly-specific IgE. Here we demonstrate that innate-like B1 cells are responsible for this IgE production during infection with the nematode parasites Nippostrongylus brasiliensis and Heligmosomoides polygyrus bakeri. In vitro analysis of B1 cell immunoglobulin class switch recombination to IgE demonstrated a requirement for anti-CD40 and IL-4 that was further enhanced when IL-5 was added or when the B1 source was helminth infected mice. An IL-25-induced upregulation of IgE in B1 cells was also demonstrated. In T cell-reconstituted RAG1−/− mice, N. brasiliensis clearance was enhanced with the addition of B2 cells in an IgE-dependent manner. This enhanced clearance was impeded by reconstitution with IgE sufficient B1 cells. Mucosal mast cells mediated the B2 cell enhancement of clearance in the absence of B1 cells. The data support B1 cell IgE secretion as a regulatory response exploited by the helminth.
In Brief
Martin et al. show that B1 cell IgE is induced during Th2 helminth infections by IL-25. This B1 cell IgE blocks parasite clearance through inhibition of mucosal mast cell activation by B2 cell IgE.

INTRODUCTION
Immunoglobulin E (IgE) is an evolutionarily conserved immunoglobulin that is well known for causing the symptoms of atopic disease. This antibody class, despite having a half-life of less than a day in plasma, can persist for weeks to months when bound to cell surface FcεRI, making it a long-lasting “gate-keeper” particularly with respect to triggering mast cells (MCs) or basophils (Oettgen, 2016). Specific IgE responses directed against innocuous particles, such as pollen, cat dander, or peanut proteins, can result in allergic disease. IgE-mediated responses range from mild to severe. They can be either site directed, such as allergic rhinitis, atopic dermatitis, urticaria, and asthma, or systemic, as in anaphylactic shock. IgE+ plasma cells generated in the germinal centers (GCs) that produce high-affinity IgE to antigens are purported to come from bone marrow (BM)-derived B cells or B2 cells through immunoglobulin class switch recombination (CSR) and somatic hyper mutation (SHM). In contrast, memory IgE responses are generated from IgG1+ memory B cells (Oettgen, 2016).
B1 cells develop early in ontogeny, prior to the first hematopoietic stem cell (HSC), and are derived initially from the fetal yolk sac and then from the fetal liver (Savage and Baumgarth, 2015). They are delineated from B2 cells by the expression of CD11b and absence of CD23. They reside primarily in the pleural and peritoneal body cavities of mice and traffic to the draining lymph nodes (LNs), spleen, and mucosal sites upon activation (Yenson and Baumgarth, 2014; Savage and Baumgarth, 2015; Waffarn et al., 2015). B1 cells are important immune effectors and regulators of adaptive immunity that bridge the innate and adaptive immune systems. The B cell receptor (BCR) repertoire in these cells is enriched for poly-specific receptors encoded in the germline with low affinities to a broad range of antigens (Baumgarth et al., 2005). B1 cells are essential Immunoglobulin M (IgM) secretors and have additionally been shown to be the definitive source of “natural” IgM. As immune effectors, they also secrete Immunoglobulin A (IgA) at mucosal sites. However, only a few reports have demonstrated IgE production by B1 cells (Takatsu et al., 1992; Vink et al., 1999; Perona-Wright et al., 2008; Savage and Baumgarth, 2015). The importance of parasite-specific IgE in controlling infection is controversial, yet there is evidence to support IgE-mediated clearance of phylogenetically distinct helminths such as Schistosoma mansoni and Trichnelia spiralis (Joseph et al., 1983; Gurish et al., 2004; Oettgen, 2016). These parasites strongly promote IgE synthesis (Wu and Zarrin, 2014). In this work, we showed that poly-specific IgE made by B1 cells was responsible for reduced MC degranulation by mechanism of IgE saturation of FcεRI that was initially proposed by Bazaral et al. (1973).
Nippostrongylus brasiliensis and Heligmosomoides polygyrus bakeri are Th2-inducing helminth parasites of mice similar to the human hookworms, Necator americanus and Ancylostoma duodenale (de Silva et al., 2003). Wild-type (WT) mice are able to clear these infections in a T cell-dependent manner, relying on the cytokines IL-13 and IL-4 for the “weep and sweep” of intestinal helminth clearance (Madden et al., 2002; Finkelman et al., 2004). This refers to the increased mucus production, goblet cell hyperplasia, and enteric nerve stimulation associated with intestinal parasite expulsion(Camberis et al., 2003; Finkelman et al., 2004). In response to these intestinal helminths, we demonstrated that B1 cells class switch to IgE. In addition, the signals that drive B1 cells to IgE production and the functional relevance of B1 cell IgE in parasite-host interactions are shown.
RESULTS
B1 Cells Make Large Amounts of IgE in Response to Helminth Infection
In our laboratory, we generated mice that lack BM-derived B cells, or B2 cells (Gibb et al., 2011), the ADAM10Tg mouse. This is due to overexpression of ADAM10 at the common lymphoid progenitor stage that leads to improper NOTCH signaling and loss of B cell development. As this defect is restricted to the BM, the B1 cell compartment is intact (Gibb et al., 2011). To study the B1 cell antibody response during helminth infection, we inoculated ADAM10Tg and WT control mice with N. brasiliensis or H. polygyrus bakeri infective third-stage larvae (L3). We measured the amount of IgE and Immunoglobulin G1 (IgG1) antibody in the serum in the naive state and on day 14 post infection. Interestingly, there was no significant difference in IgE production between WT and ADAM10Tg mice infected with either helminth (Figures 1A and 1B). This suggested that B1 cells produced significant levels of IgE post helminth infection in the absence of B2 cells. ADAM10Tg mice have an increased number of immature myeloid cells throughout the organs and circulation due to the defect in hematopoiesis (Gibb et al., 2011). These cells were selectively depleted utilizing gemcitabine (GEM) to assess whether they played a role in enhancing B1 cell IgE (Sinha et al., 2007; Saleem et al., 2012). N. brasiliensis-infected and GEM-treated mice exhibited no change in IgE levels (Figure 1A), indicating an inconsequential role of immature myeloid cells on B1 cell IgE antibody production. Myeloid-derived suppressor cell (MDSC) depletion was confirmed by flow cytometry (Figure S1A). To ensure B1 IgE production was not altered due to ADAM10 overexpression, we sorted B1 cells (Figure S1B), and ADAM10 message was not different, as measured by qPCR (Figure S1C).
Figure 1. B1 Cell IgE Is Induced with T Cell Help during Helminth Infection.
(A–D) Total serum IgE (closed circles) (A and C) and IgG1 (open circles) (A and D) was measured on day 0 (PB) and 14 post N. brasiliensis inoculation in ADAM10Tg (A10Tg, orange) and wild-type (WT; magenta) mice. Where indicated, GEM depletion of MDSCs was performed.
(B) Total serum IgE was measured on days 10 and 15 post H. polygyrus bakeri inoculation.
(C and D) Mice were T cell depleted or treated with control IgG mAb followed by N. brasiliensis infection.
*p < 0.05, **p < 0.01, ***p < 0.001. If not indicated, the comparison was not significant between groups. The error bars depict SEM. (A) was representative of three independent experiments. (B), (C), and (D) were representative of two independent experiments. One-way ANOVA with a Tukey post hoc was used to compare all IgE or all IgG1 groups in (A)–(D).
