Abstract
Background
There are conflicting recommendations and highly variable practices regarding the level of A1c to initiate insulin for individuals with newly diagnosed diabetes. This is complicated in low-income settings where adverse reactions or negative perceptions of insulin are often magnified.
Objectives
Compare the clinical outcomes of insulin and Oral Agents (OAs) in low-income settings in the United States.
Methods
We conducted a retrospective chart review in community clinics serving low -income individuals with newly diagnosed type 2 diabetes who were initiated on insulin or OAs. The primary outcome was change of hemoglobin A1c (A1c) from baseline to 12 months. Secondary outcomes consisted of other clinical measures including Emergency Department (ED) visits.
Results
A total of 18% (88/489) of patients were started on insulin. The adjusted average decrease of A1c from baseline was greater in the OA group (insulin: −1.97% vs. OA: −2.52%; p<0.001). In a subset analysis of individuals with A1cs >11%, significantly more patients were started on OAs (insulin: n=51, OA: n=93; p<0.001) and A1c improvements were similar at 12 months (insulin: −5.06% [12.94% to 7.88%] OA: −4.62% [12.57% to 7.96%]; p=0.846). Baseline A1c predicted insulin initiation (p<0.001): For every one-unit increase in baseline A1c, the odds of insulin initiation increased by 47.5%. Individuals in the insulin group had more ED visits per year (0.169 vs. 0.0025; p<0.005).
Conclusions
Given the positive clinical outcomes of OAs even with markedly elevated A1c levels in addition to the healthcare system benefits, they are a promising initial therapy for low-income adults with newly diagnosed type 2 diabetes.
Keywords: Insulin therapy, Diabetes, Efficacy
Introduction
Recommendations differ regarding the initiation of insulin for patients with newly diagnosed type 2 diabetes [1–5]. Some investigators have suggested that individuals with higher baseline Hemoglobin A1c (A1c) levels should be started on insulin but there is not a consensus for the definition of a high A1c [5,6]. Others have encouraged early insulin initiation when oral hypoglycemic agents (OAs) fail, which has led to complicated discussions of OA dosing strategies [2,4,7].
Negative perceptions and adverse events from insulin further complicate this debate. Insulin has been associated with weight gain, medication apprehension (e.g., fear of needles), and negative cultural attitudes [5,8,9]. Many patients resist starting insulin, even at A1c levels that may have led to physician recommendation to do so6. Insulin-related hypoglycemia is another barrier that contributes to resistance. According to the National Electronic Injury Surveillance System Cooperative Adverse Drug Event Surveillance Project, there are nearly 100,000 Emergency Department (ED) visits due to insulin-related hypoglycemia and medication errors annually [10]. Of those cases, nearly 30,000 (29.3%) result in hospitalizations, 60.6% of patients have severe neurologic sequelae, and 53.4% report a glucose of less than or equal to 50 mg/dl. The most common reasons for insulin-related hypoglycemia and medication errors are reduced food intake or incorrect insulin administration [10].
Other barriers to insulin, including low literacy and financial constraints, are particularly problematic in underserved populations [11]. Specifically, investigators have demonstrated negative effects of low literacy related to insulin that include identifying the correct dosage tier on a syringe and calculating carbohydrate intake for insulin titration [11]. In addition, insulin is associated with increased cost compared to OAs8 [12,13], making it difficult for underserved patients to obtain appropriate medications and supplies (e.g., test strips, lancets, syringes) [7,12,13]. Though newer insulin analogues are preferred (e.g., once daily dosing) and may be better received by patients, they are considerably more expensive [8]. Human insulin’s, particularly premixed NPH-regular insulin, are less expensive but require multiple daily dosing, increasing the risk for nonadherence, provider-patient miscommunications, and insulin stacking (inappropriately high serum levels of insulin accumulated due to repeated doses) [8,14].
Though there are multiple barriers to insulin, it is still considered the most potent hypoglycemic agent [1]. This results in a common choice when glycemic control is difficult or baseline A1c levels are elevated in spite of its risks [1,4]. However, there is little real world data comparing the clinical outcomes of the insulin and OAs, particularly with low-income populations. Of all individuals with diabetes in the world, 80% live in low-income areas [15]. Estimates predict that the number of people living with diabetes will rise from the current 336 million to 520 million by 2030 [16]. Though many diagnosed with type 2 diabetes may eventually need insulin [8,17], delaying initiation until oral agents are maximized may be an important strategy in low-income settings and in the developing world [13].
