Abstract
The mechanisms by which melanins are transferred from melanocytes and stored within keratinocytes to generate skin pigmentation are hotly debated. In this issue, Correia and Moreiras et al. and Hurbain et al. provide evidence that melanin cores of melanosomes are secreted from melanocytes and taken up and stored within non-degradative membranous organelles in keratinocytes in the basal epidermis of human skin.
Skin pigmentation reflects an interaction between two cell types within an epidermal-melanin unit. Melanocytes In the basal layer of the epidermis first synthesize melanin pigments in specialized membrane-bound compartments called melanosomes. The melanins within melanosomes polymerize on an amyloid matrix to form a melanin core, or “melanocore”. Next, melanin pigments are transferred from melanocyte dendrites to closely apposed keratinocytes in the basal epidermal layer. Keratinocytes store melanin in membrane-bound compartments and position them into a “helmet” surrounding the nucleus to protect their DNA from ultraviolet radiation (UVR) damage (Wu and Hammer 2014). Our understanding of how melanocytes synthesize and polymerize melanins in melanosomes has deepened over the last two decades (Sitaram and Marks 2012). However, we still understand very little about melanin transfer to keratinocytes or about the organelles within which they are stored in these cells – despite substantial evidence that these steps are critical determinants of skin pigmentation (Wu and Hammer 2014). Two papers in this issue of JID (Correia and Moreiras et al.; Hurbain et al.) begin to alleviate this knowledge gap and suggest a means to move forward to better understand how keratinocytes regulate pigmentation.
Many models for melanosome transfer have been proposed over the years, but recently two have curried particular favor. The shedding-phagocytosis model proposes that keratinocytes ingest nearby melanosome-rich melanocyte dendrite fragments. This model is best supported by live imaging analyses of primary mouse melanocyte/keratinocyte co-cultures and of melanocytes in developing chick embryos (Wu et al. 2012; Tadokoro et al. 2016). The exocytosis-endocytosis model proposes that melanocores are secreted by melanocytes upon melanosome fusion with the plasma membrane, and are subsequently taken up by keratinocytes into phagosome-like structures. This model is best supported by static electron microscopy images of serially sectioned human skin (Tarafder et al. 2014). Moreover, the compartments within which melanins are stored in keratinocytes have never been fully characterized; are they classical degradative phagocytic structures or a specialized organelle? In aggregate, the findings reported by Correia and Moreiras et al. and Hurbain et al. support the model that melanocores are transferred to keratinocytes via exocytosis and reside in non-degradative, membrane-bound compartments after phagocytosis by keratinocytes (Figure 1).
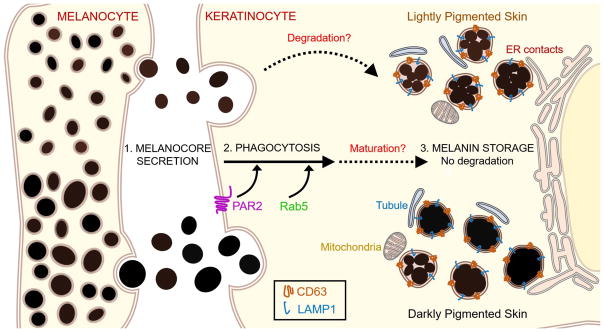
Figure 1. Melanocore uptake and storage by keratinocytes.
Melanocores are secreted by melanocytes into the extracellular space and phagocytosed by keratinocytes (step 1). PAR2 signaling and Rab5 are required for melanocore phagocytosis (step 2). Melanocores in keratinocytes are stored in non-degradative compartments bounded by single membranes that contain CD63 and LAMP2 and that contact rough and smooth ER, mitochondria, and small tubular membranes of unknown origin (step 3). These compartments harbor either clustered structures or dense, isolated structures in all skin types, but the ratio of clustered to isolated melanocore structures decreases as skin pigmentation increases (step 3, top vs. bottom). Although it is not yet clear how melanin storage organelles in keratinocytes mature, some clustered melanocores in lightly pigmented skin may undergo partial degradation (steps 2 to 3).