B1 Cell IgE Production during Helminth Is T Cell Dependent
To assess whether helminth-induced B1 cell IgE production required T cells, we depleted both CD8+ and CD4+ T cells with GK1.5 and 2.43 antibodies, respectively (Saleem et al., 2012). Both WT and ADAM10Tg mice had significantly reduced IgE production after T cell depletion (Figure 1C). WT mice had significantly reduced IgG1 after T cell depletion that was not seen in ADAM10Tg mice (Figure 1D). IgG1 levels were significantly reduced in ADAM10Tg mice (Figures 1A and 1D). The reason for this is not known; however, the ADAM10Tg mice make IgE that is equivalent or more than WT levels (Figures 1A and 1C). A baseline level of IgE and IgG1 remained in both WT and ADAM10Tg mice despite the loss of T cells (Figures 1C and 1D). This could represent a small amount of T cell-independent IgE production.
B1 Cell Antibody Production after NP-KLH Immunization Is Not NP Specific
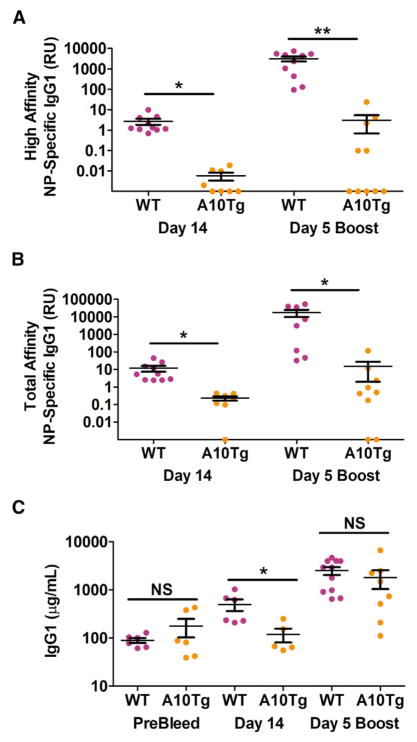
We next examined the antigen specificity of B1 cell IgG1 in the ADAM10Tg mouse. Mice were immunized intraperitoneally (i.p.) with NP32-KLH in alum and both high-affinity IgG1 antibody and total specific IgG1 antibody was assessed in serum by enzyme-linked immunosorbent assay (ELISA) (Smith et al., 1997). On day 14, ADAM10Tg mice have almost undetectable nitrophenol (NP)-specific IgG1 antibody (Figures 2A and 2B) in addition to having measurable total IgG1 that was significantly less than WT (Figure 2C). A boost at day 28 induced increased high-affinity IgG1, total specific IgG1, and total IgG1 in WT mice (Figures 2A–2C). ADAM10Tg mice, while having a large spread in total IgG1, did not increase significantly after the boost, even from the naive bleed (Figure 2C), and though some animals developed a limited amount of NP-specific IgG1, it was three to four orders of magnitude different than the WT (Figures 2A and 2B).
Figure 2. B1 Cell Antibody Responses to NP-KLH Are Not Specific.
(A–C) ADAM10Tg (A10Tg, orange) and WT (magenta) mice were immunized i.p. with NP32-KLH in alum. Serumlevels of high-affinity NP-specific (NP4BSAbinding) (A), total NP-specific IgG1 (NP25BSA-binding) (B), and total IgG1 (C) were measured by ELISA on day 0, 14, and day 5 post boost. NS, not significant; *p < 0.05, **p < 0.01. The error bars depict SEM. Significance was obtained using an unpaired Student’s t test to compare WT and A10Tg. Data are from three independent experiments.
B1 Cell-Derived IgE Fails to Induce MC Degranulation
A major function of antigen-specific IgE is to induce degranulation through FcεRI on MCs and basophils. To measure the antigen specificity of B1 cell IgE as well as examine the ability of B1 cell antibody to induce degranulation, we used a model of active cutaneous anaphylaxis (ACA) (Evans et al., 2014). MC degranulation was induced by crosslinking OVA-specific IgE molecules bound to FcεRI on skin MCs; the resulting dye leakage creates a blue spot. This degranulation can also result from and be enhanced by IgG complexes bound to FcγRIII (Strait et al., 2006). Measurement of the area of the spot and its content of Evans blue in WT mice indicated significantly increased MC degranulation compared to ADAM10Tg mice (Figures 3A and 3B) despite an equivalent total IgE (Figure 3C) and IgG1 (Figure 3D) in the serum of both groups of mice. This indicated that neither the B1 cell IgE nor the B1 cell IgG1 induced with this antigen in alum was OVA specific. Further, OVA-specific IgE was confirmed as significantly less by ELISA in the ADAM10Tg mice (Figure 3E). Next, blocking of specific IgE to FcεRI by helminth-induced B1 cell antibody was tested in a model of passive cutaneous anaphylaxis (PCA). ADAM10Tg and WT mice were infected with N. brasiliensis L3, and PCA reaction was assessed 21 days later (Figures 3F and 3G). All uninfected control mice had no statistical differences in degranulation, indicating that skin MC activation was normal (Figures 3F and 3G). Additionally, numbers of MCs in the skin between ADAM10Tg and WT mice were equivalent (data not shown), but the amount of MC degranulation seen in both ADAM10Tg and WT mice was significantly reduced by helminth infection (Figures 3F and 3G) supporting a helminth-induced blockage of MC degranulation. Total IgE levels in these mice were equivalent and increased during helminth infection (Figure 3H). Total IgG1 levels were elevated in WT mice after helminth infection, but not in ADAM10Tg mice (Figure 3I). This is similarly seen in previous infections (Figures 1A and 1C). To illustrate that this PCA suppression was IgE dependent, we utilized IgE deficient mice (IgE−/−). After N. brasiliensis infection, there was no evidence of suppression of the PCA reaction in IgE−/− mice (Figures 3F and 3G).
Figure 3. Antibody Produced by B1 Cells Blocks Antigen-Specific IgE-Mediated MC Degranulation.
(A–E) ADAM10Tg (A10Tg, orange) and WT (magenta) mice were immunized with OVA in alum for an ACA model. Post i.d. OVA challenge and i.v. injection with Evan’s blue dye solution, MC degranulation was measured by dye release into the back skin as surface area of the spot (A) and dye extraction (B) (minus control spot). Total serum IgE (C), serum IgG1 (D), and OVA-specific IgE (E) (dotted line depicts limits of detection) antibody were as measured from sera just prior to ACA test. PCA model utilized either naive or day 21 post-N. brasiliensis (Nb) mice.
(F and G) Surface area of the spot (F) or dye extraction (G) was measured in WT, A10Tg, or IgE deficient (IgE−/−) mice.
(H and I) Total serum IgE (H) and serum IgG1 (I) antibody were measured from sera collected just prior to PCA test.
*p < 0.05, **p < 0.01, ***p < 0.001. If not indicated, the comparison was not significant between groups. Error bars depict SEM. Significance was obtained as follows: Student’s t test for (A)–(E), Kruskal-Wallis test with a Dunn’s multiple comparison for (F) and (G), and a one-way ANOVA with a Tukey post hoc test for (H) and (I).