This study provided real world data related to insulin versus OA initiation and evaluated what the actual outcomes are among low-income populations. The objectives of this study were to 1) compare individuals initiated on insulin to those started exclusively on OAs, 2) evaluate variables (i.e., baseline A1c, BMI, sex, race/ethnicity) that might predict insulin versus OA initiation, and 3) analyze clinical outcomes (i.e., change of A1c, Body Mass Index [BMI], blood pressure) of each group. Our primary hypothesis was that OAs would be clinically equivalent to insulin (i.e., A1c after 12 months) but superior in terms of system burden (i.e., ED visits).
Methods
To study the initiation of diabetes medications in a low-income, largely minority US population served by public clinic system, we identified a sample of patients who were diagnosed with type 2 diabetes and then followed for at least 12 months in this system in a retrospective chart review. We evaluated predictors and outcomes of initial therapy being insulin as opposed to OAs.
Patients in the study were diagnosed with diabetes between the years 2010 to 2015 and received their healthcare at Community Clinics operated by the Harris Health System. The Harris Health System is a county owned health system that serves low-income (> 150% of the federal poverty level), mainly uninsured persons. Harris County includes Houston, Texas, the nation’s fourth largest city. Data regarding clinical information (i.e., A1c, height, weight), ethnicity, medications, and past medical history were extracted from each individual’s Electronic Medical Record (EMR).
Inclusion criteria consisted of (1) ≥18 years old, (2) newly diagnosed with type 2 diabetes (i.e., A1c >6.5%) between the years 2010 and 2015. Exclusion criteria consisted of (1) baseline A1c less than American Diabetes Association targets (<7% or <7.5% if 65 years old or greater) [1], (2) confounding disease or condition (i.e., gestational diabetes, chronic steroid therapy, chronic kidney disease at a level contraindicated to metformin initiation according to guidelines during the time of the study [Creatinine >1.4 in females; >1.5 males] [18,19], (3) not on a hypoglycemic agent or incomplete EMRs. To avoid coding errors (i.e., coded as a new diagnosis in the Harris County system but reported previously at an outside clinic), a physician performed a secondary EMR review that included reading provider notes and historic prescriptions. The study design was evaluated and approved by the Institutional Review Board at Baylor College of Medicine.
Measures
We gathered clinical information (baseline to 12 months), medication data (baseline to 24 months), and additional information from individuals with markedly elevated A1c levels.
Clinical information
Clinical information (i.e., A1c, cholesterol, height, weight), ethnicity, medications, and past medical history were extracted from each individual’s EMR. A1c was collected at baseline (date of OA/insulin initiation), 6-months, and 12-months. Lipid profile (total cholesterol, triglycerides, high-density lipoprotein, low-density lipoprotein), blood pressure, and weight were collected at baseline and 12-months. The number of ED visits per patient was also collected from baseline to 12 months. If an individual switched from OAs to insulin or vice versa, data was not gathered beyond the switching point.
Medication data
We recorded every hypoglycemic agent prescribed from baseline to 24 months for each individual.
Subset population
Since we hypothesized that A1c levels would predict insulin initiation, we wanted to analyze groups when baseline A1c levels were more comparable. In order to do this, we analyzed individuals whose A1c levels would be considered markedly elevated using current guidelines (>11%) [1,4]. Data gathered consisted of baseline and 12-month clinical outcomes (i.e., A1c, BMI, cholesterol, blood pressure).
Statistical analysis
The association between insulin initiation and baseline A1c was evaluated using logistic regression analyses. The covariates included sex, race/ethnicity, age, sequelae of type 2 diabetes (i.e., presence of at least one of the following: Coronary artery disease, congestive heart failure, chronic kidney disease, or cerebral vascular attack), history of hypertension, history of hyperlipidemia, and baseline BMI. The change of A1c, BMI, blood pressure, and lipid levels of individuals between OAs and insulin were examined using analysis of covariance (ANCOVA). In addition to the covariates listed above, the baseline A1c, systolic and diastolic blood pressure, and cholesterol were also accordingly included in the change of A1c model.