Correia and Moreiras et al. developed a new microscopy assay to quantify and differentiate melanin uptake in the form of melanocores or membrane-bound melanosomes (Correia et al.). Using a pigmented human melanoma cell line, they isolated either membrane-bound melanosomes from cell homogenates or membrane-free melanocores that had been secreted into the medium. They then added either preparation to cultures of a mouse keratinocyte cell line. The authors found that although keratinocytes internalized both melanosomes and melanocores, only melanocore uptake required PAR2 signaling, a receptor whose activity stimulates melanin transfer in response to UVR (Figure 1, step 2) (Wu and Hammer 2014). Moreover, only internalized melanocores assembled into helmets around the nuclei. These data suggest that while both melanocores and melanosomes are phagocytosed by keratinocytes in vitro, only melanocore uptake and distribution are controlled by physiologically relevant transfer signals. The data are also consistent with previous observations that melanocores are not surrounded by membranes in extracellular spaces (Figure 1, step 1) (Tarafder et al. 2014). Interestingly, synthetic melanins were inefficiently internalized and distributed within keratinocytes, suggesting that melanocore size, shape or compaction might also contribute to uptake and subsequent distribution.
Hurbain et al. also investigated the fate of internalized melanocores in keratinocytes using high resolution, quantitative electron microscopy analyses of human skin samples representative of the full range of human skin pigmentation (Hurbain et al.). Consistent with previous non-quantitative studies (Konrad and Wolff 1973; Szabó et al. 1969; Thong et al. 2003), the authors found that melanin accumulates most prominently in keratinocytes within the deepest layer of skin, the stratum basale, and that the number of melanin granules correlates with general skin pigmentation. More importantly, their analyses clarified how melanins are differentially distributed within keratinocytes across different skin phototypes. Previous limited analyses suggested that melanosomes in dark skin exist primarily in single isolated membrane-delimited structures, and in light skin exclusively as clusters each surrounded by a membrane (Szabó et al. 1969). Hurbain, et al. found that both structures exist in most skin types, but that the ratio of clusters to single melanocores decreases as skin pigmentation increases (Figure 1, step 3). To better characterize the 3-dimensional structure of membrane-bound melanocores at different pigmentation levels, the authors performed electron tomography analyses. In all skin types analyzed, isolated and clustered melanocores were enclosed by single outer membranes and embedded in a heterogeneous, electron dense matrix of unknown composition. In lighter skin types, the matrix was less dense and fuzzy at the edges, suggesting some level of breakdown (Figure 1, step 3). These data and others presented in the paper support the conclusion proposed by Correia and Moreiras et al. that keratinocytes take up melanocores into compartments surrounded by single membranes, perhaps sized to accommodate either one large melanocore or a cluster of smaller cores.
The data in both papers support the exocytosis-endocytosis model of melanin uptake. How can they be reconciled with equally strong data supporting the phagocytosis-shedding model (Wu et al. 2012)? One possibility is that each mechanism is used at a different site of melanin transfer. The human skin samples analyzed by Hurbain et al. and Tarafder et al. reflect primarily interfollicular melanocyte-keratinocyte interactions, whereas the primary mouse melanocytes studied by Wu et al. are most likely derived from the hair bulb. It is conceivable that distinct melanin transfer mechanisms predominate in these two physiologically distinct pigment transfer sites. Differences in physical properties of the hair bulb and skin, such as compliance, could impact shear stresses to favor either melanosome exocytosis or membrane shedding. Alternatively, experimental differences might account for the contrasting data. Future live cell imaging experiments performed in intact human skin cultures or primary cell lines from different pigment transfer systems would begin to address these differences.
The Hurbain et al. data raise another interesting question regarding how keratinocytes from different phototypes internalize and/or store melanocores individually or as clusters. When keratinocytes isolated from light skin are cultured with melanocytes from dark skin, transferred melanocores are organized into clusters, whereas keratinocytes from dark skin organize melanocores individually (Minwalla et al. 2001). This suggests that keratinocyte-specific signals and machinery control melanocore organization during or after transfer. The signals and protein mediators of this process remain unknown. Determinants of melanocore structuring could either be coupled to phagocytosis or arise as keratinocyte melanosomes mature and are transported to the perinuclear region. The reliance of melanocore uptake on keratinocyte PAR2 observed by Correia and Moreiras et al. raises the interesting possibility that PAR2 signaling could segregate phagocytosed melanocores into specific endocytic organelles. For example, partitioning of melanocores into a non-degradative pathway could allow keratinocytes to maintain melanin for long periods, which might be particularly useful in cases of prolonged PAR2 signaling downstream of UVR exposure. It will be interesting to employ live cell imaging approaches to compare the fates of melanocores from a range of human skin phototypes in the absence and presence of PAR2 signaling.