B1 Cells Are Primed to Make IgE during Helminth Infection
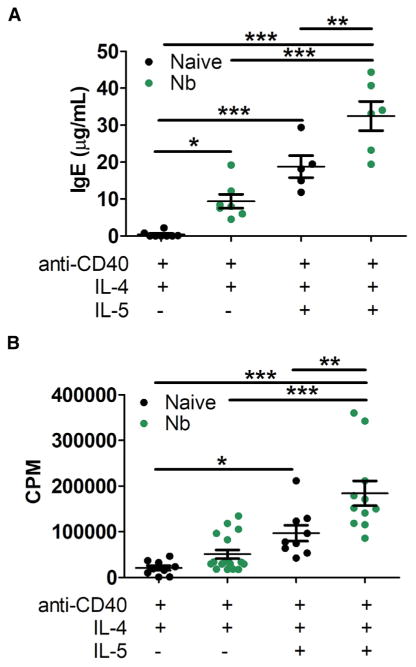
To investigate the signals necessary for the B1 cell to class switch to IgE, we sorted peritoneal cavity B1 from naive or N. brasiliensis-infected mice (Figure S1B). Naive B1 cells make little IgE when treated with anti-CD40 and IL-4, but the addition of IL-5, a known B1 proliferative agent (Erickson et al., 2001; Takatsu, 2011), significantly increased IgE production (Figure 4A). Interestingly, B1 cells from N. brasiliensis-infected mice made significantly more IgE than from B1 cells from naive mice, after anti-CD40 and IL-4 treatment. This did not correlate with increased cell proliferation (Figures 4A and 4B). When IL-5 was added, B1 cells from N. brasiliensis-infected mice had increased sensitivity to IL-5-induced proliferation, and this correlated to increased IgE production as well (Figures 4A and 4B). Overall, this indicated that B1 cell IgE was stimulated in vitro by similar signals as B2 cell-induced IgE with respect to anti-CD40 and IL-4; however, other signals may be priming B1 cells for increased IgE production during infection with N. brasiliensis. Since B1 cells are known to secrete large amounts of IgM, IgM production by both B1 and B2 cells was compared in culture. B1 cells from N. brasiliensis-infected mice treated with anti-CD40, IL-4, and ± IL-5 made significantly more IgM than similarly treated B2 cells from infected mice, reinforcing that B1 cells were being examined (Figure S2A).
Figure 4. IL-5 Enhanced IgE Production in B1 Cells.
B1 cells were sorted (CD23−B220int, CD11bint, CD138−) (Figure S1B shows gating) from the peritoneal lavage of mice infected with N. brasiliensis (day 14) or naive WT mice.
(A) 30,000 cells/mL were cultured with anti-CD40, IL-4, ± IL-5. Supernatants were harvested on day 9 of culture for total IgE ELISA.
(B) For proliferation, 60,000 cells/mL were cultured for 72 hr, followed by the addition of 1 μCi/well [H3]-thymidine with cell harvest at 24 hr later.
CPM, counts per minute. *p < 0.05, **p < 0.01, ***p < 0.001. Error bars depict SEM. Significance was obtained using a one-way ANOVA with a Tukey post hoc test.
The Alarmin IL-25, but Not IL-33, Enhances B1 Cell IgE Production during Helminth Infection
IL-25 production by intestinal tuft cells has been shown to be important for cytokine production by ILC2 and Th2 cells (von Moltke et al., 2016; Gerbe et al., 2016). Fort et al. (2001) reported that i.p. IL-25 injection induced IgE production at 10 days. These data, as well as the importance of IL-25 release by intestinal tuft cells during helminth infection, led to an examination of the effects of IL-25 on B1 cell IgE. B1 cells from N. brasiliensis-infected mice, but not naive mice, made significantly more IgE when treated with anti-CD40, IL-4, and IL-25 (in the range of 1 to 100 ng/mL) as compared to cells treated with anti-CD40 and IL-4 alone (Figures 5A and 5B). IL-25 plus IL-5 did not further increase B1 cell IgE production from either naive or N. brasiliensis-infected B1 cells (Figures 5A and 5C). In addition, IL-25 induced a moderate amount of proliferation in B1 cells treated with anti-CD40 and IL-4 (Figure 5D), but the increased baseline proliferation seen with IL-5, anti-CD40, and IL-4 treated B1 cells was not further augmented with IL-25 (Figure 5E). The increase in IgE seen after treatment with IL-25 in B1 cells from N. brasiliensis-infected mice was demonstrated by quantitative RT-PCR (qRT-PCR) analysis of secreted IgE expression (Figure 5F). Additional controls show that IL-25 alone did not induce detectable levels of IgE in culture (Figures S2G–S2I).
Figure 5. IL-25 Enhanced B1 Cell IgE Production Only from Helminth-Infected Mice.
B1 cells were sorted (Figure S1B) from mice infected with N. brasiliensis or naive mice. They were then cultured with anti-CD40, IL-4, ± IL-5 and with increasing doses of IL-25.
(A) Fold change in IgE as compared to no IL-25 was assessed.
(B and C) Total IgE from no IL-5 in cultures (B) and IL-5 added to cultures (C).
(D and E) Proliferation of B1 cells from mice infected with N. brasiliensis was examined as in Figure 4 after addition of IL-25, without (D) or with (E) IL-5. CPM, counts per minute.
(A–F) Secreted Ighe message was assessed after 4 days anti-CD40 and IL-4 culture in B1 cells ± 30 μg IL-25 and normalized to Actb from N. brasiliensis infected mice. Statistics: fold change in B1 cells from naive (black open circle, solid line) was compared to fold change in N. brasiliensis-infected mice (Nb) (black filled circle, dotted line) by unpaired Student’s t test (A); comparison was made between no IL-25 and dosages of IL-25 added to culture using an unpaired Student’s t test (B and C) and a Mann-Whitney, non-parametric comparison (D–F).
*p < 0.05, ***p < 0.001. If not indicated, the comparison was not significant. n > 7 mice per group in (A)–(E), n = 3 pooled mouse samples per group in (F). Error bars depict SEM. Experiments are the products of at least two independent repeats for all groups.
Since IgM is typically secreted by B1 cells, the regulation of IgM levels by IL-25 was examined. B1 cells from naive mice treated with anti-CD40 and IL-4 produced IgM that was not affected by the addition of IL-25, with or without IL-5 (Figures S2B and S2C). IL-5 increased IgM production overall (Figures S2B and S2C). Inversely, B1 cells from mice infected with N. brasiliensis that were treated with anti-CD40 and IL-4 had lower IgM when treated with IL-25 compared to naive. IL-25 also had no significant effect on IgG1 levels by ELISA (Figures S2D and S2E) or qRT-PCR (Figure S2F). To see whether this pathway was active only in B1 cells, we examined IgE production in B2 cells from both naive and N. brasiliensis-infected mice. IgE, IgG1, and IgM were not significantly altered beyond the addition of anti-CD40 and IL-4 (Figures S3A–S3F). In addition, IL-25 alone did not induce IgE, IgG1, or IgM secretion by B2 cells (Figures S3A–S3F).