In order to handle any missing data, multiple imputations by Markov Chain Monte Carlo method were used and 20 datasets with imputed values were created. Starting after the first 200 iterations, data were sampled with 100 iterations between successive imputations. Each dataset was analyzed independently with missing data values imputed in the pre-specified models. The coefficient estimates from the analyses of the 20 imputation-completed datasets were combined into a single set of estimates and adjusted standard errors based on Rubin’s rules. All the analyses were conducted in SAS (version 9.4, SAS Institute Inc., Cary, NC, USA). Alpha was set at 0.05.
Results
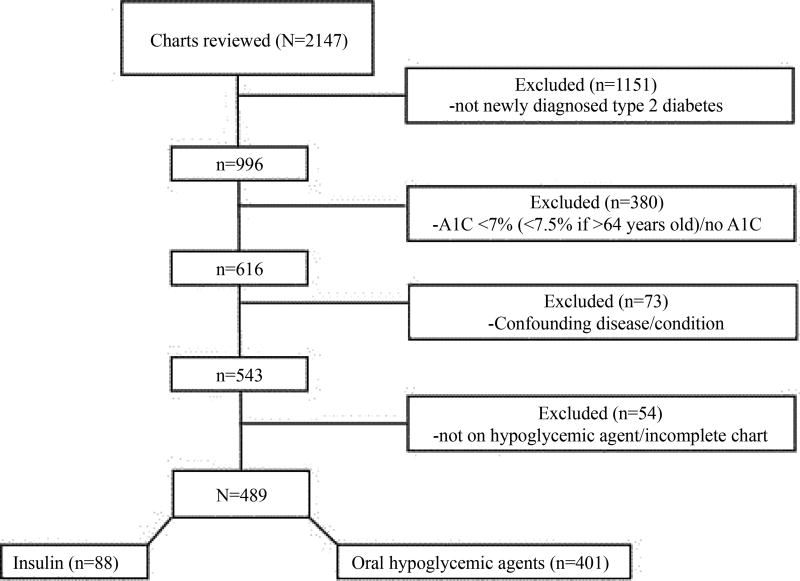
Demonstrated in Figure 1, a total of 2147 charts were reviewed. The secondary physician review assisted in identifying a large amount of individuals who contained a new ICD code of diabetes but were not a true new diagnosis (i.e., diagnosed at an outside healthcare system) (53.6%, n=1151). A total of 489 patients were included in the final analysis (insulin: 88, OA: 401).
Figure 1.
Flowchart of patients who met inclusion criteria for the study population.
Descriptive data analysis
Table 1 illustrates the baseline characteristics of individuals. A substantial number of patients (18.0%) were started on insulin therapy. The majority of patients were minorities and primarily Hispanic in both groups (insulin: 54.6%, OA: 68.3%). There were also a large number of African American individuals in the insulin group (insulin: 35.2%, OA: 17.5%). Age, comorbidities (i.e., hypertension, hyperlipidemia), and diabetes sequelae (i.e., coronary artery disease, congestive heart failure, chronic kidney disease, cerebral vascular accident) were not significantly different between groups.
Table 1.
Baseline characteristics of individuals with newly diagnosed type 2 diabetes in low-income community clinics (N=489).