Both Correia and Moreiras et al. and Hurbain et al. investigated the fate of melanocores after uptake by keratinocytes. Correia and Moreiras et al. found that proper melanocore uptake required the early endosomal effector Rab5, but not the late endosomal Rabs 7 and 9, consistent with the model that keratinocytes endocytose or phagocytose melanocores using classical common early endocytic machinery (Figure 1, step 2). Both groups found that melanocores were surrounded by membranes that contain LAMP2 and CD63, markers of late endosomes and lysosomes; Correia and Moreiras et al. additionally detected early endosome markers surrounding some melanocores (Figure 1, steps 2 and 3). These data suggest that melanocores are internalized in a phagocytosis-like process involving fusion with sequential endolysosomal compartments. Surprisingly, however, while both groups showed that keratinocytes contain abundant late endosomes and lysosomes that are acidic and degradative, Correia and Moreiras et al. found that melanin is not significantly degraded in keratinocytes and persists at initial intensity for at least seven days. Moreover, both groups observed that unlike typical degradative phagolysosomes, keratinocyte melanosomes were not acidic. Together, these data suggest that most melanocores are not actively degraded by endolysosomal hydrolases and may reside in a specialized storage organelle in keratinocytes. This contrasts with conclusions of previous work suggesting that keratinocytes degrade melanins, particularly in light skin (Thong et al. 2003; Ebanks et al. 2011; Murase et al. 2013).
One caveat to both studies is that the keratinocyte melanosomes were analyzed at steady-state only; melanosome uptake was not analyzed kinetically. Thus, although the detected melanosomes lacked degradative capacity, it is possible that they represent storage compartments at the end of a progressive series of more active endolysosomal intermediates in which melanin degradation might occur. This would be analogous to distinct populations of lysosomes that vary in luminal pH and degradative capacity according to cell position and that likely represent different stages of maturation (Johnson et al. 2016). Indeed, Hurbain et al. observed partially detached melanin deposits in clustered melanocores, suggesting that some melanins may have been degraded during or after transfer. It will be interesting to determine if melanin degradation is coupled to melanosome transport and positioning in keratinocytes. A coupled transport-degradation system could provide keratinocytes with a way to prevent degradation of perinuclear melanosomes for pronounced nuclear photoprotection.
Previous work had suggested that autophagy regulates melanin levels in keratinocytes and is more active in light skin (Ebanks et al. 2011; Murase et al. 2013). However, whereas Hurbain et al. detected abundant LC3A – a component of the autophagy machinery – in the stratum corneum, they did not detect LC3A on melanosome membranes in either keratinocytes or melanocytes. Although this result is subject to the concern about kinetics raised earlier, at face value it suggests that keratinocyte melanosomes do not contain autophagic membranes and raises questions regarding the relevance of autophagy to melanin degradation in keratinocytes. Autophagy pathways might control melanin levels indirectly in keratinocytes through downstream signaling components that could differ across skin pigmentation types.
Interestingly, using electron tomography and three-dimensional reconstruction, Hurbain, et al. observed that keratinocyte melanosomes made inter-organelle contacts with endoplasmic reticulum, mitochondria, and small, unidentified tubular membranes that might arise from endosomes or from melanosomes themselves (Figure 1, step 3). Such inter-organelle contacts likely reflect membrane remodeling of the keratinocyte melanosome, and might control local structure, content, or dynamics of melanosome membranes. Tubules emanating from melanosome membranes could allow for removal of lysosome membrane proteins that influence degradative potential, such as the vacuolar H+-ATPase responsible for acidifying endolysosomes, or proteins important for melanosome positioning. Another non-mutually exclusive possibility is that the tubules facilitate the reformation of endolysosomes from the melanosome membranes, perhaps after a short-lived phagolysosome-like stage. This would be analogous to the reformation of nascent lysosomes from tubular membranes that extend from autolysosomes after autophagy, which then undergo further maturation to become degradative lysosomes (Rong et al. 2012). A regulated maturation process like this could potentially explain the evidence for melanin degradation in light skin keratinocyte melanosomes by Hurbain, et al. despite the absence of autophagy indicators or signs of degradative activity. It would be interesting to use live cell imaging to examine the role of inter-organelle contacts in keratinocyte melanosome formation and maturation after melanocore phagocytosis.
In sum, both Correia and Moreiras et al and Hurbain et al have provided us with intriguing new insights into mechanisms involved in melanin transfer and storage within keratinocytes that hint at exciting new findings to come.
Bullet points.
Keratinocytes preferentially internalize naked “melanocores” over membranous melanosomes under physiological conditions.
Keratinocytes store melanins in non-degradative, non-acidic organelles that harbor lysosomal membrane proteins.