Komai-Koma et al. recently showed that B1 cells proliferate after daily i.p. injection with IL-33 (Komai-Koma et al., 2011) through production of IL-5 that then upregulated ST2 on B1 cells (Ahmed and Koma, 2015). To test the effect of IL-33 on B1 cell IgE production, we injected IL-33 i.p. into WT mice and compared sorted B1 cells to B1 cells from naive mice. IL-33 injected i.p. did not increase B1 cell IgE or IgG1 (Figures S4D and S4E). In addition, in vitro IL-33 treatment also failed to increase IgE or IgG1 production, either with or without IL-5 pre-treatment (Figures S4D and S4E). Finally, ex vivo IL-33 treatment did not enhance IgE secretion by B1 cells isolated from mice infected with N. brasiliensis but did increase proliferation, as described (Figures S4A–S4C). These data suggest that IL-33 does not enhance IgE production in B1 cells.
IL-25-Dependent B1 Cell-Produced IgE Blocks Enhanced B2 Cell-Produced IgE-Mediated Suppression of N. brasiliensis
To examine the physiological importance of B1 cell-produced IgE during infection with N. brasiliensis, we turned to a RAG1−/− mouse model that lacks functional T cells and B cells, including B1 cells (Paciorkowski et al., 2000). All mice were reconstituted with WT CD4+ T cells and further reconstituted with B1 cells alone, B2 cells alone, or B1 and B2 cells. One week after reconstitution, mice were inoculated with N. brasiliensis L3. The total number of lymphocytes that are reconstituted after 1 week was determined by flow cytometry and compared to that of a WT mouse (Figures S5A–S5F). Proper reconstitution was confirmed by flow cytometry after N. brasiliensis infection (Figure S5G). The level of infection was monitored by measuring fecal egg levels (Figures 6A and 6B). Both B1 cell-only and CD4+ T cell-control reconstituted mice had similar infection levels, demonstrating that B1 cells did not alter parasitic clearance mediated directly by CD4+ T cells (Figures 6A and 6B). Intriguingly, B2 cell-only reconstituted mice showed significantly decreased egg production compared to the other groups, and B1/B2 cell-reconstituted mice showed greater egg production compared to the B2 cell-only group (Figures 6A and 6B). This indicated that B1 cells were hindering B2-mediated clearance. To test whether this was B1 cell IgE mediated, we reconstituted mice with B1 cells from IgE−/− mice and WT B2 cells. These IgE−/− B1 cells did not inhibit the B2-mediated suppression of egg production (Figures 6A and 6B). To further show that the B2-mediated suppression of egg production was IgE dependent, we reconstituted RAG1−/− mice with IgE−/− B2 cells. These B2-IgE−/− mice exhibited considerably greater egg production than the mice reconstituted with WT B2 cells (Figures 6A and 6B). The total IgE levels in serum were similar between all three groups that had been reconstituted with WT B cells and were only significantly reduced in the IgE−/− mice (Figure 6C).
Figure 6. IL-25-Mediated B1 Cell IgE Blocks Parasite Clearance by B2 Cell IgE in Reconstituted Mice.
RAG1−/− mice were reconstituted with the indicated cells as described in Experimental Procedures.
(A) Eggs per gram (EPG) of feces were determined over the time course of infection with N. brasiliensis L3.
(B) Day 7 EPG.
(C) Total serum IgE was measured in serum by ELISA on day 14.
(D and E) 35 days post infection, an ACA test was induced using NES and spot size (D) and dye extraction (E) from skin was assessed.
(F) EPG was measured over the time course of infection.
(G) Day 7 EPG.
*p < 0.05, **p < 0.01, ***p < 0.001. If not indicated, the comparison was not significant. For (A)–(E), n > 7 mice per group in all groups except T cell alone, which n = 4 mice per group and data are the product of four independent repeats. For (G) and (F), n = 4 mice per group, and data are representative of two independent repeats. Error bars depict SEM. Significance was determined as follows: one-way ANOVA with a Tukey post hoc for (B), (D), (E), and (G), and a Kruskal-Wallis non-parametric test with a Dunn’s post hoc for (C).
These mice were maintained for 35 days after N. brasiliensis inoculation and then injected intradermally (i.d.) with N. brasiliensis excretory-secretory extract (NES) in a model of ACA to determine whether B1 cell IgE provided protection against IgE-mediated helminth-specific MC degranulation. We observed that the mice reconstituted with WT B2 cells generated an ACA reaction (Figures 6D and 6E). The mice that were reconstituted with WT B1 and WT B2 had reduced ACA responses reflective of their decreased parasite clearance (Figures 6D and 6E). Mice reconstituted with IgE−/− B2 cells from N. brasiliensis-infected mice failed to induce ACA responses. Mice that had been reconstituted with both IgE−/− B1 cells and WT B2 cells generated ACA skin reactions similar to WT B2 cells alone, demonstrating the importance of B1 cell-derived IgE in blocking B2 cell induced degranulation (Figures 6D and 6E).
To examine whether this B1 cell-derived IgE was dependent on the cytokine IL-25, we reconstituted RAG1−/− mice with either WT B2 cells and WT B1 cells or WT B2 cells and B1 cells from IL-25 receptor deficient mice (IL-25R−/−). These were both compared to mice with just WT B2 cells. As the IL-25R−/− mice were on a BALB/c background, this experiment used the BALB/c RAG1−/− mice. IL-25R−/− B1 cells were unable to hinder the WT B2 cell-mediated clearance (Figures 6F and 6G), strongly supporting the in vitro cytokine data for IL-25 enhancement of B1 cell IgE production.
B2 Enhancement of Helminth Clearance Is MC Dependent
To further elucidate the mechanism behind B2 cell IgE enhancement of helminth clearance, we infected T cell plus B2 cell or T cell only control reconstituted RAG1−/− mice with N. brasiliensis L3. ELISAs showed significant increases in both MC-produced histamine and MC protease-1 (MCPT-1) in the mucus of the jejunum on day 7 in B2 cell reconstituted mice (Figures 7A and 7B). qRT-PCR analysis of total jejunal tissue demonstrated that B2 cell reconstitution significantly increased MC protease genes Mcpt1, Mcpt2, Cpa3, and Cma1, consistent with a role for MCs in suppression of N. brasiliensis egg production (Figure 7C; Table S1). Expression of the Th2-associated genes Il4, Il5, Il13, Il6, and Il9 were similar between T cell only and T cell plus B2 cell reconstituted mice, indicating that differences in Il4 and Il13 are probably not responsible for the reduced fecal egg burden in mice that had received B2 cells (Figure 7C; Table S1). In contrast, the mucus-related genes Muc2, Muc3, Tff2, and Fcgbp are significantly elevated by B2 cell reconstitution, pointing to a mechanism for the increased clearance (Figure 7C; Table S1). Differences in the expression of additional genes were also examined as well as for un-reconstituted RAG1−/− mice infected with N. brasiliensis L3, naive mice (Table S1), and lung at day 2 post helminth infection (Table S2). In the lung, no gene expression was altered by B2 cell reconstitution except Ear11, an eosinophil-related gene.
Figure 7. B2 Cell Enhanced Clearance Is MC and IgE Dependent.
(A and B) Mice were reconstituted as in Figure 6. Day 7 post-N. brasiliensis infection. Histamine (A) and MCPT-1 (B) were measured by ELISA on mucus from the jejunum.
(C) Expression profiles from jejunal RNA were examined for genes potentially associated with helminth clearance (see also Tables S1 and S2).
(D) Mice were administered anti-ckit (ACK.2) antibody or control IgG daily starting at day 1 and day 7; EPG were measured.