| Insulin Initiation | OAA Initiation | p-value | |
|---|---|---|---|
| N | 18%(n=88) | 82% (n=401) | - |
| Sex | |||
| Male | 52.3% (n=46) | 42.7% (n=171) | 0.1 |
| Female | 47.7% (n=42) | 57.3% (n=230) | |
| Race/Ethnicity | |||
| AAB/non-Hispanic Black | 35.2% (n=31) | 17.5% (n=70) | 0.001 |
| Caucasian/Other | 10.2% (n=9) | 14.2% (n=58) | |
| Hispanic | 54.6% (n=48) | 68.3% (n=273) | |
| Age (years) (SD) | 47.1 (±10.3) | 48.6 (±11.2) | 0.273 |
| Comorbidities | |||
| Hypertension | 69.3% (n=61) | 59.9% (n=240) | 0.98 |
| Hyperlipidemia | 78.4% (n=69) | 76.6% (n=307) | 0.709 |
| Diabetes Sequelae | 11.2% (n=10) | 8.9% (n=36) | 0.265 |
| Coronary artery disease | 4.5% (n=4) | 4.7% (n=19) | |
| Congestive heart failure | 4.5% (n=4) | 1.5% (n=6) | |
| Chronic kidney disease | 1.1% (n=1) | 0.7% (n=3) | |
| Cerebral vascular attack | 1.1% (n=1) | 2.0% (n=8) | |
OAs: Oral Hypoglycemic Agents;
AA: African American;
ED: Emergency Department
Main analyses
Clinical outcomes
Table 2 demonstrates the clinical outcomes for the two groups. Between groups, A1c, BMI, and total cholesterol levels were significantly different at baseline. By 12 months, there were no statistical differences between groups including A1c, BMI, lipid profile (total cholesterol, triglycerides, high density lipoprotein, low density lipoprotein), and blood pressure (systolic, diastolic). In both groups, the 6-month A1c levels were lower than the 12-month values. However, there was a more marked A1c increase in the insulin group from 6 to 12 months (insulin: 0.52% (7.21 to 7.73%), OA: 0.29% (6.93% to 7.22%)). Analyses with ANCOVA showed that the difference between insulin and OA initiation on A1c change was significant (p<0.001) and that the adjusted average decrease in A1c (change from baseline) was greater with OAs (insulin: −1.97% vs. OA: −2.52%; p<0.001). Further, the insulin group had a significantly higher amount of ED visits compared to those placed on OAs (p<0.005).
Table 2.
Descriptive clinical data of individuals with newly diagnosed type 2 diabetes in low-income community clinics N=489).
| Insulin Initiation | OAA Initiation | p | |
|---|---|---|---|
| A1c (%) | |||
| Month 0 | 11.25 (+2.1) | 9.40 (+2.1) | <0.001 |
| Month 6 | 7.21 (+1.7) | 6.93 (+1.4) | 0.192 |
| Month 12 | 7.73 (+2.1) | 7.22 (+1.7) | 0.064 |
| Systolic Blood Pressure (mmHg) | |||
| Month 0 | 129.1 (+18.4) | 130.5 (+18.1) | 0.428 |
| Month 12 | 129.4 (+18.0) | 120.2 (+19.8) | 0.762 |
| Diastolic Blood Pressure (mmHg) | |||
| Month 0 | 78.9 (+11.7) | 80.5 (+17.9) | 0.493 |
| Month 12 | 77.8 (+11.4) | 77.4 (+11.9) | 0.936 |
| Cholesterol (mg/dL) | |||
| Month 0 | |||
| Total Cholesterol | 192.7 (+53.1) | 204.9 (+46.8) | 0.037 |
| High-density lipoprotein | 37.5 (+46.6) | 33.0 (+91.2) | 0.673 |
| Low-density lipoprotein | 110.0 (+60.7) | 125.0 (+99.2) | 0.208 |
| Triglycerides | 225.7 (+223.2) | 234.3 (+262.5) | 0.786 |
| Month 12 | |||
| Total Cholesterol | 180.0 (+46.2) | 184.4 (+40.8) | 0.484 |
| High-density lipoprotein | 39.5 (+18.5) | 40.8 (+18.5) | 0.642 |
| Low-density lipoprotein | 104.0 (+36.5) | 106.0 (+32.6) | 0.662 |
| Triglycerides | 182.5 (+172.5) | 188.5 (+143.3) | 0.775 |
| BMI (kg/m2) | |||
| Month 0 | 31.5 (+7.2) | 33.3 (+7.6) | 0.035 |
| Month 12 | 33.1 (+ 7.1) | 33.2 (+7.2) | 0.326 |
| EDC visits (0 to 12 months) (SD) | 0.169 (+0.43) | 0.0025 (±0.05) | 0.005 |
OAs: Oral Hypoglycemic Agents
Predictors of insulin initiation
Logistic regression analyses revealed that baseline A1c was a significant predictor of insulin initiation (p<0.001): For every one-unit increase in baseline A1c, the odds of insulin initiation increased by 47.5%. Other variables including BMI, age, comorbidities, and sex were not significant predictors.