Skin tone correlates with ratio of isolated dense melanocores to small melanocore clusters.
References
- Correia M, Moreiras H, Pereira F, Neto M, Festas T, Tarafder A, et al. Melanin transferred to keratinocytes resides in non-degradative compartments. J Invest Dermatol. doi: 10.1016/j.jid.2017.09.042. [DOI] [PubMed] [Google Scholar]
- Ebanks JP, Koshoffer A, Wickett RR, Schwemberger S, Babcock G, Hakozaki T, et al. Epidermal keratinocytes from light vs. dark skin exhibit differential degradation of melanosomes. J Invest Dermatol. 2011;131(6):1226–33. doi: 10.1038/jid.2011.22. [DOI] [PubMed] [Google Scholar]
- Hurbain I, Romao M, Sextius P, Bourreau E, Marchal C, Bernerd F, et al. Melanosome distribution in keratinocytes in different skin types: melanosome clusters are not degradative organelles. J Invest Dermatol. doi: 10.1016/j.jid.2017.09.039. [DOI] [PubMed] [Google Scholar]
- Johnson DE, Ostrowski P, Jaumouillé V, Grinstein S. The position of lysosomes within the cell determines their luminal pH. J Cell Biol. 2016;212(6):677–92. doi: 10.1083/jcb.201507112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konrad K, Wolff K. Hyperpigmentation, melanosome size, and distribution patterns of melanosomes. Arch Dermatol. 1973;107(6):853–60. [PubMed] [Google Scholar]
- Minwalla L, Zhao Y, Le Poole IC, Wickett RR, Boissy RE. Keratinocytes play a role in regulating distribution patterns of recipient melanosomes in vitro. J Invest Dermatol. 2001;117(2):341–7. doi: 10.1046/j.0022-202x.2001.01411.x. [DOI] [PubMed] [Google Scholar]
- Murase D, Hachiya A, Takano K, Hicks R, Visscher MO, Kitahara T, et al. Autophagy has a significant role in determining skin color by regulating melanosome degradation in keratinocytes. J Invest Dermatol. 2013;133(10):2416–24. doi: 10.1038/jid.2013.165. [DOI] [PubMed] [Google Scholar]
- Rong Y, Liu M, Ma L, Du W, Zhang H, Tian Y, et al. Clathrin and phosphatidylinositol-4,5-bisphosphate regulate autophagic lysosome reformation. Nat Cell Biol. 2012;14(9):924–34. doi: 10.1038/ncb2557. [DOI] [PubMed] [Google Scholar]
- Sitaram A, Marks MS. Mechanisms of protein delivery to melanosomes in pigment cells. Physiology. 2012;27(2):85–99. doi: 10.1152/physiol.00043.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabó G, Gerald AB, Pathak MA, Fitzpatrick TB. Racial differences in the fate of melanosomes in human epidermis. Nature. 1969;222(5198):1081–2. doi: 10.1038/2221081a0. [DOI] [PubMed] [Google Scholar]
- Tadokoro R, Murai H, Sakai KI, Okui T, Yokota Y, Takahashi Y. Melanosome transfer to keratinocyte in the chicken embryonic skin is mediated by vesicle release associated with Rho-regulated membrane blebbing. Sci Rep. 2016;6:38277. doi: 10.1038/srep38277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarafder AK, Bolasco G, Correia MS, Pereira FJC, Iannone L, Hume AN, et al. Rab11b mediates melanin transfer between donor melanocytes and acceptor keratinocytes via coupled exo/endocytosis. J Invest Dermatol. 2014;134(4):1056–66. doi: 10.1038/jid.2013.432. [DOI] [PubMed] [Google Scholar]
- Thong H-Y, Jee S-H, Sun C-C, Boissy RE. The patterns of melanosome distribution in keratinocytes of human skin as one determining factor of skin colour. Br J Dermatol. 2003;149(3):498–505. doi: 10.1046/j.1365-2133.2003.05473.x. [DOI] [PubMed] [Google Scholar]
- Wu X, Hammer JA. Melanosome transfer: it is best to give and receive. Curr Opin Cell Biol. 2014;29:1–7. doi: 10.1016/j.ceb.2014.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu XS, Masedunskas A, Weigert R, Copeland NG, Jenkins NA, Hammer JA. Melanoregulin regulates a shedding mechanism that drives melanosome transfer from melanocytes to keratinocytes. Proc Natl Acad Sci U S A. 2012;109(31):E2101–9. doi: 10.1073/pnas.1209397109. [DOI] [PMC free article] [PubMed] [Google Scholar]