(E) MCs were visualized in intestinal sections with chloroacetate esterase staining countered with hematoxylin (pink and arrows). Scale bar, 500 μm.
(F) PL was examined by flow for MCs (ckit+FcεRI+).
(G and H) MLN and lungs were examined by flow for ILC2 (Live,CD45+Lin−CD90.2+Sca1+ICOS+) percentage (G) and number (H). (Gating schematic SF8A, B.) Reconstituted mice were infected with 750 N. brasiliensis L3 and treated with anti-IgE (R1E4) antibody or control IgG daily starting at reconstitution.
(I and J) On day 7, EPG (I) and adult worms (J) were measured.
*p < 0.05, **p < 0.01, ***p < 0.001. n > 8 mice per group in (A), (B), and (D)–(F). n = 6 mice per group in (C). n = 3 mice per group in (G) and (H). n = 4mice per group in (I) and (J). Error bars depict SEM. Data are the product of two independent repeats. Statistics: an unpaired Studentss t test was utilized for comparison in (A), (B), and (H), a one-way ANOVA with a Tukey post hoc test was used in (D), (G), (I), and (J) and with a Bonferroni post hoc in (C).
To confirm that the mechanism was MC dependent, we injected anti-ckit antibody (ACK.2) into T cell plus B2 cell or T cell only controls to deplete MCs (Brandt et al., 2003). ACK.2-treated mice lacked the B2 enhancement of helminth clearance that was seen in control Rat IgG isotype treated mice (Figure 7D). MC depletion was confirmed in jejunal intestinal sections by chloroacetate esterase staining (Figure 7E) and in peritoneal lavage (PL) on day 7 post inoculation by flow cytometry (Figure 7F). As ckit is an important marker on ILC2s and these cells are implicated in helminth clearance, we examined the mesenteric lymph node (MLN) and lung of ACK.2 and control RAG1−/− mice after N. brasiliensis infection for ILC2 percentage and numbers and found them not statistically different (Figures 7G, 7H, S5H, and S5I). To further support the IgE-mediated mechanism, we injected the anti-IgE antibody (R1E4, which blocks IgE binding to the FcεRI) i.p. into T cell plus B2 cell or T cell only controls to prevent IgE from binding to FcεRI (Baniyash et al., 1988). As in ACK.2-treated mice, R1E4-treated mice lacked the B2 enhancement of helminth clearance that was seen in the Rat IgG isotype control treated mice (Figures 7I and 7J).
To determine whether B1 cells suppressed antigen-specific IgG1 responses, we reconstituted RAG1−/− mice with CD4+ T cells plus B1 and/or B2 cells and immunized them with NP32- KLH in alum. RAG1−/− mice reconstituted with B1 cells only have significantly reduced total-specific and high-affinity IgG1 to the NP antigen, but NP-specific IgG1 levels and total IgG1 levels were similar in mice given B2 cells regardless of the addition of B1 cells. (Figure S5J). These data recapitulate the results observed in the ADAM10Tg mouse model.
DISCUSSION
B1 cells have long been thought to be important innate immune effectors. They generate critical IgM responses to bacteria, as well as to influenza virus (Savage and Baumgarth, 2015; Waffarn et al., 2015). In the past few years, a role for B1 cells in Th2 disease has emerged. Patel and Kearney (2015) demonstrated that B1 cell IgM blocked the response to a house dust mite (HDM) antigen asthma model, with germline anti-phosphorycholine (PC) antibody. Another study suggested that B1 cell IgE might be specific for PC in an HDM model, yet this was never shown (Patel and Kearney, 2015). Both N. brasiliensis and H. polygyrus bakeri have secreted PC epitopes (Péry et al., 1979; Hewitson et al., 2011). Recent studies have emphasized that B1 cell B cell receptors (BCRs) require stimulation during development without costimulation (Kreslavsky et al., 2017). Since this requires abundant amounts of antigen, the majority of B1 cell BCRs are self reactive (Kreslavsky et al., 2017). We do not know whether self-specific B1 cell IgE is present, but B1 cell IgE clearly does not enhance parasitic clearance (Figures 6A and 6B). In addition, we did not observe significant antigen-specific IgE in the B1-only mice that were immunized with OVA (Figure 3E). Variable region mapping is an important next step in determination of the IgE-switched B1-clones that are expanded in response to N. brasiliensis infection. This will help determine whether there is a preference for enhancement of a particular B1 variable region in this infection.
Pochanke et al. (2007) has described a clone of IgE that is present on FcεRI and can degranulate MCs as early as day 7. This early IgE clone is specific for a pharynx-derived epitope on N. brasiliensis, and it does not undergo any somatic hypermutation (SHM) (Pochanke et al., 2007), which is not surprising as germinal centers are minimally developed. The result that specific IgE is made against the parasite is not unexpected, and we would anticipate that this clone would induce MC activation, as exemplified by the PCA (Pochanke et al., 2007). However, there is no evidence that this IgE enhances rejection of the parasite, as we demonstrate with our B2 cells in the RAG1−/− reconstitution model. Also, in our PCA studies (Figures 3F and 3G), N. brasiliensis infection caused an inhibition of the PCA reaction, not its complete elimination. Thus, our results are quite compatible with the Pochanke et al. (2007) findings.
The alarmin IL-25 has been shown to be an important regulator of ILC2s. These cells secrete IL-5 after IL-25 stimulation; both cytokines are important for B1 cell IgE production (Figures 4A and 5). Although we know from our depletion studies that T cells are important in the B1 cell IgE response to N. brasiliensis in vivo, there is a small basal amount of IgE that remains despite this depletion (Figure 1C). This residual B1 cell IgE may be stimulated by these cytokines directly. The role of T cells and location of interaction with B1 cells warrants further investigation, as mice with B1 cells only do not exhibit germinal centers (data not shown). The cytokine order and amount may be important for IL-5 to further increase IgE production in conjunction with IL-25.
Our studies demonstrate a role for IL-25 in the B1 cell IgE response during helminth infection. Along with other signals, such as IL-4, IL-5, and CD40L stimulation from T cells, IL-25 causes B1 cells to proliferate and enhances CSR to IgE. This IgE binds to MC FcεRI and competes with the binding of parasite- specific IgE that is produced by B2 cells. Traditionally, IgE antibody was not thought to be critical in the clearance of helminth infections since clearance of N. brasiliensis was not delayed in IgE−/− mice (Watanabe et al., 1988). In addition, MC-deficient mouse models only exhibit a small delay in N. brasiliensis clearance; thus, the role of MCs in this model has also been minimized (Mitchell et al., 1983). The importance of ILC2s (Neill et al., 2010), alternatively activated macrophages (AAMs) (Oeser et al., 2015), and basophils (Liu et al., 2010) in N. brasiliensis clearance has certainly been recognized. However, our RAG1−/− reconstitution model demonstrates that IgE activation of MCs can play a host-protective role in N. brasiliensis infection. This role is diminished by the B1 cell-produced non-specific IgE. MC degranulation is then no longer evident (Figures 6 and 7; Table S1). Figures 7G and 7H ensure that ILC2 loss was not the cause of the results seen with the anti-ckit antibody; this is in agreement with a recent separate study (Shimokawa et al., 2017). Shimokawa et al. (2017) noted with the use of the anti-ckit antibody, the loss of MCs in the intestines caused a very small reduction in ILC2 number only in the intestinal site due to MC-derived IL-33 that is lost when MCs are deleted. Questions still need to be answered about the effects of anti-ckit treatment on the interstitial cells of Cajal. These cells play a role in intestinal wave activity and are ckit+ (Ordög et al., 1999). The loss of these cells may alter helminth clearance, yet studies utilizing ACK.2 show that the loss of wave activity that leads to muscle quiescence takes 3–4 weeks of treatment (Ordög et al., 1999). Our short anti-ckit treatment regime (Figure 7D) and anti-IgE treatment (Figures 7I and 7J) combine to confirm that IgE binding to the FcεRI is crucial for the B2-mediated enhanced worm expulsion. The lower level of suppression seen with anti-IgE is simply due to a higher N. brasiliensis L3 injection in this experiment series; 750 versus 650 L3. While our data do not directly exclude a contribution by IgE binding to the FcεRI on basophils, we note that there were no significant changes in Mcpt8, the basophil-specific protease expression (Table S1) (Ugajin et al., 2009). Extrapolating from both Hepworth et al. (2012) and Shimokawa et al. (2017), who have recently highlighted the importance of mast cells in clearance of H. polygyrus bakeri, our data support MCs as the primary driver of this phenotype. Although the RAG1−/− reconstitution model has limitations due to lymphopenia (Figures S5A–S5F), there are clearly sufficient cells to give a strong IgE (Figure 6C) and equivalent IgG1 responses (Figure S5J) as well as mediate the observed clearance (Figures 6A and 6B).