Medication data
Data from 24 months of medication records revealed that 22.7% (20/88) switched from insulin to OAs while only 4.2% (17/401) changed from OAs to insulin (p<0.001).
Subset population
We analyzed a subset of individuals with baseline A1c levels >11%. A significant number were started on OAs (insulin: n=51, OA: n=93; p<0.001). Baseline A1cs were not significantly different (insulin: 12.94%, OA: 12.57%; p=0.1) and groups had equivalent decreases in A1c at 12 months (insulin: 7.88% (−5.06%), OA: 7.96% (−4.61%); p=0.846). There were no significant differences in other clinical values including A1c, BMI, lipid profile (total cholesterol, triglycerides, high density lipoprotein, low density lipoprotein), and blood pressure (systolic, diastolic).
Discussion
The current study compared real world data of insulin and OA initiation, clinical outcomes, and predicting variables for individuals with newly diagnosed type 2 diabetes in a low-income setting. We had several key findings including that patients placed on OAs had equivalent or better clinical outcomes. This was also true for patient with markedly elevated A1cs >11%. Also, the level of A1c at diagnosis strongly predicted placement on insulin.
Clinical outcomes were an important finding in the current study. Since the insulin group started at a significantly higher baseline A1c level, it was not surprising that its absolute change (−3.52%) was greater than the OA group (−2.18%). However, further analyses that accounted for these differences revealed that the adjusted average change of A1c was greater for OAs (−2.52%) than insulin (−1.97%) (p<0.0001). Further, in an additional analysis of individuals with severely elevated baseline A1cs (>11%), the insulin and OA groups had no significant difference at baselines (insulin: 12.94%, OA: 12.57%; p=0.1) and by 12 months, had similar improvements in A1c levels (insulin 7.88% (−5.06%), OA 7.96% (−4.61%); p=0.846). These analyses suggest that OAs is just as efficacious as insulin to improve glycemic control, even at markedly elevated baseline levels. This contrasts current recommendations that suggest starting insulin for A1c levels as low as 7.5% [4].
These findings also conflict with investigations that have found insulin to be the most effective hypoglycemic agent [2,5,20]. Data from these investigations likely explain why we found a practice of initiating patients on insulin with increasing A1cs. An expanding body of literature has recommended early insulin initiation for patients with elevated A1c levels in order to preserve beta-cell function [2,21]. However, disease duration varies in these studies [2,20]. Pancreatic β-cell function in individuals with newly diagnosed diabetes is vastly different than in individuals who have had diabetes for decades, resulting in a better response to OAs [2]. In addition, underserved communities may have more difficulties and barriers to insulin use [13], which would also lead to better OA outcomes.
We observed that in both groups, A1c levels substantially decreased in the first six months. However from 6 to 12 months, A1c levels increased in both groups, though this rise was more pronounced in the insulin group. One explanation for this increase in the insulin group could be hypoglycemia. Since A1c is the estimated average glucose during a 3-month period [22], recurrent hypoglycemic events would result in falsely lower A1c levels. This theory is supported by several observations during our chart review including provider encounters with concerns of insulin-induced hypoglycemia and A1c levels less than 5% at six months in the insulin group. Other explanations include increased patient motivation for lifestyle changes at the time of diagnosis that may plateau or decrease by 6-months or that individual may initially comply with insulin administration but have substantial trouble maintaining this behavior.
We also observed that there were significantly more individuals switched from insulin to OAs than vice versa. Specifically, 22% switched from insulin to OAs while 4.2% change from OAs to insulin in a two-year period (p<0.001). Negative perceptions of insulin may have contributed to this finding. Insulin also has known issues with adherence due to anxiety, discomfort, and fear [23]. On the other hand, individuals may have seen an improvement in glucose (though still not optimal) and decreased medication frequency.
Consistent with the literature, we found that the insulin group had more diabetes-related ED visits and weight gain [7]. ED use is an increasing issue in healthcare, placing large financial burdens on the patient and system [7]. The annual expenditure of ED visits due to insulin-induced hypoglycemia is $120 million10. In the current study, individuals started on insulin went to the ED significantly more than those on OAs (p<0.005). Further, the insulin group increased BMI while the OA individuals decreased (insulin: 31.5 kg/m2 to 33.1 kg/m2, OA: 33.4 kg/m2 to 33.3 kg/m2). Since hypoglycemic agent need and weight are interrelated, it is critical to use agents that do not create a vicious cycle of increased medications and BMI [24].
In addition to the clinical benefits of OAs, these findings are of vital importance for low-income populations. With limited access to healthcare, low-income settings are particularly susceptible to increased ED utilization [7]. Also, insulin is a known contributor to adverse events due to low literacy [11,13,23]. The simpler dosing of OAs in lieu of injectable administration of insulin lessens the likelihood of these adverse events [7]. Further, insulin is associated with higher resource utilization [12]. The average individual on insulin uses 764.3 self-monitoring blood glucose strips per year and spends $2850 on glucose testing, insulin, and supplies [12]. This is in contrast to OAs where many do not need self-monitoring blood glucose [25] and the two most commonly used OA classes (biguanides, sulfonylureas) have a monthly expenditure of $4 (annual $48/medication) [26].
Like in all observational data, there are possible limitations. There could be unmeasured variables in the decision to use insulin versus OA for initial therapy. In individuals with an A1c >11% it is quite possible that patient resistance was the critical factor, and that this could have an unpredictable impact on outcome. The time before diagnosis is quite variable and just how long diabetes was undiagnosed prior to treatment differs individually. The fact that we observed OAs as or more effective than insulin may actually be an underestimate in that our system has a very restricted oral formulary: Only metformin and sulfonylureas were readily available. There was limited use of meglitinides, highly restricted use thiazolidinedione’s, and no availability of Dipeptidyl Peptidase 4 (DPP-4) inhibitors, Sodium-Glucose Co-Transporters (SGLT) inhibitors, or Glucagon-Like Peptide (GLP)-1. As the DPP-4 and GLP-1 are approaching generic and becoming more economical, the oral strategy may become even more attractive in low-income populations. The 2017 AACE guidelines allow option of triple oral even for AIC>9 without symptoms [4].
Conclusion
Given the clinical outcomes and healthcare systems benefits of OAs, they may be superior to insulin for adults with newly diagnosed diabetes in low-income settings. Many individuals with type 2 diabetes will eventually need insulin [8]. However, considering the costs, complexity of management, and risks of hypoglycemia, delaying insulin in individuals until ultimately needed may be an important consideration.
Acknowledgments
National Institutes of Health. National Institute of Diabetes, Digestive, and Kidney Disorders. Federal Award Identification Number (FAIN) K23DK110341. Elizabeth Vaughan (PI).
References
- 1.American Diabetes Association. Standards of medical care in diabetes 2017. J Clin Appl Res Educ. 2017;40(S1):S1–S134. [Google Scholar]
- 2.Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: A consensus algorithm for the initiation and adjustment of therapy: A consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2009;32:193–203. doi: 10.2337/dc08-9025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Raz I. Guideline approach to therapy in patients with newly diagnosed type 2 diabetes. Diabetes Care. 2013;36(s2):s139–44. doi: 10.2337/dcS13-2035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes managment algorithm - 2017 executive summary. Endocr Pract. 2017;23(2):207–38. doi: 10.4158/EP161682.CS. [DOI] [PubMed] [Google Scholar]
- 5.Niseander K. Early and aggressive initiation of Insulin therapy in type 2 diabetes: What is the evidence? Clinical Diabetes. 2009;27(2):60–8. [Google Scholar]