IgE has been long known to be induced during helminth infection, and its role in immunity to parasites is often debated (MacDonald et al., 2002). We hypothesize that in the long evolutionary interaction between helminths and mammals, helminths have developed a mechanism of inducing large amounts of B1 cell IgE that may provide it an evolutionary survival advantage. Slower parasite clearance and increased fecundity leads to increased egg output for the parasite and improved evolutionary success (Quinnell et al., 2004). In summary, this study provides evidence for two opposing roles of IgE in helminth infection. The induction of B1 IgE represents a regulatory mechanism that inhibits MC function and in its absence reveals a previously sidelined role for B2 cell IgE and IgE-mediated MCdegranulation in the enhancement of helminth clearance.
EXPERIMENTAL PROCEDURES
Further details and an outline of resources used in this work can be found in Supplemental Experimental Procedures.
Statistical Methods
Error bars represent the standard error of the mean (SEM). A horizontal line with a symbol representing the p value indicates statistical comparison. For pairwise comparisons, Mann-Whitney tests were performed for non-normally distributed data, and Student’s t tests were performed for normally distributed data. For multiple comparisons, Kruskal-Wallis tests with Dunn’s post hoc were performed for non-normally distributed data, and one-way ANOVA tests with Tukey post hoc or Bonferroni post hoc were performed for normally distributed data. All tests are detailed in the figure legends. A p value of <0.05 was considered significant. All statistical analysis was performed with GraphPad Prism 6 (SCR_002798).
Mice
Mice were kept at Virginia Commonwealth University (VCU) in a barrier vivarium facility in accordance with the humane treatment of laboratory animals sets forth by the National Institutes of Health and the American Association for the Accreditation of Laboratory Animal Care. All mouse protocols were conducted with the permission and oversight of the VCU Institutional Animal Care and Use Committee.
Immunization Models
ACA (Evans et al., 2014) and PCA (Starkl et al., 2016) models were performed as described with the following modifications: i.d. spotting (ACA with OVA-albumin [Sigma-Aldrich] or in PCA with IgE anti-DNP) (Keegan et al., 1991) was done on the pre-shaven flank. Evan’s Blue dye (Sigma-Aldrich) was intravenously (i.v.) injected (in ACA immediately following i.d. spot or in PCA 24 hr following mixed with DNP-BSA [Sigma-Aldrich]). Spot size was measured on the back, and back skin was collected for formamide (Sigma-Aldrich) extraction (Evans et al., 2014). For NP Immunizations, NP32KLH (LCG BioSearch Technologies) in alum is injected i.p. in 200 μL saline.
RAG1−/− Reconstitution
All naive RAG1−/− mice were i.v. reconstituted with 5×106 CD4+ T cells. CD4+ T cells are isolated by first B220+ depletion, followed by anti-mouse L3T4 magnetic bead selection (Miltenyi Biotec) from the spleens of WT mice. Indicated mice were i.v. reconstituted with 10 × 106 naive B2 cells. B2 cells are isolated by depletion using either anti-mouse CD43 Miltenyi microbeads (Miltenyi Biotec) or biotinylated anti-mouse CD43 (AB_493384) followed by anti-biotin microbeads (Miltenyi Biotec) from the spleens of WT mice. Indicated mice were i.p. reconstituted with 2–4 × 105 B1 cells. B1 cells are isolated from the peritoneal and pleural cavities of WT mice (Yenson and Baumgarth, 2014). Briefly, cells were Fc blocked on ice for 10 min (2.4G2) (Unkeless, 1979) followed by the following biotinylated antibodies for 30 min on ice: anti-mouse CD23 (B3B4) (AB_312829), anti- mouse CD49b (DX5) (AB_313035), anti-mouse F4/80 (BM8) (AB_893499), anti-mouse CD90.2 (30-H12) (AB_313175), and anti-mouse GR-1 (RB6-8C5) (AB_313368). After washing, anti-biotin microbeads (Miltenyi Biotec) were added, and magnetic bead depletion was performed for B1 enrichment. Mice were not used in experiments until 1 week post reconstitution.
Supplementary Material
Highlights.
B1 cells make IgE in response to helminths
IL-25 induces B1 cell IgE production in cells harvested from helminth-infected mice
B2 cell IgE enhances helminth clearance in a mast cell-dependent manner
B1 cell IgE blocks B2 cell-mediated helminth clearance
Acknowledgments
Special thanks to Julie Farnsworth, Jared Farrar, Qingzhao Zhang, Sana Vohra, Danielle Ebelle, Sharoon Arshad, and Swe San for technical assistance. Additional thanks to John J. Ryan, PhD for guidance and feedback on this project. The study was funded by the NIH/NIAID R01AI18697A1 to D.H.C. and NIH/NIAID F32AI124502 to R.K.M. Microscopy was performed at the VCU Microscopy Facility and flow cytometry utilized the VCU Flow Cytometry core. Both cores are supported, in part, by funding from NIH-NCI Cancer Center Support Grant P30 CA016059.
Footnotes
Supplemental Information includes Supplemental Experimental Procedures, five figures, and two tables and can be found with this article online at https://doi.org/10.1016/j.celrep.2018.01.048.
DECLARATION OF INTERESTS
The authors declare no competing interests.
AUTHOR CONTRIBUTIONS
Conceptualization, R.K.M. and D.H.C.; Methodology, R.K.M., J.C.L., and D.H.C.; Validation, R.K.M. and J.C.L.; Formal Analysis, R.K.M., Y.A.V., E.H.D., and J.C.L.; Investigation, R.K.M., S.R.D., Y.A.V., M.P.Z., B.N.J., E.H.D., J.C.L., A.J.L., M.M.D., and L.M.K.; Resources, J.F.U., F.D.F., and D.H.C.; Writing – Original Draft, R.K.M.; Writing – Resources and Editing, J.F.U., F.D.F., S.R.D., D.H.C., J.C.L., and A.J.L.; Visualization, R.K.M. and Y.A.V.; Supervision, R.K.M.; Project Administration, D.H.C.; Funding Acquisition, D.H.C. and R.K.M.