- 6.Peyrot M, Rubin RR, Lauritzen T, et al. Resistance to insulin therapy among patients and providers: results of the cross-national Diabetes Attitudes, Wishes, and Needs (DAWN) study. Diabetes Care. 2005;28(11):2673–9. doi: 10.2337/diacare.28.11.2673. [DOI] [PubMed] [Google Scholar]
- 7.World Health Organization. Prevention and control of noncommunicable diseases: Guidelines for primary health care in low-resource settings. Geneva, CH: World Health Organization; 2012. [PubMed] [Google Scholar]
- 8.Hamaty M. Insulin treatment for type 2 diabetes: When to start, which to use. Cleveland Clinic Journal of Medicine. 2011;78(5):332–42. doi: 10.3949/ccjm.78a.10051. [DOI] [PubMed] [Google Scholar]
- 9.Hu J, Amirehsani K, Wallace D, et al. The meaning of insulin to Hispanic immigrants with type 2 diabetes and their families. Diabetes Educ. 2012;38(2):263–70. doi: 10.1177/0145721712437559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Geller AI, Shehab N, Lovegrove MC, et al. National estimates of insulin-related hypoglycemia and errors leading to emergency department visits and hospitalizations. JAMA. 2014;174(5):678–86. doi: 10.1001/jamainternmed.2014.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.White RO, Wolff K, Cavanaugh, et al. Addressing health literacy and numeracy to improve diabetes education and care. Diabetes Spectr. 2011;23(4):238–43. doi: 10.2337/diaspect.23.4.238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yeaw J, Lee WC, Aagren M, et al. Cost of self-monitoring of blood glucose in the United States among patients on an insulin regimen for diabetes. J Manag Care Pharm. 2012;18(1):21–32. doi: 10.18553/jmcp.2012.18.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grant P. Management of diabetes in resource-poor settings. Clin Med. 2013;13(1):27–31. doi: 10.7861/clinmedicine.13-1-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aye MM, Atkin SL. Patient safety and minimizing risk with insulin administration – role of insulin degludec. Drug Healthc Pat Saf. 2014;6:55–67. doi: 10.2147/DHPS.S59566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Horton R. Diabetes-a global threat. The Lancet. 2009;373:1735. doi: 10.1016/S0140-6736(09)60954-5. [DOI] [PubMed] [Google Scholar]
- 16.International Diabetes Federation. IDF diabetes atlas. 7. Brussels, BE: International Diabetes Federation; 2017. [Google Scholar]
- 17.UK Prospective Diabetes Study Group. Overview of 6 years of therapy of type II diabetes: A progressive disease. Diabetes & Metabolic Syndrome. 1995;44:1249–58. [PubMed] [Google Scholar]
- 18.US Food and Drug Administration. FDA Drug Safety Communication: FDA revises warnings regarding use of the diabetes medicine metformin in certain patients with reduced kidney function. 2016 [Google Scholar]
- 19.https://www.accessdata.fda.gov/drugsatfda_docs/nda/2000/20357S019_Glucophage_prntlbl.pdf
- 20.Steffes MW, Sibley S, Jackson M, et al. B-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care. 2003;26(3):832–6. doi: 10.2337/diacare.26.3.832. [DOI] [PubMed] [Google Scholar]
- 21.Henske JA, Griffith ML, Fowler MJ. Initiating and titrating insulin in patients with type 2 diabetes. Clinical Diabetes. 2009;27(2):72–6. [Google Scholar]
- 22.Saudek CD, Brick JC. The clinical use of hemoglobin A1c. J Diabetes Sci Technol. 2009;3(4):629–34. doi: 10.1177/193229680900300402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Aronson R. The role of comfort and discomfort in insulin therapy. Diabetes Technol Ther. 2012;14(8):741–7. doi: 10.1089/dia.2012.0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mitri J, Hamdy O. Diabetes medications and body weight. Expert Opin Drug Saf. 2009;8(5):573–84. doi: 10.1517/14740330903081725. [DOI] [PubMed] [Google Scholar]
- 25.Cameron C, Coyle D, Ur E, et al. Cost-effectiveness of self-monitoring of blood glucose in patients with type 2 diabetes mellitus managed without insulin. CMAJ. 2010;182(1):28–34. doi: 10.1503/cmaj.090765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lipska KJ, Krumholz HM. Is Hemoglobin A1c the right outcome for studies in diabetes? JAMA. 2017 doi: 10.1001/jama.2017.0029. Epub ahead of print:E1–E2. [DOI] [PMC free article] [PubMed] [Google Scholar]