References
- Ahmed A, Koma MK. Interleukin-33 triggers B1 cell expansion and its release of monocyte/macrophage chemoattractants and growth factors. Scand J Immunol. 2015;82:118–124. doi: 10.1111/sji.12312. [DOI] [PubMed] [Google Scholar]
- Baniyash M, Kehry M, Eshhar Z. Anti-IgE monoclonal antibodies directed at the Fc epsilon receptor binding site. Mol Immunol. 1988;25:705–711. doi: 10.1016/0161-5890(88)90106-x. [DOI] [PubMed] [Google Scholar]
- Baumgarth N, Tung JW, Herzenberg LA. Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin Immunopathol. 2005;26:347–362. doi: 10.1007/s00281-004-0182-2. [DOI] [PubMed] [Google Scholar]
- Bazaral M, Orgel HA, Hamburger RN. The influence of serum IgE levels of selected recipients, including patients with allergy, helminthiasis and tuberculosis, on the apparent P-K titre of a reaginic serum. Clin Exp Immunol. 1973;14:117–125. [PMC free article] [PubMed] [Google Scholar]
- Brandt EB, Strait RT, Hershko D, Wang Q, Muntel EE, Scribner TA, Zimmermann N, Finkelman FD, Rothenberg ME. Mast cells are required for experimental oral allergen-induced diarrhea. J Clin Invest. 2003;112:1666–1677. doi: 10.1172/JCI19785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camberis M, Le Gros G, Urban J., Jr Animal model of Nippostrongylus brasiliensis and Heligmosomoides polygyrus. Curr Protoc Immunol. 2003;Chapter 19:12. doi: 10.1002/0471142735.im1912s55. [DOI] [PubMed] [Google Scholar]
- de Silva NR, Brooker S, Hotez PJ, Montresor A, Engels D, Savioli L. Soil-transmitted helminth infections: updating the global picture. Trends Parasitol. 2003;19:547–551. doi: 10.1016/j.pt.2003.10.002. [DOI] [PubMed] [Google Scholar]
- Erickson LD, Foy TM, Waldschmidt TJ. Murine B1 B cells require IL-5 for optimal T cell-dependent activation. J Immunol. 2001;166:1531– 1539. doi: 10.4049/jimmunol.166.3.1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans H, Killoran KE, Mitre E. Measuring local anaphylaxis in mice. J Vis Exp. 2014;(92):e52005. doi: 10.3791/52005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelman FD, Shea-Donohue T, Morris SC, Gildea L, Strait R, Madden KB, Schopf L, Urban JF., Jr Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol Rev. 2004;201:139–155. doi: 10.1111/j.0105-2896.2004.00192.x. [DOI] [PubMed] [Google Scholar]
- Fort MM, Cheung J, Yen D, Li J, Zurawski SM, Lo S, Menon S, Clifford T, Hunte B, Lesley R, et al. IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity. 2001;15:985–995. doi: 10.1016/s1074-7613(01)00243-6. [DOI] [PubMed] [Google Scholar]
- Gerbe F, Sidot E, Smyth DJ, Ohmoto M, Matsumoto I, Dardalhon V, Cesses P, Garnier L, Pouzolles M, Brulin B, et al. Intestinal epithelial tuft cells initiate type 2mucosal immunity to helminth parasites. Nature. 2016;529:226–230. doi: 10.1038/nature16527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibb DR, Saleem SJ, Kang DJ, Subler MA, Conrad DH. ADAM10 overexpression shifts lympho- and myelopoiesis by dysregulating site 2/site 3 cleavage products of Notch. J Immunol. 2011;186:4244–4252. doi: 10.4049/jimmunol.1003318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurish MF, Bryce PJ, Tao H, Kisselgof AB, Thornton EM, Miller HR, Friend DS, Oettgen HC. IgE enhances parasite clearance and regulates mast cell responses in mice infected with Trichinella spiralis. J Immunol. 2004;172:1139–1145. doi: 10.4049/jimmunol.172.2.1139. [DOI] [PubMed] [Google Scholar]
- Hepworth MR, Maurer M, Hartmann S. Regulation of type 2 immunity to helminths by mast cells. Gut Microbes. 2012;3:476–481. doi: 10.4161/gmic.21507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hewitson JP, Filbey KJ, Grainger JR, Dowle AA, Pearson M, Murray J, Harcus Y, Maizels RM. Heligmosomoides polygyrus elicits a dominant nonprotective antibody response directed against restricted glycan and peptide epitopes. J Immunol. 2011;187:4764–4777. doi: 10.4049/jimmunol.1004140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph M, Auriault C, Capron A, Vorng H, Viens P. A new function for platelets: IgE-dependent killing of schistosomes. Nature. 1983;303:810–812. doi: 10.1038/303810a0. [DOI] [PubMed] [Google Scholar]
- Keegan AD, Fratazzi C, Shopes B, Baird B, Conrad DH. Characterization of new rat anti-mouse IgE monoclonals and their use along with chimeric IgE to further define the site that interacts with Fc epsilon RII and Fc epsilon RI. Mol Immunol. 1991;28:1149–1154. doi: 10.1016/0161-5890(91)90030-n. [DOI] [PubMed] [Google Scholar]
- Komai-Koma M, Gilchrist DS, McKenzie ANJ, Goodyear CS, Xu D, Liew FY. IL-33 activates B1 cells and exacerbates contact sensitivity. J Immunol. 2011;186:2584–2591. doi: 10.4049/jimmunol.1002103. [DOI] [PubMed] [Google Scholar]
- Kreslavsky T, Vilagos B, Tagoh H, Poliakova DK, Schwickert TA, Wöhner M, Jaritz M, Weiss S, Taneja R, Rossner MJ, Busslinger M. Essential role for the transcription factor Bhlhe41 in regulating the development, self-renewal and BCR repertoire of B-1a cells. Nat Immunol. 2017;18:442–455. doi: 10.1038/ni.3694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Q, Kreider T, Bowdridge S, Liu Z, Song Y, Gaydo AG, Urban JF, Jr, Gause WC. B cells have distinct roles in host protection against different nematode parasites. J Immunol. 2010;184:5213–5223. doi: 10.4049/jimmunol.0902879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDonald AS, Araujo MI, Pearce EJ. Immunology of parasitic helminth infections. Infect Immun. 2002;70:427–433. doi: 10.1128/iai.70.2.427-433.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madden KB, Whitman L, Sullivan C, Gause WC, Urban JF, Jr, Katona IM, Finkelman FD, Shea-Donohue T. Role of STAT6 and mast cells in IL-4- and IL-13-induced alterations in murine intestinal epithelial cell function. J Immunol. 2002;169:4417–4422. doi: 10.4049/jimmunol.169.8.4417. [DOI] [PubMed] [Google Scholar]
- Mitchell LA, Wescott RB, Perryman LE. Kinetics of expulsion of the nematode, Nippostrongylus brasiliensis, in mast-cell deficient W/WV mice. Parasite Immunol. 1983;5:1–12. doi: 10.1111/j.1365-3024.1983.tb00718.x. [DOI] [PubMed] [Google Scholar]
- Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TKA, Bucks C, Kane CM, Fallon PG, Pannell R, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464:1367–1370. doi: 10.1038/nature08900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oeser K, Schwartz C, Voehringer D. Conditional IL-4/IL-13-deficient mice reveal a critical role of innate immune cells for protective immunity against gastrointestinal helminths. Mucosal Immunol. 2015;8:672–682. doi: 10.1038/mi.2014.101. [DOI] [PubMed] [Google Scholar]
- Oettgen HC. Fifty years later: emerging functions of IgE antibodies in host defense, immune regulation, and allergic diseases. J Allergy Clin Immunol. 2016;137:1631–1645. doi: 10.1016/j.jaci.2016.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ordög T, Ward SM, Sanders KM. Interstitial cells of cajal generate electrical slow waves in the murine stomach. J Physiol. 1999;518:257–269. doi: 10.1111/j.1469-7793.1999.0257r.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paciorkowski N, Porte P, Shultz LD, Rajan TV. B1 B lymphocytes play a critical role in host protection against lymphatic filarial parasites. J Exp Med. 2000;191:731–736. doi: 10.1084/jem.191.4.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel PS, Kearney JF. Neonatal exposure to pneumococcal phosphorylcholine modulates the development of house dust mite allergy during adult life. J Immunol. 2015;194:5838–5850. doi: 10.4049/jimmunol.1500251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perona-Wright G, Mohrs K, Taylor J, Zaph C, Artis D, Pearce EJ, Mohrs M. Cutting edge: helminth infection induces IgE in the absence of mu- or delta-chain expression. J Immunol. 2008;181:6697–6701. doi: 10.4049/jimmunol.181.10.6697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Péry P, Luffau G, Charley J, Petit A, Rouze P, Bernard S. Phosphorylcholine antigens from Nippostrongylus brasiliensis. II.–Isolation and partial characterization of phosphorylcholine antigens from adult worm. Ann Immunol (Paris) 1979;130:889–900. [PubMed] [Google Scholar]
- Pochanke V, Koller S, Dayer R, Hatak S, Ludewig B, Zinkernagel RM, Hengartner H, McCoy KD. Identification and characterization of a novel antigen from the nematode Nippostrongylus brasiliensis recognized by specific IgE. Eur J Immunol. 2007;37:1275–1284. doi: 10.1002/eji.200737135. [DOI] [PubMed] [Google Scholar]
- Quinnell RJ, Bethony J, Pritchard DI. The immunoepidemiology of human hookworm infection. Parasite Immunol. 2004;26:443–454. doi: 10.1111/j.0141-9838.2004.00727.x. [DOI] [PubMed] [Google Scholar]
- Saleem SJ, Martin RK, Morales JK, Sturgill JL, Gibb DR, Graham L, Bear HD, Manjili MH, Ryan JJ, Conrad DH. Cutting edge: mast cells critically augment myeloid-derived suppressor cell activity. J Immunol. 2012;189:511–515. doi: 10.4049/jimmunol.1200647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savage HP, Baumgarth N. Characteristics of natural antibody-secreting cells. Ann N Y Acad Sci. 2015;1362:132–142. doi: 10.1111/nyas.12799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimokawa C, Kanaya T, Hachisuka M, Ishiwata K, Hisaeda H, Kurashima Y, Kiyono H, Yoshimoto T, Kaisho T, Ohno H. Mast cells are crucial for induction of group 2 innate lymphoid cells and clearance of helminth infections. Immunity. 2017;46:863–874. e4. doi: 10.1016/j.immuni.2017.04.017. [DOI] [PubMed] [Google Scholar]
- Sinha P, Clements VK, Bunt SK, Albelda SM, Ostrand-Rosenberg S. Cross-talk between myeloid-derived suppressor cells and macrophages subverts tumor immunity toward a type 2 response. J Immunol. 2007;179:977–983. doi: 10.4049/jimmunol.179.2.977. [DOI] [PubMed] [Google Scholar]
- Smith KG, Light A, Nossal GJ, Tarlinton DM. The extent of affinity maturation differs between the memory and antibody-forming cell compartments in the primary immune response. EMBO J. 1997;16:2996–3006. doi: 10.1093/emboj/16.11.2996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starkl P, Marichal T, Gaudenzio N, Reber LL, Sibilano R, Tsai M, Galli SJ. IgE antibodies, FcεRIα, and IgE-mediated local anaphylaxis can limit snake venom toxicity. J Allergy Clin Immunol. 2016;137:246–257. e11. doi: 10.1016/j.jaci.2015.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strait RT, Morris SC, Finkelman FD. IgG-blocking antibodies inhibit IgE-mediated anaphylaxis in vivo through both antigen interception and Fc gamma RIIb cross-linking. J Clin Invest. 2006;116:833–841. doi: 10.1172/JCI25575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takatsu K. Interleukin-5 and IL-5 receptor in health and diseases. Proc Jpn Acad, Ser B, Phys Biol Sci. 2011;87:463–485. doi: 10.2183/pjab.87.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takatsu K, Takaki S, Hitoshi Y, Mita S, Katoh S, Yamaguchi N, Tominaga A. Cytokine receptors on Ly-1 B cells. IL-5 and its receptor system. Ann N Y Acad Sci. 1992;651:241–258. doi: 10.1111/j.1749-6632.1992.tb24619.x. [DOI] [PubMed] [Google Scholar]
- Ugajin T, Kojima T, Mukai K, Obata K, Kawano Y, Minegishi Y, Eishi Y, Yokozeki H, Karasuyama H. Basophils preferentially express mouse Mast Cell Protease 11 among the mast cell tryptase family in contrast to mast cells. J Leukoc Biol. 2009;86:1417–1425. doi: 10.1189/jlb.0609400. [DOI] [PubMed] [Google Scholar]
- Unkeless JC. Characterization of a monoclonal antibody directed against mouse macrophage and lymphocyte Fc receptors. J Exp Med. 1979;150:580–596. doi: 10.1084/jem.150.3.580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vink A, Warnier G, Brombacher F, Renauld JC. Interleukin 9-induced in vivo expansion of the B-1 lymphocyte population. J Exp Med. 1999;189:1413–1423. doi: 10.1084/jem.189.9.1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Moltke J, Ji M, Liang HE, Locksley RM. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature. 2016;529:221–225. doi: 10.1038/nature16161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waffarn EE, Hastey CJ, Dixit N, Soo Choi Y, Cherry S, Kalinke U, Simon SI, Baumgarth N. Infection-induced type I interferons activate CD11b on B-1 cells for subsequent lymph node accumulation. Nat Commun. 2015;6:8991. doi: 10.1038/ncomms9991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe N, Katakura K, Kobayashi A, Okumura K, Ovary Z. Protective immunity and eosinophilia in IgE-deficient SJA/9 mice infected with Nippostrongylus brasiliensis and Trichinella spiralis. Proc Natl Acad Sci USA. 1988;85:4460–4462. doi: 10.1073/pnas.85.12.4460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu LC, Zarrin AA. The production and regulation of IgE by the immune system. Nat Rev Immunol. 2014;14:247–259. doi: 10.1038/nri3632. [DOI] [PubMed] [Google Scholar]
- Yenson V, Baumgarth N. Purification and immune phenotyping of B-1 cells from body cavities of mice. Methods Mol Biol. 2014;1190:17–34. doi: 10.1007/978-1-4939-1161-5_2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.